Submitted:
27 March 2024
Posted:
27 March 2024
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
2.1. Materials
2.1.1. Minerals and Electrodes
2.1.2. Wastewater and Reagents
2.2. Methods
2.2.1. Immersion Pretreatment
2.2.2. Bench-Scale Flotation Experiments
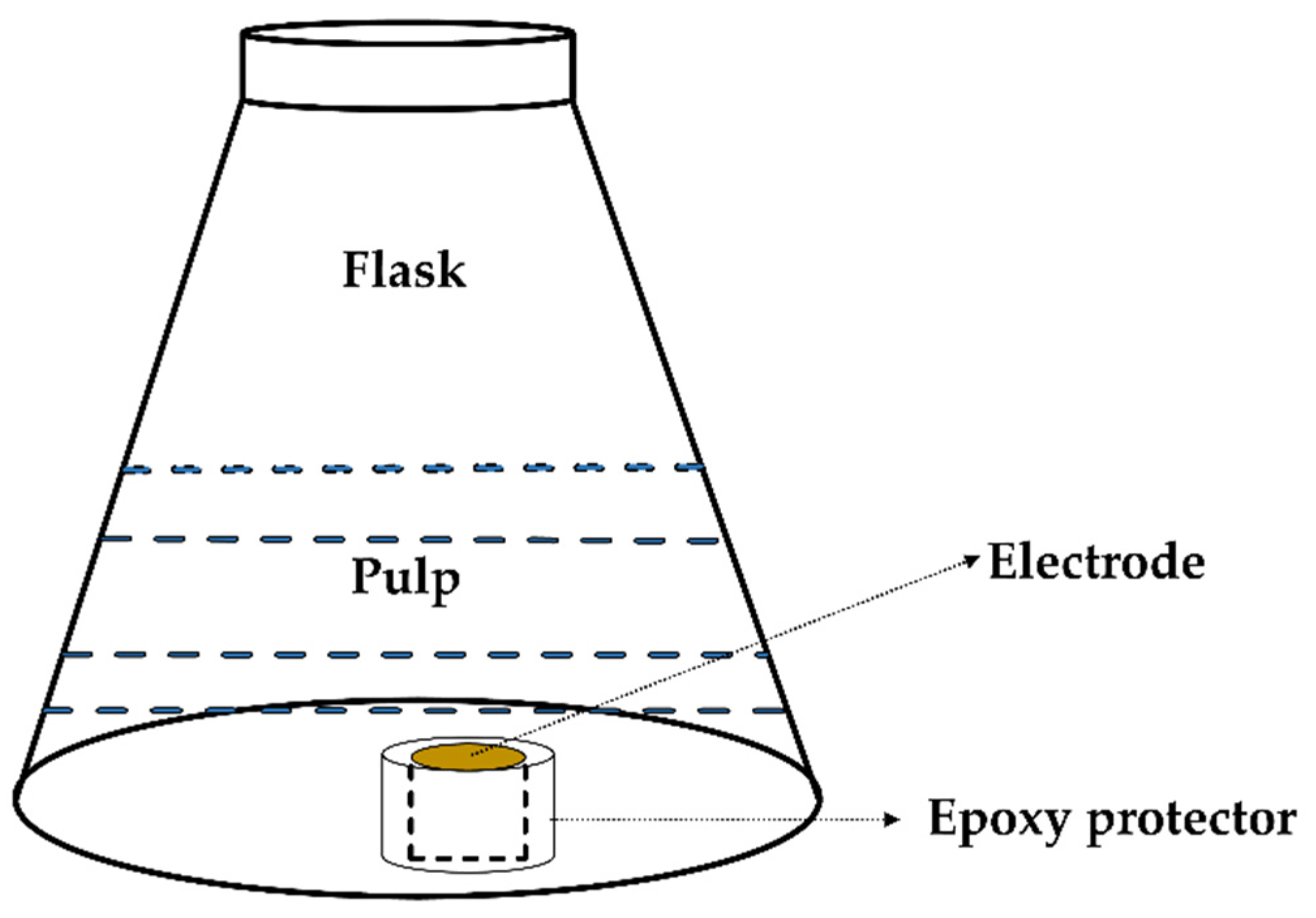
2.2.3. Electrochemical Experiments
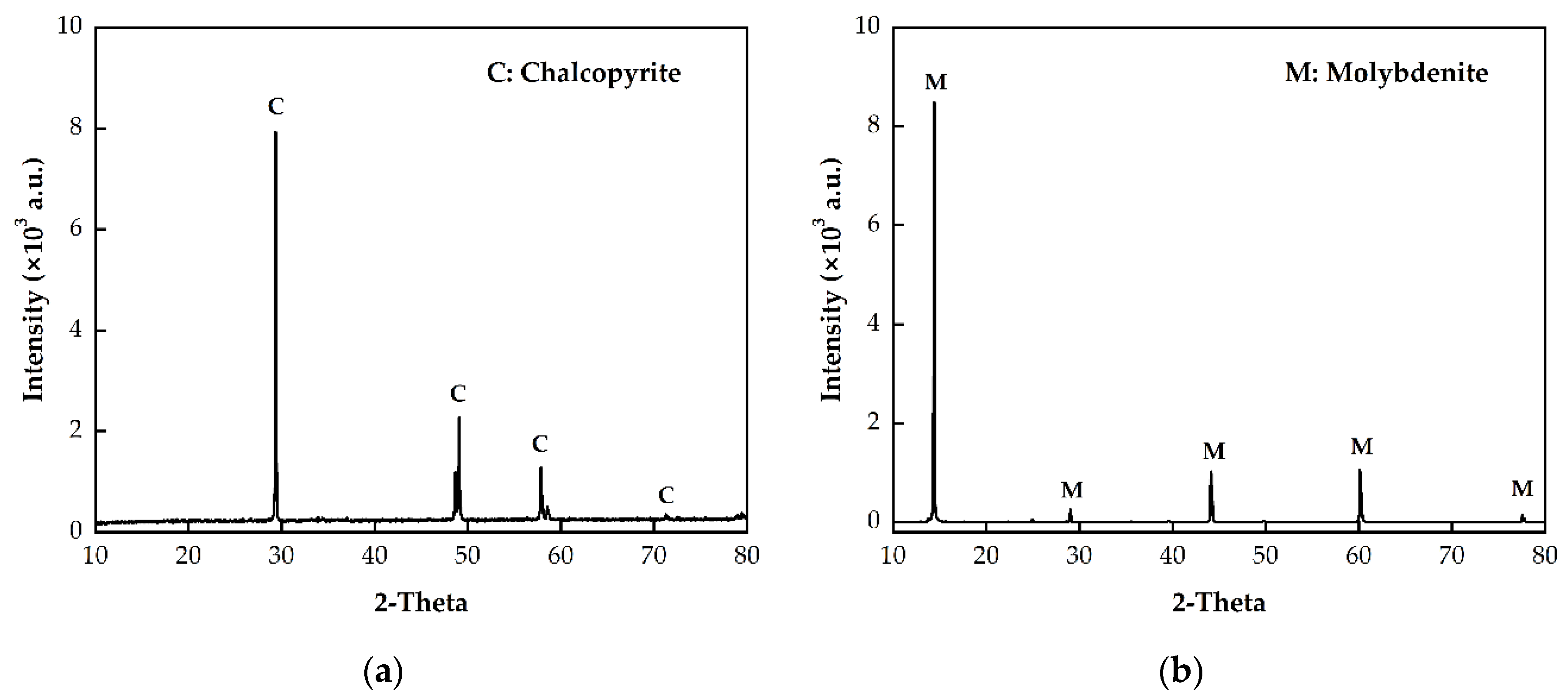
2.2.4. SEM and XRD Analysis
3. Results and Discussion
3.1. Flotation of Single Minerals
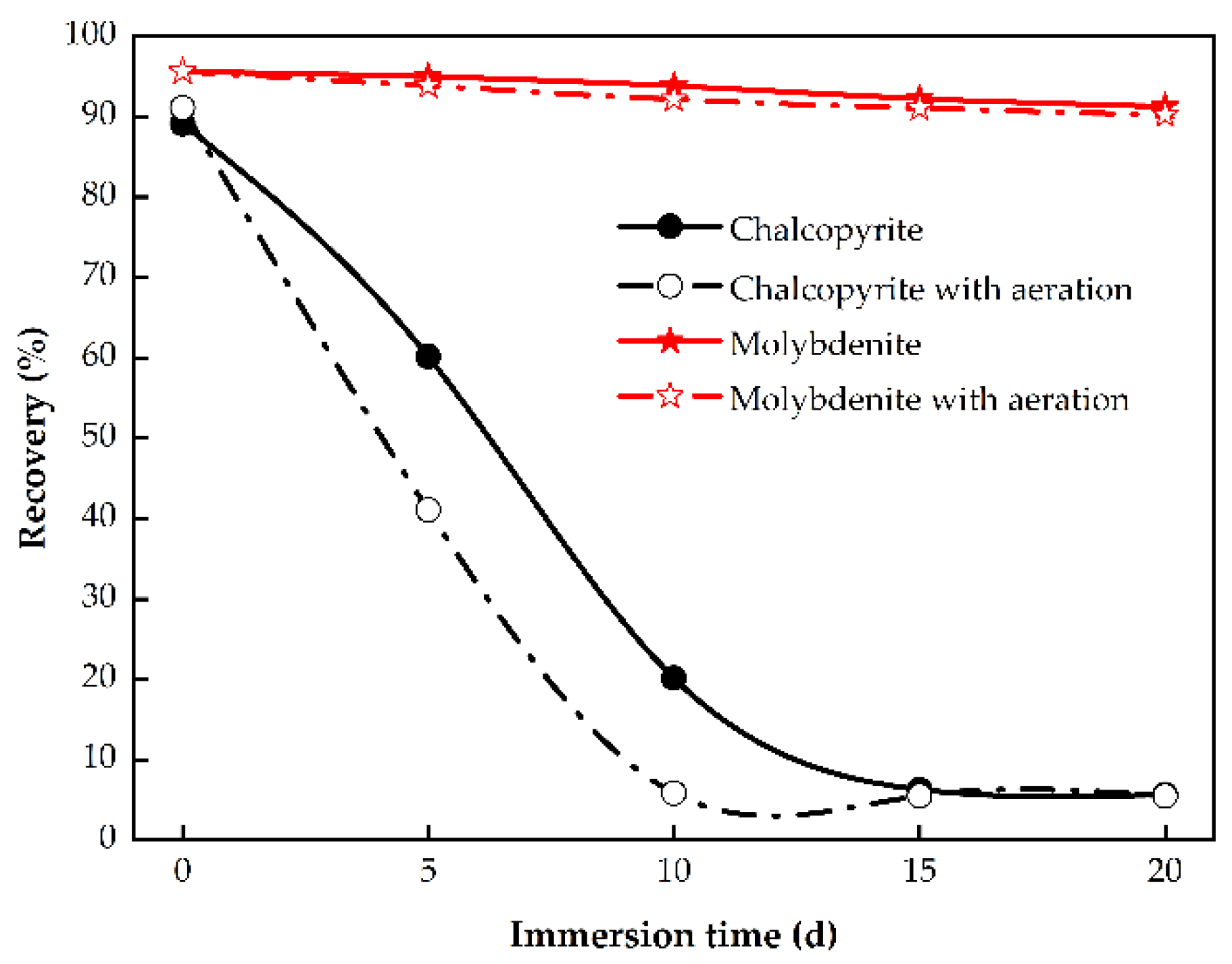
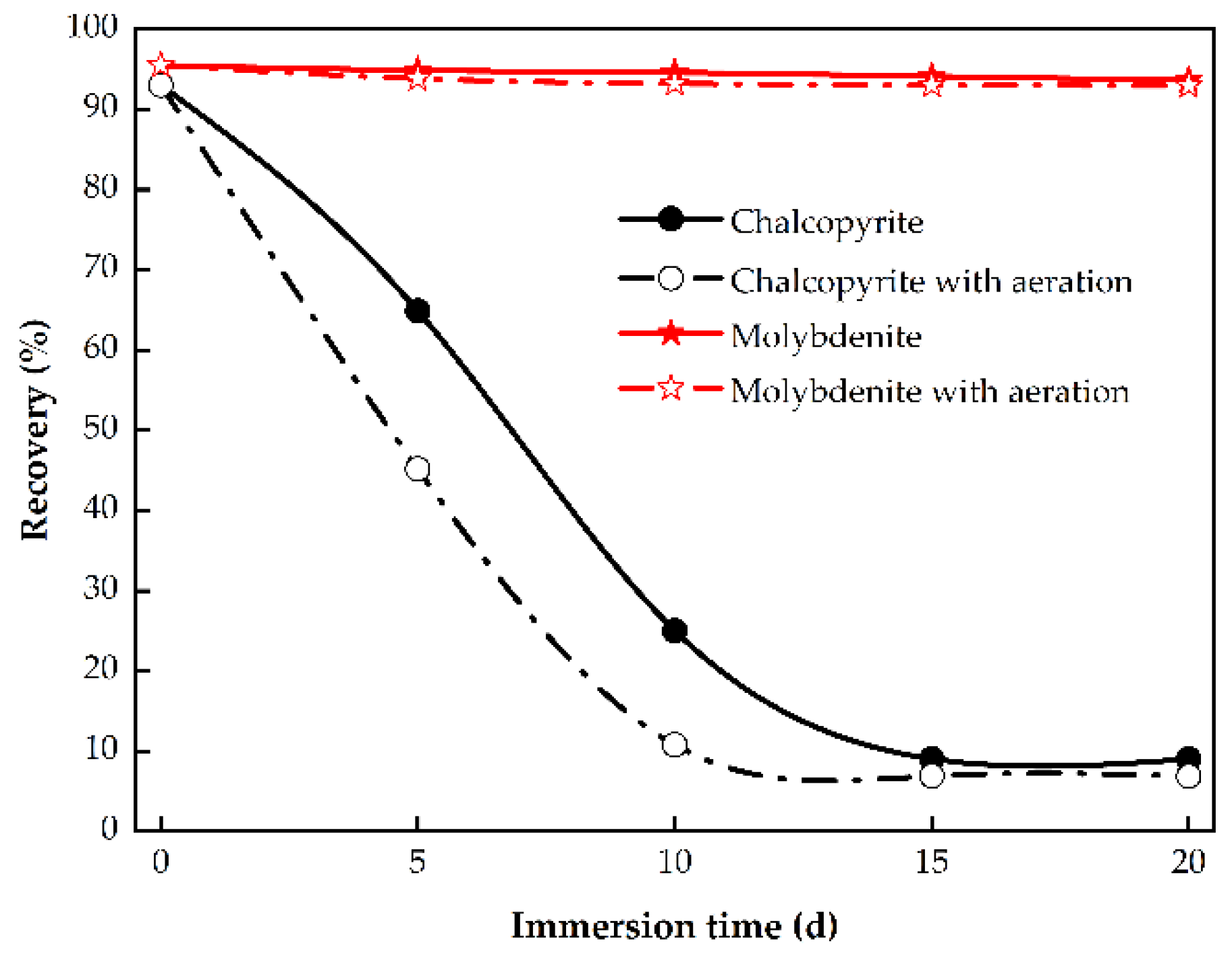
3.1.1. Effect of Immersion Time
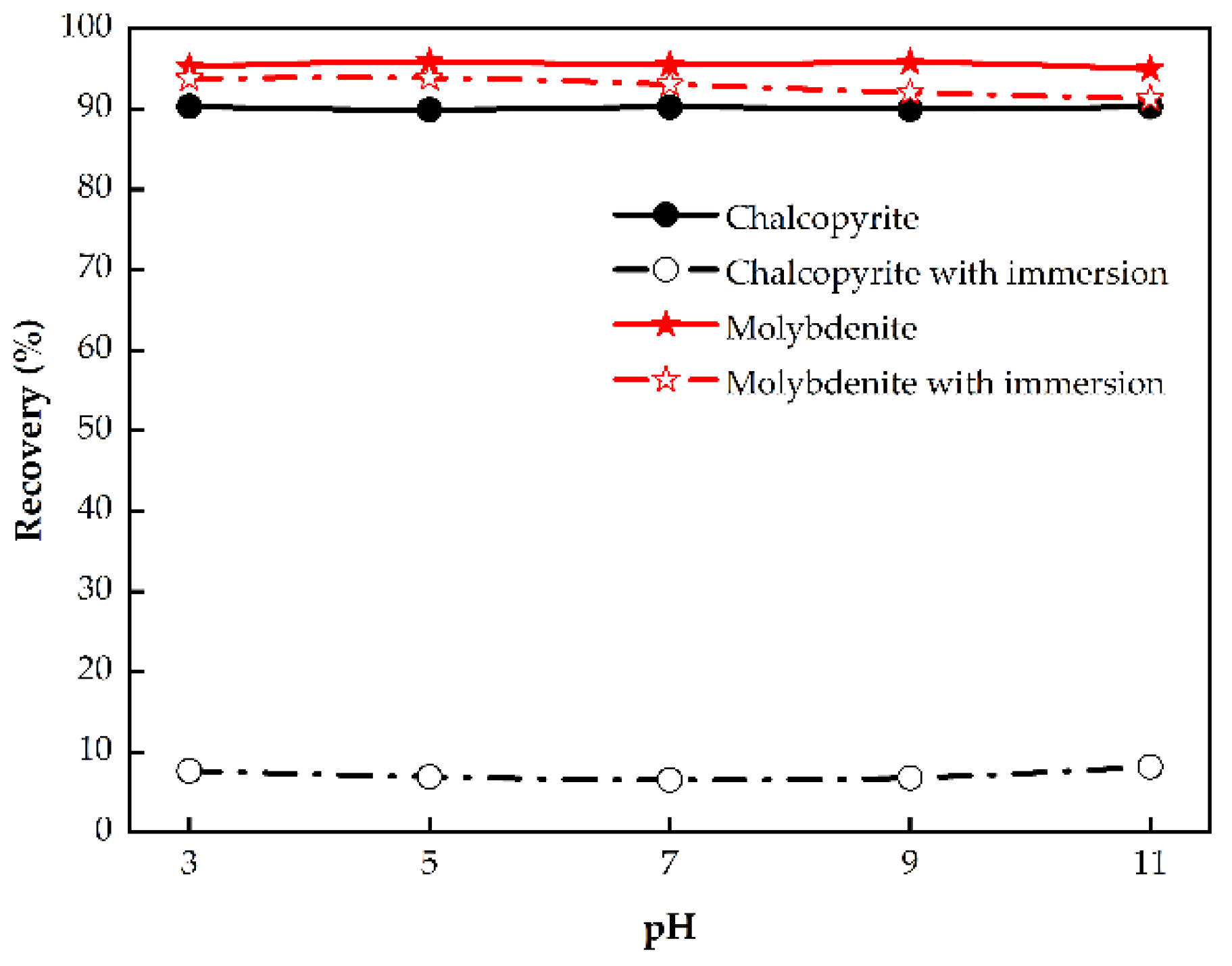
3.1.2. Effect of Pulp pH
3.1.3. Effect of Collector Dosage
3.2. Flotation of Artificially Mixed Minerals
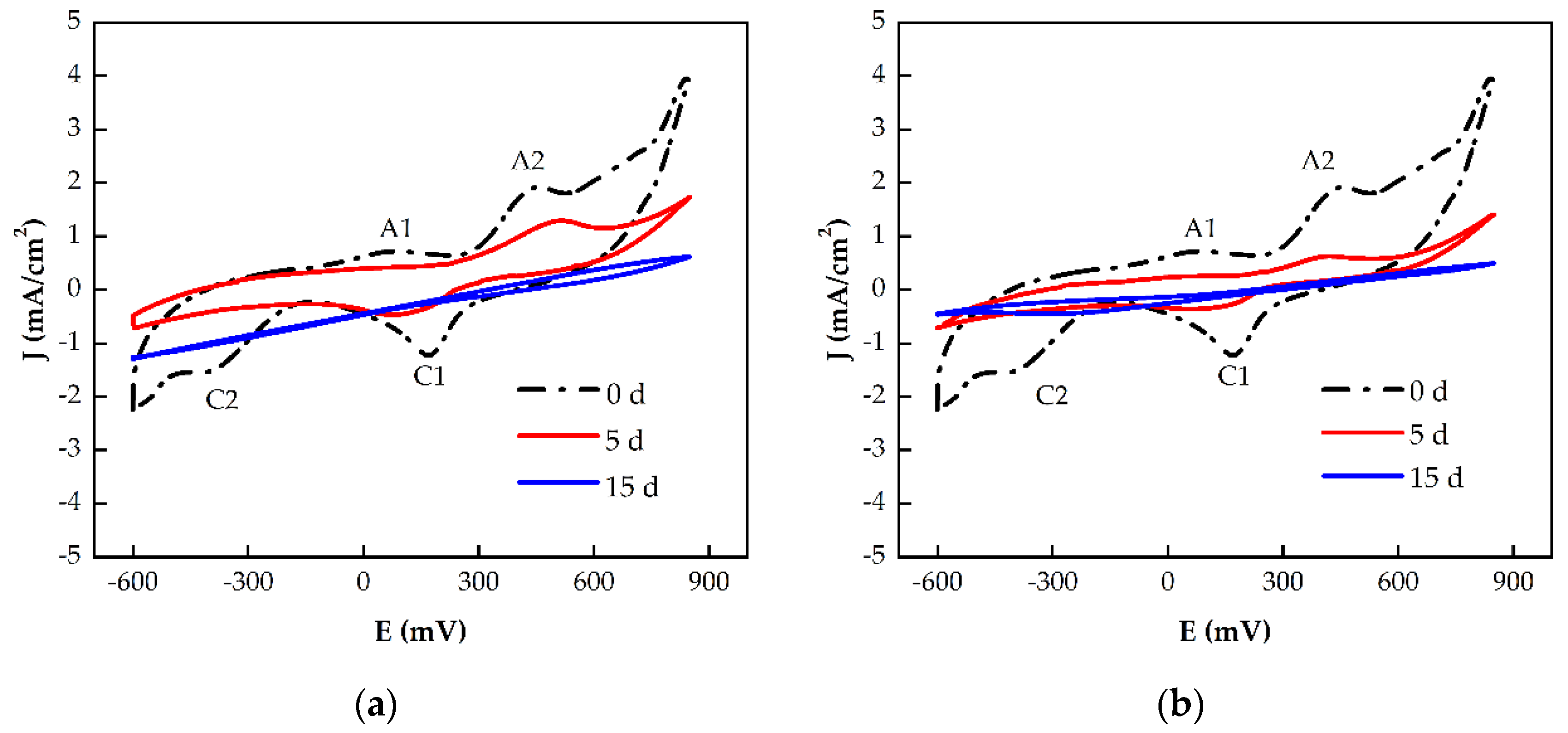
3.3. Changes in Chalcopyrite Electrochemical Properties with Immersion Pretreatment
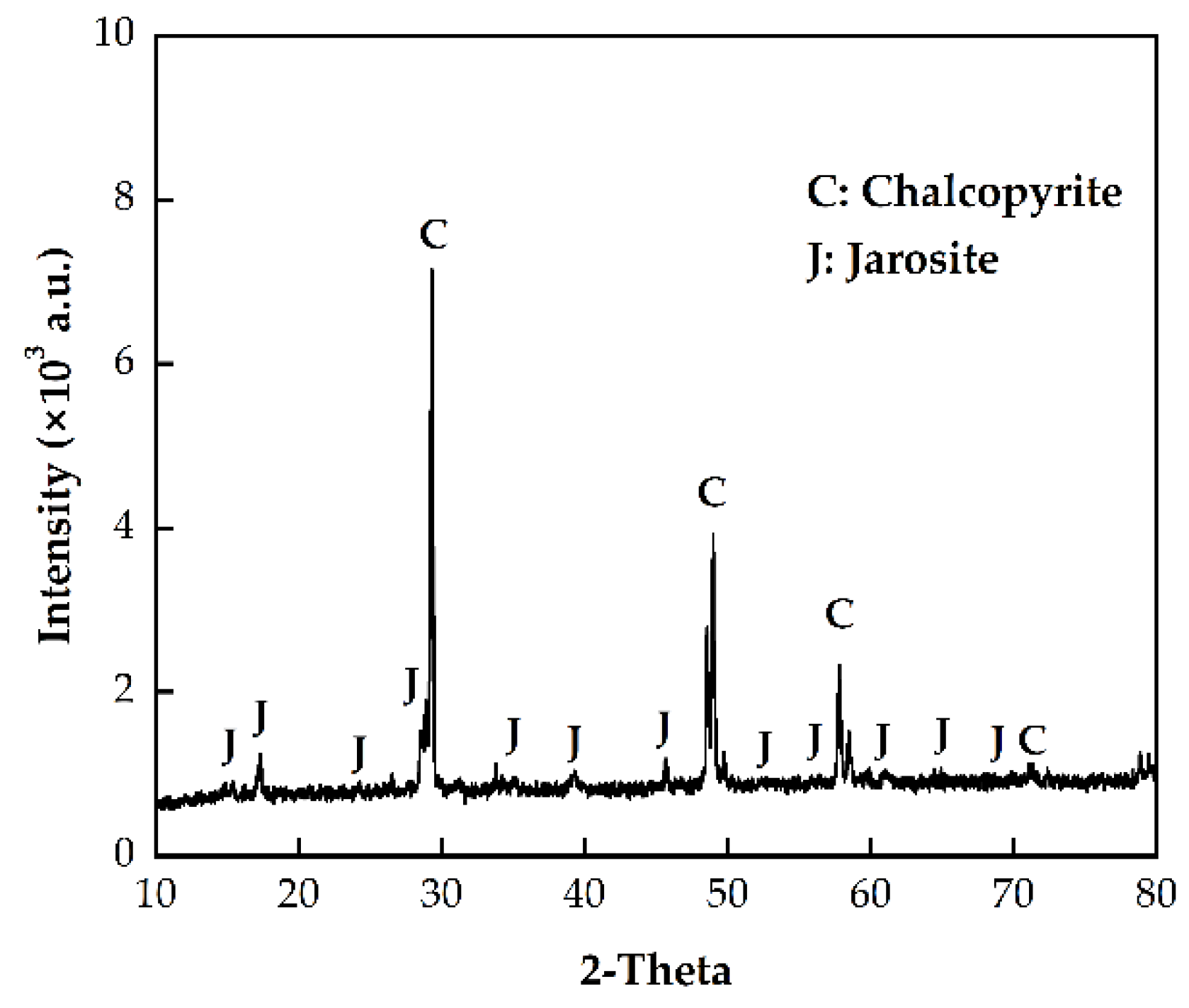
3.4. Changes in Chalcopyrite Surface Morphology and Intermediate with Immersion Pretreatment
4. Conclusions
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Z. Sun; Q. Wang. The asymmetric effect of natural resource abundance on economic growth and environmental pollution: Evidence from resource-rich economy. Resources Policy 2021, 72, 102085. [CrossRef]
- G. Jing; X. Meng; W. Sun; P.B. Kowalczuk; Z. Gao. Recent advances in the treatment and recycling of mineral processing wastewater. Environmental Science: Water Research & Technology 2023, 9(5), 1290-1304. [CrossRef]
- S. Lin; R. Liu; M. Wu; Y. Hu; W. Sun; Z. Shi; H. Han; W. Li. Minimizing beneficiation wastewater through internal reuse of process water in flotation circuit. Journal of Cleaner Production 2020, 245, 118898. [CrossRef]
- A. Azevedo; H.A. Oliveira; J. Rubio. Treatment and water reuse of lead-zinc sulphide ore mill wastewaters by high rate dissolved air flotation. Minerals Engineering 2018, 127, 114-121. [CrossRef]
- S. Meng; S. Wen; G. Han; X. Wang; Q. Feng. Wastewater Treatment in Mineral Processing of Non-Ferrous Metal Resources: A Review. Water 2022, 14(5), 726. [CrossRef]
- H. Wan; J. Qu; T. He; X. Bu; W. Yang; H. Li. A New Concept on High-Calcium Flotation Wastewater Reuse. Minerals 2018, 8(11), 496. [CrossRef]
- G. Jing; S. Ren; Y. Gao; W. Sun; Z. Gao. Electrocoagulation: A Promising Method to Treat and Reuse Mineral Processing Wastewater with High COD. Water 2020, 12(2), 595. [CrossRef]
- G. Jing; J. Wang; W. Sun; S. Pooley; D. Liao; Z. Shi; Q. Chen; Z. Gao. Reuse of mine and ore washing wastewater in scheelite flotation process to save freshwater: Lab to industrial scale. Journal of Water Process Engineering 2023, 53, 103674. [CrossRef]
- X. Yang. Beneficiation studies of tungsten ores – A review. Minerals Engineering 2018, 125, 111-119. [CrossRef]
- W. Jin; S. Yang; C. Tang; Y. Li; C. Chang; Y. Chen. Reaction Mechanism and Technical Application of Metallic Bismuth Extraction from Bismuthinite Concentrate by Low-Temperature Alkaline Smelting. ACS Sustainable Chemistry & Engineering 2023, 11(27), 9932-9946. [CrossRef]
- W.-C. Li; H.-J. Yu; X. Gao; X.-L. Liu; J.-H. Wang. Review of Mesozoic multiple magmatism and porphyry Cu–Mo (W) mineralization in the Yidun Arc, eastern Tibet Plateau. Ore Geology Reviews 2017, 90, 795-812. [CrossRef]
- B. Yang; M. Zeng; H. Zhu; P. Huang; Z. Li; S. Song. Selective depression of molybdenite using a novel eco-friendly depressant in Cu-Mo sulfides flotation system. Colloids and Surfaces A: Physicochemical and Engineering Aspects 2021, 622, 126683. [CrossRef]
- X. Wang; W.-q. Qin; F. Jiao; C.-r. Yang; W. Li; Z.-q. Zhang; J.-m. Zhou; J.-g. Guo; J. Zhang. Review on development of low-grade scheelite recovery from molybdenum tailings in Luanchuan, China: A case study of Luoyang Yulu Mining Company. Transactions of Nonferrous Metals Society of China 2022, 32(3), 980-998. [CrossRef]
- J. Kang; C. Chen; W. Sun; H. Tang; Z. Yin; R. Liu; Y. Hu; A.V. Nguyen. A significant improvement of scheelite recovery using recycled flotation wastewater treated by hydrometallurgical waste acid. Journal of Cleaner Production 2017, 151, 419-426. [CrossRef]
- H. Yan; B. Yang; M. Zeng; P. Huang; A. Teng. Selective flotation of Cu-Mo sulfides using xanthan gum as a novel depressant. Minerals Engineering 2020, 156, 106486. [CrossRef]
- G. Yi; E. Macha; J. Van Dyke; R. Ed Macha; T. McKay; M.L. Free. Recent progress on research of molybdenite flotation: A review. Advances in Colloid & Interface Science 2021, 295, 102466. [CrossRef]
- J. Hao; J. Liu; Y. Yu; H. Gao; X. Qin; X. Bai. Depressants for separation of chalcopyrite and molybdenite: Review and prospects. Minerals Engineering 2023, 201, 108209. [CrossRef]
- H. Peng; D. Wu; M. Abdalla; W. Luo; W. Jiao; X. Bie. Study of the Effect of Sodium Sulfide as a Selective Depressor in the Separation of Chalcopyrite and Molybdenite. Minerals 2017, 7(4), 51. [CrossRef]
- X. Tang; Y. Chen; K. Liu; G. Zeng; Q. Peng; Z. Li. Selective flotation separation of molybdenite and chalcopyrite by thermal pretreatment under air atmosphere. Colloids and Surfaces A: Physicochemical and Engineering Aspects 2019, 583, 123958. [CrossRef]
- T. Hirajima; M. Mori; O. Ichikawa; K. Sasaki; H. Miki; M. Farahat; M. Sawada. Selective flotation of chalcopyrite and molybdenite with plasma pre-treatment. Minerals Engineering 2014, 66-68, 102-111. [CrossRef]
- W. Peng; S. Liu; Y. Cao; W. Wang; S. Lv; Y. Huang. A novel approach for selective flotation separation of chalcopyrite and molybdenite – Electrocatalytic oxidation pretreatment and its mechanism. Applied Surface Science 2022, 597, 153753. [CrossRef]
- J. Kang; R. Fan; Y. Hu; W. Sun; R. Liu; Q. Zhang; H. Liu; X. Meng. Silicate removal from recycled wastewater for the improvement of scheelite flotation performance. Journal of Cleaner Production 2018, 195, 280-288. [CrossRef]
- F.T. Awadalla; A. Kumar. Opportunities for Membrane Technologies in the Treatment of Mining and Mineral Process Streams and Effluents∗. Separation Science & Technology 1994, 29(10), 1231-1249. [CrossRef]
- T. Hirajima; H. Miki; G.P.W. Suyantara; H. Matsuoka; A.M. Elmahdy; K. Sasaki; Y. Imaizumi; S. Kuroiwa. Selective flotation of chalcopyrite and molybdenite with H2O2 oxidation. Minerals Engineering 2017, 100, 83-92. [CrossRef]
- H. Miki; H. Matsuoka; T. Hirajima; G.P.W. Suyantara; K. Sasaki. Electrolysis Oxidation of Chalcopyrite and Molybdenite for Selective Flotation. MATERIALS TRANSACTIONS 2017, 58(5), 761-767. [CrossRef]
- Z. Yin; S. Chen; Z. Xu; C. Zhang; J. He; J. Zou; D. Chen; W. Sun. Flotation separation of molybdenite from chalcopyrite using an environmentally-efficient depressant L-cysteine and its adsoption mechanism. Minerals Engineering 2020, 156, 106438. [CrossRef]
- H. Li; W. Xiao; J. Jin; Y. Han. Influence Mechanism of Magnetized Modified Kerosene on Flotation Behavior of Molybdenite. Minerals 2022, 12(1), 2. [CrossRef]
- M. Irannajad; O. Salmani Nuri; A. Mehdilo. Surface dissolution-assisted mineral flotation: A review. Journal of Environmental Chemical Engineering 2019, 7(3), 103050. [CrossRef]
- Z. Wang; Y. Qian; L.-h. Xu; B. Dai; J.-h. Xiao; K. Fu. Selective chalcopyrite flotation from pyrite with glycerine-xanthate as depressant. Minerals Engineering 2015, 74, 86-90. [CrossRef]
- X. Wang; B. Zhao; J. Liu; Y. Zhu; Y. Han. Dithiouracil, a highly efficient depressant for the selective separation of molybdenite from chalcopyrite by flotation: Applications and mechanism. Minerals Engineering 2022, 175, 107287. [CrossRef]
- X. Zhang; L. Lu; Y. Cao; J. Yang; W. Che; J. Liu. The flotation separation of molybdenite from chalcopyrite using a polymer depressant and insights to its adsorption mechanism. Chemical Engineering Journal 2020, 395, 125137. [CrossRef]
- J. Yang; L. Chen; Z. Xue; K. Yang; Y. Shao; J. Zeng; Y. Gao. Performance evaluation of PHGMS technology for superfine chalcopyrite-molybdenite separation. Separation & Purification Technology 2024, 336, 126136. [CrossRef]
- I. Park; S. Hong; S. Jeon; M. Ito; N. Hiroyoshi. Flotation Separation of Chalcopyrite and Molybdenite Assisted by Microencapsulation Using Ferrous and Phosphate Ions: Part I. Selective Coating Formation. Metals 2020, 10(12), 1667. [CrossRef]
- M. Abdollahi; A. Bahrami; M.S. Mirmohammadi; F. Kazemi; A. Danesh; Y. Ghorbani. A process mineralogy approach to optimize molybdenite flotation in copper – molybdenum processing plants. Minerals Engineering 2020, 157, 106557. [CrossRef]
- H. Zhao; Y. Zhang; X. Zhang; L. Qian; M. Sun; Y. Yang; Y. Zhang; J. Wang; H. Kim; G. Qiu. The dissolution and passivation mechanism of chalcopyrite in bioleaching: An overview. Minerals Engineering 2019, 136, 140-154. [CrossRef]
- A. Ghahremaninezhad; E. Asselin; D.G. Dixon. Electrochemical evaluation of the surface of chalcopyrite during dissolution in sulfuric acid solution. Electrochimica Acta 2010, 55(18), 5041-5056. [CrossRef]
- Y. Zhang; H. Zhao; L. Qian; M. Sun; X. Lv; L. Zhang; J. Petersen; G. Qiu. A brief overview on the dissolution mechanisms of sulfide minerals in acidic sulfate environments at low temperatures: Emphasis on electrochemical cyclic voltammetry analysis. Minerals Engineering 2020, 158, 106586. [CrossRef]
- G. Gu; K. Hu; X. Zhang; X. Xiong; H. Yang. The stepwise dissolution of chalcopyrite bioleached by Leptospirillum ferriphilum. Electrochimica Acta 2013, 103, 50-57. [CrossRef]
- E.M. Córdoba; J.A. Muñoz; M.L. Blázquez; F. González; A. Ballester. Leaching of chalcopyrite with ferric ion. Part I: General aspects. Hydrometallurgy 2008, 93(3), 81-87. [CrossRef]
- X. Lv; J. Wang; X. Zeng; Z. Liang; D. He; Y. Zhang; H. Yuan; H. Zhao; Q. Meng. Cooperative extraction of metals from chalcopyrite by bio-oxidation and chemical oxidation. Geochemistry 2021, 81(4), 125772. [CrossRef]
- A. Ahmadi; M. Schaffie; Z. Manafi; M. Ranjbar. Electrochemical bioleaching of high grade chalcopyrite flotation concentrates in a stirred bioreactor. Hydrometallurgy 2010, 104(1), 99-105. [CrossRef]
- E.M. Córdoba; J.A. Muñoz; M.L. Blázquez; F. González; A. Ballester. Leaching of chalcopyrite with ferric ion. Part II: Effect of redox potential. Hydrometallurgy 2008, 93(3), 88-96. [CrossRef]
- L. Gunneriusson; Å. Sandström; A. Holmgren; E. Kuzmann; K. Kovacs; A. Vértes. Jarosite inclusion of fluoride and its potential significance to bioleaching of sulphide minerals. Hydrometallurgy 2009, 96(1), 108-116. [CrossRef]
- B. Yan; T. Liang; X. Yang; A.J. Gadgil. Superior removal of As(III) and As(V) from water with Mn-doped β-FeOOH nanospindles on carbon foam. Journal of Hazardous Materials 2021, 418, 126347. [CrossRef]
- Q. Chen; Y. Yao; Z. Zhao; J. Zhou; Z. Chen. Long term catalytic activity of pyrite in Heterogeneous Fenton-like oxidation for the tertiary treatment of dyeing wastewater. Journal of Environmental Chemical Engineering 2021, 9(4), 105730. [CrossRef]
- S. Karthe; R. Szargan; E. Suoninen. Oxidation of pyrite surfaces: a photoelectron spectroscopic study. Applied Surface Science 1993, 72(2), 157-170. [CrossRef]


| Items | Cu | Pb | Zn | S | Al | Fe | K | Na | Mg | Si | COD | pH |
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Data | 0.15 | 0.23 | 0.16 | 40.71 | 592 | 10.32 | 210 | 1028 | 69.12 | 226 | 85 | 4.00 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




