Submitted:
13 March 2024
Posted:
14 March 2024
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
2.1. Viruses and Cell Cultures
2.2. Animals
2.3. In Vitro Co-Infection
2.4. In Vivo Co-Infection
2.5. RNA Extraction and qPCR
2.6. Statistical Analysis
2.7. Biosafety
3. Results
3.1. Co-Infection SARS-CoV-2 and Enteroviruses in Vero E6 Cells
| (A) | ||||||||||||||||||||||||||||||||||
| Mono-infection | Co-infection | |||||||||||||||||||||||||||||||||
| SARS-CoV-2 | LEV-8 | SARS-CoV-2 + LEV-8 | ||||||||||||||||||||||||||||||||
| Time post-infection |
Viral titers, lgТCID50/mL | Viral RNA load according to dPCR, lg genome copies/mL | Viral titers, lgTCID50/mL |
Viral RNA load according to dPCR, lg genome copies/mL | Viral titers, SARS-CoV-2/LEV-8, lgTCID50/mL |
Viral RNA load according to dPCR, SARS-CoV-2/LEV-8, lg genome copies/mL |
||||||||||||||||||||||||||||
| 24 h | 4.8±0.3 | 6.6±0.4 | 5.2±0.3 | 7.0±0.3 | 2.3±0.3/3.5±0.3 | 4.6±0.3/5.6±0.3 | ||||||||||||||||||||||||||||
| 48 h | 6.9±0.4 | 8.4±0.3 | 7.3±0.4 | 8.6±0.4 | 3.8±0.4/5.8±0.3 | 5.5±0.3/7.3±0.3 | ||||||||||||||||||||||||||||
| SARS-CoV-2 | EV-A71 | SARS-CoV-2 + EV-A71 | ||||||||||||||||||||||||||||||||
| 24 h | 5.2±0.3 | 7.0±0.4 | 5.6±0.3 | 7.4±0.3 | 2.2±0.3/3.3±0.3 | 4.3±0.3/5.4±0.3 | ||||||||||||||||||||||||||||
| 48 h | 7.3±0.4 | 8.5±0.3 | 7.5±0.4 | 9.0±0.4 | 3.5±0.4/5.4±0.3 | 5.3±0.3/7.1±0.3 | ||||||||||||||||||||||||||||
| (B) | ||||||||||||||||||||||||||||||||||
| Time post-infection with SARS-CoV-2 |
Viral titers (SARS-СoV-2/LEV-8), lgТCID50/mL | Viral RNA load according to dPCR (SARS-СoV-2/LEV-8) lg genome copies/mL | Viral titers, lgТCID50/mL |
Viral RNA load by dPCR, lg genome copies/mL | ||||||||||||||||||||||||||||||
| LEV-8 pre-infection -24h- SARS-CoV-2 | Mock pre-infection -24h- SARS-CoV-2 | |||||||||||||||||||||||||||||||||
| 24 h | 2.7±0.3/6.9±0.4 | 4.4±0.3/8.2±0.4 | 5.1±0.4 | 7.7±0.3 | ||||||||||||||||||||||||||||||
| 48 h | 3.2±0.3/7.3±0.4 | 4.8±0.4/9.1±0.3 | 7.2±0.4 | 9.3±0.4 | ||||||||||||||||||||||||||||||
| SARS-CoV-2 pre-infection -24h- LEV-8 | Mock pre-infection -24h- LEV-8 | |||||||||||||||||||||||||||||||||
| 48 h | 6.3±0.3/5.9±0.4 | 8.0±0.4/7.8±0.3 | 5.3±0.3 | 7.1±0.3 | ||||||||||||||||||||||||||||||
| 72 h | 5.0±0.4/5.6±0.4 | 6.9±0.3/7.4±0.4 | 7.5±0.4 | 9.2±0.3 | ||||||||||||||||||||||||||||||
| Time post-infection with SARS-CoV-2 |
Viral titers (SARS-СoV-2/EV-A71), lgТCID50/mL | Viral RNA load according to dPCR (SARS-СoV-2/EV-A71) lg genome copies/mL | Viral titers, lgТCID50/mL |
Viral RNA load by dPCR, lg genome copies/mL | ||||||||||||||||||||||||||||||
| EV-A71 pre-infection -24h- SARS-CoV-2 | Mock pre-infection -24h- SARS-CoV-2 | |||||||||||||||||||||||||||||||||
| 24 h | 2.9±0.3/4.9±0.2 | 4.7±0.3/6.5±0.2 | 5.2±0.3 | 7.3±0.3 | ||||||||||||||||||||||||||||||
| 48 h | 3.1±0.3/6.5±0.4 | 5.0±0.4/8.2±0.3 | 7.1±0.4 | 8.7±0.4 | ||||||||||||||||||||||||||||||
| SARS-CoV-2 pre-infection -24h- EV-A71 | Mock pre-infection -24h- EV-A71 | |||||||||||||||||||||||||||||||||
| 48 h | 5.8±0.4/5.3±0.4 | 7.6±0.3/7.1±0.3 | 6.8±0.3 | 8.3±0.4 | ||||||||||||||||||||||||||||||
| 72 h | 4.7±0.3/5.1±0.3 | 7.0±0.3/7.2±0.4 | 6.5±0.4 | 8.0±0.3 | ||||||||||||||||||||||||||||||
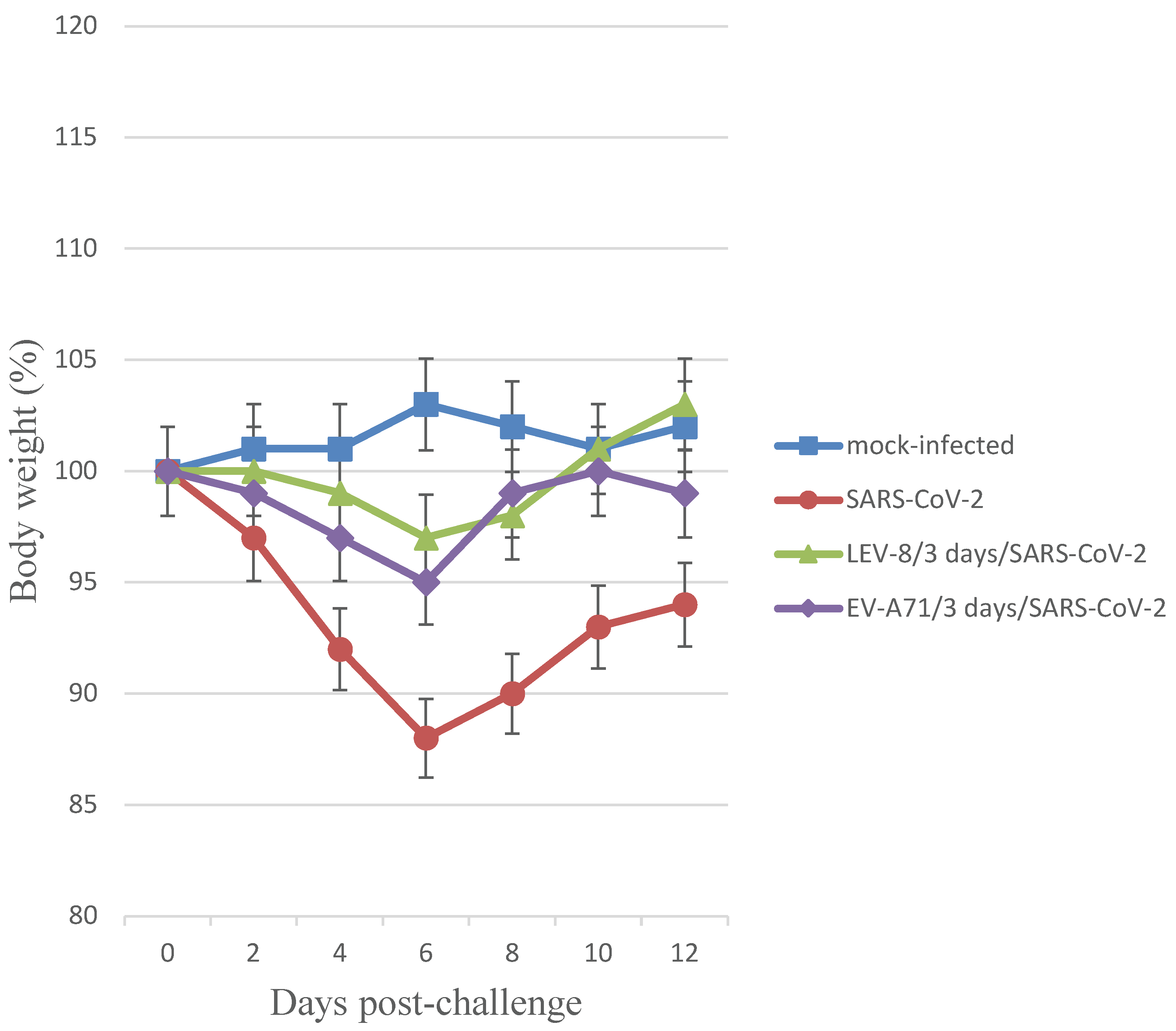
3.2. Co-Infection with SARS-CoV-2 and Enteroviruses in Animals
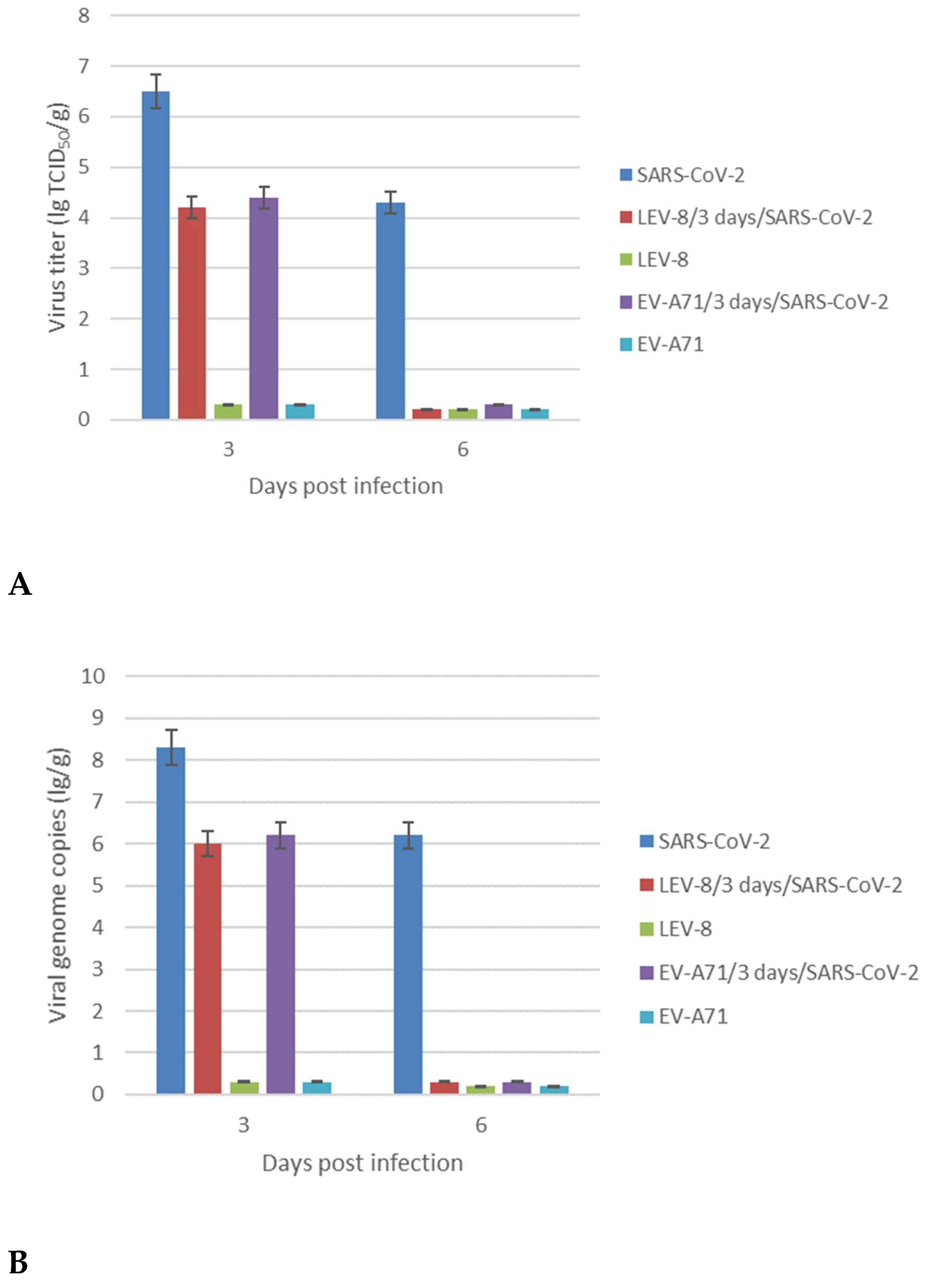
3.3. Replication of SARS-CoV-2 and Enteroviruses in the Lungs
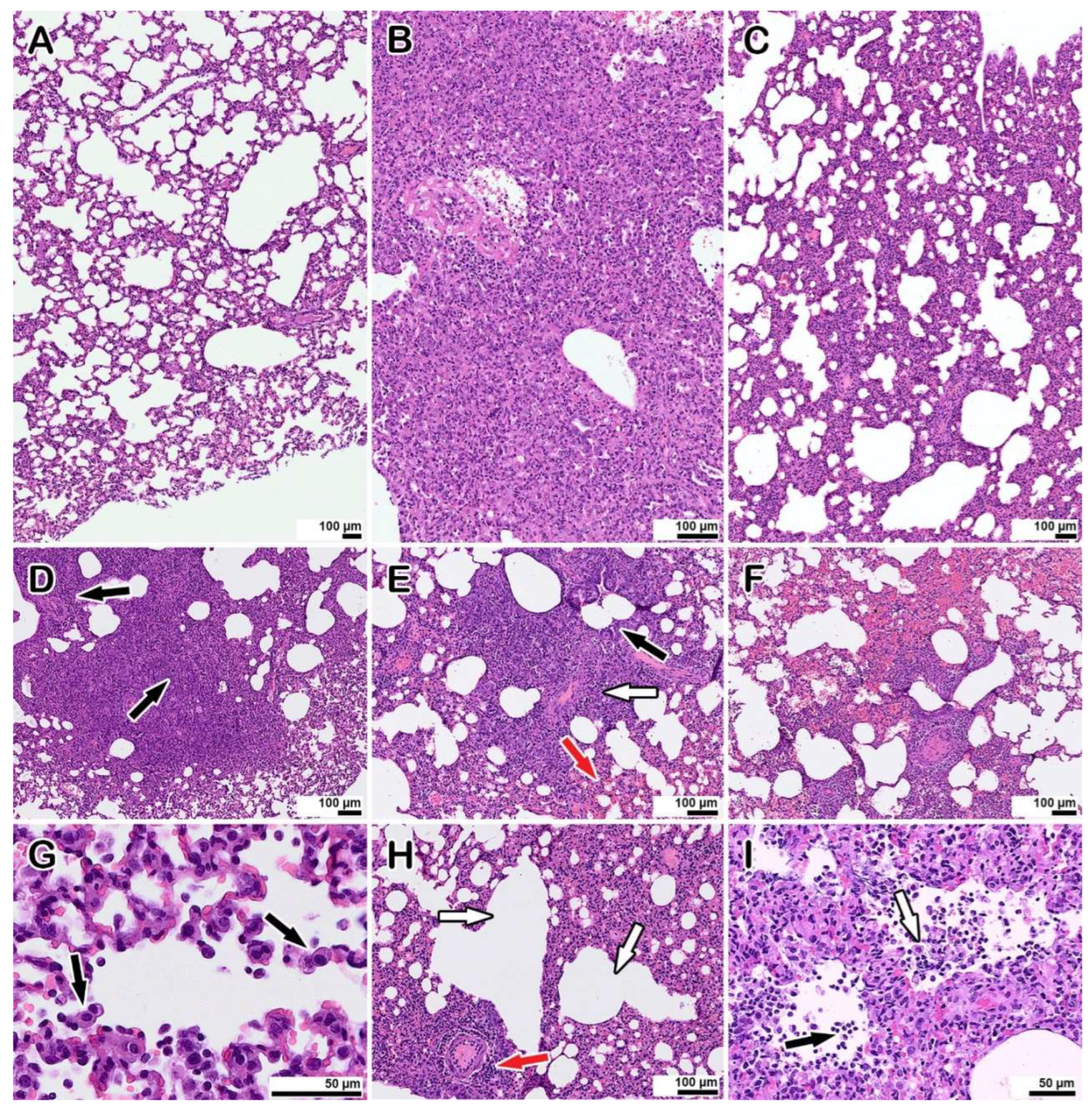
3.4. Morphological Examination of Co-Infected Animals
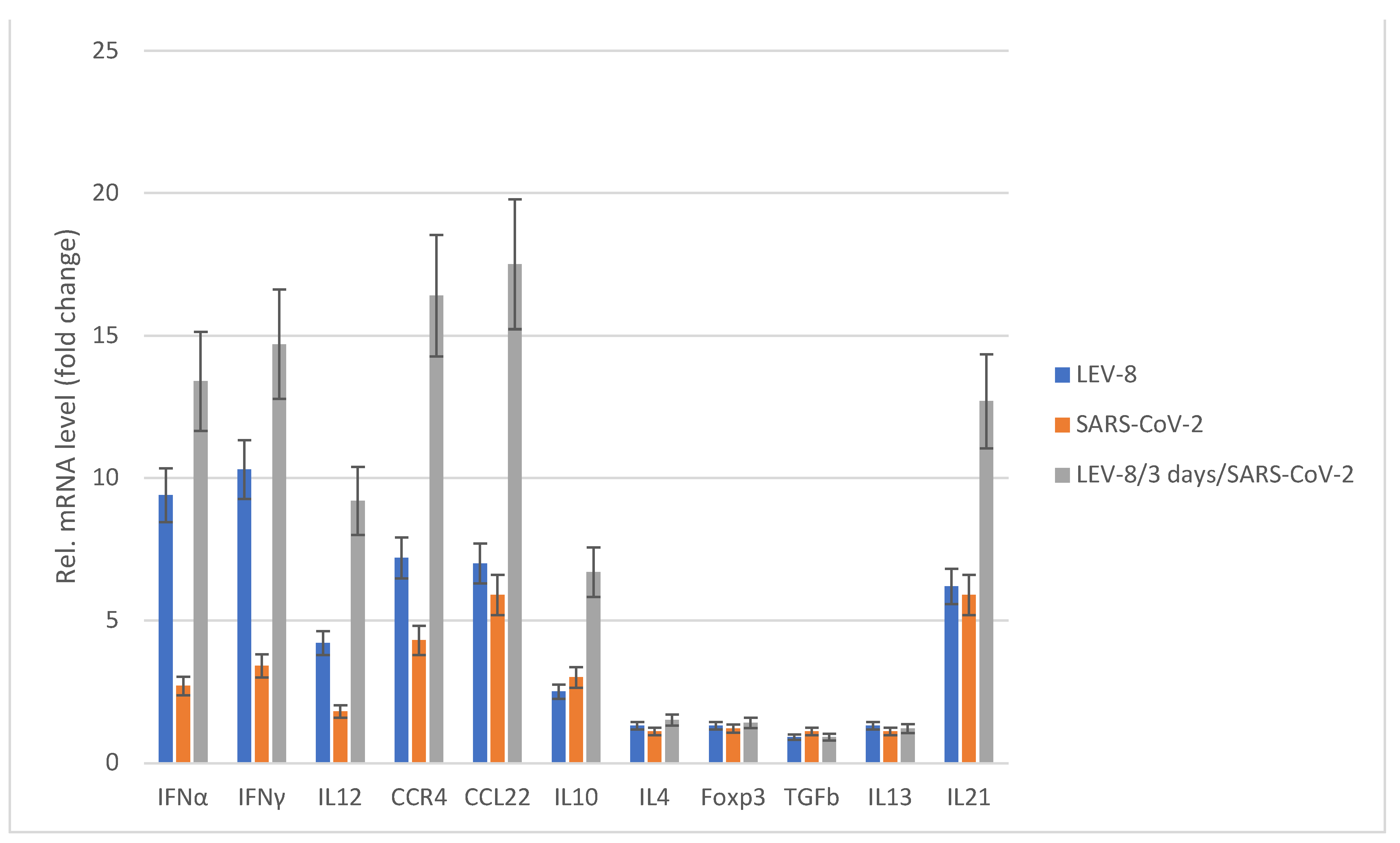
3.5. Chemokine/Cytokine Responses in Co-Infected Animals
4. Discussion
5. Conclusion
Supplementary Materials
Author statements
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Wu, F.; Zhao, S.; Yu, B.; Chen, Y.M.; Wang, W.; Song, Z.G.; Hu, Y.; Tao, Z.; Tian, J.; Pei, Y.; Yuan, M.L.; Zhang, Y.; Dai, F.; Liu, Y.; Wang, Q.; Zheng, J.; Xu, L.; Holmes, E.; Zhang, Y.Z. A new coronavirus associated with human respiratory disease in China. Nature 2020, 579, 265–269. [Google Scholar] [CrossRef]
- Zhu, N.; Zhang, D.; Wang, W.; Li, X.; Yang, B.; Song, J.; Zhao, X.; Huang, B.; Shi, W.; Lu, R.; Niu, P.; Zhan, F.; Ma, X.; Wang, D.; Xu, W.; Wu, G.; Gao, G.; Tan, W. A novel coronavirus from patients with pneumonia in China, 2019. N. Engl. J. Med. 2020, 382, 727–733. [Google Scholar] [CrossRef] [PubMed]
- Wang, C.; Horby, P.W.; Hayden, F.G.; Gao, G.F. A novel coronavirus outbreak of global health concern. Lancet 2020, 395, 470–473. [Google Scholar] [CrossRef] [PubMed]
- Kim, D.; Quinn, J.; Pinsky, B.; Shah, N.H.; Brown, I. Rates of co-infection between SARS-CoV-2 and other respiratory pathogens. J.A.M.A. 2020, 323, 2085–2086. [Google Scholar] [CrossRef] [PubMed]
- Lansbury, L.; Lim, B.; Baskaran, V.; Lim, W. Co-infections in people with COVID-19: a systematic review and meta-analysis. J. Infect. 2020, 81, 266–275. [Google Scholar] [CrossRef] [PubMed]
- Glass, E.; Hoang, V.; Boschi, C.; Ninove, L.; Zandotti, C.; Boutin, A.; Bremond, V.; Dubourg, G.; Ranque, S.; Lagier, J.; Million, M.; Fournier, P.; Drancourt, M.; Gautret, P.; Colson, F. Incidence and outcome of coinfections with SARS-CoV-2 and rhinovirus. Viruses 2021, 13, 2528. [Google Scholar] [CrossRef] [PubMed]
- Maltezou, H.; Paranikolopoulou, A.; Vassiliu, S.; Theodoridou, K.; Nikolopolou, G.; Sipsas, N. COVID-19 and respiratory virus co-Infections: a systematic review of the literature. Viruses 2023, 15, 865. [Google Scholar] [CrossRef]
- Goldberg, E.; Hasegawa, K.; Lawrence, A.; Kline, J.; Camargo, C. Viral coinfection is associated with improved outcomes in emergency department patients with SARS-CoV-2. West J Emerg Med. 2021, 22, 1262–1269. [Google Scholar] [CrossRef]
- Chan, J.; Zhang, A.; Yuan, S.; Poon, V.; Chan, C.; Lee, A.; Chan, W.; Fan, Z.; Tsoi, H.; Wen, L.; Liang, R.; Cao, J.; Chen, Y.; Tang, K.; Luo, C.; Cai, J.; Kok, K.; Chu, H.; Chan, K.; Sridhar, S.; Chen, Z.; Chen, H.; To, K.; Yuen, K. Simulation of the clinical and pathological manifestations of coronavirus disease 2019 (COVID-19) in a golden Syrian hamster model: implications for disease pathogenesis and transmissibility. Clin. Infect. Dis. 2020, 71, 2428–2446. [Google Scholar] [CrossRef]
- Imai, M.; Iwatsuki-Horimoto, K.; Hatta, M.; Loeber, S.; Halfmann, P.J.; Nakajima, N.; Watanabe, T.; Ujie, M.; Takahashi, M. Syrian hamsters as a small animal model for SARS-CoV-2 infection and countermeasure development. Proc Natl Acad Sci USA 2020, 117, 16587–16595. [Google Scholar] [CrossRef]
- Phyu, W.; Ong, K.; Won, K. A consistent orally-infected hamster model for enterovirus A71 encephalomyelitis demonstrates squamous lesions in the paws, skin and oral cavity reminiscent of Hand-Foot-and-Mouth Disease. PLoS One 2016. [Google Scholar] [CrossRef] [PubMed]
- Chumakov, M.P.; Voroshilova, M.K.; Antsupova, A.S.; Boiko, V.M.; Blinova, M.I.; Priimiagi, L.S.; Rodin, V.I.; Seibil, V.B.; Siniak, K.M.; Smorodintsev, A.A. Live enteroviral vaccines for the emergency nonspecific prevention of mass respiratory diseases during fall-winter epidemics of influenza and acute respiratory diseases. Zh Mikrobiol Epidemiol Immunobiol 1992, 37–40. [Google Scholar]
- Fujimoto, T. Hand-Foot-and-Mouth Disease, Aseptic Meningitis, and Encephalitis Caused by Enterovirus. Brain and nerve 2018, 70, 121–131. [Google Scholar] [CrossRef] [PubMed]
- Svyatchenko, V.; Nikonov, S.; Mayorov, A.; Gelfond, M.; Loktev, V. Antiviral photodynamic therapy: Inactivation and inhibition of SARS-CoV-2 in vitro using methylene blue and Radachlorin. Photodiagnosis Photodyn. Ther. 2021, 33, 102–112. [Google Scholar] [CrossRef] [PubMed]
- Toth, K.; Spencer, J.; Dhar, D.; Sagartz, J.; Buller, R.; Painter, G.; Wold, W.S. Hexadecyloxypropyl-cidofovir, CMX001, prevents adenovirus induced mortality in a permissive, immunosuppressed animal model. Proc. Natl. Acad. Sci. USA 2008, 105, 7293–7297. [Google Scholar] [CrossRef] [PubMed]
- Ryzhikov, A.B.; Ryzhikov, E.A.; Bogryantseva, M.P.; Danilenko, E.D.; Imatdinov, I.R.; Nechaeva, E.A.; Pyankov, O.; Pyankova, O.G.; Susloparov, I.; Taranov, O.; Gudymo, A.; Danilchenko, N.; Sleptsova, E.; Bodnev, S.; Onkhonova, G.; Petrov, V.; Moiseeva, A.; Torzhkova, P.; Pyankov, S.; Tregubchak, T.; Antonets, D.; Gavrilova, E.; Maksyutov, R. Immunogenicity and protectivity of the peptide vaccine against SARS-CoV-2. Annals of the Russian Academy of Medical Sciences 2021, 76, 5–19. [Google Scholar] [CrossRef]
- Sanders, R.; Mason, D.J.; Foy, C.A.; Huggett, J.F. Evaluation of digital PCR for absolute RNA quantification. PLoS One 2013, 8, e75296. [Google Scholar] [CrossRef]
- Chan, J.F.; Yuan, S.; Zhang, A.; Poon, V.K.; Chan, C.C.; Lee, A.C.; Fan, Z.; Li, C.; Liang, R.; Cao, J. Surgical mask partition reduces the risk of noncontact transmission in a Golden Syrian hamster model for coronavirus disease 2019 (COVID-19). Clin. Infect. Dis. 2020, 71, 2139–2149. [Google Scholar] [CrossRef]
- Hashemi, S.A.; Safamanesh, S.; Ghasemzadeh-Moghaddam, H.; Ghafouri, M.; Azimian, A. High prevalence of SARS-CoV-2 and influenza A virus (H1N1) coinfection in dead patients in Northeastern Iran. J. Med. Virol. 2020, 93, 1008–1012. [Google Scholar] [CrossRef]
- Stowe, J.; Tessier, E.; Zhao, H.; Guy, R.; Muller-Pebody, B.; Zambon, M.; Andrews, N.; Ramsay, M.; Lopez Bernal, J. Interactions between SARS-CoV-2 and Influenza and the impact of coinfection on disease severity: A test negative design. Int. J. Epidemiol. 2021, 50, 1124–1133. [Google Scholar] [CrossRef]
- Kumar, N.; Sharma, S.; Barua, S.; Tripathi, B.; Rouse, B. Virological and Immunological Outcomes of Coinfections. Clin. Microbiol. Rev. 2018, 31, e00111–17. [Google Scholar] [CrossRef] [PubMed]
- Bai, L.; Zhao, Y.; Dong, J.; Liang, S.; Guo, M.; Liu, X.; Wang, X.; Huang, Z.; Sun, X.; Zhang, Z.; Dong, L.; Liu, Q.; Zheng, Y.; Niu, D.; Xiang, M.; Song, K.; Ye, J.; Zheng, W.; Tang, Z.; Tang, M.; Zhou, Y.; Shen, C.; Dai, M.; Zhou, L.; Chen, Y.; Yan, H.; Lan, K.; Xu, K. Coinfection with influenza A virus enhances SARS-CoV-2 infectivity. Cell Res. 2021, 4, 395–403. [Google Scholar] [CrossRef] [PubMed]
- Svyatchenko, V.A.; Ternovoi, V.A.; Lutkovskiy, R.Y.; Protopopova, E.V.; Gudymo, A.S.; Danilchenko, N.V.; Susloparov, I.M.; Kolosova, N.P.; Ryzhikov, A.B.; Taranov, O.S.; Omigov, V.V.; Gavrilova, E.V.; Agafonov, A.P.; Maksyutov, R.A.; Loktev, V.B. Human Adenovirus and Influenza A Virus Exacerbate SARS-CoV-2 Infection in Animal Models. Microorganisms 2023, 11, 180. [Google Scholar] [CrossRef]
- Essaidi-Laziosi, M.; Alvarez, C.; Puhach, O.; Sattonnet-Roche, P.; Torriani, G.; Tapparel, C.; Kaiser, L.; Eckerle, I. Sequential infections with rhinovirus and influenza modulate the replicative capacity of SARS-CoV-2 in the upper respiratory tract. Emerg. Microbes. Infect. 2022, 11, 412–423. [Google Scholar] [CrossRef]
- Fage, C.; Henaut, M.; Carbonneau, J.; Piret, J.; Boivin, G. Influenza A(H1N1)pdm09 virus but not respiratory syncytial virus interferes with SARS-CoV-2 replication during sequential infections in human nasal epithelial cells. Viruses 2022, 14, 395. [Google Scholar] [CrossRef] [PubMed]
- Royston, L.; Tapparel, C. Rhinoviruses and respiratory enteroviruses: not as simple as ABC. Viruses 2016, 8, 16. [Google Scholar] [CrossRef] [PubMed]
- Kovesdi, I.; Sandig, V.; Slavin, S.; Renz, W.; Ranst, M.; Chumakov, P.; Bakacs, T. The clinically validated viral superinfection therapy (SIT) platform technology could cure early cases of COVID-19 disease. 2020, 2020020147. [Google Scholar] [CrossRef]
- Chumakov, K.; Benn, C.S.; Aaby, P.; Kottilil, S.; Gallo, R. Can existing live vaccines prevent COVID-19? Science 2020, 368, 1187–1188. [Google Scholar] [CrossRef]
- Kashir, J.; AlKattan, K.; Yaqinuddin, A. COVID-19: cross-immunity of viral epitopes may influence severity of infection and immune response. Sig Transduct Target Ther. 2021, 6, 102. [Google Scholar] [CrossRef]
- Shrock, E.; Fujimura, E.; Kula, T.; Timms, R.; Lee, I.; Leng, Y.; Robinson, M.; Sie, B.; Li, M. Viral epitope profiling of COVID-19 patients reveals cross-reactivity and correlates of severity. Science 2020, 370, eabd4250. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




