Submitted:
18 February 2024
Posted:
19 February 2024
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Overview of Pharmacodynamics and Pharmacokinetics in Comparison with Other ß-Blockers
- A negative bathmotropic effect (by blocking INa channels responsible for the rapid depolarization phase of the cardiac action potential).
- A negative dromotropic effect (it decreases the speed of impulse conduction through the atrioventricular node by blocking especially IKs channels).
- A negative chronotropic effect (blocking the ß-1 receptors in the sinoatrial node and modulating the activity of various ion channels, including If- funny current).
- A negative inotropic effect: due to its blockade of the ß-1 receptors in the myocardium and in this way blocking the release of calcium, necessary for cardiomyocyte contraction.
- An antiarrhythmic effect: due to its suppression of triggered activity and prolongation of refractory period by blocking IKs channels. In this way, it may help prevent the re-entry of electrical impulses that can lead to certain types of arrhythmias such as AF, AFL, atrioventricular re-entry tachycardia, or atrioventricular nodal reentry tachycardia [12].
| Property | Landiolol[13] | Esmolol[13] | Metoprolol[14] | Nebivolol[15] | Bisoprolol[15] | Atenolol[16] | Carvedilol[17] | Propranolol[18] |
| Drug class | Ultra-short-acting selective ß-1 blocker | Short-acting selective ß-1 blocker | Selective ß -1 blocker | Highly cardio selective ß -1 blocker with vasodilator properties | Highly cardio-selective ß -1 blocker | Cardio selective ß -1 blocker | Non-selective ß blocker with alpha-1 blocking activity | Non-selective ß blocker |
| Half-life | Very short (about 4 minutes) | Very short (about 9 minutes) | 3-7 hours | 10-12 hours |
10-12 hours |
6-7 hours |
7-10 hours |
4-6 hours |
| Pharmacokinetics | Rapid onset and offset of action | Rapid onset and offset of action | Rapidly and completely absorbed | Absorbed rapidly and extensively metabolized | Slowly and completely absorbed | Absorbed slowly but almost completely | Extensive l y metabolized | Rapidly and completely absorbed |
| Pharmacodynamics | Selective ß1-blocker, short-acting |
Selective ß1-blocker, short-acting |
Selective ß1-blocker, long-acting |
ß1-blocker with vasodilator effects | Selective ß1-blocker, long-acting |
Selective ß1-blocker, long-acting |
Non-selective ß-blocker with ß1-blockade |
Non-selective ß-blocker, membrane-stabilizing activity |
| Cardio selectivity | Highly | Highly | Highly | Highly with vasodilator properties | Highly | Highly | Non-selective | Non-selective |
| Dose Range | 1-40 μg/kg/min | 50-300 μg/kg/min |
25-200 mg/day |
2.5-10 mg/day |
2.5-10 mg/day |
25-100 mg/day |
6.25-25 mg/day |
20-320 mg/day |
| Vasodilator effects | Minimal | Minimal | Minimal | Significant | Minimal | Minimal | Strong | Minimal |
| Indications | Rapid heart rate control in critical care settings | Acute heart rate control, intraoperative and postoperative tachycardia | Hypertension, angina, heart failure, post-myocardial infarction | Hypertension, angina, heart failure | Hypertension, angina, heart failure | Hypertension, angina, arrhythmias | Hypertension, angina, heart failure | Hypertension, angina, arrhythmias |
| Adverse effects | Rarely Hypotension, bradycardia, | Hypotension, bradycardia, bronchospasm | Hypotension, bradycardia, fatigue, dizziness | Hypotension, bradycardia, fatigue, dizziness, headache | Hypotension, bradycardia, fatigue, dizziness | Hypotension, bradycardia, fatigue, dizziness | Hypotension, bradycardia, fatigue, dizziness, heart block | Hypotension, bradycardia, fatigue, dizziness, bronchospasm |
| Contraindication | Severe bradycardia, heart block | Heart block, severe bradycardia, heart failure, asthma | Bradycardia, heart block, heart failure, hypotension | Severe bradycardia, heart block, hepatic impairment | Heart block, severe bradycardia, heart failure, asthma | Bradycar dia, heart block, heart failure, asthma | Asthma, heart block, severe bradycard ia | Asthma, heart block, bradycardia |
3. The Use of Landiolol in Clinical Practice
3.1. Postoperative Atrial Fibrillation Prevention and Treatment in Cardiac Surgery
3.2. Postoperative Atrial Fibrillation Prevention and Treatment in Non-Cardiac Surgery
| Study name | Type of surgery | Number of patients | Design study | Outcomes |
|---|---|---|---|---|
| Nojiri et al.[33] | Lung surgery | 30 |
Study group (n=15) – landiolol 5 mcg/kg/min or 10 mcg/kg/min Control group (n=15) – 0.25 mg digoxin and 5 mg verapamil |
Rate of conversion to SR: Study group: at 2 h: 8/15 (53%) at 12 h 11/15 (73%) Control group: at 2 h: 3/15 (20%) at 12 h 8/15 (53%) Time of conversion to SR: Study group: 8.1± 11.0 h Control group: 23.0 ± 26.0 h |
| Niwa et al.[39] | Esophagectomy | 24 |
Study group (n=11) – landiolol 6.5 ± 3.4 mcg/kg/min, increased to 7.7 ± 4.4 mcg/kg/min Control group (n=13) – digoxin and calcium channel blockers. |
Rate of conversion to SR: Study group: at 2 h 5/8 (62.5%) at 12 h 8/8 (100%) Control group: at 2 h: at 2 h 1/13 (7.7%) at 12 h 7/13 (53.8%) Time of conversion to SR: Study group: 3.6 6 ±6.6 h Control group: 23.3 ± 5.2 h |
| Mori et al.[38] | Esophagectomy | 74 | Study group (n=13) – landiolol 0.01 mg/kg/min increased to 0.04 mg/kg/min | Rate of conversion to SR: 10/13 (77%) |
| Nakano et al.[32] | Pulmonary resection | 25 | Study group (n=25) - landiolol 5–10 mcg/kg/min or o 1–5 mcg/kg/min |
Rate of conversion to SR: 14/25 (56%) – at 1 h 4/14 (28.6%) and at 12 h 5/14 (35.7%) HR control: from 135 ± 24 bpm to 85 ± 19 bpm |
3.3. Treatment of Atrial Fibrillation in Patients with Cardiac Dysfunction
3.4. Treatment of Sepsis-Related Atrial Fibrillation
4. Challenges and Perspectives
| Hypotension [19,28,47,48,55,56] | Dyspnea [55] |
| Bradycardia [48,55,57] | Asthma [46] |
| Aggravation of cardiac failure [48,50,55] | Hypokalemia [54] |
| Ventricular tachycardia [55] | Hypoglycemia [54] |
| Complete atrioventricular block [48,53] | Hepatic enzyme increased [53,54] |
| Cardiorespiratory arrest [50,55] | Gamma-glutamyl transferase [54] |
| Cardiogenic shock [53,55] | Blood uric acid increased [54] |
| Embolic stroke [46] | Blood alkaline phosphatase increased [54] |
| Pneumonia [33,55] | Increased serum creatinine [48,56] |
| Respiratory failure [33,55] | Vomiting and nausea [46] |
5. Conclusion
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Oancea, A.F.; Jigoranu, R.A.; Morariu, P.C.; Miftode, R.-S.; Trandabat, B.A.; Iov, D.E.; Cojocaru, E.; Costache, I.I.; Baroi, L.G.; Timofte, D.V.; et al. Atrial Fibrillation and Chronic Coronary Ischemia: A Challenging Vicious Circle. Life 2023, 13, 1370. [Google Scholar] [CrossRef] [PubMed]
- Kawakami, A; Kobayashi, Y; Katsube,T. Effect of Landiolol for Atrial Fibrillation after Open Heart Surgery. The Japanese Journal of Thoracic Surgery 2023, 75, 339–342.
- Kinjo, T.; Kimura, M.; Kaname, N.; Horiuchi, D.; Itoh, T.; Ishida, Y.; Nishizaki, K.; Toyama, Y.; Sasaki, S.; Tomita, H. Landiolol, an Intravenous Β1-selective Blocker, Is Useful for Dissociating a Fusion of Atrial Activation via Accessory Pathway and Atrioventricular Node. J Arrhythm 2023, 39, 937–946. [Google Scholar] [CrossRef] [PubMed]
- Ojima, T.; Iwahashi, M.; Nakamori, M.; Nakamura, M.; Katsuda, M.; Iida, T.; Hayata, K.; Yamaue, H. Atrial Fibrillation after Esophageal Cancer Surgery: An Analysis of 207 Consecutive Patients. Surg Today 2014, 44, 839–847. [Google Scholar] [CrossRef] [PubMed]
- Kiyokuni, M.; Konishi, M.; Sakamaki, K.; Kawashima, C.; Narikawa, M.; Doi, H.; Iwata, K.; Tomari, S.; Nakayama, N.; Komura, N.; et al. Beneficial Effect of Early Infusion of Landiolol, a Very Short-Acting Beta-1 Adrenergic Receptor Blocker, on Reperfusion Status in Acute Myocardial Infarction. Int J Cardiol 2016, 221, 321–326. [Google Scholar] [CrossRef] [PubMed]
- Hoshi, T.; Sato, A.; Nishina, H.; Kakefuda, Y.; Wang, Z.; Noguchi, Y.; Aonuma, K. Acute Hemodynamic Effects of Landiolol, an Ultra-Short-Acting Beta-Blocker, in Patients with Acute Coronary Syndrome: Preliminary Study. J Cardiol 2012, 60, 252–256. [Google Scholar] [CrossRef] [PubMed]
- Morelli, A.; Ertmer, C.; Westphal, M.; Rehberg, S.; Kampmeier, T.; Ligges, S.; Orecchioni, A.; D’Egidio, A.; D’Ippoliti, F.; Raffone, C.; et al. Effect of Heart Rate Control With Esmolol on Hemodynamic and Clinical Outcomes in Patients With Septic Shock. JAMA 2013, 310, 1683. [Google Scholar] [CrossRef] [PubMed]
- Seki, Y.; Jesmin, S.; Shimojo, N.; Islam, Md.M.; Rahman, Md.A.; Khatun, T.; Sakuramoto, H.; Oki, M.; Sonobe, A.; Kamiyama, J.; et al. Significant Reversal of Cardiac Upregulated Endothelin-1 System in a Rat Model of Sepsis by Landiolol Hydrochloride. Life Sci 2014, 118, 357–363. [Google Scholar] [CrossRef] [PubMed]
- Schroeder, L.; Monno, P.; Unger, M.; Ackerl, J.; Shatilova, O.; Schmitt, J.; Dresbach, T.; Mueller, A.; Kipfmueller, F. Heart Rate Control with Landiolol Hydrochloride in Infants with Ventricular Dysfunction and Pulmonary Hypertension. ESC Heart Fail 2023, 10, 385–396. [Google Scholar] [CrossRef]
- Chalkias, A.; O’Donnell, E.P. Mechanisms of Landiolol-Mediated Positive Inotropy in Critical Care Settings. Eur J Clin Pharmacol 2023, 79, 1607–1612. [Google Scholar] [CrossRef]
- Oliver, E.; Mayor Jr, F.; D’Ocon, P. Beta-Blockers: Historical Perspective and Mechanisms of Action. Revista Española de Cardiología (English Edition) 2019, 72, 853–862. [Google Scholar] [CrossRef] [PubMed]
- Grandi, E.; Ripplinger, C.M. Antiarrhythmic Mechanisms of Beta Blocker Therapy. Pharmacol Res 2019, 146, 104274. [Google Scholar] [CrossRef] [PubMed]
- Poveda-Jaramillo, R.; Monaco, F.; Zangrillo, A.; Landoni, G. Ultra-Short–Acting β-Blockers (Esmolol and Landiolol) in the Perioperative Period and in Critically Ill Patients. J Cardiothorac Vasc Anesth 2018, 32, 1415–1425. [Google Scholar] [CrossRef] [PubMed]
- Morris J; Awosika AO; Dunham A Metoprolol. In StatPearls; StatPearls Publishing: Treasure Island (FL), 2023.
- AlHabeeb, W.; Mrabeti, S.; Abdelsalam, A.A.I. Therapeutic Properties of Highly Selective β-Blockers With or Without Additional Vasodilator Properties: Focus on Bisoprolol and Nebivolol in Patients With Cardiovascular Disease. Cardiovasc Drugs Ther 2022, 36, 959–971. [Google Scholar] [CrossRef] [PubMed]
- Kirch, W.; Görg, K.G. Clinical Pharmacokinetics of Atenolol — A Review. Eur J Drug Metab Pharmacokinet 1982, 7, 81–91. [Google Scholar] [CrossRef] [PubMed]
- Wisler, J.W.; DeWire, S.M.; Whalen, E.J.; Violin, J.D.; Drake, M.T.; Ahn, S.; Shenoy, S.K.; Lefkowitz, R.J. A Unique Mechanism of β-Blocker Action: Carvedilol Stimulates β-Arrestin Signaling. Proceedings of the National Academy of Sciences 2007, 104, 16657–16662. [Google Scholar] [CrossRef] [PubMed]
- Lewis, P. The Essential Action of Propranolol in Hypertension. Am J Med 1976, 60, 837–852. [Google Scholar] [CrossRef] [PubMed]
- Sezai, A.; Osaka, S.; Yaoita, H.; Ishii, Y.; Arimoto, M.; Hata, H.; Shiono, M. Safety and Efficacy of Landiolol Hydrochloride for Prevention of Atrial Fibrillation after Cardiac Surgery in Patients with Left Ventricular Dysfunction: Prevention of Atrial Fibrillation after Cardiac Surgery with Landiolol Hydrochloride for Left Ventricular Dysfunction (PLATON) Trial. Journal of Thoracic and Cardiovascular Surgery 2015, 150, 957–964. [Google Scholar]
- Sezai, A.; Minami, K.; Nakai, T.; Hata, M.; Yoshitake, I.; Wakui, S.; Shiono, M.; Hirayama, A. Landiolol Hydrochloride for Prevention of Atrial Fibrillation after Coronary Artery Bypass Grafting: New Evidence from the PASCAL Trial. J Thorac Cardiovasc Surg 2011, 141, 1478–1487. [Google Scholar] [CrossRef]
- Sezai, A.; Nakai, T.; Hata, M.; Yoshitake, I.; Shiono, M.; Kunimoto, S.; Hirayama, A. Feasibility of Landiolol and Bisoprolol for Prevention of Atrial Fibrillation after Coronary Artery Bypass Grafting: A Pilot Study. J Thorac Cardiovasc Surg 2012, 144, 1241–1248. [Google Scholar] [CrossRef]
- Hagiwara, S.; Iwasaka, H.; Maeda, H.; Noguchi, T. LANDIOLOL, AN ULTRASHORT-ACTING Β1-ADRENOCEPTOR ANTAGONIST, HAS PROTECTIVE EFFECTS IN AN LPS-INDUCED SYSTEMIC INFLAMMATION MODEL. Shock 2009, 31, 515–520. [Google Scholar] [CrossRef]
- Oprea, A.D.; Lombard, F.W.; Kertai, M.D. Perioperative β-Adrenergic Blockade in Noncardiac and Cardiac Surgery: A Clinical Update. J Cardiothorac Vasc Anesth 2019, 33, 817–832. [Google Scholar] [CrossRef] [PubMed]
- January, C.T.; Wann, L.S.; Alpert, J.S.; Calkins, H.; Cigarroa, J.E.; Cleveland, J.C.; Conti, J.B.; Ellinor, P.T.; Ezekowitz, M.D.; Field, M.E.; et al. 2014 AHA/ACC/HRS Guideline for the Management of Patients With Atrial Fibrillation: Executive Summary. Circulation 2014, 130, 2071–2104. [Google Scholar] [CrossRef] [PubMed]
- Hindricks, G.; Potpara, T.; Dagres, N.; Arbelo, E.; Bax, J.J.; Blomström-Lundqvist, C.; Boriani, G.; Castella, M.; Dan, G.-A.; Dilaveris, P.E.; et al. 2020 ESC Guidelines for the Diagnosis and Management of Atrial Fibrillation Developed in Collaboration with the European Association for Cardio-Thoracic Surgery (EACTS). Eur Heart J 2021, 42, 373–498. [Google Scholar] [CrossRef]
- Arsenault, K.A.; Yusuf, A.M.; Crystal, E.; Healey, J.S.; Morillo, C.A.; Nair, G.M.; Whitlock, R.P. Interventions for Preventing Post-Operative Atrial Fibrillation in Patients Undergoing Heart Surgery. Cochrane Database of Systematic Reviews 2013, 2021. [Google Scholar]
- Thein, P.M.; White, K.; Banker, K.; Lunny, C.; Mirzaee, S.; Nasis, A. Preoperative Use of Oral Beta-Adrenergic Blocking Agents and the Incidence of New-Onset Atrial Fibrillation After Cardiac Surgery. A Systematic Review and Meta-Analysis. Heart Lung Circ 2018, 27, 310–321. [Google Scholar] [CrossRef]
- Sasaki, K.; Kumagai, K.; Maeda, K.; Akiyama, M.; Ito, K.; Matsuo, S.; Katahira, S.; Suzuki, T.; Suzuki, Y.; Kaiho, Y.; et al. Preventive Effect of Low-Dose Landiolol on Postoperative Atrial Fibrillation Study (PELTA Study). Gen Thorac Cardiovasc Surg 2020, 68, 1240–1251. [Google Scholar] [CrossRef] [PubMed]
- Hao, J.; Zhou, J.; Xu, W.; Chen, C.; Zhang, J.; Peng, H.; Liu, L. Beta-Blocker Landiolol Hydrochloride in Preventing Atrial Fibrillation Following Cardiothoracic Surgery: A Systematic Review and Meta-Analysis. Annals of Thoracic and Cardiovascular Surgery 2022, 28, 18–31. [Google Scholar] [CrossRef]
- Sakamoto, A.; Kitakaze, M.; Takamoto, S.; Namiki, A.; Kasanuki, H.; Hosoda, S. Landiolol, an Ultra-Short-Acting Β1-Blocker, More Effectively Terminates Atrial Fibrillation than Diltiazem after Open Heart Surgery: Prospective, Multicenter, Randomized, Open-Label Study (JL-KNIGHT Study). Circulation Journal 2012, 76, 1097–1101. [Google Scholar] [CrossRef]
- Balik, M.; Sander, M.; Trimmel, H.; Heinz, G. Landiolol for Managing Post-Operative Atrial Fibrillation. European Heart Journal Supplements 2018, 20, A10–A14. [Google Scholar] [CrossRef]
- Nakano, T.; Shimizu, K.; Kawashima, O.; Kamiyoshihara, M.; Nagashima, T.; Ibe, T.; Takeyoshi, I. Effect of Landiolol Hydrochloride, an Ultra-Short-Acting Beta 1-Selective Blocker, on Supraventricular Tachycardia, Atrial Fibrillation and Flutter after Pulmonary Resection. J Clin Pharm Ther 2012, 37, 431–435. [Google Scholar] [CrossRef]
- Nojiri, T.; Yamamoto, K.; Maeda, H.; Takeuchi, Y.; Funakoshi, Y.; Maekura, R.; Okumura, M. Efficacy of Low-Dose Landiolol, an Ultrashort-Acting β-Blocker, on Postoperative Atrial Fibrillation in Patients Undergoing Pulmonary Resection for Lung Cancer. Gen Thorac Cardiovasc Surg 2011, 59, 799–805. [Google Scholar] [CrossRef] [PubMed]
- Ito, K.; Nozaki, M.; Sakamoto, R.; Suzuki, T.; Masuda, R.; Iwazaki, M. Safety of Landiolol Infusion in Patients Undergoing Lung Resection. Open J Anesthesiol 2014, 04, 183–190. [Google Scholar] [CrossRef]
- Aoyama, H.; Otsuka, Y.; Aoyama, Y. Landiolol Infusion during General Anesthesia Does Not Prevent Postoperative Atrial Fibrillation in Patients Undergoing Lung Resection. Gen Thorac Cardiovasc Surg 2016, 64, 735–741. [Google Scholar] [CrossRef]
- Yoshida T; Furukita Y; Yamamoto Y; Nishino T; Inoue S; Morimoto M; Okumura K; Toba H; Yoshida M; Takizawa H; et al. A Randomized, Open Label Study of the Efficacy of Prophylactic 24-h Low-Dose Landiolol for Atrial Fibrillation in Transthoracic Esophagectomy. Esophagus 2017, 14, 97–103.
- Okita, T.; Uji, M.; Shinjo, T.; Morioka, M.; Kumano, H.; Ishimura, N.; Nishiwada, M. [Use of Landiolol Hydrochloride for the Prevention of Atrial Fibrillation after Lung Resection]. Masui 2008, 57, 953–958. [Google Scholar] [PubMed]
- Mori, K.; Yamada, K.; Fukuda, T.; Mitsui, T.; Kitamura, T.; Yamaguchi, D.; Ando, J.; Wada, I.; Nomura, S.; Shimizu, N.; et al. Landiolol Hydrochloride for Early Postoperative Tachycardia after Transthoracic Esophagectomy. Surg Today 2014, 44, 848–854. [Google Scholar] [CrossRef]
- Niwa, Y.; Koike, M.; Iwata, N.; Kobayashi, D.; Tanaka, C.; Fujii, T.; Nakayama, G.; Sugimoto, H.; Fujiwara, M.; Kodera, Y. Effect of Landiolol Hydrochloride on Tachyarrhythmia after Esophagectomy. Hepatogastroenterology 2014, 61, 1546–1551. [Google Scholar]
- Wariishi, S.; Yamashita, K.; Nishimori, H.; Fukutomi, T.; Yamamoto, M.; Radhakrishnan, G.; Sasaguri, S. Postoperative Administration of Landiolol Hydrochloride for Patients with Supraventricular Arrhythmia: The Efficacy of Sustained Intravenous Infusion at a Low Dose☆. Interact Cardiovasc Thorac Surg 2009, 9, 811–813. [Google Scholar] [CrossRef]
- Ojima, T.; Nakamori, M.; Nakamura, M.; Katsuda, M.; Hayata, K.; Kato, T.; Kitadani, J.; Tabata, H.; Takeuchi, A.; Yamaue, H. Randomized Clinical Trial of Landiolol Hydrochloride for the Prevention of Atrial Fibrillation and Postoperative Complications after Oesophagectomy for Cancer. British Journal of Surgery 2017, 104, 1003–1009. [Google Scholar] [CrossRef]
- Bezati, S.; Velliou, M.; Polyzogopoulou, E.; Boultadakis, A.; Parissis, J. The Role of Landiolol in the Management of Atrial Tachyarrhythmias in Patients with Acute Heart Failure and Cardiogenic Shock: Case Reports and Review of Literature. European Heart Journal, Supplement 2022, 24, D22–D33. [Google Scholar] [CrossRef]
- McDonagh, T.A.; Metra, M.; Adamo, M.; Gardner, R.S.; Baumbach, A.; Böhm, M.; Burri, H.; Butler, J.; Čelutkienė, J.; Chioncel, O.; et al. 2021 ESC Guidelines for the Diagnosis and Treatment of Acute and Chronic Heart Failure. Eur Heart J 2021, 42, 3599–3726. [Google Scholar] [CrossRef]
- Okajima, M.; Takamura, M.; Taniguchi, T. Landiolol, an Ultra-Short-Acting Β1-Blocker, Is Useful for Managing Supraventricular Tachyarrhythmias in Sepsis. World J Crit Care Med 2015, 4, 251. [Google Scholar] [CrossRef]
- Rehberg, S.; Joannidis, M.; Whitehouse, T.; Morelli, A. Landiolol for Managing Atrial Fibrillation in Intensive Care. European Heart Journal, Supplement 2018, 20, A15–A18. [Google Scholar] [CrossRef]
- Nagai, R.; Kinugawa, K.; Inoue, H.; Atarashi, H.; Seino, Y.; Yamashita, T.; Shimizu, W.; Aiba, T.; Kitakaze, M.; Sakamoto, A.; et al. Urgent Management of Rapid Heart Rate in Patients with Atrial Fibrillation/Flutter and Left Ventricular Dysfunction - Comparison of the Ultra-Short-Acting Β1-Selective Blocker Landiolol with Digoxin (J-Land Study) -. Circulation Journal 2013, 77, 908–916. [Google Scholar] [CrossRef] [PubMed]
- Wada, Y.; Aiba, T.; Tsujita, Y.; Itoh, H.; Wada, M.; Nakajima, I.; Ishibashi, K.; Okamura, H.; Miyamoto, K.; Noda, T.; et al. Practical Applicability of Landiolol, an Ultra-Short-Acting Β1-Selective Blocker, for Rapid Atrial and Ventricular Tachyarrhythmias with Left Ventricular Dysfunction. J Arrhythm 2016, 32, 82–88. [Google Scholar] [CrossRef] [PubMed]
- Iwahashi, N.; Takahashi, H.; Abe, T.; Okada, K.; Akiyama, E.; Matsuzawa, Y.; Konishi, M.; Maejima, N.; Hibi, K.; Kosuge, M.; et al. Urgent Control of Rapid Atrial Fibrillation by Landiolol in Patients With Acute Decompensated Heart Failure With Severely Reduced Ejection Fraction. Circ Rep 2019, 1, 422–430. [Google Scholar] [CrossRef]
- Kobayashi, S.; Myoren, T.; Kajii, T.; Kohno, M.; Nanno, T.; Ishiguchi, H.; Nishimura, S.; Fukuda, M.; Hino, A.; Fujimura, T.; et al. Addition of a Β1-Blocker to Milrinone Treatment Improves Cardiac Function in Patients with Acute Heart Failure and Rapid Atrial Fibrillation. Cardiology (Switzerland) 2019, 142, 195–202. [Google Scholar] [CrossRef] [PubMed]
- Kakihana, Y.; Nishida, O.; Taniguchi, T.; Okajima, M.; Morimatsu, H.; Ogura, H.; Yamada, Y.; Nagano, T.; Morishima, E.; Matsuda, N. Efficacy and Safety of Landiolol, an Ultra-Short-Acting Β1-Selective Antagonist, for Treatment of Sepsis-Related Tachyarrhythmia (J-Land 3S): A Multicentre, Open-Label, Randomised Controlled Trial. Lancet Respir Med 2020, 8, 863–872. [Google Scholar] [CrossRef]
- Matsuda, N.; Nishida, O.; Taniguchi, T.; Okajima, M.; Morimatsu, H.; Ogura, H.; Yamada, Y.; Nagano, T.; Ichikawa, A.; Kakihana, Y. Impact of Patient Characteristics on the Efficacy and Safety of Landiolol in Patients with Sepsis-Related Tachyarrhythmia: Subanalysis of the J-Land 3S Randomised Controlled Study. EClinicalMedicine 2020, 28. [Google Scholar] [CrossRef]
- Unger, M.; Morelli, A.; Singer, M.; Radermacher, P.; Rehberg, S.; Trimmel, H.; Joannidis, M.; Heinz, G.; Cerny, V.; Dostál, P.; et al. Landiolol in Patients with Septic Shock Resident in an Intensive Care Unit (LANDI-SEP): Study Protocol for a Randomized Controlled Trial. Trials 2018, 19. [Google Scholar] [CrossRef] [PubMed]
- Syed, Y.Y. Landiolol: A Review in Tachyarrhythmias. Drugs 2018, 78, 377–388. [Google Scholar] [CrossRef] [PubMed]
- Yamashita, T.; Nakasu, Y.; Mizutani, H.; Sumitani, K. A Prospective Observational Survey on Landiolol in Atrial Fibrillation/Atrial Flutter Patients with Chronic Heart Failure – AF-CHF Landiolol Survey. J Cardiol 2019, 74, 418–425. [Google Scholar] [CrossRef] [PubMed]
- Yamashita, T.; Saitoh, T.; Matsushita, M. Design of a Prospective Observational Survey on Landiolol in Atrial Fibrillation/Atrial Flutter Patients with Chronic Heart Failure - AF-CHF Landiolol Survey. J Cardiol 2015, 66, 69–72. [Google Scholar] [CrossRef] [PubMed]
- Stix, G.; Wolzt, M.; Domanovits, H.; Kadlecová, P.; Husch, B.; Trebs, M.; Hodisch, J.; Unger, M.; Krumpl, G. Open-Label Two-Dose Pilot Study of Landiolol for the Treatment of Atrial Fibrillation/Atrial Flutter in Caucasian Patients. Circulation Journal 2019, 84, 33–42. [Google Scholar] [CrossRef] [PubMed]
- Shibata, S.C.; Uchiyama, A.; Ohta, N.; Fujino, Y. Efficacy and Safety of Landiolol Compared to Amiodarone for the Management of Postoperative Atrial Fibrillation in Intensive Care Patients. J Cardiothorac Vasc Anesth 2016, 30, 418–422. [Google Scholar] [CrossRef] [PubMed]
- Horikoshi, Y.; Goyagi, T.; Kudo, R.; Kodama, S.; Horiguchi, T.; Nishikawa, T. The Suppressive Effects of Landiolol Administration on the Occurrence of Postoperative Atrial Fibrillation and Tachycardia, and Plasma IL-6 Elevation in Patients Undergoing Esophageal Surgery: A Randomized Controlled Clinical Trial. J Clin Anesth 2017, 38, 111–116. [Google Scholar] [CrossRef] [PubMed]
- Schnaubelt, S.; Eibensteiner, F.; Oppenauer, J.; Tihanyi, D.; Neymayer, M.; Brock, R.; Kornfehl, A.; Veigl, C.; Al Jalali, V.; Anders, S.; et al. Hemodynamic and Rhythmologic Effects of Push-Dose Landiolol in Critical Care—A Retrospective Cross-Sectional Study. Pharmaceuticals 2023, 16. [Google Scholar] [CrossRef]
- Matsuishi, Y.; Jesmin, S.; Kawano, S.; Hideaki, S.; Shimojo, N.; Mowa, C.N.; Akhtar, S.; Zaedi, S.; Khatun, T.; Tsunoda, Y.; et al. Landiolol Hydrochloride Ameliorates Acute Lung Injury in a Rat Model of Early Sepsis through the Suppression of Elevated Levels of Pulmonary Endothelin-1. Life Sci 2016, 166, 27–33. [Google Scholar] [CrossRef]
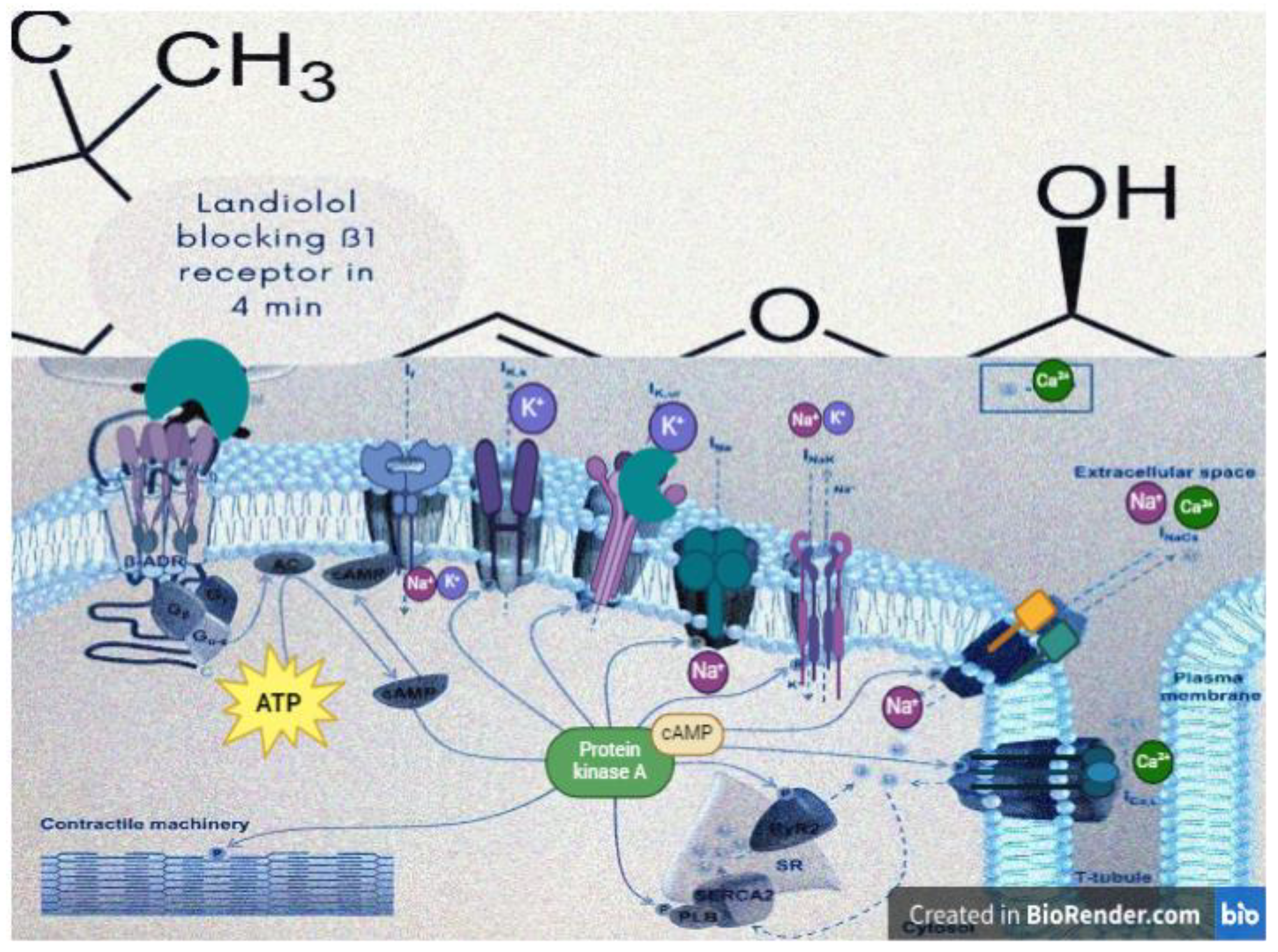
| Current channel | If | IKs | INa | INaK |
|---|---|---|---|---|
| Role | A mixed cation current is carried by both Na+ and K+. It is mainly involved in the pacemaker activity of the sinoatrial node, contributing to the diastolic depolarization and spontaneous firing of action potentials. | A delayed rectifier potassium current plays a role in repolarizing the cardiac action potential. It contributes to the plateau phase of the action potential in cardiac myocytes. | The fast inward sodium current is responsible for the rapid depolarization phase of the cardiac action potential. It plays a critical role in initiating and propagating action potentials in cardiac myocytes. | The sodium-potassium pump (Na+/K+-ATPase) plays a crucial role in maintaining the resting membrane potential of cardiac myocytes by actively transporting 3 atoms of sodium out of the cell and 2 atoms of potassium into the cell. |
| The influence of landiolol1 | Indirect- by blocking ß-1 receptors, landiolol reduces the stimulatory effects of endogenous catecholamines, leading to a decrease in If activity and a subsequent decrease in heart rate. | May have minor effects on IKs, primarily through downstream signaling pathways influenced by the blockade of ß-1 adrenergic receptors. | Indirect- by blocking ß-1 receptors, landiolol reduces the stimulatory effects of catecholamines on INa, leading to a decrease in the rate of rise of the action potential and a reduction in myocardial excitability. | Not well-documented, its effects on intracellular calcium levels and ion handling in cardiac myocytes, mediated through ß-1 receptor blockade, may indirectly influence the activity of the sodium-potassium pump. |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




