Submitted:
06 February 2024
Posted:
07 February 2024
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Results
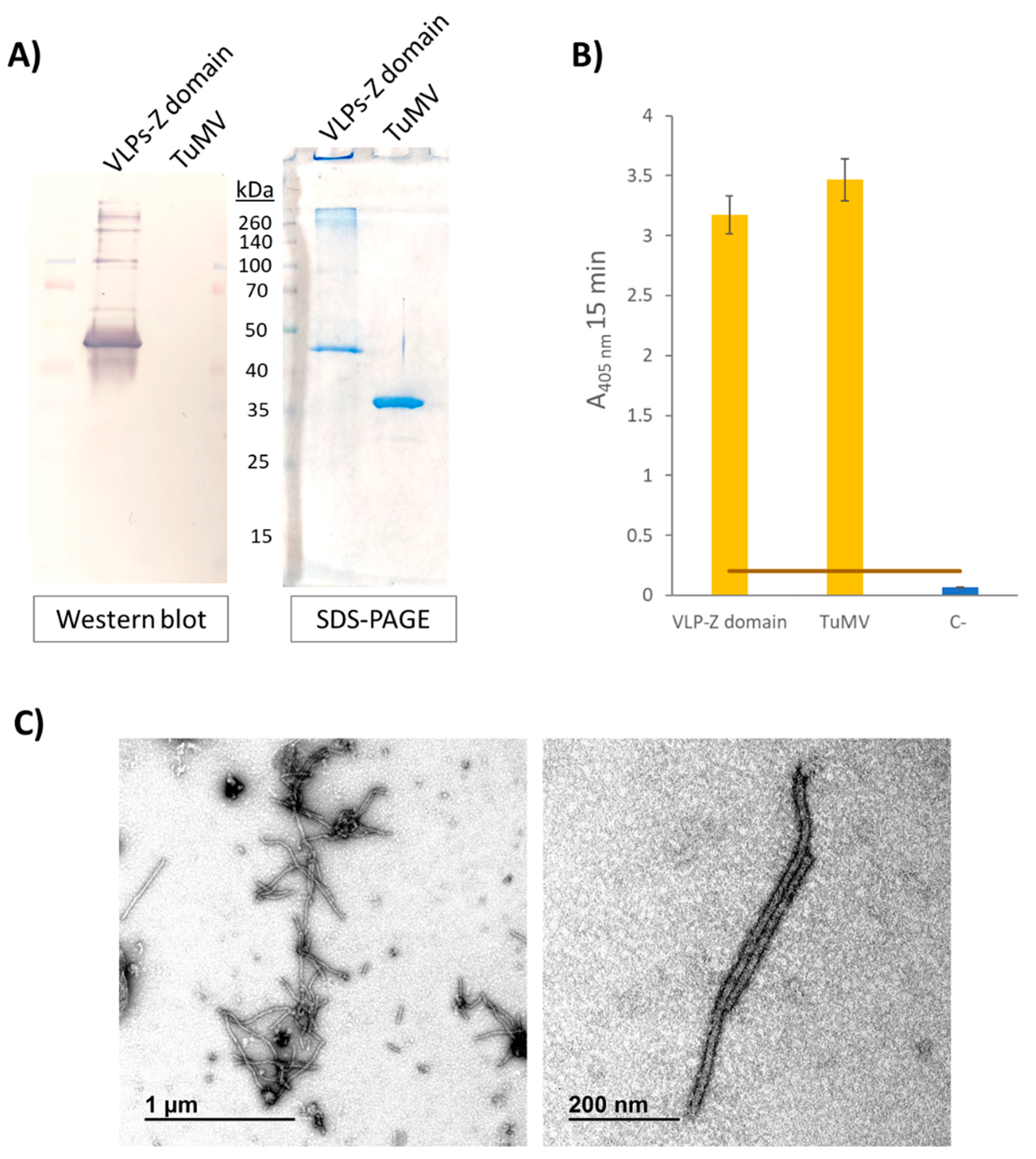
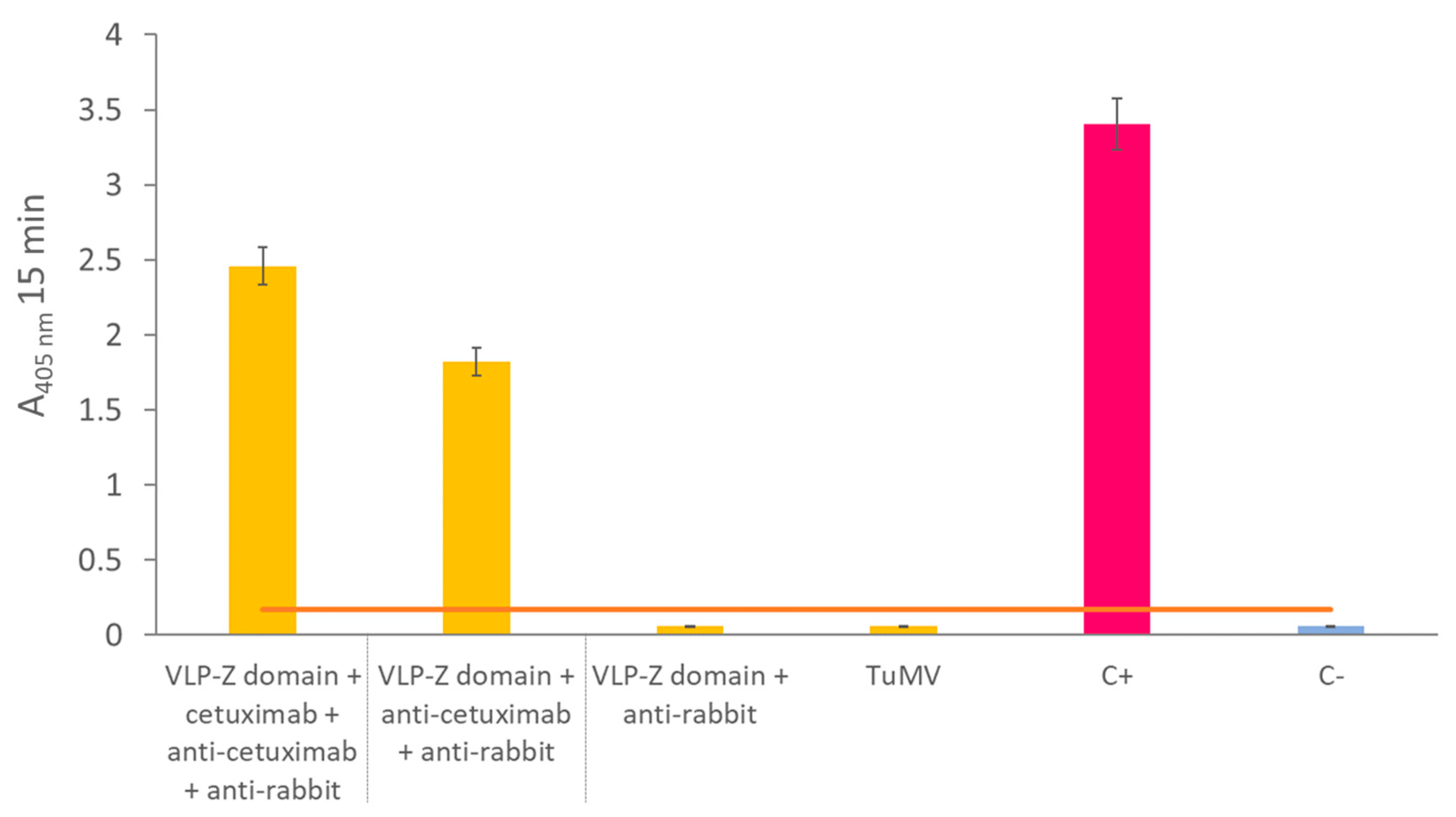
2.1. VLP Purification and Cetuximab Functionalization Check
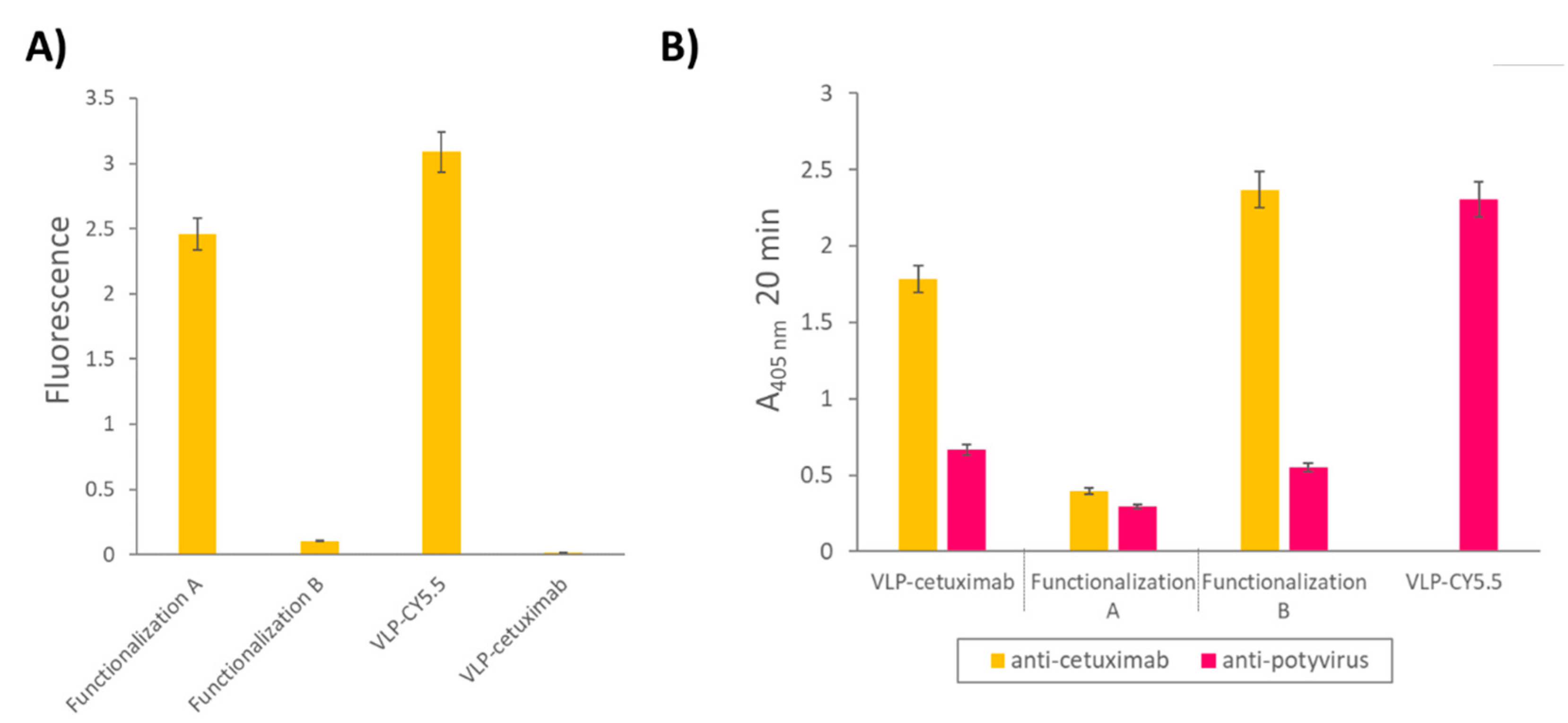
2.2. Triple Functionalization of VLP-Z Domain with Cetuximab and Cy5.5
2.3. Cell Viability Assay
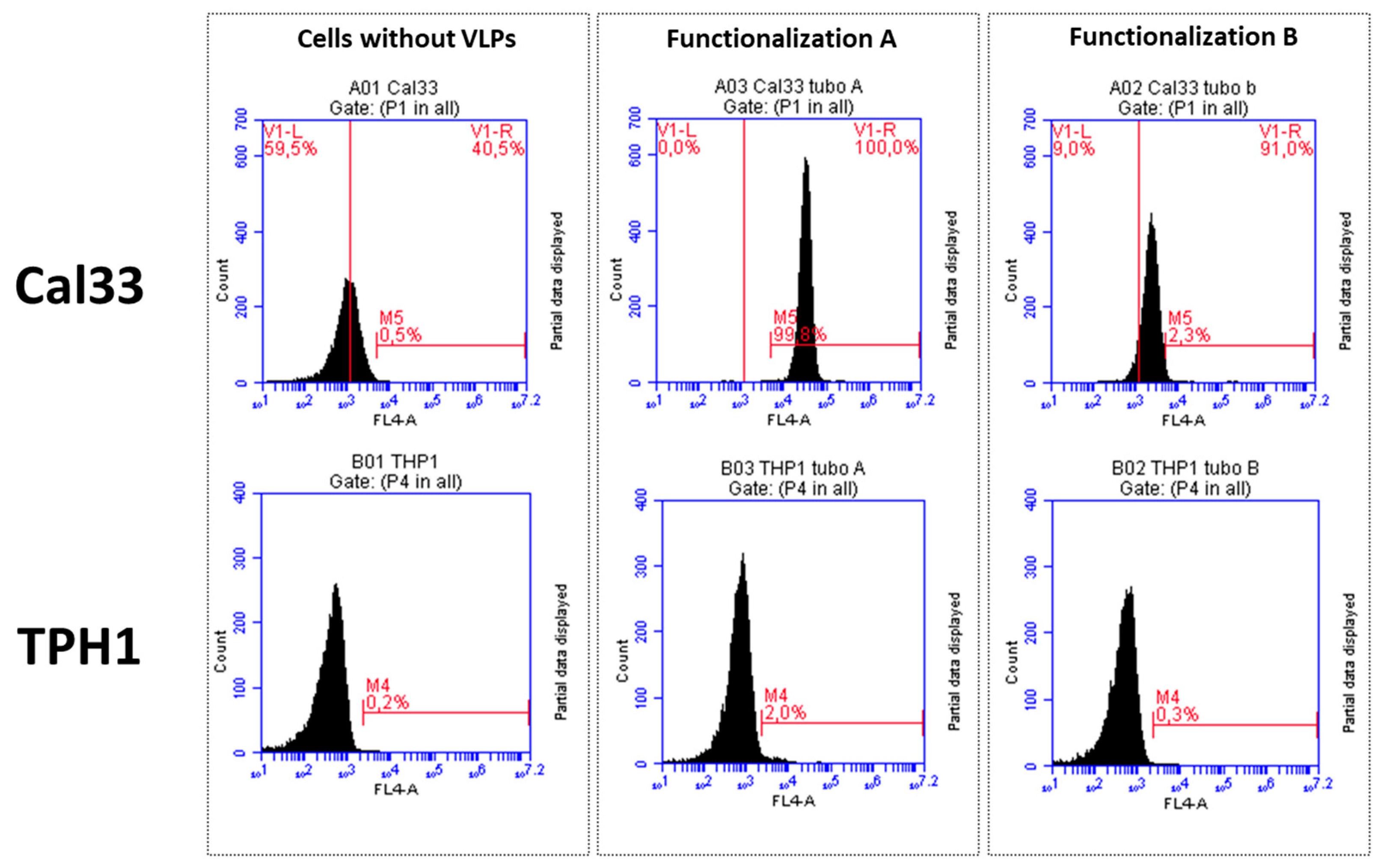
2.4. Flow Cytometry
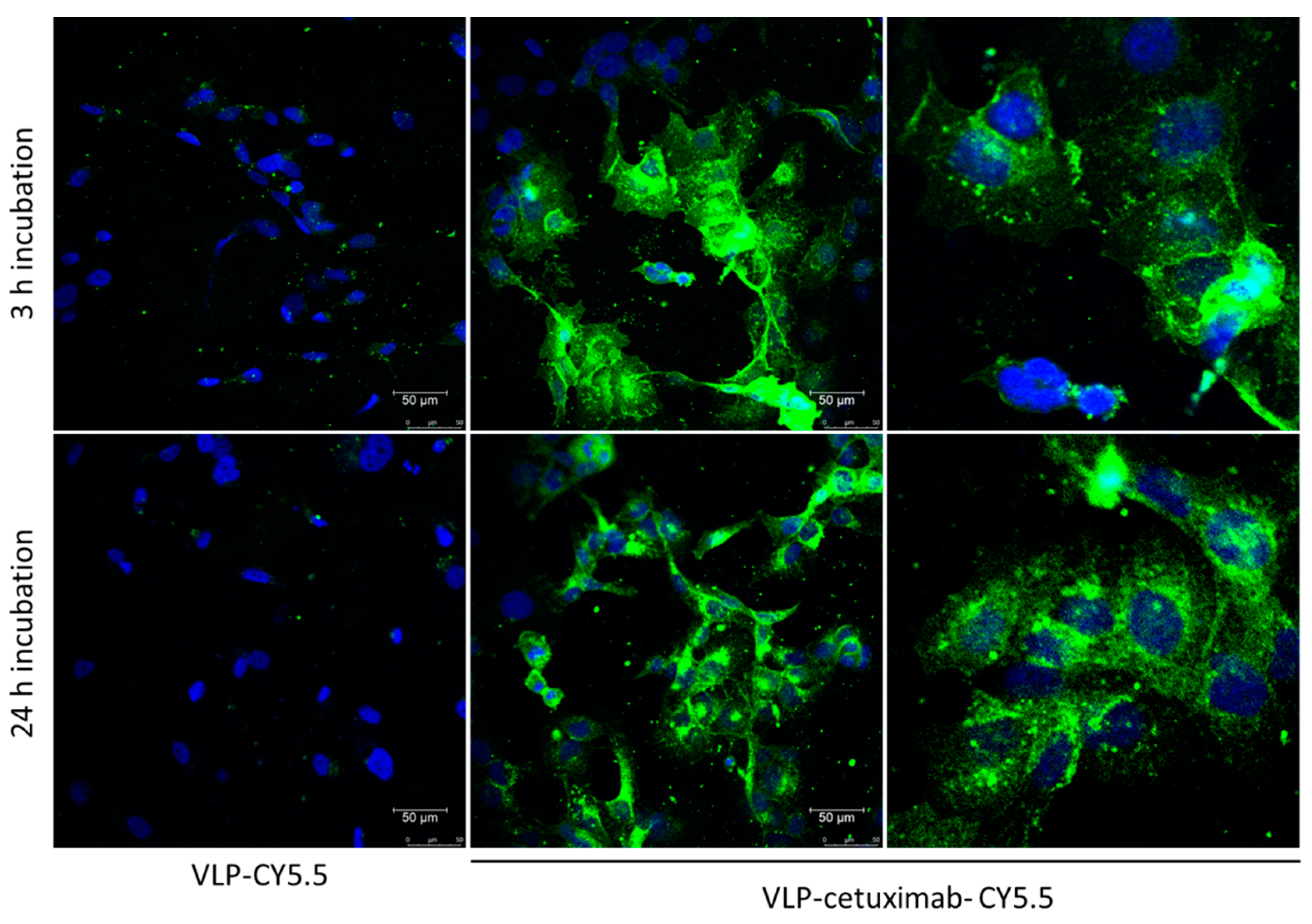
2.5. Confocal Microscopy
3. Discussion
4. Materials and Methods
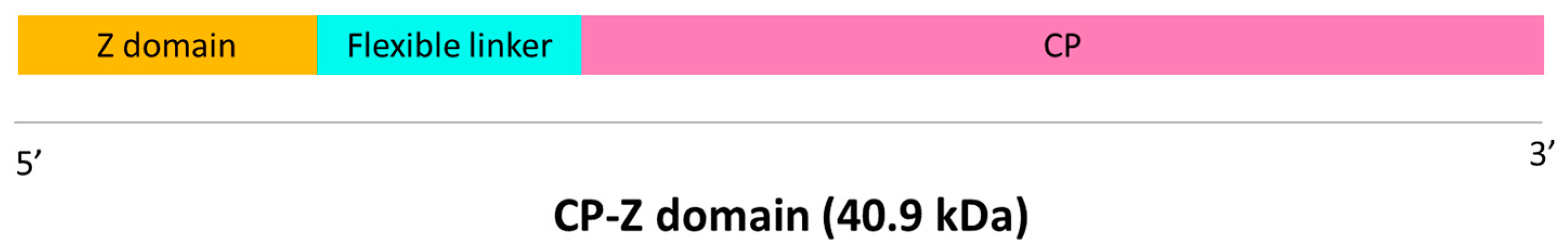
4.1. Cloning in Expression Vectors and Agroinfiltration
4.2. Purification of VLP-Z Domain
4.3. Characterization of Purified VLPs-Z Domain
4.4. VLP-Z Domain Functionalization with Cetuximab
4.5. Triple Functionalization of VLP-Z Domain with Cetuximab and Cy5.5
4.6. Cell Viability Assay
4.7. Flow Cytometry
4.8. Confocal Microscopy
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Jeevanandam, J.; Barhoum, A.; Chan, Y.S.; Dufresne, A.; Danquah, M.K. Review on Nanoparticles and Nanostructured Materials: History, Sources, Toxicity and Regulations. Beilstein J. Nanotechnol. 2018, 9, 1050–1074. [Google Scholar] [CrossRef] [PubMed]
- Chung, Y.H.; Cai, H.; Steinmetz, N.F. Viral Nanoparticles for Drug Delivery, Imaging, Immunotherapy, and Theranostic Applications. Adv. Drug Deliv. Rev. 2020, 156, 214–235. [Google Scholar] [CrossRef] [PubMed]
- Rybicki, E.P. Plant Molecular Farming of Virus-like Nanoparticles as Vaccines and Reagents. Wiley Interdiscip. Rev. Nanomedicine Nanobiotechnology 2020, 12. [Google Scholar] [CrossRef]
- Shukla, S.; Hu, H.; Cai, H.; Chan, S.-K.; Boone, C.E.; Beiss, V.; Chariou, P.L.; Steinmetz, N.F. Annual Review of Virology Plant Viruses and Bacteriophage-Based Reagents for Diagnosis and Therapy. Annu. Rev. Virol. 2020, 7, 559–587. [Google Scholar] [CrossRef] [PubMed]
- Truchado, D.A.; Rincón, S.; Zurita, L.; Ponz, F. Turnip Mosaic Virus Nanoparticles: A Versatile Tool in Biotechnology. In Tools & Techniques of Plant Molecular Farming. Concepts and Strategies in Plant Sciences; Kole, C., Chaurasia, A., Hefferon, K.L.P.J., Eds.; Springer: Singapore, 2023; pp. 235–249. [Google Scholar]
- Velázquez-Lam, E.; Tome-Amat, J.; Segrelles, C.; Yuste-Calvo, C.; Asensio, S.; Peral, J.; Ponz, F.; Lorz, C. Antitumor Applications of Polyphenol-Conjugated Turnip Mosaic Virus-Derived Nanoparticles. Nanomedicine 2022, 999–1012. [Google Scholar] [CrossRef] [PubMed]
- Hanawa, M.; Suzuki, S.; Dobashi, Y.; Yamane, T.; Kono, K.; Enomoto, N.; Ooi, A. EGFR Protein Overexpression and Gene Amplification in Squamous Cell Carcinomas of the Esophagus. Int. J. Cancer 2006, 118, 1173–1180. [Google Scholar] [CrossRef]
- Ryott, M.; Wangsa, D.; Heselmeyer-Haddad, K.; Lindholm, J.; Elmberger, G.; Auer, G.; Lundqvist, E.Å.; Ried, T.; Munck-Wikland, E. EGFR Protein Overexpression and Gene Copy Number Increases in Oral Tongue Squamous Cell Carcinoma. Eur. J. Cancer 2009, 45, 1700–1708. [Google Scholar] [CrossRef] [PubMed]
- Uribe, P.; Gonzalez, S. Epidermal Growth Factor Receptor (EGFR) and Squamous Cell Carcinoma of the Skin: Molecular Bases for EGFR-Targeted Therapy. Pathol. Res. Pract. 2011, 207, 337–342. [Google Scholar] [CrossRef]
- Baselga, J. The EGFR as a Target for Anticancer Therapy-Focus on Cetuximab. Eur. J. Cancer 2001, 37, 16–22. [Google Scholar] [CrossRef]
- Sunada, H.; Magunt, B.E.; Mendelsohn, J.; Macleod, C.L. Monoclonal Antibody against Epidermal Growth Factor Receptor Is Internalized without Stimulating Receptor Phosphorylation (Phosphotyrosine/Percoll Gradient/Epidermoid Carcinoma Cell). Cell Biol. 1986, 83, 3825–3829. [Google Scholar]
- Bou-Assaly, W.; Mukherji, S. Cetuximab (Erbitux). Am. J. Neuroradiol. 2010, 31, 626–627. [Google Scholar] [CrossRef]
- Rigi, G.; Ghaedmohammadi, S.; Ahmadian, G. A Comprehensive Review on Staphylococcal Protein A (SpA): Its Production and Applications. Biotechnol. Appl. Biochem. 2019, 66, 454–464. [Google Scholar] [CrossRef] [PubMed]
- Nilsson, B.; Moks, T.; Jansson, B.; Abrahmsen, L.; Elmblad, A.; Holmgren, E.; Henrichson, C.; Jones, T.A.; Uhlen, M. A Synthetic IgG-Binding Domain Based on Staphylococcal Protein A; 1987; Vol. 1.
- Myrhammar, A.; Rosik, D.; Karlström, A.E. Photocontrolled Reversible Binding between the Protein A-Derived Z Domain and Immunoglobulin G. Bioconjug. Chem. 2020, 31, 622–630. [Google Scholar] [CrossRef]
- Wendlandt, T.; Koch, C.; Britz, B.; Liedek, A.; Schmidt, N.; Werner, S.; Gleba, Y.; Vahidpour, F.; Welden, M.; Poghossian, A.; et al. Facile Purification and Use of Tobamoviral Nanocarriers for Antibody-Mediated Display of a Two-Enzyme System. Viruses 2023, 15, 1951. [Google Scholar] [CrossRef] [PubMed]
- Werner, S.; Marillonnet, S.; Hause, G.; Klimyuk, V.; Gleba, Y. Immunoabsorbent Nanoparticles Based on a Tobamovirus Displaying Protein A. Proc. Natl. Acad. Sci. 2006, 103, 17678–17683. [Google Scholar] [CrossRef]
- Kalnciema, I.; Balke, I.; Skrastina, D.; Ose, V.; Zeltins, A. Potato Virus M-like Nanoparticles: Construction and Characterization. Mol. Biotechnol. 2015, 57, 982–992. [Google Scholar] [CrossRef]
- Truchado, D.A.; Rincón, S.; Zurita, L.; Sánchez, F.; Ponz, F. Isopeptide Bonding in Planta Allows Functionalization of Elongated Flexuous Proteinaceous Viral Nanoparticles, Including Non-Viable Constructs by Other Means. Viruses 2023, 15, 375. [Google Scholar] [CrossRef]
- Czapar, A.E.; Steinmetz, N.F. Plant Viruses and Bacteriophages for Delivery in Medicine and Biotechnology. Curr. Opin. Chem. Biol. 2017, 38, 108–116. [Google Scholar] [CrossRef] [PubMed]
- Eiben, S.; Koch, C.; Altintoprak, K.; Southan, A.; Tovar, G.; Laschat, S.; Weiss, I.M.; Wege, C. Plant Virus-Based Materials for Biomedical Applications: Trends and Prospects. Adv. Drug Deliv. Rev. 2019, 145, 96–118. [Google Scholar] [CrossRef]
- Azizi, M.; Shahgolzari, M.; Fathi-Karkan, S.; Ghasemi, M.; Samadian, H. Multifunctional Plant Virus Nanoparticles: An Emerging Strategy for Therapy of Cancer. Wiley Interdiscip. Rev. Nanomedicine Nanobiotechnology 2023, 15. [Google Scholar] [CrossRef]
- Le, D.H.T.; Lee, K.L.; Shukla, S.; Commandeur, U.; Steinmetz, N.F. Potato Virus X, a Filamentous Plant Viral Nanoparticle for Doxorubicin Delivery in Cancer Therapy. Nanoscale 2017, 9, 2348–2357. [Google Scholar] [CrossRef]
- Zhang, W.; Yang, S.; Shan, T.; Hou, R.; Liu, Z.; Li, W.; Guo, L.; Wang, Y.; Chen, P.; Wang, X.; et al. Virome Comparisons in Wild-Diseased and Healthy Captive Giant Pandas. Microbiome 2017, 5, 90. [Google Scholar] [CrossRef] [PubMed]
- Lee, K.L.; Hubbard, L.C.; Hern, S.; Yildiz, I.; Gratzl, M.; Steinmetz, N.F.; Author, B.S. Shape Matters: The Diffusion Rates of TMV Rods and CPMV Icosahedrons in a Spheroid Model of Extracellular Matrix Are Distinct NIH Public Access Author Manuscript. Biomater Sci 2013, 1. [Google Scholar] [CrossRef]
- Yarden, Y.; Pines, G. The ERBB Network: At Last, Cancer Therapy Meets Systems Biology. Nat. Rev. Cancer 2012, 12, 553–563. [Google Scholar] [CrossRef] [PubMed]
- Hendler, F.J.; Ozanne, B.W. Human Squamous Cell Lung Cancers Express Increased Epidermal Growth Factor Receptors. J. Clin. Invest. 1984, 74, 647–651. [Google Scholar] [CrossRef] [PubMed]
- Sigismund, S.; Avanzato, D.; Lanzetti, L. Emerging Functions of the EGFR in Cancer. Mol. Oncol. 2018, 12, 3–20. [Google Scholar] [CrossRef]
- Steinmetz, N.F. Viral Nanoparticles as Platforms for Next-Generation Therapeutics and Imaging Devices. Nanomedicine Nanotechnology, Biol. Med. 2010, 6, 634–641. [Google Scholar] [CrossRef]
- Wen, A.M.; Lee, K.L.; Yildiz, I.; Bruckman, M.A.; Shukla, S.; Steinmetz, N.F. Viral Nanoparticles for in Vivo Tumor Imaging. J. Vis. Exp. 2012. [Google Scholar] [CrossRef]
- Beatty, P.H.; Lewis, J.D. Cowpea Mosaic Virus Nanoparticles for Cancer Imaging and Therapy. Adv. Drug Deliv. Rev. 2019, 145, 130–144. [Google Scholar] [CrossRef]
- Park, J.; Chariou, P.L.; Steinmetz, N.F. Site-Specific Antibody Conjugation Strategy to Functionalize Virus-Based Nanoparticles. Bioconjug. Chem. 2020, 31, 1408–1416. [Google Scholar] [CrossRef]
- Martí, M.; Merwaiss, F.; Butković, A.; Daròs, J.A. Production of Potyvirus-Derived Nanoparticles Decorated with a Nanobody in Biofactory Plants. Front. Bioeng. Biotechnol. 2022, 10. [Google Scholar] [CrossRef] [PubMed]
- Sainsbury, F.; Thuenemann, E.C.; Lomonossoff, G.P. PEAQ: Versatile Expression Vectors for Easy and Quick Transient Expression of Heterologous Proteins in Plants. Plant Biotechnol. J. 2009, 7, 682–693. [Google Scholar] [CrossRef] [PubMed]
- Sánchez, F.; Ponz, F. Presenting Peptides at the Surface of Potyviruses in Planta. In Methods in Molecular Biology; Humana Press Inc., 2018; Vol. 1776, pp. 471–485.
- Yuste-Calvo, C.; González-Gamboa, I.; Pacios, L.F.; Sánchez, F.; Ponz, F. Structure-Based Multifunctionalization of Flexuous Elongated Viral Nanoparticles. ACS Omega 2019, 4, 5019–5028. [Google Scholar] [CrossRef]

Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).



