Submitted:
02 April 2024
Posted:
03 April 2024
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Results
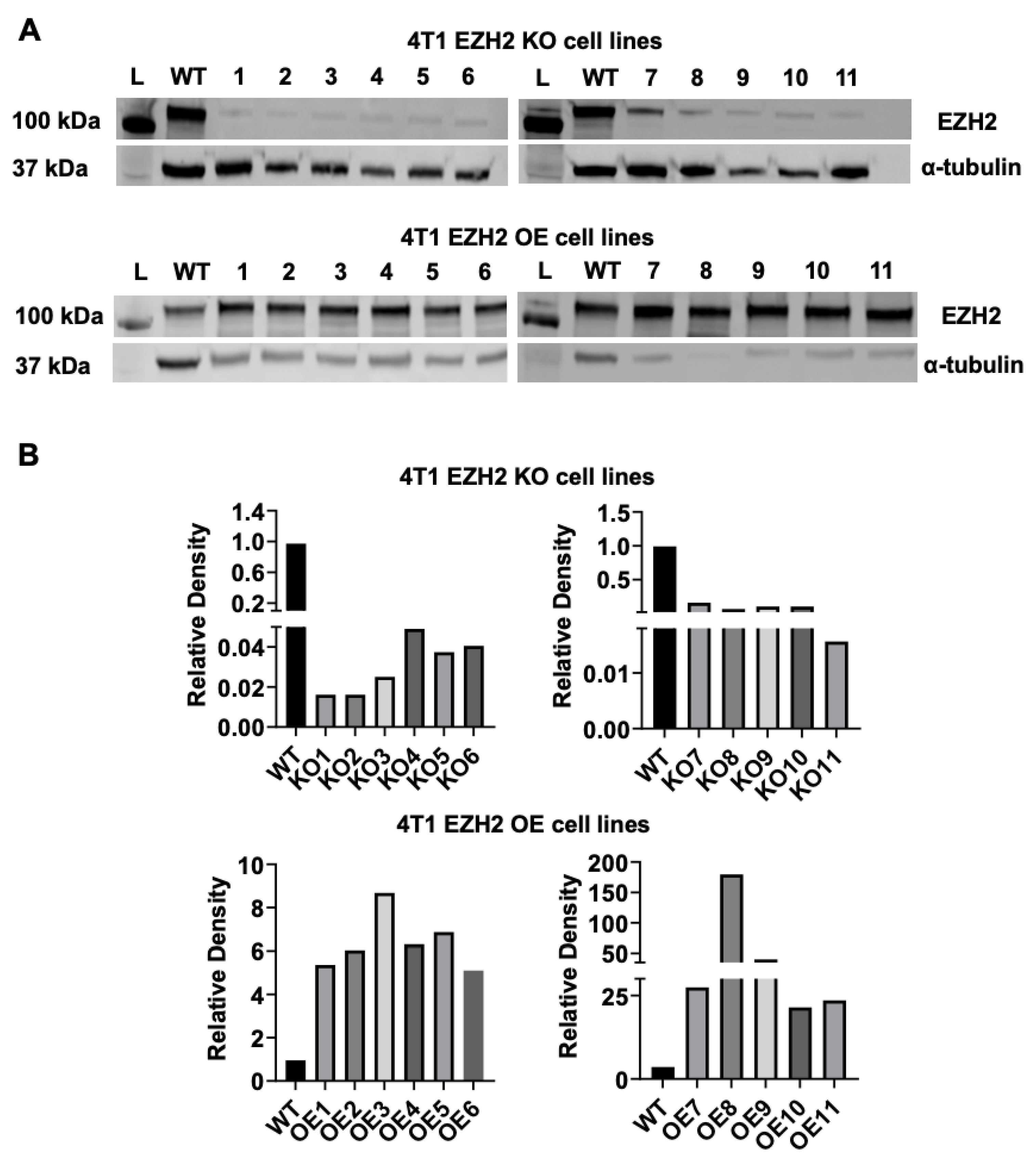
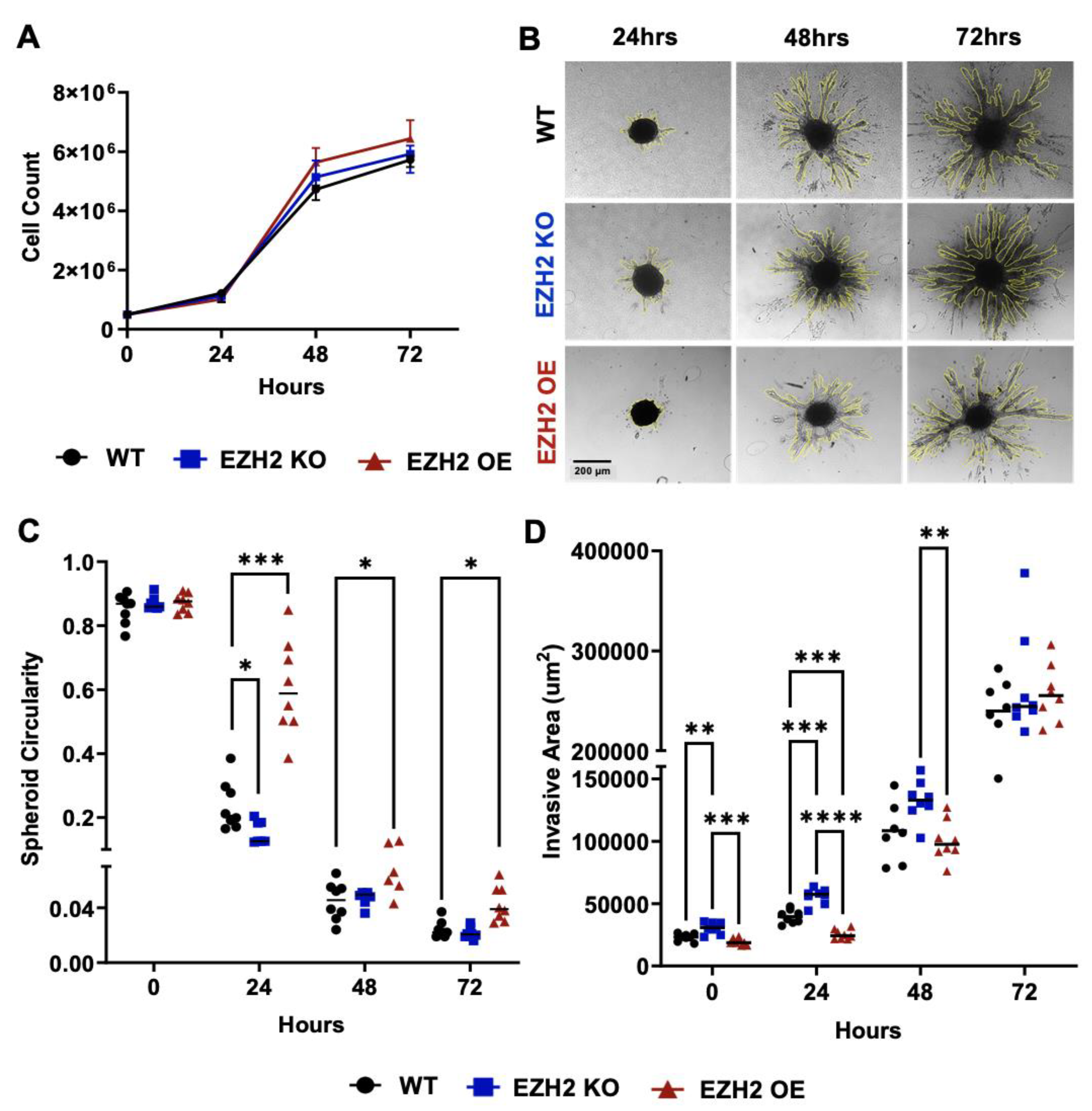
2.1. EZH2 Knockout and Overexpressing Lines Derived from the 4T1 TNBC Model Behave Similarly to the Parent Line In Vitro
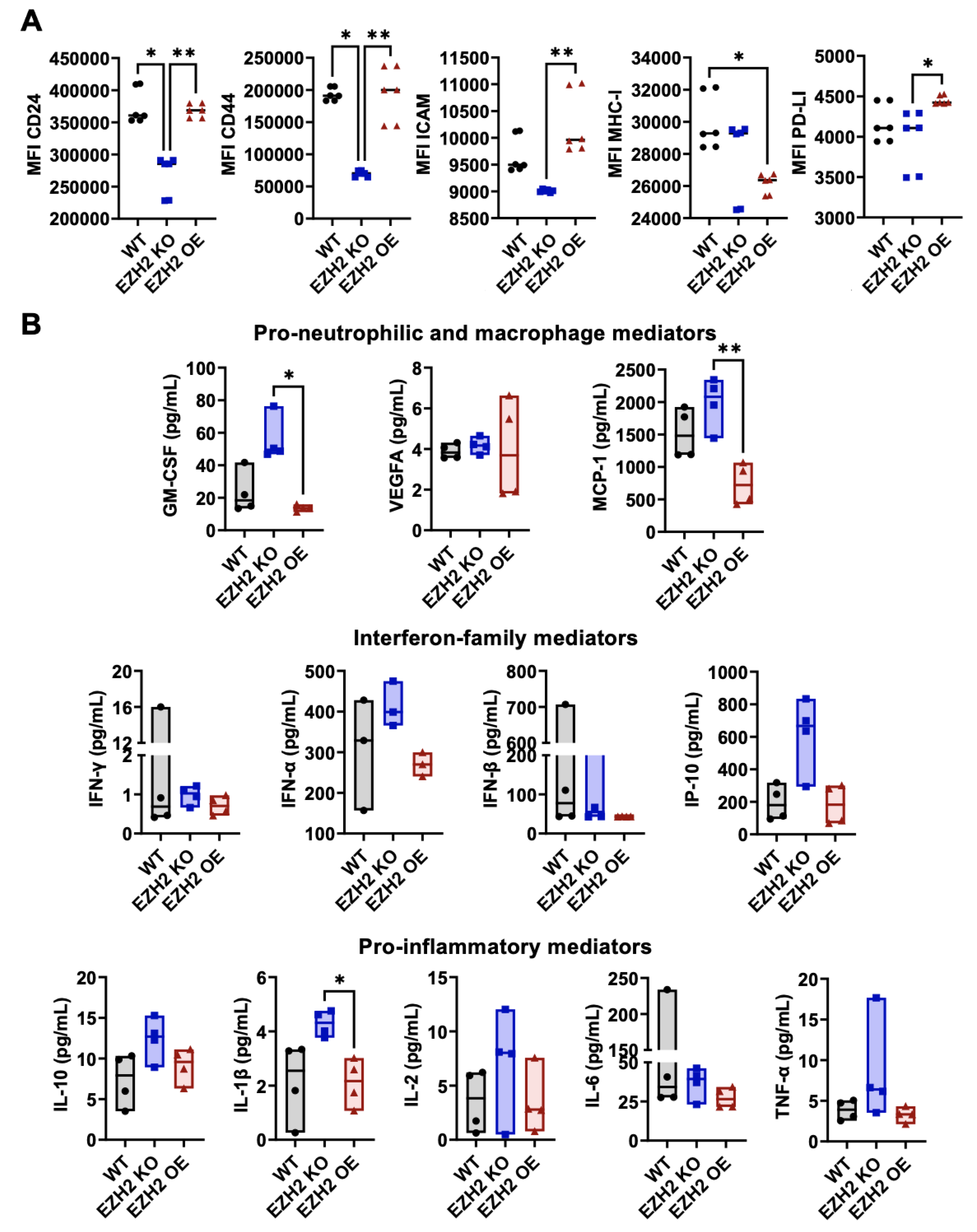
2.2. EZH2 Expression Impacts Surface Phenotype and Secreted Mediators of 4T1 Cells In Vitro
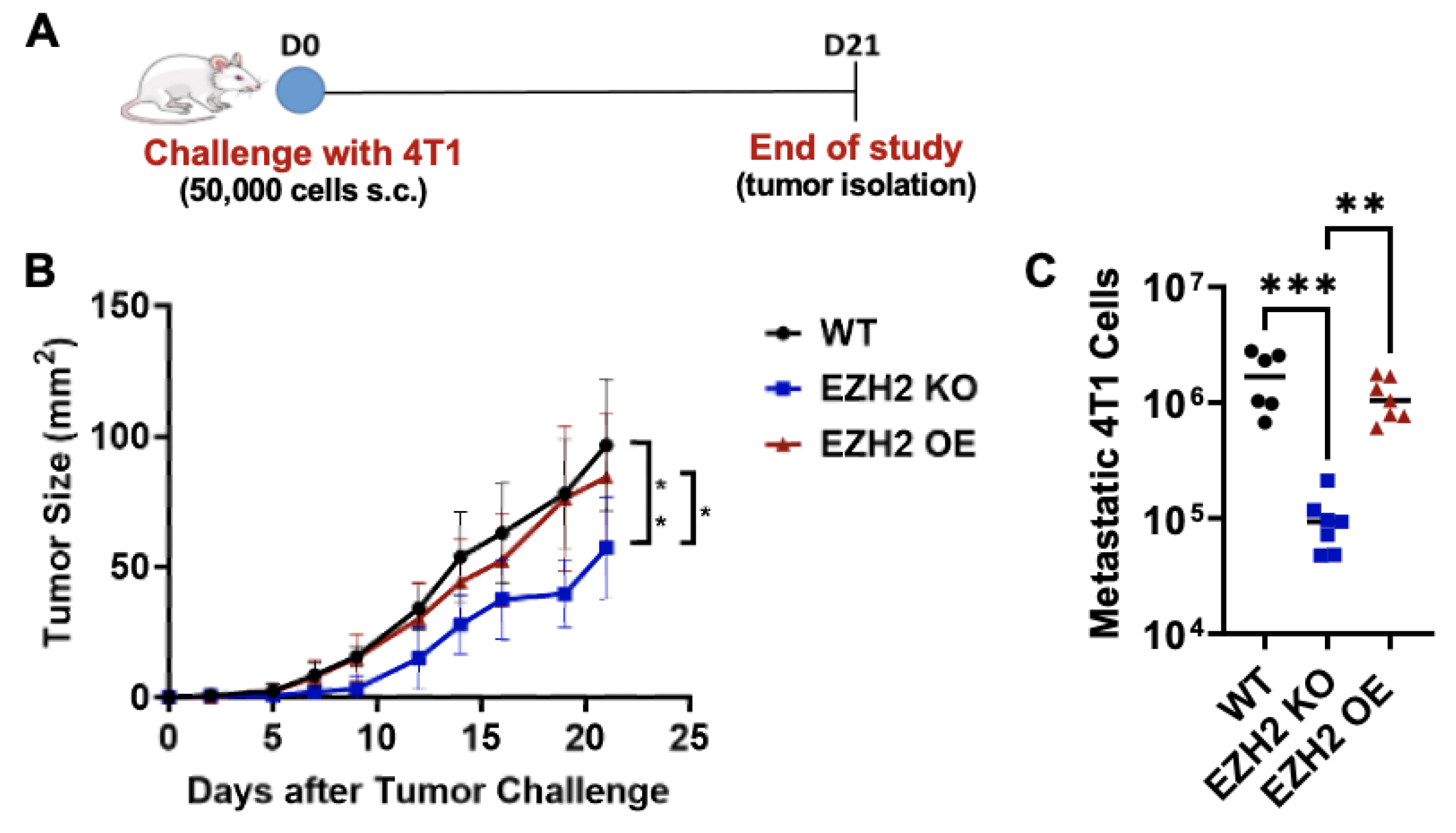
2.3. EZH2 Knockout Reduces Primary Tumor Growth and Lung Metastasis of 4T1 Cells In Vivo
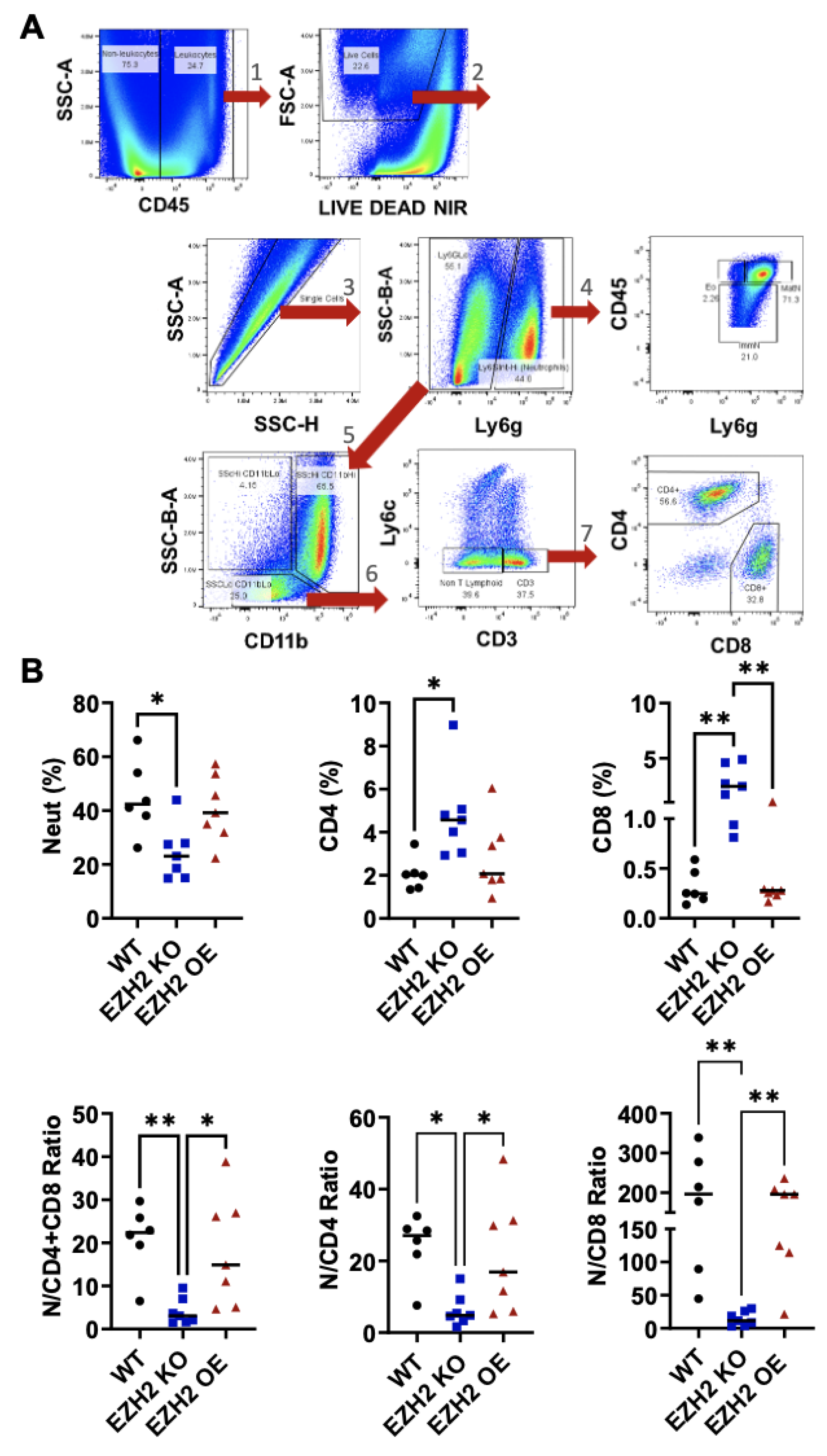
2.4. Tumor-Intrinsic EZH2 Knockout Alters the Balance of Neutrophils, CD4+ and CD8+ T-Cells in Primary 4T1 Tumors
3. Discussion
4. Materials and Methods
4.1. Cell Lines
4.2. Western Blot
4.3. Cell Proliferation Assay
4.4. 3D Spheroid Invasion Assay
4.5. Extracellular Mediator Assay
4.6. Animals
4.7. Lung Metastasis Assay
4.8. Flow Cytometry Staining and Data Acquisition
4.9. Statistical Analysis
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Arnold, M.; et al. Current and future burden of breast cancer: Global statistics for 2020 and 2040. Breast 2022, 66, 15–23. [Google Scholar] [CrossRef] [PubMed]
- Zagami, P.; Carey, L.A. Triple negative breast cancer: Pitfalls and progress. NPJ Breast Cancer 2022, 8, 95. [Google Scholar] [CrossRef] [PubMed]
- Aysola, K.; et al. Triple Negative Breast Cancer - An Overview. Hereditary Genet, 2013. 2013(Suppl 2).
- Wang, L.; et al. Hot and cold tumors: Immunological features and the therapeutic strategies. MedComm (2020) 2023, 4, e343. [Google Scholar] [CrossRef] [PubMed]
- Hong, R. and B. Xu, Breast cancer: an up-to-date review and future perspectives. Cancer Commun (Lond) 2022, 42, 913–936. [Google Scholar] [CrossRef] [PubMed]
- Peddi, P.F., M. J. Ellis, and C. Ma, Molecular basis of triple negative breast cancer and implications for therapy. Int J Breast Cancer 2012, 2012, 217185. [Google Scholar] [CrossRef] [PubMed]
- Sun, S.; et al. EZH2, a prominent orchestrator of genetic and epigenetic regulation of solid tumor microenvironment and immunotherapy. Biochim Biophys Acta Rev Cancer 2022, 1877, 188700. [Google Scholar] [CrossRef] [PubMed]
- Adibfar, S.; et al. The molecular mechanisms and therapeutic potential of EZH2 in breast cancer. Life Sci 2021, 286, 120047. [Google Scholar] [CrossRef]
- Duan, D.; et al. EZH2-CCF-cGAS Axis Promotes Breast Cancer Metastasis. Int J Mol Sci. 2022, 23. [Google Scholar] [CrossRef]
- Zhang, L.; et al. EZH2 engages TGFbeta signaling to promote breast cancer bone metastasis via integrin beta1-FAK activation. Nat Commun 2022, 13, 2543. [Google Scholar] [CrossRef]
- Liu, Y. and Q. Yang, The roles of EZH2 in cancer and its inhibitors. Med Oncol 2023, 40, 167. [Google Scholar] [CrossRef]
- Pulaski, B.A. and S. Ostrand-Rosenberg, Mouse 4T1 breast tumor model. Curr Protoc Immunol, 2001. Chapter 20: p. Unit 20 2.
- Wagner, T.E.; et al. Small-molecule-based regulation of RNA-delivered circuits in mammalian cells. Nat Chem Biol 2018, 14, 1043–1050. [Google Scholar] [CrossRef] [PubMed]
- Beck, D.B.; et al. In vivo proximity labeling for the detection of protein-protein and protein-RNA interactions. J Proteome Res 2014, 13, 6135–6143. [Google Scholar] [CrossRef] [PubMed]
- Vo, B.T.; et al. Inactivation of Ezh2 Upregulates Gfi1 and Drives Aggressive Myc-Driven Group 3 Medulloblastoma. Cell Rep 2017, 18, 2907–2917. [Google Scholar] [CrossRef] [PubMed]
- Ran, F.A.; et al. Genome engineering using the CRISPR-Cas9 system. Nat Protoc 2013, 8, 2281–2308. [Google Scholar] [CrossRef] [PubMed]
- Summerbell, E.R.; et al. Epigenetically heterogeneous tumor cells direct collective invasion through filopodia-driven fibronectin micropatterning. Sci Adv 2020, 6, eaaz6197. [Google Scholar] [CrossRef] [PubMed]
- Munoz, L.E.; et al. Metformin reduces PD-L1 on tumor cells and enhances the anti-tumor immune response generated by vaccine immunotherapy. J Immunother Cancer, 2021. 9(11).
- Mathenge, E.G.; et al. Core needle biopsy of breast cancer tumors increases distant metastases in a mouse model. Neoplasia 2014, 16, 950–960. [Google Scholar] [CrossRef] [PubMed]
- Flood, B.A.; et al. STING pathway agonism as a cancer therapeutic. Immunol Rev 2019, 290, 24–38. [Google Scholar] [CrossRef] [PubMed]
- Walker Ii, W.H.; et al. Mammary Tumors Induce Central Pro-inflammatory Cytokine Expression, but Not Behavioral Deficits in Balb/C Mice. Sci Rep 2017, 7, 8152. [Google Scholar] [CrossRef] [PubMed]
- DuPre, S.A., D. Redelman, and K.W. Hunter, Jr., The mouse mammary carcinoma 4T1: characterization of the cellular landscape of primary tumours and metastatic tumour foci. Int J Exp Pathol 2007, 88, 351–360. [Google Scholar] [CrossRef] [PubMed]
- Zhang, L.; et al. Blocking immunosuppressive neutrophils deters pY696-EZH2-driven brain metastases. Sci Transl Med, 2020. 12(545).
- Liu, X.; et al. Epithelial-type systemic breast carcinoma cells with a restricted mesenchymal transition are a major source of metastasis. Sci Adv 2019, 5, eaav4275. [Google Scholar] [CrossRef]
- Zheng, C.; et al. Neutrophils in triple-negative breast cancer: an underestimated player with increasingly recognized importance. Breast Cancer Res 2023, 25, 88. [Google Scholar] [CrossRef] [PubMed]
- Arpinati, L.; et al. Tumor-Derived Factors Differentially Affect the Recruitment and Plasticity of Neutrophils. Cancers (Basel), 2021. 13(20).
- Uribe-Querol, E. and C. Rosales, Neutrophils in Cancer: Two Sides of the Same Coin. J Immunol Res 2015, 2015, 983698. [Google Scholar] [CrossRef] [PubMed]
- Hurt, B.; et al. Cancer-promoting mechanisms of tumor-associated neutrophils. Am J Surg 2017, 214, 938–944. [Google Scholar] [CrossRef]
- Brandau, S., C. A. Dumitru, and S. Lang, Protumor and antitumor functions of neutrophil granulocytes. Semin Immunopathol 2013, 35, 163–176. [Google Scholar] [CrossRef]
- Liu, Y.; et al. Advances in immunotherapy for triple-negative breast cancer. Mol Cancer 2023, 22, 145. [Google Scholar] [CrossRef]
- Kim, I.S.; et al. Immuno-subtyping of breast cancer reveals distinct myeloid cell profiles and immunotherapy resistance mechanisms. Nat Cell Biol 2019, 21, 1113–1126. [Google Scholar] [CrossRef]
- Kitchen, G.B.; et al. The histone methyltransferase Ezh2 restrains macrophage inflammatory responses. Faseb j 2021, 35, e21843. [Google Scholar] [CrossRef] [PubMed]
- Koss, B.; et al. Epigenetic Control of Cdkn2a.Arf Protects Tumor-Infiltrating Lymphocytes from Metabolic Exhaustion. Cancer Res 2020, 80, 4707–4719. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).



