Submitted:
08 March 2024
Posted:
12 March 2024
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
2.1. Sampling, Isolation and Maintenance of Halophilic Bacteria
2.2. Screening of H. gomseomemsis for PHA Production
2.3. Confocal Microscopy Analysis
2.4. Identification of Halophilic Isolates
2.5. Polymer Extraction
2.6. Characterization of Obtained Polyhydroxyalkanoates
3. Results and Discussion
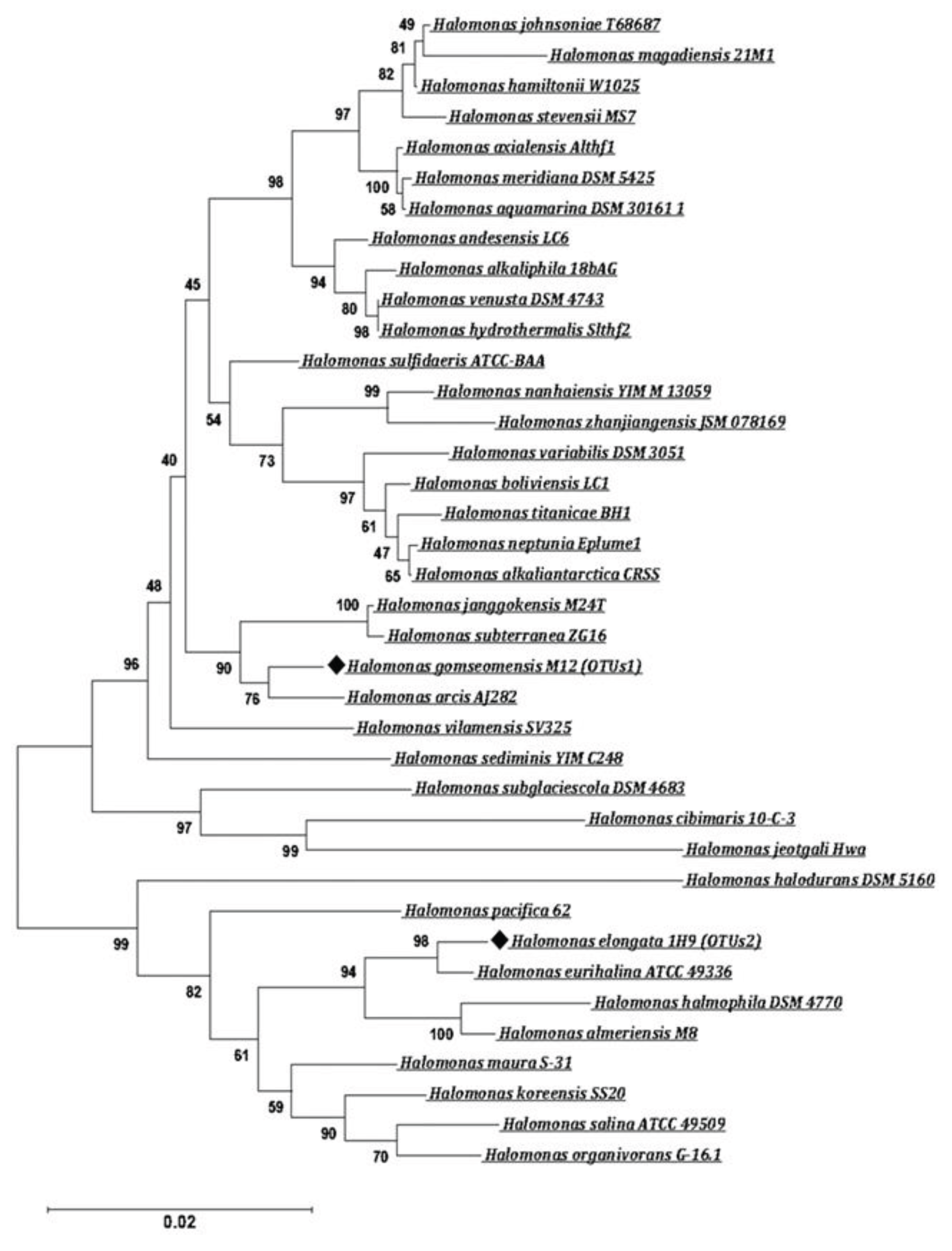
3.1. Identification of Halophile Microorganism through 16S rRNA
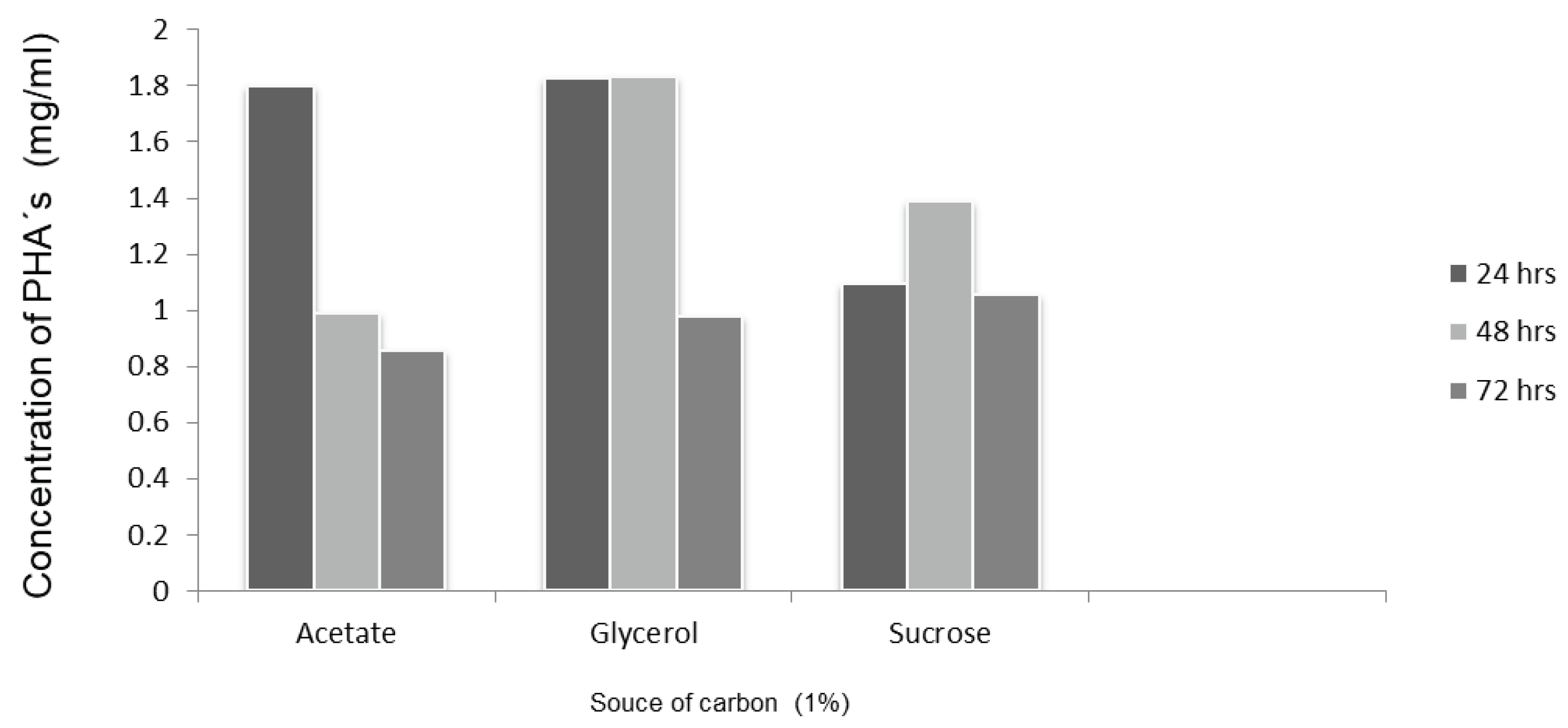
3.2. Effect of Carbon Sources in Intracellular PHA Accumulation
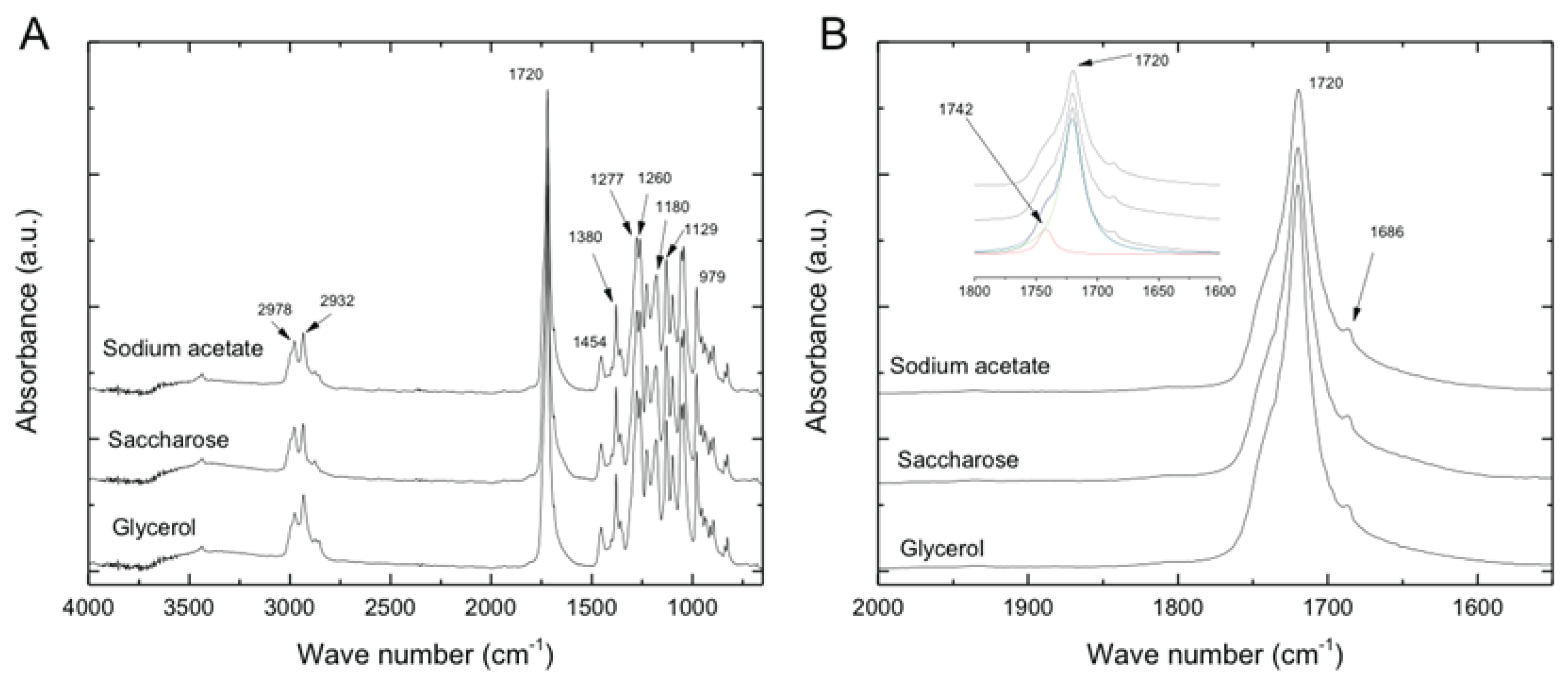
3.3. Fourier Transform Infrared Spectroscopy
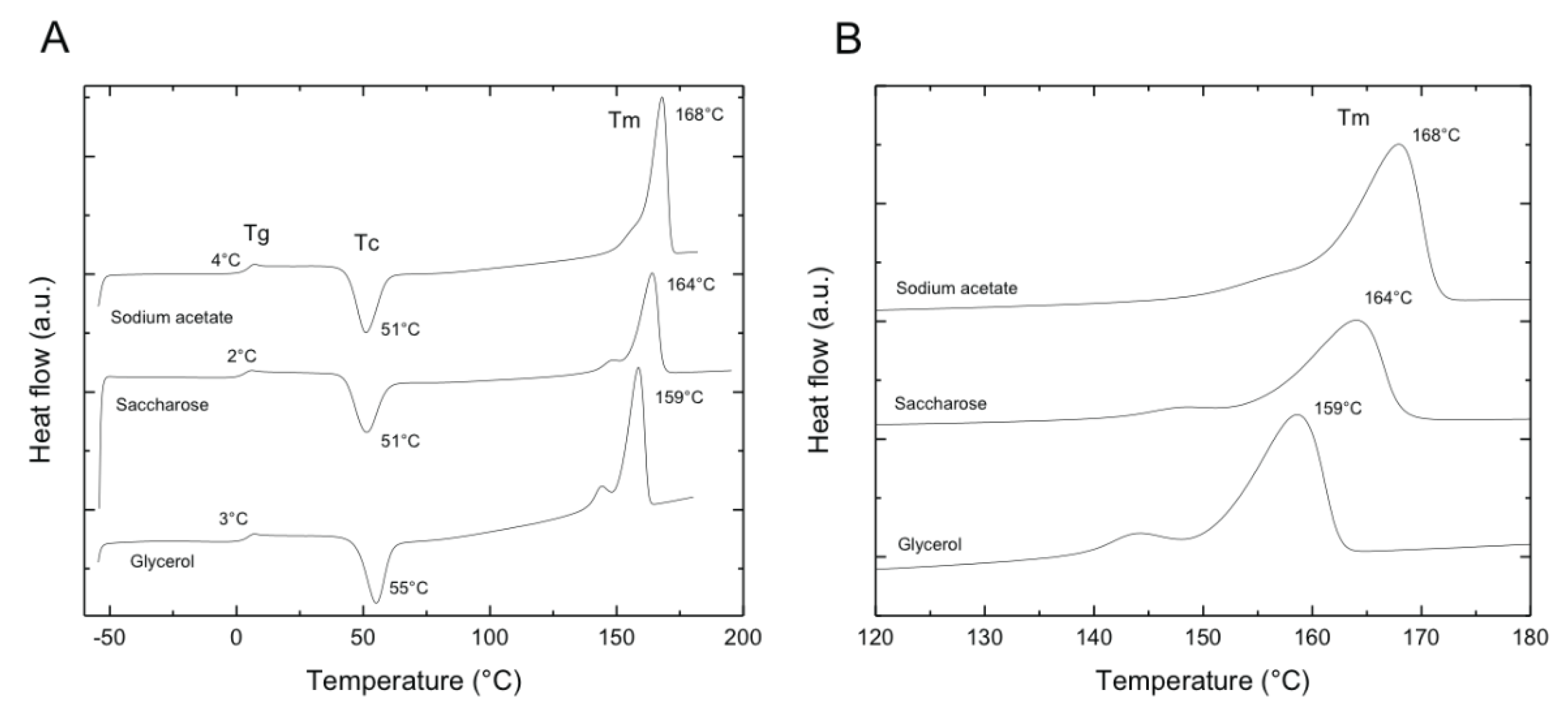
3.4. Diferential Scanning Calorimetry
3.5. Thermogravimetric Analysis
4. Conclusions
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
References
- Hye-Young, S.; Hyeoncheol, F.; So, Y.; Sang, Y.; Up, L. Structural Insights into Polyhydroxyalkanoates Biosynthesis. Trends Biochem Sci. 2018, 43 (10), 790–805. [Google Scholar] [CrossRef]
- de Souza Machado, A.A.; Lau, C.W.; Till, J.; Kloas, W.; Lehmann, A.; Becker, R.; Rillig, M.C. Impacts of Microplastics on the Soil Biophysical Environment. GlobalChange Biol. 2018, 24, 1405–1405. [Google Scholar] [CrossRef]
- Zhang, X.; Rongcong, L.; Zhen, W., Yuan; Guo-Qiang, Ch. Application of (R)-3-hydroxyalkanoate methyl esters derived from microbial polyhydroxyalkanoates as novel biofuels. Biomacromolecules 2009, 10(4), 707-11. [Google Scholar] [CrossRef]
- Anbreen, A.; Mohammad, Z.; Khalid, M.; Aqdas, N.; Muhammad, N.; Shazia, T. Microbial production of polyhydroxyalkanoates (PHAs) and its copolymers: A review of recent advancements. Int J Biol Macromol 2016, 89, 161-74. [Google Scholar] [CrossRef]
- Laycock, B.; Halley, P.; Pratt, S.; Werker, A.; Lant, P. The chemomechanical properties of microbial polyhydroxyalkanoates. Prog. Polym. Sci. 2014, 39, 397–442. [Google Scholar] [CrossRef]
- Zulfiqar, A.; Muhammad, R.; Muhammad, I.; Ibrahim, M. Recent developments in bioreactor scale production of bacterial polyhydroxyalkanoates. Bioprocess and Biosystems Engineering 2019, 42, 901–919. [Google Scholar] [CrossRef]
- Zulfiqar, A.; Shahina, R.; Ibrahim, M. Polyhydroxyalkanoates: Properties and chemical modification approaches for their functionalization. Biotechnol Prog. 2018, 34(1), 29-4. [Google Scholar] [CrossRef]
- Chethana, M.; Kona, M.; Urvashi, S.; Vimal, K. Production of Polyhydroxyalkanoates and Its Potential Applications. Advances in Sustainable Polymers 2019, 131–164. [Google Scholar] [CrossRef]
- Subhasree, R.; Vipin, Ch. Biomedical Applications of Polyhydroxyalkanoates. Indian J Microbiol. 2017, 57(3), 261–269. [Google Scholar] [CrossRef]
- Tiago, M.; Amaro, D.; Giuseppe, C.; Lucilla, I. Prospects for the Use of Whey for Polyhydroxyalkanoate (PHA). Production. Frontiers in Microbiology 2019, 10, 1.12. [Google Scholar] [CrossRef]
- Bhatia, S. K.; Gurav, R.; Choi, T.-R.; Jung, H.-R.; Yang, S.-Y.; Moon, Y.-M. et al.; et al. Bioconversion of plant biomass hydrolysare into bioplastic (polyhydroxyalkanoates) using Ralstonia eutropha 5119. Bioresour. Technol. 2019, 271, 306–315. [Google Scholar] [CrossRef]
- Bhatia, S. K.; Kim, J.-H.; Kim, M.-S.; Kim, J.; Wong, J. W.; Hong, Y. G. et al.; et al. Production of (3-hydroxybutyrate-co-3-hydroxyhexanoate) copolymer from coffee waste oil using engineered Ralstonia eutropha. Bioprocess Biosyst. Eng. 2018, 41, 229–235. [Google Scholar] [CrossRef] [PubMed]
- Cervantes-Uc, J.M.; Catzin, J.; Vargas, I.; Herrera-Kao, W.; Miguel, F.; Rincón-Arriaga, S.; Lizama-Uc, G. Biosynthesis and characterization of polyhydroxyalkanoates produced by an extreme halophilic bacterium, Halomonas nitroreducens, isolated from hypersaline ponds. J Appl Microbiol 2014, 117, 1056–1065. [Google Scholar] [CrossRef] [PubMed]
- Gunaratne, L.; Shanks, R.A.; Amarasinghe, G. Thermal history effects on crystallisation and melting of poly(3-hydroxybutyrate). Thermochimica Acta 2004, 423, 127–135. [Google Scholar] [CrossRef]
- Toh, P.; Jau, M.; Saw-Peng, Y.; Abed, R.; Sudesh, K. Comparison Of Polyhydroxyalkanoates Biosynthesis, Mobilization And The Effects On Cellular Morphology In Spirulina Platensis And Synechocystis Sp. Uniwg. 2008. Journal of Bioscience 19(2), 21–38. https://www.researchgate.net/publication/237628395_Comparison_of_polyhydroxyalkonates_biosynthesis_mobilization_and_the_effects_of_cellular_morphology_in_Spirulina_platensis_and_Synechocystis_sp_UNIWG_J_Biosci.
- Thomson, N.; Roy, I.; Summers, D.; Sivaniah, E. In vitro production of polyhydroxyalkanoates. 2009. Achievements and applications. [CrossRef]
- Ikejima, T.; Yagi, K.; Inoue, Y. Thermal properties and crystallization behavior of poly (3hydroxybutyric acid) in blends with chitin and chitosan. Macromol. Chem. Phys. 1999, 200, 413–421. [Google Scholar] [CrossRef]
- Xu, J.; Guo, B-H.; Yang, R.; Wu, Q.; Chen, G-Q.; Zhang, Z-M. In situ FTIR study on melting and crystallization of polyhydroxyalkanoates. Polymer 2002, 43, 6893–6899. [Google Scholar] [CrossRef]
- Padermshoke, A.; Katsumoto, Y.; Sato, H.; Ekgasit, S.; Noda, I.; Ozaki, Y. Surface melting and crystallization behavior of polyhydroxyalkanoates studied by attenuated total reflection infrared spectroscopy. Polymer https://www.academia.edu/18874033/Surface_melting_and_crystallization_behavior_of_polyhydroxyalkanoates_studied_by_attenuated_total_reflection_infrared_spectroscopy. 2004, 45, 6547–6554. [Google Scholar] [CrossRef]
- Sato, H.; Dybal, J.; Murakami, R.; Noda, I.; Ozaki, Y. Infrared and Raman spectroscopy and quantum chemistry calculation studies of C–H/O hydrogen bondings and thermal behavior of biodegradable polyhydroxyalkanoate. Journal of Molecular Structure 2005, 744–747, 35–46. [Google Scholar] [CrossRef]
- Xiao, N.; Jiao, N. Formation of Polyhydroxyalkanoate in Aerobic Anoxygenic Phototrophic Bacteria and Its Relationship to Carbon Source and Light Availability. Applied and Environmental Microbiology 2011, 77, 7445–6450. [Google Scholar] [CrossRef]
- Shah, K.R. FTIR analysis of polyhydroxyalkanoates by novel Bacillus sp. AS 3-2 from soil of Kadi region, North Gujarat, India. J Biochem Tech. https://jbiochemtech.com/storage/models/article/jhGf8ERXqctMxostMWavZcj7BfBV9PEpcOVvw63gEXqsO8PefA1i3PPYJimo/ftir-analysis-of-polyhydroxyalkanoates-by-a-locally-isolated-novel-bacillus-sp-as-3-2-from-soil-of.pdf. 2012, 380–383. [Google Scholar]
- Bera, A.; Dubey, S.; Bhayani, K.; Mondal, D.; Mishra, S.; Ghosh, P.K. Microbial synthesis of polyhydroxyalkanoate using seaweed-derived crude levulinic Acid as co-nutrient. International Journal of Biological Macromolecules 2015, 72, 487–494. [Google Scholar] [CrossRef]
- Tanahashi, N.; Doi, Y. Thermal properties and stereoregularity of poly(3-hydroxybutyrate) prepared from optically active -butyrolactone with a zinc-based catalyst. Macromolecules 1991, 24, 5732–5733. [Google Scholar] [CrossRef]
- Kunioka, M.; Doi, Y. Thermal degradation of microbial copolyesters: poly(3-hydroxybutyrate-co-3-hydroxyvalerate) and poly(3-hydroxybutyrate-co-4-hydroxybutyrate. Macromolecules 1990, 23, 1933–1936. [Google Scholar] [CrossRef]
- Anderson, A.J.; Dawes, E.A. Ocurrence, Metabolism, Metaboic Role, and Industrial Uses of Bacterial Polyhydroxyalkanoates. Microbiological Reviews 1990, 54, 450–427. [Google Scholar] [CrossRef] [PubMed]
- Martino, L.; Cruz, M.V.; Scoma, A.; Freitas, F.; Bertin, L.; Scandola, M.; Reis, M. Recovery of amorphous polyhydroxybutyrate granules from Cupriavidus necator cells grown on used cooking oil. International Journal of Biological Macromolecules 2014, 71, 117–123. [Google Scholar] [CrossRef] [PubMed]
- Zheng, Z.; Bei, F-F.; Tian, H-L.; Chen, G-Q. Effects of crystallization of polyhydroxyalkanoate blend on Surface physicochemical properties and interactions with rabbit articular cartilage chondrocytes. Biomaterials 2005, 26, 3537–3548. [Google Scholar] [CrossRef] [PubMed]
- Corre, Y-M.; Bruzaud, S.; Audic, J-L.; Grohens, Y. Morphology and functional properties of comercial polyhydroxyalkanoates: A comprehensive and comparative study. Polymer Testing https://www.academia. 2012, 31, 226–235. [Google Scholar] [CrossRef]
- Porter, M.; Yu, J. Monitoring the in situ crystallization of native biopolyester granules in Ralstonia eutropha via infrared spectroscopy. Journal of Microbiological Methods 2011, 87, 49–55. [Google Scholar] [CrossRef] [PubMed]
- Padermshoke, A.; Katsumoto, Y.; Sato, H.; Ekgasit, S.; Noda, I.; Ozaki, Y. Surface melting and crystallization behavior of polyhydroxyalkanoates studied by attenuated total reflection infrared spectroscopy. Polymer 2004, 45, 6547–6554. [Google Scholar] [CrossRef]
- Teeka, J., Yuliani; Higuchi, T.; Yamamoto, K. Characterization of polyhydroxyalkanoates (PHAs) biosynthesis by isolated Novosphingobium sp. THA_AIK7 using crude glycerol. J Ind Microbiol Biotechnol 2012, 39, 749–758. [Google Scholar] [CrossRef]
- Xu, J.; Guo, B-H.; Yang, R.; Wu, Q.; Chen, G-Q.; Zhang, Z-M. In situ FTIR study on melting and crystallization of polyhydroxyalkanoates. Polymer 2002, 43, 6893–6899. [Google Scholar] [CrossRef]

| Carbon source | Tg (°C) | Tc (°C) | Tm (°C) | Crystallinity (%) |
|---|---|---|---|---|
| Sodium acetate | 4 | 51 | 168 | 60 |
| Saccharose | 2 | 51 | 164 | 49 |
| Glycerol | 3 | 55 | 159 | 50 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




