1. Introduction
Protein materials, that is, supramolecular protein complexes with defined physicochemical and biological properties, are gaining interest in biomedicine because of their potent applications in drug delivery and in regenerative medicine [
1,
2,
3,
4,
5]. Several approaches allow the controlled oligomerization of selected polypeptides into fibrils, layers, matrices, nanoparticles or microparticles [
6,
7,
8,
9]. One of the most versatile, promising and technologically simple strategy for protein assembly is the exploitation of the coordination capabilities between divalent cations (such as Zn
2+, Ca
2+, Mg
2+ and Mn2+) and histidine (His) residues [
10]. This category of interactivity allows the cross-molecular binding of poly-His-tagged proteins into nanoparticles and progressively [
11], as granular microscale particles [
12], in a process that can be reversed by the exposure to chelating agents or, more slowly, by the mere physiological, equilibrium-based dilution of the gluing cation (
Figure 1A). This fact results in the progressive disintegration of the material into their forming building blocks, either nanoparticles or their protomers, that are released to the media [
13,
14]. In addition, it is known that the formation of the nanoparticle building blocks is favoured if the N-terminal segment of the polypeptide is cationic [
15,
16]. Taking this approach and by regulating the cation/His ratio in the coordination mixture, both nanoparticles and microparticles have been generated in a controlled way through robust and highly efficient protocols that keep the folding status and functionality of the forming polypeptides [
17]. While the intermediate nanoparticles have been mostly adapted as vehicles for the cell-targeted delivery of small molecular weight drugs and cytotoxic protein in oncology [
18,
19], microparticles show appealing properties as slow drug delivery systems [
20,
21,
22].
Once these micron-scale materials are administered subcutaneously, they leak the forming polypeptides that reach the bloodstream [
22]. Notably, if fused to specific ligands of cell surface receptors, the released protein accumulates in receptor-overexpressing target organs [
22,
23]. This principle, which clearly mimics the secretion process of peptide hormones in the mammalian endocrine system [
24,
25], has been explored for the delivery of cytotoxic proteins [
22] and nanoconjugates [
26] in oncology, growth factors in regenerative medicine [
27] and nanobodies in neurodegenerative diseases [
21]. A main advantage of this system over other slow drug delivery platforms is that the protein drug is self-contained as mechanically stable but dynamic depots, in the absence of any chemically heterogeneous scaffold. Thus, inert holding materials or matrices, that might pose compatibility issues, are not needed here to support the endocrine-like character of these protein materials.
The fact that these secretory microparticles undergo a disintegration process in vivo results in a time-prolonged release of functional, properly folded proteins, in contrast to a conventional single shot. The leakage profile is dependent on protein properties [
14] and on the used gluing cation [
23]. This concept might be highly appealing in vaccinology as a way to expose a given antigen to the immune system during a prolonged period upon a single administration, mimicking the immune stimulation during a natural infection. Therefore, secretory microparticles might represent a novel approach to clinical immunization based on the subcutaneous implantation of antigen-delivering protein materials. This possibility has been explored here through the preparation and immunogenicity evaluation of protein-only secretory materials, utilizing two distinct antigens and in two different experimental models. Firstly, the in vivo safety and immunogenicity of protein-only microparticles based on p30 [
28], a main antigen of the African swine fever virus (ASFV) [
29], were examined in pigs. This structurally complex virus [
30] causes severe haemorrhagic disease in domestic pigs [
31]. Being a significant global veterinary concern [
32] primarily due to the lack of effective vaccine prototypes [
33], it is particularly worrying in large countries such as China in which the pig industry is a strong economic supporter [
34,
35]. Secondly, a mouse model was employed to assess the safety and immunogenicity of secretory granules based on GFP. Additionally, to obtain deeper understanding of the immunological mechanisms triggered by these materials, the specific cytokine profile induced in vitro in primary cells was examined. The administration of these secretory microparticles and the further detailed characterization of the triggered immune responses allow envisaging such a depot-based immunization approach as suited for further exploration and development of this concept. In this regard, the obtained data have been evaluated in the context of simple, cost-effective, and efficient new generation protein-based vaccination platforms that might offer appealing properties over the current immunization methods.
2. Materials and Methods
2.1. Ethics Statement
Animal care and procedures were performed in accordance with the guidelines of the Good Experimental Practice and with the approval of the Ethics Committee on Animal Experimentation of the Generalitat de Catalunya (pig experiments project codes: CEA-OH/11580/1 and CEA-OH/10298/2; mice experiment project code: CEA-OH/11691/2).
2.2. Protein Design, Production and Purification
In-house designed synthetic gene for the modular protein RK4-P30-H6 was provided by Gene Art (Thermo Fisher), subcloned in a pET22b plasmid (Novagen). The construct contains four repetitions of RK at the N-terminal that serve as a cationic peptide to stimulate nanoparticle formation together with the poly-His tag at the C-terminal end [
16]. The production of the protein was tested at 20 ºC (overnight) and 37 ºC (3 hours) with two different concentrations of IPTG (0.1 mM, 1 mM) in an
Escherichia coli BL21 strain. Protein production was optimized at 20ºC overnight with 0.1 mM IPTG. After the production, cells were harvested at 5000 g for 15 minutes and the pellets were washed with PBS and stored at –80 ºC until further use. Prior to the purification, cells were resuspended in Wash Buffer (20 mM Tris-HCl pH 8, 500 mM NaCl and 10 mM imidazole) with a cOmplete Protease Inhibitor Cocktail EDTA-free tablet (Roche Diagnostics). Cells were then disrupted in an EmulsiFlex-C5 system (Avestin) for 3 rounds at approximately 7500 psi. After the disruption, the mixture was centrifuged at 15000 g for 45 minutes and the soluble fraction was retained and filtered through sterile 0.22 µm filters (Millipore). Next, the filtered soluble fraction was loaded into a HisTrap HP 5 mL column (GE Healthcare) using an ÄKTA Pure chromatography system (GE Healthcare). Elution was performed through a linear gradient of Elution Buffer (20 mM Tris-HCl pH 8, 500 mM NaCl, 500 mM imidazole). The fractions were analyzed by SDS-PAGE and Western blot (anti-His tag, Genscript) and those containing RK-P30-H6 were dialyzed thrice against a saline buffer (166 mM NaCO3H, 333 mM NaCl).
2.3. Dynamic Light Scattering
The volume size distribution of nanoparticles was determined by Dynamic Light Scattering (DLS) at 633 nm (Zetasizer Pro, Malvern). Samples were diluted in their respective buffer to a concentration of 1 mg/mL. The samples were measured in triplicate.
2.4. Microparticle Formation and Protein Release
RK4-p30-H6 and GFP-H6 (at 1 mg/mL) were mixed, individually, with a zinc chloride solution at a 200:1 divalent cation:protein ratio. The mixture was incubated at room temperature for 10 minutes and then the microparticles were recovered by centrifugation (10000g, 5 minutes). The supernatant was discarded. To assess the release of protein from these microparticles, these materials were resuspended in PBS (diluted to a final concentration of 1 mg/mL) and further incubated at 37ºC for seven days. Samples were extracted on day 0, 1, 3 and 7. Then, we loaded the samples into a polyacrylamide gel which was later transferred to a PVDF membrane. The percentage of protein released during the experiment was assessed by Western Blot using an anti-polyhistidine tag antibody. Images were processed with the Image Lab (BioRad) software.
2.5. Scanning Electron Microscopy
High-resolution images of cation-induced microparticles were obtained by field emission scanning electron microscopy (FESEM). 10 μL of each microparticle sample (0.5 mg/mL) were deposited on silicon wafers (Ted Pella Inc.) overnight and then observed without coating in a FESEM Zeiss Merlin (Zeiss) operating at 1 kV and equipped with a high-resolution secondary electron detector.
2.6. Study Design of the Pig Experiments
Landrace x Large White male piglets at 8 weeks of age at arrival were used. Pigs were housed in the experimental farm of IRTA Monells (Girona, Spain). Animals were fed ad libitum, and an acclimation period of one week was allowed before the initiation of the study. In the first experiment, animals were subcutaneously inoculated once or twice three weeks apart with 50 µg (final volume of 0.5 ml) of the soluble or protein-only-microparticles (POM) form of the ASFV protein p30 in sodium carbonate buffer (166 mM NaHCO3, 333 mM NaCl buffer, pH 8), in the presence or absence of CAF01 (half of the volume, following manufacturer’s instructions). A group of three pigs received PBS following the same procedure as control. Blood samples were collected weekly. In the second experiment, pigs received either a high dose of 150 µg of p30 POM or PBS (control group), following the same immunization regimen than experiment 1.
2.7. Study Design of the Mice Experiment
Sixteen BALB/c mice at 7 weeks of age at arrival were used (half females and half males). Mice were allocated in cages according to sex and fed ad libitum during the experiment. Ear cuts were used to differentiate the animals. After one week of acclimation, six mice (3 females and 3 males) were subcutaneously inoculated twice 3 weeks apart with 50 µg of GFP POM, while six other (3 females and 3 males) received 5 µg of the same antigen following the same regimen. GFP POM was suspended in sodium carbonate buffer (166 mM NaHCO3, 333 mM NaCl buffer, pH 8), and the inoculated volume was 0.3 ml. Four mice (2 females and 2 males) were used as control and received 0.3 ml of PBS in each administration. Blood for the generation of serum was taken from the facial vein before the first inoculation (SD0), and 2 and 9 weeks after the second inoculation (SD35 and SD85, respectively). Two weeks after the second immunization, three mice receiving 50 µg/dose of GFP POM, three receiving 5 µg/dose and two controls were euthanized to assess the cellular response induced by GFP POM using splenocytes. The six remaining animals were kept for seven more weeks to analyse long-term immunogenicity.
2.8. ELISA
ASFV-specific antibodies were assessed by indirect ELISA based on soluble extracts of ASFV-infected cells approved by the WOAH [
36]. For porcine IgG and IgA, peroxidase-conjugated rabbit anti-pig IgG (Sigma-Aldrich, A5670) at 1/20,000 or peroxidase-conjugated goat anti pig-IgA (Invitrogen, PA1-84625) at 1/2,500 were used. For specific isotypes, mouse anti-pig IgG1 (BioRad, MCA635GA) at 1/1,000, mouse anti-pig IgG2 (BioRad, MCA636GA) at 1/1,000, followed by peroxidase-conjugated goat anti-mouse polyvalent immunoglobulins (Sigma-Aldrich, A4012) at 1/2,500 were used. In the case of GFP-specific antibodies, COSTAR 3590 Corning High binding 96-well plates (Cultek, 153590) were coated with the soluble form of the protein at 5 µg/ml in carbonate-bicarbonate buffer overnight at 4 ºC. Plates were washed 3x with PBS-Tween20 0.05% (PBS-T), and blocked for 1 hour at 37 ºC, 5 % CO2 with PBS-T 2 % BSA. Afterwards, mouse serum samples in PBS-T 2 % BSA were added and incubated for 1 hour at 37 ºC, 5 % CO2. After incubation with the tested mice serum, the corresponding antibody in PBS-T 2 % BSA was used: HRP-conjugated goat anti-mouse polyvalent immunoglobulins (Sigma-Aldrich, A4012) at 1/1,000, HRP anti-mouse IgG1 (Abcam, ab97240) at 1/1,000, HRP anti-mouse IgG2a heavy chain (Abcam, ab97245) at 1/1,000, or HRP anti-mouse IgA alpha chain (Abcam, ab97235) at 1/1,000. In both cases, soluble 3,3′,5,5′-tetramethylbenzidine (TMB, Sigma-Aldrich, T4444) was used as specific chromogenic substrate for the HRP, and the reaction was stopped with 1 N H2SO4. Plates were finally read at a wavelength of 450 nm, and average absorbance [optical density (OD) values] of duplicates are represented in the graphs.
2.9. Mouse Splenocytes Collection and Flow Cytometry
Mice splenocytes were obtained by mechanically dissociation of spleens and filtration through a 40 µm cell strainer. Red blood cells were then lysed using NH4Cl for 5 min at room temperature, and splenocytes were finally suspended in RPMI 1640 medium (Gibco) supplemented with 10% FBS (Cultek), 2 mM L-glutamine (Invitrogen), 100 IU/ml penicillin/streptomycin (Invitrogen), 0.05 mM β-mercaptoethanol and 1 mM sodium pyruvate. Fresh mouse splenocytes were used for flow cytometry analysis. One million cells were used per condition in U-bottom 96-well plates. Splenocytes were stimulated for 5 days with 5 µg/ml of GFP POM. Complete RPMI was used as negative control, while stimulation with phorbol myristate acetate (PMA) plus ionomycin (at 5 ng/ml and 500 ng/ml, respectively) was used as positive control. After stimulation, cells were stained with Zombie NIR fixable viability kit (Biolegend, 423106) following manufacturer’s instructions, and then a blockage of Fc receptors was performed with PBS 5 % FBS for 15 min on ice. Extracellular staining was performed for 20 min on ice in PBS 2 % FBS using 50 µl of a mix of the following antibodies: APC hamster anti-mouse CD3e at 1/20 dilution (BD Biosciences, #553066), PerCP-Cy5.5 rat anti-mouse CD4 at 1/300 dilution (BD Biosciences, #550954) and PE-Cy7 rat anti-mouse CD8a at 1/150 dilution (BD Biosciences, #552877). Afterwards, the BD Cytofix/Cytoperm Kit (BD Biosciences) was used according to the manufacturer’s protocol to fix and permeabilize the cells. Intracellular staining using BV421 mouse anti-Ki67 at 1/150 dilution (BD Biosciences #652411) was then performed for 30 min on ice in Perm/Wash buffer (BD Biosciences). Samples were acquired on a BD FACSAria IIu flow cytometer (BD Biosciences) and data was analyzed using FlowJo v10.7.1 software (Tree Star Inc).
2.10. Multiplex Luminex Assay
PAMs were seeded in 24-well plates (106 cells/well) in RPMI 1640 medium supplemented with 10% FBS, 100 IU/ml of penicillin/streptomycin, 2 mM L-glutamine and 0.5% nystatin (sigma-MERCK) and incubated overnight at 37 ºC and 5 % CO2. Afterwards, the medium was removed and replenished with fresh RPMI either with or without 5 µg/ml of GFP POM. After a 24-hour incubation at 37 ºC, 5% CO2, cell supernatants were collected and cytokine were determined using the Luminex xMAP technology and the ProcartaPlex Porcine Cytokine & Chemokine Panel 1 (ThermoFisher Scientific). Cytokine concentrations were calculated using The xPONENT software (Luminex) and expressed as pg/ml (except for TNF, for which a technical problem with the standard curve occurred and only the mean fluorescence intensity (MFI) values could be assessed).
2.11. Statistical Analyses
Prism version 8.3.0. software (GraphPad) was used to plot the results and perform statistical analyses. The tests used are specified on figure legends. Statistical significance was set at p < 0.05 and is displayed in GraphPad style (p > 0.05 ns, * p ≤ 0.05, ** p ≤ 0.01, *** p ≤ 0.001).
3. Results
3.1. Design and Construction of an ASFV-Antigen Nanoparticle
A full-length, proteolytically stable version of the antigen p30 was engineered and produced in
Escherichia coli as a multidomain protein, placed between a cationic N-terminal peptide (RK4) and a C-terminal hexahistidine (H6) tail (RK4-P30-H6,
Figure 1B). The combination of these end terminal peptides was expected to promote the self-assembling of the construct as oligomeric nanoparticles [
16] assisted by the low amount of divalent cations from the media [
15]. A simpler GFP-H6 construct [
37] was used as a control for some experiments (
Figure 1B). Upon bioproduction, RK4-P30-H6 was recovered, in a single step, by immobilized metal affinity chromatography (IMAC). The size and integrity of the construct were confirmed through Western Blot (
Figure 1C, left). Notably, we observed that, as predicted, the protein spontaneously organized as monodisperse nanoparticle populations of around 52 nanometers in size, which could be disassembled by submitting the materials to denaturing plus chelating conditions (
Figure 1C, right). In contrast, the control GFP-H6, also well produced (
Figure 1D, left), remained unassembled because of the non-cationic character of its N-terminal end (
Figure 1D, right).
3.2. RK4-p30-H6 Microparticles Promote Consistent and Prolonged Antigen Release
Using pure solutions of RK4-p30-H6 and GFP-H6, we generated secretory microscale granules by mixing the stored pure protein with a zinc chloride solution at a 200:1 divalent cation:protein ratio. The formation of these granules occurs through the Zn-mediated cross-molecular clustering [
10] via the overhanging H6 tails [
12], and it results in mechanically stable particles that leak the forming protein, in vitro and in vivo, during prolonged time periods [
13,
20,
21,
22,
27]. The size of the resulting materials, named p30- or GFP-POM (from Protein-Only Microparticles), was determined by both SEM and DLS (
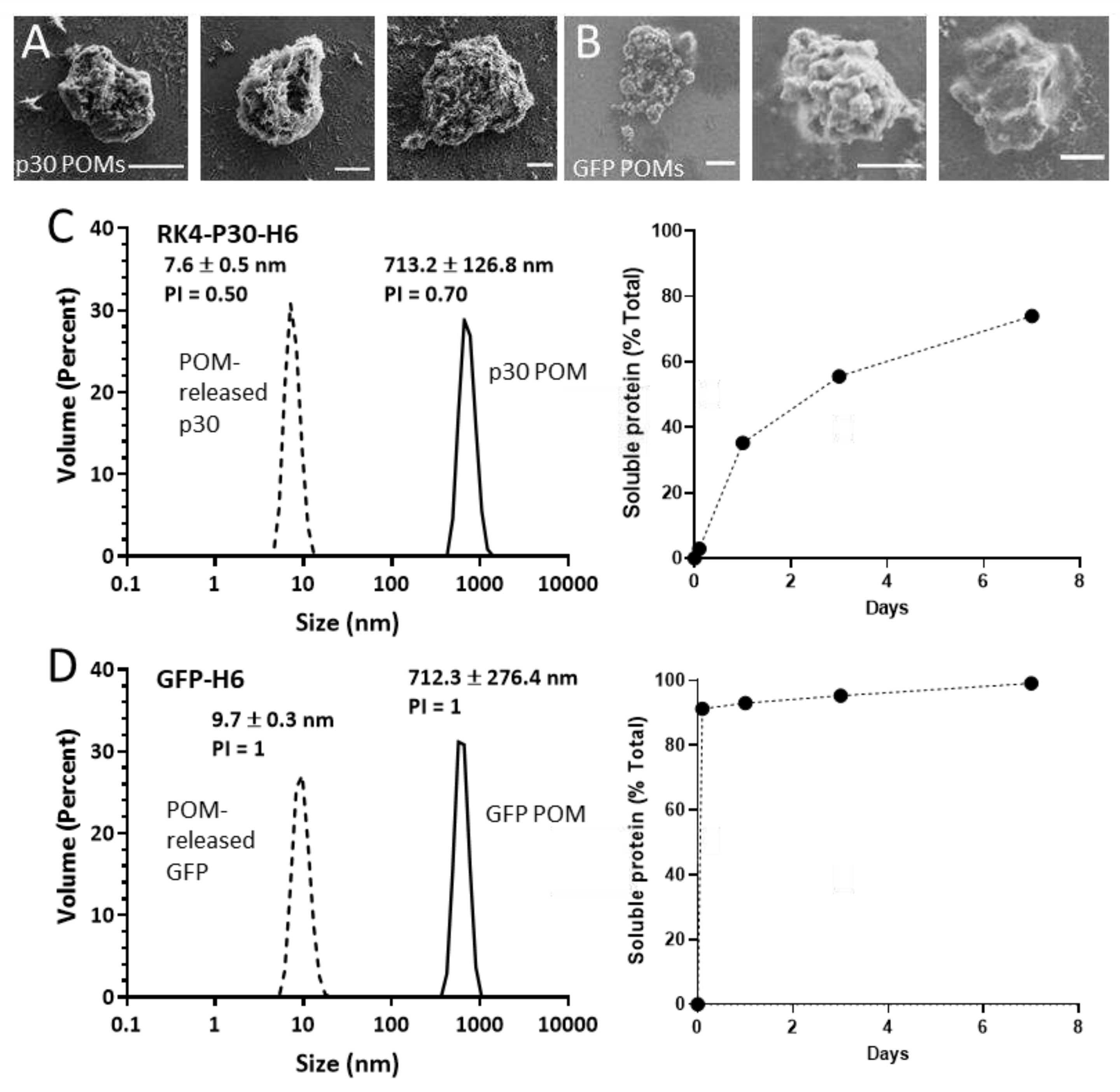
Figure 2A,B), resulting in values ranging from 0.5 µm to 4 µm. We also evaluated the protein released from these POMs linked to their slow disintegration, in vitro, under physiological conditions, for seven days. Under this experimental setting, a highly regular and sustained protein leakage was observed during the entire experiment, in the case of p30-POM (
Figure 2C) but faster for a large fraction of the GFP-POM content (
Figure 2D). A small fraction of GFP-H6 was, however, released progressively for a few days. The differential leakage pattern of GFP-H6 was probably related to the missing N-terminal cationic peptide, which in this type of modular protein constructions shown architectonic roles through electrostatic interactions between protein monomers [
16,
38]. The size of the released RK4-p30-H6 and GFP-H6 proteins was compatible with their dimeric forms (
Figure 2, C-D), that appeared as highly stable. This fact indicated that the building block nanoparticles (sizing around 50 nm) out of which the p30 POMs were formed (
Figure 1A) were unstable upon their transit through POMs, disassembling in lower order structures in parallel or immediately after leakage in vitro. In other parallel platforms based on different proteins of oncological interest, the leaked nanoparticles have been shown more stable under in vitro testing [
22]. However, we assumed that the material produced here might be more robust in vivo in a more crowded ionic environment, and that the nanoparticle version of the antigen might be at least temporarily available for immune stimulation.
3.3. Subcutaneous Administration of p30 POM Is Safe and Immunogenic in Pigs
In previous studies, the subcutaneous inoculation of protein-only secretory microparticles formed by therapeutic proteins did not show any side toxicities while the released protein was fully functional [
22,
23,
39,
40]. Here we aimed to assess the safety and immunogenicity of protein-only secretory microparticles for their potential use as slow-release systems for antigens. To do so, the soluble and POM forms of the ASFV p30 were subcutaneously administered to pigs, and the induced antibody response was analysed. In each group, pigs received one dose of 50 µg at SD0, and a booster was administered at SD14 only in half of them. For comparative purposes, two additional groups of pigs received the same protein versions but formulated with the commercial adjuvant CAF01. No adverse effects were noticed in the animals after administration of any of the formulations. Neither control pigs injected with PBS nor pigs receiving one single dose of any of the tested protein versions and formulations showed detectable ASFV-specific IgGs at any of the tested time points (
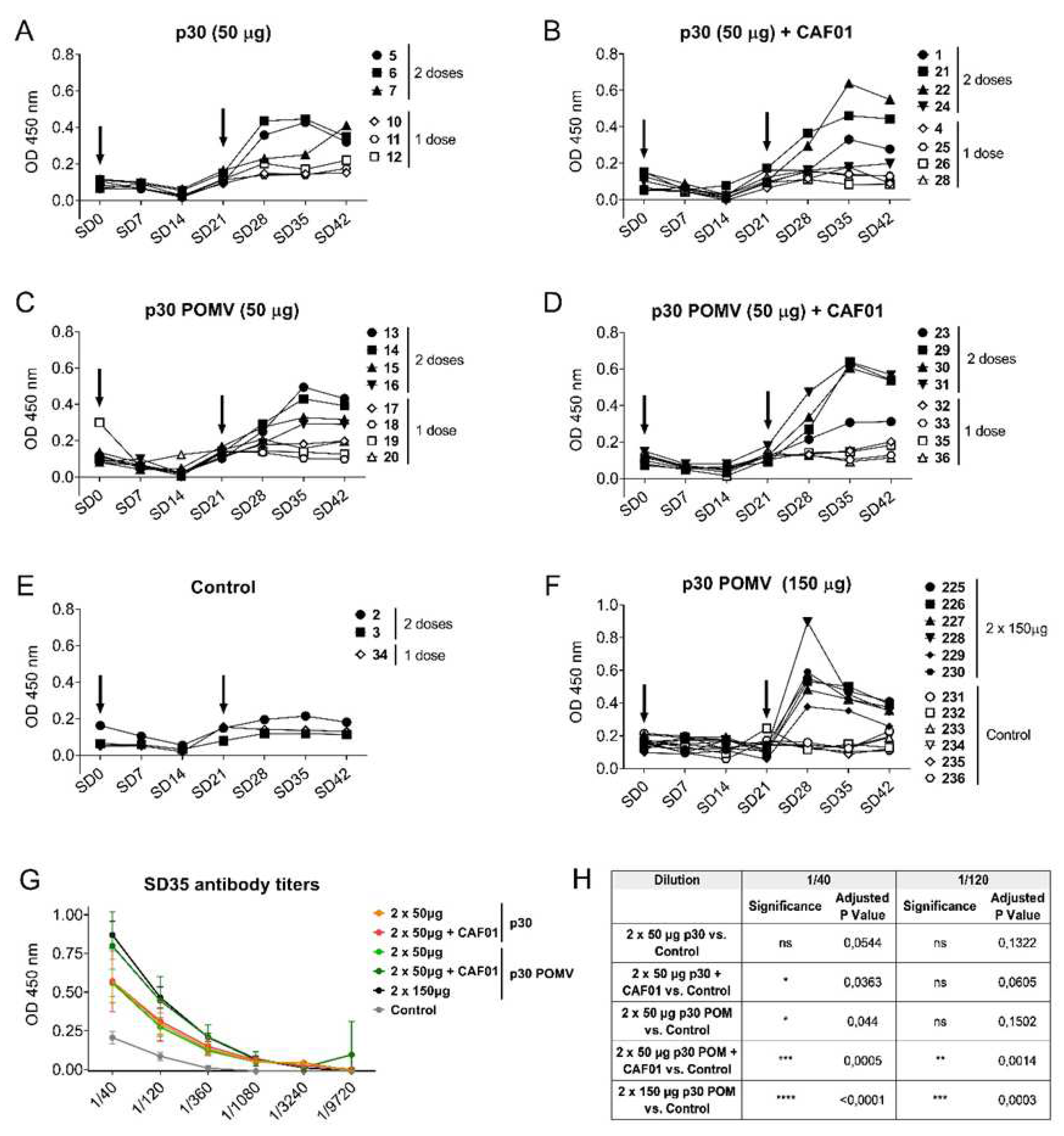
Figure 3, A-E). In contrast, administration of two doses of p30 resulted in seroconversion in all cases, detectable from day 7 after the second inoculation, peaking after 14 days, and slowly declining by day 21 (
Figure 3, A-D).
In previous characterization studies of secretory granules, 0.5-1 mg/doses were routinely administered to mouse models with a good accumulation of the tumor-targeted released protein in target tissues [
22,
23]. Considering the small amount of protein used here (50 µg/dose), we wondered whether increasing the injected POM amount could optimize the antigen formulation, and even trigger a potent immune response that might prevent the booster or the use of adjuvant. To test this hypothesis, six pigs were inoculated twice with 150 µg of p30 POM, while another group of six control pigs received PBS. Again, no seroconversion was observed after one single shot, but detectable ASFV-specific IgGs were observed after a second administration (
Figure 3F). Remarkably, two weeks after the second administration (SD35), the ASFV-specific IgG titres showed that immunization with soluble p30 alone did not render significant levels compared to the control group, while 50 µg of soluble p30+CAF01 showed a slight but significant increase of specific antibodies compared to the control group at dilution 1/40 (
Figure 3, G-H). Notably, the use of p30 POMV without CAF01 also resulted in a significant increase of the antibody levels at this dilution (
Figure 3, G-H), thus suggesting that the particulate nature of the antigen was capable to enhance vaccine immunogenicity without the need of an adjuvant. Indeed, while the inoculation of CAF01with 50 µg of p30 POM resulted in more significantly higher levels of ASFV-specific IgGs at both tested dilutions, 150 µg of p30 POM without CAF01 rendered even more significantly higher ASFV-specific antibody levels (
Figure 3, G-H).
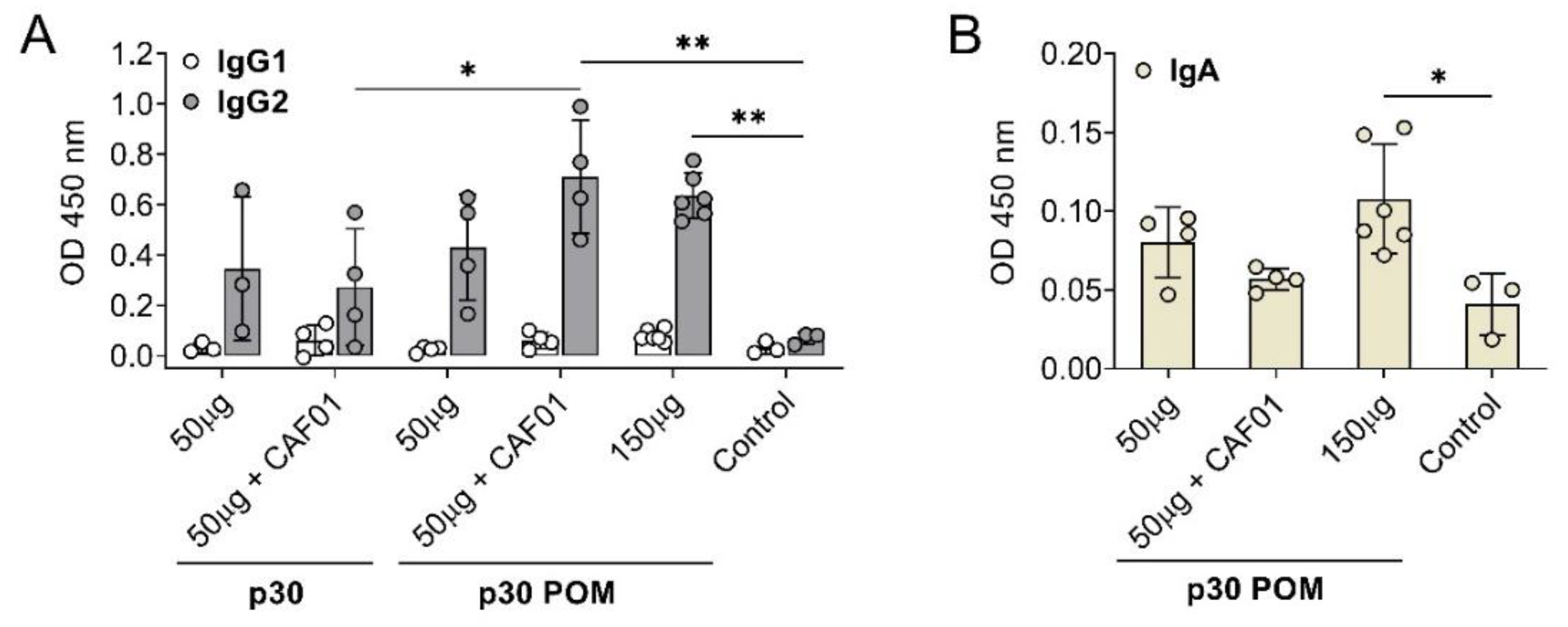
To further characterize the antibody response triggered by p30 POM, the induced IgG isotypes as well as the presence of specific IgA in serum were assessed by ELISA. Immunization with p30, both as soluble protein version and in the POM form, induced IgG2 anti-ASFV antibodies but no detectable IgG1 (
Figure 4A), indicating a Th1-like bias. This total bias towards a Th1-like response is in contrast with the balanced IgG1/IgG2 profile typically observed in sera from ASF-immune pigs after vaccination with the live attenuated ASFV BA71ΔCD2, a protective vaccine prototype developed by us [
41]. Again, only the groups receiving 50 µg of p30 POM+CAF01 or 150 µg of p30 POM showed statistically significant higher levels of IgG2 compared to the controls, and more uniform levels were found in animals inoculated with the high dose of p30 POM (
Figure 4A).
Notably, significant levels of ASFV-specific IgA were only found in serum after administration of the higher 150 µg p30 POM amount (
Figure 4B), stressing the importance of an adequate dose.
3.4. POM Proteins Stimulate the Immune System in a Non-Specific Manner
As showed above, the molecular architecture of POMs might has an immunostimulatory capability that overcomes the lack of adjuvant (
Figure 4G). This is likely due to the capacity of particulate antigens to enhance vaccine immunogenicity compared to soluble antigens [
42]. Thus, to gain further insight on the immunomodulatory properties of POMs, pulmonary alveolar macrophages (PAMs) were in vitro stimulated for 24 hours with GFP POM or left untreated (RPMI), and a multiplex Luminex assay was used to assess levels of cytokines in supernatants. Our results showed that GFP POM was able to significantly stimulate the secretion of several pro-inflammatory cytokines, including TNF, IFNγ, IL-1β and IL-4 (
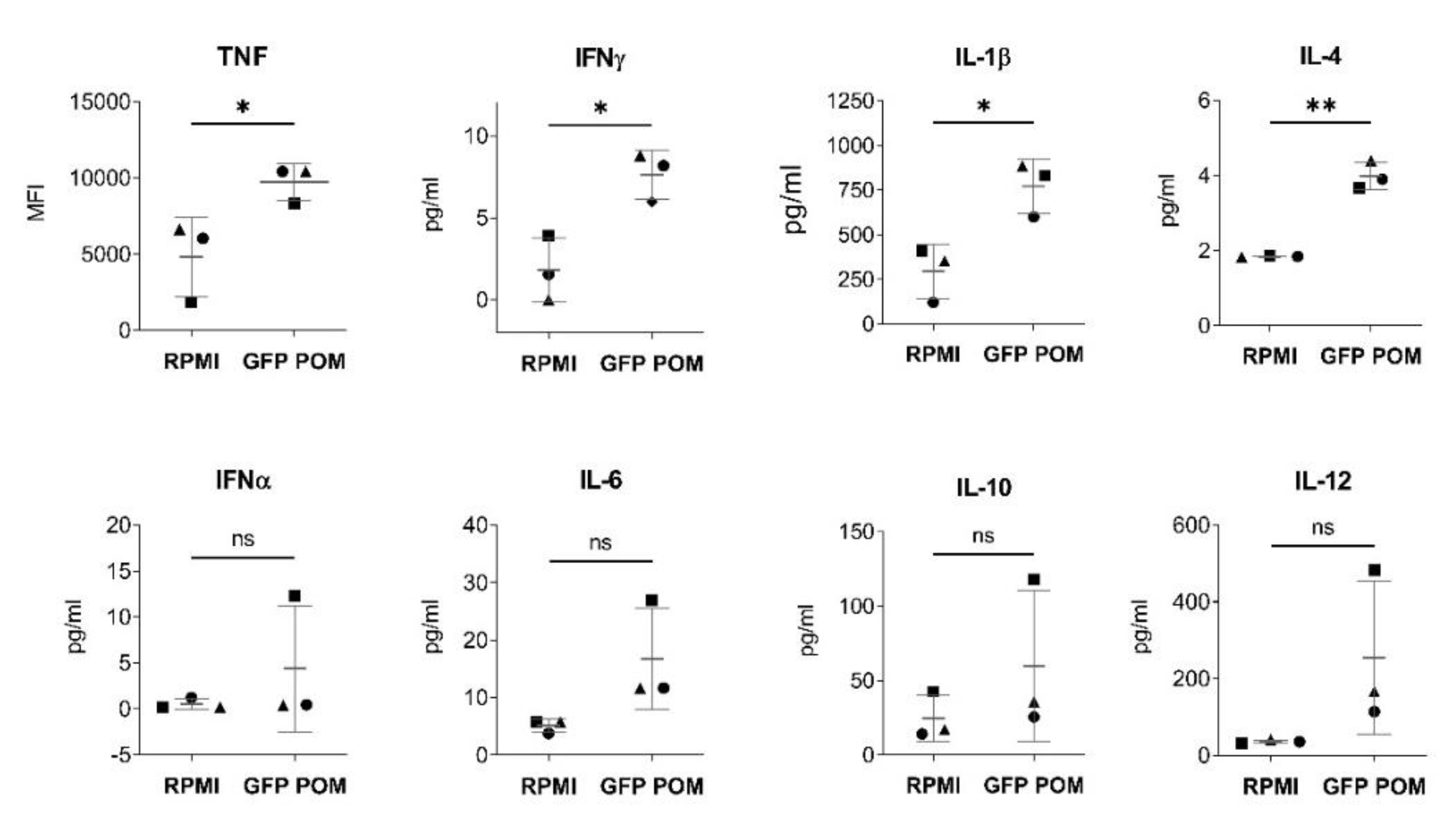
Figure 5). No deleterious effect was observed on PAMs after in vitro stimulation with POM, supporting their safety profile for future applications.
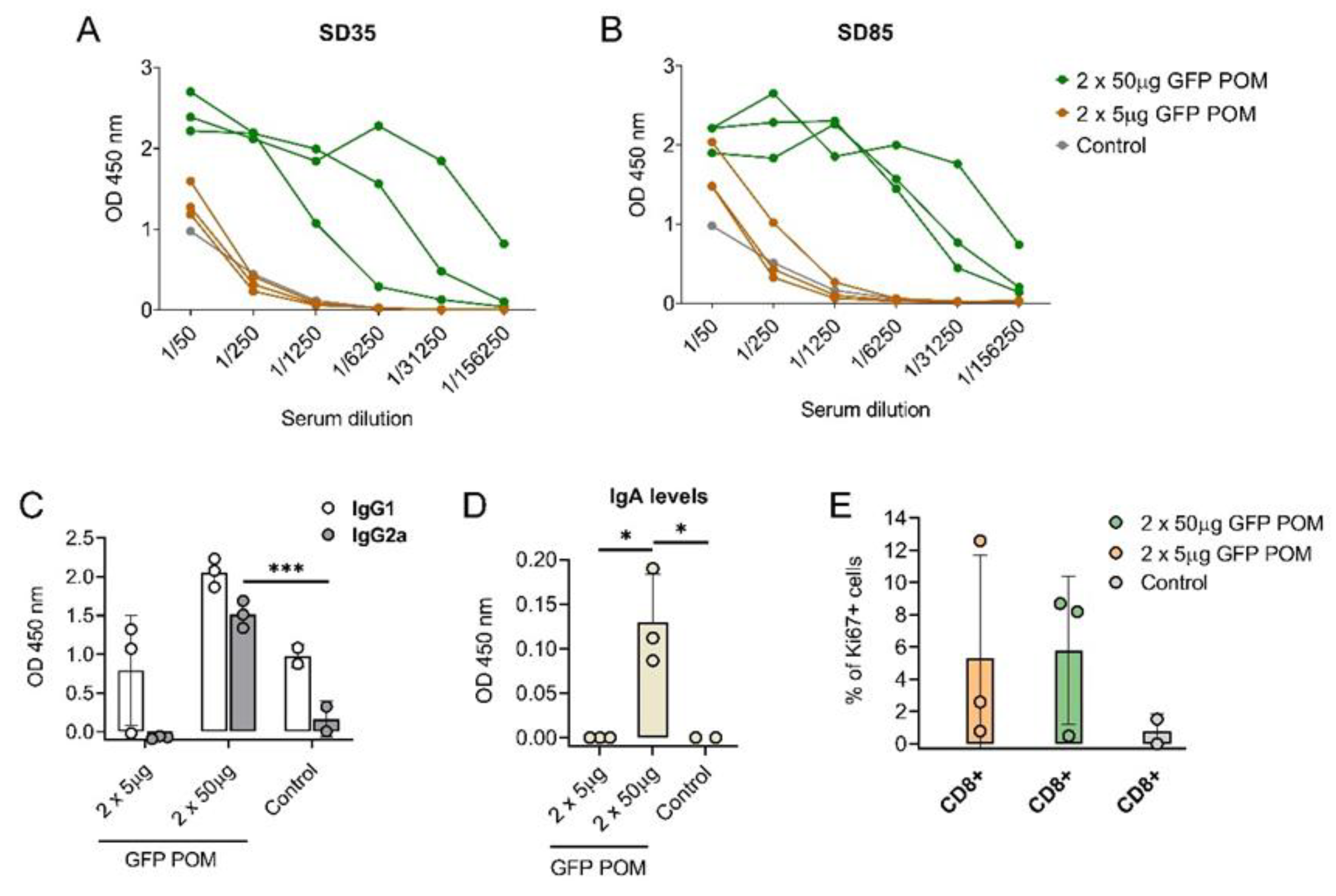
3.5. Subcutaneous Administration of GFP POM Is Safe and Immunogenic in Mice
With the intention of compiling more data regarding the immunogenicity and the immune response triggered by POM formulations, our next step was to test them in another animal model, specifically in mice. To do so, BALB/c mice were inoculated twice 2 weeks apart with either 5 or 50 µg of GFP POM. Administration of GFP POM in mice induced long-term GFP-specific antibodies in a dose dependent manner. Thus, only the 50 µg high dose induced significant levels of GFP-specific antibodies that were detectable two weeks after the second shot (SD35, first time point tested) and lasted at least nine weeks (SD85, last time point tested) (
Figure 6, A-B). The POM formulation of GFP induced significantly higher levels of GFP-specific IgG2 antibodies (
Figure 6C), in accordance with what was observed in pigs receiving p30 POM. Moreover, in this case levels of IgG2 were also augmented, although not significantly, in mice receiving the high dose of 50 µg of GFP POM (
Figure 6C). Also, in line with what was observed in pigs, anti-GFP IgA was detected in mice serum only when prime-boosting with 50 µg of GFP POM, but not with 5 µg doses (
Figure 6D). These results confirmed the potential of POM formulations to induce mucosal immunity, as well as the dose dependence of the immune response induced. Two weeks after the second administration, half of the mice were sacrificed to analyze the cellular response induced by GFP POM. Thus, splenocytes were isolated and in vitro stimulated for 5 days with 5 µg/ml of GFP POM to be assayed for lymphoproliferative activity by flow cytometry, using the Ki67 proliferation marker. In vitro stimulation of splenocytes obtained from mice immunized with GFP POM, either with 5 or 50 µg/dose, resulted in the specific proliferation of CD8
+ T cells (
Figure 6E). Despite the proliferation extent being not significantly higher, this data provides the first evidence that GFP POM can induce memory CD8
+ T cells in mice, even at low concentrations of antigen.
4. Discussion
In the context of global health and facing potential pandemic treats, developing new and more efficient immunization systems is an inexcusable need. Much has been learned about vaccination strategies from the still ongoing Covid-19 pandemics, but a consensus has been reached about the fact that both the classical and the recently developed vaccination strategies are still far to be optimal [
43,
44,
45,
46]. A main and generic problem of the current vaccination boosts is the limited time of exposure to the antigen. In natural infections, the sensing elements of the immune system perceive the immunogens during days or weeks, while the pathogen multiplication is active. Among other studies, a recent analysis of the response against the human immunodeficiency virus (HIV) Env protein has determined the relevance of prolonged exposure to relevant antigens in the development of vaccination strategies [
47], versus the conventional single antigen shot. In fact, a slow-delivery immunization strategy based on Env over 12 days dramatically improved the immunological outcomes, as such prolonged dosage mimic features of the immune response to natural infections and expands the durability and effectiveness of such response [
47]. Therefore, time-sustained dosage of immunogens is now observed as a ground-breaking and highly promising alternative to the boost-based current approaches [
48].
Linked to this scenario, divalent cations as Zn
+2 are excellent protein clustering agents that exploit its interactivity with His residues to crosslink His-tagged polypeptides as nanoparticles or, at higher metal concentrations, nanoparticle-secreting microparticles [
10,
12,
13] (
Figure 1A). The microparticle version of the resulting materials show an amyloidal architecture, inner organization and protein-leaking properties [
20,
21,
22,
27]. This is similar to the properties observed in the secretory granules from the mammalian endocrine system, that support the secretion of peptide hormones from such granular depots [
24,
25,
49,
50]. This category of protein-based dynamic repositories, in the synthetic version described here, had been so far adapted to the release of protein drugs in oncology [
23], degenerative disorders [
21] and regenerative medicine [
27]. Of course, it might be very convenient for the slow-style approach to antigen release in vaccinology. In addition, compared to other sustained drug delivery systems that require holding materials, non-functional scaffolding materials [
5], bioactive polypeptides, formulated as artificial secretory granules, are self-contained and self-released in absence of potentially toxic assisting containers. Apart from the time-sustained vaccination ideology [
48], the removal of any drug vehicle is also a main goal in the emerging conceptual setting around nanomedical drug-delivery [
51]. This of course would be of perfect applicability to immunization approaches based on selected antigens, under the umbrella of subunit vaccines in contrast to the use of the whole pathogen.
By testing here the above principles with an innovative POM formulation, based on metal-assisted, self-assembling nanoparticles (
Figure 1 and Figure 2), we demonstrate the induction of a balanced immune response, including the activation of the innate immunity as well as antigen-specific antibody and cellular responses (
Figure 3,
Figure 4,
Figure 5 and
Figure 6). The activation of the innate immune system by POM most probably provides an optimal environment for the induction of antigen-specific adaptive immune responses in the moment when the POM slowly disintegrates in the form of soluble protein components, namely unstable nanoparticles that progressively disassemble, at least in vitro, until stable dimeric protein forms. Furthermore, the induction of specific IgAs after parenteral immunization in both pig and mice (
Figure 4 and
Figure 6), opens new expectations for using the secretory granules as a transversal platform to generate broad systemic and probably mucosal immunity in any animal species. Further studies are required to test the efficacy of POM to induce mucosal immunity upon parenteral or intranasal administration.
In vaccinology, the activation of specific CD8 T-cell responses is of high importance to achieve sterilizing immunity against intracellular pathogens. Thus, while antibodies can block the pathogens in fluids, CD8 T-cells are the only ones capable to specifically kill the infected cells, destroying also the intracellular pathogen replicating within them. In fact, subunit and inactivated vaccines are traditionally not efficient at inducing CD8+ T cells. The ability of POMVs to induce a CD8+ T cell response observed here might be linked to their original, aggregated microscale nature, which can facilitate cross-presentation and therefore the priming of CD8 T-cell responses [
52,
53].
Different antibody isotypes play distinct roles in antiviral immunity. Among them, IgG2 antibodies are specifically triggered during Th1-type immune responses [
54] and exhibit superior capabilities in activating Fc receptor-mediated effector responses, which are crucial for resolving infections caused by intracellular pathogens. On the other hand, elevated levels of IgG1 are associated with Th2 immune responses and do not stimulate Fc receptor-mediated immune responses as effectively [
55]. Both POM formulations tested in this study induced IgG2a antibodies, regardless of the animal species used. This observation aligns with the activation of CD8
+ T cells by cross priming and the induction of a Th1 adaptive response because of the aggregated nature of the microparticles.
5. Conclusions
As summarized from the present data, the combination of several intrinsic features of nanoparticle-based POMs, described here, make them a promising platform for further development of vaccine candidates. Among them, it is important to stress that (i) the supramolecular architecture of the antigens might be protective from proteolytic degradation in vivo; (ii) the time-extended release of the protein mimics the constant antigen stimulation during a natural infection; (iii) the supramolecular structure of the antigen might act as an adjuvant and promote antigen presentation; (iv) the delivered antigens might transit from nanoparticles to dimeric forms, through intermediate oligomers during the disintegration of the depot. Such disintegration process might induce specific antibodies and T-helper responses, and also cytotoxic T-cells by cross-priming as already shown for conventional nanoparticle-based vaccines [
56,
57]. Together with these features, the easy and cost-effective manufacturing, as well as the safety profile due to their intrinsic molecular purity, make POMs a very promising immunization platform with a high versatility in its further adaptation for new generation vaccine formulation.
Author Contributions
Conceptualization, J.A., A.V., F.R. and E.V.; methodology, L.B.C, C.M.T. and H.L.L; L.B.C, C.M.T., H.L.L. and J.L, investigation, data curation, L.B.C and C.M.T.; writing—original draft preparation, A.V.; writing—review and editing, all authors; supervision, J.A., A.V., F.R. and E.V.; funding acquisition, J.A., A.V., F.R. and E.V. All authors have read and agreed to the published version of the manuscript.
Funding
This project was mainly funded by the proof-of-concept project NANOSWINE, granted by UAB-Generalitat de Catalunya to EV and AV. The authors are indebted to the Agencia Española de Investigación (AEI) for granting projects on the construction of protein materials of clinical interest (PID2019-105416RB-I00/AEI/10.13039/501100011033 and PDC2022-133858-I00 to EV and PID2020-116174RB-I00 to AV), to AGAUR for project SGR 2021 00092 to AV and to the Spanish Ministry of Science and Innovation (grant reference PID2019-107616RB-I00 to FR). C.M.T. was supported by a Margarita Salas grant from Universitat Autònoma de Barcelona and Ministerio de Universidades (funded by the European Union – NextGenerationEU). We also appreciate the support from CIBER -Consorcio Centro de Investigación Biomédica en Red- (CB06/01/0014), Instituto de Salud Carlos III, Ministerio de Ciencia e Innovación, and Red de Investigación en Sanidad Animal (RISA). A.V. received an ICREA ACADEMIA award.
Data Availability Statement
Data will be made available on request.
Conflicts of Interest
The authors are co-inventors on a patent claiming the use of synthetic protein depots as vaccination platforms.
References
- Harrington, M.J.; Fratzl, P. Natural load-bearing protein materials. Progress in Materials Science 2021, 120, 100767. [Google Scholar] [CrossRef]
- Miserez, A.; Yu, J.; Mohammadi, P. Protein-Based Biological Materials: Molecular Design and Artificial Production. Chemical Reviews 2023, 123, 2049–2111. [Google Scholar] [CrossRef] [PubMed]
- Xu, X.; Chen, X.; Li, J. Natural protein bioinspired materials for regeneration of hard tissues. Journal of materials chemistry. B 2020, 8, 2199–2215. [Google Scholar] [CrossRef] [PubMed]
- Shen, Y.; Levin, A.; Kamada, A.; Toprakcioglu, Z.; Rodriguez-Garcia, M.; Xu, Y.; Knowles, T.P.J. From Protein Building Blocks to Functional Materials. ACS Nano 2021, 15, 5819–5837. [Google Scholar] [CrossRef]
- Cano-Garrido, O.; Serna, N.; Unzueta, U.; Parladé, E.; Mangues, R.; Villaverde, A.; Vázquez, E. Protein scaffolds in human clinics. Biotechnology Advances 2022, 61, 108032. [Google Scholar] [CrossRef]
- Kobayashi, N.; Arai, R. Design and construction of self-assembling supramolecular protein complexes using artificial and fusion proteins as nanoscale building blocks. Current opinion in biotechnology 2017, 46, 57–65. [Google Scholar] [CrossRef]
- Zhu, J.; Avakyan, N.; Kakkis, A.; Hoffnagle, A.M.; Han, K.; Li, Y.; Zhang, Z.; Choi, T.S.; Na, Y.; Yu, C.-J.; et al. Protein Assembly by Design. Chemical Reviews 2021, 121, 13701–13796. [Google Scholar] [CrossRef]
- King, N.P.; Lai, Y.-T. Practical approaches to designing novel protein assemblies. Current Opinion in Structural Biology 2013, 23, 632–638. [Google Scholar] [CrossRef]
- Yang, L.; Liu, A.; Cao, S.; Putri, R.M.; Jonkheijm, P.; Cornelissen, J.J.L.M. Self-Assembly of Proteins: Towards Supramolecular Materials. Chemistry – A European Journal 2016, 22, 15570–15582. [Google Scholar] [CrossRef]
- Lopez-Laguna, H.; Sanchez, J.; Unzueta, U.; Mangues, R.; Vazquez, E.; Villaverde, A. Divalent Cations: A Molecular Glue for Protein Materials. Trends in biochemical sciences 2020, 45, 992–1003. [Google Scholar] [CrossRef]
- López-Laguna, H.; Sánchez, J.M.; Carratalá, J.V.; Rojas-Peña, M.; Sánchez-García, L.; Parladé, E.; Sánchez-Chardi, A.; Voltà-Durán, E.; Serna, N.; Cano-Garrido, O.; et al. Biofabrication of functional protein nanoparticles through simple His-tag engineering. ACS sustainable chemistry & engineering 2021, 9, 12341–12354. [Google Scholar] [CrossRef]
- López-Laguna, H.; Voltà-Durán, E.; Parladé, E.; Villaverde, A.; Vázquez, E.; Unzueta, U. Insights on the emerging biotechnology of histidine-rich peptides. Biotechnology Advances 2022, 54, 107817. [Google Scholar] [CrossRef]
- López-Laguna, H.; Parladé, E.; Álamo, P.; Sánchez, J.M.; Voltà-Durán, E.; Serna, N.; Sánchez-García, L.; Cano-Garrido, O.; Sánchez-Chardi, A.; Villaverde, A.; et al. In Vitro Fabrication of Microscale Secretory Granules. Advanced Functional Materials 2021, 31, 2100914. [Google Scholar] [CrossRef]
- Parlade, E.; Sanchez, J.M.; Lopez-Laguna, H.; Unzueta, U.; Villaverde, A.; Vazquez, E. Protein features instruct the secretion dynamics from metal-supported synthetic amyloids. International journal of biological macromolecules 2023, 250, 126164. [Google Scholar] [CrossRef]
- Lopez-Laguna, H.; Unzueta, U.; Conchillo-Sole, O.; Sanchez-Chardi, A.; Pesarrodona, M.; Cano-Garrido, O.; Volta, E.; Sanchez-Garcia, L.; Serna, N.; Saccardo, P.; et al. Assembly of histidine-rich protein materials controlled through divalent cations. Acta Biomater 2019, 83, 257–264. [Google Scholar] [CrossRef]
- Unzueta, U.; Ferrer-Miralles, N.; Cedano, J.; Zikung, X.; Pesarrodona, M.; Saccardo, P.; Garcia-Fruitos, E.; Domingo-Espin, J.; Kumar, P.; Gupta, K.C.; et al. Non-amyloidogenic peptide tags for the regulatable self-assembling of protein-only nanoparticles. Biomaterials 2012, 33, 8714–8722. [Google Scholar] [CrossRef] [PubMed]
- López-Laguna, H.; Sánchez-García, L.; Serna, N.; Voltà-Durán, E.; Sánchez, J.M.; Sánchez-Chardi, A.; Unzueta, U.; Łoś, M.; Villaverde, A.; Vázquez, E. Engineering Protein Nanoparticles Out from Components of the Human Microbiome. Small 2020, 16, 2001885. [Google Scholar] [CrossRef]
- Núñez, Y.; Garcia-León, A.; Falgàs, A.; Serna, N.; Sánchez-García, L.; Garrido, A.; Sierra, J.; Gallardo, A.; Unzueta, U.; Vázquez, E.; et al. T22-PE24-H6 Nanotoxin Selectively Kills CXCR4-High Expressing AML Patient Cells In Vitro and Potently Blocks Dissemination In Vivo. Pharmaceutics 2023, 15, 727. [Google Scholar] [CrossRef]
- Pallarès, V.; Unzueta, U.; Falgàs, A.; Aviñó, A.; Núñez, Y.; García-León, A.; Sánchez-García, L.; Serna, N.; Gallardo, A.; Alba-Castellón, L.; et al. A multivalent Ara-C-prodrug nanoconjugate achieves selective ablation of leukemic cells in an acute myeloid leukemia mouse model. Biomaterials 2022, 280, 121258. [Google Scholar] [CrossRef]
- Chen, T.-Y.; Cheng, W.-J.; Horng, J.-C.; Hsu, H.-Y. Artificial peptide-controlled protein release of Zn2+-triggered, self-assembled histidine-tagged protein microparticle. Colloids and Surfaces B: Biointerfaces 2020, 187, 110644. [Google Scholar] [CrossRef]
- Zhao, L.; Luo, L.; Meng, F. Metal-mediated nanobody assemblies as potent alleviators of human islet amyloid polypeptide aggregation. Materials Chemistry Frontiers 2023, 7, 2068–2077. [Google Scholar] [CrossRef]
- Sánchez, J.M.; López-Laguna, H.; Álamo, P.; Serna, N.; Sánchez-Chardi, A.; Nolan, V.; Cano-Garrido, O.; Casanova, I.; Unzueta, U.; Vazquez, E.; et al. Artificial Inclusion Bodies for Clinical Development. Advanced Science 2020, 7, 1902420. [Google Scholar] [CrossRef]
- Álamo, P.; Parladé, E.; López-Laguna, H.; Voltà-Durán, E.; Unzueta, U.; Vazquez, E.; Mangues, R.; Villaverde, A. Ion-dependent slow protein release from in vivo disintegrating micro-granules. Drug Delivery 2021, 28, 2383–2391. [Google Scholar] [CrossRef]
- Maji, S.K.; Schubert, D.; Rivier, C.; Lee, S.; Rivier, J.E.; Riek, R. Amyloid as a depot for the formulation of long-acting drugs. PLoS biology 2008, 6, e17. [Google Scholar] [CrossRef] [PubMed]
- Maji, S.K.; Perrin, M.H.; Sawaya, M.R.; Jessberger, S.; Vadodaria, K.; Rissman, R.A.; Singru, P.S.; Nilsson, K.P.R.; Simon, R.; Schubert, D.; et al. Functional Amyloids As Natural Storage of Peptide Hormones in Pituitary Secretory Granules. Science 2009, 325, 328–332. [Google Scholar] [CrossRef] [PubMed]
- Serna, N.; Falgàs, A.; García-León, A.; Unzueta, U.; Núñez, Y.; Sánchez-Chardi, A.; Martínez-Torró, C.; Mangues, R.; Vazquez, E.; Casanova, I.; et al. Time-Prolonged Release of Tumor-Targeted Protein–MMAE Nanoconjugates from Implantable Hybrid Materials. Pharmaceutics 2022, 14, 192. [Google Scholar]
- Serna, N.; Cano-Garrido, O.; Sánchez, J.M.; Sánchez-Chardi, A.; Sánchez-García, L.; López-Laguna, H.; Fernández, E.; Vázquez, E.; Villaverde, A. Release of functional fibroblast growth factor-2 from artificial inclusion bodies. Journal of Controlled Release 2020, 327, 61–69. [Google Scholar] [CrossRef]
- Prados, F.J.; Viñuela, E.; Alcamí, A. Sequence and characterization of the major early phosphoprotein p32 of African swine fever virus. Journal of Virology 1993, 67, 2475–2485. [Google Scholar] [CrossRef]
- Tulman, E.R.; Delhon, G.A.; Ku, B.K.; Rock, D.L. African Swine Fever Virus. In Lesser Known Large dsDNA Viruses; Van Etten, J.L., Ed.; Springer Berlin Heidelberg: Berlin, Heidelberg, 2009; pp. 43–87. [Google Scholar]
- Wang, Y.; Kang, W.; Yang, W.; Zhang, J.; Li, D.; Zheng, H. Structure of African Swine Fever Virus and Associated Molecular Mechanisms Underlying Infection and Immunosuppression: A Review. Frontiers in Immunology 2021, 12. [Google Scholar] [CrossRef]
- Netherton, C.L.; Connell, S.; Benfield, C.T.O.; Dixon, L.K. The Genetics of Life and Death: Virus-Host Interactions Underpinning Resistance to African Swine Fever, a Viral Hemorrhagic Disease. Frontiers in Genetics 2019, 10. [Google Scholar] [CrossRef]
- Gallardo, M.C.; Reoyo, A.d.l.T.; Fernández-Pinero, J.; Iglesias, I.; Muñoz, M.J.; Arias, M.L. African swine fever: a global view of the current challenge. Porcine Health Management 2015, 1, 21. [Google Scholar] [CrossRef]
- Bosch-Camós, L.; López, E.; Rodriguez, F. African swine fever vaccines: a promising work still in progress. Porcine Health Management 2020, 6, 17. [Google Scholar] [CrossRef]
- Vergne, T.; Chen-Fu, C.; Li, S.; Cappelle, J.; Edwards, J.; Martin, V.; Pfeiffer, D.U.; Fusheng, G.; Roger, F.L. Pig empire under infectious threat: risk of African swine fever introduction into the People's Republic of China. Veterinary Record 2017, 181, 117–117. [Google Scholar] [CrossRef]
- Wu, K.; Liu, J.; Wang, L.; Fan, S.; Li, Z.; Li, Y.; Yi, L.; Ding, H.; Zhao, M.; Chen, J. Current State of Global African Swine Fever Vaccine Development under the Prevalence and Transmission of ASF in China. Vaccines 2020, 8, 531. [Google Scholar] [CrossRef] [PubMed]
- Gallardo, C.; Soler, A.; Nieto, R.; Carrascosa, A.L.; De Mia, G.M.; Bishop, R.P.; Martins, C.; Fasina, F.O.; Couacy-Hymman, E.; Heath, L.; et al. Comparative evaluation of novel African swine fever virus (ASF) antibody detection techniques derived from specific ASF viral genotypes with the OIE internationally prescribed serological tests. Veterinary microbiology 2013, 162, 32–43. [Google Scholar] [CrossRef] [PubMed]
- Vazquez, E.; Roldan, M.; Diez-Gil, C.; Unzueta, U.; Domingo-Espin, J.; Cedano, J.; Conchillo, O.; Ratera, I.; Veciana, J.; Daura, X.; et al. Protein nanodisk assembling and intracellular trafficking powered by an arginine-rich (R9) peptide. Nanomedicine (Lond) 2010, 5, 259–268. [Google Scholar] [CrossRef] [PubMed]
- Cespedes, M.V.; Unzueta, U.; Tatkiewicz, W.; Sanchez-Chardi, A.; Conchillo-Sole, O.; Alamo, P.; Xu, Z.; Casanova, I.; Corchero, J.L.; Pesarrodona, M.; et al. In vivo architectonic stability of fully de novo designed protein-only nanoparticles. ACS Nano 2014, 8, 4166–4176. [Google Scholar] [CrossRef] [PubMed]
- Corchero, J.L.; Favaro, M.T.P.; Márquez-Martínez, M.; Lascorz, J.; Martínez-Torró, C.; Sánchez, J.M.; López-Laguna, H.; de Souza Ferreira, L.C.; Vázquez, E.; Ferrer-Miralles, N.; et al. Recombinant Proteins for Assembling as Nano- and Micro-Scale Materials for Drug Delivery: A Host Comparative Overview. Pharmaceutics 2023, 15, 1197. [Google Scholar] [CrossRef] [PubMed]
- Alamo, P.; Parlade, E.; Favaro, M.T.P.; Gallardo, A.; Mendoza, R.; Ferreira, L.C.S.; Roher, N.; Mangues, R.; Villaverde, A.; Vazquez, E. Probing the Biosafety of Implantable Artificial Secretory Granules for the Sustained Release of Bioactive Proteins. ACS applied materials & interfaces 2023, 15, 39167–39175. [Google Scholar] [CrossRef]
- Monteagudo, P.L.; Lacasta, A.; López, E.; Bosch, L.; Collado, J.; Pina-Pedrero, S.; Correa-Fiz, F.; Accensi, F.; Navas, M.J.; Vidal, E. BA71ΔCD2: a new recombinant live attenuated African swine fever virus with cross-protective capabilities. Journal of virology 2017, 91, e01058–01017. [Google Scholar] [CrossRef] [PubMed]
- Snapper, C.M. Distinct Immunologic Properties of Soluble Versus Particulate Antigens. Front Immunol 2018, 9, 598. [Google Scholar] [CrossRef]
- Tennenbaum, S. Simple criteria for finding (nearly) optimal vaccination strategies. Journal of Theoretical Biology 2008, 250, 673–683. [Google Scholar] [CrossRef]
- Hotez, P.J. SARS-CoV-2 variants offer a second chance to fix vaccine inequities. Nature Reviews Microbiology 2023, 21, 127–128. [Google Scholar] [CrossRef]
- Morens, D.M.; Taubenberger, J.K.; Fauci, A.S. Rethinking next-generation vaccines for coronaviruses, influenzaviruses, and other respiratory viruses. Cell Host & Microbe 2023, 31, 146–157. [Google Scholar] [CrossRef]
- Hameed, S.A.; Paul, S.; Dellosa, G.K.Y.; Jaraquemada, D.; Bello, M.B. Towards the future exploration of mucosal mRNA vaccines against emerging viral diseases; lessons from existing next-generation mucosal vaccine strategies. NPJ vaccines 2022, 7, 71. [Google Scholar] [CrossRef]
- Lee, J.H.; Sutton, H.J.; Cottrell, C.A.; Phung, I.; Ozorowski, G.; Sewall, L.M.; Nedellec, R.; Nakao, C.; Silva, M.; Richey, S.T.; et al. Long-primed germinal centres with enduring affinity maturation and clonal migration. Nature 2022, 609, 998–1004. [Google Scholar] [CrossRef] [PubMed]
- Quast, I.; Tarlinton, D. Time is of the essence for vaccine success. Nature Immunology 2022, 1–3. [Google Scholar] [CrossRef]
- Jacob, R.S.; Anoop, A.; Maji, S.K. Protein nanofibrils as storage forms of peptide drugs and hormones. Adv Exp Med Biol 2019, 1174, 265–290. [Google Scholar] [PubMed]
- Jacob, R.S.; Das, S.; Ghosh, S.; Anoop, A.; Jha, N.N.; Khan, T.; Singru, P.; Kumar, A.; Maji, S.K. Amyloid formation of growth hormone in presence of zinc: relevance to its storage in secretory granules. Scientific reports 2016, 6, 1–18. [Google Scholar] [CrossRef]
- Shen, J.; Wolfram, J.; Ferrari, M.; Shen, H. Taking the vehicle out of drug delivery. Materials today 2017, 20, 95–97. [Google Scholar] [CrossRef]
- Muntjewerff, E.M.; Meesters, L.D.; van den Bogaart, G. Antigen Cross-Presentation by Macrophages. Front Immunol 2020, 11, 1276. [Google Scholar] [CrossRef]
- Flinsenberg, T.W.; Boes, M. Application of antigen cross-presentation research into patient care. Front Immunol 2014, 5, 287. [Google Scholar] [CrossRef] [PubMed]
- Snapper, C.M.; Paul, W.E. Interferon-γ and B Cell Stimulatory Factor-1 Reciprocally Regulate Ig Isotype Production. Science 1987, 236, 944–947. [Google Scholar] [CrossRef] [PubMed]
- Nimmerjahn, F.; Ravetch, J.V. Divergent Immunoglobulin G Subclass Activity Through Selective Fc Receptor Binding. Science 2005, 310, 1510–1512. [Google Scholar] [CrossRef] [PubMed]
- Favaro, M.T.P.; Rodrigues-Jesus, M.J.; Venceslau-Carvalho, A.A.; Alves, R.; Pereira, L.R.; Pereira, S.S.; Andreata-Santos, R.; de Souza Ferreira, L.C. Nanovaccine based on self-assembling nonstructural protein 1 boosts antibody responses to Zika virus. Nanomedicine: nanotechnology, biology, and medicine 2021, 32, 102334. [Google Scholar] [CrossRef]
- de Pinho Favaro, M.T.; Atienza-Garriga, J.; Martinez-Torro, C.; Parlade, E.; Vazquez, E.; Corchero, J.L.; Ferrer-Miralles, N.; Villaverde, A. Recombinant vaccines in 2022: a perspective from the cell factory. Microbial cell factories 2022, 21, 203. [Google Scholar] [CrossRef]
Figure 1.
Construction and characterization of RK4-P30-H6 and GFP-H6. (A) Architectonic principles governing the POM principle, namely microparticle generation out of nanoparticles and further nanoparticle release. His tagged proteins tend to self-assemble, upon recombinant production and Ni2+-based purification, into oligomeric nanoparticles, assisted by divalent cations. A cationic amino acid N-terminal stretch favours this process. The addition of a molar excess of cationic Zn produces the immediate formation of microscale particles. Upon in vivo administration and upon Zn dilution, these materials release differently stable nanoparticles. (B) Schematic representation of RK4-P30-H6 and GFP-H6 constructs. (C) RK4-P30-H6 characterization by H6 immunodetection in Western blot with anti-His monoclonal antibody (C, left). Size of the purified protein determined by DLS. The protein size was also measured under chelating conditions (2 mM EDTA + 1% SDS) (C, right). (D) Immunodetection of GFP-H6 by Western Blot (D, left) and size of the construct analyzed by DLS (D, right).
Figure 1.
Construction and characterization of RK4-P30-H6 and GFP-H6. (A) Architectonic principles governing the POM principle, namely microparticle generation out of nanoparticles and further nanoparticle release. His tagged proteins tend to self-assemble, upon recombinant production and Ni2+-based purification, into oligomeric nanoparticles, assisted by divalent cations. A cationic amino acid N-terminal stretch favours this process. The addition of a molar excess of cationic Zn produces the immediate formation of microscale particles. Upon in vivo administration and upon Zn dilution, these materials release differently stable nanoparticles. (B) Schematic representation of RK4-P30-H6 and GFP-H6 constructs. (C) RK4-P30-H6 characterization by H6 immunodetection in Western blot with anti-His monoclonal antibody (C, left). Size of the purified protein determined by DLS. The protein size was also measured under chelating conditions (2 mM EDTA + 1% SDS) (C, right). (D) Immunodetection of GFP-H6 by Western Blot (D, left) and size of the construct analyzed by DLS (D, right).
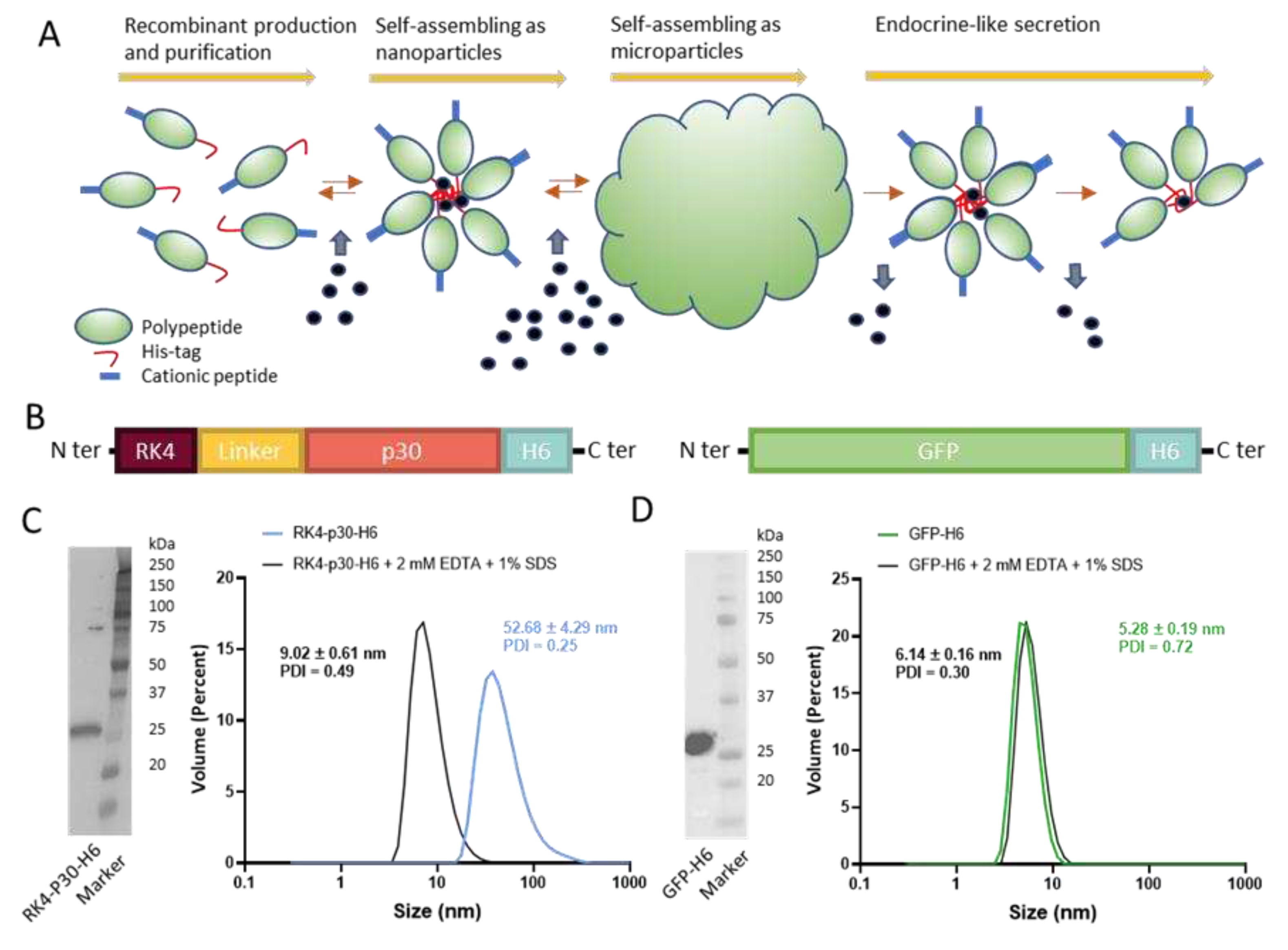
Figure 2.
Formation and characterization of p30 POMs and GFP POMs. Representative micrographs of p30 POMs (A) and GFP POMs (B) obtained by SEM (scale bar represents 1 µm). (C) Size of p30 POMs and of the soluble protein released in vitro after seven days as determined by DLS (left). The relative amount of soluble protein released from p30 POM for seven days is also shown (right). (D) Size of GFP POMs and of the soluble protein released from these microparticles as determined by DLS at day seven (left). The relative amount of soluble protein released from GFP POM for seven days is also depicted (right).
Figure 2.
Formation and characterization of p30 POMs and GFP POMs. Representative micrographs of p30 POMs (A) and GFP POMs (B) obtained by SEM (scale bar represents 1 µm). (C) Size of p30 POMs and of the soluble protein released in vitro after seven days as determined by DLS (left). The relative amount of soluble protein released from p30 POM for seven days is also shown (right). (D) Size of GFP POMs and of the soluble protein released from these microparticles as determined by DLS at day seven (left). The relative amount of soluble protein released from GFP POM for seven days is also depicted (right).
Figure 3.
Subcutaneous inoculation of p30 secretory granules in pigs induces ASFV-specific antibodies. ASFV-specific IgGs in sera from pigs inoculated with 50 µg of p30 Soluble (A) without or (B) with CAF01, or with 50 µg of p30 POM (C) without or (D) with CAF01, and (E) the control group. (F) ASFV-specific IgGs in sera from pigs receiving 150 µg of p30 POM or PBS as control from the second experiment. (G) ASFV-specific IgG titres in pig sera two weeks after the second administration (SD35). (H) Statistical analyses of ASFV-specific IgG titres in pig sera two weeks after the second administration (SD35). Statistical significance was determined by one-way ANOVA followed by Tukey’s multiple comparisons test and is displayed in GraphPad style (ns p > 0.05, * p ≤ 0.05, ** p ≤ 0.01, *** p ≤ 0.001, **** p ≤ 0.0001).
Figure 3.
Subcutaneous inoculation of p30 secretory granules in pigs induces ASFV-specific antibodies. ASFV-specific IgGs in sera from pigs inoculated with 50 µg of p30 Soluble (A) without or (B) with CAF01, or with 50 µg of p30 POM (C) without or (D) with CAF01, and (E) the control group. (F) ASFV-specific IgGs in sera from pigs receiving 150 µg of p30 POM or PBS as control from the second experiment. (G) ASFV-specific IgG titres in pig sera two weeks after the second administration (SD35). (H) Statistical analyses of ASFV-specific IgG titres in pig sera two weeks after the second administration (SD35). Statistical significance was determined by one-way ANOVA followed by Tukey’s multiple comparisons test and is displayed in GraphPad style (ns p > 0.05, * p ≤ 0.05, ** p ≤ 0.01, *** p ≤ 0.001, **** p ≤ 0.0001).
Figure 4.
Subcutaneous inoculation of p30 POM in pigs induces an IgG2 bias and detectable IgA in serum. (A) ASFV-specific IgG1 and IgG2 in sera (1/100 dilution) from pigs receiving p30 or p30 POM two weeks after the second inoculation (SD35) assessed by ELISA. (B) ASFV-specific IgA in sera (1/100 dilution) from pigs receiving p30 POM two weeks after the second inoculation (SD35) determined by ELISA. Statistical significance was determined by one-way ANOVA followed by Tukey’s multiple comparisons test and is displayed in GraphPad style (* p ≤ 0.05, ** p ≤ 0.01).
Figure 4.
Subcutaneous inoculation of p30 POM in pigs induces an IgG2 bias and detectable IgA in serum. (A) ASFV-specific IgG1 and IgG2 in sera (1/100 dilution) from pigs receiving p30 or p30 POM two weeks after the second inoculation (SD35) assessed by ELISA. (B) ASFV-specific IgA in sera (1/100 dilution) from pigs receiving p30 POM two weeks after the second inoculation (SD35) determined by ELISA. Statistical significance was determined by one-way ANOVA followed by Tukey’s multiple comparisons test and is displayed in GraphPad style (* p ≤ 0.05, ** p ≤ 0.01).
Figure 5.
In vitro stimulation with GFP POM induces activation of PAMs. Cytokine levels in culture supernatants of PAMs in vitro stimulated for 24 hours with 5 µg/ml of GFP POM or left unstimulated (RPMI) quantified by Luminex-based multiplex assay. Statistical significance was determined by unpaired two-tailed t-test for normally distributed data, or two-tailed Mann-Whitney U test for not normally distributed data and is displayed in GraphPad style (p > 0.05 ns, * p ≤ 0.05, ** p ≤ 0.01).
Figure 5.
In vitro stimulation with GFP POM induces activation of PAMs. Cytokine levels in culture supernatants of PAMs in vitro stimulated for 24 hours with 5 µg/ml of GFP POM or left unstimulated (RPMI) quantified by Luminex-based multiplex assay. Statistical significance was determined by unpaired two-tailed t-test for normally distributed data, or two-tailed Mann-Whitney U test for not normally distributed data and is displayed in GraphPad style (p > 0.05 ns, * p ≤ 0.05, ** p ≤ 0.01).
Figure 6.
Subcutaneous inoculation of microparticulated GFP induces long-lasting dose-dependent production of GFP-specific antibodies and CD8 T cells in mice. (A, B) GFP-specific antibody levels in sera from mice receiving GFP POM assessed by ELISA two (SD35, A) and nine (SD85, B) weeks after the second inoculation. (C) GFP-specific IgG1 and IgG2a in sera from mice receiving GFP POM nine weeks after the second inoculation. (D) GFP-specific IgA in sera from mice receiving GFP POM nine weeks after the second inoculation. (E) Percentage of proliferating (Ki67+) CD4+ and CD8+ T cells in splenocytes after in vitro stimulation for five days with 5 µg/ml of GFP POM. Percentages obtained from untreated cells were subtracted. Statistical significance was assessed by one-way ANOVA followed by Tukey’s multiple comparisons test and is displayed in GraphPad style (* p ≤ 0.05, *** p ≤ 0.001).
Figure 6.
Subcutaneous inoculation of microparticulated GFP induces long-lasting dose-dependent production of GFP-specific antibodies and CD8 T cells in mice. (A, B) GFP-specific antibody levels in sera from mice receiving GFP POM assessed by ELISA two (SD35, A) and nine (SD85, B) weeks after the second inoculation. (C) GFP-specific IgG1 and IgG2a in sera from mice receiving GFP POM nine weeks after the second inoculation. (D) GFP-specific IgA in sera from mice receiving GFP POM nine weeks after the second inoculation. (E) Percentage of proliferating (Ki67+) CD4+ and CD8+ T cells in splenocytes after in vitro stimulation for five days with 5 µg/ml of GFP POM. Percentages obtained from untreated cells were subtracted. Statistical significance was assessed by one-way ANOVA followed by Tukey’s multiple comparisons test and is displayed in GraphPad style (* p ≤ 0.05, *** p ≤ 0.001).
|
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).