Submitted:
13 February 2024
Posted:
14 February 2024
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Results
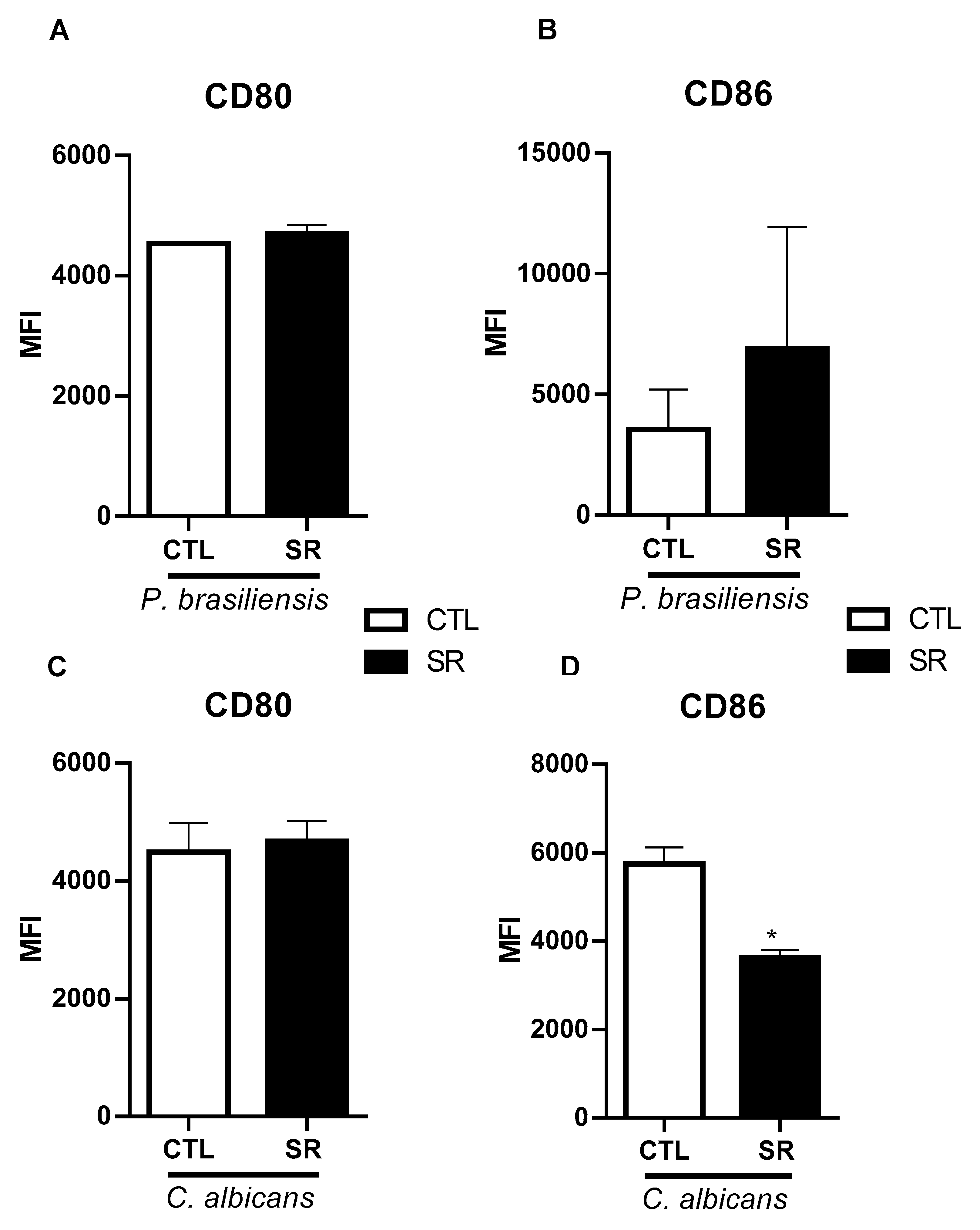
2.1. Expression of B-1 cell activation molecules following mice intraperitoneal stimulation with P. brasiliensis or C. albicans
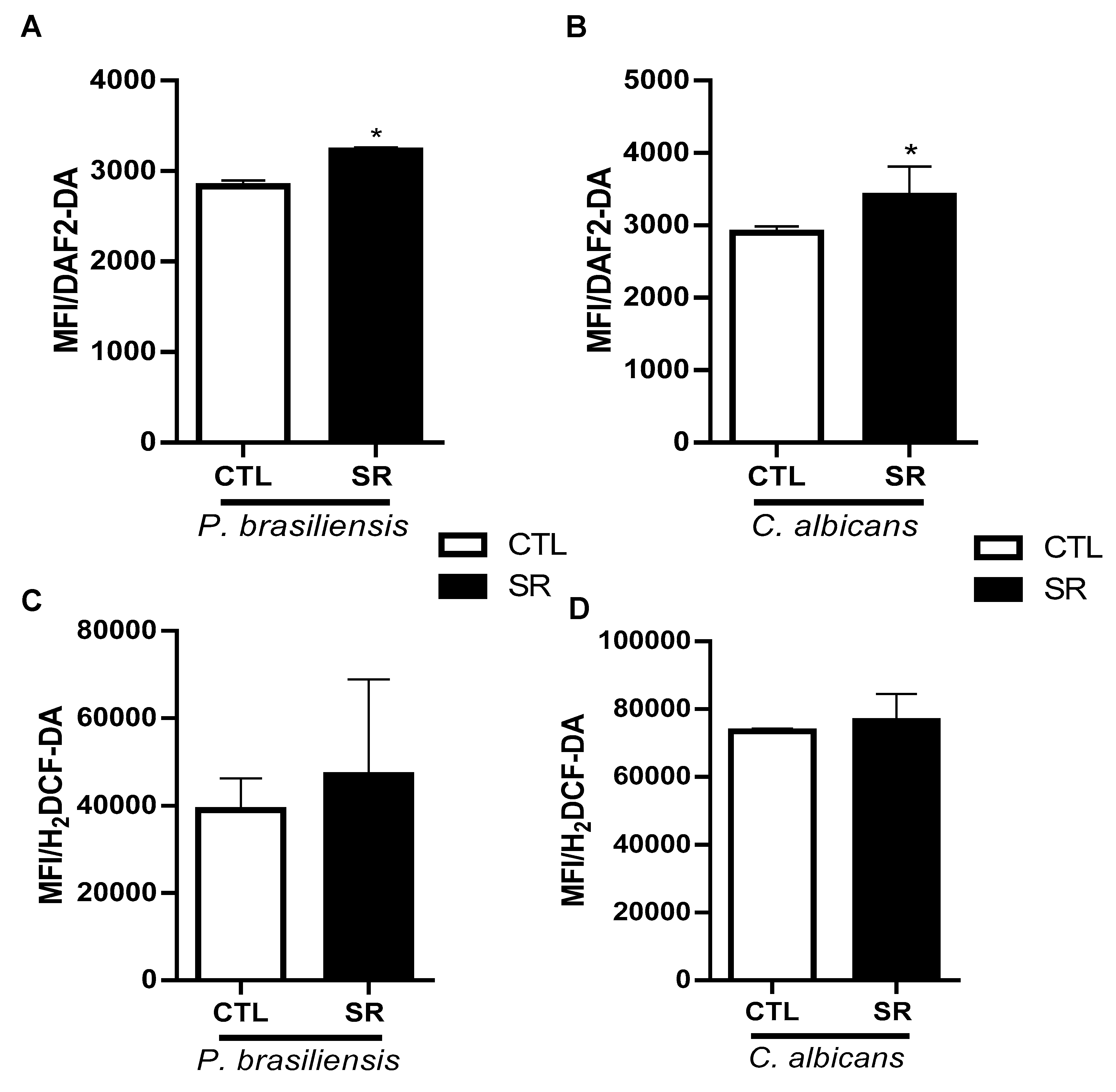
2.2. Evaluation of intracellular NO, ROS production after intraperitoneal stimulation of C57BL/6J mice with P. brasiliensis or C. albicans
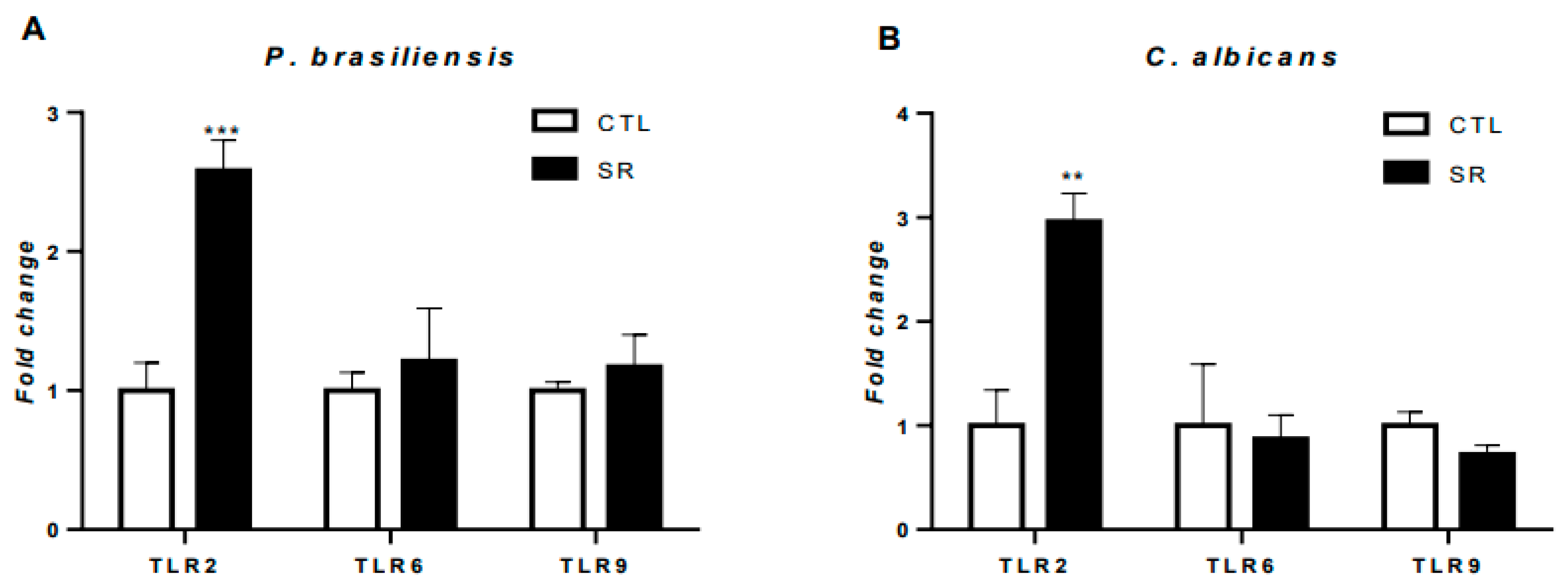
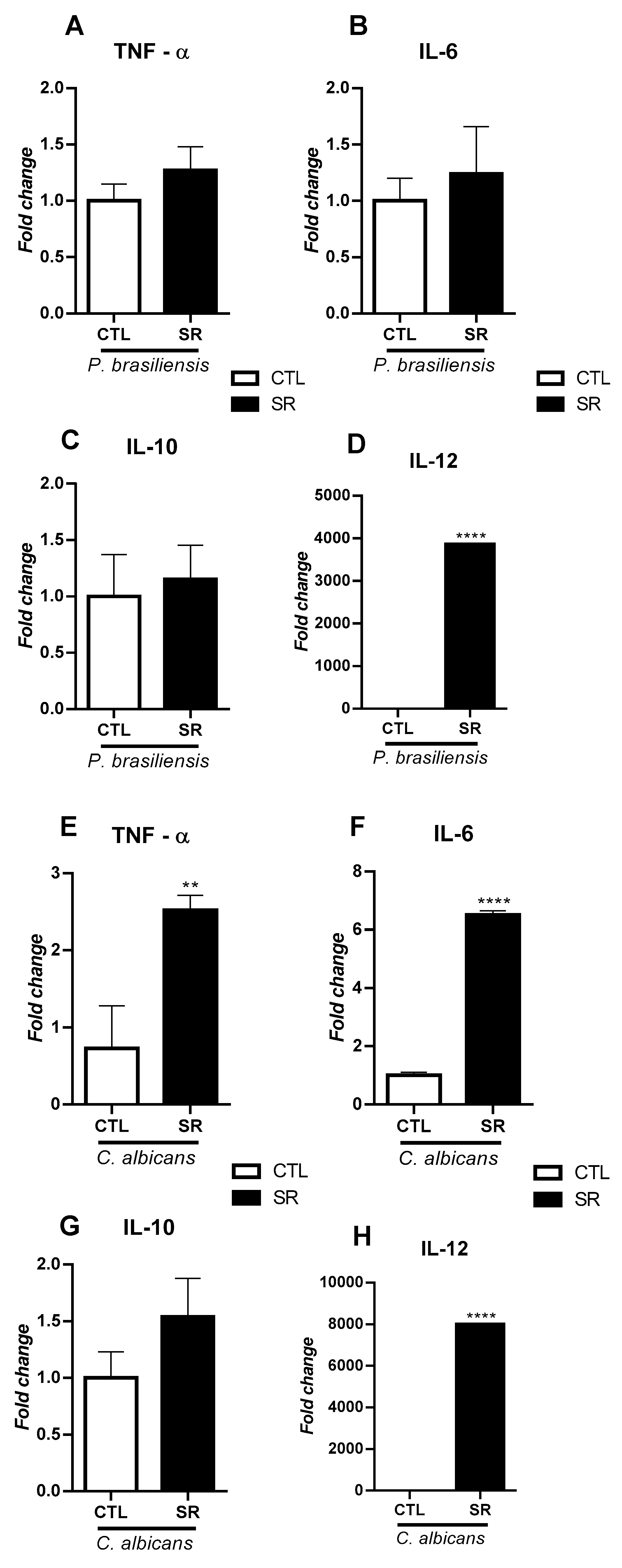
2.3. Expression of TLR and cytokine genes in B-1 cells after murine intraperitoneal stimulation with P. brasiliensis or C. albicans
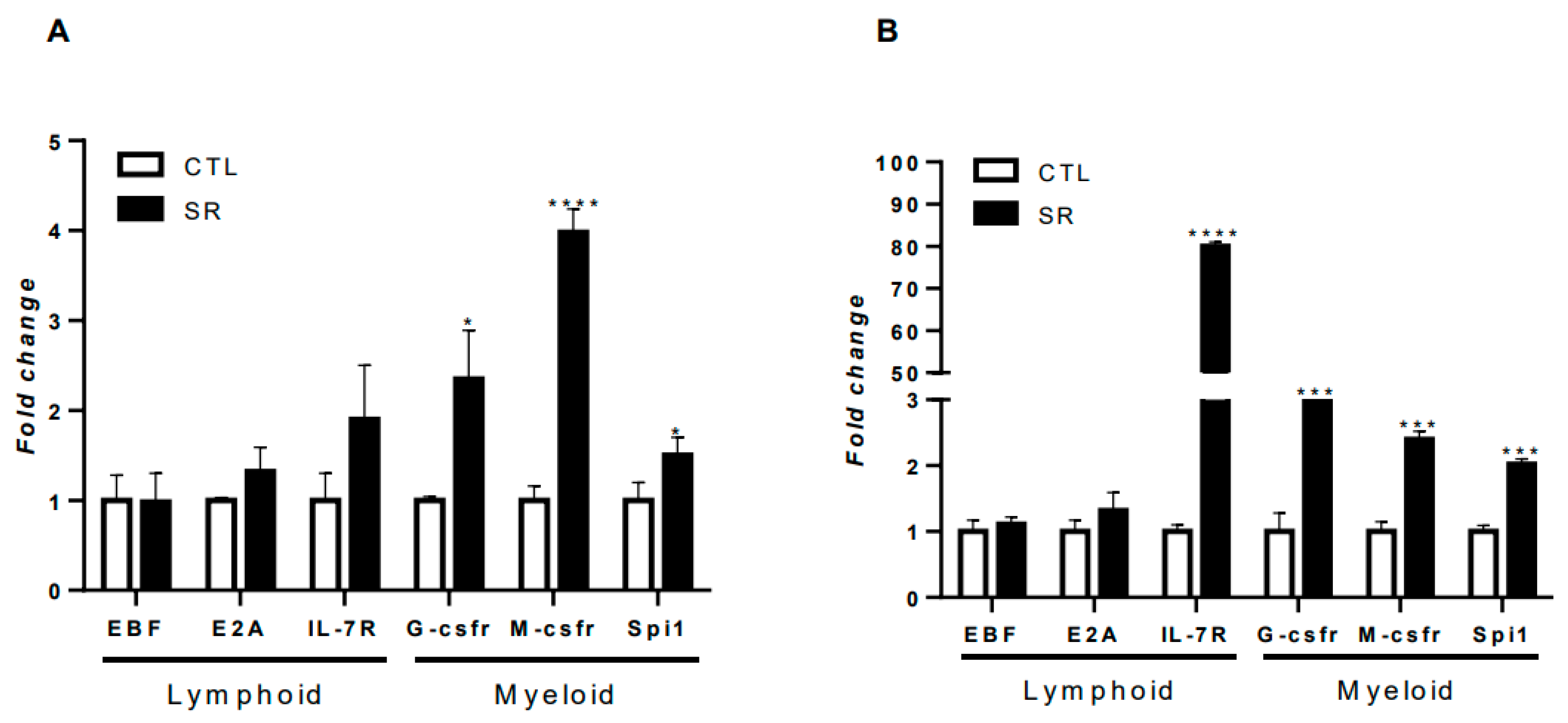
2.4. Expression of lymphoid and myeloid commitment genes in B-1 cells from sleep-restricted C57BL/6J mice and intraperitoneally stimulated with P. brasiliensis or C. albicans fungi
3. Discussion
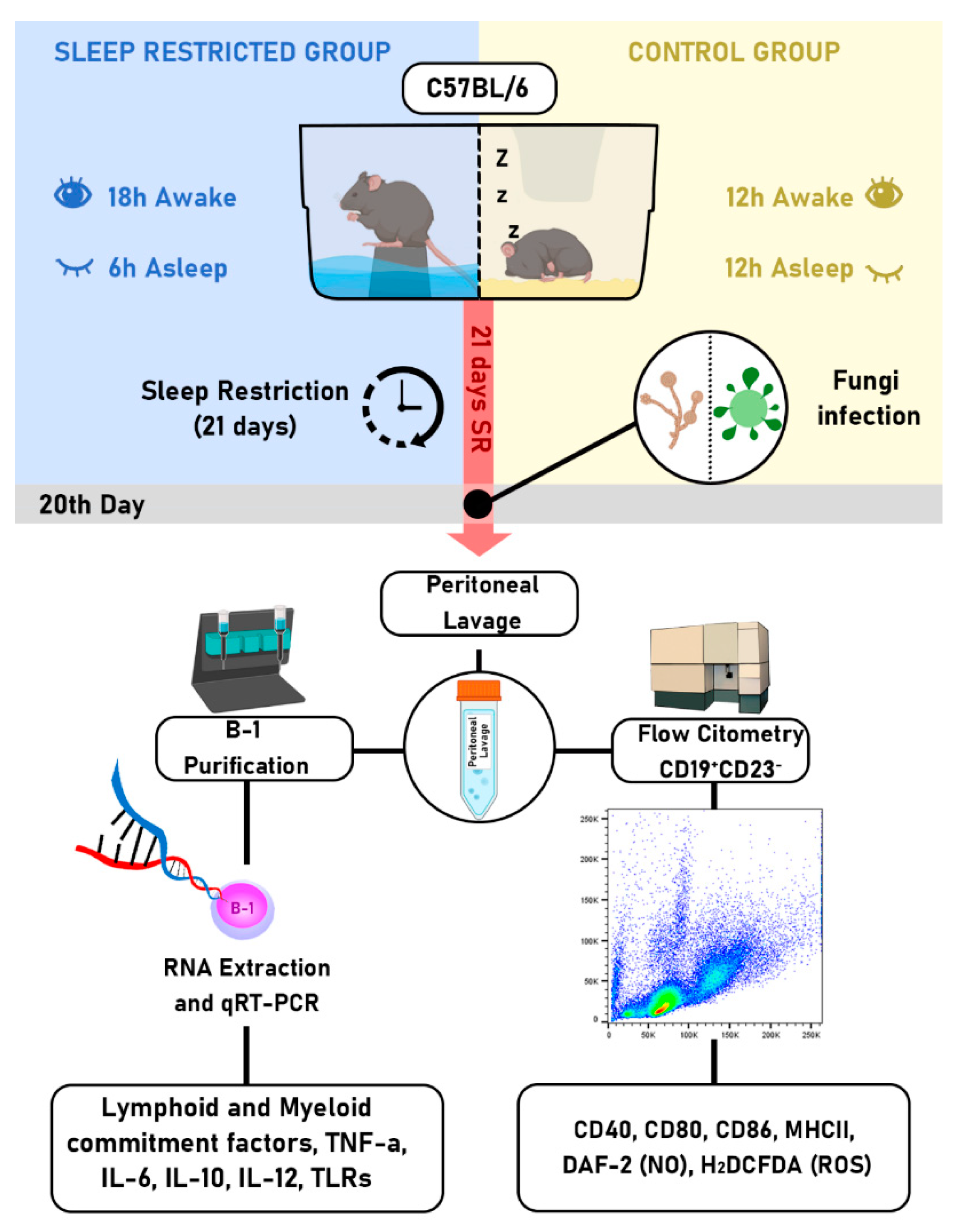
4. Material and Methods
4.1. Animals
4.2. Sleep Restriction Protocol
4.3. Flow cytometry analysis
4.4. Enrichment of peritoneal B-1 lymphocytes
4.5. Quantitative reverse transcriptase-polymerase chain reaction (qRT-PCR)
4.6. Fungus
4.7. Statistical Analysis
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Hardy, R.R. and K. Hayakawa. B cell development pathways. Annu Rev Immunol. 2001, 19, 595–621. [CrossRef]
- Berland, R. and H.H. Wortis. Origins and functions of B-1 cells with notes on the role of CD5. Annu Rev Immunol. 2002, 20, 253–300. [CrossRef]
- Baumgarth, N. The double life of a B-1 cell: self-reactivity selects for protective effector functions. Nat Rev Immunol. 2011, 11, 34–46. [Google Scholar] [CrossRef]
- Montecino-Rodriguez, E. and K. Dorshkind. New perspectives in B-1 B cell development and function. Trends Immunol. 2006, 27, 428–433. [CrossRef] [PubMed]
- Montecino-Rodriguez, E., et al. Distinct Genetic Networks Orchestrate the Emergence of Specific Waves of Fetal and Adult B-1 and B-2 Development. Immunity 2016, 45, 527–539. [CrossRef]
- Jin, W., Z. Luo, and H. Yang, Peripheral B Cell Subsets in Autoimmune Diseases: Clinical Implications and Effects of B Cell-Targeted Therapies. J Immunol Res. 2020, 2020, 9518137. [CrossRef]
- Novaes E Brito, R.R., et al., B-1 cell response in immunity against parasites. Parasitol Res. 2019, 118, 1343–1352. [CrossRef] [PubMed]
- Almeida, S.R., et al., Mouse B-1 cell-derived mononuclear phagocyte, a novel cellular component of acute non-specific inflammatory exudate. Int Immunol. 2001, 13, 1193–1201. [CrossRef]
- Ghosn, E.E., M. Russo, and S.R. Almeida, Nitric oxide-dependent killing of Cryptococcus neoformans by B-1-derived mononuclear phagocyte. J Leukoc Biol. 2006, 80, 36–44. [CrossRef]
- Novaes e Brito, R.R., et al., In Vitro and In Vivo Phagocytic Ability of Mouse B-1 Cells. Immunology and Immunogenetics Insights 2010, 2, 31. [CrossRef]
- Geraldo, M.M., et al., In vivo and in vitro phagocytosis of Leishmania (Leishmania) amazonensis promastigotes by B-1 cells. Parasite Immunol. 2016, 38, 365–376. [CrossRef]
- Popi, A.F., et al., Co-ordinated expression of lymphoid and myeloid specific transcription factors during B-1b cell differentiation into mononuclear phagocytes in vitro. Immunology 2009, 126, 114–122. [CrossRef] [PubMed]
- Popi, A.F., et al., Could a B-1 cell derived phagocyte "be one" of the peritoneal macrophages during LPS-driven inflammation? PLoS One 2012, 7, 345–370. [CrossRef]
- Reis, N.F.C., et al., Promastigotes or Extracellular Vesicles Modulate B-1 Cell Activation and Differentiation. Front Cell Infect Microbiol. 2020, 10, 573–813. [CrossRef]
- De Lorenzo, B.H., et al., Sleep-deprivation reduces NK cell number and function mediated by β-adrenergic signalling. Psychoneuroendocrinology 2015, 57, 134–143. [CrossRef]
- Sousa, M.E.P., et al., Invariant Natural Killer T cells resilience to paradoxical sleep deprivation-associated stress. Brain Behav Immun. 2020, 90, 208–215. [CrossRef] [PubMed]
- Besedovsky, H.O. and A. del Rey, Immune-neuro-endocrine interactions: facts and hypotheses. Endocr Rev. 1996, 17, 64–102. [CrossRef]
- Ransohoff, R.M. Chemokines and chemokine receptors: standing at the crossroads of immunobiology and neurobiology. Immunity 2009, 31, 711–721. [Google Scholar] [CrossRef]
- Vidal, A.S., et al., Impact of sleep restriction in B-1 cells activation and differentiation. Immunobiology 2022, 227, 152–280. [CrossRef]
- Palmblad, J., et al., Stressor exposure and immunological response in man: interferon-producing capacity and phagocytosis. J Psychosom Res. 1976, 20, 193–199. [CrossRef]
- Casey, F.B., et al., Altered antigen uptake and distribution due to exposure to extreme environmental temperatures or sleep deprivation. J Reticuloendothel Soc. 1974, 15, 87–95.
- Spiegel, K., J.F. Sheridan, and E. Van Cauter, Effect of sleep deprivation on response to immunization. JAMA 2002, 288, 1471–1472. [CrossRef]
- Lange, T., et al., Sleep enhances the human antibody response to hepatitis A vaccination. Psychosom Med. 2003, 65, 831–835. [CrossRef]
- Palma, B.D., et al., Effects of sleep deprivation on the development of autoimmune disease in an experimental model of systemic lupus erythematosus. Am J Physiol Regul Integr Comp Physiol. 2006, 291, 1527–1532. [CrossRef]
- da Costa, L.F.V., et al., B-1 cell decreases susceptibility to encephalitozoonosis in mice. Immunobiology 2017, 222, 218–227. [CrossRef]
- Noal, V., et al., Infection with Paracoccidioides brasiliensis induces B-1 cell migration and activation of regulatory T cells. Microbes Infect. 2016, 18, 798–803. [CrossRef] [PubMed]
- Kumar, H., T. Kawai, and S. Akira, Toll-like receptors and innate immunity. Biochem Biophys Res Commun. 2009, 388, 621–625. [CrossRef] [PubMed]
- Capitani, N. and C.T. Baldari, The Immunological Synapse: An Emerging Target for Immune Evasion by Bacterial Pathogens. Front Immunol. 2022, 13, 943344. [CrossRef] [PubMed]
- Horta, M.F., et al., Reactive oxygen species and nitric oxide in cutaneous leishmaniasis. J Parasitol Res. 2012, 2012, 203818. [CrossRef]
- Swindle, E.J. and D.D. Metcalfe, The role of reactive oxygen species and nitric oxide in mast cell-dependent inflammatory processes. Immunol Rev. 2007, 217, 186–205. [CrossRef] [PubMed]
- Chittora, R., et al., Cytomorphological Analysis and Interpretation of Nitric Oxide-Mediated Neurotoxicity in Sleep-Deprived Mice Model. Ann Neurosci. 2022, 29, 7–15. [CrossRef] [PubMed]
- Jiang, J., et al., REM sleep deprivation induces endothelial dysfunction and hypertension in middle-aged rats: Roles of the eNOS/NO/cGMP pathway and supplementation with L-arginine. PLoS One 2017, 12, 0182746. [CrossRef]
- Maragno-Correa, J.M., et al., Sleep deprivation increases mortality in female mice bearing ehrlich ascitic tumor. Neuroimmunomodulation 2013, 20, 134–140. [CrossRef]
- Kreuk, L.S., et al., B cell receptor and Toll-like receptor signaling coordinate to control distinct B-1 responses to both self and the microbiota. Elife 2019, 8. [CrossRef]
- Genestier, L., et al., TLR agonists selectively promote terminal plasma cell differentiation of B cell subsets specialized in thymus-independent responses. J Immunol. 2007, 178, 7779–7786. [CrossRef] [PubMed]
- Acorci-Valério, M.J., et al., Role of TLR2 and TLR4 in human neutrophil functions against Paracoccidioides brasiliensis. Scand J Immunol. 2010, 71, 99–108. [CrossRef]
- Rodriguez-Echeverri, C., J.D. Puerta-Arias, and Á. González, Paracoccidioides brasiliensis activates mesenchymal stem cells through TLR2, TLR4, and Dectin-1. Med Mycol. 2021, 59, 149–157. [CrossRef]
- Nakaira-Takahagi, E., et al., Interactions between TLR2, TLR4, and mannose receptors with gp43 from Paracoccidioides brasiliensis induce cytokine production by human monocytes. Med Mycol. 2011, 49, 694–703. [CrossRef]
- Bonfim, C.V., R.L. Mamoni, and M.H. Blotta, TLR-2, TLR-4 and dectin-1 expression in human monocytes and neutrophils stimulated by Paracoccidioides brasiliensis. Med Mycol. 2009, 47, 722–733. [CrossRef]
- Arango Duque, G. and A. Descoteaux, Macrophage cytokines: involvement in immunity and infectious diseases. Front Immunol. 2014, 5, 491. [CrossRef]
- Baumgarth, N. A Hard(y) Look at B-1 Cell Development and Function. J Immunol, 2017, 199, 3387–3394. [Google Scholar] [CrossRef]
- Popi, A.F., I.M. Longo-Maugéri, and M. Mariano, An Overview of B-1 Cells as Antigen-Presenting Cells. Front Immunol. 2016, 7, 138. [CrossRef]
- Arruda, C., et al., Interleukin-12 protects mice against disseminated infection caused by Paracoccidioides brasiliensis but enhances pulmonary inflammation. Clin Immunol. 2002, 103, 185–195. [CrossRef] [PubMed]
- Ashman, R.B., D. Vijayan, and C.A. Wells, IL-12 and related cytokines: function and regulatory implications in Candida albicans infection. Clin Dev Immunol. 2011, 2011, 686597. [CrossRef]
- Kawai, T. and S. Akira, The role of pattern-recognition receptors in innate immunity: update on Toll-like receptors. Nat Immunol. 2010, 11, 373–384. [CrossRef] [PubMed]
- Garbarino, S., et al., Role of sleep deprivation in immune-related disease risk and outcomes. Commun Biol. 2021, 4, 1304. [CrossRef]
- Axelsson, J., et al., Effects of sustained sleep restriction on mitogen-stimulated cytokines, chemokines and T helper 1/ T helper 2 balance in humans. PLoS One 2013, 8, e82291. [CrossRef]
- Dimitrov, S., et al., Sleep associated regulation of T helper 1/T helper 2 cytokine balance in humans. Brain Behav Immun. 2004, 18, 341–348. [CrossRef]
- Mussalem, J.S., et al., Adjuvant effect of killed Propionibacterium acnes on mouse peritoneal B-1 lymphocytes and their early phagocyte differentiation. PLoS One 2012, 7, e33955. [CrossRef]
- Bergmann, B.M., et al., Effect of extended sleep deprivation on tumor growth in rats. Am J Physiol. 1996, 271, R1460–R1464. [CrossRef]
- Silva, R.H., et al., Role of hippocampal oxidative stress in memory deficits induced by sleep deprivation in mice. Neuropharmacology 2004, 46, 895–903. [CrossRef]
- Machado, R.B., et al., Sleep deprivation induced by the modified multiple platform technique: quantification of sleep loss and recovery. Brain Res. 2004, 1004, 45–51. [CrossRef] [PubMed]
- Brito, R.R.N.e., et al., Role of distinct immune components in the radiation-induced abrogation of systemic lupus erythematosus development in mice. Lupus 2007, 16, 947–954. [CrossRef] [PubMed]
- Bustin, S.A., et al., The MIQE guidelines: minimum information for publication of quantitative real-time PCR experiments. Clin Chem. 2009, 55, 611–622. [CrossRef] [PubMed]
- Spandidos, A., et al., PrimerBank: a resource of human and mouse PCR primer pairs for gene expression detection and quantification. Nucleic Acids Res. 2010, 38, D792–D799. [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).



