Submitted:
07 March 2024
Posted:
08 March 2024
You are already at the latest version
Abstract
Keywords:
1. Introduction
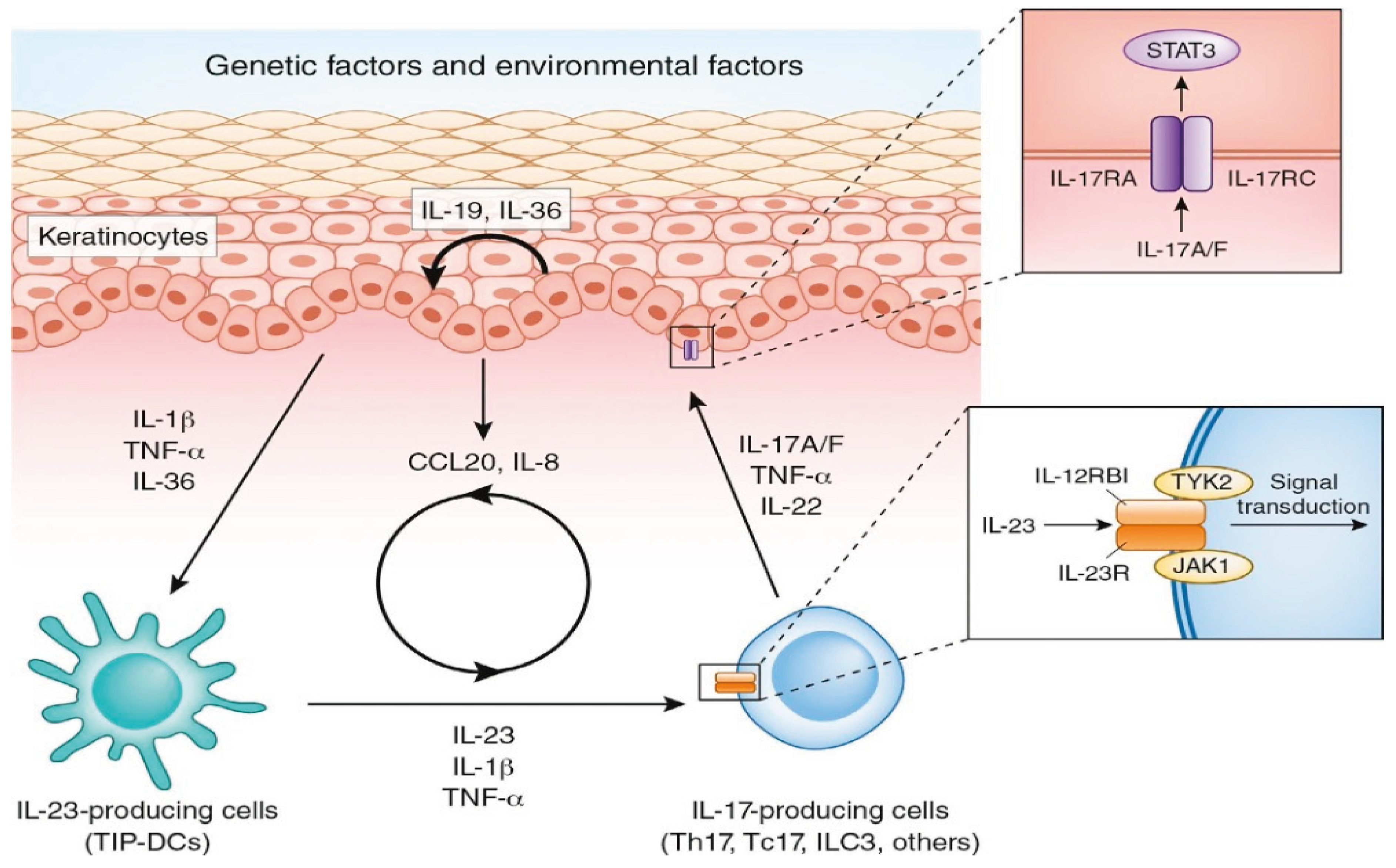
2. Materials and Methods
2.1. Materials
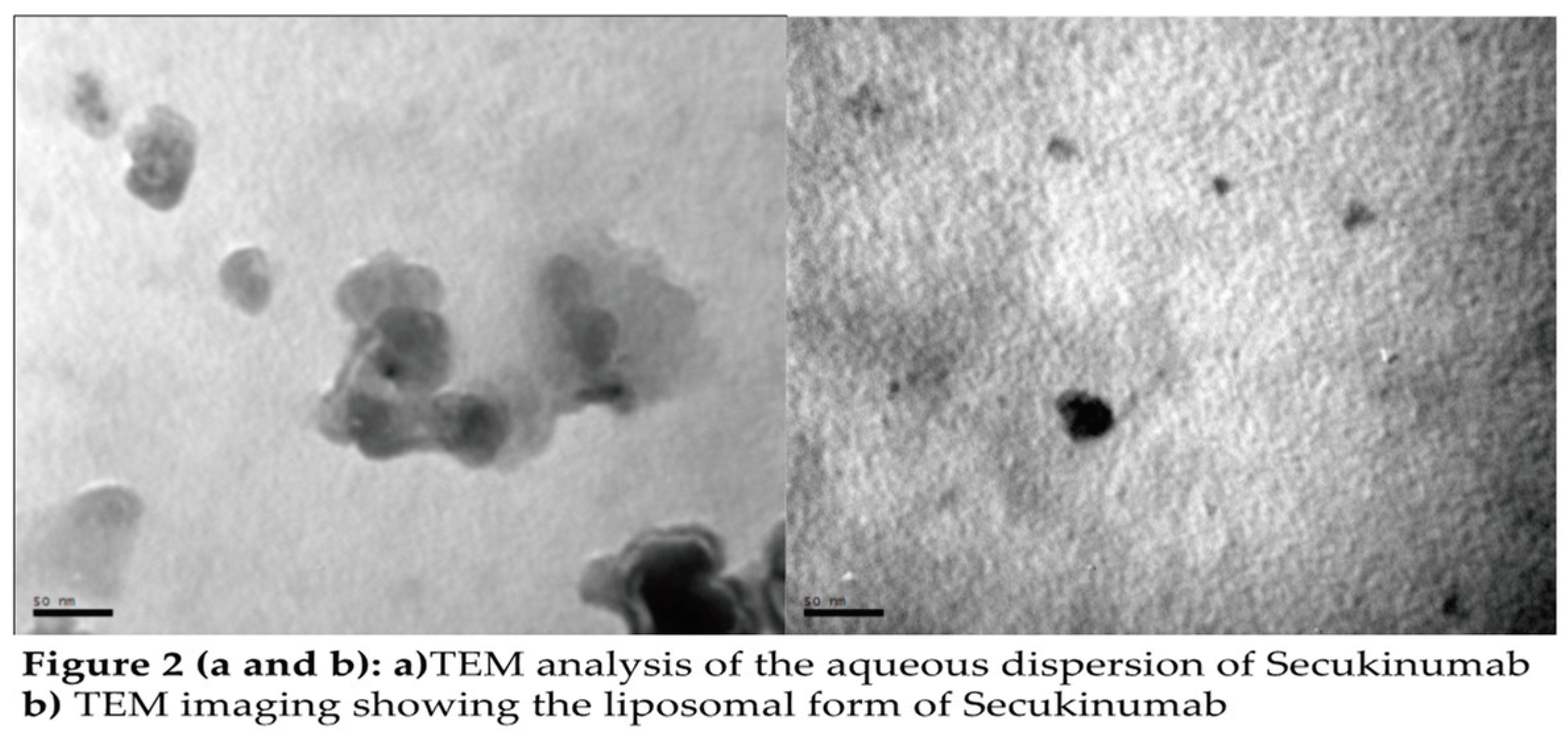
2.2. Preparation and Characterisation of Peptide-Loaded Liposome
2.3. Ethical Approval
2.4. Blood Collection
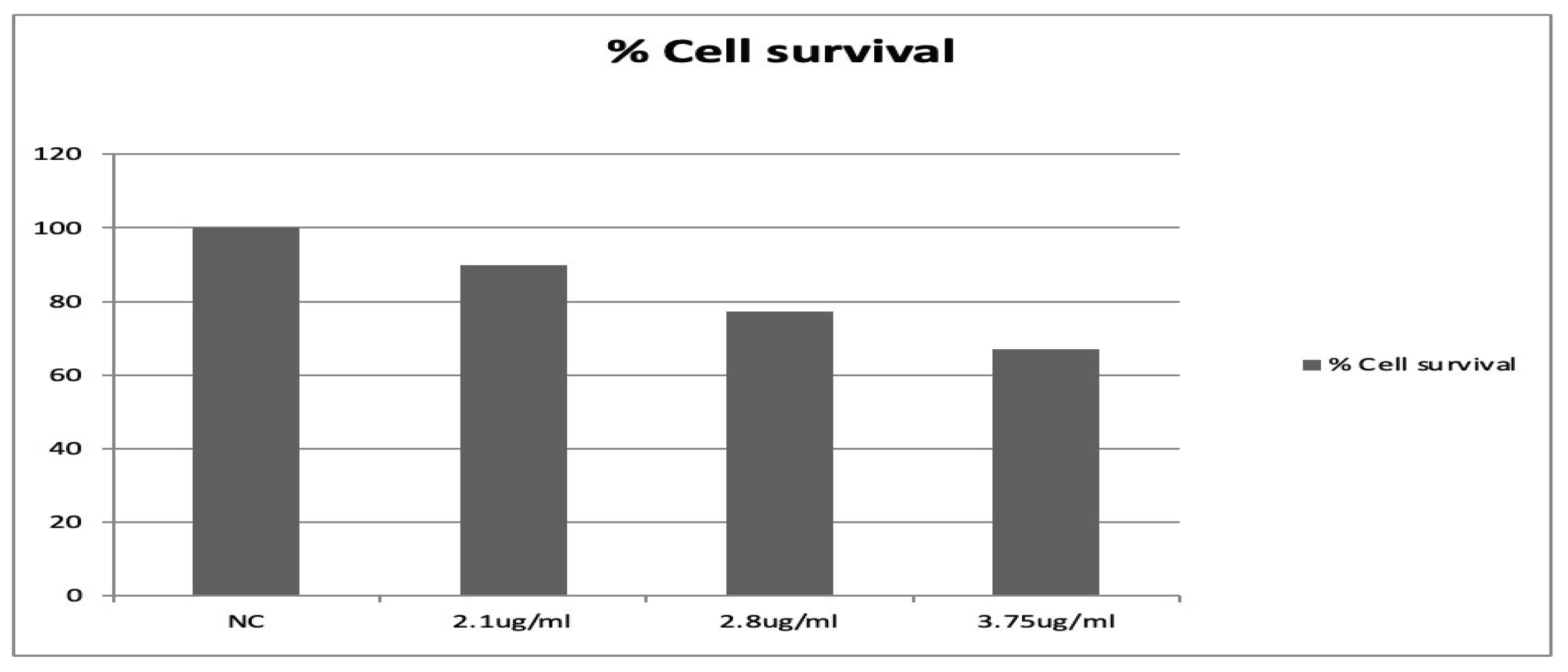
2.5. Cell Viability Determination
| No | Sample Code | Age | Ethnicity | Gender | Smoking History |
|---|---|---|---|---|---|
| 1 | P3 26719 | 31 | Caucasian | M | Ex S |
| 2 | P1 26719 | 41 | Asian | M | N |
| 3 | P3 26719 | 65 | Caucasian | F | Ex S |
| 4 | P4 25719 | 64 | Asian | F | N |
| 5 | P2 21519 | 23 | Caucasian | F | N |
| 6 | P3 21519 | 47 | Caucasian | M | Ex S |
| 7 | P1 21519 | 50 | Caucasian | M | N |
| 8 | P128519 | 35 | Caucasian | F | N |
| 9 | P228519 | 38 | Caucasian | M | Y |
| 10 | P125619 | 80 | Caucasian | F | N |
| 11 | P2 25619 | 64 | Caucasian | M | N |
| 12 | P2 18619 | 62 | Caucasian | F | N |
| 13 | P1 18619 | 44 | Caucasian | M | N |
| 14 | P1 2719 | 80 | Caucasian | F | Ex S |
| 15 | P2 2719 | 53 | Caucasian | F | N |
| 16 | P3 2719 | 63 | Caucasian | F | Ex S |
| 17 | P1 9719 | 35 | Caucasian | M | Y |
| 18 | P2 9719 | 55 | Caucasian | F | N |
| 19 | P3 9719 | 42 | Caucasian | F | N |
| 20 | P4 9719 | 35 | Asian | M | N |
| No | Age | Ethnicity | Gender | Smoking History |
|---|---|---|---|---|
| 1 | 33 | Caucasian | F | Non-smoker |
| 2 | 34 | Asian | M | Past smoker |
| 3 | 20 | Asian | F | Non-smoker |
| 4 | 42 | Asian | F | Non-smoker |
| 5 | 21 | Caucasian | F | Non-smoker |
| 6 | 20 | Asian | M | Non-smoker |
| 7 | 21 | Asian | M | Non-smoker |
| 8 | 22 | Asian | F | Non-smoker |
| 9 | 21 | Caucasian | F | Non-smoker |
| 10 | 22 | Asian | F | Non-smoker |
| 11 | 24 | Asian | F | Non-smoker |
| 12 | 35 | Caucasian | F | Non-smoker |
| 13 | 37 | Caucasian | M | Non-smoker |
| 14 | 40 | Asian | M | Non-smoker |
| 15 | 39 | African | F | Non-smoker |
| 16 | 33 | Asian | M | Non-smoker |
| 17 | 39 | Caucasian | M | Non-smoker |
| 18 | 28 | Caucasian | M | Non-smoker |
| 19 | 35 | Arab | Male | Non-smoker |
| 20 | 48 | Arab | Male | Non-smoker |
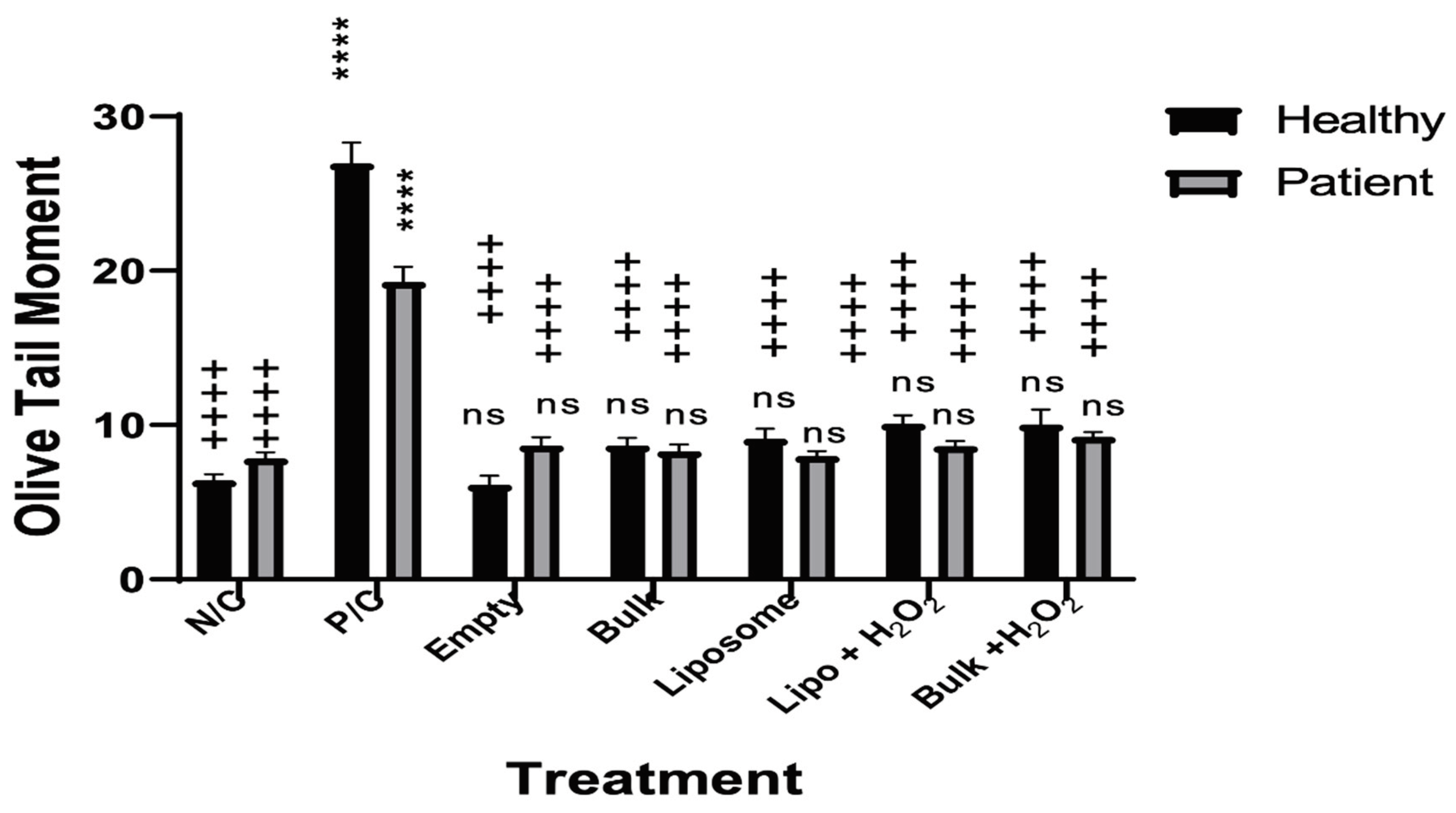
2.6. Cell Treatment and the Comet Assay
2.7. Cytokinesis Block Micronucleus Assay (CBMN)
2.8. Statistical Analysis
3. Results
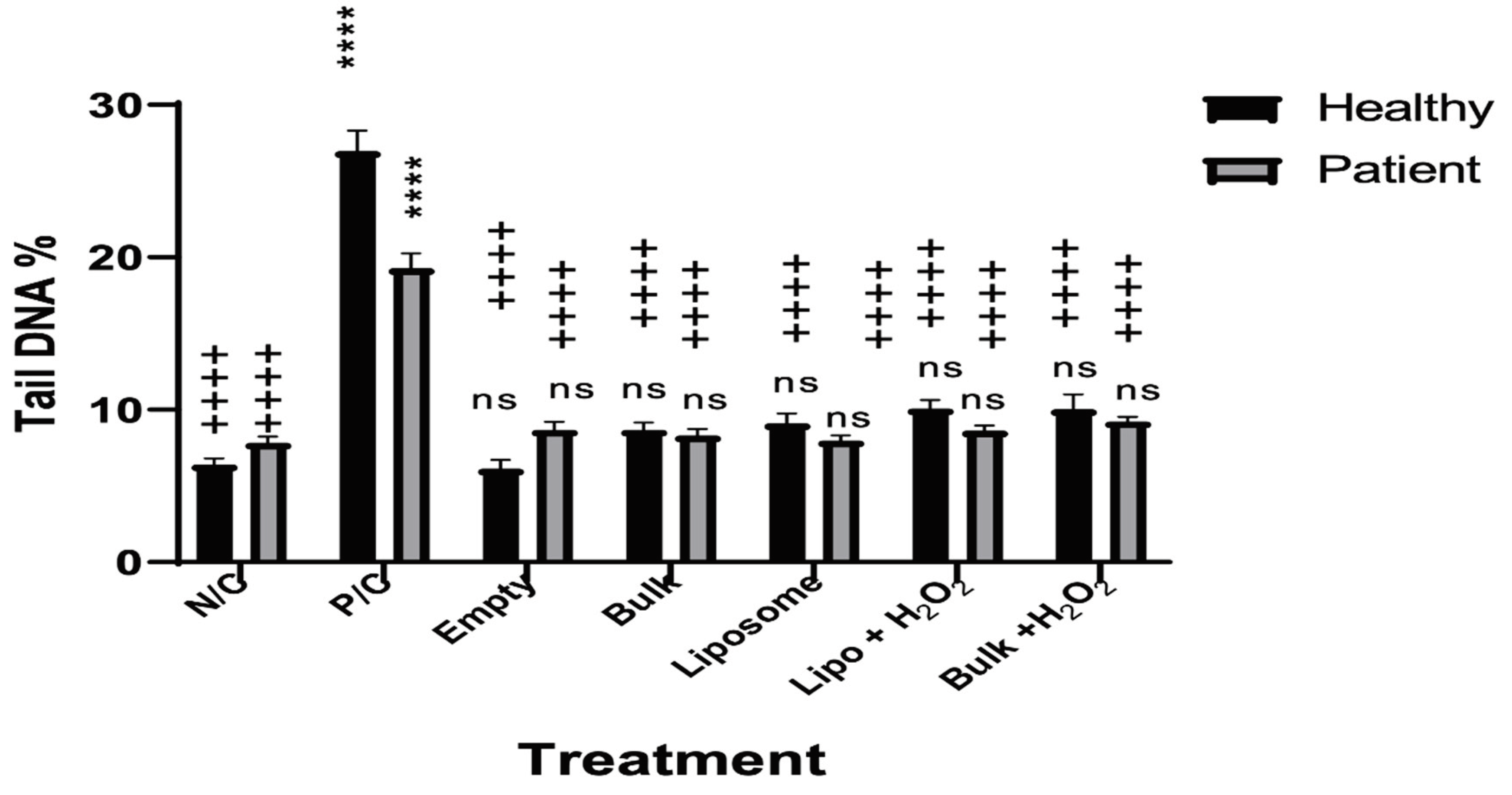
3.1. The Effects of Secukinumab and H2O2 on the Lymphocytes from Healthy
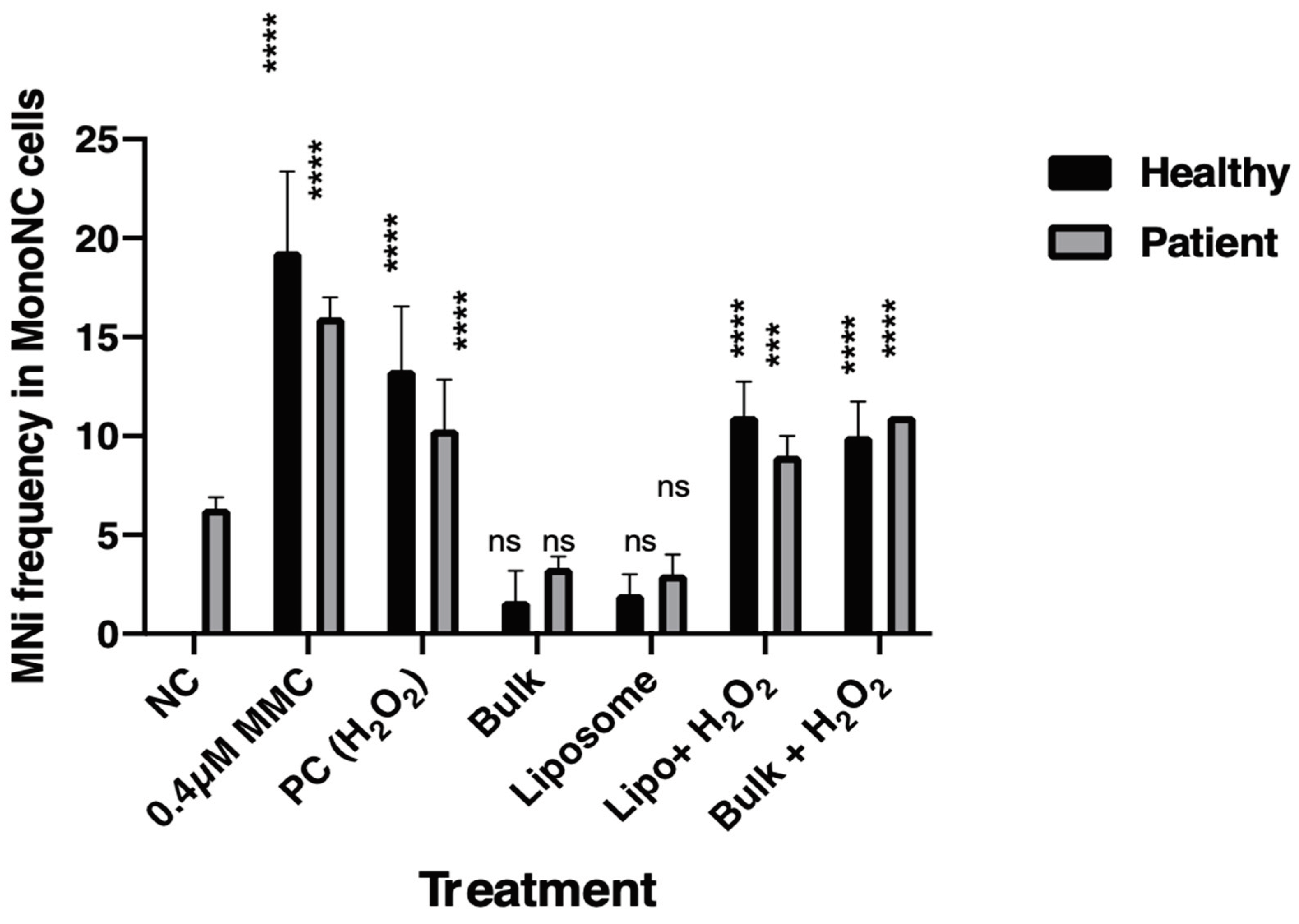
3.2. Cytokinesis Block Micronucleus Assay (CBMN)
4. Discussion
5. Conclusion
Abbreviations
| CHO | Chinese Hamster Ovary cells |
| Chol | Cholesterol |
| CBMN | cytokinesis-block micronucleus |
| DCM | methanol dichloromethane |
| DOPE | Dioleoyl phsphoethanolamine |
| DPPG | 1,2-Dipalmitoyl-sn-glycerol sodium salt |
| IL-17 | Interleukin 17 |
| mAb | Monoclonal antibody |
| PSAI | Psoriasis Area and Severity Index |
| Th | T-Helper cells |
| TNFa | Tumor necrosis factor-alpha |
| RPMI | Mammalian cell culture media/ Roswell Park Memorial Institute |
References
- ANDERSON, D., NAJAFZADEH, M., GOPALAN, R., GHADERI, N., SCALLY, A. J., BRITLAND, S. T., JACOBS, B. K., REYNOLDS, P. D., DAVIES, J. & WRIGHT, A. L. 2014. Sensitivity and specificity of the empirical lymphocyte genome sensitivity (LGS) assay: implications for improving cancer diagnostics. The FASEB Journal, 28, 4563-4570.
- BAGEL, J.; BLAUVELT, A.; NIA, J.; HASHIM, P.; PATEKAR, M.; DE VERA, A.; AHMAD, K.; PAGUET, B.; XIA, S.; MUSCIANISI, E.; et al. Secukinumab maintains superiority over ustekinumab in clearing skin and improving quality of life in patients with moderate to severe plaque psoriasis: 52-week results from a double-blind phase 3b trial (CLARITY). J Eur Acad Dermatol Venereol 2021, 35, 135–142. [Google Scholar] [CrossRef]
- BISSONNETTE, R.; LUGER, T.; THACI, D.; TOTH, D.; LACOMBE, A.; XIA, S.; MAZUR, R.; PATEKAR, M.; CHAREF, P.; MILUTINOVIC, M.; LEONARDI, C.; MROWIETZ, U. Secukinumab demonstrates high sustained efficacy and a favourable safety profile in patients with moderate-to-severe psoriasis through 5 years of treatment (SCULPTURE Extension Study). J Eur Acad Dermatol Venereol 2018, 32, 1507–1514. [Google Scholar] [CrossRef]
- BRIDGEWOOD, C.; WITTMANN, M.; MACLEOD, T.; WATAD, A.; NEWTON, D.; BHAN, K.; AMITAL, H.; DAMIANI, G.; GIRYES, S.; BRAGAZZI, N. L.; MCGONAGLE, D. T Helper 2 IL-4/IL-13 Dual Blockade with Dupilumab Is Linked to Some Emergent T Helper 17Type Diseases, Including Seronegative Arthritis and Enthesitis/Enthesopathy, but Not to Humoral Autoimmune Diseases. J Invest Dermatol 2022, 142, 2660–2667. [Google Scholar] [CrossRef]
- CIAZYNSKA, M.; OLEJNICZAK-STARUCH, I.; SOBOLEWSKA-SZTYCHNY, D.; NARBUTT, J.; SKIBINSKA, M.; LESIAK, A. Ultraviolet Radiation and Chronic Inflammation-Molecules and Mechanisms Involved in Skin Carcinogenesis: A Narrative Review. Life (Basel) 2021, 11. [Google Scholar]
- DAMIANI, G.; ODORICI, G.; PACIFICO, A.; MORRONE, A.; CONIC, R.R.Z.; DAVIDSON, T.; WATAD, A.; PIGATTO, P.D.M.; COLOMBO, D.; MALAGOLI, P.; FIORE, M. Secukinumab Loss of Efficacy Is Perfectly Counteracted by the Introduction of Combination Therapy (Rescue Therapy): Data from a Multicenter Real-Life Study in a Cohort of Italian Psoriatic Patients That Avoided Secukinumab Switching. Pharmaceuticals (Basel) 2022, 15. [Google Scholar] [CrossRef] [PubMed]
- ÉCONOMIQUES, O. D. C. E. D. D. 2016. Test No. 487: In vitro mammalian cell micronucleus test, OECD Publishing.
- ELDER, J. T. Expanded genome-wide association study meta-analysis of psoriasis expands the catalog of common psoriasis-associated variants. Journal of Investigative Dermatology Symposium Proceedings, 2018. Elsevier, S77-S78.
- FENECH, M. 2007. Cytokinesis-block micronucleus cytome assay. Nat Protoc 2007, 2, 1084–1104. [Google Scholar] [CrossRef]
- FENECH, M. 2009. A lifetime passion for micronucleus cytome assays—reflections from Down Under. Mutation Research/Reviews in Mutation Research 2009, 681, 111–117. [Google Scholar] [CrossRef]
- FENECH, M.; KNASMUELLER, S.; BOLOGNESI, C.; BONASSI, S.; HOLLAND, N.; MIGLIORE, L.; PALITTI, F.; NATARAJAN, A. T.; KIRSCH-VOLDERS, M. Molecular mechanisms by which in vivo exposure to exogenous chemical genotoxic agents can lead to micronucleus formation in lymphocytes in vivo and ex vivo in humans. Mutat Res 2016, 770, 12–25. [Google Scholar] [CrossRef]
- GRIFFITHS, C.E.; BARKER, J.N. Pathogenesis and clinical features of psoriasis. Lancet 2007, 370, 263–271. [Google Scholar] [CrossRef]
- HEIDENREICH, R.; ROCKEN, M.; GHORESCHI, K. Angiogenesis drives psoriasis pathogenesis. Int J Exp Pathol 2009, 90, 232–248. [Google Scholar] [CrossRef]
- KADAM, D.P.; SURYAKAR, A.N.; ANKUSH, R.D.; KADAM, C.Y.; DESHPANDE, K.H. Role of oxidative stress in various stages of psoriasis. Indian J Clin Biochem 2010, 25, 388–392. [Google Scholar] [CrossRef] [PubMed]
- LAI, R.; XIAN, D.; XIONG, X.; YANG, L.; SONG, J.; ZHONG, J. Proanthocyanidins: novel treatment for psoriasis that reduces oxidative stress and modulates Th17 and Treg cel. Redox Report 2018, 23, 130–135. [Google Scholar] [CrossRef] [PubMed]
- LIN, X.; HUANG, T. Oxidative stress in psoriasis and potential therapeutic use of antioxidants. Free radical research 2016, 50, 585–595. [Google Scholar] [CrossRef] [PubMed]
- LOMBARDO, D.; KISELEV, M.A. Methods of Liposomes Preparation: Formation and Control Factors of Versatile Nanocarriers for Biomedical and Nanomedicine Application. Pharmaceutics 2022, 14. [Google Scholar] [CrossRef] [PubMed]
- MAGDOLENOVA, Z.; LORENZO, Y.; COLLINS, A.; DUSINSKA, M. Can standard genotoxicity tests be applied to nanoparticles? Journal of Toxicology and Environmental Health, Part A 2012, 75, 800–806. [Google Scholar] [CrossRef] [PubMed]
- MATSUZAKI, G.; UMEMURA, M. Interleukin-17 family cytokines in protective immunity against infections: role of hematopoietic cell-derived and non-hematopoietic cell-derived interleukin-17s. Microbiol Immunol 2018, 62, 1–13. [Google Scholar] [CrossRef] [PubMed]
- MIYAGAWA, F. Pathogenesis of Paradoxical Reactions Associated with Targeted Biologic Agents for Inflammatory Skin Diseases. Biomedicines 2022, 10. [Google Scholar] [CrossRef]
- MOLDEN, R.; HU, M.; YEN, E.S.; SAGGESE, D.; REILLY, J.; MATTILA, J.; QIU, H.; CHEN, G.; BAK, H.; LI, N. Host cell protein profiling of commercial therapeutic protein drugs as a benchmark for monoclonal antibody-based therapeutic protein development. MAbs 2021, 13, 1955811. [Google Scholar] [CrossRef]
- NAJAFZADEH, M.; GUEIDAN, C.; BADALI, H.; VAN DEN ENDE, A.G.; XI, L.; DE HOOG, G. Genetic diversity and species delimitation in the opportunistic genus Fonsecaea. Medical mycology 2009, 47, 17–25. [Google Scholar] [CrossRef]
- NAJAFZADEH, M.A.D. The use of isolated peripheral lymphocytes and human whole blood in the comet assay. Nature Protocolexchange 2016. [Google Scholar] [CrossRef]
- NESTLE, F.O.; TURKA, L.A.; NICKOLOFF, B.J. Characterization of dermal dendritic cells in psoriasis. Autostimulation of T lymphocytes and induction of Th1 type cytokines. J Clin Invest 1994, 94, 202–209. [Google Scholar] [CrossRef]
- NO, O.T. 489: In vivo Mammalian alkaline Comet assay. OECD Guidelines for the Testing of Chemicals. OECD Publ 2014, 4, 1–21. [Google Scholar]
- NSAIRAT, H.; KHATER, D.; SAYED, U.; ODEH, F.; AL BAWAB, A.; ALSHAER, W. Liposomes: structure, composition, types, and clinical applications. Heliyon 2022, 8, e09394. [Google Scholar] [CrossRef]
- REICH, K.; BLAUVELT, A.; ARMSTRONG, A.; LANGLEY, R.G.; DE VERA, A.; KOLBINGER, F.; SPINDELDREHER, S.; REN, M.; BRUIN, G. Secukinumab, a fully human anti-interleukin-17A monoclonal antibody, exhibits low immunogenicity in psoriasis patients treated up to 5 years. J Eur Acad Dermatol Venereol 2019, 33, 1733–1741. [Google Scholar] [CrossRef]
- RENDON, A.; SCHÄKEL, K. Psoriasis Pathogenesis and Treatment. International journal of molecular sciences 2019, 20, 1475. [Google Scholar] [CrossRef] [PubMed]
- SCHWEIKERT, B.; MALMBERG, C.; AKERBORG, O.; KUMAR, G.; NOTT, D.; KIRI, S.; SAPIN, C.; HARTZ, S. Cost-Effectiveness Analysis of Sequential Biologic Therapy with Ixekizumab Versus Secukinumab in the Treatment of Active Psoriatic Arthritis with Concomitant Moderate-to-Severe Psoriasis in the UK. Pharmacoecon Open 2020, 4, 635–648. [Google Scholar] [CrossRef] [PubMed]
- SERCOMBE, L.; VEERATI, T.; MOHEIMANI, F.; WU, S.Y.; SOOD, A.K.; HUA, S. Advances and Challenges of Liposome Assisted Drug Delivery. Front Pharmacol 2015, 6, 286. [Google Scholar] [CrossRef] [PubMed]
- STANIĆ, D.; PLEĆAŠ-SOLAROVIĆ, B.; PETROVIĆ, J.; BOGAVAC-STANOJEVIĆ, N.; SOPIĆ, M.; KOTUR-STEVULJEVIĆ, J.; IGNJATOVIĆ, S.; PEŠIĆ, V. Hydrogen peroxide-induced oxidative damage in peripheral blood lymphocytes from rats chronically treated with corticosterone: The protective effect of oxytocin treatment. Chemico-biological interactions 2016, 256, 134–141. [Google Scholar] [CrossRef]
- TICE, R.R.; AGURELL, E.; ANDERSON, D.; BURLINSON, B.; HARTMANN, A.; KOBAYASHI, H.; MIYAMAE, Y.; ROJAS, E.; RYU, J.C.; SASAKI, Y.F. Single cell gel/comet assay: guidelines for in vitro and in vivo genetic toxicology testing. Environ Mol Mutagen 2000, 35, 206–221. [Google Scholar] [CrossRef]
- WAGENER, F.A.; CARELS, C.E.; LUNDVIG, D. Targeting the redox balance in inflammatory skin conditions. International journal of molecular sciences 2013, 14, 9126–9167. [Google Scholar] [CrossRef]
- YAMANAKA, K. Special Issue: "Skin Disease and Comorbidities. J Clin Med 2021, 10. [Google Scholar] [CrossRef] [PubMed]
- ZOU, Y.; MENG, Z. Literature Overview of the IL-17 Inhibition from Psoriasis to COVID-19. J Inflamm Res 2021, 14, 5611–5618. [Google Scholar] [CrossRef] [PubMed]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




