Submitted:
04 March 2024
Posted:
05 March 2024
You are already at the latest version
Abstract
Keywords:
Introduction
Materials and Methods
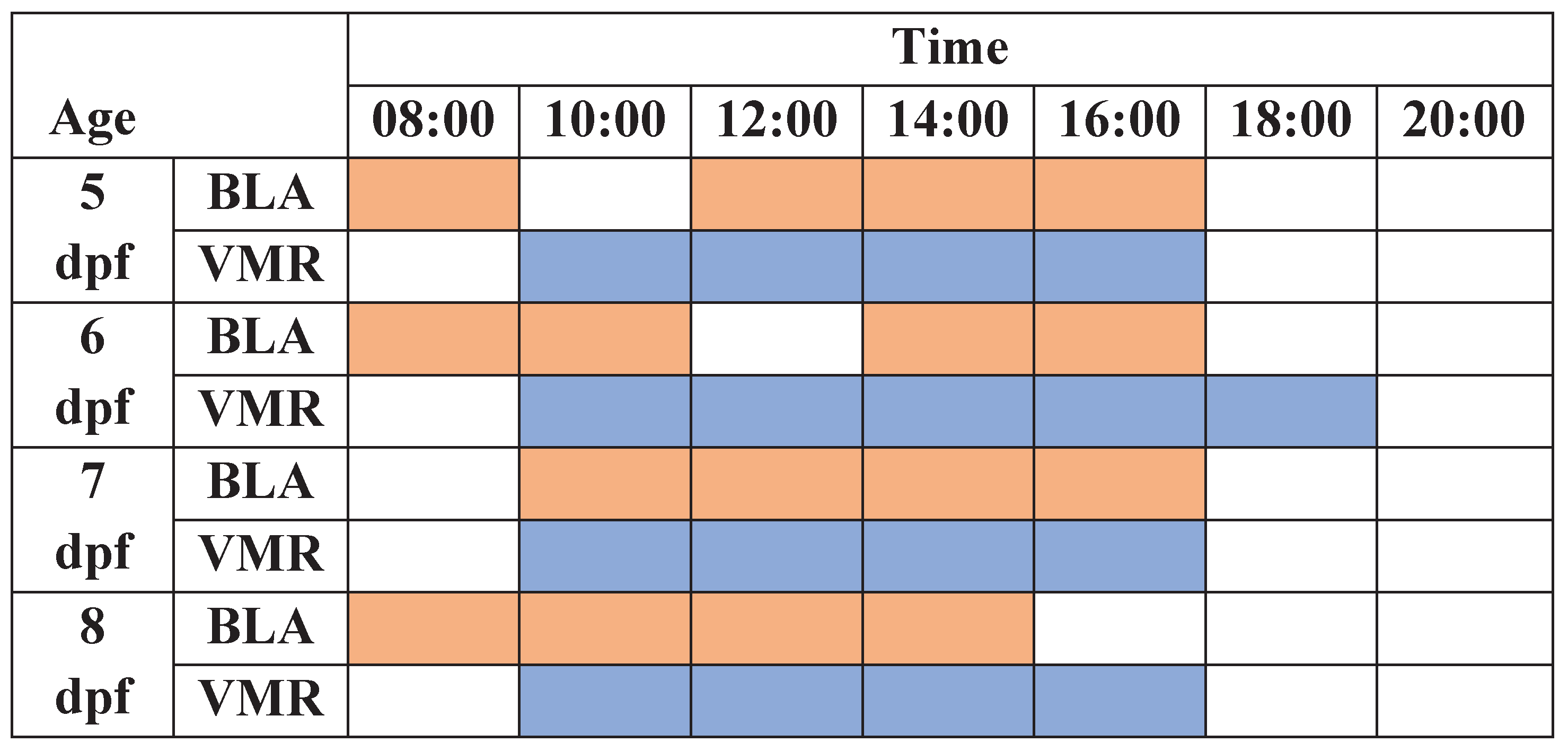
Behavioral analysis
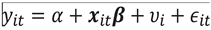
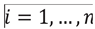 and for each i,
and for each i, 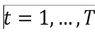 , of which periods were actually observed
, of which periods were actually observed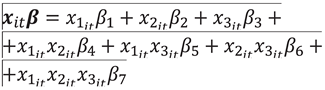
Results
Discussion
Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Ilie, O.D.; Duta, R.; Jijie, R.; Nita, I.B.; Nicoara, M.; Faggio, C.; Dobrin, R.; Mavroudis, I.; Ciobica, A.; Doroftei, B. Assessing Anti-Social and Aggressive Behavior in a Zebrafish (Danio rerio) Model of Parkinson’s Disease Chronically Exposed to Rotenone. Brain Sci. 2022, 12, 1–12. [Google Scholar] [CrossRef]
- Rashidian, G.; Mohammadi-Aloucheh, R.; Hosseinzadeh-Otaghvari, F.; Chupani, L.; Stejskal, V.; Samadikhah, H.; Zamanlui, S.; Multisanti, C.R.; Faggio, C. Long-term exposure to small-sized silica nanoparticles (SiO2-NPs) induces oxidative stress and impairs reproductive performance in adult zebrafish (Danio rerio). Comp. Biochem. Physiol. Part C Toxicol. Pharmacol. 2023, 273, 109715. [Google Scholar] [CrossRef]
- Ricarte, M.; Prats, E.; Montemurro, N.; Bedrossiantz, J.; Bellot, M.; Gómez-Canela, C.; Raldúa, D. Environmental concentrations of tire rubber-derived 6PPD-quinone alter CNS function in zebrafish larvae. Sci. Total Environ. 2023, 896, 165240. [Google Scholar] [CrossRef] [PubMed]
- Basnet, R.M.; Zizioli, D.; Taweedet, S.; Finazzi, D.; Memo, M. Zebrafish larvae as a behavioral model in neuropharmacology. Biomedicines 2019, 7. [Google Scholar] [CrossRef] [PubMed]
- Strähle, U.; Scholz, S.; Geisler, R.; Greiner, P.; Hollert, H.; Rastegar, S.; Schumacher, A.; Selderslaghs, I.; Weiss, C.; Witters, H.; et al. Zebrafish embryos as an alternative to animal experiments-A commentary on the definition of the onset of protected life stages in animal welfare regulations. Reprod. Toxicol. 2012, 33, 128–132. [Google Scholar] [CrossRef] [PubMed]
- Hill, B.N.; Coldsnow, K.D.; Hunter, D.L.; Hedge, J.M.; Korest, D.; Jarema, K.A.; Padilla, S. Assessment of Larval Zebrafish Locomotor Activity for Developmental Neurotoxicity Screening. Neuromethods 2021, 172, 327–351. [Google Scholar] [CrossRef]
- Faria, M.; Bellot, M.; Bedrossiantz, J.; Ramírez, J.R.R.; Prats, E.; Garcia-Reyero, N.; Gomez-Canela, C.; Mestres, J.; Rovira, X.; Barata, C.; et al. Environmental levels of carbaryl impair zebrafish larvae behaviour: The potential role of ADRA2B and HTR2B. J. Hazard. Mater. 2022, 431. [Google Scholar] [CrossRef] [PubMed]
- Faria, M.; Prats, E.; Rosas Ramírez, J.R.; Bellot, M.; Bedrossiantz, J.; Pagano, M.; Valls, A.; Gomez-Canela, C.; Porta, J.M.; Mestres, J.; et al. Androgenic activation, impairment of the monoaminergic system and altered behavior in zebrafish larvae exposed to environmental concentrations of fenitrothion. Sci. Total Environ. 2021, 775, 145671. [Google Scholar] [CrossRef]
- Hill, B.N.; Britton, K.N.; Hunter, D.L.; Olin, J.K.; Lowery, M.; Hedge, J.M.; Knapp, B.R.; Jarema, K.A.; Rowson, Z.; Padilla, S. Inconsistencies in variable reporting and methods in larval zebrafish behavioral assays. Neurotoxicol. Teratol. 2023, 96, 107163. [Google Scholar] [CrossRef]
- Cassar, S.; Adatto, I.; Freeman, J.L.; Gamse, J.T.; Iturria, I.; Lawrence, C.; Muriana, A.; Peterson, R.T.; Van Cruchten, S.; Zon, L.I. Use of Zebrafish in Drug Discovery Toxicology. Chem. Res. Toxicol. 2020, 33, 95–118. [Google Scholar] [CrossRef]
- Rosa, J.G.S.; Lima, C.; Lopes-Ferreira, M. Zebrafish Larvae Behavior Models as a Tool for Drug Screenings and Pre-Clinical Trials: A Review. Int. J. Mol. Sci. 2022, 23. [Google Scholar] [CrossRef]
- Padilla, S.; Hunter, D.L.; Padnos, B.; Frady, S.; MacPhail, R.C. Assessing locomotor activity in larval zebrafish: Influence of extrinsic and intrinsic variables. Neurotoxicol. Teratol. 2011, 33, 624–630. [Google Scholar] [CrossRef]
- Fero, K.; Yokogawa, T.; Burgess, H.A. The behavioral repertoire of larval zebrafish. In Zebrafish models in neurobehavioral research; Springer, 2011; pp. 249–291. [Google Scholar]
- Tian, N.; Copenhagen, D.R. Visual stimulation is required for refinement on ON and OFF pathways in postnatal retina. Neuron 2003, 39, 85–96. [Google Scholar] [CrossRef]
- Tufi, S.; Leonards, P.; Lamoree, M.; De Boer, J.; Legler, J.; Legradi, J. Changes in Neurotransmitter Profiles during Early Zebrafish (Danio rerio) Development and after Pesticide Exposure. Environ. Sci. Technol. 2016, 50, 3222–3230. [Google Scholar] [CrossRef]
- Wolter, M.E.; Svoboda, K.R. Doing the locomotion: Insights and potential pitfalls associated with using locomotor activity as a readout of the circadian rhythm in larval zebrafish. J. Neurosci. Methods 2020, 330. [Google Scholar] [CrossRef] [PubMed]
- Poupard, G.; André, M.; Durliat, M.; Ballagny, C.; Boeuf, G.; Babin, P.J. Apolipoprotein E gene expression correlates with endogenous lipid nutrition and yolk syncytial layer lipoprotein synthesis during fish development. Cell Tissue Res. 2000, 300, 251–261. [Google Scholar] [CrossRef] [PubMed]
- Kimmel, C.B.; Ballard, W.W.; Kimmel, S.R.; Ullmann, B.; Schilling, T.F. Stages of embryonic development of the zebrafish. Dev. Dyn. 1995, 203, 253–310. [Google Scholar] [CrossRef] [PubMed]
- Bedrossiantz, J.; Faria, M.; Prats, E.; Barata, C.; Cachot, J.; Raldúa, D. Heart rate and behavioral responses in three phylogenetically distant aquatic model organisms exposed to environmental concentrations of carbaryl and fenitrothion. Sci. Total Environ. 2023, 865, 161268. [Google Scholar] [CrossRef] [PubMed]
- Cameron, A.; Trivedi, P.K. Microeconometrics Using Stata, 2nd ed.; Stata Press: Texas, 2022; ISBN 978-1-59718-359-8. [Google Scholar]
- Armitage, P. Statistical methods in medical research; John Wiley and Sons: New York, NY, 1971. [Google Scholar]
- StataCorp, L. Stata statistical software: Release 17. Coll. Stn. StataCorp LP 2021.
- de Esch, C.; van der Linde, H.; Slieker, R.; Willemsen, R.; Wolterbeek, A.; Woutersen, R.; De Groot, D. Locomotor activity assay in zebrafish larvae: Influence of age, strain and ethanol. Neurotoxicol. Teratol. 2012, 34, 425–433. [Google Scholar] [CrossRef] [PubMed]
- Maximino, C.; Meinerz, D.L.; Fontana, B.D.; Mezzomo, N.J.; Stefanello, F. V.; de, S. Prestes, A.; Batista, C.B.; Rubin, M.A.; Barbosa, N. V.; Rocha, J.B.T.; et al. Extending the analysis of zebrafish behavioral endophenotypes for modeling psychiatric disorders: Fear conditioning to conspecific alarm response. Behav. Processes 2018, 149, 35–42. [Google Scholar] [CrossRef] [PubMed]
- Lima, M.G.; Silva, R.X.d.C.; Silva, S. de N. dos S.; Rodrigues, L. do S. dos S.; Oliveira, K.R.H.M.; Batista, E. de J.O.; Maximino, C.; Herculano, A.M. Time-dependent sensitization of stress responses in zebrafish: A putative model for post-traumatic stress disorder. Behav. Processes 2016, 128, 70–82. [Google Scholar] [CrossRef] [PubMed]
- Selderslaghs, I.W.T.; Hooyberghs, J.; De Coen, W.; Witters, H.E. Locomotor activity in zebrafish embryos: A new method to assess developmental neurotoxicity. Neurotoxicol. Teratol. 2010, 32, 460–471. [Google Scholar] [CrossRef] [PubMed]
- Ingebretson, J.J.; Masino, M.A. Quantification of locomotor activity in larval Zebrafish: Considerations for the design of high-throughput behavioral studies. Front. Neural Circuits 2013, 7, 1–9. [Google Scholar] [CrossRef] [PubMed]
- MacPhail, R.C.; Brooks, J.; Hunter, D.L.; Padnos, B.; Irons, T.D.; Padilla, S. Locomotion in larval zebrafish: Influence of time of day, lighting and ethanol. Neurotoxicology 2009, 30, 52–58. [Google Scholar] [CrossRef] [PubMed]
- Kristofco, L.A.; Cruz, L.C.; Haddad, S.P.; Behra, M.L.; Chambliss, C.K.; Brooks, B.W. Age matters: Developmental stage of Danio rerio larvae influences photomotor response thresholds to diazinion or diphenhydramine. Aquat. Toxicol. 2016, 170, 344–354. [Google Scholar] [CrossRef] [PubMed]
- Fernandes, A.M.; Fero, K.; Arrenberg, A.B.; Bergeron, S.A.; Driever, W.; Burgess, H.A. Deep brain photoreceptors control light-seeking behavior in zebrafish larvae. Curr. Biol. 2012, 22, 2042–2047. [Google Scholar] [CrossRef]
- Haigis, A.C.; Ottermanns, R.; Schiwy, A.; Hollert, H.; Legradi, J. Getting more out of the zebrafish light dark transition test. Chemosphere 2022, 295. [Google Scholar] [CrossRef]
- Ogungbemi, A.; Leuthold, D.; Scholz, S.; Küster, E. Hypo- or hyperactivity of zebrafish embryos provoked by neuroactive substances: a review on how experimental parameters impact the predictability of behavior changes. Environ. Sci. Eur. 2019, 31. [Google Scholar] [CrossRef]
- Rock, S.; Rodenburg, F.; Schaaf, M.J.M.; Tudorache, C. Detailed Analysis of Zebrafish Larval Behaviour in the Light Dark Challenge Assay Shows That Diel Hatching Time Determines Individual Variation. Front. Physiol. 2022, 13, 1–11. [Google Scholar] [CrossRef]
- Fraser, T.W.K.; Khezri, A.; Jusdado, J.G.H.; Lewandowska-Sabat, A.M.; Henry, T.; Ropstad, E. Toxicant induced behavioural aberrations in larval zebrafish are dependent on minor methodological alterations. Toxicol. Lett. 2017, 276, 62–68. [Google Scholar] [CrossRef]
- Fitzgerald, J.A.; Kirla, K.T.; Zinner, C.P.; vom Berg, C.M. Emergence of consistent intra-individual locomotor patterns during zebrafish development. Sci. Rep. 2019, 9, 1–14. [Google Scholar] [CrossRef]
- Nüßer, L.K.; Skulovich, O.; Hartmann, S.; Seiler, T.B.; Cofalla, C.; Schuettrumpf, H.; Hollert, H.; Salomons, E.; Ostfeld, A. A sensitive biomarker for the detection of aquatic contamination based on behavioral assays using zebrafish larvae. Ecotoxicol. Environ. Saf. 2016, 133, 271–280. [Google Scholar] [CrossRef] [PubMed]
- Maeda, H.; Fukushima, N.; Hasumi, A. Standardized method for the assessment of behavioral responses of zebrafish larvae. Biomedicines 2021, 9, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Faria, M.; Prats, E.; Novoa-Luna, K.A.; Bedrossiantz, J.; Gómez-Canela, C.; Gómez-Oliván, L.M.; Raldúa, D. Development of a vibrational startle response assay for screening environmental pollutants and drugs impairing predator avoidance. Sci. Total Environ. 2019, 650, 87–96. [Google Scholar] [CrossRef] [PubMed]
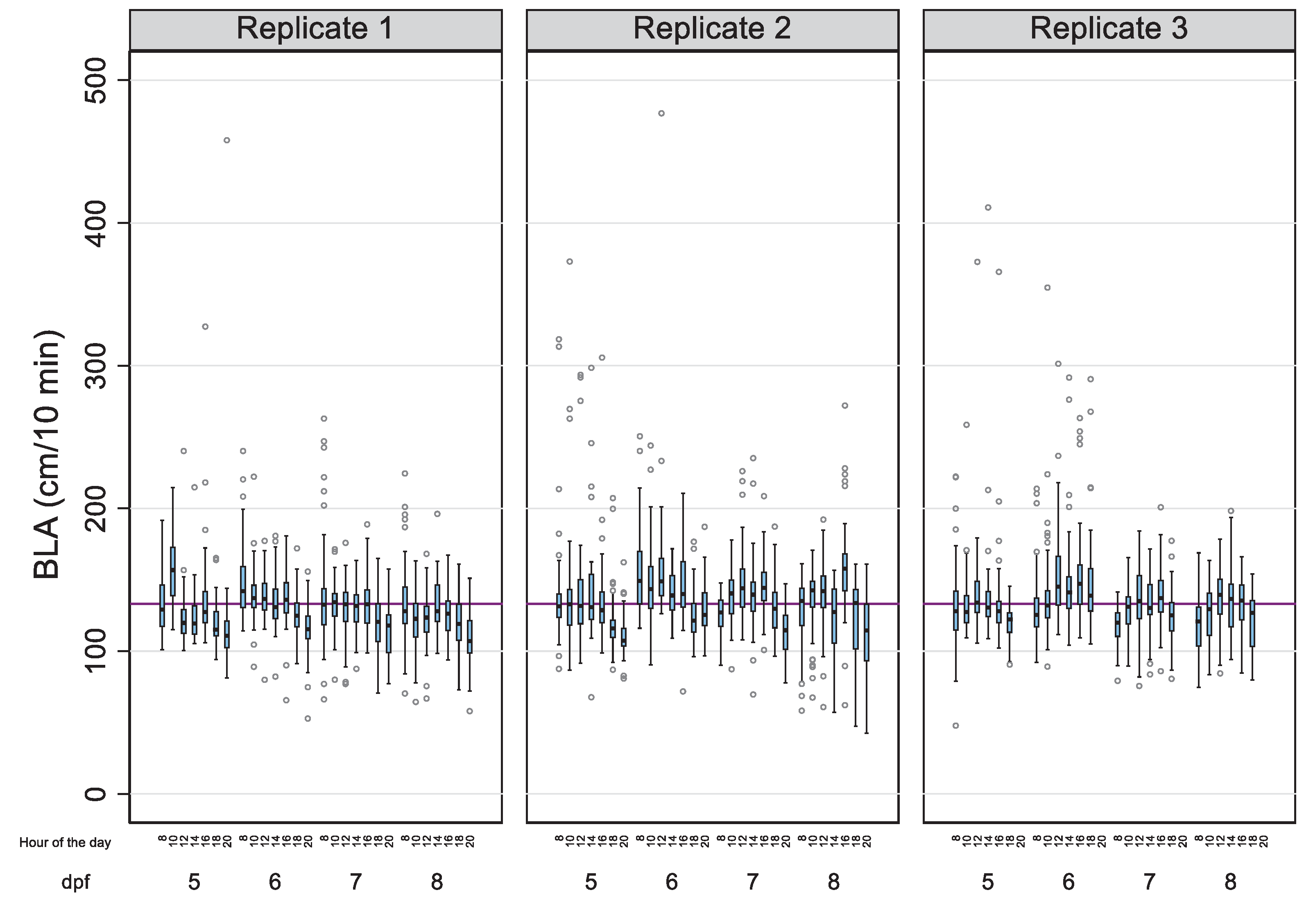
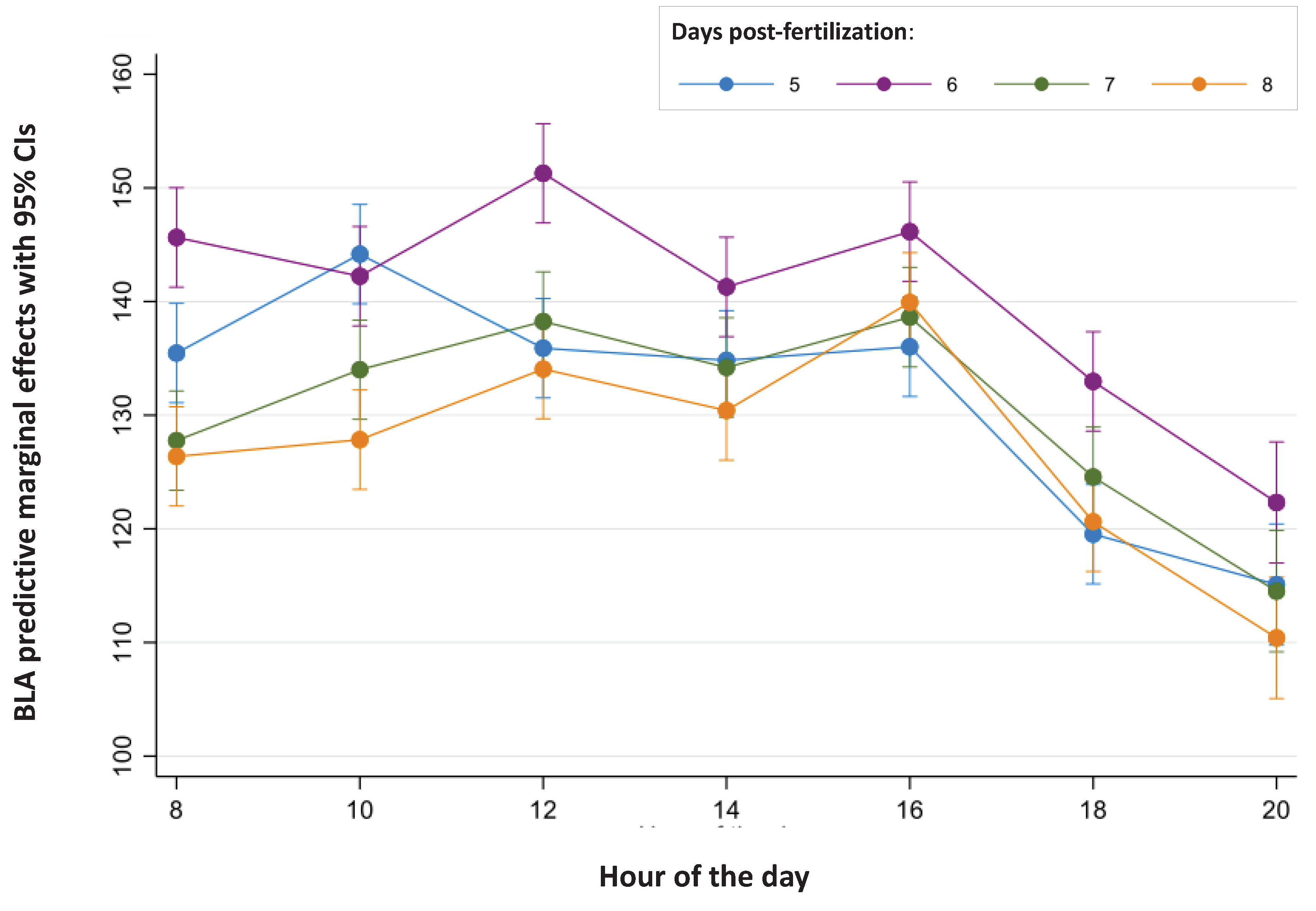
| N | n | SD | SDBetween | SDWithin | Rho | 1-Rho | ||
|---|---|---|---|---|---|---|---|---|
| 3,814 | 143 | 133.14 | 28.99 | 11.75 | 26.51 | 16.43% | 83.57% | |
| Day (age) | ||||||||
| 5 | 954 | 143 | 132.38 | 34.42 | 22.36 | 26.17 | 42.19% | 57.81% |
| 6 | 954 | 143 | 141.15 | 30.20 | 19.28 | 23.55 | 40.13% | 59.87% |
| 7 | 952 | 143 | 131.09 | 22.99 | 14.10 | 18.21 | 37.48% | 62.52% |
| 8 | 954 | 143 | 127.91 | 25.31 | 16.37 | 19.31 | 41.82% | 58.18% |
| Time (hours) | ||||||||
| 08:00 | 572 | 143 | 133.81 | 30.30 | 18.07 | 24.36 | 35.51% | 64.49% |
| 10:00 | 572 | 143 | 137.06 | 27.94 | 16.83 | 22.34 | 36.20% | 63.80% |
| 12:00 | 572 | 143 | 139.87 | 31.68 | 19.33 | 25.14 | 37.15% | 62.85% |
| 14:00 | 572 | 143 | 135.18 | 26.78 | 15.03 | 22.19 | 31.43% | 68.57% |
| 16:00 | 571 | 143 | 140.22 | 28.60 | 17.12 | 22.97 | 35.71% | 64.29% |
| 18:00 | 571 | 143 | 124.46 | 22.63 | 12.67 | 18.77 | 31.29% | 68.71% |
| 20:00 | 384 | 96 | 115.58 | 26.32 | 14.38 | 22.09 | 29.76% | 70.24% |
| Experiment | ||||||||
| 1 | 1,344 | 48 | 129.17 | 25.77 | 9.89 | 23.84 | 14.67% | 85.33% |
| 2 | 1,342 | 48 | 135.80 | 31.14 | 13.02 | 28.36 | 17.40% | 82.60% |
| 3 | 1,128 | 47 | 134.69 | 29.45 | 11.28 | 27.25 | 14.64% | 85.36% |
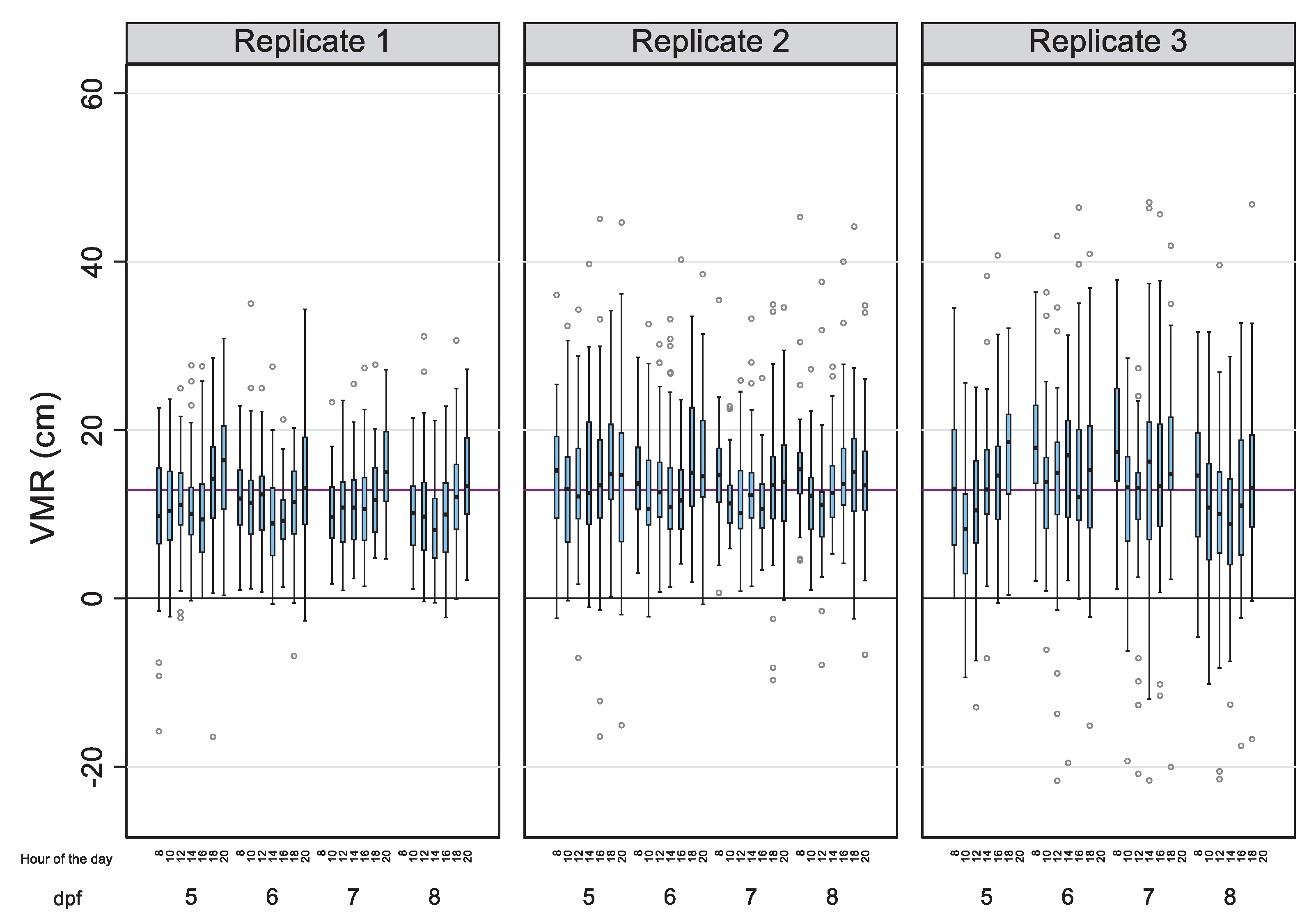
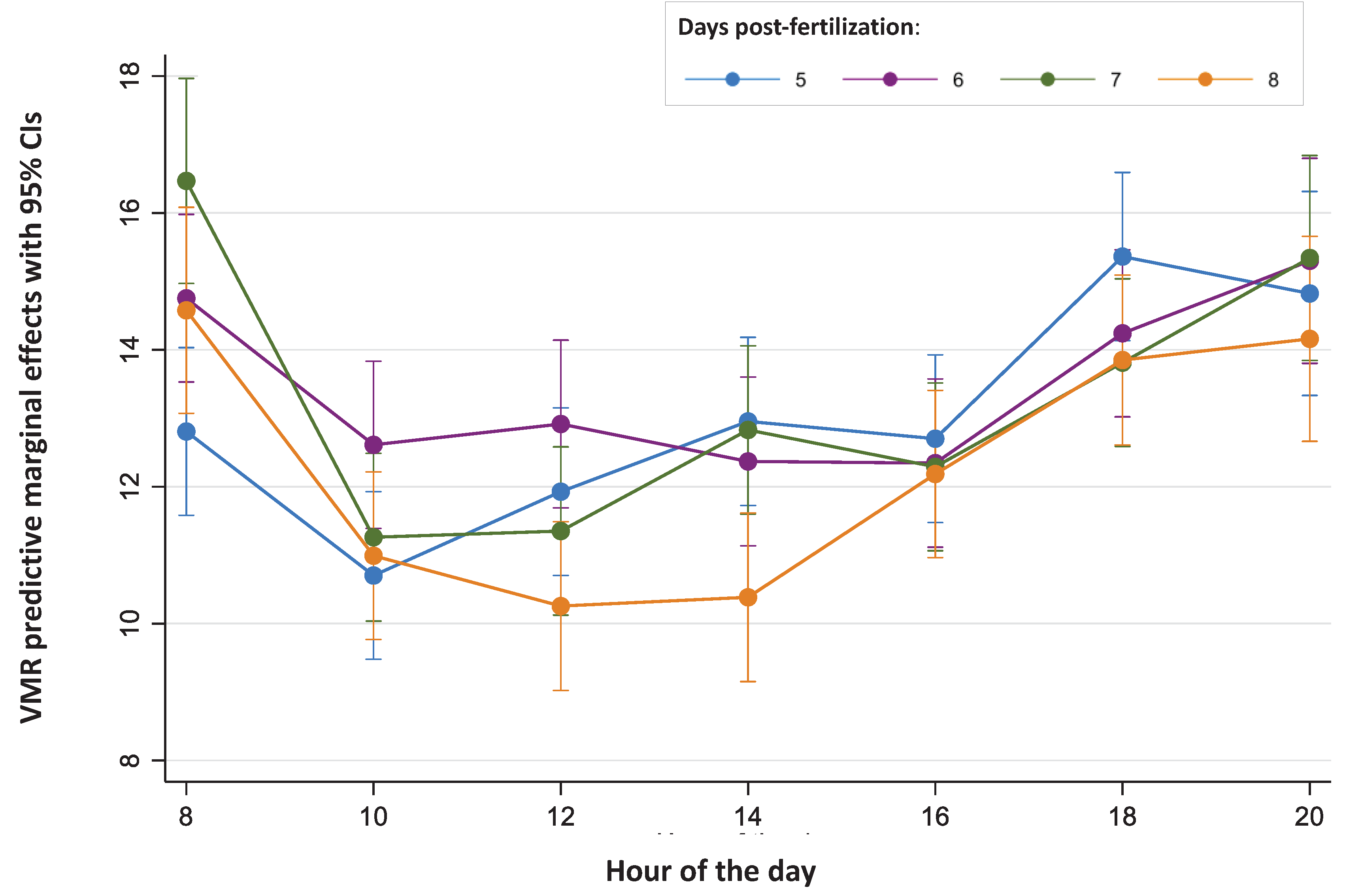
| N | n | SDBetween | SDWithin | Rho | 1-Rho | |||
|---|---|---|---|---|---|---|---|---|
| 3,699 | 143 | 12.79 | 8.56 | 3.65 | 7.76 | 18.14% | 81.86% | |
| Day (age) | ||||||||
| 5 | 951 | 143 | 12.90 | 8.75 | 5.14 | 7.13 | 34.14% | 65.86% |
| 6 | 950 | 143 | 13.33 | 8.54 | 5.66 | 6.50 | 43.10% | 56.90% |
| 7 | 900 | 143 | 12.96 | 8.05 | 5.44 | 6.11 | 44.26% | 55.74% |
| 8 | 898 | 143 | 11.92 | 8.82 | 6.27 | 6.39 | 49.09% | 50.91% |
| Time (hours) | ||||||||
| 08:00 | 473 | 143 | 14.45 | 7.67 | 4.96 | 6.05 | 40.16% | 59.84% |
| 10:00 | 570 | 143 | 11.28 | 7.46 | 4.42 | 6.02 | 35.04% | 64.96% |
| 12:00 | 568 | 143 | 11.59 | 8.28 | 4.45 | 6.98 | 28.85% | 71.15% |
| 14:00 | 567 | 143 | 11.94 | 9.34 | 5.22 | 7.76 | 31.20% | 68.80% |
| 16:00 | 571 | 143 | 12.13 | 8.61 | 5.15 | 6.94 | 35.51% | 64.49% |
| 18:00 | 569 | 143 | 14.18 | 9.60 | 5.61 | 7.80 | 34.12% | 65.88% |
| 20:00 | 381 | 96 | 14.91 | 7.57 | 5.03 | 5.68 | 43.91% | 56.09% |
| Experiment | ||||||||
| 1 | 1,244 | 48 | 11.41 | 6.21 | 2.83 | 5.55 | 20.61% | 79.39% |
| 2 | 1,341 | 48 | 13.75 | 7.29 | 3.41 | 6.46 | 21.73% | 78.27% |
| 3 | 1,114 | 47 | 13.17 | 11.53 | 4.26 | 10.74 | 13.57% | 86.43% |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




