Submitted:
08 February 2024
Posted:
09 February 2024
Read the latest preprint version here
Abstract
Keywords:
1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Cell Culture
2.3. Splenocyte Isolation
2.4. Cell Calcium Assays
2.5. Permeabilized Cell Assays
2.6. Statistical Analysis
3. Results
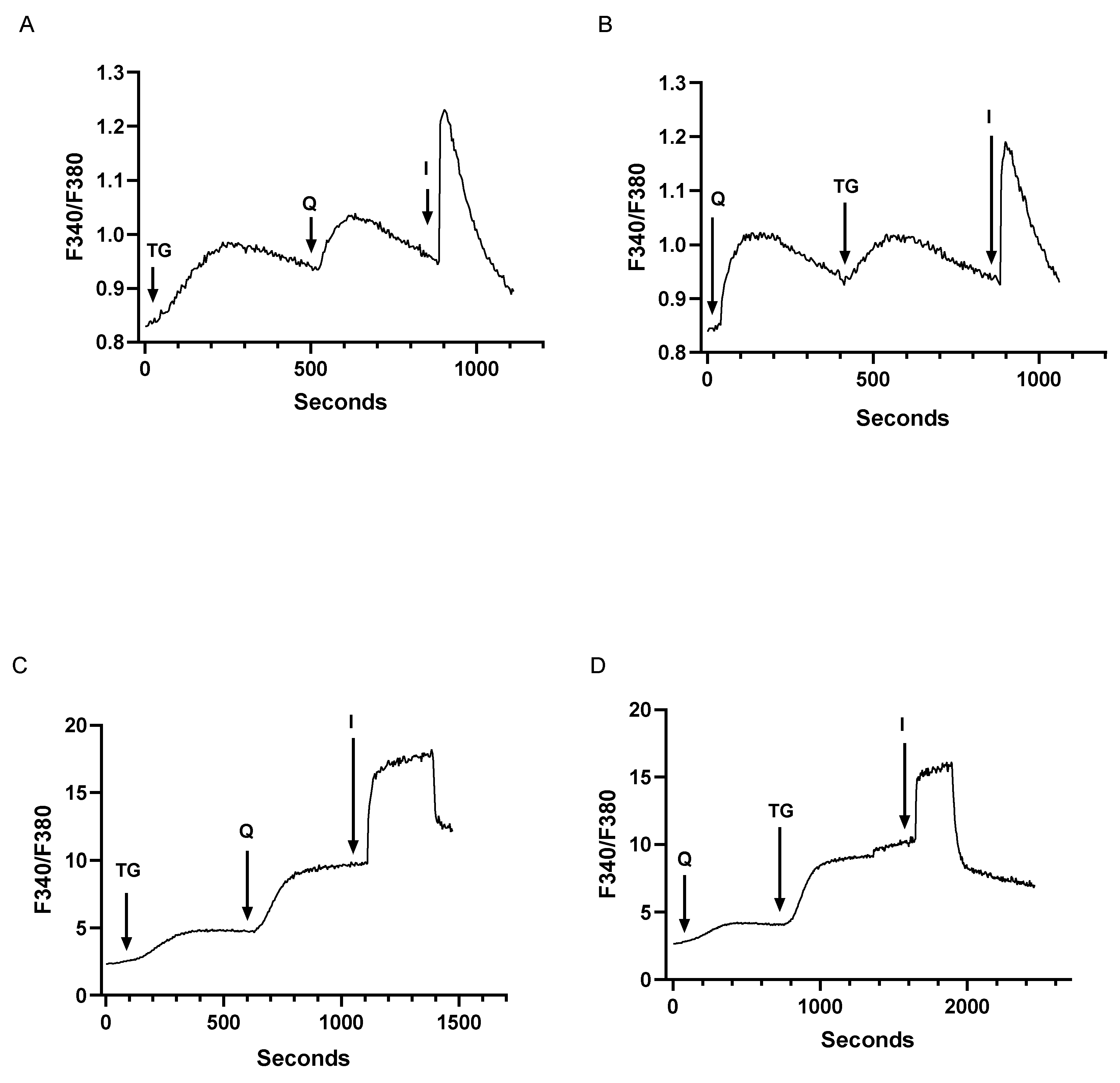
3.1. Low Concentrations of TG and tBHQ Induce Ca2+ Release in T Cells and Establish a Pharmacological Regimen for Specific Blockade of SERCA 2b and SERCA 3
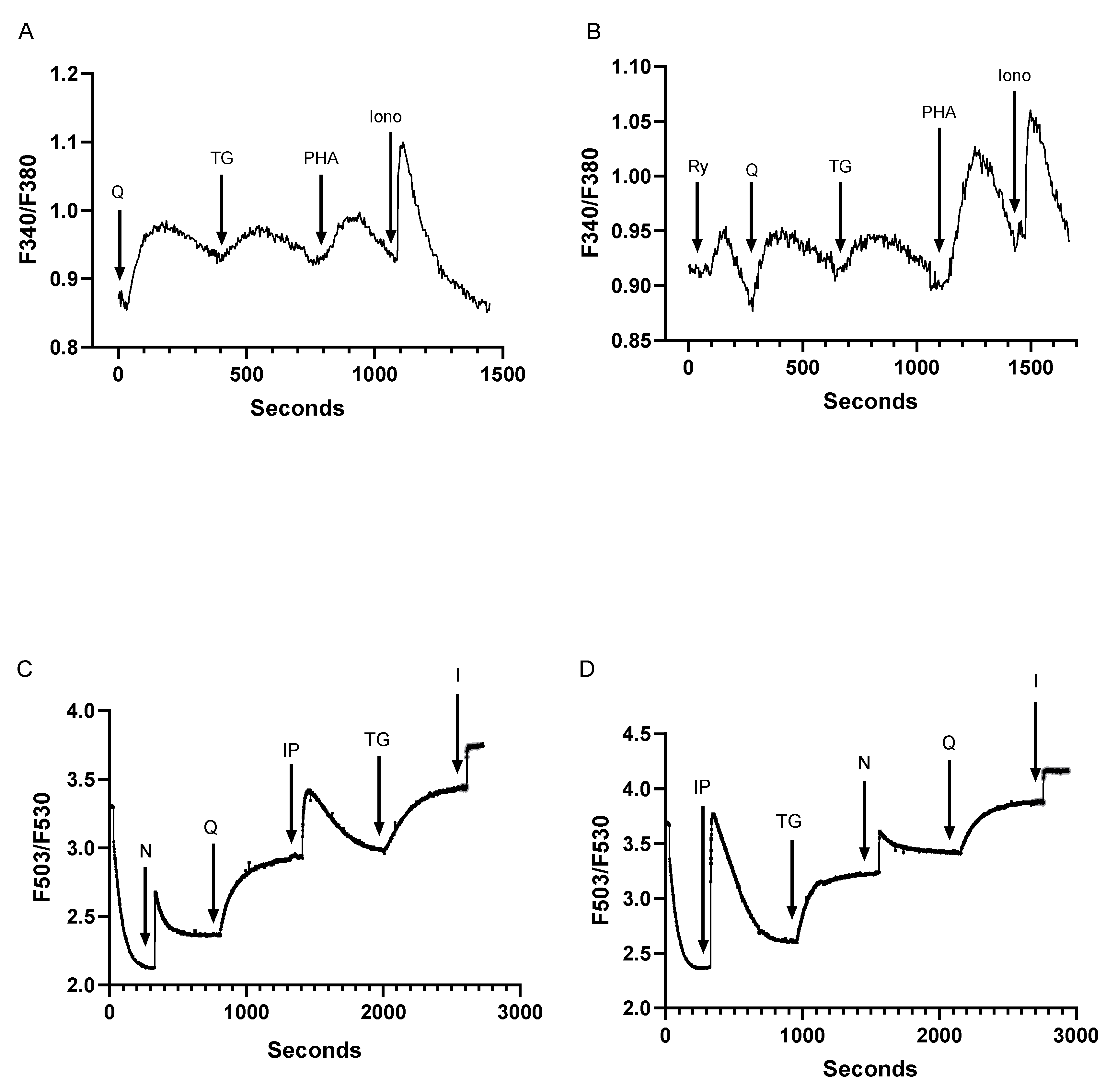
3.2. Low Dose SERCA Blockers and Agonist-Induced Ca2+ Release Patterns Suggest a Complex T Cell Ca2+ Pool Profile with up to Five Distinct Ca2+ Store Compartments
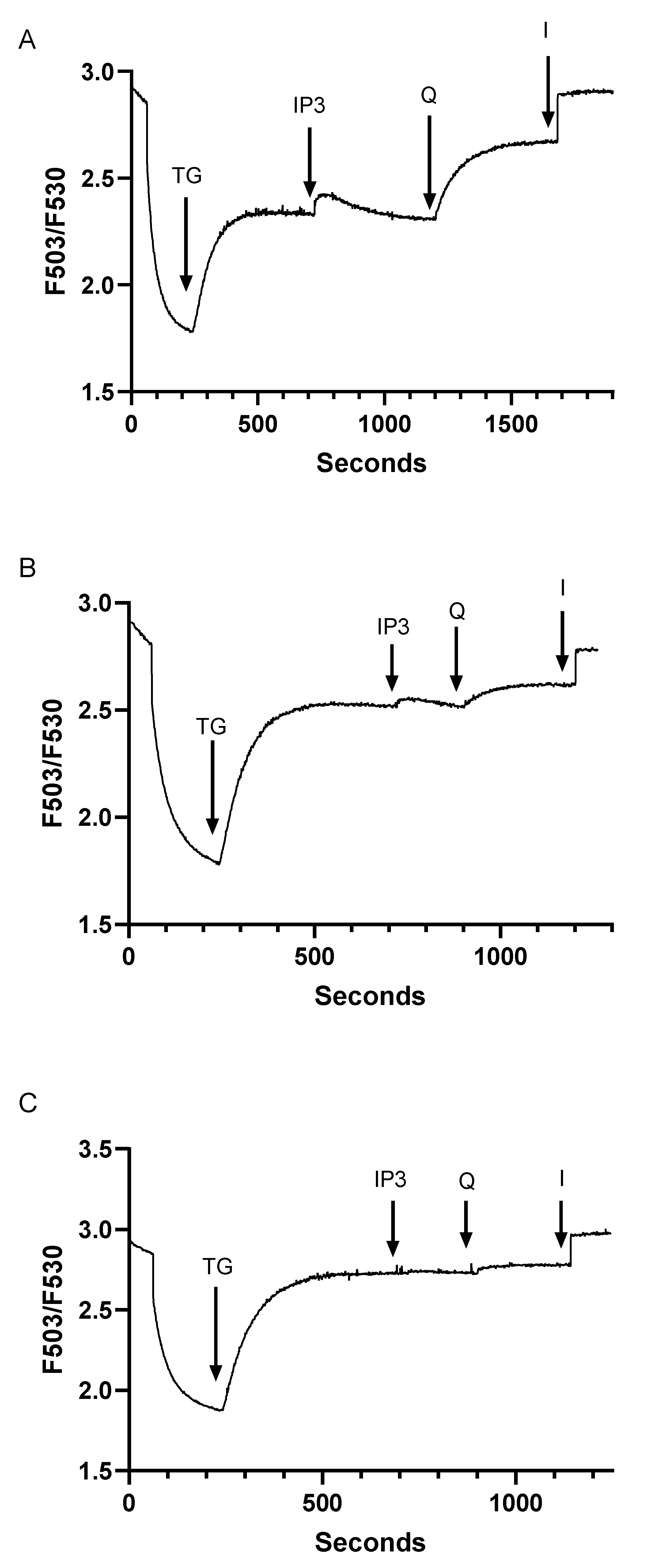
3.3. Assessment of Inter-Relationships of Agonist Releasable Ca2+ Pools in T Lymphocytes
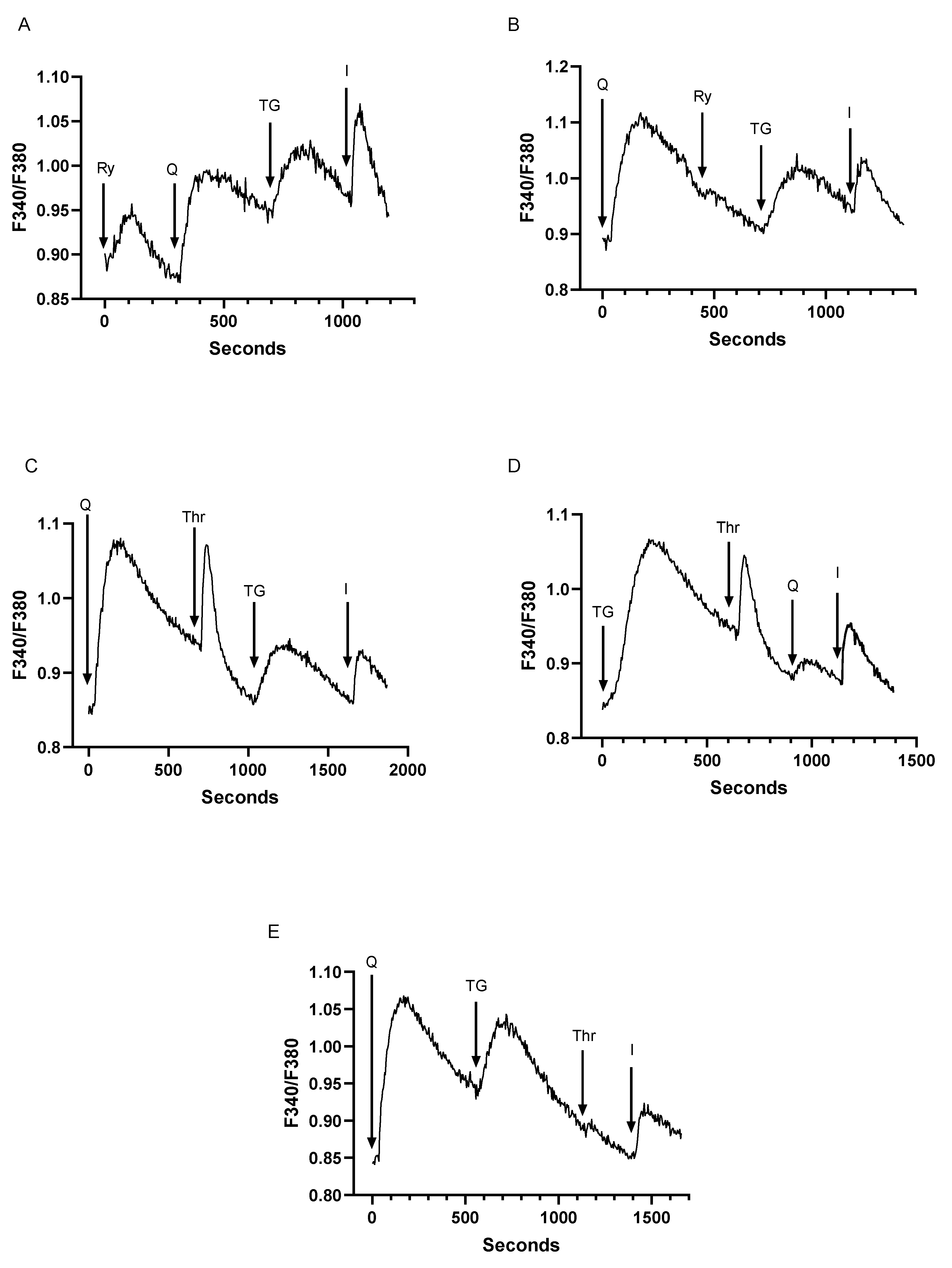
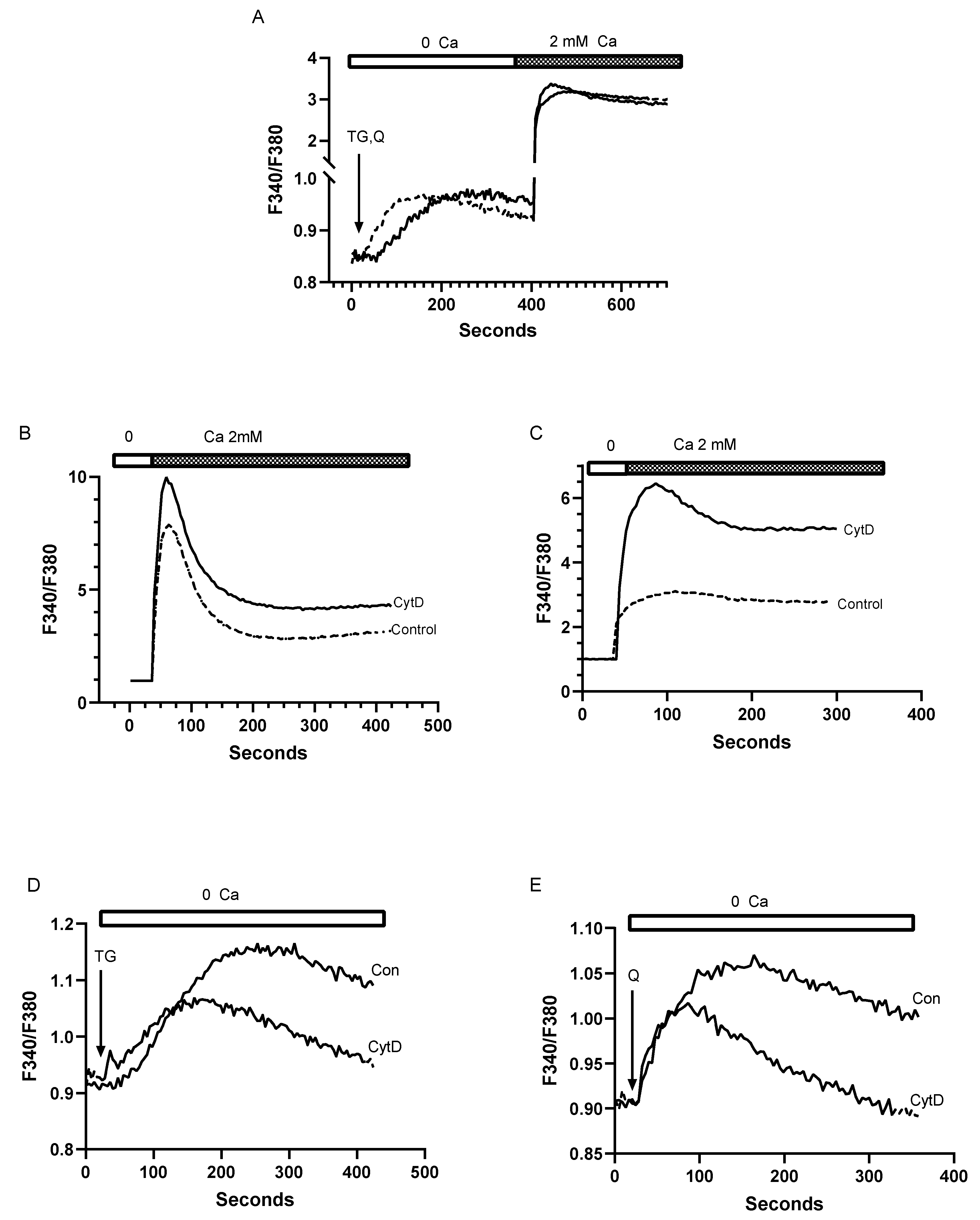
3.4. Despite Differences in SERCA Blocker Sensitivities and Agonist-Mobilizable Ca2+ Responses, the SERCA 2b and SERCA 3-Regulated Ca2+ Stores Exhibit Similar Ca2+ Influx Coupling Actions with Similar Sensitivity to Actin Cytoskeletal Disruption
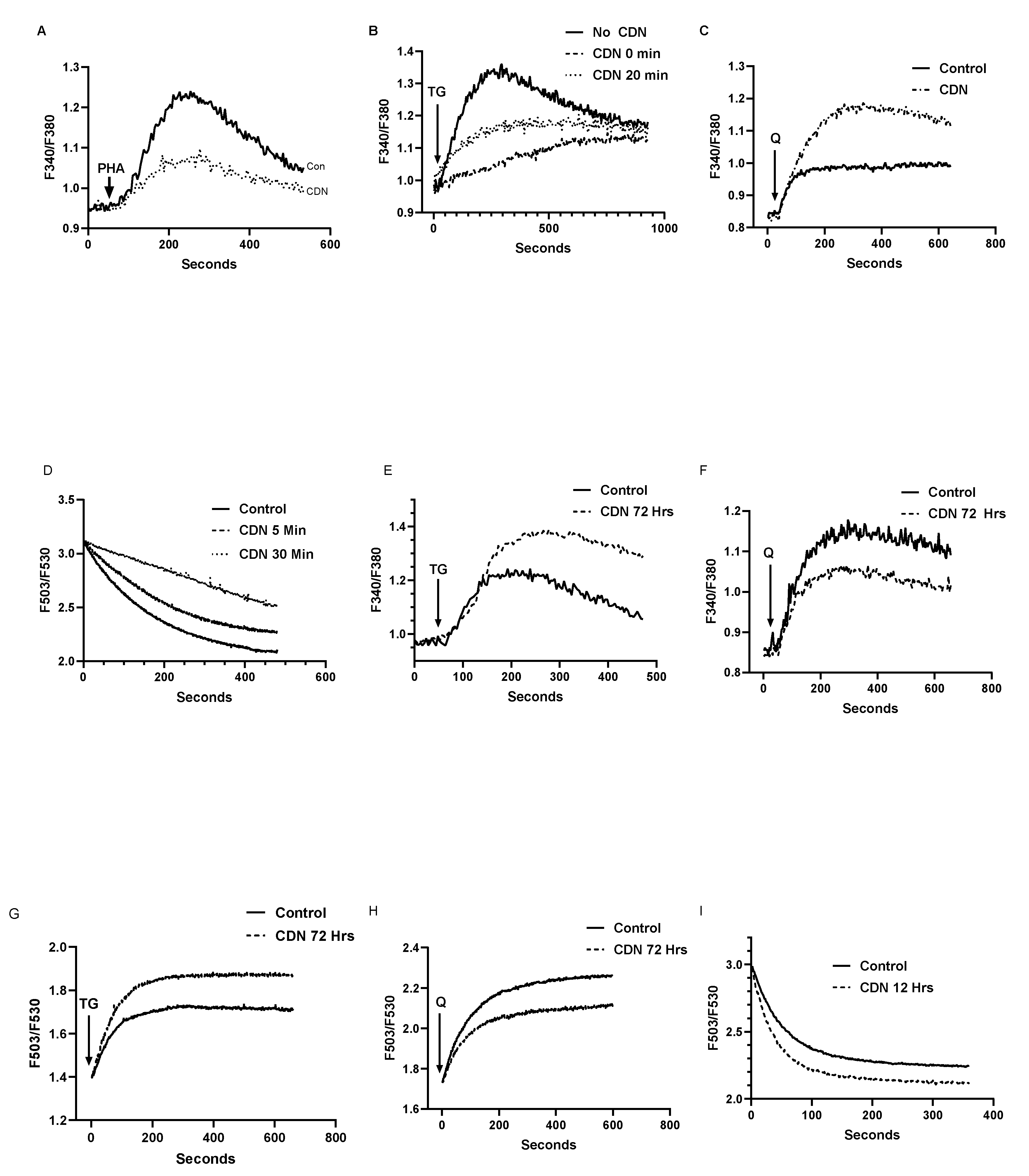
3.5. The SERCA Activator CDN1163 Exerts Complex Short and Long-Term Effects on T Cell Ca2+ Stores Revealing a Differential Regulatory Action on SERCA 2b versus SERCA 3 Ca2+ Pools
4. Discussion
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Conflicts of Interest
References
- Janeway, C.A.; Bottomly, K. Signals and signs for lymphocyte responses. Cell 1994, 76, 275–285. [Google Scholar] [CrossRef]
- Lewis, R.S.; Cahalan, M.D. Ion Channels and Signal Transduction in Lymphocytes. Annu. Rev. Physiol. 1990, 52, 415–430. [Google Scholar] [CrossRef]
- Lewis, R.S. Calcium Signaling Mechanisms in T Lymphocytes. Annu. Rev. Immunol. 2001, 19, 497–521. [Google Scholar] [CrossRef] [PubMed]
- Gil, D.; Diercks, B.-P.; Guse, A.H.; Dupont, G. Three-Dimensional Model of Sub-Plasmalemmal Ca2+ Microdomains Evoked by T Cell Receptor/CD3 Complex Stimulation. Front. Mol. Biosci. 2022, 9, 811145. [Google Scholar] [CrossRef] [PubMed]
- Berridge, M.J.; Lipp, P.; Bootman, M.D. The versatility and universality of calcium signalling. Nat. Rev. Mol. Cell Biol. 2000, 1, 11–21. [Google Scholar] [CrossRef] [PubMed]
- Berridge, M.J. Lymphocyte Activation in Health and Disease. Crit. Rev. Immunol. 2017, 37, 439–462. [Google Scholar] [CrossRef] [PubMed]
- Trebak, M.; Kinet, J.-P. Calcium signalling in T cells. Nat. Rev. Immunol. 2019, 19, 154–169. [Google Scholar] [CrossRef] [PubMed]
- Cornea, R.L.; Gruber, S.J.; Lockamy, E.L.; Muretta, J.M.; Jin, D.; Chen, J.; Dahl, R.; Bartfai, T.; Zsebo, K.M.; Gillispie, G.D.; et al. High-Throughput FRET Assay Yields Allosteric SERCA Activators. SLAS Discov. Adv. Sci. Drug Discov. 2013, 18, 97–107. [Google Scholar] [CrossRef] [PubMed]
- Bidwell, P.A.; Yuen, S.L.; Li, J.; Berg, K.; Rebbeck, R.T.; Aldrich, C.C.; Roopnarine, O.; Cornea, R.L.; Thomas, D.D. A Large-Scale High-Throughput Screen for Modulators of SERCA Activity. Biomolecules 2022, 12, 1789. [Google Scholar] [CrossRef]
- Zhang, C.; Bose, D.D.; Thomas, D.W. Paradoxical effects of sarco/endoplasmic reticulum Ca2+-ATPase (SERCA) activator gingerol on NG115-401L neuronal cells: Failure to augment ER Ca2+ uptake and protect against ER stress-induced cell death. Eur. J. Pharmacol. 2015, 762, 165–173. [Google Scholar] [CrossRef]
- Rahate, K.; Bhatt, L.K.; Prabhavalkar, K.S. SERCA stimulation: A potential approach in therapeutics. Chem. Biol. Drug Des. 2020, 95, 5–15. [Google Scholar] [CrossRef]
- Kang, S.; Dahl, R.; Hsieh, W.; Shin, A.; Zsebo, K.M.; Buettner, C.; Hajjar, R.J.; Lebeche, D. Small Molecular Allosteric Activator of the Sarco/Endoplasmic Reticulum Ca2+-ATPase (SERCA) Attenuates Diabetes and Metabolic Disorders. J. Biol. Chem. 2016, 291, 5185–5198. [Google Scholar] [CrossRef]
- Tadini-Buoninsegni, F.; Smeazzetto, S.; Gualdani, R.; Moncelli, M.R. Drug Interactions With the Ca2+-ATPase From Sarco(Endo)Plasmic Reticulum (SERCA). Front. Mol. Biosci. 2018, 5, 36. [Google Scholar] [CrossRef] [PubMed]
- Rakovskaya, A.; Erofeev, A.; Vinokurov, E.; Pchitskaya, E.; Dahl, R.; Bezprozvanny, I. Positive Allosteric Modulators of SERCA Pump Restore Dendritic Spines and Rescue Long-Term Potentiation Defects in Alzheimer’s Disease Mouse Model. Int. J. Mol. Sci. 2023, 24, 13973. [Google Scholar] [CrossRef]
- Krajnak, K.; Dahl, R. A new target for Alzheimer’s disease: A small molecule SERCA activator is neuroprotective in vitro and improves memory and cognition in APP/PS1 mice. Bioorganic Med. Chem. Lett. 2018, 28, 1591–1594. [Google Scholar] [CrossRef] [PubMed]
- Sordi, G., et al., Stimulation of Ca(2+) -ATPase Transport Activity by a Small-Molecule Drug. ChemMedChem, 2021. 16(21): p. 3293-3299.
- Berridge, M.J. The endoplasmic reticulum: a multifunctional signaling organelle. Cell Calcium 2002, 32, 235–249. [Google Scholar] [CrossRef]
- Berridge, M.J.; Bootman, M.D.; Roderick, H.L. Calcium signalling: Dynamics, homeostasis and remodelling. Nat. Rev. Mol. Cell Biol. 2003, 4, 517–529. [Google Scholar] [CrossRef]
- Chemaly, E.R.; Troncone, L.; Lebeche, D. SERCA control of cell death and survival. Cell Calcium 2018, 69, 46–61. [Google Scholar] [CrossRef] [PubMed]
- Inesi, G.; Sagara, Y. Specific inhibitors of intracellular Ca2+ transport ATPases. J. Membr. Biol. 1994, 141, 1–6. [Google Scholar] [CrossRef]
- Inesi, G.; Hua, S.; Xu, C.; Ma, H.; Seth, M.; Prasad, A.M.; Sumbilla, C. Studies of Ca2+ ATPase (SERCA) Inhibition. J. Bioenerg. Biomembr. 2005, 37, 365–368. [Google Scholar] [CrossRef]
- Lytton, J.; Westlin, M.; Hanley, M.R. Thapsigargin inhibits the sarcoplasmic or endoplasmic reticulum Ca-ATPase family of calcium pumps. J. Biol. Chem. 1991, 266, 17067–17071. [Google Scholar] [CrossRef] [PubMed]
- Thomas, D. and M.R. Hanley, Pharmacological tools for perturbing intracellular calcium storage. Methods Cell Biol, 1994. 40: p. 65-89. [CrossRef]
- Mengeste, A.M.; Lund, J.; Katare, P.; Ghobadi, R.; Bakke, H.G.; Lunde, P.K.; Eide, L.; Mahony, G.O.; Göpel, S.; Peng, X.-R.; et al. The small molecule SERCA activator CDN1163 increases energy metabolism in human skeletal muscle cells. Curr. Res. Pharmacol. Drug Discov. 2021, 2, 100060. [Google Scholar] [CrossRef] [PubMed]
- Britzolaki, A.; Cronin, C.C.; Flaherty, P.R.; Rufo, R.L.; Pitychoutis, P.M. Chronic but not acute pharmacological activation of SERCA induces behavioral and neurochemical effects in male and female mice. Behav. Brain Res. 2021, 399, 112984–112984. [Google Scholar] [CrossRef] [PubMed]
- Nguyen, H.T., et al., CDN1163, an activator of sarco/endoplasmic reticulum Ca(2+) ATPase, up-regulates mitochondrial functions and protects against lipotoxicity in pancreatic beta-cells. Br J Pharmacol, 2023. 180(21): p. 2762-2776. [CrossRef]
- Guse, A.H.; Roth, E.; Emmrich, F. Intracellular Ca2+ pools in Jurkat T-lymphocytes. Biochem. J. 1993, 291 Pt 2, 447–451. [Google Scholar] [CrossRef] [PubMed]
- Dadsetan, S.; Zakharova, L.; Molinski, T.F.; Fomina, A.F. Store-operated Ca2+ Influx Causes Ca2+ Release from the Intracellular Ca2+ Channels That Is Required for T Cell Activation. J. Biol. Chem. 2008, 283, 12512–12519. [Google Scholar] [CrossRef]
- Astoul, E. Astoul, E., et al., PI 3-K and T-cell activation: limitations of T-leukemic cell lines as signaling models. Trends Immunol, 2001. 22(9): p. 490-6. [CrossRef]
- Benson, J.C.; Romito, O.; Abdelnaby, A.E.; Xin, P.; Pathak, T.; Weir, S.E.; Kirk, V.; Castaneda, F.; Yoast, R.E.; Emrich, S.M.; et al. A multiple-oscillator mechanism underlies antigen-induced Ca2+ oscillations in Jurkat T-cells. J. Biol. Chem. 2023, 299, 105310. [Google Scholar] [CrossRef]
- Zani, F.; Blagih, J.; Gruber, T.; Buck, M.D.; Jones, N.; Hennequart, M.; Newell, C.L.; Pilley, S.E.; Soro-Barrio, P.; Kelly, G.; et al. The dietary sweetener sucralose is a negative modulator of T cell-mediated responses. Nature 2023, 615, 705–711. [Google Scholar] [CrossRef]
- Wuytack, F.; Papp, B.; Verboomen, H.; Raeymaekers, L.; Dode, L.; Bobe, R.; Enouf, J.; Bokkala, S.; Authi, K.; Casteels, R. A sarco/endoplasmic reticulum Ca(2+)-ATPase 3-type Ca2+ pump is expressed in platelets, in lymphoid cells, and in mast cells. J. Biol. Chem. 1994, 269, 1410–1416. [Google Scholar] [CrossRef]
- Wu, K.D.; Lee, W.S.; Wey, J.; Bungard, D.; Lytton, J. Localization and quantification of endoplasmic reticulum Ca(2+)-ATPase isoform transcripts. Am. J. Physiol. Physiol. 1995, 269 Pt 1, C775–C784. [Google Scholar] [CrossRef]
- Poch, E.; Leach, S.; Snape, S.; Cacic, T.; MacLennan, D.H.; Lytton, J.; Wray, S.; Burdyga, T.; Chandrasekera, P.C.; Kargacin, M.E.; et al. Functional characterization of alternatively spliced human SERCA3 transcripts. Am. J. Physiol. 1998, 275, C1449–C1458. [Google Scholar] [CrossRef]
- Kovacs, T., et al., All three splice variants of the human sarco/endoplasmic reticulum Ca2+-ATPase 3 gene are translated to proteins: a study of their co-expression in platelets and lymphoid cells. Biochem J, 2001. 358(Pt 3): p. 559-68. [CrossRef]
- Cavallini, L.; Coassin, M.; Alexandre, A. Two classes of agonist-sensitive Ca2+ stores in platelets, as identified by their differential sensitivity to 2,5-di-(tert-butyl)-1,4-benzohydroquinone and thapsigargin. Biochem. J. 1995, 310 Pt 2, 449–452. [Google Scholar] [CrossRef]
- Papp, B.; Enyedi, A.; Pászty, K.; Kovács, T.; Sarkadi, B.; Gárdos, G.; Magnier, C.; Wuytack, F.; Enouf, J. Simultaneous presence of two distinct endoplasmic-reticulum-type calcium-pump isoforms in human cells. Characterization by radio-immunoblotting and inhibition by 2,5-di-(t-butyl)-1,4-benzohydroquinone. Biochem. J. 1992, 288 Pt 1, 297–302. [Google Scholar] [CrossRef]
- López, J.J.; Camello-Almaraz, C.; Pariente, J.A.; Salido, G.M.; Rosado, J.A. Ca2+ accumulation into acidic organelles mediated by Ca2+- and vacuolar H+-ATPases in human platelets. Biochem. J. 2005, 390 Pt 1, 243–252. [Google Scholar] [CrossRef]
- Jardín, I.; López, J.J.; Pariente, J.A.; Salido, G.M.; Rosado, J.A. Intracellular Calcium Release from Human Platelets: Different Messengers for Multiple Stores. Trends Cardiovasc. Med. 2008, 18, 57–61. [Google Scholar] [CrossRef]
- Jardin, I.; Ben Amor, N.; Bartegi, A.; Pariente, J.A.; Salido, G.M.; Rosado, J.A. Differential involvement of thrombin receptors in Ca2+ release from two different intracellular stores in human platelets. Biochem. J. 2007, 401, 167–174. [Google Scholar] [CrossRef]
- López, J.J.; Redondo, P.C.; Salido, G.M.; Pariente, J.A.; Rosado, J.A. Two distinct Ca2+ compartments show differential sensitivity to thrombin, ADP and vasopressin in human platelets. Cell. Signal. 2006, 18, 373–381. [Google Scholar] [CrossRef] [PubMed]
- Elaïb, Z.; Adam, F.; Berrou, E.; Bordet, J.-C.; Prévost, N.; Bobe, R.; Bryckaert, M.; Rosa, J.-P. Full activation of mouse platelets requires ADP secretion regulated by SERCA3 ATPase–dependent calcium stores. Blood 2016, 128, 1129–1138. [Google Scholar] [CrossRef]
- Fomina, A.F. Neglected wardens: T lymphocyte ryanodine receptors. J. Physiol. 2021, 599, 4415–4426. [Google Scholar] [CrossRef]
- Thakur, P.; Dadsetan, S.; Fomina, A.F. Bidirectional Coupling between Ryanodine Receptors and Ca2+ Release-activated Ca2+ (CRAC) Channel Machinery Sustains Store-operated Ca2+ Entry in Human T Lymphocytes. J. Biol. Chem. 2012, 287, 37233–37244. [Google Scholar] [CrossRef] [PubMed]
- Guse, A.H.; Diercks, B. Integration of nicotinic acid adenine dinucleotide phosphate (NAADP)-dependent calcium signalling. J. Physiol. 2018, 596, 2735–2743. [Google Scholar] [CrossRef] [PubMed]
- Wolf, I.M.A.; Guse, A.H. Ca2+ Microdomains in T-Lymphocytes. Front. Oncol. 2017, 7, 73. [Google Scholar] [CrossRef]
- Schwarzmann, N.; Kunerth, S.; Weber, K.; Mayr, G.W.; Guse, A.H. Knock-down of the Type 3 Ryanodine Receptor Impairs Sustained Ca2+ Signaling via the T Cell Receptor/CD3 Complex. J. Biol. Chem. 2002, 277, 50636–50642. [Google Scholar] [CrossRef] [PubMed]
- Hohenegger, M.; Berg, I.; Weigl, L.; Mayr, G.W.; Potter, B.V.L.; Guse, A.H. Pharmacological activation of the ryanodine receptor in Jurkat T-lymphocytes. Br. J. Pharmacol. 1999, 128, 1235–1240. [Google Scholar] [CrossRef] [PubMed]
- Dammermann, W.; Guse, A.H. Functional Ryanodine Receptor Expression Is Required for NAADP-mediated Local Ca2+ Signaling in T-lymphocytes. J. Biol. Chem. 2005, 280, 21394–21399. [Google Scholar] [CrossRef] [PubMed]
- Steen, M.; Kirchberger, T.; Guse, A.H. NAADP Mobilizes Calcium from the Endoplasmic Reticular Ca2+ Store in T-lymphocytes. J. Biol. Chem. 2007, 282, 18864–18871. [Google Scholar] [CrossRef] [PubMed]
- Rosado, J. Acidic Ca2+ stores in platelets. Cell Calcium 2011, 50, 168–174. [Google Scholar] [CrossRef] [PubMed]
- Feng, M.; Elaïb, Z.; Borgel, D.; Denis, C.V.; Adam, F.; Bryckaert, M.; Rosa, J.-P.; Bobe, R. NAADP/SERCA3-Dependent Ca 2+ Stores Pathway Specifically Controls Early Autocrine ADP Secretion Potentiating Platelet Activation. Circ. Res. 2020, 127, E166–E183. [Google Scholar] [CrossRef] [PubMed]
- Rosado, J.A.; López, J.J.; Harper, A.G.S.; Harper, M.T.; Redondo, P.C.; Pariente, J.A.; Sage, S.O.; Salido, G.M. Two Pathways for Store-mediated Calcium Entry Differentially Dependent on the Actin Cytoskeleton in Human Platelets. J. Biol. Chem. 2004, 279, 29231–29235. [Google Scholar] [CrossRef] [PubMed]
- Huang, Y.-P.; Huang, W.-W.; Tsai, K.-F.; Shiao, L.-R.; Yang, Z.-H.; Tseng, S.-Y.; Lin, Y.-H.; Chen, C.-Y.; Chan, P.; Leung, Y.-M. CDN1163, a SERCA activator, causes intracellular Ca2+ leak, mitochondrial hyperpolarization and cell cycle arrest in mouse neuronal N2A cells. NeuroToxicology 2023, 98, 9–15. [Google Scholar] [CrossRef]
- Wu, K.-C.; Chen, C.-Y.; Chuang, C.-M.; Shiao, L.-R.; Chan, P.; Leung, Y.-M. Suppression of Ca2+ oscillations by SERCA inhibition in human alveolar type 2 A549 cells: rescue by ochratoxin A but not CDN1163. Life Sci. 2022, 308, 120913. [Google Scholar] [CrossRef]
- Kunerth, S.; Mayr, G.W.; Koch-Nolte, F.; Guse, A.H. Analysis of subcellular calcium signals in T-lymphocytes. Cell. Signal. 2003, 15, 783–792. [Google Scholar] [CrossRef] [PubMed]
- Launay, S.; Bobe, R.; Lacabaratz-Porret, C.; Bredoux, R.; Kovàcs, T.; Enouf, J.; Papp, B. Modulation of Endoplasmic Reticulum Calcium Pump Expression during T Lymphocyte Activation. J. Biol. Chem. 1997, 272, 10746–10750. [Google Scholar] [CrossRef] [PubMed]
- Bobe, R.; Bredoux, R.; Corvazier, E.; Andersen, J.P.; Clausen, J.D.; Dode, L.; Kovács, T.; Enouf, J. Identification, Expression, Function, and Localization of a Novel (Sixth) Isoform of the Human Sarco/Endoplasmic Reticulum Ca2+ATPase 3 Gene. J. Biol. Chem. 2004, 279, 24297–24306. [Google Scholar] [CrossRef] [PubMed]
- Arbabian, A.; Brouland, J.-P.; Gélébart, P.; Kovàcs, T.; Bobe, R.; Enouf, J.; Papp, B. Endoplasmic reticulum calcium pumps and cancer. BioFactors 2011, 37, 139–149. [Google Scholar] [CrossRef]
- Papp, B.; Brouland, J.-P.; Arbabian, A.; Gélébart, P.; Kovács, T.; Bobe, R.; Enouf, J.; Varin-Blank, N.; Apáti. Endoplasmic Reticulum Calcium Pumps and Cancer Cell Differentiation. Biomolecules 2012, 2, 165–186. [Google Scholar] [CrossRef]
- López, J.J.; Jardín, I.; Bobe, R.; Pariente, J.A.; Enouf, J.; Salido, G.M.; Rosado, J.A. STIM1 regulates acidic Ca2+ store refilling by interaction with SERCA3 in human platelets. Biochem. Pharmacol. 2008, 75, 2157–2164. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).



