Submitted:
07 March 2024
Posted:
07 March 2024
You are already at the latest version
Abstract
Keywords:
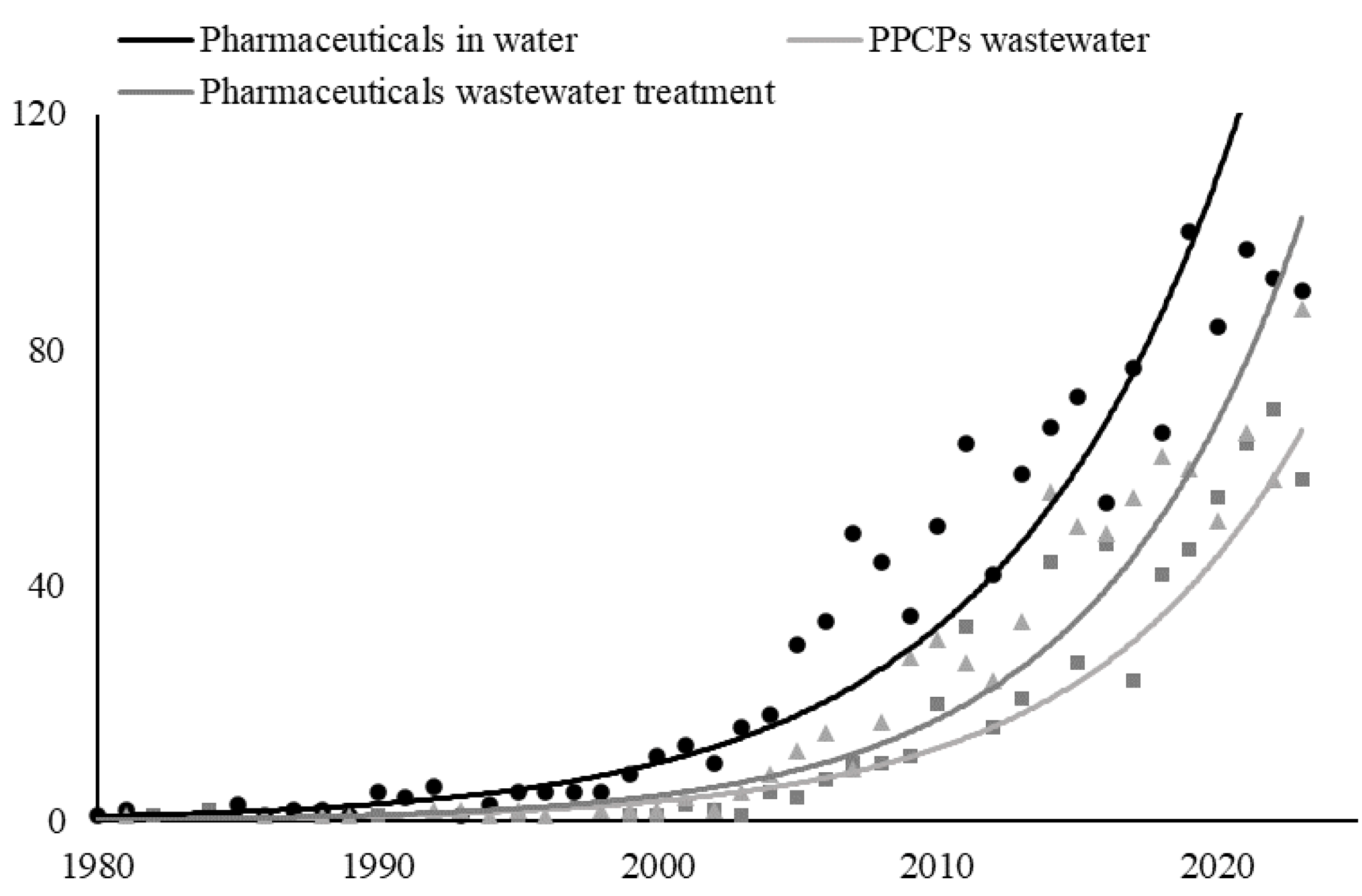
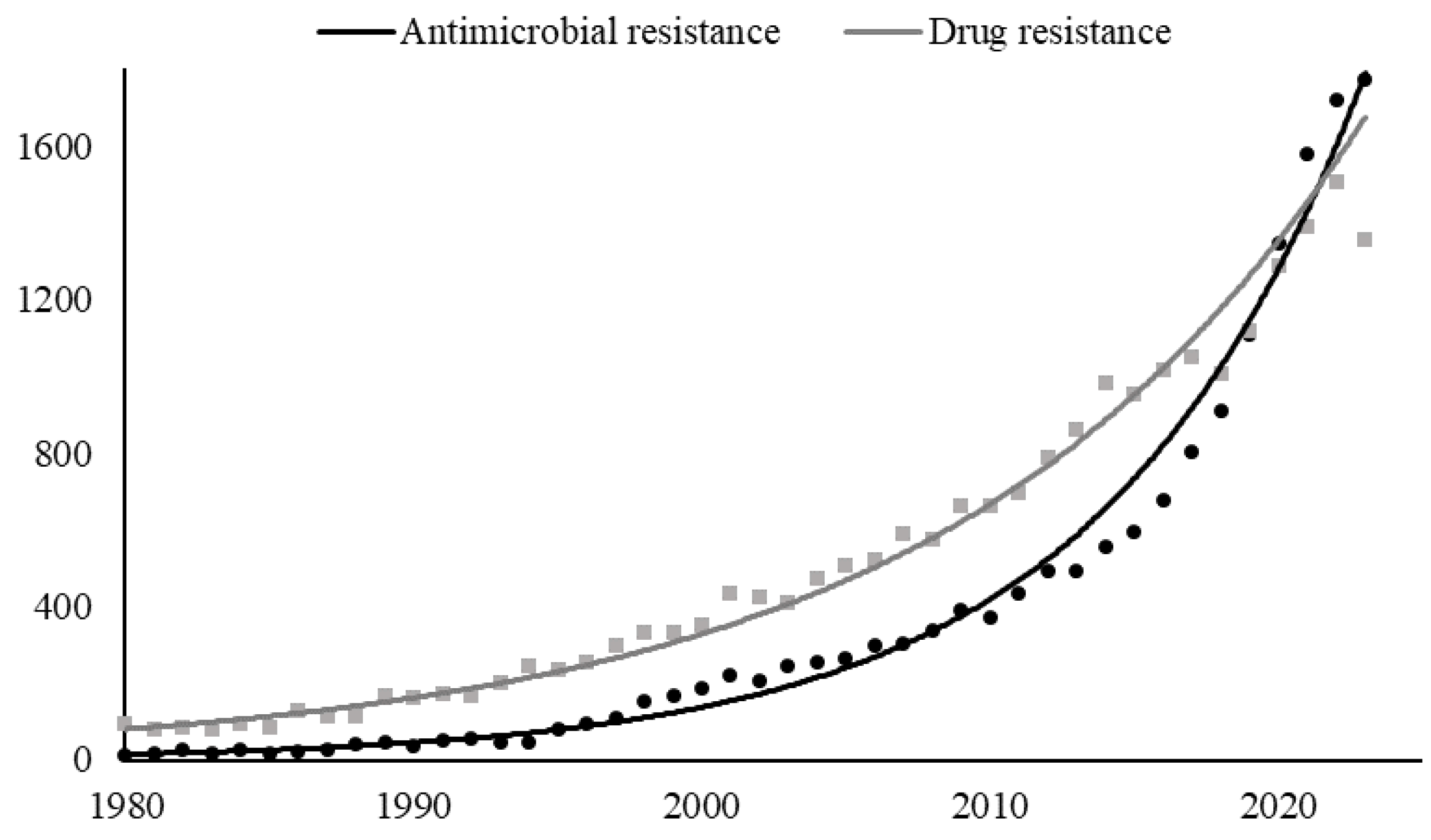
1. Introduction
2. Materials and Methods
2.1. Materials
2.2. CaCO3 Production Method
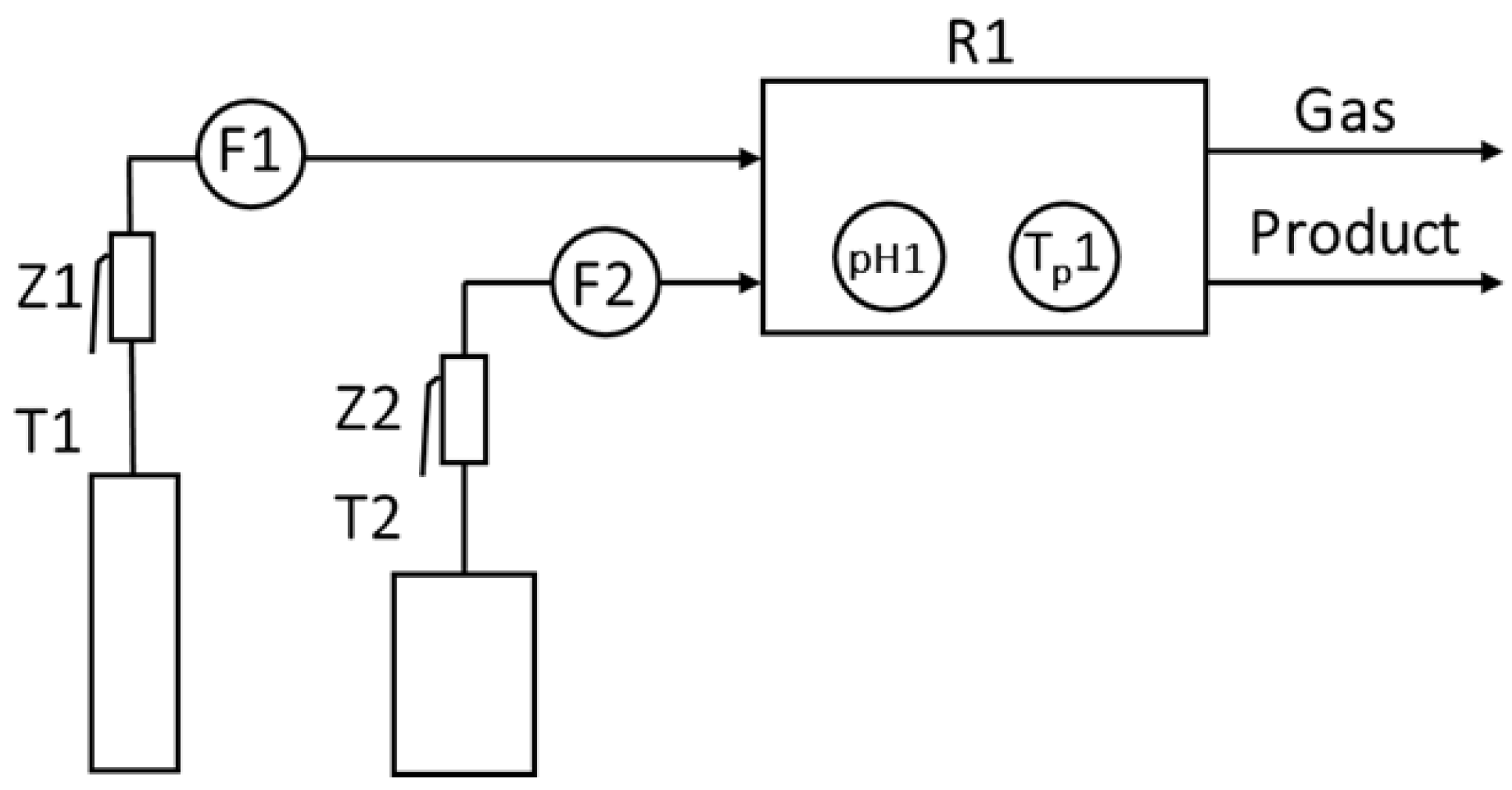
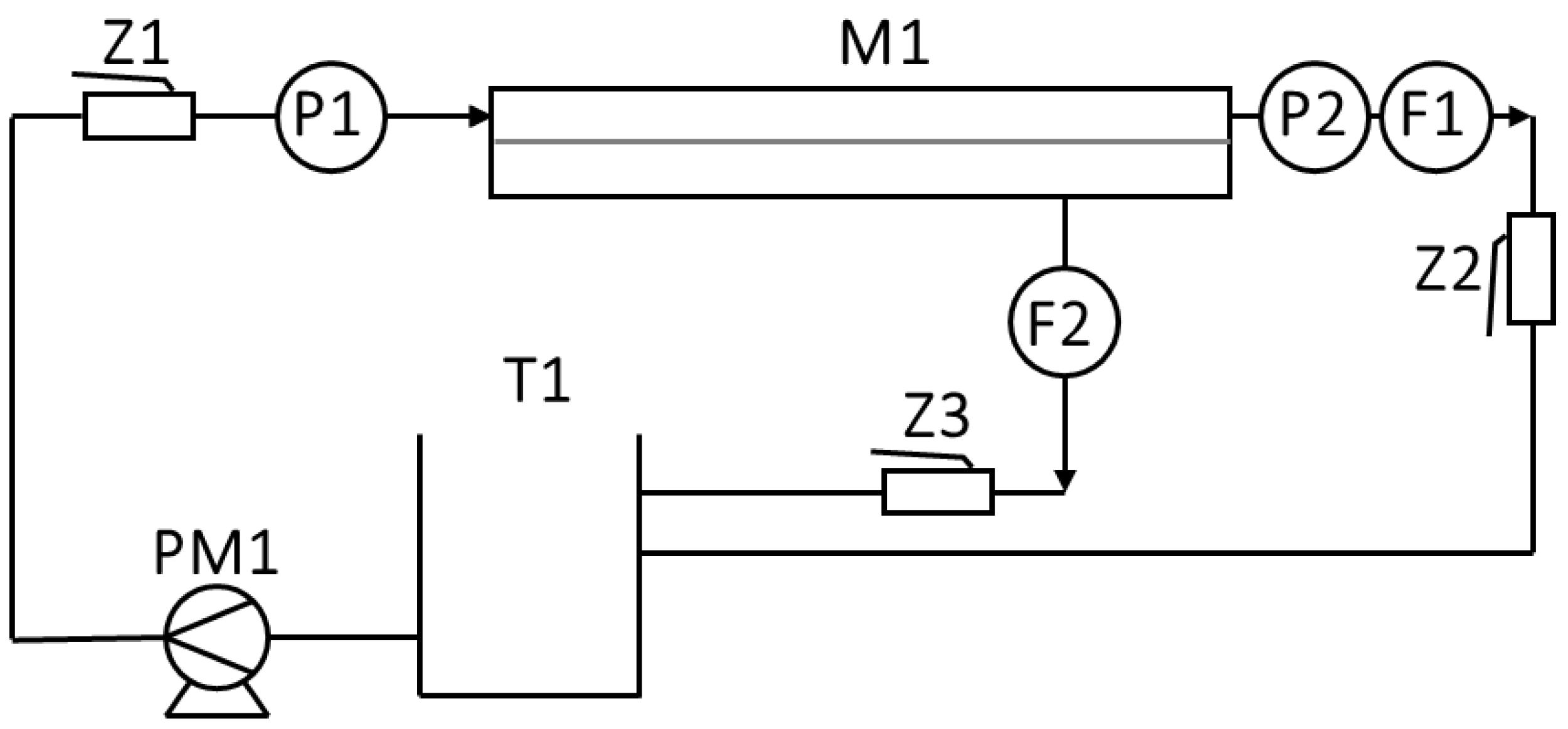
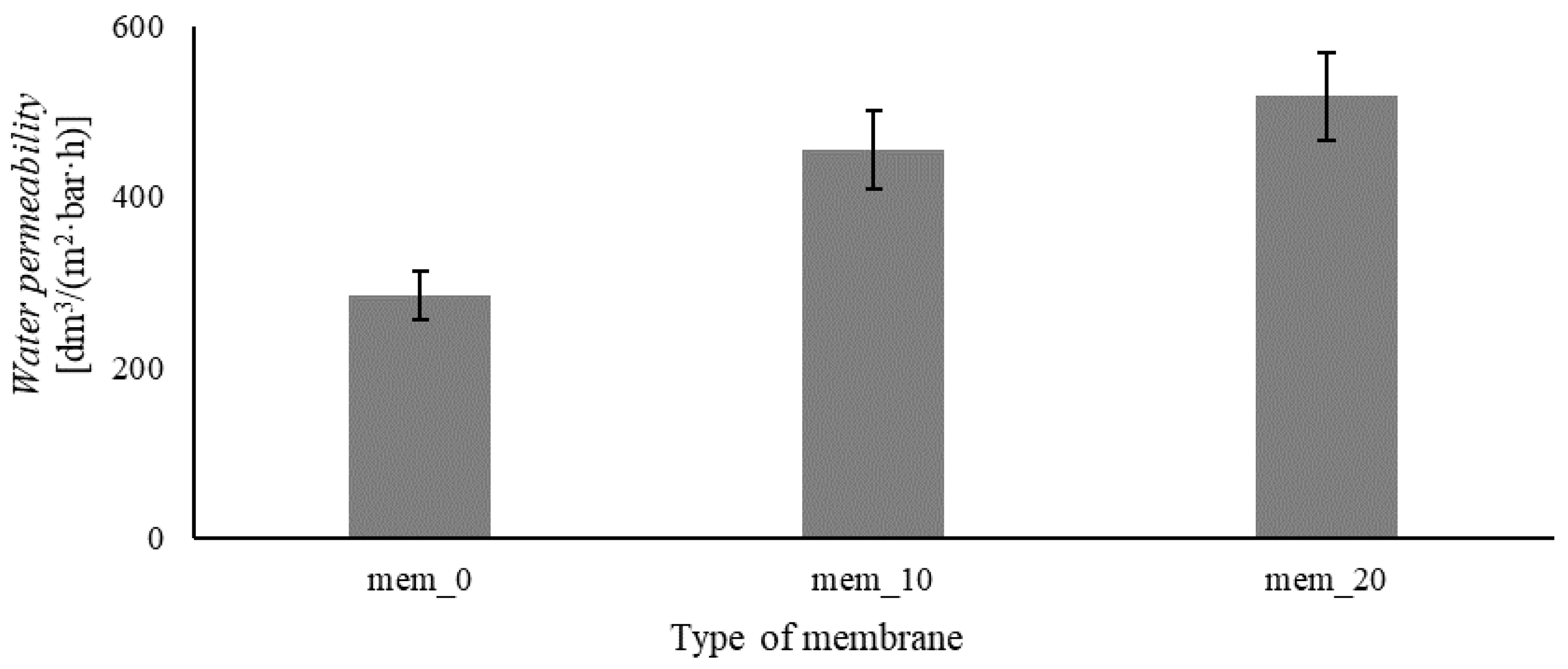
2.3. Method of Membrane Preparation
2.4. Testing of CaCO3 Adsorption Properties
2.5. Morphology of Membranes Testing
2.6. Testing of CaCO3 Antibacterial Properties
2.7. Mechanisms of Adsorption
3. Results and Discussion
3.1. Adsorption Stationary System
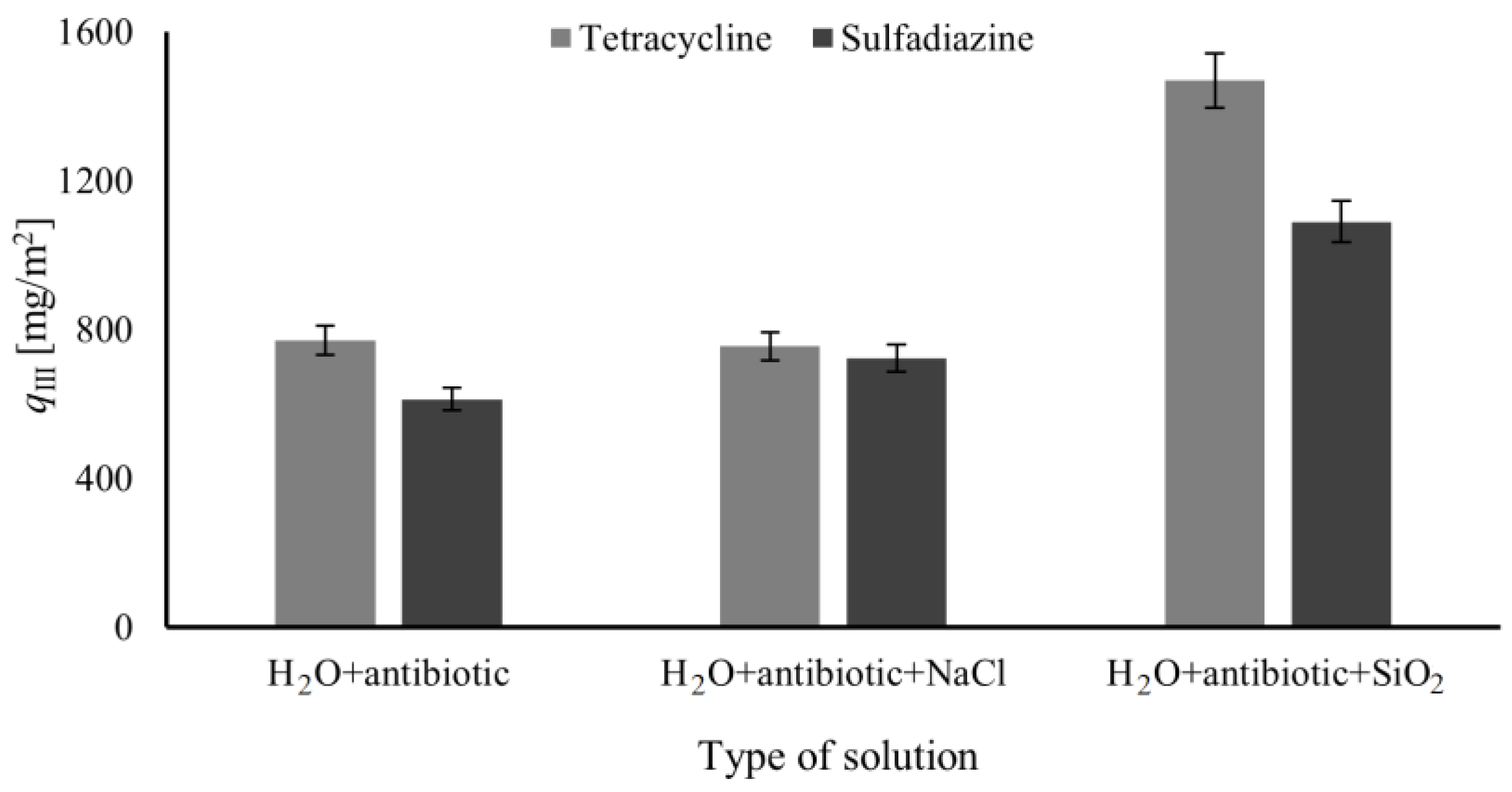
3.2. Adsorption Flow System
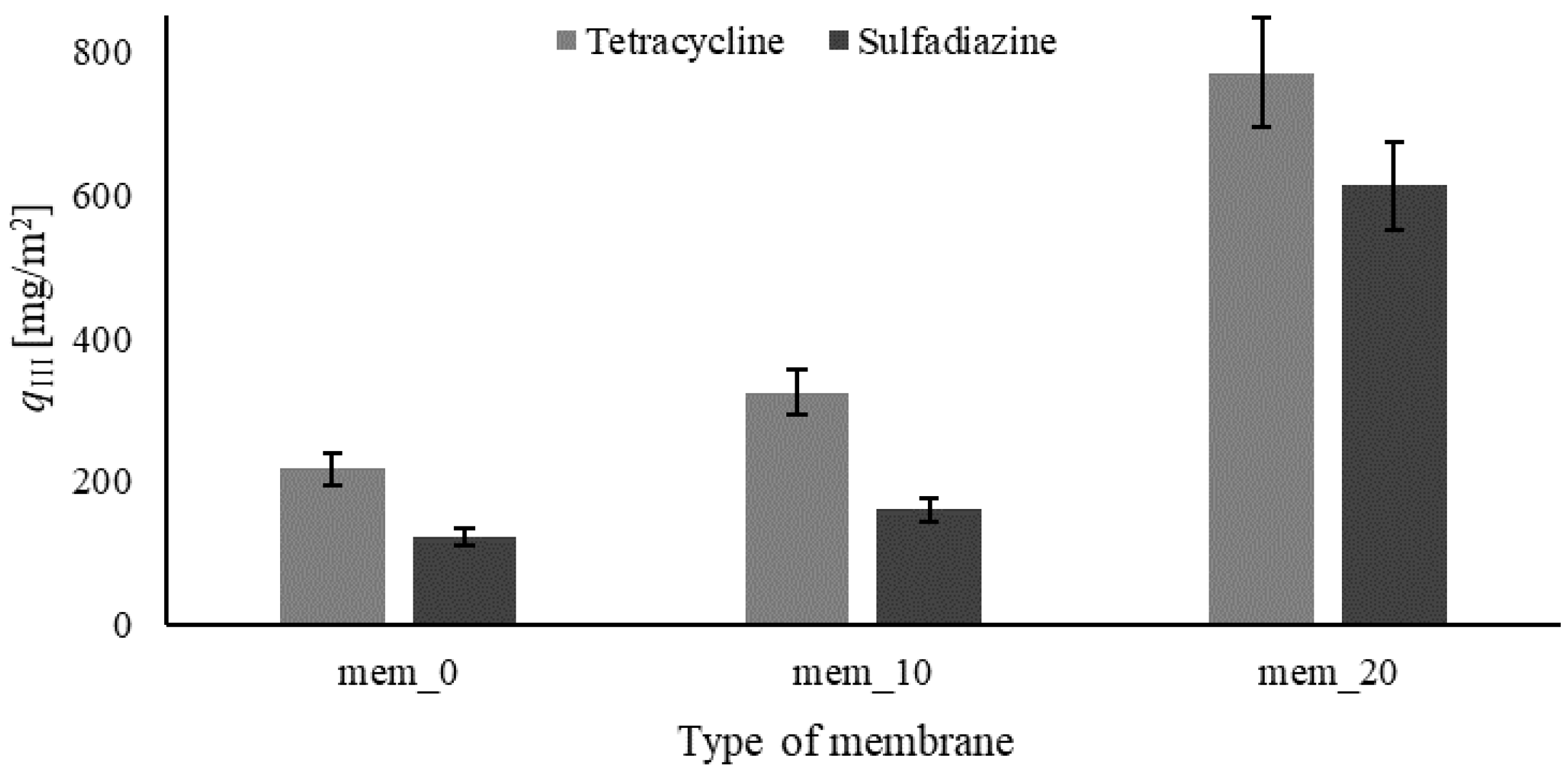
3.3. Filtration-Adsorption System
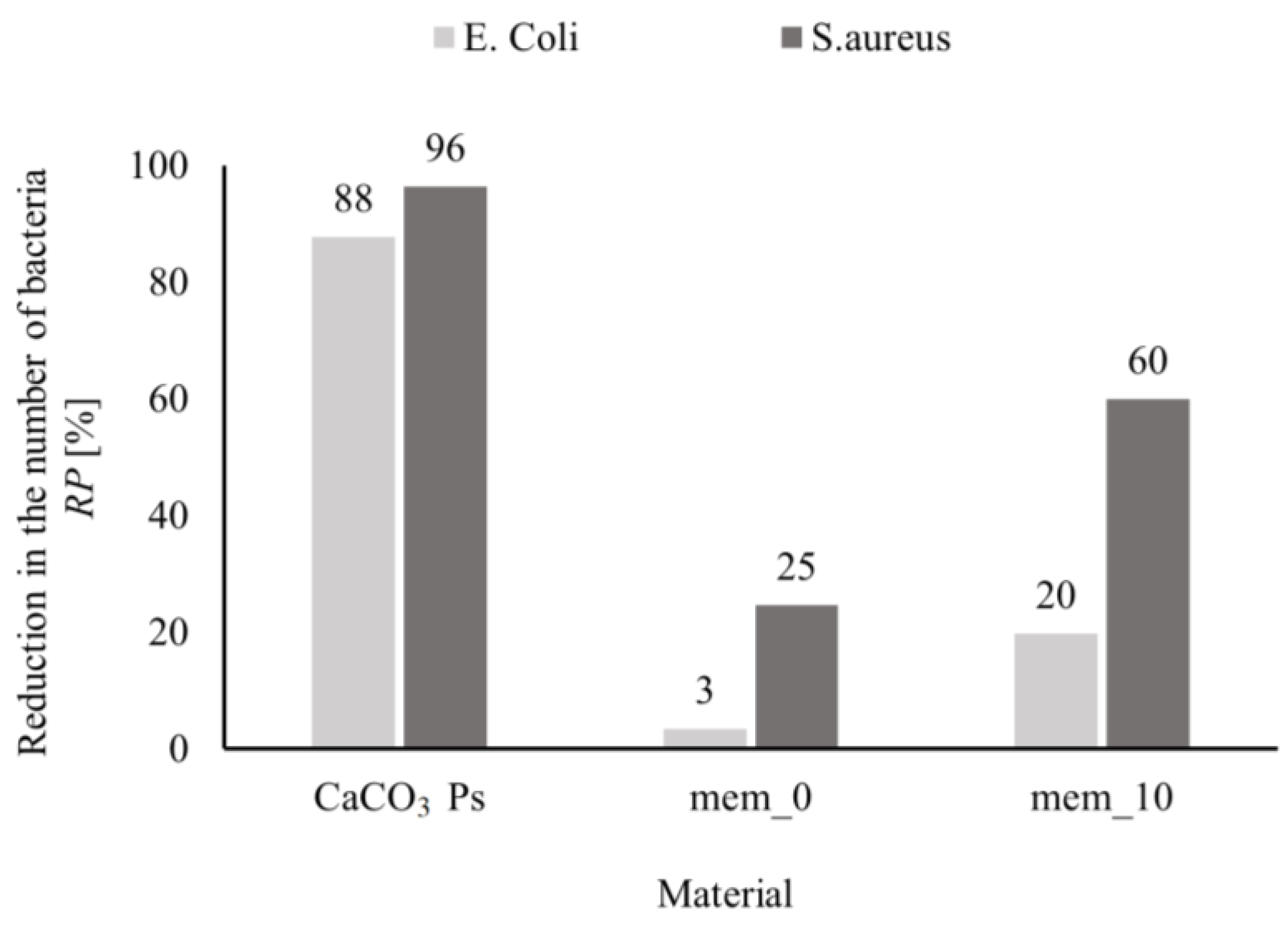
3.4. Antibacterial Properties
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Rodriguez-Narvaez, O. M., Peralta-Hernandez, J. M., Goonetilleke, A., Bandala, E. R., Treatment technologies for emerging contaminants in water: A review. Chemical Engineering Journal, 2017, 323, 361-380. [CrossRef]
- O'Flynn, D., Lawler, J., Yusuf, A., Parle-McDermott, A., Harold, D., Mc Cloughlin, T., Holland, L., Regan, F., White, B., A review of pharmaceutical occurrence and pathways in the aquatic environment in the context of a changing climate and the COVID-19 pandemic. Analitical Methods, 2021, 13, 575-594. [CrossRef]
- Xu, L., Zhang, H., Xiong, P., Zhu, Q., Liao, C., Jiang, G. Occurrence, fate, and risk assessment of typical tetracycline antibiotics in the aquatic environment: A review. Science of The Total Environment, 2021, 753. 141975. [CrossRef]
- Angeles L.F., Mullen, R.A., Huang, I.J., Wilson, C., Khunjar, W., Sirotkin, H.I., McElroy, A. E., Aga, D.S. Assessing pharmaceutical removal and reduction in toxicity provided by advanced wastewater treatment systems. Environmental Science: Water Research & Technology, 2020, 6. 62-77. [CrossRef]
- Dadgostar, P., Antimicrobial resistance: implications and costs. Infection and drug resistance, 2019, 3903-3910. [CrossRef]
- Holmes, A. H., Moore, L. S., Sundsfjord, A., Steinbakk, M., Regmi, S., Karkey, A., Understanding the mechanisms and drivers of antimicrobial resistance. The Lancet, 2015, 387(10014), 176-187. [CrossRef]
- Salam, M. A., Al-Amin, M. Y., Salam, M. T., Pawar, J. S., Akhter, N., Rabaan, A. A., Alqumber, M. A., Antimicrobial resistance: a growing serious threat for global public health. In Healthcare, 2023, Vol. 11, No. 13, p. 1946. [CrossRef]
- Mompelat, S., Le Bot, B.,Thomas, O., Occurrence and fate of pharmaceutical products and by-products, from resource to drinking water. Environment international, 2019, 35(5), 803-814. [CrossRef]
- Nikolaou, A., Meric, S., Fatta, D., Occurrence patterns of pharmaceuticals in water and wastewater environments. Analytical and bioanalytical chemistry, 2017, 387, 1225-1234. [CrossRef]
- Bottoni, P., Caroli, S., Caracciolo, A. B., Pharmaceuticals as priority water contaminants. Toxicological & Environmental Chemistry, 2010, 92(3), 549-565. [CrossRef]
- Ranjan, N., Singh, P. K., Maurya, N. S., Pharmaceuticals in water as emerging pollutants for river health: A critical review under Indian conditions. Ecotoxicology and Environmental Safety, 2022, 247, 114220. [CrossRef]
- Grassi, M., Kaykioglu, G., Belgiorno, V., Lofrano, G. 2012. Removal of Emerging Contaminants from Water and Wastewater by Adsorption Process. Emerging Compounds Removal from Wastewater. SpringerBriefs in Molecular Science, 2012, 15-37. [CrossRef]
- Gadipelly, C., Pérez-González, A., Yadav, G.D., Ortiz, I., Ibáñez, R., Rathod, V.K., Marathe, K.V., Pharmaceutical industry wastewater: review of the technologies for water treatment and reuse. Industrial & Engineering Chemistry Research, 2014, 53(29). 11571-11592.
- Homem, V., Santos, L. Degradation and removal methods of antibiotics from aqueous matrices – A review. Journal of Environmental Management, 2011, 92. 10. 2304-2347. [CrossRef]
- Grassi, M., Kaykioglu, G., Belgiorno, V., Lofrano, G., Removal of Emerging Contaminants from Water and Wastewater by Adsorption Process. Emerging Compounds Removal from Wastewater, 2012, 15-37. [CrossRef]
- Gadipelly, C., Pérez-González, A., Yadav, G.D., Ortiz, I., Ibáñez, R., Rathod, V.K., Marathe, K.V., Pharmaceutical industry wastewater: review of the technologies for water treatment and reuse, Industrial & Engineering Chemistry Research, 2014, 53(29). 11571-11592.
- Homem, V., Santos, L., Degradation and removal methods of antibiotics from aqueous matrices – A review. Journal of Environmental Management, 2011, 92. 10. 2304-2347. [CrossRef]
- N’diaye, A.D., Kankou, M.S., Modeling of adsorption isotherms of pharmaceutical products onto various adsorbents: A Short Review. Journal of Materials and Environmental Science, 2020, 11(8). 1264-1276.
- Al-Khateeb, L. A., Almotiry, S., Salam, M. A., Adsorption of pharmaceutical pollutants onto graphene nanoplatelets. Chemical Engineering Journal, 2014, 248, 191-199. [CrossRef]
- Zielinska, I., Polak, D., Szwast, M., Analysis of the adsorption of selected pharmaceuticals on a composite material PEBAX/GO, Journal of Water Process Engineering, 2021, 44, 1-7. [CrossRef]
- Kim, H., Hwang, Y. S., & Sharma, V. K., Adsorption of antibiotics and iopromide onto single-walled and multi-walled carbon nanotubes, Chemical engineering journal, 2014, 255, 23-27. [CrossRef]
- Zhu, X., He, M., Sun, Y., Xu, Z., Wan, Z., Hou, D., Tsang, D. C., Insights into the adsorption of pharmaceuticals and personal care products (PPCPs) on biochar and activated carbon with the aid of machine learning. Journal of Hazardous Materials, 2022, 423, 127060. [CrossRef]
- Crini, G., Non-conventional low-cost adsorbents for dye removal: A review. Bioresource Technology, 2006, 97. 9. 1061-1085. [CrossRef]
- de Andrade, J. R., Oliveira, M. F., da Silva, M. G. C. , Vieira, M. G. A., Adsorption of Pharmaceuticals from Water and Wastewater Using Nonconventional Low-Cost Materials: A Review. Industrial & Engineering Chemistry Research, 2018, 57. 9. 3103–3127. [CrossRef]
- Song X., Cao, Y., Bu, X., Luo, X. Porous vaterite and cubic calcite aggregated calcium carbonate obtained from steamed ammonia liquid waste for Cu2+ heavy metal ions removal by adsorption process. Applied Surface Science. 2021, 536. 147958. [CrossRef]
- Shamsuri, A. A., & Sumadin, Z. A., Influence of hydrophobic and hydrophilic mineral fillers on processing, tensile and impact properties of LDPE/KCF biocomposites. Composites Communications, 2018, 9, 65-69. [CrossRef]
- Wszelaka-Rylik, M., Piotrowska, K., Gierycz, P., Simulation, aggregation and thermal analysis of nanostructured calcite obtained in a controlled multiphase process. Journal of Thermal Analysis and Calorimetry, 2015, 119, 1323–1338. [CrossRef]
- Schlomach, J., Quarch, K., Kind, M. 2006. Investigation of precipitation of calcium carbonate at high supersaturations. Chemical Engineering&Technology, 2006, 29. 2. 215–220. DOI : 10.1002/ceat.200500390.
- Rehage, H., Orthey, J., Kind, M., On the complete similitude of technical precipitation. Part II: Stirred-tank reactors. Chemical Engineering Journal, 2021, 426. 131788. DOI : 10.1016/j.cej.2021.131788.
- Jahani, D., Nazari, A., Ghourbanpour, J. Ameli, A., Polyvinyl alcohol/calcium carbonate nanocomposites as efficient and cost-effective cationic dye adsorbents. Polymers, 2020, 12(10), 2179. [CrossRef]
- Zhou, L., Peng, T., Sun, H., Guo, X., Fu, D., The characterization and amoxicillin adsorption activity of mesopore CaCO3 microparticles prepared using rape flower pollen. Minerals, 2019, 9(4), 254. [CrossRef]
- Hui, T. S., Ibrahim, N. A., Zhi, L. L., Zaini, M. A. A., Endocrine-disrupting chemical removal by carbon nanocomposites. Handbook of Advanced Approaches Towards Pollution Prevention and Control, 2021, 45-74. [CrossRef]
- Wang, C., He, C., Tong, Z., Liu, X., Ren, B., Zeng, F., Combination of adsorption by porous CaCO3 microparticles and encapsulation by polyelectrolyte multilayer films for sustained drug delivery. International journal of pharmaceutics, 2006, 308(1-2), 160-167. [CrossRef]
- Blanco, J. F., Sublet, J., Nguyen, Q. T., Schaetzel, P., Formation and morphology studies of different polysulfones-based membranes made by wet phase inversion process. Journal of membrane science, 2006, 283(1-2), 27-37.
- Kędra-Królik, K., Gierycz P., Obtaining calcium carbonate in a multiphase system by the use of new rotating disc precipitation reactor. Journal of Thermal Analysis and Calorimetry, 2006, 83. 579-582. [CrossRef]
- Ruthven, D. M., Adsorption (Chemical Engineering). 2003. Encyclopedia of Physical Science and Technology (Third Edition). Academic Press. 251 – 271. [CrossRef]
- Chang, Q., Chapter 10 - Surface of Solids. Colloid and Interface Chemistry for Water Quality Control. Academic Press, 2016, 175 – 225. [CrossRef]
- Oba, S. N., Ighalo, J. O., Aniagor, C. O., & Igwegbe, C. A. Removal of ibuprofen from aqueous media by adsorption: A comprehensive review. Science of The Total Environment, 2021, 780, 146608. doi:10.1016/j.scitotenv.2021.1466.
- Zhou, Y., Liu, X., Xiang, Y., Wang, P., Zhang, J., Zhang, F., Wei, J., Luo, L., Lei, M., Tang, L., Modification of biochar derived from sawdust and its application in removal of tetracycline and copper from aqueous solution: adsorption mechanism and modelling, Bioresource Technology, 2017, 245, 266 - 273. doi:10.1016/j.biortech.2017.08.178.
- Hu, S., Zhang, Y., Shen, G., Zhang, H., Yuan, Z., Zhang, W., Adsorption/desorption behaviour and mechanisms of sulfadiazine and sulfamethoxazole in agricultural soil systems. Soil and Tillage Research, 2019, 186, 233-241. [CrossRef]
- Zhang, L., Wang, Y., Jin, S., Lu, Q., Ji, J., Adsorption isotherm, kinetic and mechanism of expanded graphite for sulfadiazine antibiotics removal from aqueous solutions. Environmental technology, 2017, 38(20), 2629-2638. [CrossRef]
- Boxall, A. B., Blackwell, P., Cavallo, R., Kay, P., & Tolls, J., The sorption and transport of a sulphonamide antibiotic in soil systems. Toxicology letters, 2002, 131(1-2), 19-28. [CrossRef]
- Ersan, G., Apul, O. G., Perreault, F., & Karanfil, T., Adsorption of organic contaminants by graphene nanosheets: A review. Water Research, 2017, 126, 385-398. [CrossRef]
- Gao, Y., Li, Y., Zhang, L., Huang, H., Hu, J., Shah, S. M., Su, X., Adsorption and removal of tetracycline antibiotics from aqueous solution by graphene oxide. Journal of colloid and interface science, 2012, 368(1), 540-546. [CrossRef]
- M. Xiaoming et al, Ma, X., Li, L., Yang, L., Su, C., Wang, K., Yuan, S., Zhou, J., Adsorption of heavy metal ions using hierarchical CaCO3–maltose meso/macroporous hybrid materials: Adsorption isotherms and kinetic studies. Journal of hazardous materials, 2012, 209, 467-477. [CrossRef]
- Conde-Cid, M., Núñez-Delgado, A., Fernández-Sanjurjo, M. J., Álvarez-Rodríguez, E., Fernández-Calviño, D., & Arias-Estévez, M., Tetracycline and sulfonamide antibiotics in soils: presence, fate and environmental risks. Processes, 2020, 8(11), 1479. [CrossRef]
- Kuo, C. Y., Lin, H. N., Tsai, H. A., Wang, D. M., Lai, J. Y., Fabrication of a high hydrophobic PVDF membrane via nonsolvent induced phase separation. Desalination, 2008, 233(1-3), 40-47. DOI 10.1016/j.desal.2007.09.025.
- Li, Q., Xu, Z. L., & Liu, M., Preparation and characterization of PVDF microporous membrane with highly hydrophobic surface. Polymers for Advanced Technologies, 2011, 22(5), 520-531. [CrossRef]
- Wang, C., Piao, C., Zhai, X., Hickman, F. N., Li, J., Synthesis and characterization of hydrophobic calcium carbonate particles via a dodecanoic acid inducing process. Powder Technology, 2010, 198(1), 131-134. [CrossRef]
- Abedini, R., Mousavi, S. M., & Aminzadeh, R., A novel cellulose acetate (CA) membrane using TiO2 nanoparticles: Preparation, characterization and permeation study. Desalination, 2011, 277(1-3), 40-45. [CrossRef]
- Blanco, J. F., Sublet, J., Nguyen, Q. T., Schaetzel, P., Formation and morphology studies of different polysulfones-based membranes made by wet phase inversion process. Journal of membrane science, 2006, 283(1-2), 27-37. [CrossRef]
- Hu, S., Zhang, Y., Shen, G., Zhang, H., Yuan, Z., & Zhang, W. Adsorption/desorption behavior and mechanisms of sulfadiazine and sulfamethoxazole in agricultural soil systems. Soil and Tillage Research, 2019, 186, 233-241.
- Wang, S., Wang, H., Adsorption behavior of antibiotic in soil environment: a critical review. Frontiers of Environmental Science & Engineering, 2015, 9, 565-574. [CrossRef]
- Liu, S., Xu, W. H., Liu, Y. G., Tan, X. F., Zeng, G. M., Li, X., Cai, X. X., Facile synthesis of Cu (II) impregnated biochar with enhanced adsorption activity for the removal of doxycycline hydrochloride from water. Science of the Total Environment, 2017, 592, 546-553. [CrossRef]
- Schmitt, M. O., & Schneider, S., Spectroscopic investigation of complexation between various tetracyclines and Mg 2+ or Ca 2+. PhysChemComm, 2000, 3(9), 42-55. [CrossRef]
- Lotfi, R., Hayati, B., Rahimi, S., Shekarchi, A. A., Mahmoodi, N. M., Bagheri, A., Synthesis and characterization of PAMAM/SiO2 nanohybrid as a new promising adsorbent for pharmaceuticals. Microchemical Journal, 2019, 146, 1150-1159. [CrossRef]
- Balarak, D., Baniasadi, M., Removal of Ciprofloxacin from of pharmaceutical wastewater by adsorption on SiO2 nanoparticle. Journal of Pharmaceutical Research International, 2018, 25(6): 1-9.
- Ataee, R.A, Derakhshanpour, J., Mehrabi Tavana, A., Eydi, A., Antibacterial effect of calcium carbonate nanoparticles on Agrobacterium tumefaciens, Iranian Journal of Military Medicin, 2011, 13, 2, 65-70.
- Sabri, S., Najjar, A., Manawi, Y., Eltai, N.M., Al.-Thani, A., Atieh, M.A., Kochkodan, V., Antibacterial Properties of Polysulfone Membranes Blended with Arabic Gum, Membranes, 2019, 9(2), 29. [CrossRef]
- Prihandana, G.S., Sriani, T., Muthi’ah, A.D., Machmudah, A., Mahardika, M., Miki, N., Nanomaterials, 2022, 12(3), 388. [CrossRef]

| The concentration of calcium carbonate in suspension [%] | 0.05 | 0.1 | 0.5 | |
| Turbidity [NTU] | Initial | >1000 | >1000 | >1000 |
| After all stages | 2.97 | 3.31 | 4.16 | |
| Substance | Suspension concentration [%] |
Efficiency, η [%] |
Adsorbed mass, qI [mg/g] |
|---|---|---|---|
| Tetracycline | 0.05 0.10 0.50 |
1.07 ± 0.07 4.03 ± 0.03 10.01 ± 0.07 |
0.81 ± 0.01 1.52 ± 0.04 0.75 ± 0.04 |
| Sulfadiazine | 0.05 0.10 0.50 |
19.87 ± 0.21 17.66 ± 0.17 21.92 ± 0.19 |
15.40 ± 4.27 6.85 ± 1.96 1.69 ± 0.44 |
| Substance | Suspension concentration [%] | Adsorbed mass, |
| Tetracycline | 0.00 | 3477 ± 56 |
| 0.05 | 3742 ± 76 | |
| Sulfadiazine | 0.00 | 494 ± 61 |
| 0.05 | 954 ± 85 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




