Submitted:
15 March 2024
Posted:
15 March 2024
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
2.1. Raw Materials
2.2. Chemical Analytical Methods
2.3. Pretreatment Procedure
2.4. Crystallinity and Morphology Analysis
2.5. Enzymatic Hydrolysis
2.6. Yeast Strain
2.7. Preparation of Seed Culture
2.8. Preparation of T. latifolia Biomass Hydrolysate
2.9. Medium for Bioethanol
2.10. Ethanol Production
2.11. Determination of Yeast Cell Growth
2.12. Statistical Analysis
3. Results
3.1. Characterization of T. latifolia Biomass
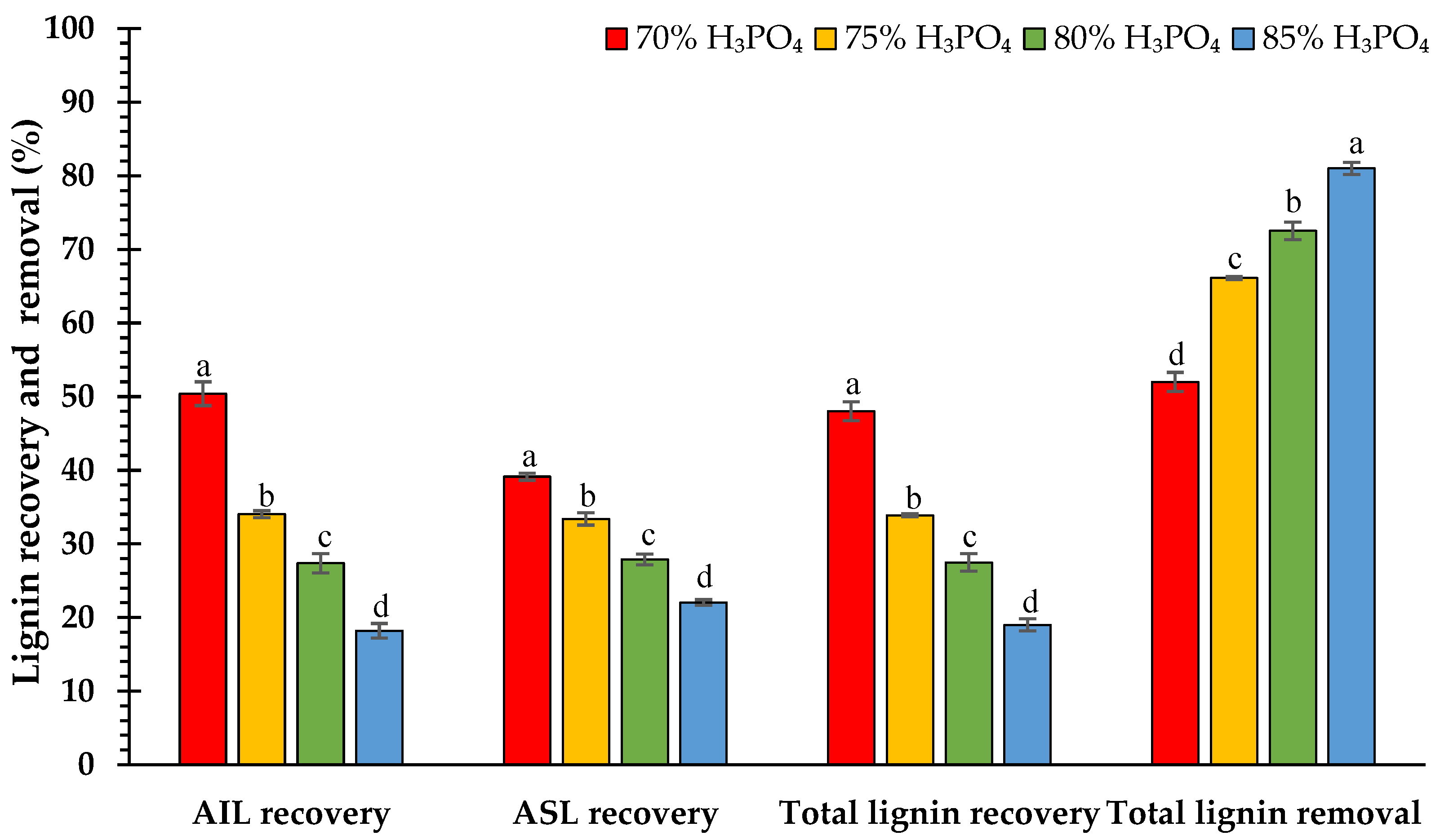
3.2. Effect of H3PO4 Concentrations on Chemical Composition
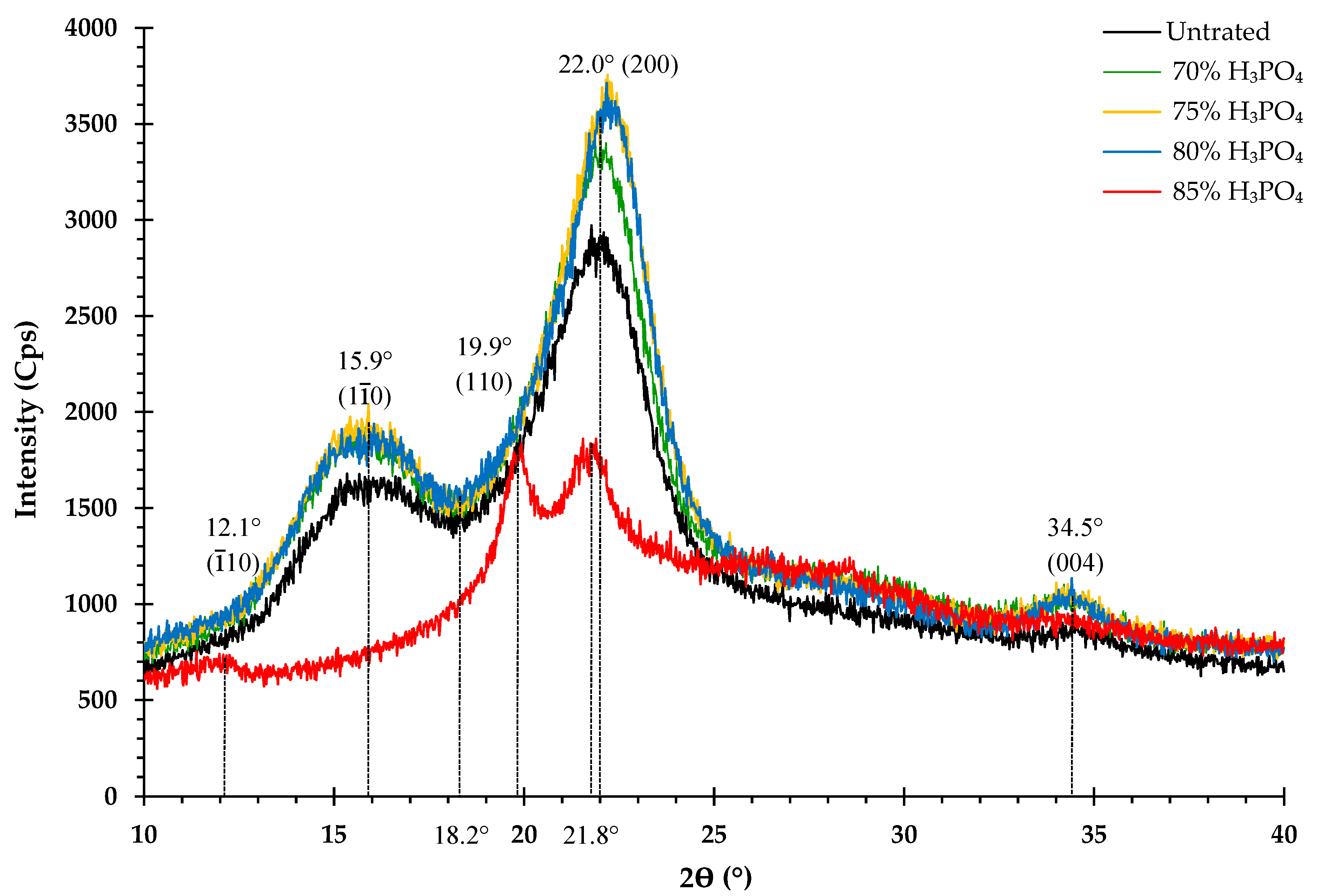
3.3. Impact of H3PO4 Concentration on Cellulose Crystallinity
3.4. Impact of H3PO4 Concentration on Biomass Morphology
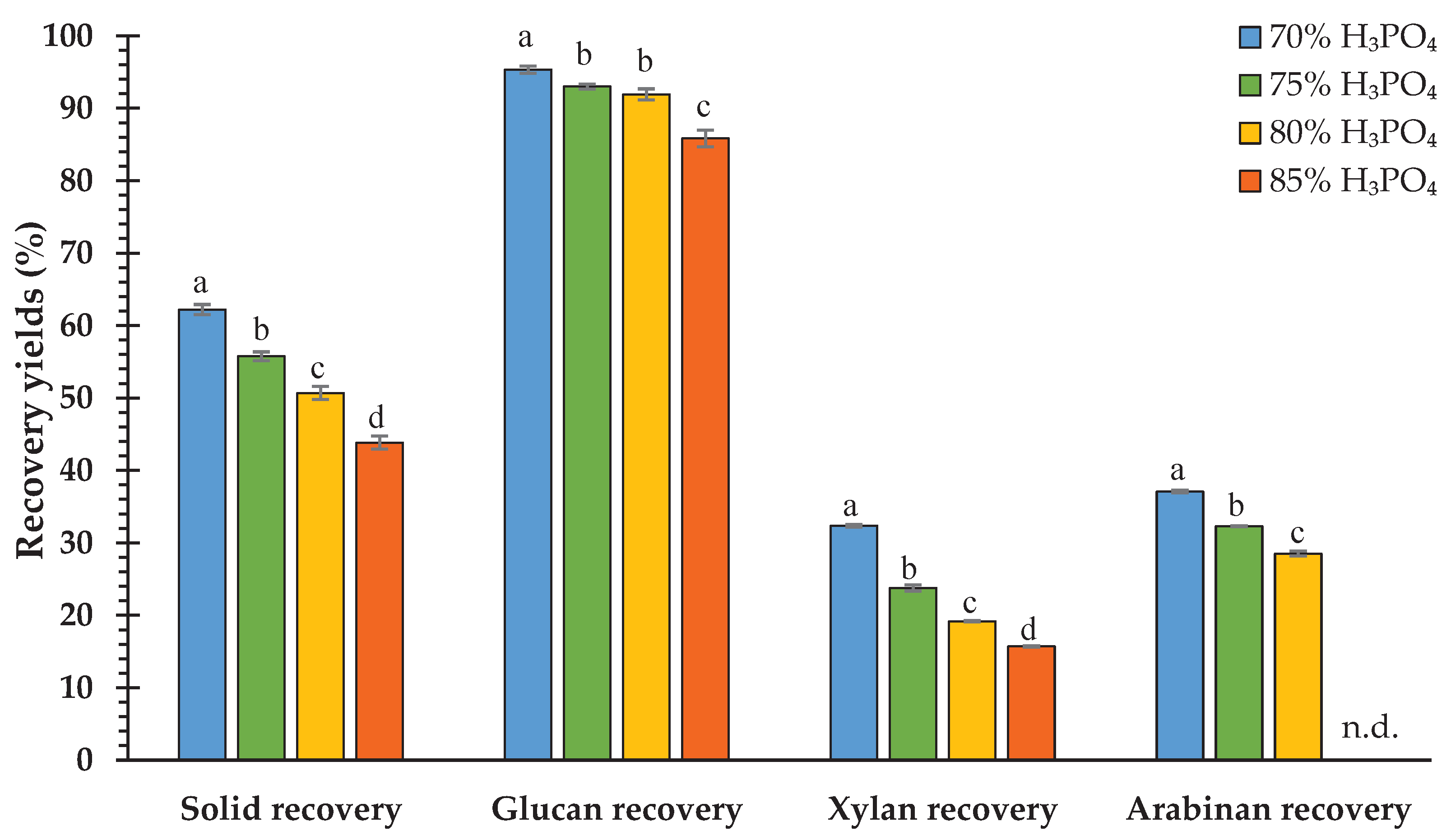
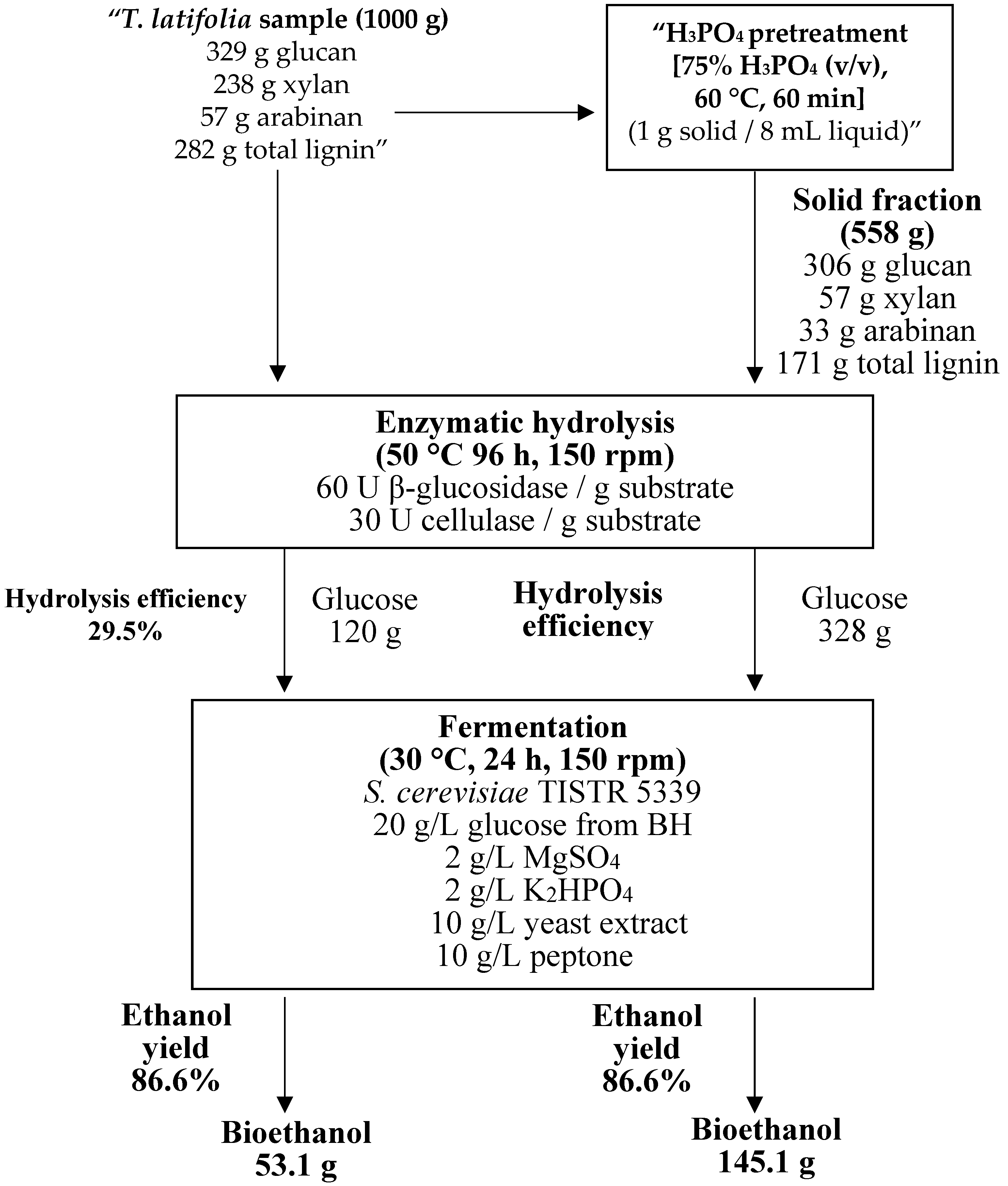
3.5. Enzymatic Saccharification Yields
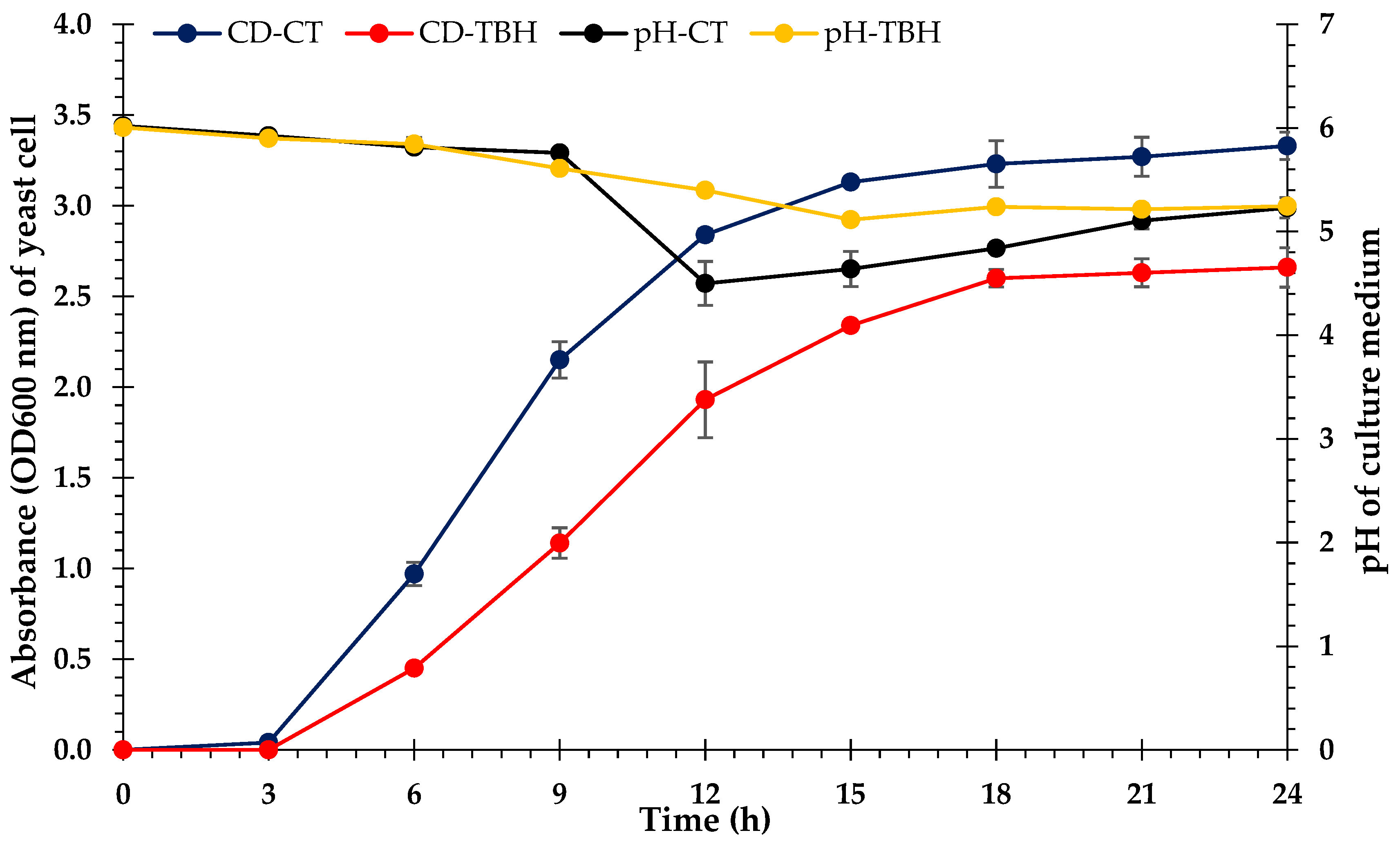
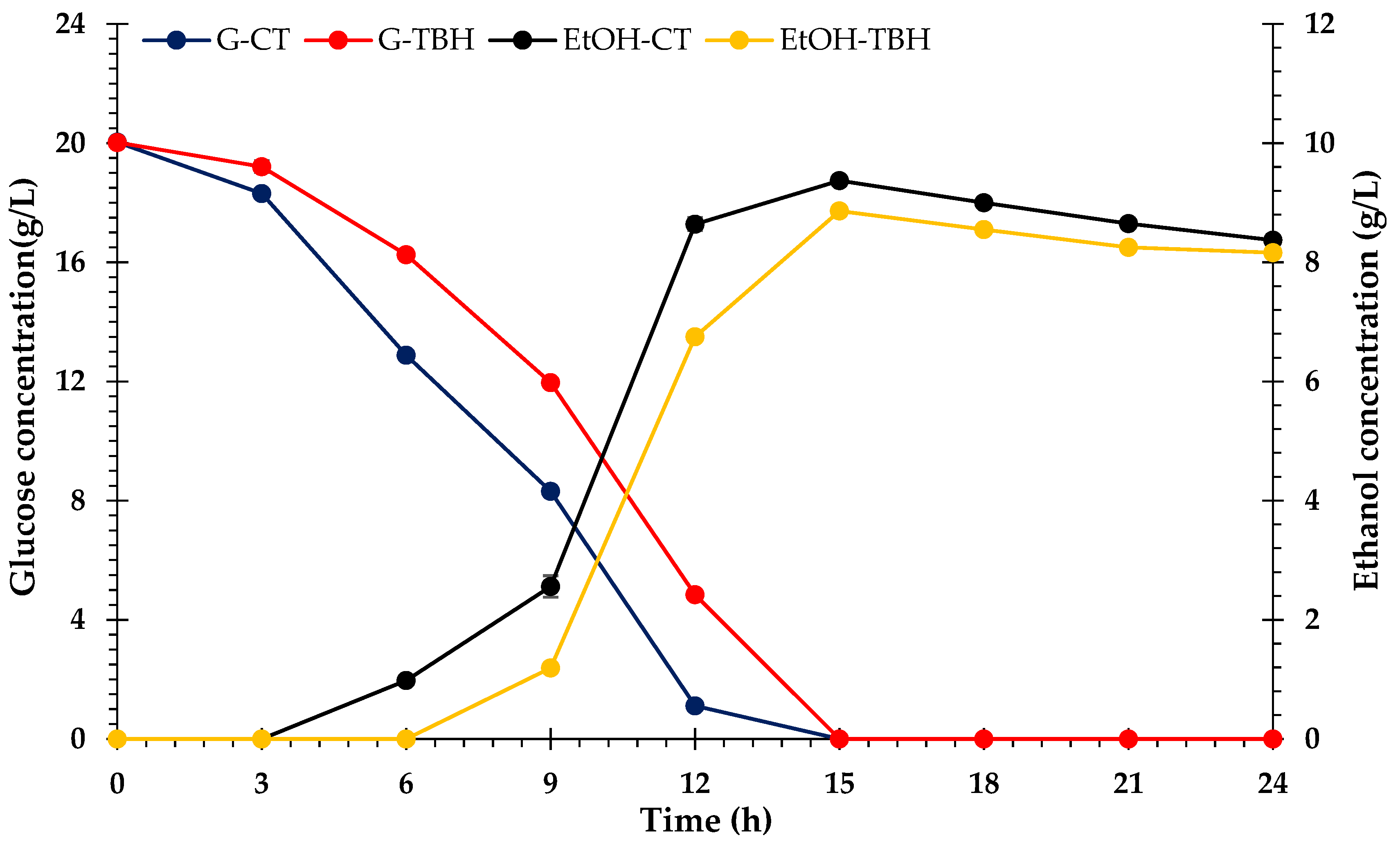
3.6. Bioethanol Fermentation
4. Discussion
4.1. Characterization of T. latifolia Biomass
4.2. Effect of H3PO4 Concentrations on Chemical Composition
4.3. Impact of H3PO4 Concentration on Cellulose Crystallinity
4.4. Impact of H3PO4 Concentration on Biomass Morphology
4.5. Enzymatic Saccharification Yields
4.6. Bioethanol Fermentation
5. Conclusions
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Afraz, M.; Muhammad, F.; Nisar, J.; Shah, A.; Munir, S.; Ali, G.; Ahmad, A. Production of value added products from biomass waste by pyrolysis: An updated review. Waste Management Bulletin. 2024, 1, 30–40. [Google Scholar] [CrossRef]
- Mankar, A.R.; Pandey, A.; Modak, A.; Pant, K.K. Pretreatment of lignocellulosic biomass: A review on recent advances. Bioresour. Technol. 2021, 334, 125235. [Google Scholar] [CrossRef]
- Zadeh, Z.E.; Abdulkhani, A.; Aboelazayem, O.; Saha, B. Recent insights into lignocellulosic biomass pyrolysis: A critical review on pretreatment, characterization, and products upgrading. Processes. 2020, 8. [Google Scholar] [CrossRef]
- Zhao, L.; Sun, Z.F.; Zhang, C.C.; Nan, J.; Ren, N.Q.; Lee, D.J.; Chen, C. Advances in pretreatment of lignocellulosic biomass for bioenergy production: Challenges and perspectives. Bioresour. Technol. 2022, 343, 126123. [Google Scholar] [CrossRef]
- Shukla, A.; Kumar, D.; Girdhar, M.; Kumar, A.; Goyal, A.; Malik, T.; Mohan, A. Strategies of pretreatment of feedstocks for optimized bioethanol production: Distinct and integrated approaches. Biotechnol. Biofuels Bioprod. 2023, 16, 44. [Google Scholar] [CrossRef]
- Toor, M.; Kumar, S.S.; Malyan, S.K.; Bishnoi, N.R.; Mathimani, T.; Rajendran, K.; Pugazhendhi, A. An overview on bioethanol production from lignocellulosic feedstocks. Chemosphere. 2020, 242, 125080. [Google Scholar] [CrossRef]
- Bhatia, S.K.J.; Jagtap, S.S.; Bedekar, A.A.; Bhatia, R.K.; Patel, A.K.; Pant, D.; Rajesh Banu, J.; Rao, C.V.; Kim, Y.G.; Yang, Y.H. Recent developments in pretreatment technologies on lignocellulosic biomass: Effect of key parameters, technological improvements, and challenges. Bioresour. Technol. 2020, 300, 122724. [Google Scholar] [CrossRef]
- Sharma, S.; Tsai, M.-L.; Sharma, V.; Sun, P.-P.; Nargotra, P.; Bajaj, B.K.; Chen, C.-W.; Dong, C.-D. Environment friendly pretreatment approaches for the bioconversion of lignocellulosic biomass into biofuels and value-added products. Environments. 2023, 10. [Google Scholar] [CrossRef]
- Hoang, A.T.; Nizetic, S.; Ong, H.C.; Chong, C.T.; Atabani, A.E.; Pham, V.V. Acid-based lignocellulosic biomass biorefinery for bioenergy production: Advantages, application constraints, and perspectives. J. Environ. Manage. 2021, 296, 113194. [Google Scholar] [CrossRef] [PubMed]
- Sun, W.; Li, X.; Zhao, J.; Qin, Y. Pretreatment strategies to enhance enzymatic hydrolysis and cellulosic ethanol production for biorefinery of corn stover. Int. J. Mol. Sci. 2022, 23. [Google Scholar] [CrossRef]
- Xiao, K.; Li, H.; Liu, L.; Liu, X.; Lian, Y. Quantitative comparison of the delignification performance of lignocellulosic biomass pretreatment technologies for enzymatic saccharification. Environ. Sci. Pollut. Res. Int. 2023, 30, 22929–22940. [Google Scholar] [CrossRef]
- Guo, Z.; Zhang, Q.; You, T.; Ji, Z.; Zhang, X.; Qin, Y.; Xu, F. Heteropoly acids enhanced neutral deep eutectic solvent pretreatment for enzymatic hydrolysis and ethanol fermentation of Miscanthus x giganteus under mild conditions. Bioresour. Technol. 2019, 293, 122036. [Google Scholar] [CrossRef]
- Larnaudie, V.; Ferrari, M.D.; Lareo, C. Switchgrass as an alternative biomass for ethanol production in a biorefinery: Perspectives on technology, economics and environmental sustainability. Renew. Sustain. Energy Rev. 2022, 158, 112115. [Google Scholar] [CrossRef]
- Wongleang, S.; Premjet, D.; Premjet, S. Cellulosic ethanol production from weed biomass hydrolysate of Vietnamosasa pusilla. Polymers. 2023, 15, 1103. [Google Scholar] [CrossRef]
- Sespene, J.; Fetalvero, E.; Faminial, T. Tiger grass industry in Marigondon Norte, San Andres, Romblon: Implications for research and development. Romblon State University Research Journal. 2011, 1, 81–95. [Google Scholar]
- Shrestha, S.; Park, J.H.; Cho, J.G.; Lee, D.Y.; Jeong, R.H.; Song, M.C.; Cho, S.K.; Lee, D.S.; Baek, N.I. Phytochemical constituents from the florets of tiger grass Thysanolaena latifolia from Nepal. J. Asian Nat. Prod. Res. 2016, 18, 206–213. [Google Scholar] [CrossRef]
- Pandiyan, K.; Singh, A.; Singh, S.; Saxena, A.K.; Nain, L. Technological interventions for utilization of crop residues and weedy biomass for second generation bio-ethanol production. Renew. Energy. 2019, 132, 723–741. [Google Scholar] [CrossRef]
- Saikia, D.C.; Goswami, T.; Chaliha, B.P. Paper from Thysanolaena maxima. Bioresour. Technol. 1992, 40, 245–248. [Google Scholar] [CrossRef]
- Flora & fauna, Thysanolaena latifolia. Available online: https://www.nparks.gov.sg/florafaunaweb/flora/2/5/2512# (accessed on 20 Jan 2024).
- Komolwanich, T.; Prasertwasu, S.; Khumsupan, D.; Tatijarern, P.; Chaisuwan, T.; Luengnaruemitchai, A.; Wongkasemjit, S. Evaluation of highly efficient monomeric sugar yield from Thai Tiger grass (Thysanolaena maxima). Mater. Res. Innov. 2016, 20, 259–267. [Google Scholar] [CrossRef]
- Kumar, B.; Bhardwaj, N.; Agrawal, K.; Chaturvedi, V.; Verma, P. Current perspective on pretreatment technologies using lignocellulosic biomass: An emerging biorefinery concept. Fuel Process. Technol. 2020, 199, 106244. [Google Scholar] [CrossRef]
- Rollin, J.A.; Zhu, Z.; Sathitsuksanoh, N.; Zhang, Y.H.P. Increasing cellulose accessibility is more important than removing lignin: A comparison of cellulose solvent-based lignocellulose fractionation and soaking in aqueous ammonia. Biotechnol. Bioeng. 2011, 108, 22–30. [Google Scholar] [CrossRef]
- Satari, B.; Karimi, K.; Kumar, R. Cellulose solvent-based pretreatment for enhanced second-generation biofuel production: A review. Sustainable Energy Fuels. 2019, 3, 11–62. [Google Scholar] [CrossRef]
- Obeng, A.; Premjet, D.; Premjet, S. Fermentable sugar production from the peels of two durian (Durio zibethinus Murr.) cultivars by phosphoric acid pretreatment. Resources. 2018, 7, 60. [Google Scholar] [CrossRef]
- Premjet, S.; Dana, S.; Obeng, A.K.; Premjet, D. Enzymatic response to structural and chemical transformations in Hibiscus sabdariffa var. altissima bark and core during phosphoric acid pretreatment. BioRes. 2018, 13, 6778–6789. [Google Scholar] [CrossRef]
- Premjet, D.; Wongleang, S.; Premjet, S. Enhancing glucose recovery from Hibiscus cannabinus L. through phosphoric acid pretreatment. Energies. 2022, 15, 7573. [Google Scholar] [CrossRef]
- Yu, H.; Xiao, W.; Han, L.; Huang, G. Characterization of mechanical pulverization/phosphoric acid pretreatment of corn stover for enzymatic hydrolysis. Bioresour. Technol. 2019, 282, 69–74. [Google Scholar] [CrossRef]
- Jin, Y.; Liu, J.; Yang, H.; Shi, Z.; Zhao, P.; Yang, J. Improving enzymatic saccharification and ethanol production of bamboo residues with sulfomethylation-aided phosphoric acid pretreatment. Ind. Crops Prod. 2021, 170, 113733. [Google Scholar] [CrossRef]
- Siripong, P.; Duangporn, P.; Takata, E.; Tsutsumi, Y. Phosphoric acid pretreatment of Achyranthes aspera and Sida acuta weed biomass to improve enzymatic hydrolysis. Bioresour. Technol. 2016, 203, 303–308. [Google Scholar] [CrossRef]
- Chen, D.; Tang, W.; Wang, H.; Sheng, Y.; Tan, X.; Shi, Y.; Fan, W.; Ge, S. Phosphoric acid pretreatment of poplar to optimize fermentable sugars production based on orthogonal experimental design. Front. Chem. 2023, 11, 1119215. [Google Scholar] [CrossRef]
- Mund, N.K.; Dash, D.; Barik, C.R.; Goud, V.V.; Sahoo, L.; Mishra, P.; Nayak, N.R. Evaluation of efficient glucose release using sodium hydroxide and phosphoric acid as pretreating agents from the biomass of Sesbania grandiflora (L.) pers.: A fast growing tree legume. Bioresour. Technol. 2017, 236, 97–105. [Google Scholar] [CrossRef]
- Sluiter, A.; Hames, B.; Ruiz, R.; Scarlata, C.; Sluiter, J.; Templeton, D. Determination of ash in biomass. In Technical Report NREL/TP-510-42622; National Renewable Energy Laboratory (National Renewable Energy Laboratory, Office of Energy Efficiency and Renewable Energy): Golden, CO, USA, 2008; pp. 1–8. [Google Scholar]
- Sluiter, A.; Hames, B.; Ruiz, R.; Scarlata, C.; Sluiter, J.; Templeton, D.; Crocker, D. Determination of structural carbohydrates and lignin in biomass. In Technical Report NREL/TP-510-42618; National Renewable Energy Laboratory (National Renewable Energy Laboratory, Office of Energy Efficiency and Renewable Energy): Golden, CO, USA, 2012; pp. 1–15. [Google Scholar]
- Sluiter, A.; Ruiz, R.; Scarlata, C.; Sluiter, J.; Templeton, D. Determination of extractives in biomass. In Technical Report NREL/TP-510-42619; National Renewable Energy Laboratory (National Renewable Energy Laboratory, Office of Energy Efficiency and Renewable Energy): Golden, CO, USA, 2008; pp. 1–12. [Google Scholar]
- Segal, L.; Creely, J.J.; Martin, A.E.; Conrad, C.M. An empirical method for estimating the degree of crystallinity of native cellulose using the X-ray diffractometer. Text. Res. J. 1959, 29, 786–794. [Google Scholar] [CrossRef]
- Siripong, P.; Doungporn, P.; Yoo, H.Y.; Kim, S.W. Improvement of sugar recovery from Sida acuta (Thailand weed) by NaOH pretreatment and application to bioethanol production. Korean J. Chem. Eng. 2018, 35, 2413–2420. [Google Scholar] [CrossRef]
- Wongleang, S.; Premjet, D.; Premjet, S. Physicochemical pretreatment of Vietnamosasa pusilla for bioethanol and xylitol production. Polymers. 2023, 15, 3990. [Google Scholar] [CrossRef]
- Binod, P.; Sindhu, R.; Singhania, R.R.; Vikram, S.; Devi, L.; Nagalakshmi, S.; Kurien, N.; Sukumaran, R.K.; Pandey, A. Bioethanol production from rice straw: An overview. Bioresour. Technol. 2010, 101, 4767–4774. [Google Scholar] [CrossRef]
- Khan, M.A.; Dharmalingam, B.; Chuetor, S.; Cheng, Y.-S.; Sriariyanun, M. Comprehensive review on effective conversion of lignocellulosic biomass to levulinic acid. Biomass Convers. Biorefin. 2023. [Google Scholar] [CrossRef]
- Manyi-Loh, C.E.; Lues, R. Anaerobic digestion of lignocellulosic biomass: Substrate characteristics (challenge) and innovation. Fermentation. 2023, 9. [Google Scholar] [CrossRef]
- Li, H.Y.; Chen, X.; Wang, C.Z.; Sun, S.N.; Sun, R.C. Evaluation of the two-step treatment with ionic liquids and alkali for enhancing enzymatic hydrolysis of Eucalyptus: Chemical and anatomical changes. Biotechnol. Biofuels. 2016, 9, 166. [Google Scholar] [CrossRef]
- Pan, S.; Chi, Y.; Zhou, L.; Li, Z.; Du, L.; Wei, Y. Evaluation of squeezing pretreatment for improving methane production from fresh banana pseudo-stems. Waste Manag. 2020, 102, 900–908. [Google Scholar] [CrossRef]
- Montoya-Rosales, J.J.; Peces, M.; González-Rodríguez, L.M.; Alatriste-Mondragón, F.; Villa-Gómez, D.K. A broad overview comparing a fungal, thermal and acid pre-treatment of bean straw in terms of substrate and anaerobic digestion effect. Biomass Bioenergy. 2020, 142, 105775. [Google Scholar] [CrossRef]
- Bonfiglio, F.; Cagno, M.; Yamakawa, C.K.; Mussatto, S.I. Production of xylitol and carotenoids from Switchgrass and Eucalyptus globulus hydrolysates obtained by intensified steam explosion pretreatment. Ind. Crops Prod. 2021, 170, 113800. [Google Scholar] [CrossRef]
- Fonseca, B.G.; Mateo, S.; Roberto, I.C.; Sánchez, S.; Moya, A.J. Bioconversion in batch bioreactor of olive-tree pruning biomass optimizing treatments for ethanol production. Biochem. Eng. J. 2020, 164, 107793. [Google Scholar] [CrossRef]
- Liu, Y.; Zheng, X.; Tao, S.; Hu, L.; Zhang, X.; Lin, X. Process optimization for deep eutectic solvent pretreatment and enzymatic hydrolysis of sugar cane bagasse for cellulosic ethanol fermentation. Renew. Energy. 2021, 177, 259–267. [Google Scholar] [CrossRef]
- Broda, M.; Yelle, D.J.; Serwańska, K. Bioethanol production from lignocellulosic biomass—Challenges and solutions. Molecules. 2022, 27. [Google Scholar] [CrossRef]
- Li, T.; Fang, Q.; Chen, H.; Qi, F.; Ou, X.; Zhao, X.; Liu, D. Solvent-based delignification and decrystallization of wheat straw for efficient enzymatic hydrolysis of cellulose and ethanol production with low cellulase loadings. RSC Adv. 2017, 7, 10609–10617. [Google Scholar] [CrossRef]
- Rahmati, S.; Doherty, W.; Dubal, D.; Atanda, L.; Moghaddam, L.; Sonar, P.; Hessel, V.; Ostrikov, K. Pretreatment and fermentation of lignocellulosic biomass: Reaction mechanisms and process engineering. React. Chem. Eng. 2020, 5, 2017–2047. [Google Scholar] [CrossRef]
- Arora, A.; Nandal, P.; Singh, J.; Verma, M.L. Nanobiotechnological advancements in lignocellulosic biomass pretreatment. Mater. Sci. Energy Technol. 2020, 3, 308–318. [Google Scholar] [CrossRef]
- Yoo, C.G.; Meng, X.; Pu, Y.; Ragauskas, A.J. The critical role of lignin in lignocellulosic biomass conversion and recent pretreatment strategies: A comprehensive review. Bioresour. Technol. 2020, 301, 122784. [Google Scholar] [CrossRef]
- Kundu, C.; Samudrala, S.P.; Kibria, M.A.; Bhattacharya, S. One-step peracetic acid pretreatment of hardwood and softwood biomass for platform chemicals production. Sci. Rep. 2021, 11, 11183. [Google Scholar] [CrossRef]
- Lorenci Woiciechowski, A.; Dalmas Neto, C.J.; Porto de Souza Vandenberghe, L.; de Carvalho Neto, D.P.; Novak Sydney, A.C.; Letti, L.A.J.; Karp, S.G.; Zevallos Torres, L.A.; Soccol, C.R. Lignocellulosic biomass: Acid and alkaline pretreatments and their effects on biomass recalcitrance—Conventional processing and recent advances. Bioresour. Technol. 2020, 304, 122848. [Google Scholar] [CrossRef]
- Zhang, Y.H.; Ding, S.Y.; Mielenz, J.R.; Cui, J.B.; Elander, R.T.; Laser, M.; Himmel, M.E.; McMillan, J.R.; Lynd, L.R. Fractionating recalcitrant lignocellulose at modest reaction conditions. Biotechnol. Bioeng. 2007, 97, 214–223. [Google Scholar] [CrossRef]
- Prasad, S.; Malav, M.K.; Kumar, S.; Singh, A.; Pant, D.; Radhakrishnan, S. Enhancement of bio-ethanol production potential of wheat straw by reducing furfural and 5-hydroxymethylfurfural (HMF). Bioresour. Technol. Rep. 2018, 4, 50–56. [Google Scholar] [CrossRef]
- Sathitsuksanoh, N.; Zhu, Z.; Zhang, Y.H.P. Cellulose solvent- and organic solvent-based lignocellulose fractionation enabled efficient sugar release from a variety of lignocellulosic feedstocks. Bioresour. Technol. 2012, 117, 228–233. [Google Scholar] [CrossRef]
- Zhang, Y.; Guo, Y.; Xie, X.; Chernyshev, V.M.; Liu, Y.; Qi, W. Effects of phosphoric acid/hydrogen peroxide, ammonia/hydrogen peroxide and deep eutectic solvent pretreatments on component separation and enzymatic saccharification of Glycyrrhiza residue. Ind. Crops Prod. 2023, 196, 116525. [Google Scholar] [CrossRef]
- Wongleang, S.; Dana, S.; Premjet, D.; Premjet, S. Phosphoric acid pretreatment of Corchorus capsularis L. biomass for enhancing glucose recovery. NU. Int. J. Sci. 2023, 20, 1–13. [Google Scholar]
- Wang, Q.; Shen, F.; Yang, G.; Zhang, Y.; Deng, S.; Hu, Y.; Zhang, J.; Song, C.; Zeng, Y. Pretreating Luffa sponge (Luffa cylindrica L.) with concentrated phosphoric acid and subsequent enzymatic saccharification. BioResources. 2016, 11, 14. [Google Scholar] [CrossRef]
- Ling, Z.; Chen, S.; Zhang, X.; Xu, F. Exploring crystalline-structural variations of cellulose during alkaline pretreatment for enhanced enzymatic hydrolysis. Bioresour. Technol. 2017, 224, 611–617. [Google Scholar] [CrossRef]
- Zoghlami, A.; Paës, G. Lignocellulosic biomass: Understanding recalcitrance and predicting hydrolysis. Front. Chem. 2019, 7, 874. [Google Scholar] [CrossRef]
- Hossain, A.; Rahaman, M.S.; Lee, D.; Phung, T.K.; Canlas, C.G.; Simmons, B.A.; Renneckar, S.; Reynolds, W.; George, A.; Tulaphol, S.; et al. Enhanced softwood cellulose accessibility by H3PO4 pretreatment: High sugar yield without compromising lignin integrity. Ind. Eng. Chem. Res. 2020, 59, 1010–1024. [Google Scholar] [CrossRef]
- Wang, Q.; Tian, D.; Hu, J.; Shen, F.; Yang, G.; Zhang, Y.; Deng, S.; Zhang, J.; Zeng, Y.; Hu, Y. Fates of hemicellulose, lignin and cellulose in concentrated phosphoric acid with hydrogen peroxide (PHP) pretreatment. RSC Adv. 2018, 8, 12714–12723. [Google Scholar] [CrossRef]
- Cai, D.; Li, P.; Chen, C.; Wang, Y.; Hu, S.; Cui, C.; Qin, P.; Tan, T. Effect of chemical pretreatments on corn stalk bagasse as immobilizing carrier of Clostridium acetobutylicum in the performance of a fermentation-pervaporation coupled system. Bioresour. Technol. 2016, 220, 68–75. [Google Scholar] [CrossRef]
- Obeng, A.K.; Premjet, D.; Premjet, S. Improved glucose recovery from durian peel by alkaline-catalyzed steam pretreatment. PeerJ. 2021, 9, e12026. [Google Scholar] [CrossRef]
- Zheng, Q.; Zhou, T.; Wang, Y.; Cao, X.; Wu, S.; Zhao, M.; Wang, H.; Xu, M.; Zheng, B.; Zheng, J.; et al. Pretreatment of wheat straw leads to structural changes and improved enzymatic hydrolysis. Sci. Rep. 2018, 8, 1321. [Google Scholar] [CrossRef]
- Du, X.; Lucia, L.A.; Ghiladi, R.A. Development of a highly efficient pretreatment sequence for the enzymatic saccharification of Loblolly pine wood. ACS Sustainable Chem. Eng. 2016, 4, 3669–3678. [Google Scholar] [CrossRef]
- Sathitsuksanoh, N.; Zhu, Z.; Wi, S.; Zhang, Y.H. Cellulose solvent-based biomass pretreatment breaks highly ordered hydrogen bonds in cellulose fibers of switchgrass. Biotechnol. Bioeng. 2011, 108, 521–529. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.H.; Cui, J.; Lynd, L.R.; Kuang, L.R. A transition from cellulose swelling to cellulose dissolution by o-phosphoric acid: Evidence from enzymatic hydrolysis and supramolecular structure. Biomacromolecules. 2006, 7, 644–648. [Google Scholar] [CrossRef]
- Zhang, J.; Zhang, J.; Lin, L.; Chen, T.; Zhang, J.; Liu, S.; Li, Z.; Ouyang, P. Dissolution of microcrystalline cellulose in phosphoric acid—Molecular changes and kinetics. Molecules. 2009, 14, 5027–5041. [Google Scholar] [CrossRef]
- Gabhane, J.; Prince William, S.P.M.; Vaidya, A.N.; Mahapatra, K.; Chakrabarti, T. Influence of heating source on the efficacy of lignocellulosic pretreatment—A cellulosic ethanol perspective. Biomass Bioenergy. 2011, 35, 96–102. [Google Scholar] [CrossRef]
- Gourlay, K.; Arantes, V.; Saddler, J.N. Use of substructure-specific carbohydrate binding modules to track changes in cellulose accessibility and surface morphology during the amorphogenesis step of enzymatic hydrolysis. Biotechnol. Biofuels. 2012, 5, 51. [Google Scholar] [CrossRef]
- Jackson de Moraes Rocha, G.; Martin, C.; Soares, I.B.; Souto Maior, A.M.; Baudel, H.M.; Moraes de Abreu, C.A. Dilute mixed-acid pretreatment of sugarcane bagasse for ethanol production. Biomass Bioenergy. 2011, 35, 663–670. [Google Scholar] [CrossRef]
- Tong, D.; Zhan, P.; Zhang, W.; Zhou, Y.; Huang, Y.; Qing, Y.; Chen, J. Surfactant-assisted dilute phosphoric acid plus steam explosion of poplar for fermentable sugar production. ChemistrySelect. 2022, 7, e202200423. [Google Scholar] [CrossRef]
- Tong, W.; Fang, H.; Song, K.; Xie, X.; Wang, J.; Jin, Y.; Wu, S.; Hu, J.; Chu, Q. Modified acid pretreatment to alter physicochemical properties of biomass for full cellulose/hemicellulose utilization. Carbohydr. Polym. 2023, 299, 120182. [Google Scholar] [CrossRef]
- Haldar, D.; Purkait, M.K. Thermochemical pretreatment enhanced bioconversion of Elephant grass (Pennisetum purpureum): Insight on the production of sugars and lignin. Biomass Convers. Biorefin. 2022, 12, 1125–1138. [Google Scholar] [CrossRef]
- Shiva, R.-J., R. M.; Rodríguez-Jasso, R.M.; Rosero-Chasoy, G.; López-Sandin, I.; Morais, A.R.C.; Ruiz, H.A. Enzymatic hydrolysis, kinetic modeling of hemicellulose fraction, and energy efficiency of autohydrolysis pretreatment using agave bagasse. Bioenergy Res. 2023, 16, 75–87. [Google Scholar] [CrossRef]
- Karimi, K.; Taherzadeh, M.J. A critical review of analytical methods in pretreatment of lignocelluloses: Composition, imaging, and crystallinity. Bioresour. Technol. 2016, 200, 1008–1018. [Google Scholar] [CrossRef]
- Sathitsuksanoh, N.; George, A.; Zhang, Y.-H.P. New lignocellulose pretreatments using cellulose solvents: A review. J. Chem. Technol. Biotechnol. 2013, 88, 169–180. [Google Scholar] [CrossRef]
- Yoo, H.Y.; Lee, J.H.; Kim, D.S.; Lee, J.H.; Lee, S.K.; Lee, S.J.; Park, C.; Kim, S.W. Enhancement of glucose yield from canola agricultural residue by alkali pretreatment based on multi-regression models. J. Ind. Eng. Chem. 2017, 51, 303–311. [Google Scholar] [CrossRef]
- Arisht, S.N.; Abdul, P.M.; Liu, C.-M.; Lin, S.-K.; Maaroff, R.M.; Wu, S.-Y.; Jahim, J.M. Biotoxicity assessment and lignocellulosic structural changes of phosphoric acid pre-treated young coconut husk hydrolysate for biohydrogen production. Int. J. Hydrog. Energy. 2019, 44, 5830–5843. [Google Scholar] [CrossRef]
- Lee, J.; Kim, S.; Lee, K.H.; Lee, S.K.; Chun, Y.; Kim, S.W.; Park, C.; Yoo, H.Y. Improvement of bioethanol production from waste chestnut shells via evaluation of mass balance-based pretreatment and glucose recovery process. Environ. Technol. Innov. 2022, 28, 102955. [Google Scholar] [CrossRef]
- Nandal, P.; Sharma, S.; Arora, A. Bioprospecting non-conventional yeasts for ethanol production from rice straw hydrolysate and their inhibitor tolerance. Renew. Energy. 2020, 147, 1694–1703. [Google Scholar] [CrossRef]


| Composition (% dw) | T. latifolia (%) |
|---|---|
| Glucan | 32.9 ± 0.5 |
| Xylan | 23.8 ± 0.2 |
| Arabinan | 5.7 ± 0.0 |
| AIL* | 22.2 ± 0.5 |
| ASL** | 5.9 ± 0.1 |
| Ash | 9.4 ± 0.1 |
| Extractive | 4.3 ± 0.2 |
| Composition (% dw) |
Raw material |
H3PO4 concentration (% v/v) | |||
|---|---|---|---|---|---|
| 70 | 75 | 80 | 85 | ||
| Glucan | 32.9 ± 0.5e | 50.4 ± 0.3d | 54.9 ± 0.2c | 59.7 ± 0.5b | 64.4 ± 0.9a |
| Xylan | 23.8 ± 0.2a | 12.4 ± 0.1b | 10.2 ± 0.2c | 9.0 ± 0.0d | 8.5 ± 0.1e |
| Arabinan | 5.7 ± 0.0a | 3.4 ± 0.0b | 3.3 ± 0.0c | 3.2 ± 0.0d | n.d. |
| AIL* | 22.2 ± 0.5a | 18.0 ± 0.6b | 13.6 ± 0.2c | 12.0 ± 0.6d | 9.2 ± 0.5e |
| ASL** | 5.9 ± 0.1a | 3.7 ± 0.0b | 3.6 ± 0.1b | 3.3 ± 0.1c | 3.0 ± 0.1d |
| Total lignin | 28.2 ± 0.4a | 21.7 ± 0.6b | 17.1 ± 0.1c | 15.3 ± 0.7d | 12.2 ± 0.5e |
| CrI (%) | Concentration of phosphoric acid | ||||
| Untreated | 70% | 75% | 80% | 85% | |
| 54.1 | 57.7 | 61.2 | 59.1 | 49.1 | |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).




