Submitted:
08 March 2024
Posted:
11 March 2024
You are already at the latest version
Abstract
Keywords:
Introduction
Method & Results
- A.
- Benefit/risk of a second ATO cycle
- 1-
- Overall clinical responses
- 2-
- Maintained response over time / introduction second line systemic therapy
- 3-
- Safety (SAE/AE ATO-related)
|
Patient study number |
Adverse Event (AE) | Severity | Duration of AE | Number of ATO infusions before AE/SAE | Action Taken with Study Treatment |
| 21 | Hepatotoxicity | Severe | 7 days | 2 | Definitive discontinuation |
| 1 | Hepatotoxicity | Severe | 10 days | 4 | Temporary discontinuation (10 days) |
| 8 | QTc prolongation | Moderate | 2 days | 2 | Temporary discontinuation (1day) |
|
Patient study number |
Adverse Event (AE) | Severity | Number of ATO infusions before AE |
|
13 |
Vomiting | Moderate | 1 |
| Diarrhea | Moderate | 2 | |
| Abdominal pain | Moderate | 3 | |
| Anemia | Moderate | 3 | |
| Diarrhea | Moderate | 10 | |
| Nausea | Moderate | 10 | |
| 6 | Hypokalemia | Mild | 8 |
|
4 |
Thrombopenia | Moderate | 8 |
| Muscular pain | Mild | 9 |
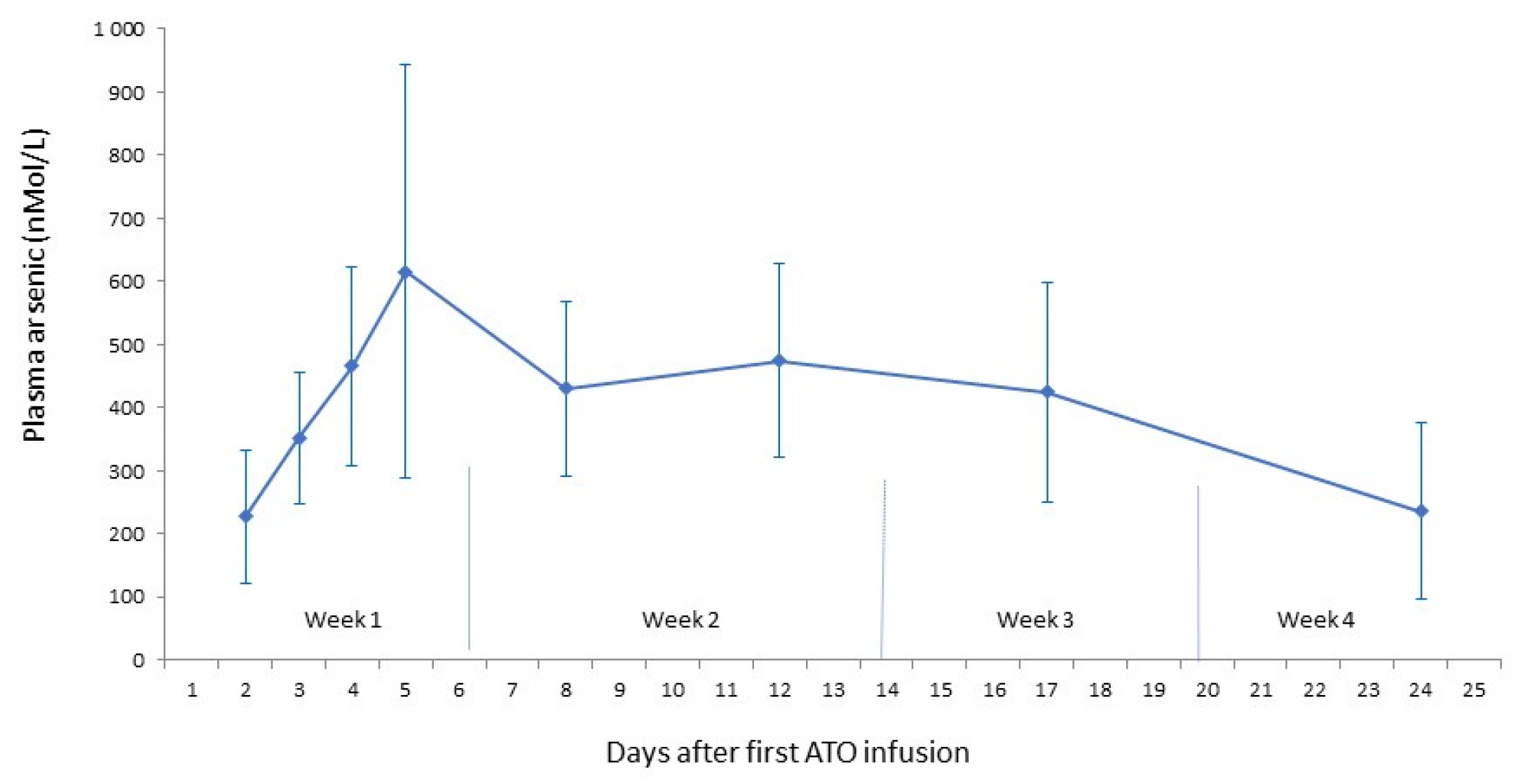
- B.
- Residual arsenic plasma concentrations
Discussion
Conclusion
Acknowledgments
References
- Shen ZX, Chen GQ, Ni JH, Li XS, Xiong SM, Qiu QY, et al. Use of arsenic trioxide (As2O3) in the treatment of acute promyelocytic leukemia (APL): II. Clinical efficacy and pharmacokinetics in relapsed patients. Blood. 1997 May 1;89(9):3354–60.
- Wang XJ, Sun Z, Villeneuve NF, Zhang S, Zhao F, Li Y, et al. Nrf2 enhances resistance of cancer cells to chemotherapeutic drugs, the dark side of Nrf2. Carcinogenesis. 2008 Jun;29(6):1235–43. [CrossRef]
- Lu J, Yu K, Fan S, Liu W, Dong Z, Li J, et al. Influence of AS3MT polymorphisms on arsenic metabolism and liver injury in APL patients treated with arsenic trioxide. Toxicol Appl Pharmacol. 2019 Sep 15;379:114687. [CrossRef]
- Huynh TT, Sultan M, Vidovic D, Dean CA, Cruickshank BM, Lee K, et al. Retinoic acid and arsenic trioxide induce lasting differentiation and demethylation of target genes in APL cells. Sci Rep. 2019 Jul 1;9(1):9414. [CrossRef]
- Mathews V, George B, Lakshmi KM, Viswabandya A, Bajel A, Balasubramanian P, et al. Single-agent arsenic trioxide in the treatment of newly diagnosed acute promyelocytic leukemia: durable remissions with minimal toxicity. Blood. 2006 Apr 1;107(7):2627–32. [CrossRef]
- Ghavamzadeh A, Alimoghaddam K, Ghaffari SH, Rostami S, Jahani M, Hosseini R, et al. Treatment of acute promyelocytic leukemia with arsenic trioxide without ATRA and/or chemotherapy. Annals of Oncology. 2006 Jan;17(1):131–4. [CrossRef]
- Lee SJ, Onstad L, Chow EJ, Shaw BE, Jim HSL, Syrjala KL, et al. Patient-reported outcomes and health status associated with chronic graft-versus-host disease. Haematologica. 2018 Sep;103(9):1535–41. [CrossRef]
- Jagasia M, Arora M, Flowers MED, Chao NJ, McCarthy PL, Cutler CS, et al. Risk factors for acute GVHD and survival after hematopoietic cell transplantation. Blood. 2012 Jan 5;119(1):296–307. [CrossRef]
- Jeanne M, Lallemand-Breitenbach V, Ferhi O, Koken M, Le Bras M, Duffort S, et al. PML/RARA oxidation and arsenic binding initiate the antileukemia response of As2O3. Cancer Cell. 2010 Jul 13;18(1):88–98. [CrossRef]
- Chêne C, Jeljeli MM, Rongvaux-Gaïda D, Thomas M, Rieger F, Batteux F, et al. A Fenton-like cation can improve arsenic trioxide treatment of sclerodermatous chronic Graft-versus-Host Disease in mice. Front Immunol. 2022;13:917739. [CrossRef]
- Chêne C, Rongvaux-Gaïda D, Thomas M, Rieger F, Nicco C, Batteux F. Optimal combination of arsenic trioxide and copper ions to prevent autoimmunity in a murine HOCl-induced model of systemic sclerosis. Front Immunol. 2023 Mar 30;14:1149869. [CrossRef]
- Medsenic. Phase II Study of First Line Treatment of Chronic Graft Versus Host Disease With Arsenic Trioxide [Internet]. clinicaltrials.gov; 2022 Apr [cited 2023 Jan 1]. Report No.: NCT02966301. Available from: https://clinicaltrials.gov/study/NCT02966301.
- Rongvaux-Gaïda D, Dupuis M, Poupon J, Djebrani-Oussedik N, Lemonnier C, Rieger F. High Response Rate and Corticosteroid Sparing with Arsenic Trioxide-Based First-Line Therapy in Chronic Graft-versus-Host Disease after Allogeneic Hematopoietic Stem Cell Transplantation. Transplant Cell Ther. 2022 Jul 10;S2666-6367(22)01469-5. [CrossRef]
- Lee SJ, Wolff D, Kitko C, Koreth J, Inamoto Y, Jagasia M, et al. Measuring Therapeutic Response in Chronic Graft-versus-Host Disease. National Institutes of Health Consensus Development Project on Criteria for Clinical Trials in Chronic Graft-versus-Host Disease: IV. The 2014 Response Criteria Working Group Report. Biology of Blood and Marrow Transplantation. 2015 Jun 1;21(6):984–99. [CrossRef]
- Liu B, Pan S, Dong X, Qiao H, Jiang H, Krissansen GW, et al. Opposing effects of arsenic trioxide on hepatocellular carcinomas in mice. Cancer Sci. 2006 Jul;97(7):675–81. [CrossRef]
- Cicconi L, Platzbecker U, Avvisati G, Paoloni F, Thiede C, Vignetti M, et al. Long-term results of all-trans retinoic acid and arsenic trioxide in non-high-risk acute promyelocytic leukemia: update of the APL0406 Italian-German randomized trial. Leukemia. 2020 Mar;34(3):914–8. [CrossRef]
- Platzbecker U, Avvisati G, Cicconi L, Thiede C, Paoloni F, Vignetti M, et al. Improved Outcomes With Retinoic Acid and Arsenic Trioxide Compared With Retinoic Acid and Chemotherapy in Non–High-Risk Acute Promyelocytic Leukemia: Final Results of the Randomized Italian-German APL0406 Trial. JCO. 2017 Feb 20;35(6):605–12. [CrossRef]
- Raffoux E, Rousselot P, Poupon J, Daniel MT, Cassinat B, Delarue R, et al. Combined treatment with arsenic trioxide and all-trans-retinoic acid in patients with relapsed acute promyelocytic leukemia. J Clin Oncol. 2003 Jun 15;21(12):2326–34. [CrossRef]
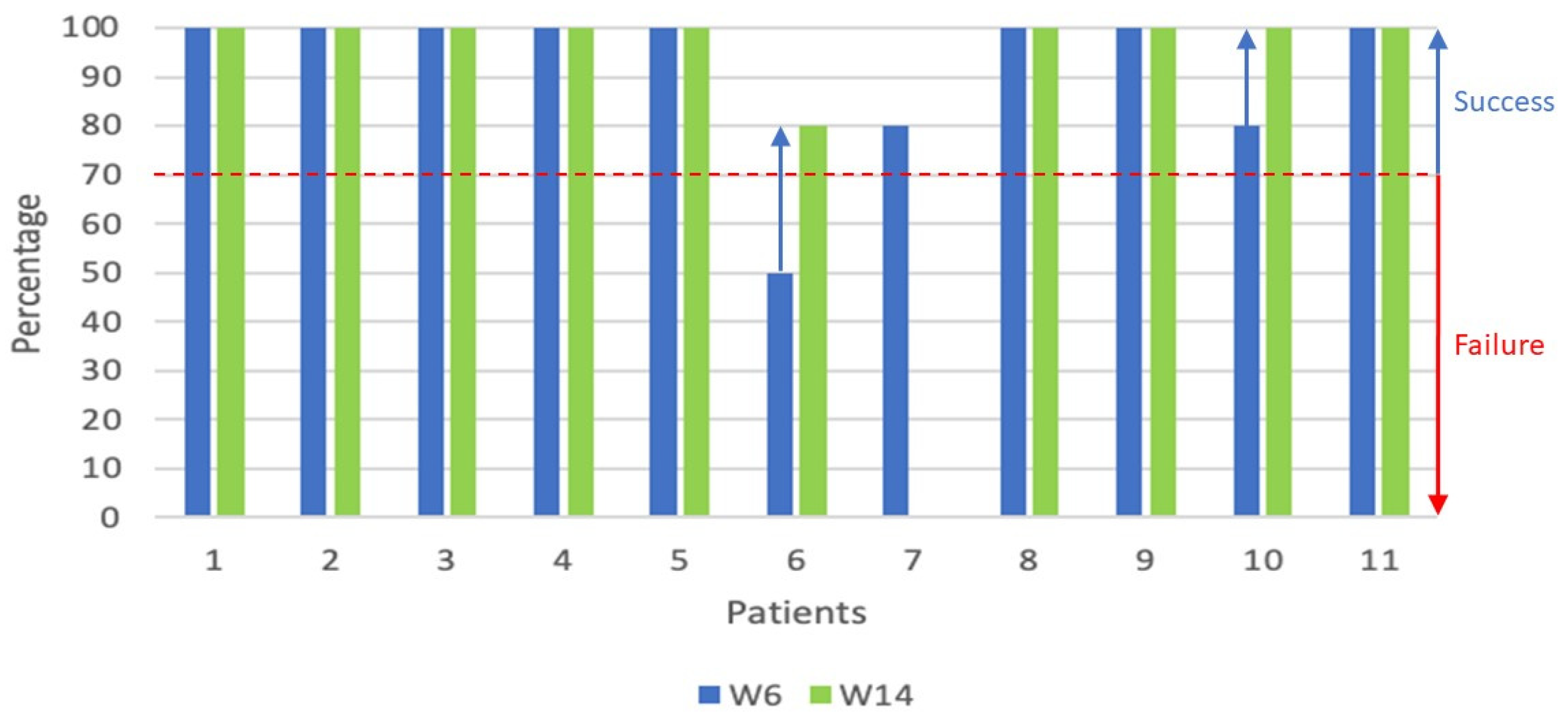
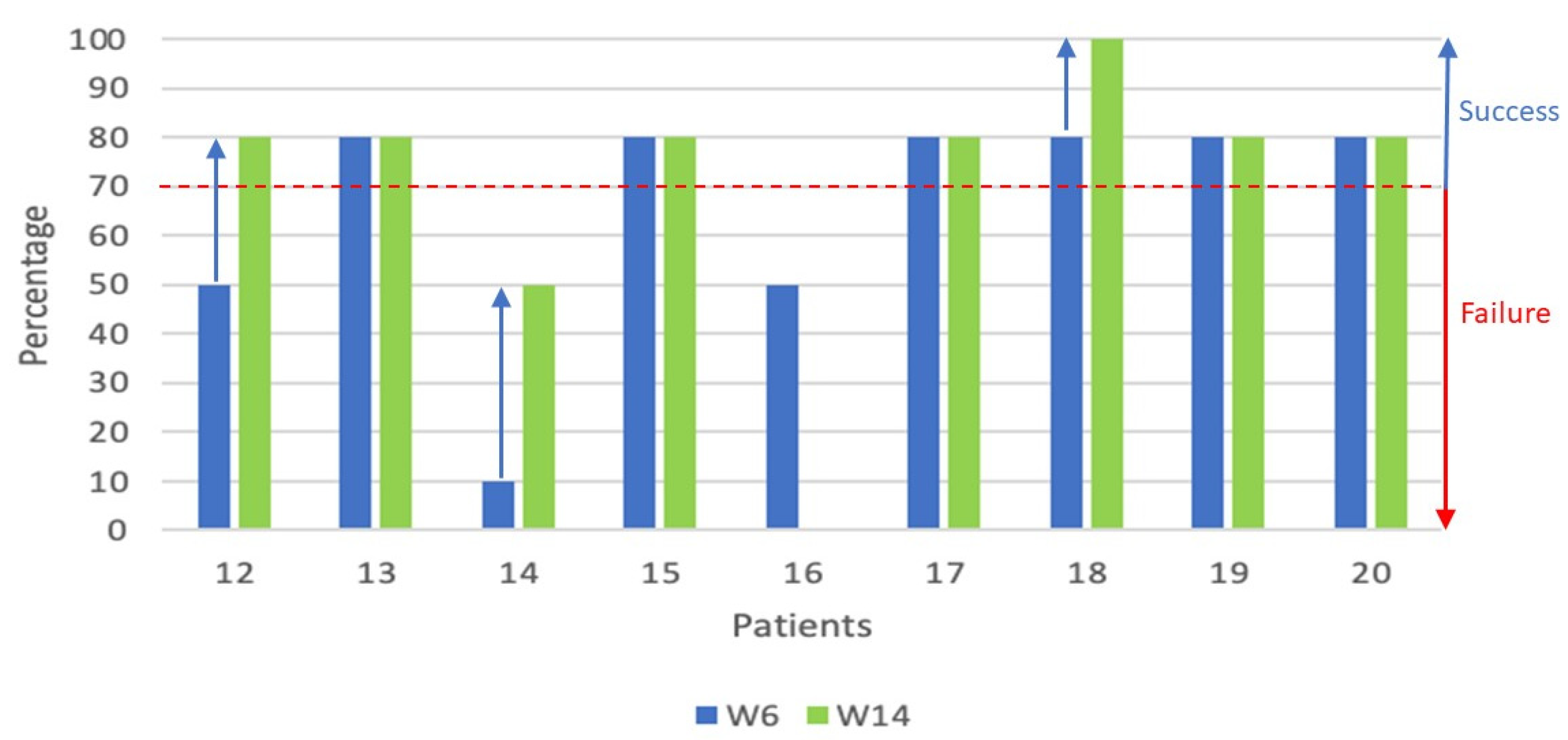
| Patient study number | Response at W6 | Response at W14 | Response at M6 | Response at M9 | Response at M12 | Second line therapy |
| 6 | MR | PR | PD* | PD* | MR* | Ruxolitinib started at M5 (5, 10 and 15 mg/day) |
| 1 | CR | CR | PR | PR* | PR* | Photopheresis started at M6 |
| 2 | CR | CR | PR | PR* | CR* | Photopheresis started at M6 |
| 4 | CR | CR | PR | PR | PR* | Ruxolitinib started at M9 (5 and 10 mg/day) |
| 5 | CR | CR | CR | PD | PR* | Ruxolitinib started after M9 (5 and 10 mg/day) |
| Patient study number | Response at W6 | Response at W14 | Response at M6 |
Response at M9 | Response at M12 | Second line therapy |
| 12 | MR | PR | MR* | PR* | PR* | Photopheresis started at M4 (ongoing at the end of study), Rituximab started at M5 (175mg/m2 and during 1M), Ruxolitinib (5 mg) started at M11 (ongoing at the end of study) |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




