Submitted:
02 February 2024
Posted:
02 February 2024
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Cell Culture
2.3. Cell Viability Analysis Using the MTT Assay
2.4. Flow Cytometry Analysis for Apoptosis
2.5. Immunofluorescence Staining
2.6. Caspase-3/7 Activity Assay
2.7. In Vitro Acetylation Assay
2.8. Western Blotting
2.9. Statistical Analysis
3. Results
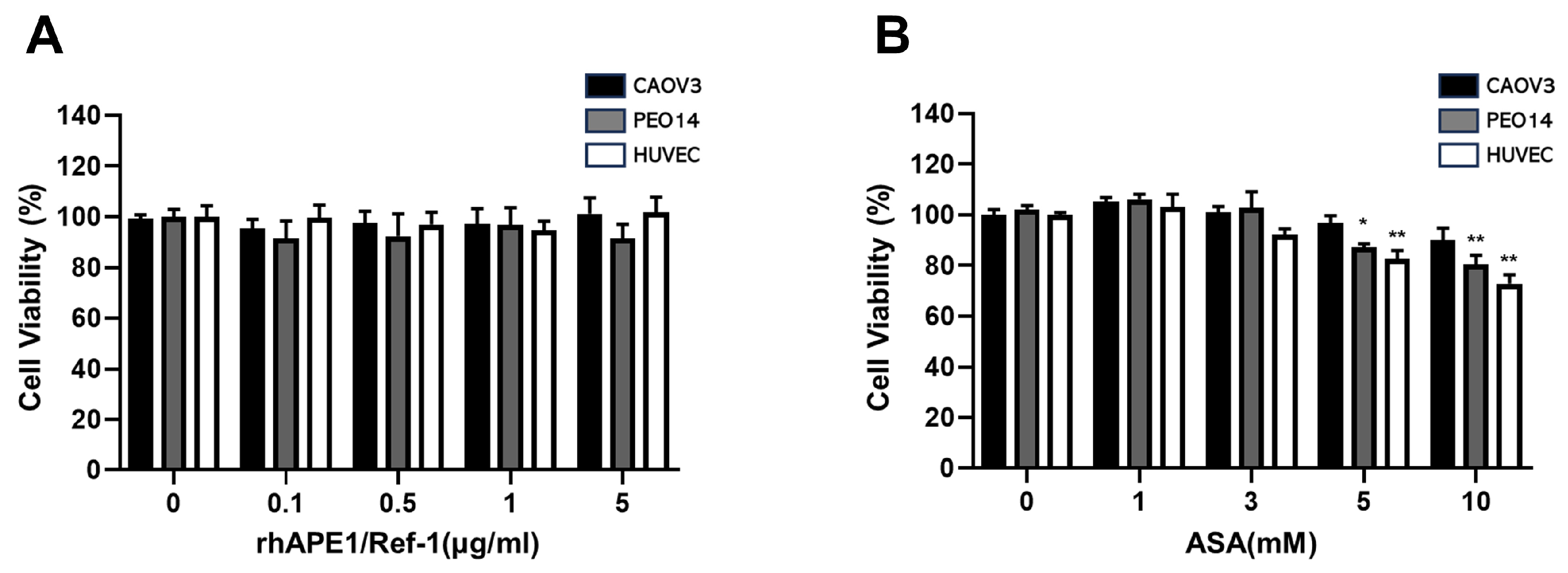
3.1. Effect of rhAPE1/Ref-1 or ASA on Cell Viability in Ovarian Cancer Cells and HUVECs
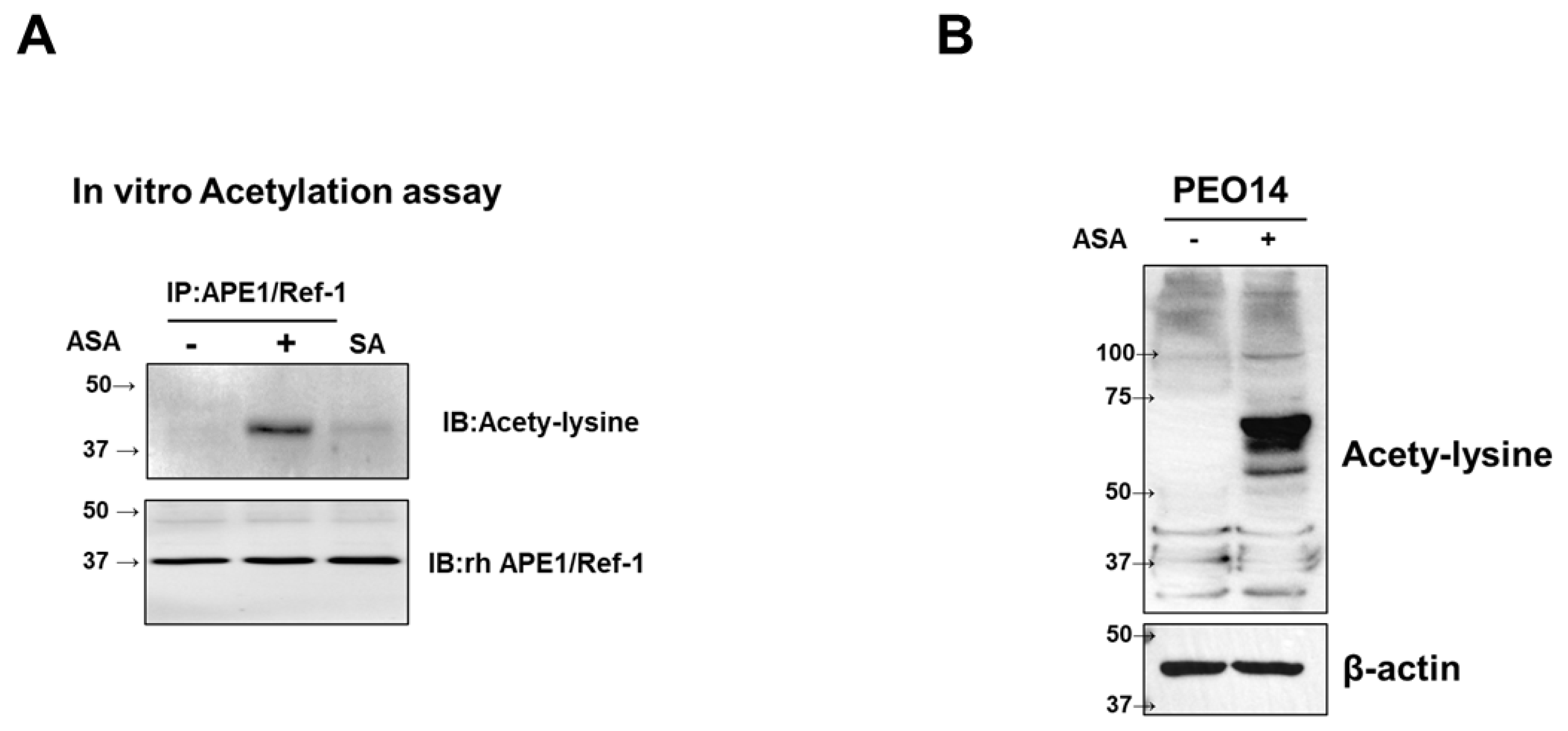
3.2. Aspirin-Induced Acetylation of rhAPE1/Ref-1 and Intracellular Shyperacetylation in PEO14 Cells
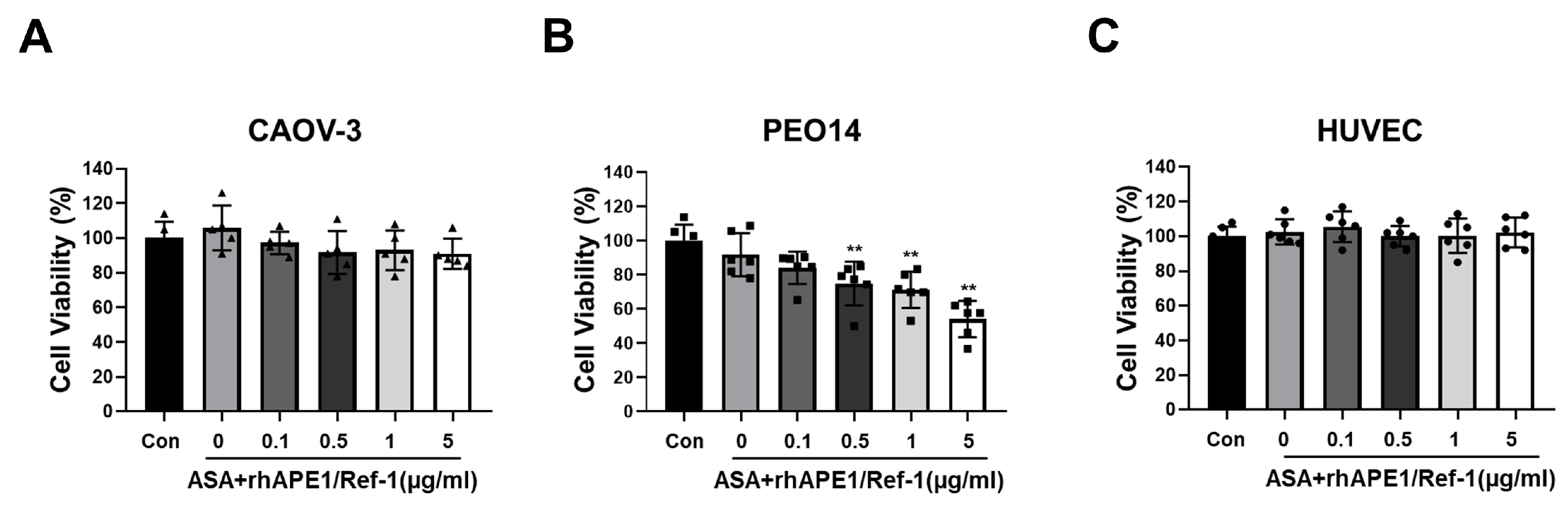
3.3. Co-Treatment of rhAPE1/Ref-1 with ASA Reduces Ovarian Cancer Cell Viability
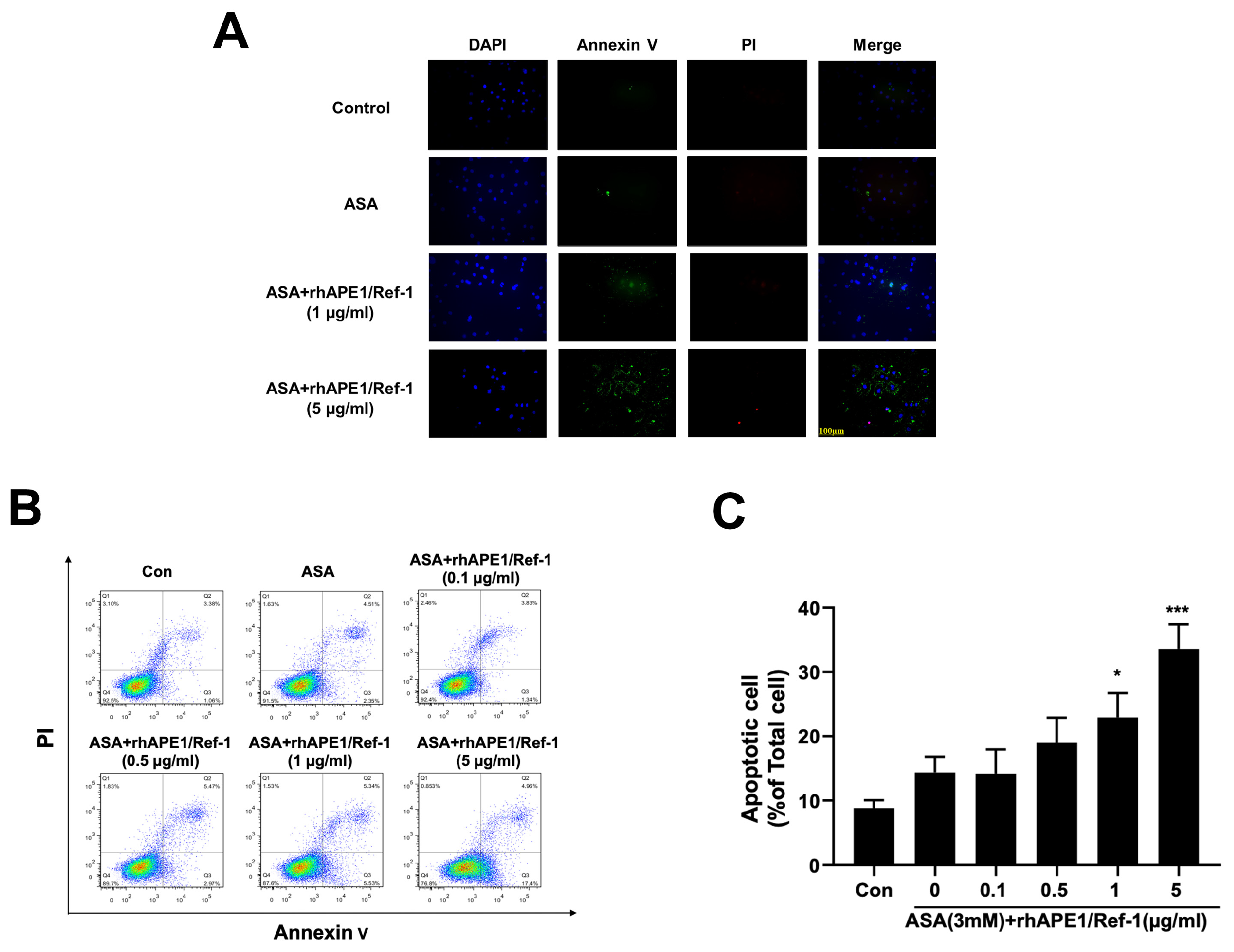
3.4. PEO14 Cell Apoptosis was Induced by Co-treatment with rhAPE1/Ref-1 and ASA
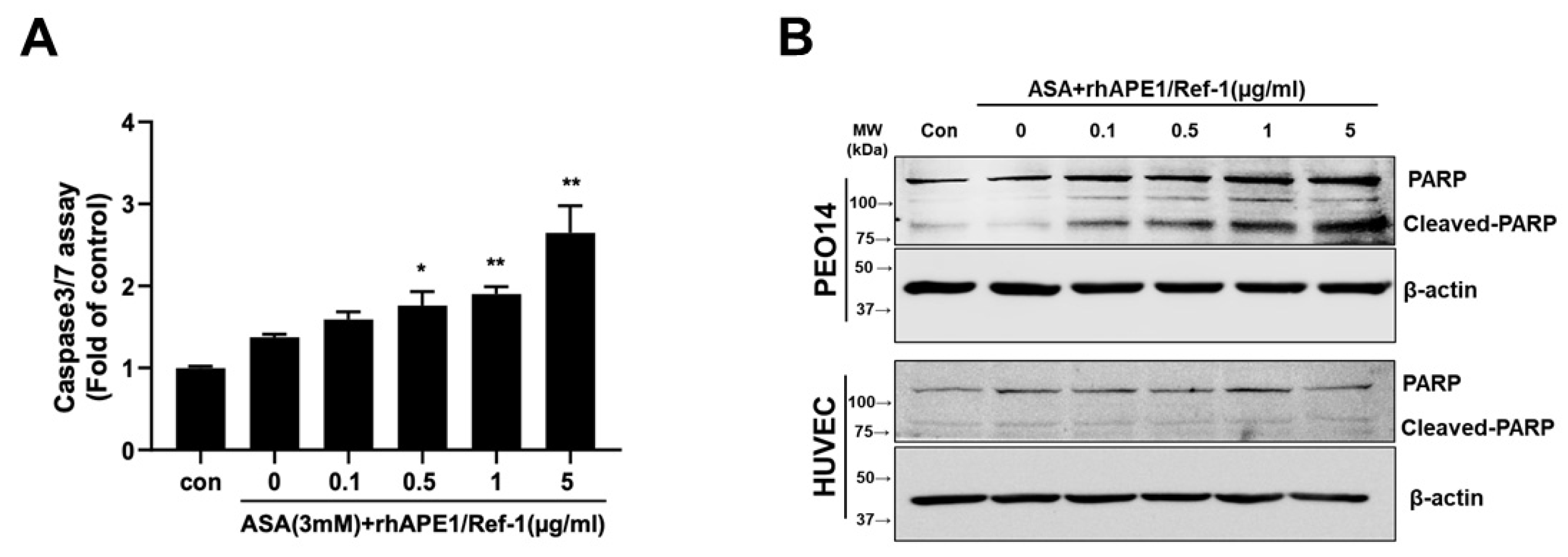
3.5. Co-treatment with rhAPE1/Ref-1 and ASA Induces Caspase Activation and PARP Cleavage in PEO14 Cells
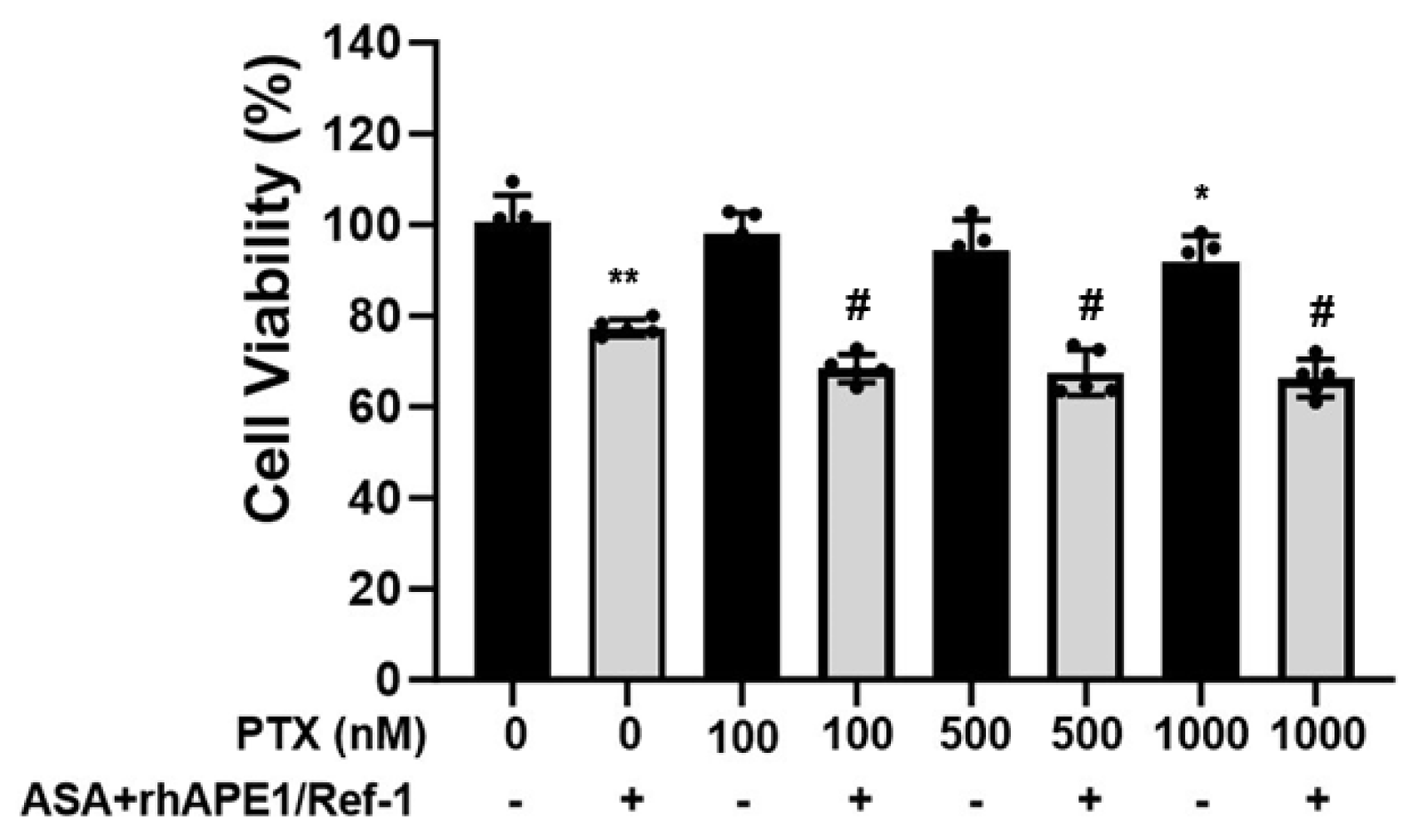
3.6. Enhanced the Anti-cancer Efficacy of PTX in PEO14 Ovarian Cancer Cells Upon Co-treatment with rhAPE1/Ref-1 and ASA
4. Discussion
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Cabasag CJ, Fagan PJ, Ferlay J, Vignat J, Laversanne M, Liu L, et al. Ovarian cancer today and tomorrow: A global assessment by world region and Human Development Index using GLOBOCAN 2020. Int J Cancer. 2022;151(9):1535-41. [CrossRef]
- Shih Ie M, Kurman RJ. Ovarian tumorigenesis: a proposed model based on morphological and molecular genetic analysis. Am J Pathol. 2004;164(5):1511-8. [CrossRef]
- Cannistra SA. Cancer of the ovary. N Engl J Med. 2004;351(24):2519-29.
- Evans AR, Limp-Foster M, Kelley MR. Going APE over ref-1. Mutat Res. 2000;461(2):83-108. [CrossRef]
- Lee YR, Joo HK, Jeon BH. The Biological Role of Apurinic/Apyrimidinic Endonuclease1/Redox Factor-1 as a Therapeutic Target for Vascular Inflammation and as a Serologic Biomarker. Biomedicines. 2020;8(3).
- Choi S, Lee YR, Park MS, Joo HK, Cho EJ, Kim HS, et al. Histone deacetylases inhibitor trichostatin A modulates the extracellular release of APE1/Ref-1. Biochem Biophys Res Commun. 2013;435(3):403-7. [CrossRef]
- Tell G, Quadrifoglio F, Tiribelli C, Kelley MR. The many functions of APE1/Ref-1: not only a DNA repair enzyme. Antioxid Redox Signal. 2009;11(3):601-20. [CrossRef]
- Joo HK, Lee YR, Lee EO, Park MS, Choi S, Kim CS, et al. The extracellular role of Ref-1 as anti-inflammatory function in lipopolysaccharide-induced septic mice. Free Radic Biol Med. 2019;139:16-23. [CrossRef]
- Park MS, Choi S, Lee YR, Joo HK, Kang G, Kim CS, et al. Secreted APE1/Ref-1 inhibits TNF-alpha-stimulated endothelial inflammation via thiol-disulfide exchange in TNF receptor. Sci Rep. 2016;6:23015. [CrossRef]
- Lee YR, Kim KM, Jeon BH, Choi S. Extracellularly secreted APE1/Ref-1 triggers apoptosis in triple-negative breast cancer cells via RAGE binding, which is mediated through acetylation. Oncotarget. 2015;6(27):23383-98. [CrossRef]
- Choi S, Lee YR, Kim KM, Choi E, Jeon BH. Dual Function of Secreted APE1/Ref-1 in TNBC Tumorigenesis: An Apoptotic Initiator and a Regulator of Chronic Inflammatory Signaling. Int J Mol Sci. 2022;23(16). [CrossRef]
- Lee YR, Park MS, Joo HK, Kim KM, Kim J, Jeon BH, Choi S. Therapeutic positioning of secretory acetylated APE1/Ref-1 requirement for suppression of tumor growth in triple-negative breast cancer in vivo. Sci Rep. 2018;8(1):8701. [CrossRef]
- Steering Committee of the Physicians' Health Study Research G. Final report on the aspirin component of the ongoing Physicians' Health Study. N Engl J Med. 1989;321(3):129-35. [CrossRef]
- Pinckard RN, Hawkins D, Farr RS. In vitro acetylation of plasma proteins, enzymes and DNA by aspirin. Nature. 1968;219(5149):68-9. [CrossRef]
- Hurwitz LM, Pinsky PF, Huang WY, Freedman ND, Trabert B. Aspirin use and ovarian cancer risk using extended follow-up of the PLCO Cancer Screening Trial. Gynecol Oncol. 2020;159(2):522-6. [CrossRef]
- Brasky TM, Liu J, White E, Peters U, Potter JD, Walter RB, et al. Non-steroidal anti-inflammatory drugs and cancer risk in women: results from the Women's Health Initiative. Int J Cancer. 2014;135(8):1869-83. [CrossRef]
- Joharatnam-Hogan N, Cafferty FH, Macnair A, Ring A, Langley RE. The role of aspirin in the prevention of ovarian, endometrial and cervical cancers. Womens Health (Lond). 2020;16:1745506520961710. [CrossRef]
- Guo J, Zhu Y, Yu L, Li Y, Guo J, Cai J, et al. Aspirin inhibits tumor progression and enhances cisplatin sensitivity in epithelial ovarian cancer. PeerJ. 2021;9:e11591. [CrossRef]
- Kampan NC, Madondo MT, McNally OM, Quinn M, Plebanski M. Paclitaxel and Its Evolving Role in the Management of Ovarian Cancer. Biomed Res Int. 2015;2015:413076. [CrossRef]
- Li S, Shi B, Liu X, An HX. Acetylation and Deacetylation of DNA Repair Proteins in Cancers. Front Oncol. 2020;10:573502. [CrossRef]
- Lage H, Denkert C. Resistance to chemotherapy in ovarian carcinoma. Recent Results Cancer Res. 2007;176:51-60.
- Spinner DM. MTT growth assays in ovarian cancer. Methods Mol Med. 2001;39:175-7.
- Lakshmanan I, Batra SK. Protocol for Apoptosis Assay by Flow Cytometry Using Annexin V Staining Method. Bio Protoc. 2013;3(6). [CrossRef]
- Taylor RC, Cullen SP, Martin SJ. Apoptosis: controlled demolition at the cellular level. Nat Rev Mol Cell Biol. 2008;9(3):231-41. [CrossRef]
- Langdon SP, Hawkes MM, Lawrie SS, Hawkins RA, Tesdale AL, Crew AJ, et al. Oestrogen receptor expression and the effects of oestrogen and tamoxifen on the growth of human ovarian carcinoma cell lines. Br J Cancer. 1990;62(2):213-6. [CrossRef]
- Marnett LJ, Kalgutkar AS. Cyclooxygenase 2 inhibitors: discovery, selectivity and the future. Trends Pharmacol Sci. 1999;20(11):465-9. [CrossRef]
- Jara-Gutierrez A, Baladron V. The Role of Prostaglandins in Different Types of Cancer. Cells. 2021;10(6). [CrossRef]
- Dibra HK, Brown JE, Hooley P, Nicholl ID. Aspirin and alterations in DNA repair proteins in the SW480 colorectal cancer cell line. Oncol Rep. 2010;24(1):37-46. [CrossRef]
- Bossard C, Busson M, Vindrieux D, Gaudin F, Machelon V, Brigitte M, et al. Potential role of estrogen receptor beta as a tumor suppressor of epithelial ovarian cancer. PLoS One. 2012;7(9):e44787. [CrossRef]
- Zheng J, Zhou J, Xie X, Xie B, Lin J, Xu Z, Zhang W. Estrogen decreases anoikis of ovarian cancer cell line Caov-3 through reducing release of Bit1. DNA Cell Biol. 2014;33(12):847-53. [CrossRef]
- Gao B, Russell A, Beesley J, Chen XQ, Healey S, Henderson M, et al. Paclitaxel sensitivity in relation to ABCB1 expression, efflux and single nucleotide polymorphisms in ovarian cancer. Sci Rep. 2014;4:4669. [CrossRef]
- Julien O, Wells JA. Caspases and their substrates. Cell Death Differ. 2017;24(8):1380-9. [CrossRef]
- Kellokumpu-Lehtinen P, Tuunanen T, Asola R, Elomaa L, Heikkinen M, Kokko R, et al. Weekly paclitaxel--an effective treatment for advanced breast cancer. Anticancer Res. 2013;33(6):2623-7.
- van Vuuren RJ, Visagie MH, Theron AE, Joubert AM. Antimitotic drugs in the treatment of cancer. Cancer Chemother Pharmacol. 2015;76(6):1101-12. [CrossRef]
- Lehoczky O, Bagameri A, Udvary J, Pulay T. Side-effects of paclitaxel therapy in ovarian cancer patents. Eur J Gynaecol Oncol. 2001;22(1):81-4.
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).



