Submitted:
25 January 2024
Posted:
25 January 2024
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
2.1. Subjects and Materials
2.2. Study Design
2.3. Outcome Measures
2.4. Statistical Analysis
3. Results
3.1. Baseline Characteristics of the Patients
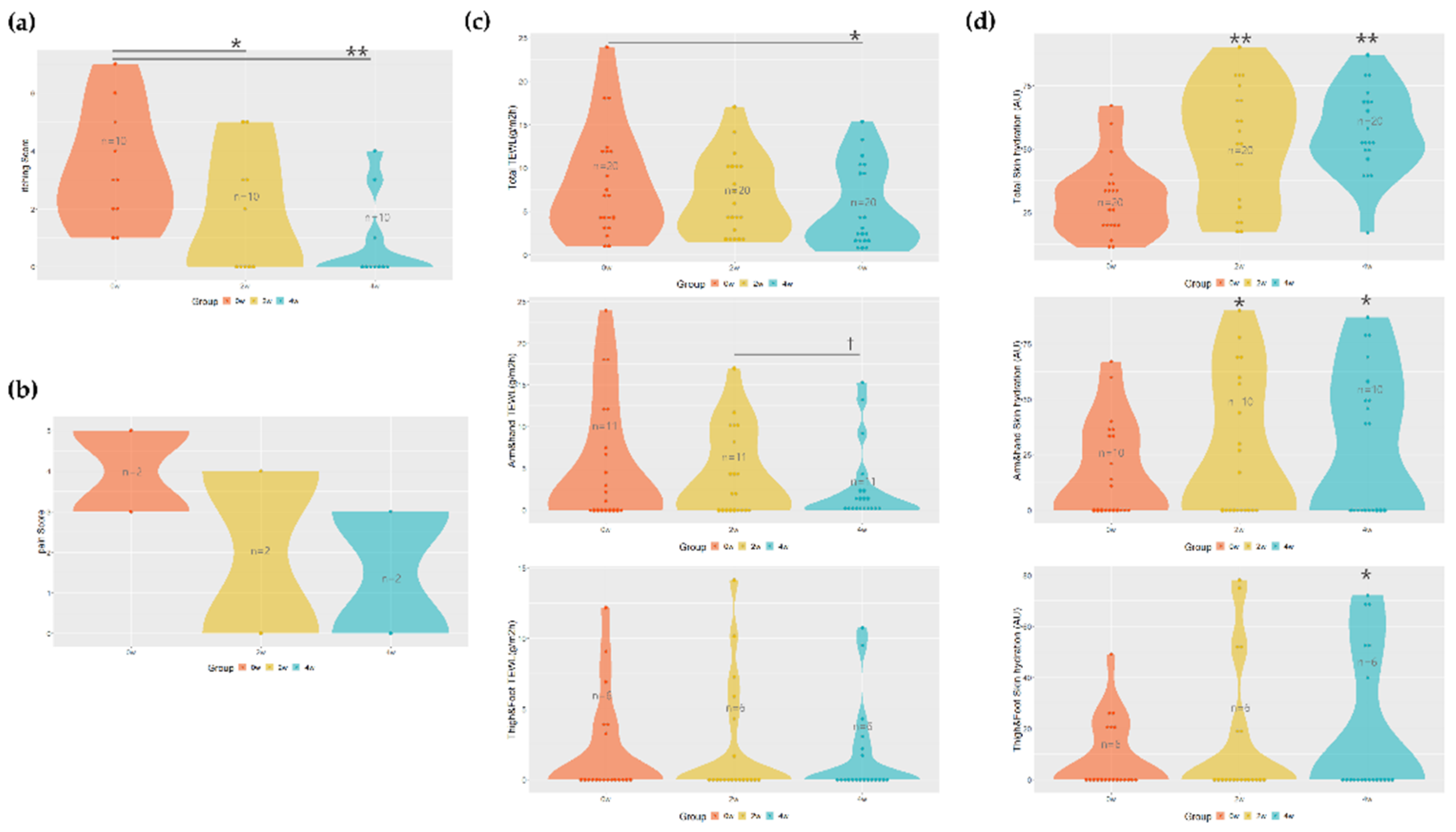
3.2. The Efficacy of the RE:Pair Formula for Relieving Itching and Pain
3.3. Changes in Transepidermal Water Loss (TEWL) and Skin Hydration with the RE:Pair Formula
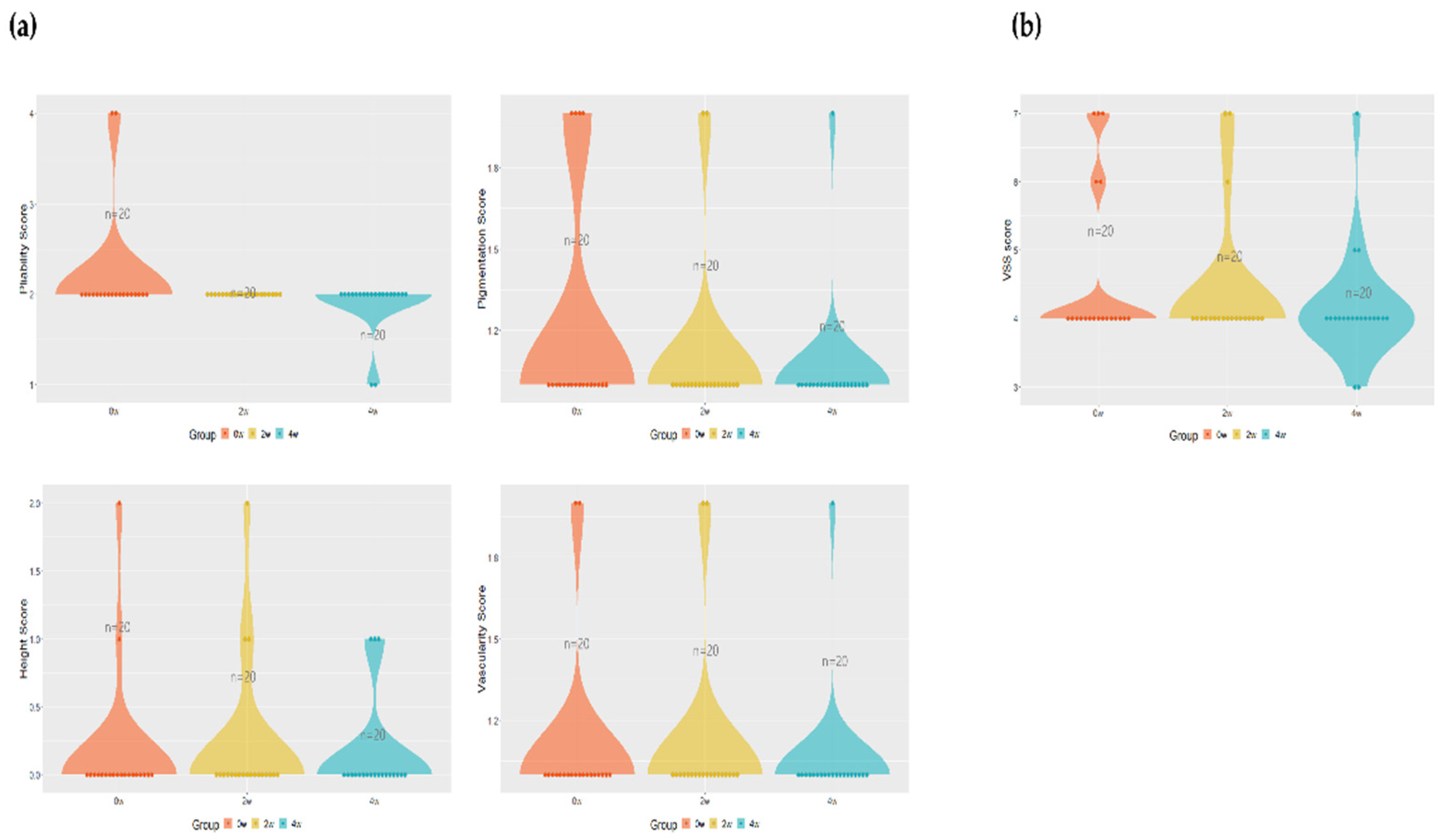
3.4. Comparison of Vancouver Scar Scale (VSS) before and after application
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Barss, P. Injury prevention: an international perspective epidemiology, surveillance, and policy. USA: Oxford University Press; 1998.
- Rossignol, A.M.; Locke, J.; Burke, J. Paediatric burn injuries in New England, USA. Burns. 1990, 16, 41–8. [Google Scholar] [CrossRef]
- Research NRCCoT, Research CoT, Medicine Io. Injury in America: a continuing public health problem: Natl Academy Pr. 1985.
- Smolle, C.; Cambiaso-Daniel, J.; Forbes, A.A.; Wurzer, P.; Hundeshagen, G.; Branski, L.K.; Huss, F.; Kamolz, L.P. Recent trends in burn epidemiology worldwide: A systematic review. Burns. 2017, 43, 249–257. [Google Scholar] [CrossRef]
- Lodha, P.; Shah, B.; Karia, S.; De Sousa, A. Post-Traumatic Stress Disorder (Ptsd) Following Burn Injuries: A Comprehensive Clinical Review. Ann Burns Fire Disasters. 2020, 33, 276–287. [Google Scholar]
- Van Loey, N.E.; Bremer, M.; Faber, A.W.; Middelkoop, E.; Nieuwenhuis, M.K. Itching following burns: Epidemiology and predictors. Br. J. Dermatol. 2008, 158, 95–100. [Google Scholar] [CrossRef] [PubMed]
- Stumpf, A.; Sta¨nder, S. Neuropathic itch: diagnosis and management. Dermatol Ther. 2013, 26, 104–9. [Google Scholar] [CrossRef] [PubMed]
- Vitale, M.; Fields-Blache, C.; Luterman, A. Severe itching in the patient with burns. J Burn Care Rehabil. 1991, 12, 330–333. [Google Scholar] [CrossRef] [PubMed]
- Chung, B.Y.; Kim, H.B.; Jung, M.J.; Kang, S.Y.; Kwak, I.S.; Park, C.W.; Kim, H.O. Post-Burn Pruritus. Int J Mol Sci. 2020, 21, 3880. [Google Scholar] [CrossRef] [PubMed]
- Jaffe, S.E.; Patterson, D.R. Treating sleep problems in patients with burn injuries: practical considerations. J Burn Care Rehabil. 2004, 25, 294–305. [Google Scholar] [CrossRef] [PubMed]
- Bell, L.; McAdams, T.; Morgan, R.; Parshley, P.F.; Pike, R.C.; Riggs, P.; Carpenter, J.E. Pruritus in burns: a descriptive study. J Burn Care Rehabil. 1988, 9, 305–8. [Google Scholar] [PubMed]
- Willebrand, M.; Low, A.; Dyster-Aas, J.; Kildal, M.; Andersson, G.; Ekselius, L.; Gerdin, B. Pruritus, personality traits and coping in long-term follow-up of burn-injured patients. Acta Derm. Venereol. 2004, 84, 375–380. [Google Scholar] [CrossRef] [PubMed]
- Gorski, J.; Proksch, E.; Baron, J.M.; Schmid, D.; Zhang, L. Dexpanthenol in Wound Healing after Medical and Cosmetic Interventions (Postprocedure Wound Healing). Pharmaceuticals. 2020, 13, 138. [Google Scholar] [CrossRef]
- Proksch, E.; Nissen, H.P. Dexpanthenol enhances skin barrier repair and reduces inflammation after sodium lauryl sulphate-induced irritation. J Dermatol Treat. 2002, 13, 173–178. [Google Scholar] [CrossRef] [PubMed]
- Maenthaisong, R.; Chaiyakunapruk, N.; Niruntraporn, S.; Kongkaew, C. The efficacy of aloe vera used for burn wound healing: A systematic review. Burns. 2007, 33, 713–718. [Google Scholar] [CrossRef]
- Bunyapraphatsara, N.; Jirakulchaiwong, S.; Thirawarapan, S.; Manonukul, J. The efficacy of Aloe vera cream in the treatment of first, second and third degree burns in mice. Phytomedicine. 1996, 2, 247–251. [Google Scholar] [CrossRef]
- Hekmatpou, D.; Mehrabi, F.; Rahzani, K.; Aminiyan, A. The Effect of Aloe Vera Clinical Trials on Prevention and Healing of Skin Wound: A Systematic Review. Iran J Med Sci. 2019, 44, 1–9. [Google Scholar] [CrossRef]
- Strashilov, S.; Slavchev, S.; Aljowder, A.; Vasileva, P.; Postelnicu-Gherasim, S.; Kostov, S.; Yordanov, A. Austrian natural ointment (Theresienöl®) with a high potential in wound healing—A European review. Wound Med. 2020, 30, 100191. [Google Scholar] [CrossRef]
- Kwon, K.C.; Won, J.G.; Seo, J.H.; Kwon, O.S.; Kim, E.H.; Kim, M.S.; Park, S.W. Effects of arginine glutamate (RE:pair) on wound healing and skin elasticity improvement after CO2 laser irradiation. J Cosmet Dermatol. 2022, 21, 5037–5048. [Google Scholar] [CrossRef]
- Karna, E.; Miltyk, W.; Wołczyński, S.; Pałka, J.A. The potential mechanism for glutamine-induced collagen biosynthesis in cultured human skin fibroblasts. Comp Biochem Physiol B Biochem Mol Biol. 2001, 130, 23–32. [Google Scholar] [CrossRef]
- Candi, E.; Knight, R.A.; Panatta, E.; Smirnov, A.; Melino, G. Cornification of the skin: A non-apoptotic cell death mechanism. In eLS; John Wiley & Sons, Ltd.: Chichester, UK, 2016. [Google Scholar]
- Markiewicz-Gospodarek, A.; Kozioł, M.; Tobiasz, M.; Baj, J.; Radzikowska-Büchner, E.; Przekora, A. Burn Wound Healing: Clinical Complications, Medical Care, Treatment, and Dressing Types: The Current State of Knowledge for Clinical Practice. Int J Environ Res Public Health. 2022, 19(3), 1338. [Google Scholar] [CrossRef]
- Singer, A.J.; Boyce, S.T. Burn Wound Healing and Tissue Engineering. J Burn Care Res. 2017, 38, e605–e613. [Google Scholar] [CrossRef]
- Akdeniz, M.; Gabriel, S.; Lichterfeld-Kottner, A.; Blume-Peytavi, U.; Kottner, J. Transepidermal water loss in healthy adults: a systematic review and meta-analysis update. Br J Dermatol. 2018, 179, 1049–1055. [Google Scholar] [CrossRef]
- Park, C.S. The Skin Barrier and Moisturizer. J Skin Barrier Res. 2007, 9, 11–17. [Google Scholar]
- Klotz, T.; Kurmis, R.; Munn, Z.; Heath, K.; Greenwood, J. Moisturisers in scar management following burn: A survey report. Burns. 2017, 43, 965–972. [Google Scholar] [CrossRef] [PubMed]
- Matheson, J.D.; Clayton, J.; Muller, M.J. The reduction of itch during burn wound healing. J Burn Care Rehabil. 2001, 22, 76–81. [Google Scholar] [CrossRef] [PubMed]
- Zachariah, J.R.; Rao, A.L.; Prabha, R.; Gupta, A.K.; Paul, M.K.; Lamba, S. Post burn pruritus - a review of current treatment options. Burns. 2012, 38, 621–9. [Google Scholar] [CrossRef] [PubMed]
- Fowler, E.; Yosipovitch, G. Post-Burn Pruritus and Its Management—Current and New Avenues for Treatment. Curr Trauma Rep. 2019, 5, 90–98. [Google Scholar] [CrossRef]
- Ahuja, R.B.; Gupta, G.K. A four-arm, double-blind, randomized and placebo- controlled study of pregabalin in the management of post-burn pruritus. Burns. 2013, 39, 24–29. [Google Scholar] [CrossRef]
- Klotz, T.; Kurmis, R.; Munn, Z.; Heath, K.; Greenwood, J.E. The effectiveness of moisturizers in the management of burn scars following burn injury: a systematic review. JBI Database System Rev Implement Rep. 2015, 13, 291–315. [Google Scholar] [CrossRef] [PubMed]
- Al-Koussa, H.; El Mais, N.; Maalouf, H.; Abi-Habib, R.; El-Sibai, M. Arginine deprivation: a potential therapeutic for cancer cell metastasis? A review. Cancer Cell Int. 2020, 20, 150. [Google Scholar] [CrossRef]
- Deliconstantinos, G.; Villiotou, V.; Stavrides, J.C. Inhibition of ultraviolet B-induced skin erythema by N-nitro-L-arginine and N-monomethyl-L-arginine. J Dermatol Sci. 1997, 15, 23–35. [Google Scholar] [CrossRef]
- Yan, H.; Peng, X.; Wang, P.; Huang, Y.S.; Wang, S.L. [Effects of different doses of L-arginine on the serum levels of helper T lymphocyte 1 (Th1)/Th2 cytokines in severely burned patients]. Zhonghua Shao Shang Za Zhi. 2009, 25, 331–334. [Google Scholar] [PubMed]
- Nieweld, C.; Summer, R. Activated Fibroblasts: Gluttonous for Glutamine. Am J Respir Cell Mol Biol. 2019, 61, 554–555. [Google Scholar] [CrossRef] [PubMed]
- Hamanaka, R.B.; O'Leary, E.M.; Witt, L.J.; Tian, Y.; Gökalp, G.A.; Meliton, A.Y.; Dulin, N.O.; Mutlu, G.M. Glutamine Metabolism Is Required for Collagen Protein Synthesis in Lung Fibroblasts. Am J Respir Cell Mol Biol. 2019, 61, 597–606. [Google Scholar] [CrossRef] [PubMed]
- Berger, M.M.; Binz, P.A.; Roux, C.; Charrière, M.; Scaletta, C.; Raffoul, W.; Applegate, L.A.; Pantet, O. Exudative glutamine losses contribute to high needs after burn injury. J Parenter Enteral Nutr. 2022, 46, 782–788. [Google Scholar] [CrossRef]
| Variables | Total (n=20) | |
| Age, Mean±SD | 48.35 ± 13.13 | |
| Sex, n (%) | Male | 15 (75 %) |
| Female | 5 (25 %) | |
| Application site | Arm + hand | 10 (50 %) |
| Thigh + foot | 6 (30 %) | |
| thrunk | 2 (10 %) | |
| elbow | 1 (5 %) | |
| face | 1 (5 %) | |
| Baseline transepidermal water loss (TEWL), mean ± standard deviation (SD) (normal) |
4.766 ± 3.27 | |
| Baseline TEWL, mean ± SD (Lesional) | 8.276 ± 6.15 | |
| Baseline Skin hydration, mean ± SD (normal) | 35.85 ± 14.43 | |
| Baseline Skin hydration, mean ± SD (Lesional) | 30.7 ± 14.69 | |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




