Submitted:
01 April 2024
Posted:
02 April 2024
You are already at the latest version
Abstract
Keywords:
1. Introduction
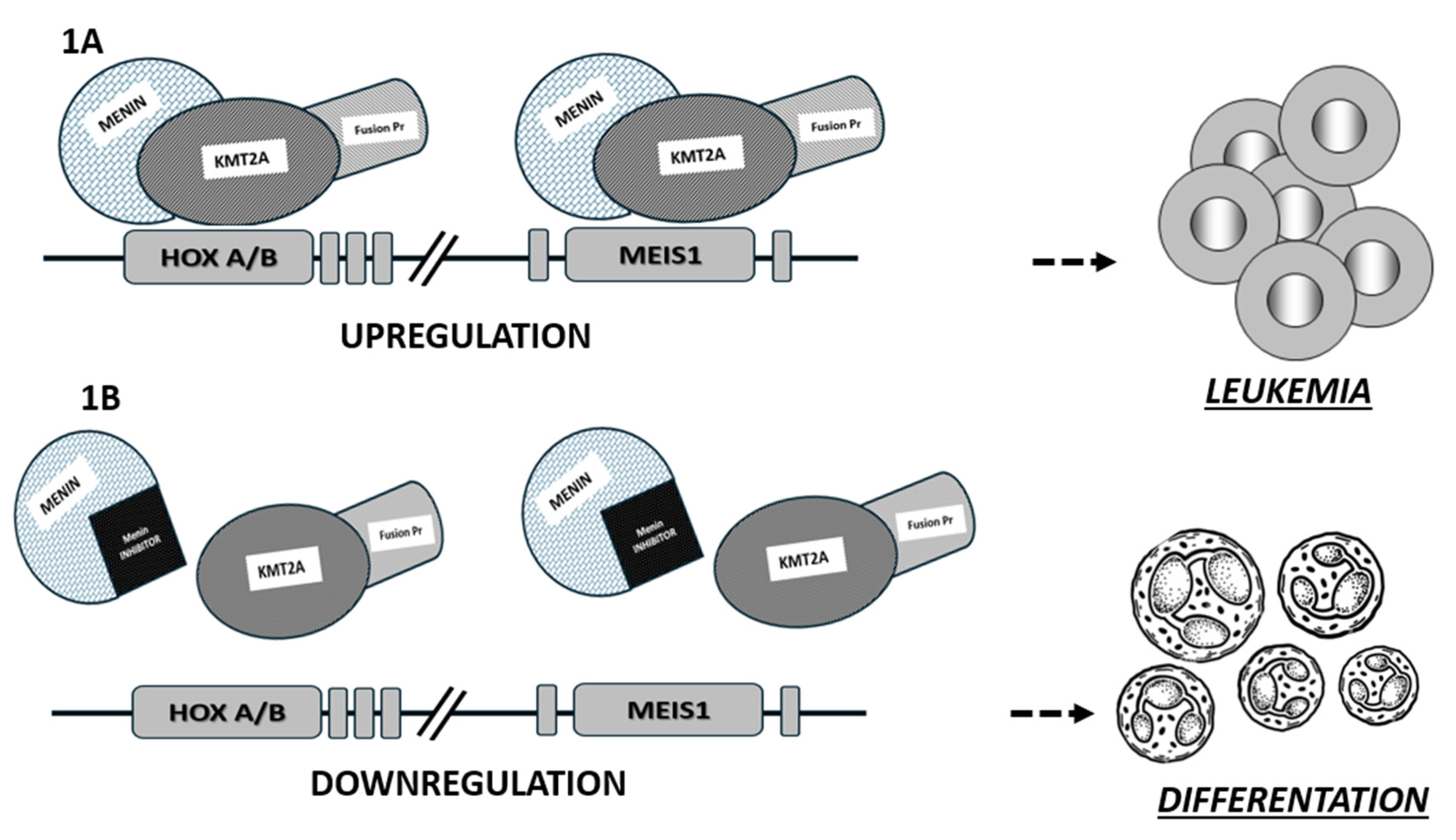
2. Role of Menin in KMT2Ar and NPM1mut AML
3. Targeting Menin in AML
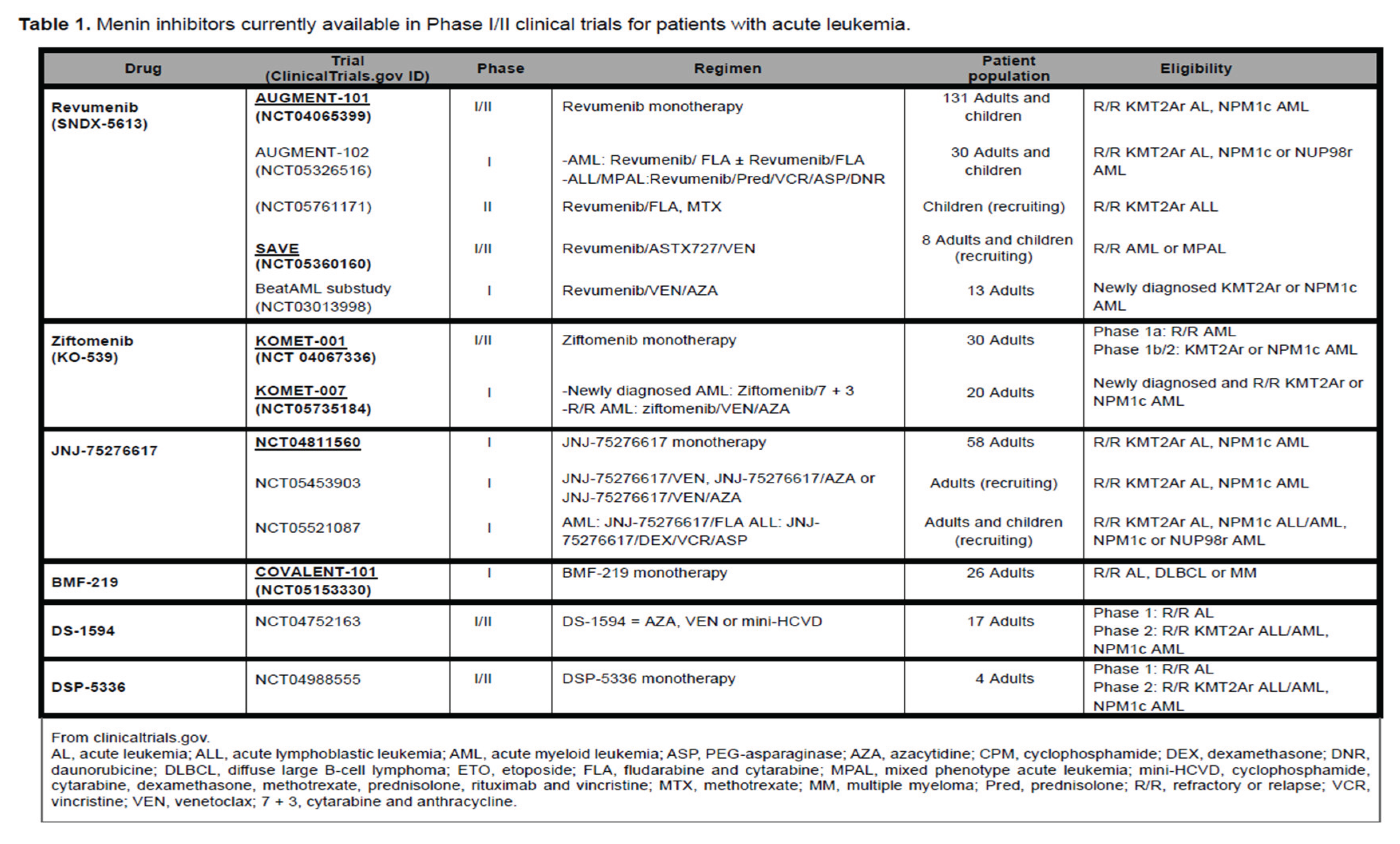
4. Menin Inhibitors: Ongoing Clinical Trials in AML
4.a. Revumenib
4.b. Ziftomenib
4.c. Other Menin Inhibitors under Investigation
4.d. Menin Inhibitors and Venetoclax Combinations
4.e. Menin Inhibitors and Other Combinations
5. Conclusions
References
- DiNardo, C.D.; Erba, H.P.; Freeman, S.D.; Wei, A.H. Acute myeloid leukaemia. The Lancet. 2023, 401(10393), 2073–86. [Google Scholar] [CrossRef] [PubMed]
- Shimony S; Stahl M; Stone RM. Acute myeloid leukemia: 2023 update on diagnosis, risk-stratification, and management. Am J Hematol. 2023, 98(3), 502–26. [CrossRef]
- Döhner H; Wei AH; Appelbaum FR; Craddock C; DiNardo CD; Dombret H; et al. Diagnosis and management of AML in adults: 2022 recommendations from an international expert panel on behalf of the ELN. Blood. 2022, 140(12), 1345–77. [CrossRef]
- Wolffhardt TM; Ketzer F; Telese S; Wirth T; Ushmorov A. Dependency of B-Cell Acute Lymphoblastic Leukemia and Multiple Myeloma Cell Lines on MEN1 Extends beyond MEN1–KMT2A Interaction. Int J Mol Sci. 2023, 24(22), 16472. [CrossRef]
- Issa GC; Ravandi F; DiNardo CD; Jabbour E; Kantarjian HM; Andreeff M. Therapeutic implications of menin inhibition in acute leukemias. Leukemia. 2021, 35(9), 2482–95. [CrossRef]
- Krivtsov AV; Armstrong SA. MLL translocations, histone modifications and leukaemia stem-cell development. Nat Rev Cancer. 2007, 7(11), 823–33. [CrossRef]
- Li X; Song Y. Structure, function and inhibition of critical protein–protein interactions involving mixed lineage leukemia 1 and its fusion oncoproteins. J Hematol Oncol. 2021, 14, 56. [CrossRef]
- Sahasrabudhe KD; Mims AS. Novel investigational approaches for high-risk genetic subsets of AML: TP53, KMT2A, FLT3. Hematology. 2022, 2022(1), 15–22. [CrossRef]
- Caslini C; Yang Z; El-Osta M; Milne TA; Slany RK; Hess JL. Interaction of MLL amino terminal sequences with menin is required for transformation. Cancer Res. 2007, 67(15), 7275–83. [CrossRef]
- Cierpicki T; Grembecka J. Challenges and opportunities in targeting the menin–MLL interaction. Future Med Chem. 2014, 6(4), 447–62. [CrossRef]
- Uckelmann HJ, Haarer EL, Takeda R, Wong EM, Hatton C, Marinaccio C, Perner F, Rajput M, Antonissen NJC, Wen YYang L, Brunetti L, Chen CW, Armstrong SA. Mutant NPM1 Directly Regulates Oncogenic Transcription in Acute Myeloid Leukemia. Cancer Discov. 2023,13(3), 746-765. [CrossRef]
- Fiskus W; Daver N; Boettcher S; Mill CP; Sasaki K; Birdwell CE; et al. Activity of menin inhibitor ziftomenib (KO-539) as monotherapy or in combinations against AML cells with MLL1 rearrangement or mutant NPM1. Leukemia. 2022, 36(11), 2729–33. [CrossRef]
- Krivtsov AV; Evans K; Gadrey JY; Eschle BK; Hatton C; Uckelmann HJ; et al. A Menin-MLL Inhibitor Induces Specific Chromatin Changes and Eradicates Disease in Models of MLL-Rearranged Leukemia. Cancer Cell. 2019, 36(6), 660-673.e11. [CrossRef]
- Sharma N; Liesveld JL. NPM 1 Mutations in AML—The Landscape in 2023. Cancers. 2023, 15(4), 1177. [CrossRef]
- Yu Y; Maggi LB; Brady SN; Apicelli AJ; Dai MS; Lu H; et al. Nucleophosmin is essential for ribosomal protein L5 nuclear export. Mol Cell Biol. 2006, 26(10), 3798–809. [CrossRef]
- Falini B; Brunetti L; Sportoletti P; Martelli MP. NPM1-mutated acute myeloid leukemia: from bench to bedside. Blood. 2020, 136(15), 1707–21. [CrossRef]
- Barajas JM; Rasouli M; Umeda M; Hiltenbrand RL; Abdelhamed S; Mohnani R; et al. Acute myeloid leukemias with UBTF tandem duplications are sensitive to Menin inhibitors. Blood. 2023, blood.2023021359. [CrossRef]
- Heikamp EB, Henrich JA, Perner F, et al. The menin-MLL1 interaction is a molecular dependency in NUP98-rearranged AML. Blood. 2022, 139, 894-906. [CrossRef]
- Papaemmanuil E; Gerstung M; Bullinger L; Gaidzik VI; Paschka P; Roberts ND; et al. Genomic Classification and Prognosis in Acute Myeloid Leukemia. N Engl J Med. 2016, 374(23), 2209–21. [CrossRef]
- Grembecka J; Belcher AM; Hartley T; Cierpicki T. Molecular basis of the mixed lineage leukemia-menin interaction: implications for targeting mixed lineage leukemias. J Biol Chem. 2010, 285(52), 40690–8. [CrossRef]
- Perner F; Stein EM; Wenge DV; Singh S; Kim J; Apazidis A; et al. MEN1 mutations mediate clinical resistance to menin inhibition. Nature. 2023, 615(7954), 913–9. [CrossRef]
- Stein EM; Aldoss I; DiPersio JF; Stone RM; Arellano ML; Rosen G; et al. Safety and Efficacy of Menin Inhibition in Patients (Pts) with MLL-Rearranged and NPM1 Mutant Acute Leukemia: A Phase (Ph) 1, First-in-Human Study of SNDX-5613 (AUGMENT 101). Blood. 2021,138(Supplement 1), 699. [CrossRef]
- Issa GC; Aldoss I; DiPersio JF; Cuglievan B; Stone RM; Arellano ML; et al. The Menin Inhibitor SNDX-5613 (revumenib) Leads to Durable Responses in Patients (Pts) with KMT2A-Rearranged or NPM1 Mutant AML: Updated Results of a Phase (Ph) 1 Study. Blood. 2022, 140(Supplement 1), 150–2. [CrossRef]
- Premnath N; Madanat YF. Novel Investigational Agents and Pathways That May Influence the Future Management of Acute Myeloid Leukemia. Cancers. 2023, 15(11), 2958. [CrossRef]
- Aldoss I; Issa GC; Thirman MJ; DiPersio J; Arellano M; Blachly JS; et al. Revumenib Monotherapy in Patients with Relapsed/Refractory KMT2Ar Acute Leukemias: Efficacy and Safety Results from the Augment-101 Phase 1/2 Study. Blood. 2023, 142(Supplement 1), 2907–2907. [CrossRef]
- Wang E; Altman J; Pettit K; Botton S; Walter R; Fenaux P; et al. Preliminary Data on a Phase 1/2A First in Human Study of the Menin-KMT2A (MLL) Inhibitor KO-539 in Patients with Relapsed or Refractory Acute Myeloid Leukemia. Blood. 2020, 136, 7–8. [CrossRef]
- Erba H; Fathi A; Issa G; Altman J; Montesinos P; Patnaik M; et al. Update on a Phase 1/2 First-in-Human Study of the Menin-KMT2A (MLL) Inhibitor Ziftomenib (KO-539) in Patients with Relapsed or Refractory Acute Myeloid Leukemia. Blood. 2022, 140, 153–6. [CrossRef]
- DiNardo KW; LeBlanc TW; Chen H. Novel agents and regimens in acute myeloid leukemia: latest updates from 2022 ASH Annual Meeting. J Hematol Oncol. 2023, 16(1) ,17. [CrossRef]
- Buchrits S; Wolach O. Non-Immunotherapy Approaches for Relapsed or Refractory AML: An Update for 2024. Acta Haematol. 2023, 1–16. [CrossRef]
- Numata M; Haginoya N; Shiroishi M; Hirata T; Sato-Otsubo A; Yoshikawa K; et al. A novel Menin-MLL1 inhibitor, DS-1594a, prevents the progression of acute leukemia with rearranged MLL1 or mutated NPM1. Cancer Cell Int. 2023, 23(1), 36. [CrossRef]
- Daver N; Zeidner JF; Yuda J; Watts JM; Levis MJ; Fukushima K; et al. Phase 1/2 First-in-Human Study of the Menin-MLL Inhibitor DSP-5336 in Patients with Relapsed or Refractory Acute Leukemia. Blood. 2023, 142(Supplement 1), 2911–2911. [CrossRef]
- Kwon MC; Querolle O; Dai X; Thuring JW; Verhulst T; Marien A; et al. Pharmacological Characterization of JNJ-75276617, a Menin-KMT2A Inhibitor, As Targeted Treatment for KMT2A-Altered and NPM1-Mutant Acute Leukemia. Blood. 2022, 140(Supplement 1), 5928–9. [CrossRef]
- Jabbour E; Searle E; Abdul-Hay M; Abedin S; Aldoss I; Alfonso Piérola A; et al. A First-in-Human Phase 1 Study of the Menin-KMT2A (MLL1) Inhibitor JNJ-75276617 in Adult Patients with Relapsed/Refractory Acute Leukemia Harboring KMT2A or NPM1 Alterations. Blood. 2023, 142(Supplement 1), 57. [CrossRef]
- Lancet J; Ravandi F; Montesinos P; Barrientos JC; Badar T; Alegre A; et al. Covalent Menin Inhibitor Bmf-219 in Patients with Relapsed or Refractory (R/R) Acute Leukemia (AL): Preliminary Phase 1 Data from the Covalent-101 Study. Blood. 2023, 142(Supplement 1), 2916. [CrossRef]
- Pei S; Pollyea DA; Gustafson A; Stevens BM; Minhajuddin M; Fu R, et al. Monocytic Subclones Confer Resistance to Venetoclax-Based Therapy in Patients with Acute Myeloid Leukemia. Cancer Discov. 2020,10(4), 536–51. [CrossRef]
- Rausch J; Dzama MM; Dolgikh N; Stiller HL; Bohl SR; Lahrmann C; et al. Menin inhibitor ziftomenib (KO-539) synergizes with drugs targeting chromatin regulation or apoptosis and sensitizes acute myeloid leukemia with MLL rearrangement or NPM1 mutation to venetoclax. Haematologica. 2023, 108(10), 2837–43. [CrossRef]
- Zeidan AM; Fathi A; Issa G; Erba H; Mackey JA; Corum D; et al. PB1885: Phase 1 study of ziftomenib in combination with venetoclax, venetoclax/azacitidine or standard induction (7+3) chemotherapy in patients with acute myeloid leukemia. HemaSphere. 2023, 7(Suppl), e56956b1. [CrossRef]
- Issa GC; Cuglievan B; DiNardo CD; Short NJ; McCall D; Gibson A; et al. Early Results of the Phase I/II Study Investigating the All-Oral Combination of the Menin Inhibitor Revumenib (SNDX-5613) with Decitabine/Cedazuridine (ASTX727) and Venetoclax in Acute Myeloid Leukemia (SAVE). Blood. 2023,142(Supplement 1), 58. [CrossRef]
- Goldberg AD; Corum D; Ahsan J; Nie K; Kozlek T; Leoni M; et al. Komet-008: A Phase 1 Study to Determine the Safety and Tolerability of Ziftomenib Combinations for the Treatment of KMT2A-Rearranged or NPM1-Mutant Relapsed/Refractory Acute Myeloid Leukemia. Blood. 2023, 142(Supplement 1), 1553–1553. [CrossRef]
- Dzama MM; Steiner M; Rausch J; Sasca D; Schönfeld J; Kunz K; et al. Synergistic targeting of FLT3 mutations in AML via combined menin-MLL and FLT3 inhibition. Blood. 2020, 136(21), 2442–56. [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).




