Submitted:
15 March 2024
Posted:
15 March 2024
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Methodology
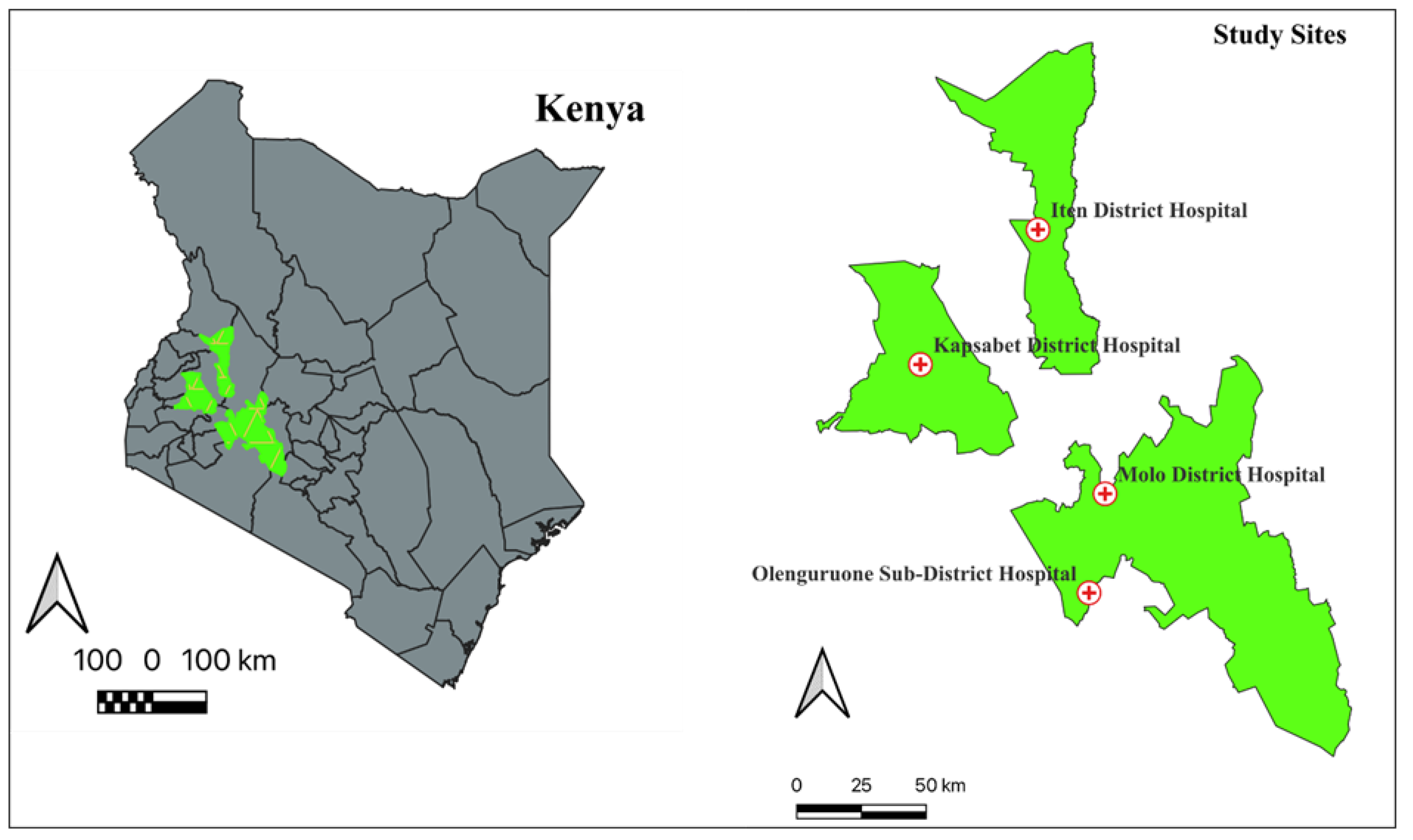
2.1. Study Area
2.2. Study Design and Sample Collection
2.3. Case Definition
2.4. Laboratory Analysis
2.5. Data Analysis
3. Results
3.1. Seropositivity across Age Groups
3.2. Predictors of SARS-CoV-2 Antibody Seroprevalence
4. Discussion
5. Conclusion
Ethics statement
Supplementary Materials
Author Contributions
Funding
Data availability statement
Acknowledgments
Conflicts of interest
References
- Gupta, A., Marzook, H. & Ahmad, F. Comorbidities and clinical complications associated with SARS-CoV-2 infection: an overview. Clinical and Experimental Medicine 2022 23:2 23, 313–331 (2022). [CrossRef]
- Deepanshi, Budhiraja, I., Garg, D., Kumar, N. & Sharma, R. A comprehensive review on variants of SARS-CoVs-2: Challenges, solutions and open issues. Comput Commun 197, 34–51 (2023). [CrossRef]
- Mathieu, E. et al. Coronavirus Pandemic (COVID-19). Our World in Data (2020).
- Mercer, T. R. & Salit, M. Testing at scale during the COVID-19 pandemic. Nature Reviews Genetics 2021 22:7 22, 415–426 (2021). [CrossRef]
- Usuf, E. & Roca, A. Seroprevalence surveys in sub-Saharan Africa: what do they tell us? Lancet Glob Health 9, e724–e725 (2021). [CrossRef]
- Bergeri, I. et al. Global SARS-CoV-2 seroprevalence from January 2020 to April 2022: A systematic review and meta-analysis of standardized population-based studies. PLoS Med 19, (2022). [CrossRef]
- True extent of SARS-CoV-2 Infection through seroprevalence studies. https://www.who.int/news/item/03-02-2022-true-extent-of-sars-cov-2-infection-through-seroprevalence-studies.
- Kenya completes its first round of COVID-19 vaccinations | Gavi, the Vaccine Alliance. https://www.gavi.org/vaccineswork/kenya-completes-its-first-round-covid-19-vaccinations.
- Mulabbi, E. N. et al. Seroprevalence of human coronaviruses among patients visiting hospital-based sentinel sites in Uganda. BMC Infect Dis 21, 1–8 (2021). [CrossRef]
- Uyoga, S. et al. Prevalence of SARS-CoV-2 Antibodies From a National Serosurveillance of Kenyan Blood Donors, January-March 2021. JAMA 326, 1436–1438 (2021).
- Zaballa, M. E. et al. Seroprevalence of anti-SARS-CoV-2 antibodies and cross-variant neutralization capacity after the Omicron BA.2 wave in Geneva, Switzerland: a population-based study. The Lancet Regional Health - Europe 24, 100547 (2023).
- Kagucia, E. W. et al. SARS-CoV-2 seroprevalence and implications for population immunity: Evidence from two Health and Demographic Surveillance System sites in Kenya, February-December 2022. Influenza Other Respir Viruses 17, (2023).
- Han, D., Li, R., Han, Y., Zhang, R. & Li, J. COVID-19: Insight into the asymptomatic SARS-COV-2 infection and transmission. Int J Biol Sci 16, 2803 (2020). [CrossRef]
- Rostami, A. et al. SARS-CoV-2 seroprevalence worldwide: a systematic review and meta-analysis. Clin Microbiol Infect 27, 331–340 (2021). [CrossRef]
- Bobrovitz, N. et al. Global seroprevalence of SARS-CoV-2 antibodies: A systematic review and meta-analysis. PLoS One 16, e0252617 (2021). [CrossRef]
- Briggs, J. et al. Seroprevalence of Antibodies to SARS-CoV-2 in Rural Households in Eastern Uganda, 2020-2022. JAMA Netw Open 6, e2255978–e2255978 (2023). [CrossRef]
- Gibbons, C. L. et al. Measuring underreporting and under-ascertainment in infectious disease datasets: A comparison of methods. BMC Public Health 14, 1–17 (2014). [CrossRef]
- Adebisi, Y. A. et al. SARS-CoV-2 diagnostic testing in Africa: needs and challenges. Pan Afr Med J 35, 4 (2020). [CrossRef]
- Dzinamarira, T., Dzobo, M. & Chitungo, I. COVID-19: A perspective on Africa’s capacity and response. J Med Virol 92, 2465–2472 (2020). [CrossRef]
- Wu, S. L. et al. Substantial underestimation of SARS-CoV-2 infection in the United States. Nature Communications 2020 11:1 11, 1–10 (2020). [CrossRef]
- Sewpaul, R. et al. Vaccine hesitancy and related factors among South African adults in 2021: unpacking uncertainty versus unwillingness. Front Public Health 11, (2023). [CrossRef]
- Troiano, G. & Nardi, A. Vaccine hesitancy in the era of COVID-19. Public Health 194, 245–251 (2021). [CrossRef]
- Sallam, M. COVID-19 Vaccine Hesitancy Worldwide: A Concise Systematic Review of Vaccine Acceptance Rates. Vaccines 2021, Vol. 9, Page 160 9, 160 (2021). [CrossRef]
- Sinnei, D. K., Karimi, P. N., Maru, S. M., Karengera, S. & Bizimana, T. Evaluation of vaccine storage and distribution practices in rural healthcare facilities in Kenya. J Pharm Policy Pract 16, 1–7 (2023). [CrossRef]
- Ortiz, J. R. et al. The potential effects of deploying SARS-Cov-2 vaccines on cold storage capacity and immunization workload in countries of the WHO African Region. Vaccine 39, 2165–2176 (2021). [CrossRef]
- Cheetham, N. J. et al. Antibody levels following vaccination against SARS-CoV-2: associations with post-vaccination infection and risk factors in two UK longitudinal studies. Elife 12, (2023).
- Rajshekhar, N. et al. Original research: Exploring COVID-19 vaccine hesitancy and uptake in Nairobi’s urban informal settlements: an unsupervised machine learning analysis of a longitudinal prospective cohort study from 2021 to 2022. BMJ Open 13, (2023).
- Bakibinga, P. et al. Demand and supply-side barriers and opportunities to enhance access to healthcare for urban poor populations in Kenya: a qualitative study. BMJ Open 12, e057484 (2022). [CrossRef]
- Otieno, P. O. et al. Access to primary healthcare services and associated factors in urban slums in Nairobi-Kenya. BMC Public Health 20, 1–9 (2020). [CrossRef]
- Kirkland, E. et al. Patient Demographics and Clinic Type Are Associated With Patient Engagement Within a Remote Monitoring Program. Telemedicine Journal and e-Health 27, 843 (2021). [CrossRef]
- Lewis, D. J. & Longley, P. A. Patterns of Patient Registration with Primary Health Care in the UK National Health Service. Annals of the Association of American Geographers 102, 1135–1145 (2012). [CrossRef]
| Demographic characteristics | Numbers (%) | Mean (Stdev) |
|---|---|---|
| Male | 218 (39.1) | |
| Female | 339 (60.9) | |
| Total sample size | 557 | |
| Age groups | ||
| 0-18 | 67 (12.0) | 15.1 (3.4) |
| 19-30 | 127 (22.8) | 25.2 (3.6) |
| 31-40 | 99 (17.8) | 35.7 (2.9) |
| 41-50 | 96 (17.2) | 46.1 (3.1) |
| 51-60 | 65 (11.7) | 54.6 (2.6) |
| 61> | 103 (18.5) | 70.5 (7.9) |
| Vaccinated | ||
| Male | 127(22.8) | |
| Female | 181(32.5) | |
| Prior COVID-19 infection | ||
| Female | 22(4.0) | |
| Male | 17(3.1) | |
| Positivity | ||
| Negative | 12 (2.2) | |
| Positive | 545 (97.8) |
| Age Groups | ICRH[1] | KCRH[2] | Molo | Olenguruone |
|---|---|---|---|---|
| 0-18 (n=67) | 28 | 14 | 15 | 10 |
| 19-30 (n=127) | 52 | 25 | 35 | 15 |
| 31-40 (n=99) | 32 | 24 | 23 | 20 |
| 41-50 (n=96) | 40 | 14 | 16 | 26 |
| 51-60 (n=65) | 32 | 7 | 16 | 10 |
| 61> (n=103) | 53 | 19 | 21 | 10 |
| All | 237 | 103 | 126 | 91 |
| Vaccination | ||||
|---|---|---|---|---|
| Female | Male | |||
| No (%) | Yes (%) | No (%) | Yes (%) | |
| ICRH (n=237) | 77(32.5) | 74 (31.2) | 41 (17.3) | 45 (19) |
| KCRH (n=103) | 21 (20.4) | 29 (28.2) | 15 (14.6) | 38 (36.9) |
| Molo (n=126) | 20 (15.9) | 62 (49.2) | 13 (10.3) | 31 (24.6) |
| Olenguruone (n=91) | 40 (44) | 16 (17.6) | 22 (24.2) | 13 (14.3) |
| Prior COVID-19 Infection | ||||
|---|---|---|---|---|
| Female | Male | |||
| No (%) | Yes (%) | No (%) | Yes (%) | |
| ICRH (n=237) | 149 (62.9) | 2 (0.8) | 86 (36.3) | 0 (0) |
| KCRH (n=103) | 32 (31.1) | 18 (17.5) | 37 (35.9) | 16 (15.5) |
| Molo (n=126) | 80 (63.5) | 2 (1.6) | 43 (34.1) | 1 (0.8) |
| Olenguruone (n=91) | 56 (61.5) | 0 (0) | 35(38.5) | 0 (0) |
| Facility | Age Groups | Negative | Positive | % Positivity prevalence |
|---|---|---|---|---|
| ICRH[3] | ||||
| 0-18 (n=28) | 1 | 27 | 96.4 | |
| 19-30(n=52) | 0 | 52 | 100 | |
| 31-40(n=32) | 0 | 32 | 100 | |
| 41-50(n=40) | 0 | 40 | 100 | |
| 51-60(n=32) | 1 | 31 | 96.9 | |
| >61(n=53) | 1 | 52 | 98.1 | |
| KCRH[4] | ||||
| 0-18(n=14) | 0 | 14 | 100 | |
| 19-30(n=25) | 0 | 25 | 100 | |
| 31-40(n=24) | 0 | 24 | 100 | |
| 41-50(14) | 0 | 14 | 100 | |
| 51-60(n=7) | 0 | 7 | 100 | |
| 61>(n=19) | 0 | 19 | 100 | |
| Molo | ||||
| 0-18(n=15) | 0 | 15 | 100 | |
| 19-30(n=35) | 1 | 34 | 97.1 | |
| 31-40(n=23) | 1 | 22 | 95.7 | |
| 41-50(n=16) | 2 | 14 | 87.5 | |
| 51-60(n=16) | 1 | 15 | 93.8 | |
| 61>(n=21) | 2 | 19 | 90.5 | |
| Olenguruone | ||||
| 0-18(n=10) | 0 | 10 | 100 | |
| 19-30(n=15) | 0 | 15 | 100 | |
| 31-40(n=20) | 1 | 19 | 95 | |
| 41-50(n=26) | 1 | 25 | 96.2 | |
| 51-60(n=10) | 0 | 10 | 100 | |
| 61>(n=10) | 0 | 10 | 100[5] |
| Predictors | Odds Ratio | 95% CI | P-values |
|---|---|---|---|
| Gender | 0.784 | ||
| Male (Ref) vs. Female | 0.76 | (0.10, 5.59) | |
| Age Group | |||
| 0 -18 | Ref | ||
| 19-30 | 7.3448 | (0.14, 386.33) | 0.324 |
| 31-40 | 7.89 | (0.04, 1868.54) | 0.459 |
| 41-50 | 13.36 | (0.01, 21352.24) | 0.491 |
| 51-60 | 5.44 | (0.00, 35775.08) | 0.706 |
| >61 | 26.62 | (0.00, 5.04422E+06) | 0.597 |
| Age | 0.95 | (0.78, 1.17) | 0.649 |
| Elisa Quantification | 1.0 | (1.0, 1.0) | 0.000 |
| Prior SARS CoV-2 Infection | |||
| Yes vs. No | 3285.29 | (0.00, 1.62E+248) | 0.978 |
| Vaccinated | |||
| Yes vs. No | 3.33 | (0.4, 27.87) | 0.267 |
| Facility | |||
| ICRH | Ref | ||
| KCRH | 21939.78 | (0.00, 6.49507E+175) | 0.960 |
| Molo | 0.17 | (0.01, 1.55) | 0.116 |
| Olenguruone | 4.99 | (0.08, 309.27) | 0.445 |
| 1 | ICRH – Iten County Referral Hospital. |
| 2 | KCRH - Kapsabet County Referral Hospital. |
| 3 | ICRH – Iten County Referral Hospital. |
| 4 | KCRH - Kapsabet County Referral Hospital. |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




