Submitted:
24 March 2024
Posted:
26 March 2024
Read the latest preprint version here
Abstract
Keywords:
1. Introduction
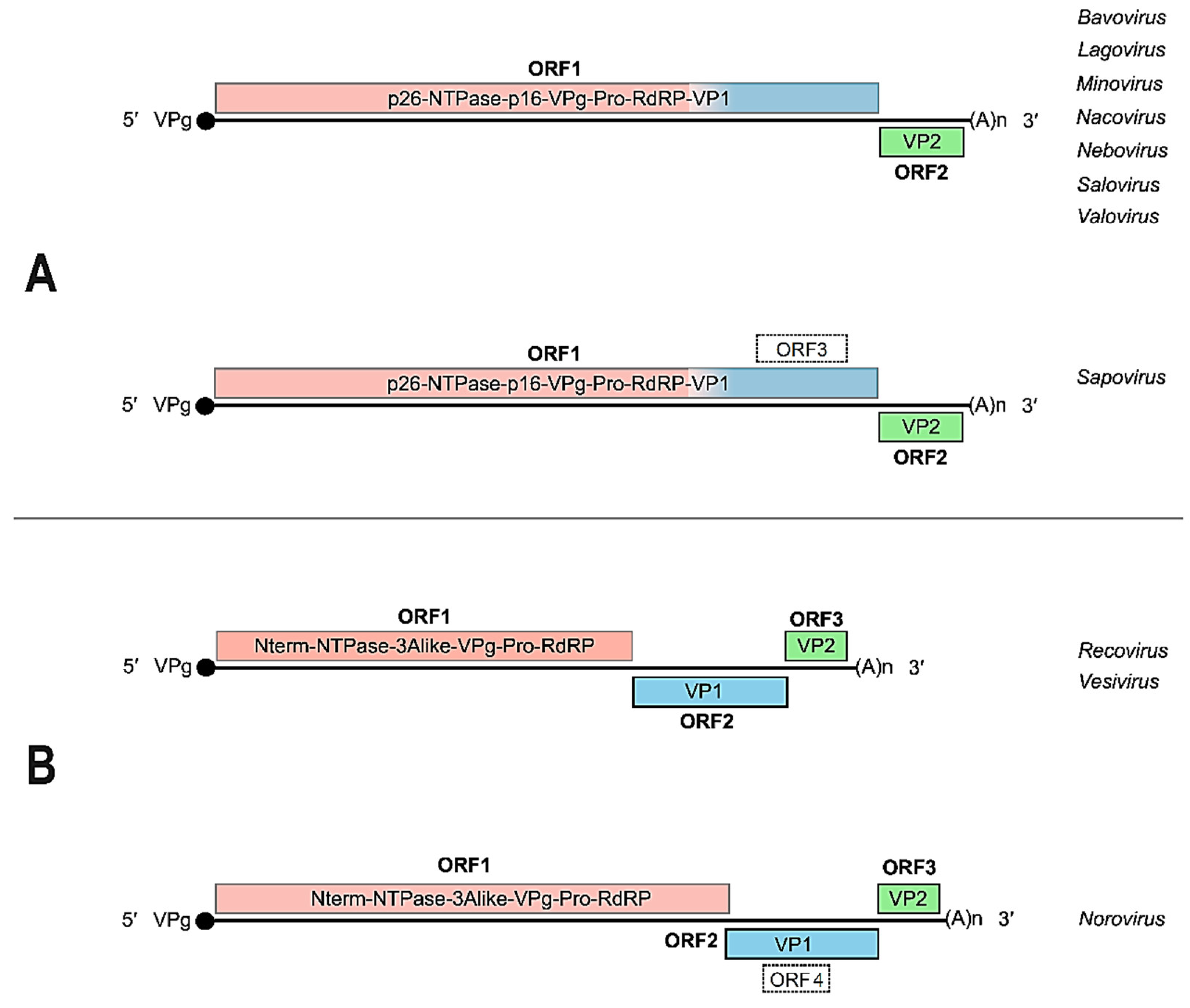
2. The Caliciviridae: Genome Organization, Gene Expression and Replication Strategies
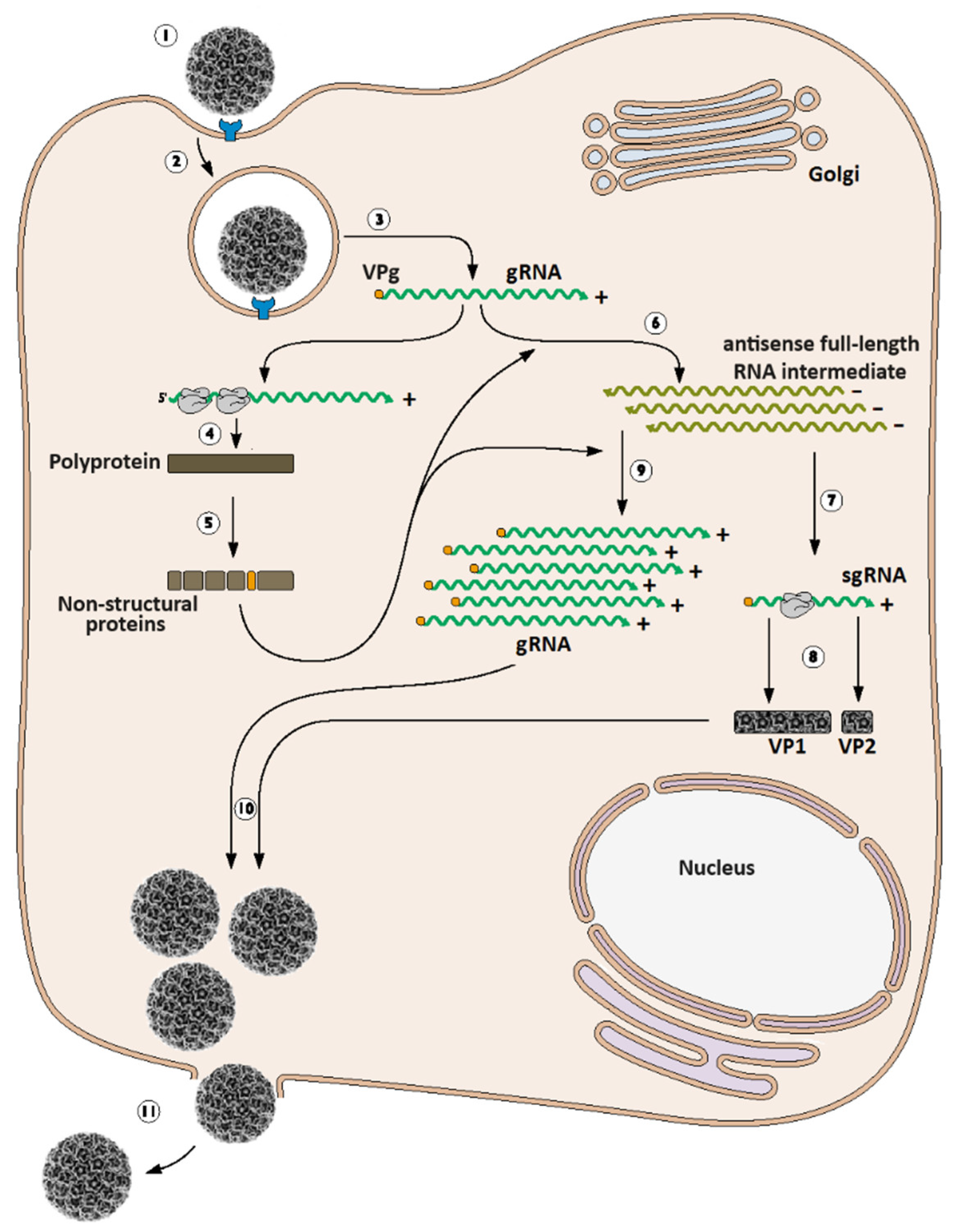
3. Calicivirus Replication Cycle
4. In Vitro Study of Caliciviruses
4.1. The challenges of Reverse Genetics
4.2. The Hallmarks of a Promising Virus-Expressing cDNA
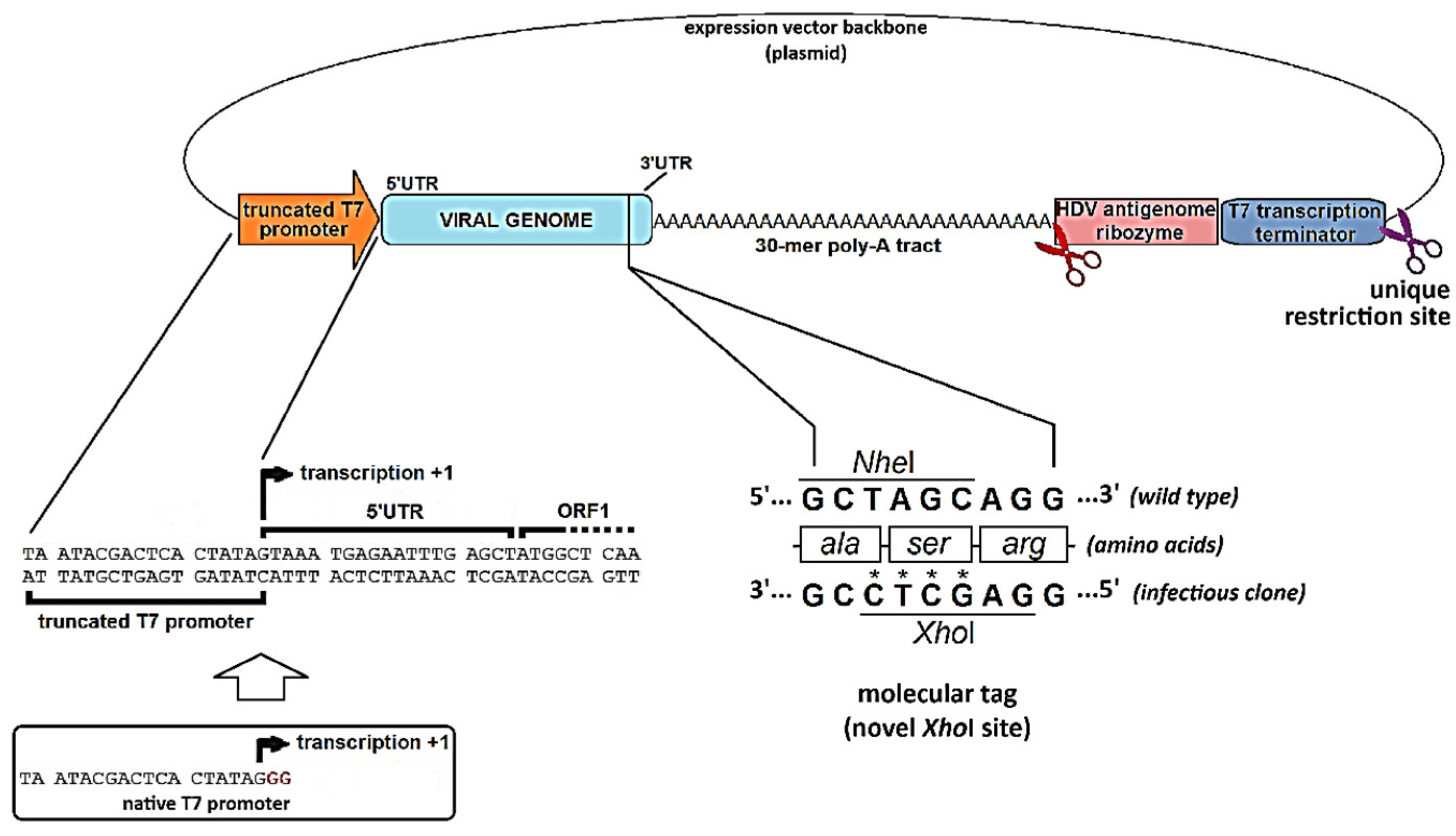
4.3. When Things Go Wrong: Interrogating the Viral Genome for the Occurrence of Replication-Critical Events
5. Chronology of the Calicivirus Reverse Genetics
5.1. The Rabbit Vesivirus Reverse Genetics Journey
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- J.C. Boyer, and A.L. Haenni, Infectious transcripts and cDNA clones of RNA viruses. Virology 198 (1994) 415-426. [CrossRef]
- S. Sosnovtsev, and K.Y. Green, RNA transcripts derived from a cloned full-length copy of the feline calicivirus genome do not require VpG for infectivity. Virology 210 (1995) 383-390. [CrossRef]
- Goodfellow, and S. Taube, Chapter 3.2 - Calicivirus Replication and Reverse Genetics. in: L. Svensson, U. Desselberger, H.B. Greenberg, and M.K. Estes, (Eds.), Viral Gastroenteritis, Academic Press, Boston, 2016, pp. 355-378.
- U. Desselberger, Caliciviridae Other Than Noroviruses. Viruses 11 (2019).
- H. Ebihara, A. Groseth, G. Neumann, Y. Kawaoka, and H. Feldmann, The role of reverse genetics systems in studying viral hemorrhagic fevers. Thromb Haemost 94 (2005) 240-53. [CrossRef]
- H.L. Cai, and Y.W. Huang, Reverse genetics systems for SARS-CoV-2: Development and applications. Virol Sin 38 (2023) 837-850.
- R. Andino, D. Silvera, S.D. Suggett, P.L. Achacoso, C.J. Miller, D. Baltimore, and M.B. Feinberg, Engineering poliovirus as a vaccine vector for the expression of diverse antigens. Science 265 (1994) 1448-51. [CrossRef]
- A.A. Khromykh, and E.G. Westaway, Subgenomic replicons of the flavivirus Kunjin: construction and applications. Journal of virology 71 (1997) 1497-1505. [CrossRef]
- P. Liljeström, Alphavirus expression systems. Current Opinion in Biotechnology 5 (1994) 495-500.
- L. Enjuanes, I. Sola, F. Almazan, A. Izeta, J.M. Gonzalez, and S. Alonso, Coronavirus Derived Expression Systems. in: E. Lavi, S.R. Weiss, and S.T. Hingley, (Eds.), The Nidoviruses: Coronaviruses and Arteriviruses, Springer US, Boston, MA, 2001, pp. 309-321.
- A.A. Amarilla, J.D.J. Sng, R. Parry, J.M. Deerain, J.R. Potter, Y.X. Setoh, D.J. Rawle, T.T. Le, N. Modhiran, X. Wang, N.Y.G. Peng, F.J. Torres, A. Pyke, J.J. Harrison, M.E. Freney, B. Liang, C.L.D. McMillan, S.T.M. Cheung, D. Guevara, J.M. Hardy, M. Bettington, D.A. Muller, F. Coulibaly, F. Moore, R.A. Hall, P.R. Young, J.M. Mackenzie, J. Hobson-Peters, A. Suhrbier, D. Watterson, and A.A. Khromykh, A versatile reverse genetics platform for SARS-CoV-2 and other positive-strand RNA viruses. Nat Commun 12 (2021) 3431.
- K.Y. Green, Chapter 20. Caliciviridae: The Noroviruses. in: D.M. Knipe, Howley, P.M., (Ed.), Fields Virology, Philadelphia, PA, 2013, pp. 582-608.
- J. Vinjé, M.K. Estes, P. Esteves, K.Y. Green, K. Katayama, N.J. Knowles, Y. L’Homme, V. Martella, H. Vennema, P.A. White, and I.R. Consortium, ICTV Virus Taxonomy Profile: Caliciviridae. Journal of General Virology 100 (2019) 1469-1470.
- I.N. Clarke, and P.R. Lambden, Organization and expression of calicivirus genes. J.Infect.Dis. 181 (Suppl 2) (2000) S309-S316. [CrossRef]
- B. Alhatlani, S. Vashist, and I. Goodfellow, Functions of the 5’ and 3’ ends of calicivirus genomes. Virus Res. (2015).
- T.P. Herbert, I. Brierley, and T.D.K. Brown, Identification of a protein linked to the genomic and subgenomic mRNAs of feline calicivirus and its role in translation. Journal of General Virology 78 (1997) 1033-1040. [CrossRef]
- G. Meyers, C. Wirblich, and H.J. Thiel, Genomic and subgenomic RNAs of rabbit hemorrhagic disease virus are both protein-linked and packaged into particles. Virology 184 (1991) 677-686. [CrossRef]
- S.V. Sosnovtsev, S.A. Sosnovtseva, and K.Y. Green, Cleavage of the feline calicivirus capsid precursor is mediated by a virus-encoded proteinase. J.Virol. 72 (1998) 3051-3059. [CrossRef]
- M.A. Yunus, L.M. Chung, Y. Chaudhry, D. Bailey, and I. Goodfellow, Development of an optimized RNA-based murine norovirus reverse genetics system. J.Virol.Methods 169 (2010) 112-118. [CrossRef]
- B. Boniotti, C. Wirblich, M. Sibilia, G. Meyers, H.J. Thiel, and C. Rossi, Identification and characterization of a 3C-like protease from rabbit hemorrhagic disease virus, a calicivirus. Journal of Virology 68 (1994) 6487-6495. [CrossRef]
- F. Parra, J.A. Boga, M.S. Marin, and R. Casais, The amino terminal sequence of VP60 from rabbit hemorrhagic disease virus supports its putative subgenomic origin. Virus Research 27 (1993) 219-228. [CrossRef]
- G. Meyers, Translation of the minor capsid protein of a calicivirus is initiated by a novel termination-dependent reinitiation mechanism. J.Biol.Chem. 278 (2003) 34051-34060. [CrossRef]
- T.A. Poyry, A. Kaminski, E.J. Connell, C.S. Fraser, and R.J. Jackson, The mechanism of an exceptional case of reinitiation after translation of a long ORF reveals why such events do not generally occur in mammalian mRNA translation. Genes Dev. 21 (2007) 3149-3162. [CrossRef]
- I.N. Clarke, M.K. Estes, K.Y. Green, G.S. Hansman, N.J. Knowles, M.K. Koopmans, D.O. Matson, G. Meyers, J.D. Neill, A. Radford, A.W. Smith, M.J. Studdert, H.J. Thiel, and J. Vinje, Family Caliciviridae. in: A.M.Q. King, M.J. Adams, E.B. Carstens, and E.J. Lefkowitz, (Eds.), Virus Taxonomy. Classification and Nomenclature of Viruses: Ninth Report of the International Committee on Taxonomy of Viruses, Elsevier, San Diego, 2011, pp. 977-986.
- Machin, J.M. Martin Alonso, and F. Parra, Identification of the amino acid residue involved in rabbit hemorrhagic disease virus VPg uridylylation. J.Biol.Chem. 276 (2001) 27787-27792. [CrossRef]
- Goodfellow, Y. Chaudhry, I. Gioldasi, A. Gerondopoulos, A. Natoni, L. Labrie, J.F. Laliberte, and L. Roberts, Calicivirus translation initiation requires an interaction between VPg and eIF 4 E. EMBO Rep. 6 (2005) 968-972. [CrossRef]
- Y. Chaudhry, A. Nayak, M.E. Bordeleau, J. Tanaka, J. Pelletier, G.J. Belsham, L.O. Roberts, and I.G. Goodfellow, Caliciviruses differ in their functional requirements for eIF4F components. J.Biol.Chem. 281 (2006) 25315-25325. [CrossRef]
- Goodfellow, The genome-linked protein VPg of vertebrate viruses - a multifaceted protein. Curr Opin Virol 1 (2011) 355-62. [CrossRef]
- K.F. Daughenbaugh, C.E. Wobus, and M.E. Hardy, VPg of murine norovirus binds translation initiation factors in infected cells. Virol.J. 3 (2006) 33. [CrossRef]
- Á.L. Álvarez, A. García-Manso, K.P. Dalton, J.M. Martín-Alonso, I. Nicieza, A. Podadera, M. Acosta-Zaldívar, D. De Llano, and F. Parra, Reverse Genetics System for Rabbit vesivirus. Frontiers in Microbiology 11 (2020). [CrossRef]
- R. Casais, L.G. Molleda, A. Machin, B.G. del, A.G. Manso, K.P. Dalton, A. Coto, J.M. Alonso, M. Prieto, and F. Parra, Structural and functional analysis of virus factories purified from Rabbit vesivirus-infected Vero cells. Virus Res. 137 (2008) 112-121. [CrossRef]
- E. Smertina, R.N. Hall, N. Urakova, T. Strive, and M. Frese, Calicivirus Non-structural Proteins: Potential Functions in Replication and Host Cell Manipulation. Front Microbiol 12 (2021) 712710. [CrossRef]
- Y. Matsuura, Y. Tohya, M. Onuma, F. Roerink, M. Mochizuki, and T. Sugimura, Expression and processing of the canine calicivirus capsid precursor. Microbiology 81 (2000) 195-199. [CrossRef]
- K.O. Chang, D.W. George, J.B. Patton, K.Y. Green, and S.V. Sosnovtsev, Leader of the capsid protein in feline calicivirus promotes replication of Norwalk virus in cell culture. J.Virol. 82 (2008) 9306-9317. [CrossRef]
- E.J. Abente, S.V. Sosnovtsev, C. Sandoval-Jaime, G.I. Parra, K. Bok, and K.Y. Green, The feline calicivirus leader of the capsid protein is associated with cytopathic effect. J.Virol. 87 (2013) 3003-3017. [CrossRef]
- G.S. Hansman, T. Oka, K. Katayama, and N. Takeda, Human sapoviruses: genetic diversity, recombination, and classification. Rev.Med Virol. 17 (2007) 133-141. [CrossRef]
- N. McFadden, D. Bailey, G. Carrara, A. Benson, Y. Chaudhry, A. Shortland, J. Heeney, F. Yarovinsky, P. Simmonds, A. Macdonald, and I. Goodfellow, Norovirus Regulation of the Innate Immune Response and Apoptosis Occurs via the Product of the Alternative Open Reading Frame 4. PLoS Pathogens 7 (2011) e1002413. [CrossRef]
- C.V. Subba-Reddy, M.A. Yunus, I.G. Goodfellow, and C.C. Kao, Norovirus RNA synthesis is modulated by an interaction between the viral RNA-dependent RNA polymerase and the major capsid protein, VP1. Journal of Virology 86 (2012) 10138-10149. [CrossRef]
- C.J. McCormick, O. Salim, P.R. Lambden, and I.N. Clarke, Translation termination reinitiation between open reading frame 1 (ORF1) and ORF2 enables capsid expression in a bovine norovirus without the need for production of viral subgenomic RNA. J.Virol. 82 (2008) 8917-8921. [CrossRef]
- R. Wennesz, C. Luttermann, F. Kreher, and G. Meyers, Structure–function relationship in the ‘termination upstream ribosomal binding site’ of the calicivirus rabbit hemorrhagic disease virus. Nucleic Acids Research 47 (2019) 1920-1934.
- K. Geissler, K. Schneider, A. Fleuchaus, C.R. Parrish, G. Sutter, and U. Truyen, Feline calicivirus capsid protein expression and capsid assembly in cultured feline cells. J.Virol. 73 (1999) 834-838. [CrossRef]
- B. Di Martino, and F. Marsilio, Feline calicivirus VP2 is involved in the self-assembly of the capsid protein into virus-like particles. Res.Vet.Sci. 89 (2010) 279-281. [CrossRef]
- L.G. Thorne, and I.G. Goodfellow, Norovirus gene expression and replication. The Journal of general virology 95 Pt 2 (2014) 278-91.
- S.J. Flint, L.W. Enquist, R.M. Krug, V.R. Racaniello, and A.M. Skalka, Picornaviruses, Principles of Virology. Molecular Biology, Pathogenesis and Control, ASM, Washington, 2000, pp. 752.
- C.E. Wobus, S.M. Karst, L.B. Thackray, K.-O. Chang, S.V. Sosnovtsev, G. Belliot, A. Krug, J.M. Mackenzie, K.Y. Green, and H.W.I.V. Virgin, Replication of Norovirus in Cell Culture Reveals a Tropism for Dendritic Cells and Macrophages. PLOS Biology 2 (2004) e432. [CrossRef]
- C. Cox, S. Cao, and Y. Lu, Enhanced detection and study of murine norovirus-1 using a more efficient microglial cell line. Virol J 6 (2009) 196. [CrossRef]
- L.C. Kreutz, B.S. Seal, and W.L. Mengeling, Early interaction of feline calicivirus with cells in culture. Arch Virol 136 (1994) 19-34. [CrossRef]
- T. Farkas, K. Sestak, C. Wei, and X. Jiang, Characterization of a rhesus monkey calicivirus representing a new genus of Caliciviridae. J.Virol. 82 (2008) 5408-5416. [CrossRef]
- M.K. Jones, K.R. Grau, V. Costantini, A.O. Kolawole, M. de Graaf, P. Freiden, C.L. Graves, M. Koopmans, S.M. Wallet, S.A. Tibbetts, S. Schultz-Cherry, C.E. Wobus, J. Vinjé, and S.M. Karst, Human norovirus culture in B cells. Nat Protoc 10 (2015) 1939-47. [CrossRef]
- L.C. van Dinten, J.A. den Boon, A.L. Wassenaar, W.J. Spaan, and E.J. Snijder, An infectious arterivirus cDNA clone: identification of a replicase point mutation that abolishes discontinuous mRNA transcription. Proc Natl Acad Sci U S A 94 (1997) 991-6. [CrossRef]
- G. van Marle, L.C. van Dinten, W.J. Spaan, W. Luytjes, and E.J. Snijder, Characterization of an equine arteritis virus replicase mutant defective in subgenomic mRNA synthesis. J Virol 73 (1999) 5274-81. [CrossRef]
- F. Almazan, I. Sola, S. Zuniga, S. Marquez-Jurado, L. Morales, M. Becares, and L. Enjuanes, Coronavirus reverse genetic systems: infectious clones and replicons. Virus Res 189 (2014) 262-70. [CrossRef]
- R. Casais, V. Thiel, S.G. Siddell, D. Cavanagh, and P. Britton, Reverse genetics system for the avian coronavirus infectious bronchitis virus. J Virol 75 (2001) 12359-69. [CrossRef]
- C.J. Lai, B.T. Zhao, H. Hori, and M. Bray, Infectious RNA transcribed from stably cloned full-length cDNA of dengue type 4 virus. Proc.Natl.Acad.Sci.U.S.A 88 (1991) 5139-5143. [CrossRef]
- N. Ruggli, J.D. Tratschin, C. Mittelholzer, and M.A. Hofmann, Nucleotide sequence of classical swine fever virus strain Alfort/187 and transcription of infectious RNA from stably cloned full-length cDNA. J.Virol. 70 (1996) 3478-3487. [CrossRef]
- M. Yanagi, R.H. Purcell, S.U. Emerson, and J. Bukh, Transcripts from a single full-length cDNA clone of hepatitis C virus are infectious when directly transfected into the liver of a chimpanzee. Proc.Natl.Acad.Sci.U.S.A 94 (1997) 8738-8743. [CrossRef]
- D.G. Gibson, L. Young, R.-Y. Chuang, J.C. Venter, C.A. Hutchison, and H.O. Smith, Enzymatic assembly of DNA molecules up to several hundred kilobases. Nature Methods 6 (2009) 343-345.
- Y. Chaudhry, M.A. Skinner, and I.G. Goodfellow, Recovery of genetically defined murine norovirus in tissue culture by using a fowlpox virus expressing T7 RNA polymerase. J.Gen.Virol. 88 (2007) 2091-2100. [CrossRef]
- C. Sandoval-Jaime, K.Y. Green, and S.V. Sosnovtsev, Recovery of murine norovirus and feline calicivirus from plasmids encoding EMCV IRES in stable cell lines expressing T7 polymerase. J.Virol.Methods 217 (2015) 1-7. [CrossRef]
- Bridgen, and R.M. Elliot, Chapter 9. Reverse genetics of RNA viruses. in: A.J. Cann, (Ed.), RNA viruses: a practical approach, Oxford University Press, New York, 2000, pp. 201-227.
- K.O. Chang, S.V. Sosnovtsev, G. Belliot, Q. Wang, L.J. Saif, and K.Y. Green, Reverse genetics system for porcine enteric calicivirus, a prototype sapovirus in the Caliciviridae. J.Virol. 79 (2005) 1409-1416. [CrossRef]
- K. Katayama, G.S. Hansman, T. Oka, S. Ogawa, and N. Takeda, Investigation of norovirus replication in a human cell line. Arch.Virol. 151 (2006) 1291-1308. [CrossRef]
- S.V. Sosnovtsev, S.A. Sosnovtseva, and K.Y. Green, Recovery of feline calicivirus from plasmid DNA containing a full-length copy of the genome. in: D. Chasey, R.M. Gaskell, and I.N. Clarke, (Eds.), The first international symposium on calicivirus, European Society for Veterinary Virology and Central Veterinary Laboratory, Reading, United Kingdom, 1996, pp. 125-130.
- T. Mitra, S.V. Sosnovtsev, and K.Y. Green, Mutagenesis of tyrosine 24 in the VPg protein is lethal for feline calicivirus. J.Virol. 78 (2004) 4931-4935. [CrossRef]
- C. Morgan, S.A. Ellison, H.M. Rose, and D.H. Moore, Structure and development of viruses observed in the electron microscope: II. vaccinia and fowl pox viruses. The Journal of Experimental Medicine 100 (1954) 301-310.
- P. Somogyi, J. Frazier, and M.A. Skinner, Fowlpox virus host range restriction: gene expression, DNA replication, and morphogenesis in nonpermissive mammalian cells. Virology 197 (1993) 439-444. [CrossRef]
- E. Scotto-Lavino, G. Du, and M.A. Frohman, 5’ end cDNA amplification using classic RACE. Nat Protoc 1 (2006) 2555-62.
- C.T. Ranjith-Kumar, Y. Wen, N. Baxter, K. Bhardwaj, and C.C. Kao, A cell-based assay for RNA synthesis by the HCV polymerase reveals new insights on mechanism of polymerase inhibitors and modulation by NS5A. PLoS.One. 6 (2011) e22575. [CrossRef]
- C.V. Subba-Reddy, I. Goodfellow, and C.C. Kao, VPg-primed RNA synthesis of norovirus RNA-dependent RNA polymerases by using a novel cell-based assay. J.Virol. 85 (2011) 13027-13037. [CrossRef]
- R.B. Seth, L. Sun, and Z.J. Chen, Antiviral innate immunity pathways. Cell Res 16 (2006) 141-147. [CrossRef]
- S.V. Sosnovtsev, M. Garfield, and K.Y. Green, Processing map and essential cleavage sites of the nonstructural polyprotein encoded by ORF1 of the feline calicivirus genome. J.Virol. 76 (2002) 7060-7072. [CrossRef]
- J.D. Neill, S.V. Sosnovtsev, and K.Y. Green, Recovery and altered neutralization specificities of chimeric viruses containing capsid protein domain exchanges from antigenically distinct strains of feline calicivirus. J.Virol. 74 (2000) 1079-1084. [CrossRef]
- S.V. Sosnovtsev, G. Belliot, K.O. Chang, O. Onwudiwe, and K.Y. Green, Feline calicivirus VP2 is essential for the production of infectious virions. J.Virol. 79 (2005) 4012-4024. [CrossRef]
- E.J. Abente, S.V. Sosnovtsev, K. Bok, and K.Y. Green, Visualization of feline calicivirus replication in real-time with recombinant viruses engineered to express fluorescent reporter proteins. Virology 400 (2010) 18-31. [CrossRef]
- Karakasiliotis, S. Vashist, D. Bailey, E.J. Abente, K.Y. Green, L.O. Roberts, S.V. Sosnovtsev, and I.G. Goodfellow, Polypyrimidine tract binding protein functions as a negative regulator of feline calicivirus translation. PLoS.One. 5 (2010) e9562. [CrossRef]
- J.R.O. Thumfart, and G. Meyers, Feline Calicivirus: Recovery of Wild-Type and Recombinant Viruses after Transfection of cRNA or cDNA Constructs. Journal of Virology 76 (2002) 6398-6407. [CrossRef]
- C. Wei, T. Farkas, K. Sestak, and X. Jiang, Recovery of infectious virus by transfection of in vitro-generated RNA from tulane calicivirus cDNA. J.Virol. 82 (2008) 11429-11436. [CrossRef]
- M. Asanaka, R.L. Atmar, V. Ruvolo, S.E. Crawford, F.H. Neill, and M.K. Estes, Replication and packaging of Norwalk virus RNA in cultured mammalian cells. Proc.Natl.Acad.Sci.U.S.A 102 (2005) 10327-10332. [CrossRef]
- S. Guix, M. Asanaka, K. Katayama, S.E. Crawford, F.H. Neill, R.L. Atmar, and M.K. Estes, Norwalk Virus RNA Is Infectious in Mammalian Cells. Journal of Virology 81 (2007) 12238-12248. [CrossRef]
- V.K. Ward, C.J. McCormick, I.N. Clarke, O. Salim, C.E. Wobus, L.B. Thackray, H.W. Virgin, and P.R. Lambden, Recovery of infectious murine norovirus using pol II-driven expression of full-length cDNA. Proc.Natl.Acad.Sci.U.S.A 104 (2007) 11050-11055. [CrossRef]
- T. Oka, H. Takagi, and Y. Tohya, Development of a novel single step reverse genetics system for feline calicivirus. J.Virol.Methods 207 (2014) 178-181. [CrossRef]
- D. Jackson, A. Cadman, T. Zurcher, and W.S. Barclay, A reverse genetics approach for recovery of recombinant influenza B viruses entirely from cDNA. J.Virol. 76 (2002) 11744-11747. [CrossRef]
- S. Komoto, J. Sasaki, and K. Taniguchi, Reverse genetics system for introduction of site-specific mutations into the double-stranded RNA genome of infectious rotavirus. Proc.Natl.Acad.Sci.U.S.A 103 (2006) 4646-4651. [CrossRef]
- P. Simmonds, I. Karakasiliotis, D. Bailey, Y. Chaudhry, D.J. Evans, and I.G. Goodfellow, Bioinformatic and functional analysis of RNA secondary structure elements among different genera of human and animal caliciviruses. Nucleic Acids Res. 36 (2008) 2530-2546. [CrossRef]
- D. Bailey, I. Karakasiliotis, S. Vashist, L.M. Chung, J. Rees, N. McFadden, A. Benson, F. Yarovinsky, P. Simmonds, and I. Goodfellow, Functional analysis of RNA structures present at the 3’ extremity of the murine norovirus genome: the variable polypyrimidine tract plays a role in viral virulence. J.Virol. 84 (2010) 2859-2870. [CrossRef]
- D. Bailey, L.B. Thackray, and I.G. Goodfellow, A single amino acid substitution in the murine norovirus capsid protein is sufficient for attenuation in vivo. J.Virol. 82 (2008) 7725-7728. [CrossRef]
- N. McFadden, A. Arias, I. Dry, D. Bailey, J. Witteveldt, D.J. Evans, I. Goodfellow, and P. Simmonds, Influence of genome-scale RNA structure disruption on the replication of murine norovirus—similar replication kinetics in cell culture but attenuation of viral fitness in vivo. Nucleic Acids Research 41 (2013) 6316-6331. [CrossRef]
- L. Thorne, D. Bailey, and I. Goodfellow, High-resolution functional profiling of the norovirus genome. J.Virol. 86 (2012) 11441-11456. [CrossRef]
- E. Lopez-Manriquez, S. Vashist, L. Urena, I. Goodfellow, P. Chavez, J.E. Mora-Heredia, C. Cancio-Lonches, E. Garrido, and A.L. Gutierrez-Escolano, Norovirus Genome Circularization and Efficient Replication Are Facilitated by Binding of PCBP2 and hnRNP A1. Journal of Virology 87 (2013) 11371-11387. [CrossRef]
- J. Cheng, A. Tang, J. Chen, D. Zhang, C. Meng, C. Li, H. Wei, and G. Liu, A cDNA-based reverse genetics system for feline calicivirus identifies the 3’ untranslated region as an essential element for viral replication. Arch Virol 168 (2023) 33.
- K. Katayama, K. Murakami, T.M. Sharp, S. Guix, T. Oka, R. Takai-Todaka, A. Nakanishi, S.E. Crawford, R.L. Atmar, and M.K. Estes, Plasmid-based human norovirus reverse genetics system produces reporter-tagged progeny virus containing infectious genomic RNA. Proc.Natl.Acad.Sci.U.S.A 111 (2014) E4043-E4052. [CrossRef]
- K.O. Chang, S.V. Sosnovtsev, G. Belliot, A.D. King, and K.Y. Green, Stable expression of a Norwalk virus RNA replicon in a human hepatoma cell line. Virology 353 (2006) 463-73. [CrossRef]
- K.-O. Chang, and D.W. George, Interferons and Ribavirin Effectively Inhibit Norwalk Virus Replication in Replicon-Bearing Cells. Journal of Virology 81 (2007) 12111-12118. [CrossRef]
- K.-O. Chang, Role of Cholesterol Pathways in Norovirus Replication. Journal of Virology 83 (2009) 8587-8595. [CrossRef]
- K. Bok, V.G. Prikhodko, K.Y. Green, and S.V. Sosnovtsev, Apoptosis in murine norovirus-infected RAW264.7 cells is associated with downregulation of survivin. J.Virol. 83 (2009) 3647-3656. [CrossRef]
- K.C. Tiew, G. He, S. Aravapalli, S.R. Mandadapu, M.R. Gunnam, K.R. Alliston, G.H. Lushington, Y. Kim, K.O. Chang, and W.C. Groutas, Design, synthesis, and evaluation of inhibitors of Norwalk virus 3C protease. Bioorg Med Chem Lett 21 (2011) 5315-9. [CrossRef]
- J.M. Martín-Alonso, D.E. Skilling, L. González-Molleda, G. del Barrio, A. Machín, N.K. Keefer, D.O. Matson, P.L. Iversen, A.W. Smith, and F. Parra, Isolation and characterization of a new Vesivirus from rabbits. Virology 337 (2005) 373-383. [CrossRef]
- Arias, L. Urena, L. Thorne, M.A. Yunus, and I. Goodfellow, Reverse genetics mediated recovery of infectious murine norovirus. J.Vis.Exp. (2012).

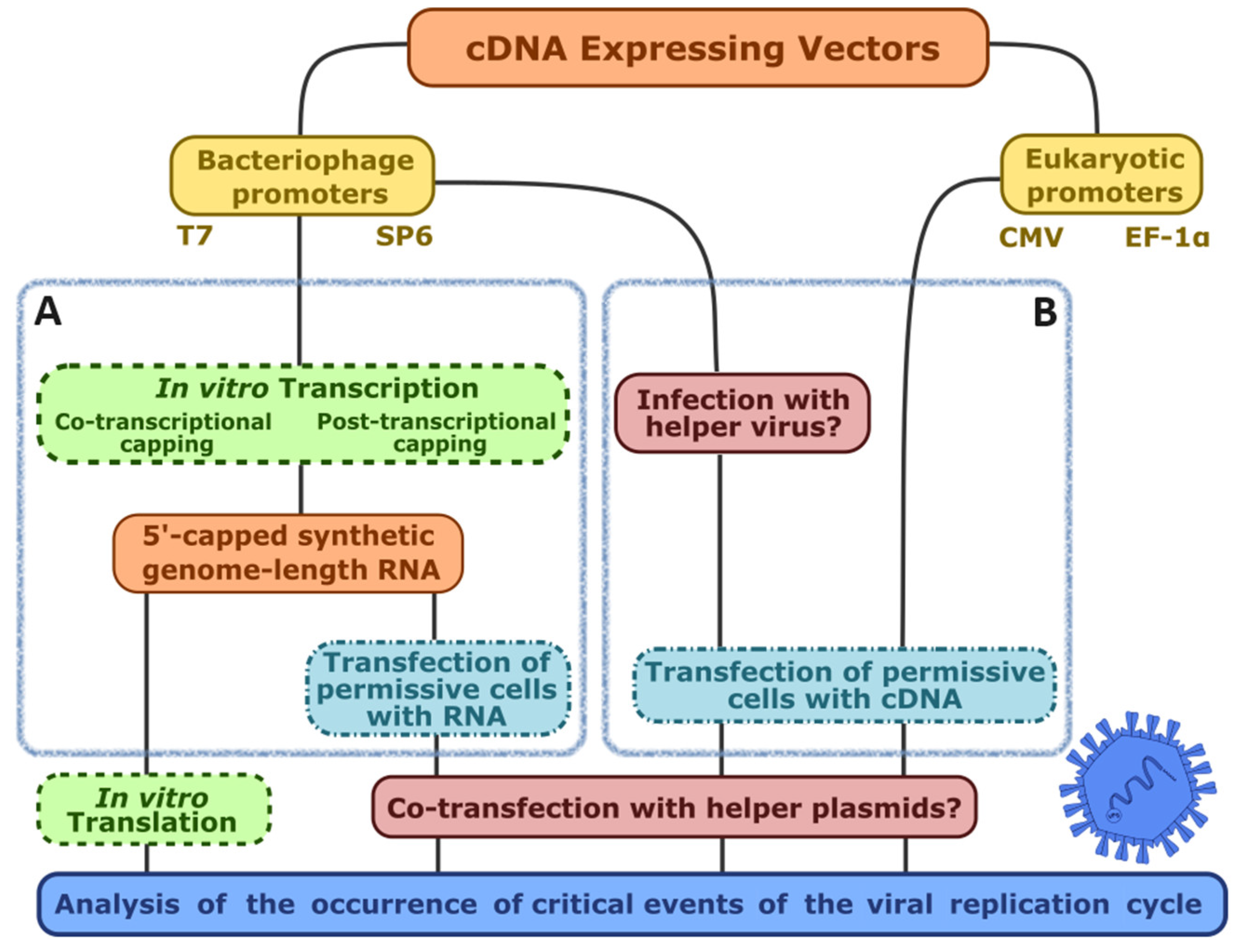
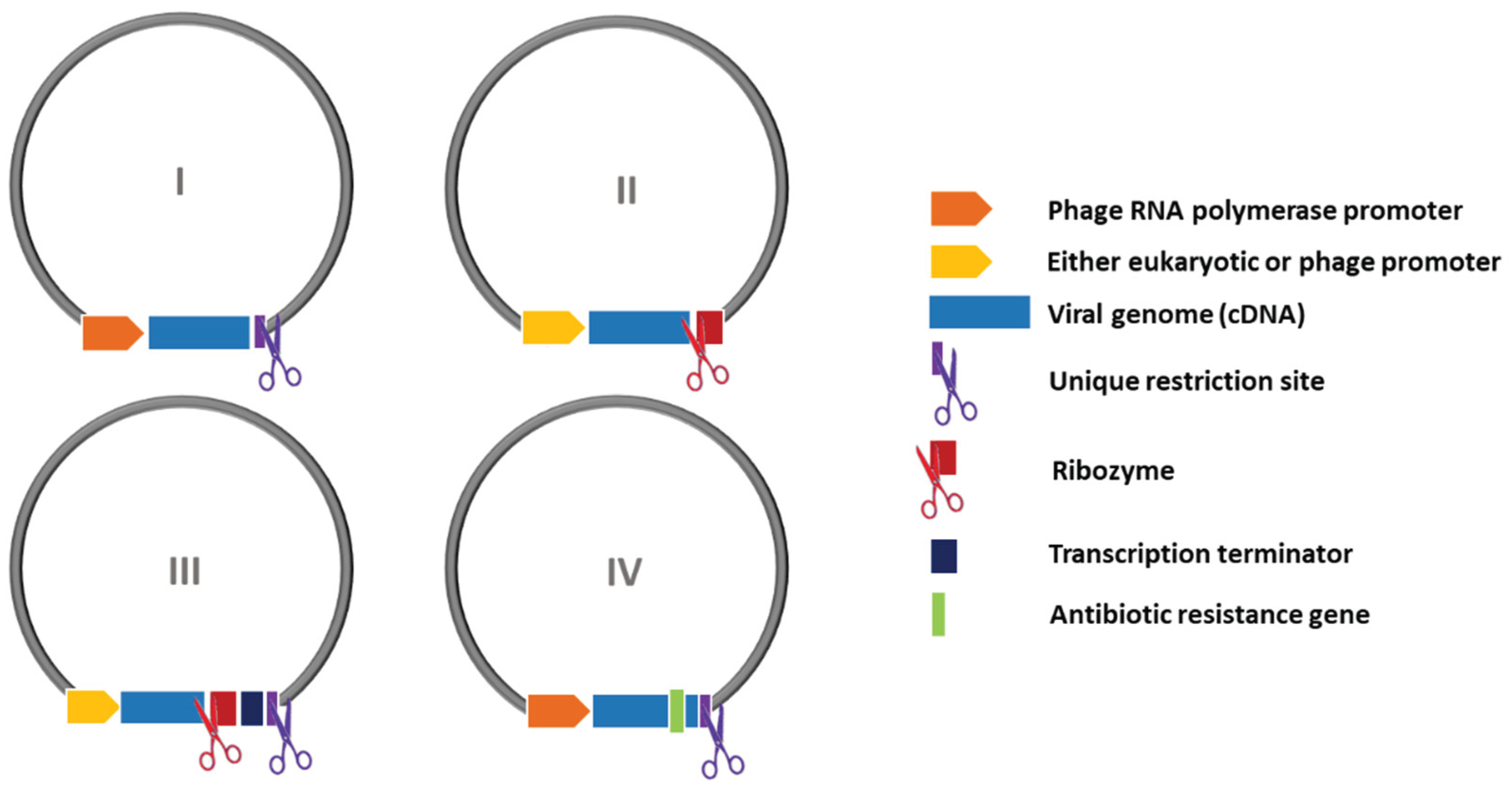
| Promoter | Bacteriophage (T3, T7, SP6, etc.) | Eukaryotic (CMV, EF-1α, SV40, etc.) |
||
|---|---|---|---|---|
| Reverse genetics strategy | Transfection of in vitro-transcribed RNA | cDNA transfection of helper virus-infected cells | cDNA transfection of phage RNA pol-expressing cell line (no helper virus) | RNA pol II-driven nuclear transcription of cDNA |
| Is there virus-induced CPE in transfected cell monolayers (passage 0)? |
|
|||
| Does the supernatant from transfected cells (passage 0) contain infectious virions? |
|
|||
| Is the genome-emulating RNA transcript present in the cytosol? |
|
|||
| Is there any non-viral sequence added to the 5′-end? |
|
|||
| Is ORF 1 being expressed? |
|
|||
| Is the viral protease functional? |
|
|||
| Is the viral RdRp functional? |
|
|||
| Is the negative strand being synthesized? |
|
|||
| Are VP1 or VP2 being synthesized? |
|
|||
| Virus name | Virus recovery strategy and infectious clone features | Design | Year of publication [Reference] |
|---|---|---|---|
| Feline calicivirus | T7 RNA polymerase-driven IVT with co-transcriptional capping, followed by RNA transfection of CRFK cells | I | 1995 {Sosnovtsev, 1995 #1355} |
| Feline calicivirus | T7 RNA polymerase-driven cDNA expression; Poly-(A)32. Two delivery methods:
|
I | 2002 {Thumfart, 2002 #2200} |
| Porcine enteric calicivirus | T7 RNA polymerase-driven cDNA expression. Poly-(A)35. Two delivery methods:
|
I | 2005 {Chang, 2005 #1226} |
| Human norovirus | T7 RNA polymerase promoter: transfection of rMVA-T7-infected 293T cells; Poly-(A)26 | III | 2005 {Asanaka, 2005 #1467} |
| Human norovirus | T7 RNA polymerase promoter: transfection of rVV-T7-infected 293T cells; Poly-(A)30 | III | 2006 {Katayama, 2006 #1468} |
| Human norovirus | No virus rescue: neomycin-resistance gene replacing part of ORF2. Transfection of BHK21 and Huh7 cells with IVT-generated RNA led to the establishment of a VP1-defective replicon that persisted beyond cell passages. Apparently, the replicon further extracted from cells had covalently acquired the 5′-linked VPg. G418 was used for colony selection | IV | 2006 {Chang, 2006 #2207} |
| Murine norovirus-1 | Pol-II-driven: viral cDNA controlled by minCMV promoter; Poly-(A)31; two delivery methods:
|
II | 2007 {Ward, 2007 #1472} |
| Murine norovirus-1 | T7 RNA polymerase-driven cDNA expression. Poly-(A)26. Two helper viruses tested for providing T7 pol:
|
II | 2007 {Chaudhry, 2007 #1117} |
| Tulane virus | T7 RNA polymerase-driven IVT with co-transcriptional capping, followed by RNA transfection of LLC-MK2 cells; poly-(A)17 | I | 2008 {Wei, 2008 #1187} |
| Murine norovirus-1 |
|
II | 2010, 2012 {Yunus, 2010 #1111;Arias, 2012 #2170} |
| Human norovirus | Pol-II-driven cDNA expression: EF-1α promoter. cDNA plasmid was transfected into COS7 cells in the absence of helper virus. Poly-(A)26 | II | 2014 {Katayama, 2014 #1469} |
| Feline calicivirus | Pol-II-driven cDNA expression: EF-1α promoter. cDNA plasmid was transfected into CRFK cells in the absence of helper virus; Poly-(A)30 | II | 2014 {Oka, 2014 #1137} |
| Rabbit vesivirus | T7 RNA polymerase-driven cDNA expression. Poly-(A)30. Two delivery methods:
|
III | 2020 {Álvarez, 2020 #2183} |
| Human norovirus Murine norovirus |
Full-length cDNA with a linker fragment containing CMV promoter synthesized by circular polymerase extension reaction (CPER); transfected in NIH3T3 cells. Poly-(A)30. | II | 2021 {Amarilla, 2021 #2395} |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




