Submitted:
06 February 2024
Posted:
07 February 2024
Read the latest preprint version here
Abstract
Keywords:
Introduction
2. Methods
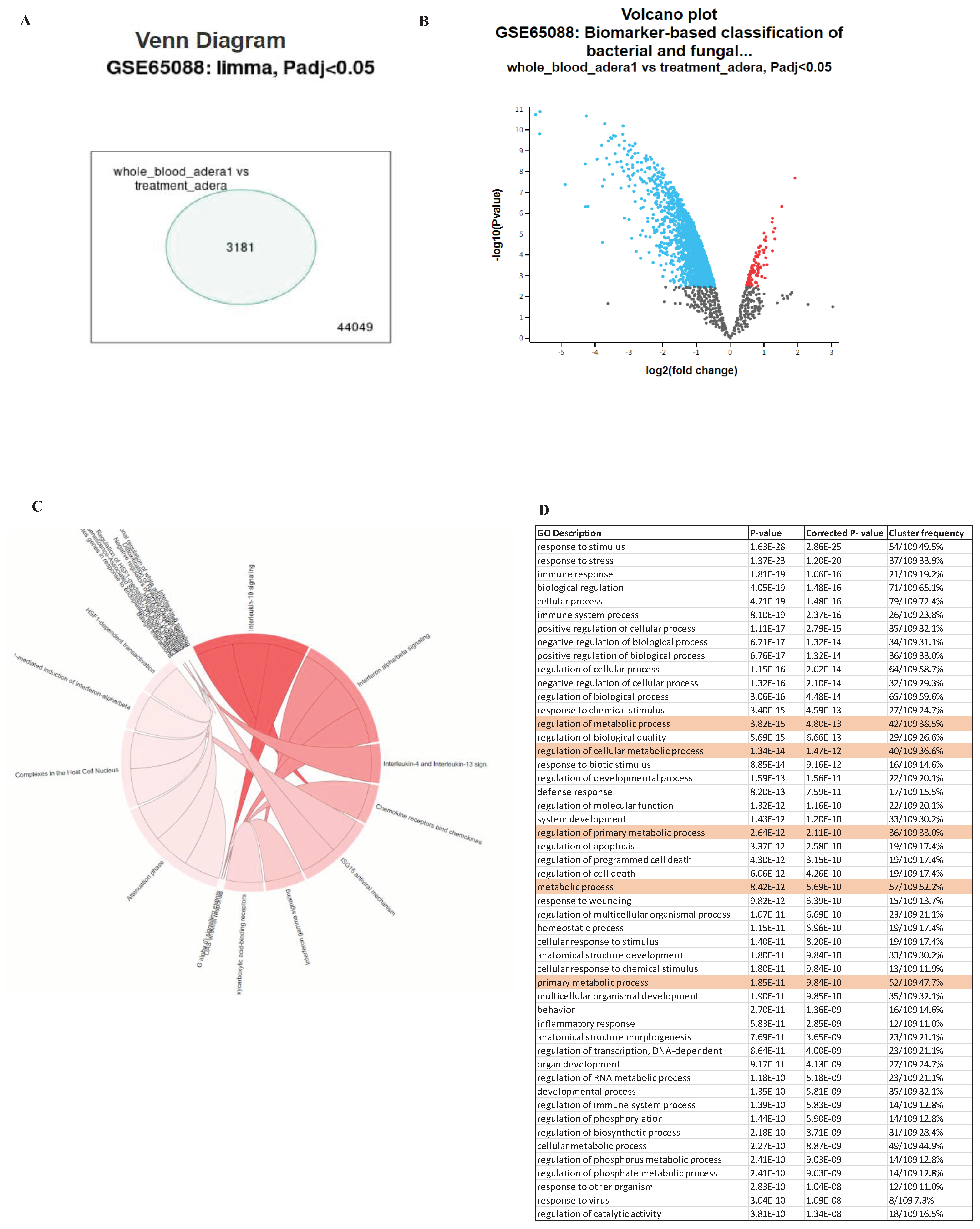
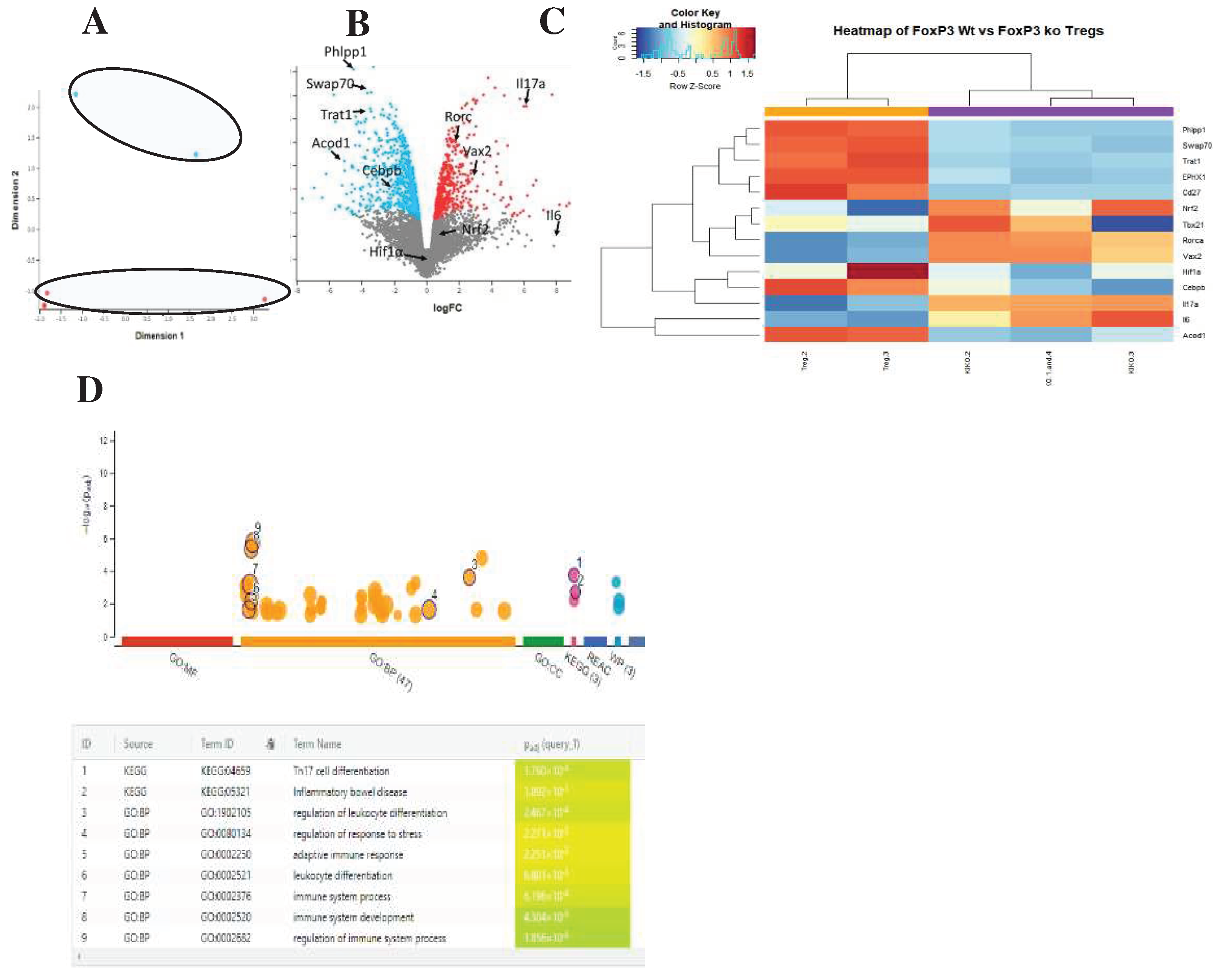
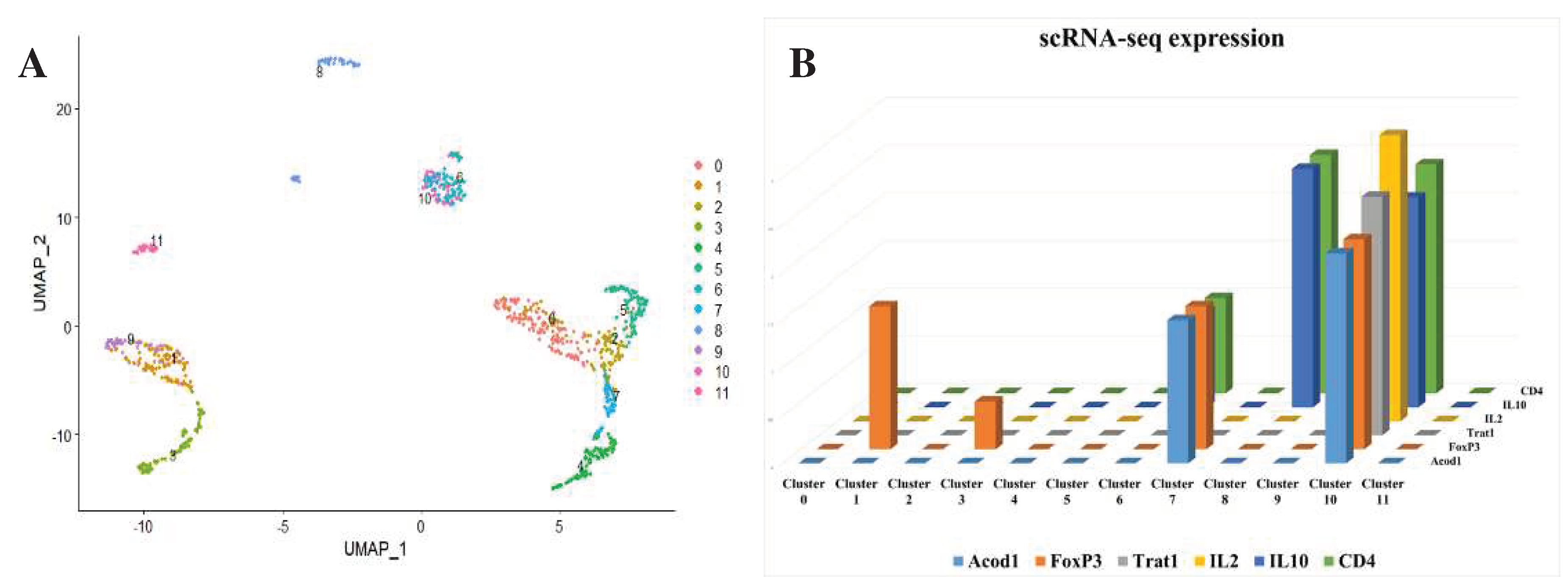
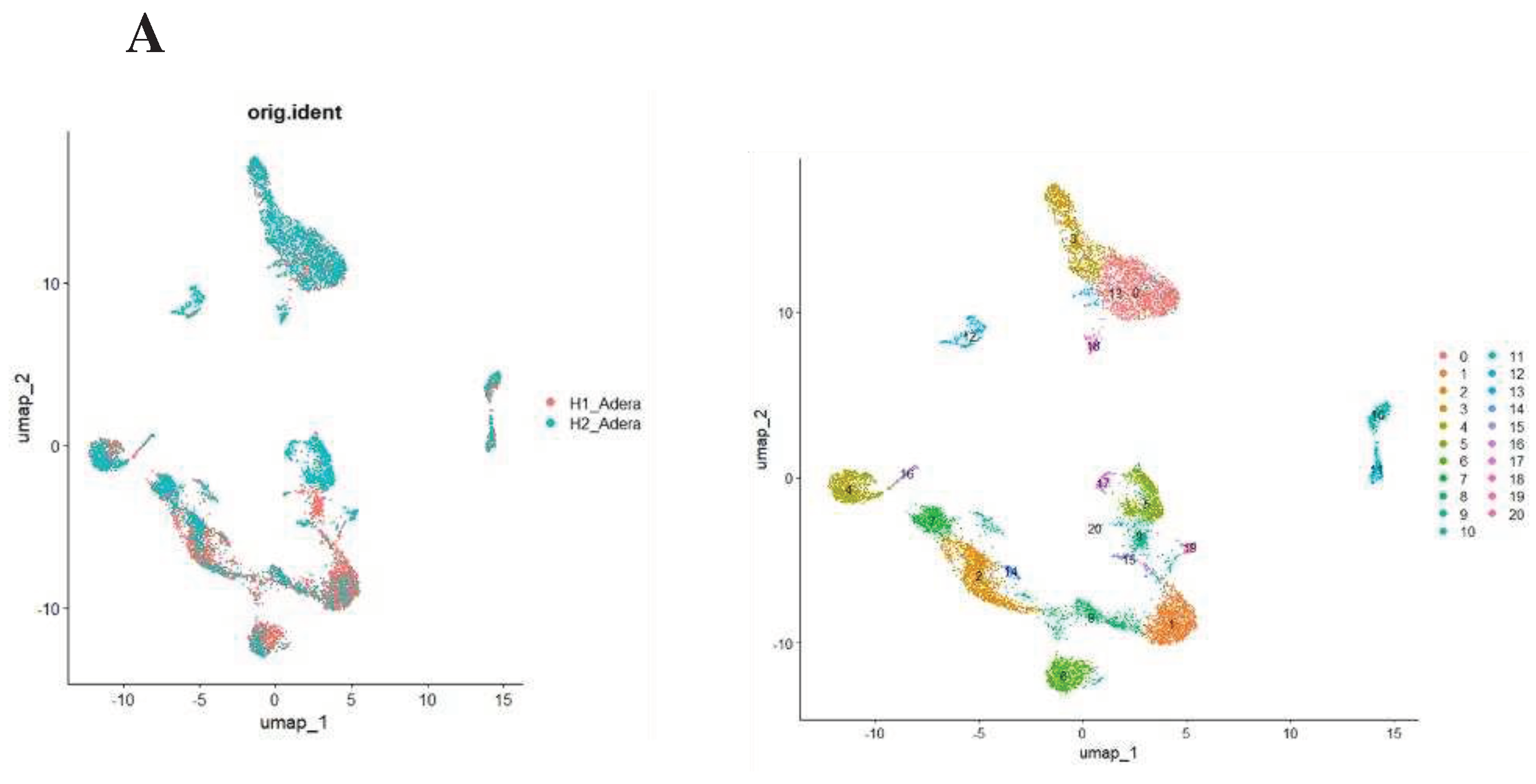
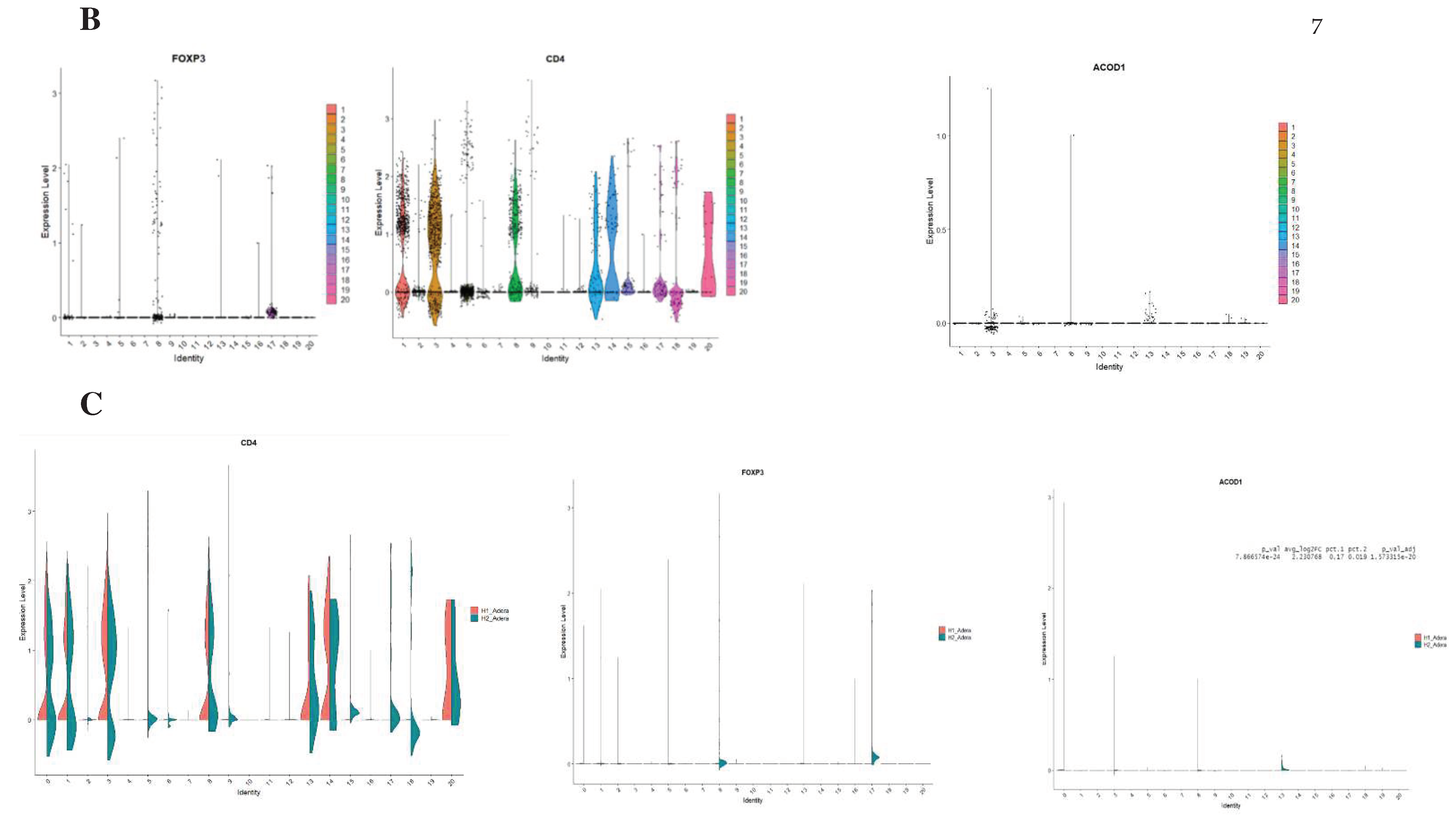
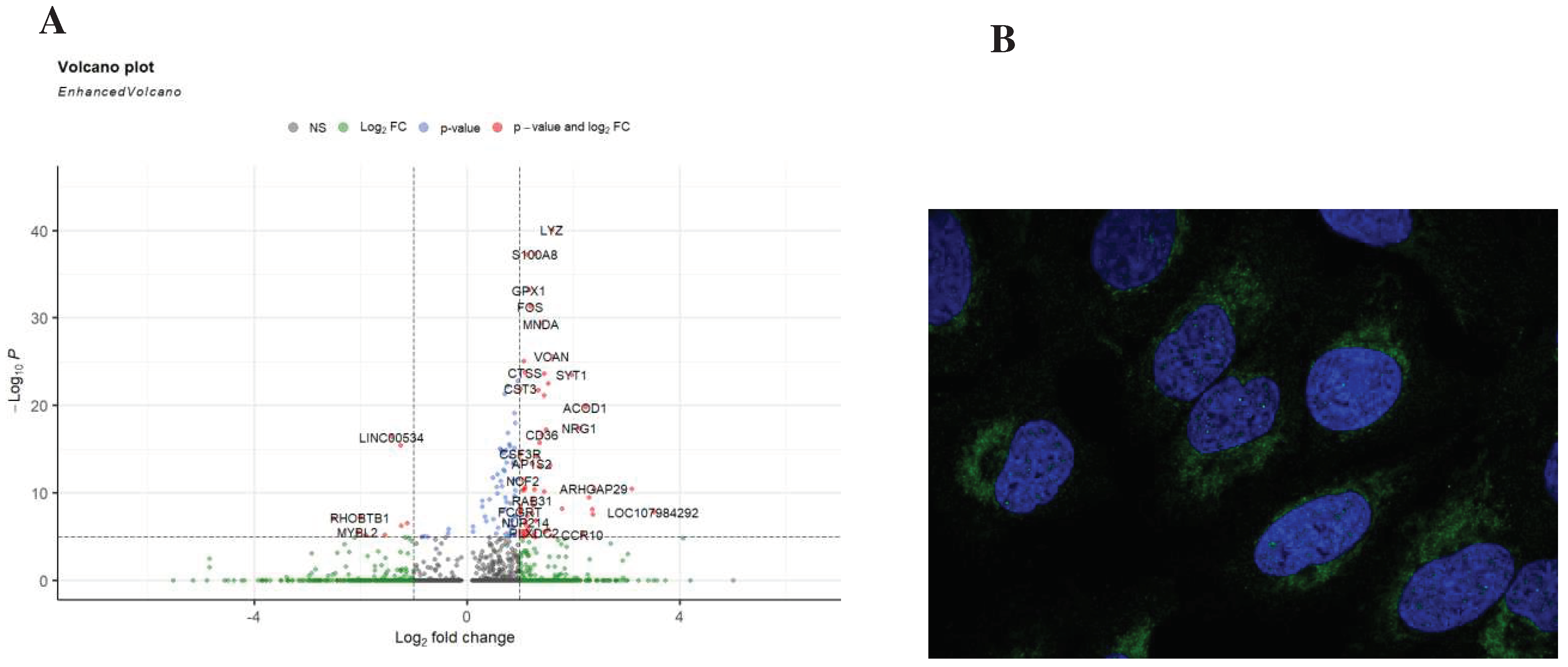
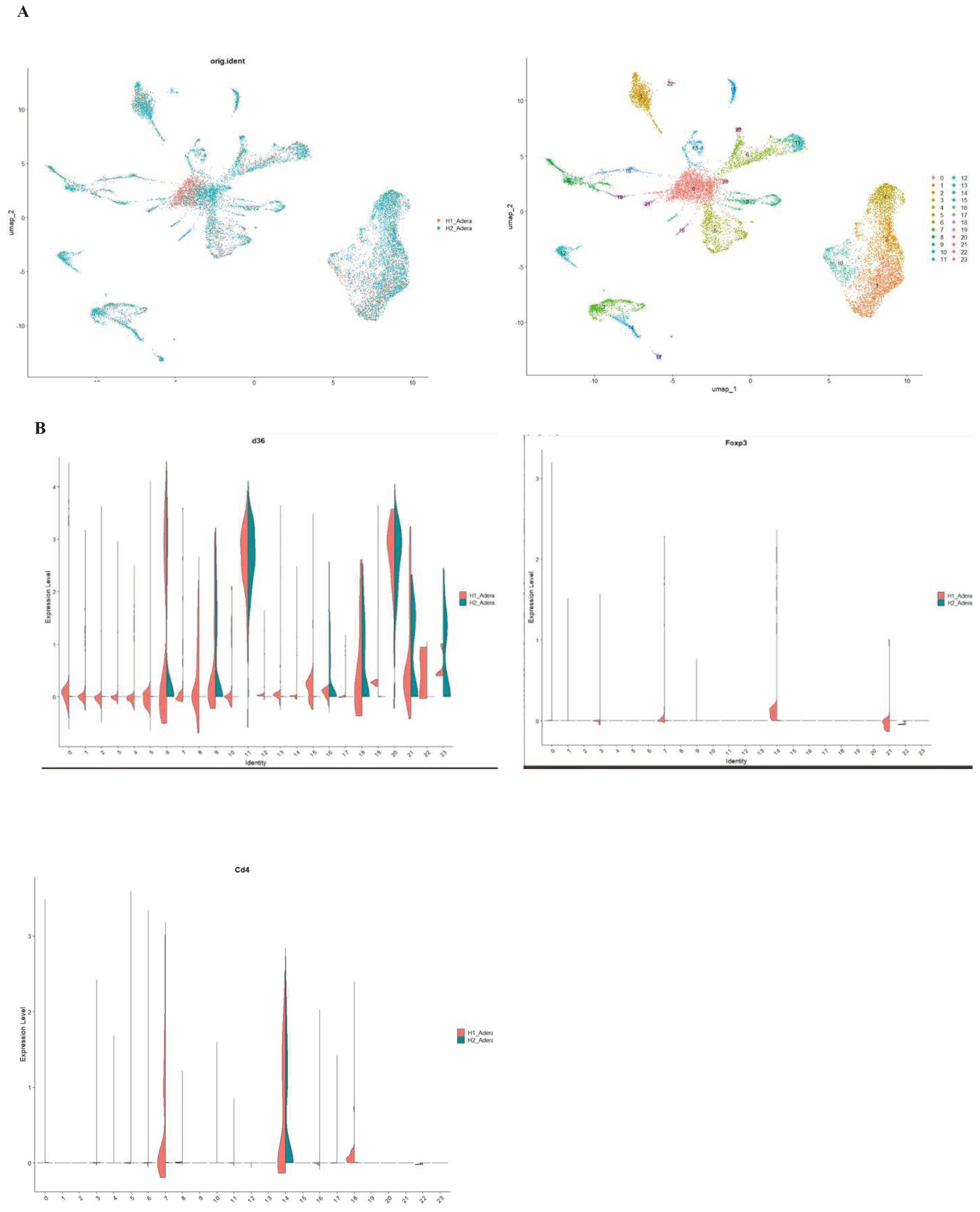
Results
Discussion
References
- Montero-Jodra, A.; de la Fuente, M.Á.; Gobelli, D.; Martín-Fernández, M.; Villar, J.; Tamayo, E.; Simarro, M. The Mitochondrial Signature of Cultured Endothelial Cells in Sepsis: Identifying Potential Targets for Treatment. Biochim. Biophys. Acta - Mol. Basis Dis. 2024, 1870, 166946. [Google Scholar] [CrossRef]
- Polat, G.; Ugan, R.A.; Cadirci, E.; Halici, Z. Sepsis and Septic Shock: Current Treatment Strategies and New Approaches. Eurasian J. Med. 2017. [Google Scholar] [CrossRef]
- Grondman, I.; Pirvu, A.; Riza, A.; Ioana, M.; Netea, M.G. Biomarkers of Inflammation and the Etiology of Sepsis. Biochem. Soc. Trans. 2021. [Google Scholar] [CrossRef]
- Ebrahim, G.J. Sepsis, Septic Shock and the Systemic Inflammatory Response Syndrome. J. Trop. Pediatr. 2011. [Google Scholar] [CrossRef]
- Delano, M.J.; Ward, P.A. The Immune System’s Role in Sepsis Progression, Resolution, and Long-Term Outcome. Immunol. Rev. 2016. [Google Scholar] [CrossRef]
- Boomer, J.S.; Green, J.M.; Hotchkiss, R.S. The Changing Immune System in Sepsis. Virulence 2014. [Google Scholar] [CrossRef]
- Christaki, E.; Anyfanti, P.; Opal, S.M. Immunomodulatory Therapy for Sepsis: An Update. Expert Rev. Anti. Infect. Ther. 2011. [Google Scholar] [CrossRef]
- Hein, F.; Massin, F.; Cravoisy-Popovic, A.; Barraud, D.; Levy, B.; Bollaert, P.-E.; Gibot, S. The Relationship between CD4+CD25+CD127- Regulatory T Cells and Inflammatory Response and Outcome during Shock States. Crit. Care 2010, 14, R19. [Google Scholar] [CrossRef]
- Cao, C.; Ma, T.; Chai, Y.; Shou, S. The Role of Regulatory T Cells in Immune Dysfunction during Sepsis. World J. Emerg. Med. 2015. [Google Scholar] [CrossRef]
- Drechsler, S.; Osuchowski, M. Cecal Ligation and Puncture. In Methods in Molecular Biology; 2021. [Google Scholar]
- Taylor, A.L.; Llewelyn, M.J. Superantigen-Induced Proliferation of Human CD4+CD25− T Cells Is Followed by a Switch to a Functional Regulatory Phenotype. J. Immunol. 2010. [Google Scholar] [CrossRef]
- Bhaumik, S.; Łazarczyk, M.; Kubick, N.; Klimovich, P.; Gurba, A.; Paszkiewicz, J.; Teodorowicz, P.; Kocki, T.; Horbańczuk, J.O.; Manda, G.; et al. Investigation of the Molecular Evolution of Treg Suppression Mechanisms Indicates a Convergent Origin. Curr. Issues Mol. Biol. 2023, 45, 628–648. [Google Scholar] [CrossRef]
- Jiang, L.N.; Yao, Y.M.; Sheng, Z.Y. The Role of Regulatory T Cells in the Pathogenesis of Sepsis and Its Clinical Implication. J. Interf. Cytokine Res. 2012. [CrossRef]
- Bergmann, C.B.; Beckmann, N.; Salyer, C.E.; Hanschen, M.; Crisologo, P.A.; Caldwell, C.C. Potential Targets to Mitigate Trauma- or Sepsis-Induced Immune Suppression. Front. Immunol. 2021. [Google Scholar] [CrossRef]
- Monneret, G.; Venet, F.; Pachot, A.; Lepape, A. Monitoring Immune Dysfunctions in the Septic Patient: A New Skin for the Old Ceremony. Mol. Med. 2008. [CrossRef]
- Rizzo, A.; Giovangiulio, M. Di; Stolfi, C.; Franze, E.; Fehling, H.J.; Carsetti, R.; Giorda, E.; Colantoni, A.; Ortenzi, A.; Rugge, M.; et al. RORGT-Expressing Tregs Drive the Growth of Colitis-Associated Colorectal Cancer by Controlling IL6 in Dendritic Cells. Cancer Immunol. Res. 2018, 6, 1082–1092. [Google Scholar] [CrossRef]
- Mickael, M.E.; Bhaumik, S.; Chakraborti, A.; Umfress, A.A.; van Groen, T.; Macaluso, M.; Totenhagen, J.; Sorace, A.G.; Bibb, J.A.; Standaert, D.G.; et al. RORγt-Expressing Pathogenic CD4 + T Cells Cause Brain Inflammation during Chronic Colitis. J. Immunol. 2022, 208, 2054–2066. [Google Scholar] [CrossRef]
- Cakir, T.; Pir, P.; Vis, D.; Linde, J.; Dix, A.; Hünniger, K.; Weber, M.; Guthke, R.; Kurzai, O. Biomarker-Based Classification of Bacterial and Fungal Whole-Blood Infections in a Genome-Wide Expression Study. Front. Microbiol. | www.frontiersin.org 2015, 6, 171. [Google Scholar] [CrossRef]
- van der Veeken, J.; Campbell, C.; Pritykin, Y.; Schizas, M.; Verter, J.; Hu, W.; Wang, Z.-M.; Matheis, F.; Mucida, D.; Charbonnier, L.-M.; et al. Genetic Tracing Reveals Transcription Factor Foxp3-Dependent and Foxp3-Independent Functionality of Peripherally Induced Treg Cells. Immunity 2022, 55, 1173–1184.e7. [Google Scholar] [CrossRef]
- Kubick, N.; Pajares, M.; Enache, I.; Manda, G.; Mickael, M.-E. Repurposing Zileuton as a Depression Drug Using an AI and In Vitro Approach. Molecules 2020, 25, 2155. [Google Scholar] [CrossRef]
- Mickael, M.E.; Kubick, N.; Łazarczyk, M.; Sacharczuk, M.; Marchewka, J.; Urbański, P.; Horbańczuk, J.O. Transcriptome Analysis of the Th17/Treg Axis Reveals Multiple Pathways That Ensure Distinct Differentiation Patterns. Anim. Sci. Pap. Reports 2023, 41, 79–93. [Google Scholar]
- Satija, R.; Farrell, J.A.; Gennert, D.; Schier, A.F.; Regev, A. Spatial Reconstruction of Single-Cell Gene Expression Data. Nat. Biotechnol. 2015. [Google Scholar] [CrossRef] [PubMed]
- Wu, R.; Chen, F.; Wang, N.; Tang, D.; Kang, R. ACOD1 in Immunometabolism and Disease. Cell. Mol. Immunol. 2020, 17, 822–833. [Google Scholar] [CrossRef]
- Wang, H.; Franco, F.; Tsui, Y.C.; Xie, X.; Trefny, M.P.; Zappasodi, R.; Mohmood, S.R.; Fernández-García, J.; Tsai, C.H.; Schulze, I.; et al. CD36-Mediated Metabolic Adaptation Supports Regulatory T Cell Survival and Function in Tumors. Nat. Immunol. 2020. [Google Scholar] [CrossRef]
- Horton, B.L.; Spranger, S. CD36 — the Achilles’ Heel of Treg Cells. Nat. Immunol. 2020. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).



