Submitted:
25 January 2024
Posted:
26 January 2024
You are already at the latest version
Abstract
Keywords:
1. Introduction
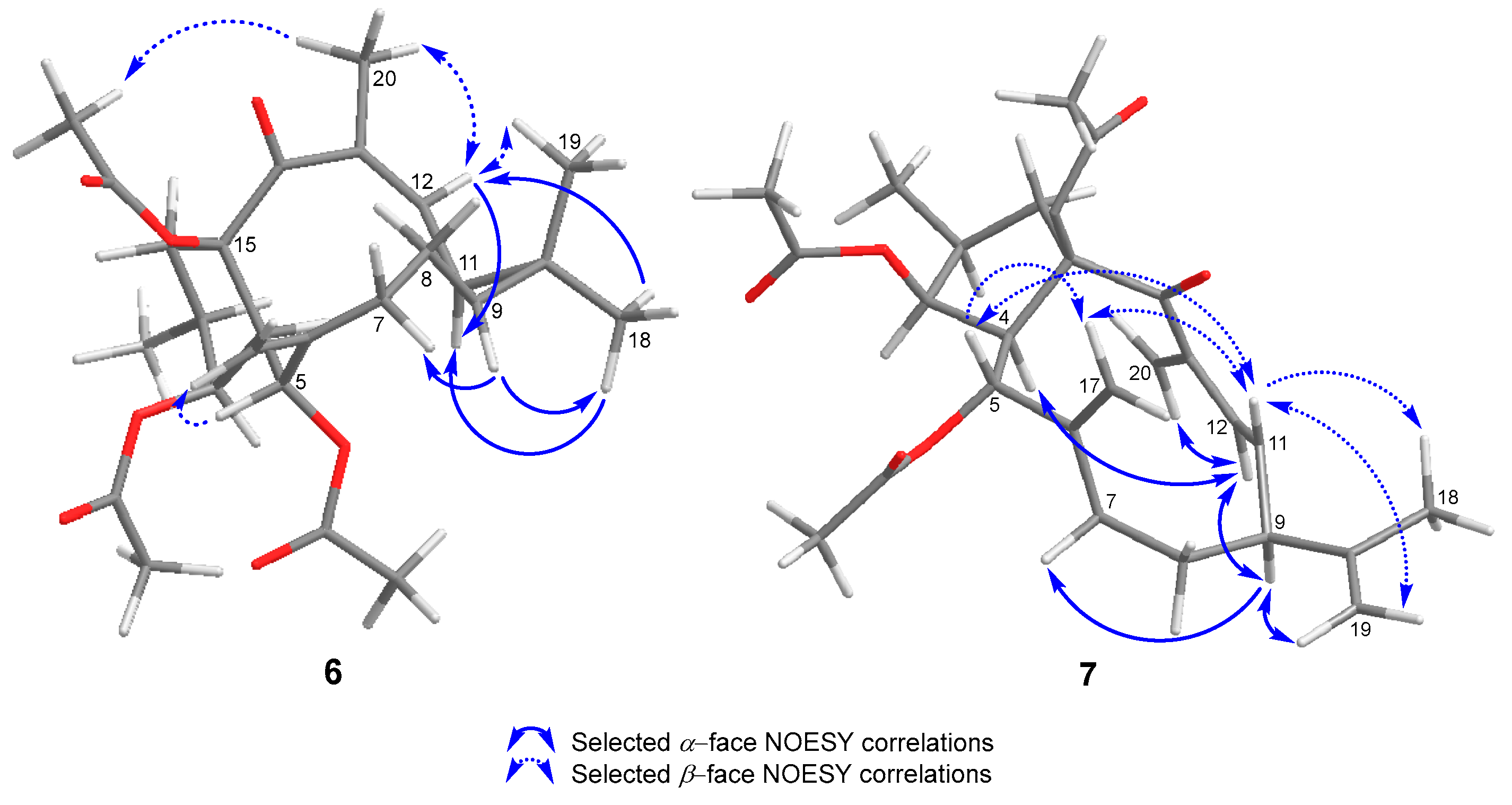
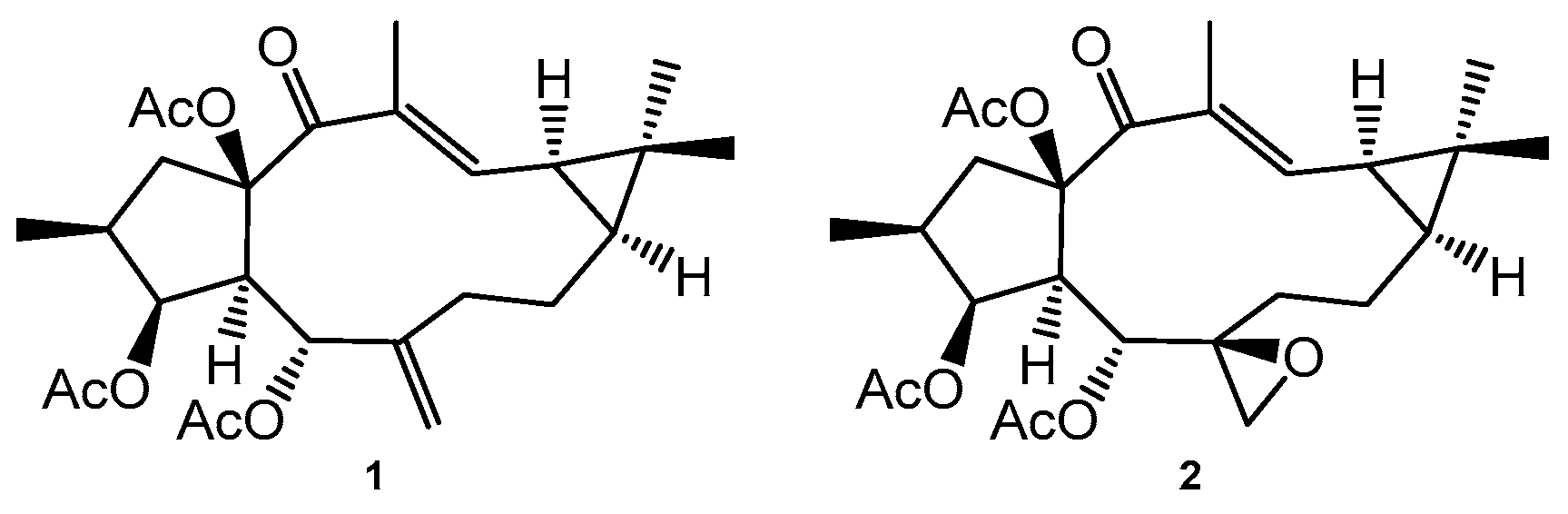
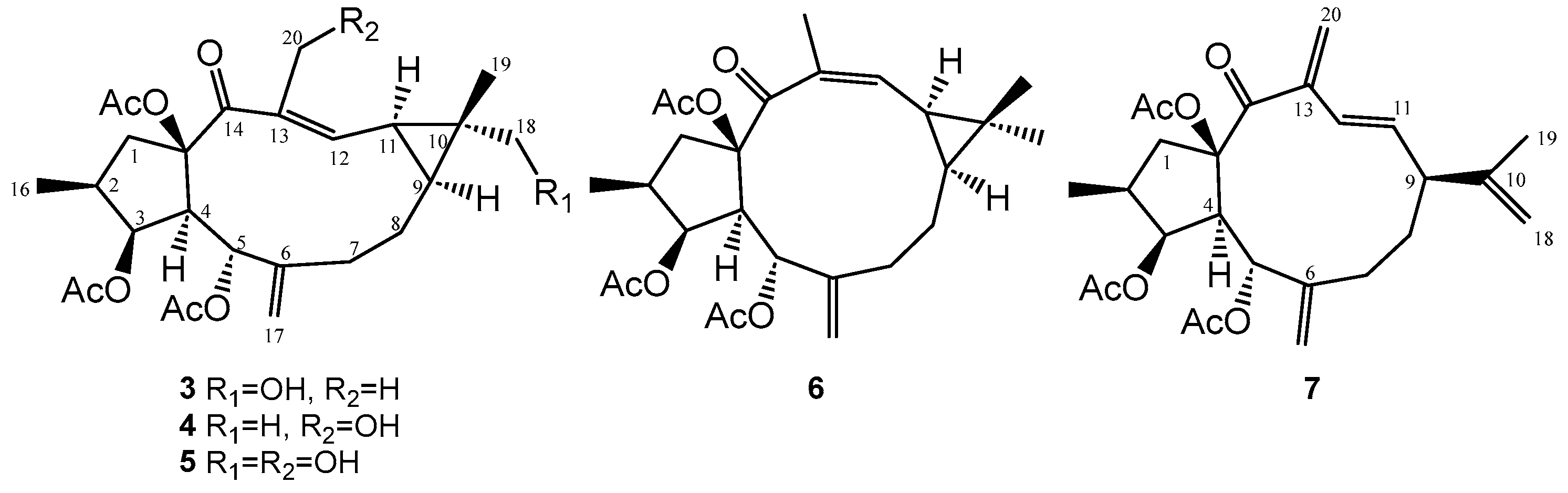
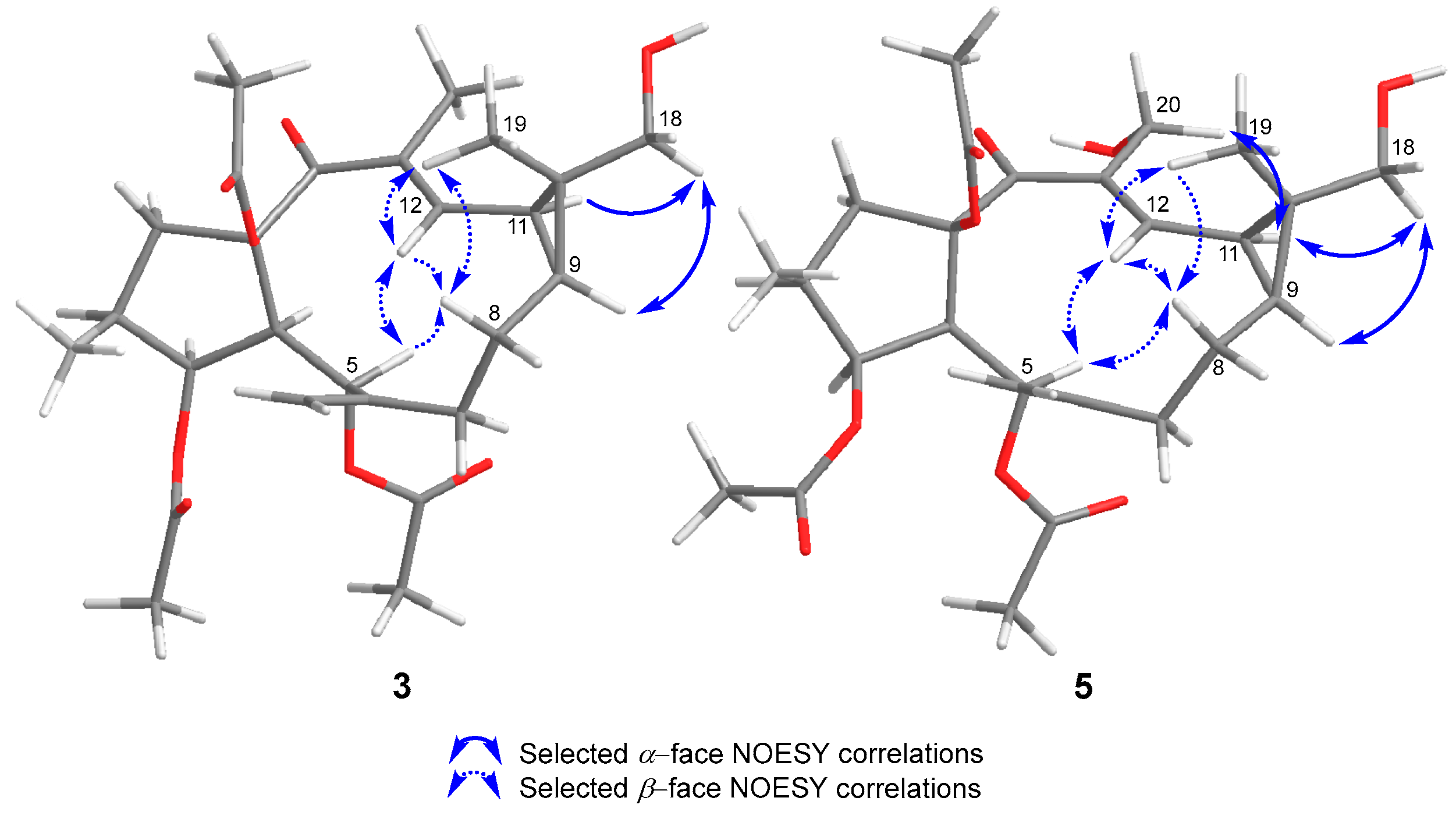
2. Results and Discussion
3. Materials and Methods
3.1. General Experimental Procedures
3.2. Plant material
3.3. Microorganism and Identification
3.4. Extraction and isolation of euphoboetirane A (1)
3.5. Biotransformation of euphoboetirane A (1)
3.6. Extraction, Isolation, and Characterization of biotransformation products
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Zhan, Z.; Li, S.; Chu, W.; Yin, S. Euphorbia Diterpenoids: Isolation, Structure, Bioactivity, Biosynthesis, and Synthesis (2013–2021). Nat. Prod. Rep. 2022, 39, 2132–2174. [Google Scholar] [CrossRef] [PubMed]
- Durán-Peña, M.J.; Botubol Ares, J.M.; Collado, I.G.; Hernández-Galán, R. Biologically Active Diterpenes Containing a Gem-Dimethylcyclopropane Subunit: An Intriguing Source of PKC Modulators. Nat. Prod. Rep. 2014, 31, 940–952. [Google Scholar] [CrossRef] [PubMed]
- Vasas, A.; Hohmann, J. Euphorbia Diterpenes: Isolation, Structure, Biological Activity, and Synthesis (2008–2012). Chem. Rev. 2014, 114, 8579–8612. [Google Scholar] [CrossRef] [PubMed]
- Vela, F.; Ezzanad, A.; Hunter, A.C.; Macías-Sánchez, A.J.; Hernández-Galán, R. Pharmacological Potential of Lathyrane-Type Diterpenoids from Phytochemical Sources. Pharmaceuticals 2022, 15, 780. [Google Scholar] [CrossRef] [PubMed]
- Flores-Giubi, E.; Geribaldi-Doldán, N.; Murillo-Carretero, M.; Castro, C.; Durán-Patrón, R.; Macías-Sánchez, A.J.; Hernández-Galán, R. Lathyrane, Premyrsinane, and Related Diterpenes from Euphorbia boetica: Effect on in Vitro Neural Progenitor Cell Proliferation. J. Nat. Prod. 2019, 82, 2517–2528. [Google Scholar] [CrossRef] [PubMed]
- Domínguez-García, S.; Geribaldi-Doldán, N.; Gómez-Oliva, R.; Ruiz, F.A.; Carrascal, L.; Bolívar, J.; Verástegui, C.; Garcia-Alloza, M.; Macías-Sánchez, A.J.; Hernández-Galán, R.; et al. A Novel PKC Activating Molecule Promotes Neuroblast Differentiation and Delivery of Newborn Neurons in Brain Injuries. Cell Death Dis. 2020, 11, 262. [Google Scholar] [CrossRef] [PubMed]
- Murillo-Carretero, M.; Geribaldi-Doldán, N.; Flores-Giubi, E.; García-Bernal, F.; Navarro-Quiroz, E.A.; Carrasco, M.; Macías-Sánchez, A.J.; Herrero-Foncubierta, P.; Delgado-Ariza, A.; Verástegui, C.; Domínguez-Riscart, J.; Daoubi, M.; Hernández-Galán, R.; Castro, C. ELAC (3,12-Di-O-Acetyl-8-O-Tigloilingol), a Plant-Derived Lathyrane Diterpene, Induces Subventricular Zone Neural Progenitor Cell Proliferation through PKCβ Activation. Brit. J. Pharmacol. 2017, 174, 2373–2392. [Google Scholar] [CrossRef] [PubMed]
- de Souza, G.G.; Oliveira, T.S.; Takahashi, J.A.; Collado, I.G.; Macías-Sánchez, A.J.; Hernández-Galán, R. Biotransformation of Clovane Derivatives. Whole Cell Fungi Mediated Domino Synthesis of Rumphellclovane A. Org. Biomol. Chem. 2012, 10, 3315–3320. [Google Scholar] [CrossRef] [PubMed]
- Wu, Y.; Yin, C.; Cao, Y.; Liu, X.; Yu, J.; Zhang, J.; Yu, B.; Cheng, Z. Regioselective Hydroxylation of Lathyrane Diterpenoids Biocatalyzed by Microorganisms and Its Application in Integrated Synthesis. J. Mol. Catal. B Enzym. 2016, 133, S352–S359. [Google Scholar] [CrossRef]
- Valentová, K. Biotransformation of Natural Products and Phytochemicals: Metabolites, Their Preparation, and Properties. Int. J. Mol. Sci. 2023, 24, 8030. [Google Scholar] [CrossRef] [PubMed]
- Liu, X.; Cheng, Z. Microbial and Chemical Transformations of Euphorbia Factor L1 and the P-Glycoprotein Inhibitory Activity in Zebrafishes. Nat. Prod. Res. 2023, 37, 871–881. [Google Scholar] [CrossRef] [PubMed]
- Lu, J.; Li, G.; Huang, J.; Zhang, C.; Zhang, L.; Zhang, K.; Li, P.; Lin, R.; Wang, J. Lathyrane-Type Diterpenoids from the Seeds of Euphorbia lathyris. Phytochemistry 2014, 104, 79–88. [Google Scholar] [CrossRef] [PubMed]
- Engel, D.W. The Determination of Absolute Configuration and Δf” Values for Light-Atom Structures. Acta Cryst. B 1972, 28, 1496–1509. [Google Scholar] [CrossRef]
- Engel, D.W.; Zechmeister, K.; Hoppe, W. Experience in the Determination of Absolute Configuration of Light Atom Compounds by X-Ray Anomalous Scattering. Tetrahedron Lett. 1972, 13, 1323–1326. [Google Scholar] [CrossRef]
- Jiao, W.; Mao, Z.; Dong, W.; Deng, M.; Lu, R. Euphorbia Factor L8: A Diterpenoid from the Seeds of Euphorbia lathyris. Acta Cryst. E 2008, 64, o331. [Google Scholar] [CrossRef] [PubMed]
- Li, L.; Huang, J.; Lyu, H.; Guan, F.; Li, P.; Tian, M.; Xu, S.; Zhao, X.; Liu, F.; Paetz, C.; Feng, X.; Chen, Y. Two Lathyrane Diterpenoid Stereoisomers Containing an Unusual trans-gem-Dimethylcyclopropane from the Seeds of Euphorbia lathyris. RSC Adv. 2021, 11, 3183–3189. [Google Scholar] [CrossRef]

| 3 | 4 | 5 | ||||
|---|---|---|---|---|---|---|
| Position | δH, mult (J in Hz) | δC, Type | δH, mult (J in Hz) | δC, Type | δH, mult (J in Hz) | δC, Type |
| 1α | 3.40, dd (14.6, 8.4) | 48.3, CH2 | 3.41, dd (14.5, 8.7) | 48.0, CH2 | 3.40, dd (14.5, 8.7) | 48.0, CH2 |
| 1β | 1.56, dd (14.6, 11.4) | 1.63-1.56, m | 1.60, dd (14.5, 11.5) | |||
| 2 | 2.30–2.19, m | 37.1, CH | 2.33–2.24, m | 37.2, CH | 2.30–2.20, m | 37.2, CH |
| 3 | 5.53, t (3.5) | 80.2, CH | 5.57, t (3.5) | 80.1, CH | 5.57, t (3.5) | 80.1, CH |
| 4 | 2.74, dd (10.4, 3.5) | 52.2, CH | 2.83, dd (10.3, 3.5) | 52.2, CH | 2.83, dd (10.3, 3.5) | 52.3, CH |
| 5 | 6.05, d (10.4) | 65.7, CH | 6.06, d (10.3) | 65.6, CH | 6.07, d (10.3) | 65.5, CH |
| 6 | - | 144.2, C | - | 143.2, C | - | 143.2, C |
| 7α | 2.12–2.04, m | 34.9, CH2 | 2.15–2.09, m | 34.7, CH2 | 2.16-2.10, m | 34.7, CH2 |
| 7β | 2.30–2.19, m | 2.24–2.17, m | 2.30–2.20, m | |||
| 8α | 1.95–1.87, m | 21.2, CH2 | 2.05–1.99, m | 21.6, CH2 | 2.06-1.99, m | 21.2, CH2 |
| 8β | 1.75, m | 1.72, tdd (14.0, 11.9, 2.0) | 1.84-1.71, m | |||
| 9 | 1.25, ddd (12.2, 8.6, 3.9) | 30.8 CH | 1.27, ddd (14.0, 8.8, 4.0) | 36.4, CH | 1.39, ddd (12.1, 8.4, 3.9) | 31.8 CH |
| 10 | - | 30.8, C | - | 26.8, C | - | 32.0, C |
| 11 | 1.53, dd (11.3, 8.6) | 24.7 CH | 1.59, dd (11.6, 8.2) | 28.3, CH | 1.77, dd (11.6, 8.4) | 24.7 CH |
| 12 | 6.46, dd (11.3, 1.3) | 144.9, CH | 6.52, d (11.6) | 150.8, CH | 6.53, d (11.6) | 149.0, CH |
| 13 | - | 135.0, C | - | 135.5, C | - | 136.4, C |
| 14 | - | 196.9 C | - | 198.7, C | - | 198.7 C |
| 15 | - | 92.3, C | - | 92.1, C | - | 92.0, C |
| 16 | 0.88, d (6.7) | 14.1, CH3 | 0.91, d (6.7) | 14.1, CH3 | 0.91, d (6.7) | 14.1, CH3 |
| 17a | 4.98, s | 115.6, CH2 | 5.07, d (1.2) | 117.6, CH2 | 5.09, s | 117.7, CH2 |
| 17b | 4.71, s | 4.84, s | 4.85, s | |||
| 18a | 3.46, d (11.1) | 71.7, CH2 | 1.16, s | 16.8, CH3 | 3.51, d (11.1) | 71.7, CH2 |
| 18b | 3.39, d (11.1) | 3.40, d (11.1) | ||||
| 19 | 1.20, s | 12.5,a CH3 | 1.14, s | 28.9, CH3 | 1.22, s | 12.5, CH3 |
| 20a | 1.67, d (1.2) | 12.4,a CH3 | 4.32, d (12.2) | 58.3, CH2 | 4.31, d (12.4) | 58.2, CH2 |
| 20b | 4.17, dd (12.2, 5.9) | 4.20, d (12.4) | ||||
| 20-OH | - | - | 2.42, brs | - | - | - |
| 3-OCOCH3 | 2.02, s | 20.9, CH3 | 2.03, s | 20.9, CH3 | 2.04,b s | 20.9,c CH3 |
| 3-OCOCH3 | - | 170.7, C | - | 170.7, C | - | 170.6, C |
| 5-OCOCH3 | 1.96, s | 21.2, CH3 | 1.98, s | 21.2, CH3 | 1.99,b s | 21.2,c CH3 |
| 5-OCOCH3 | - | 170.6, C | - | 170.6, C | - | 170.6, C |
| 15-OCOCH3 | 2.08, s | 22.0, CH3 | 2.08, s | 21.9, CH3 | 2.10, s | 22.0, CH3 |
| 15-OCOCH3 | - | 169.8, C | - | 169.8, C | - | 169.8, C |
| 6 | 7 | |||
|---|---|---|---|---|
| Position | δH, mult (J in Hz) | δC, type | δH, mult (J in Hz) | δC, type |
| 1α | 3.08, dd (13.6, 6.7) | 45.6, CH2 | 2.96, dd (14.5, 6.4) | 45.9, CH2 |
| 1β | 1.76, t (13.6) | 2.24-2.17, m | ||
| 2 | 2.18–2.10, m | 37.5, CH | 2.18–2.07, m | 38.2, CH |
| 3 | 5.51, t (4.0) | 78.5, CH | 5.46, t (3.7) | 77.2, CH |
| 4 | 2.77, dd (9.0, 4.0) | 51.6, CH | 2.87, dd (10.2, 3.7) | 51.2, CH |
| 5 | 5.83, d (9,0) | 73.0, CH | 5.77, d (10.2) | 73.2, CH |
| 6 | - | 145.1, C | - | 142.8, C |
| 7α | 1.97–1.84, m | 29.7, CH2 | 1.76–1.67, m | 26.5, CH2 |
| 7β | 2.29, m | 2.09–1.99, m | ||
| 8α | 1.97–1.84, m | 23.0, CH2 | 1.91–1.76, m | 26.2, CH2 |
| 8β | 1.11, m | 1.91–1.76, m | ||
| 9 | 0.62, ddd (11.0, 8.7, 1.9) | 34.8, CH | 2.63, td (10.5, 5.7) | 52.4, CH |
| 10 | - | 20.8, C | - | 146.5, C |
| 11 | 1.50, t (8.7) | 23.9, CH | 5.59, dd (15.8, 10.5) | 138.5, CH |
| 12 | 5.28, dd (8.7, 1.6) | 134.8, CH | 5.94, d (15.8) | 124.5, CH |
| 13 | - | 138.0, C | - | 144.3, C |
| 14 | - | 204.4, C | - | 198.0, C |
| 15 | - | 92.0, C | - | 91.4, C |
| 16 | 0.91, d (6.6) | 13.3, CH3 | 0.91, d (6.4) | 13.4, CH3 |
| 17a | 5.33, s | 115.5, CH2 | 5.31, d (2.7) | 114.5,CH2 |
| 17b | 4.92, s | 4.99, bs | ||
| 18 | 1.03, s | 15.5, CH3 | 1.71, s | 21.1, CH3 |
| 19 | 1.04, s | 28.2, CH3 | 4.72, d (5.9) | 110.2, CH2 |
| 20a | 1.83, s | 22.2, CH3 | 5.29, s | 115.4, CH2 |
| 20b | 5.25, s | |||
| 3-OCOCH3 | 2.04, s | 20.9, CH3 | 2.02, s | 20.7, CH3 |
| 3-OCOCH3 | - | 170.4, C | - | 170.4, C |
| 5-OCOCH3 | 1.98, s | 21.1, CH3 | 1.96, s | 20.9, CH3 |
| 5-OCOCH3 | - | 169.5, C | - | 169.5, C |
| 15-OCOCH3 | 2.14, s | 21.9, CH3 | 2.23, s | 21.7, CH3 |
| 15-OCOCH3 | - | 170.3, C | - | 169.1, C |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).





