Submitted:
23 January 2024
Posted:
24 January 2024
You are already at the latest version
Abstract
Keywords:
1. Introduction
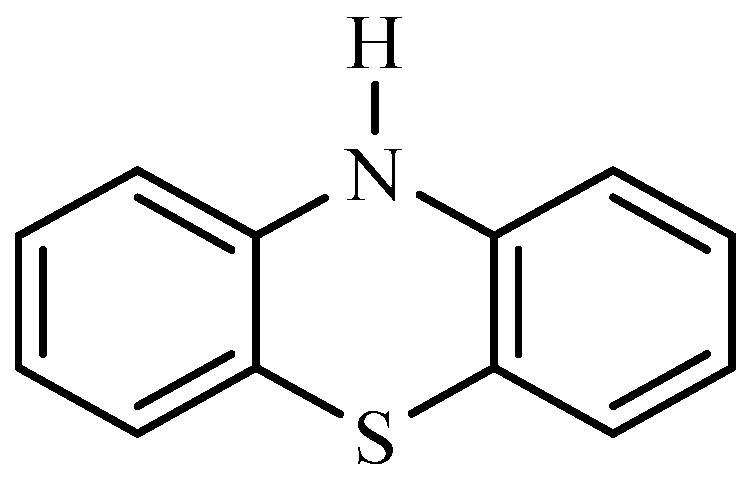
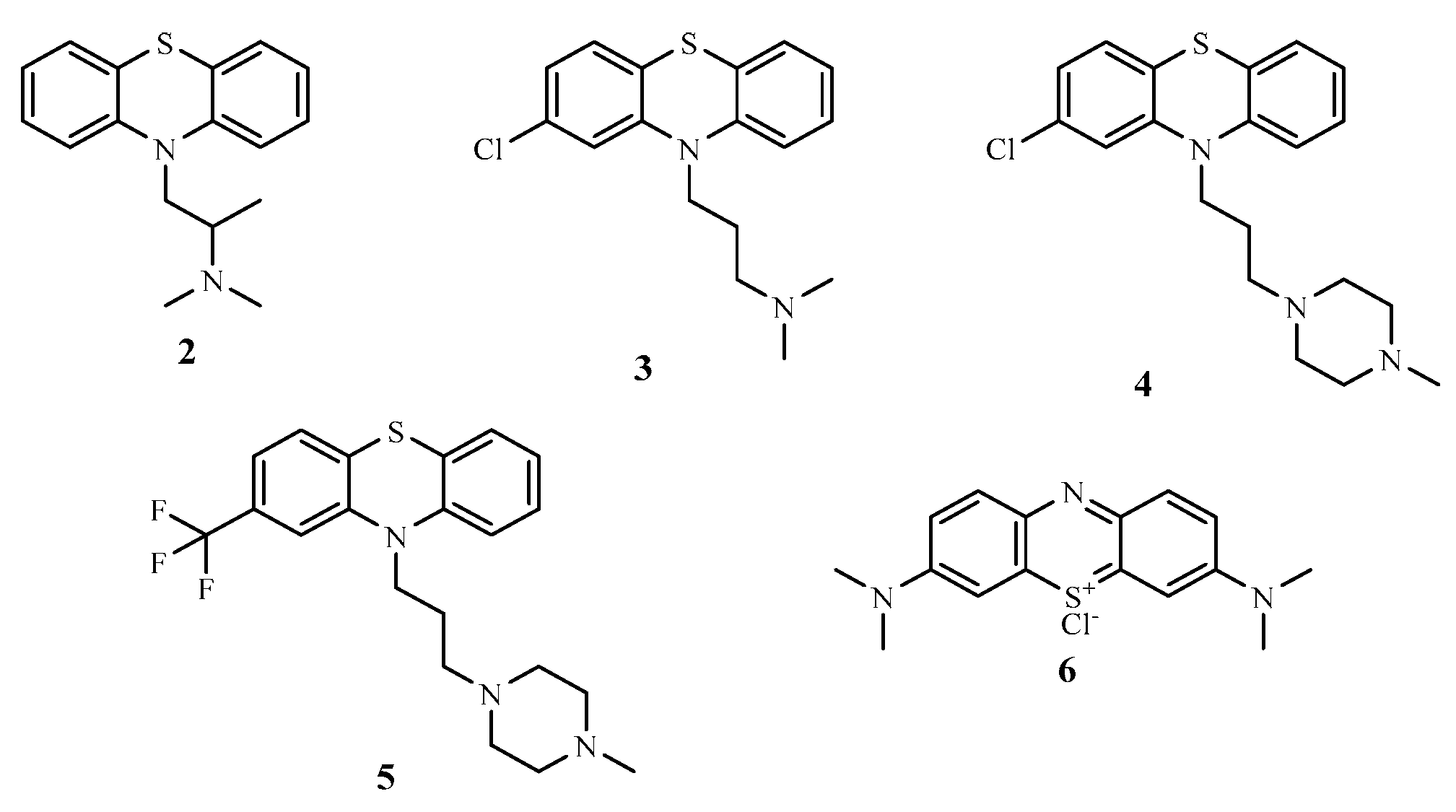
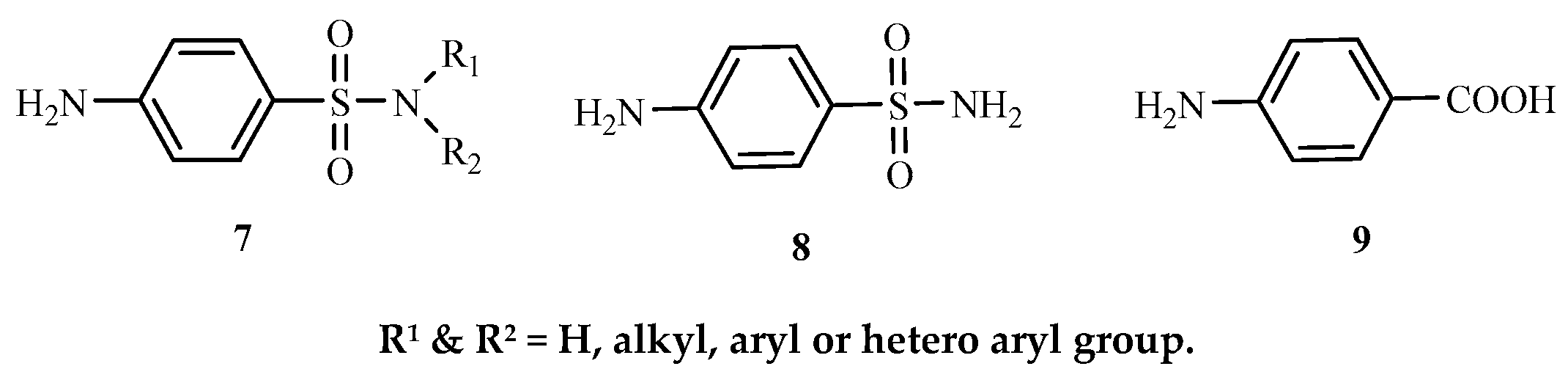
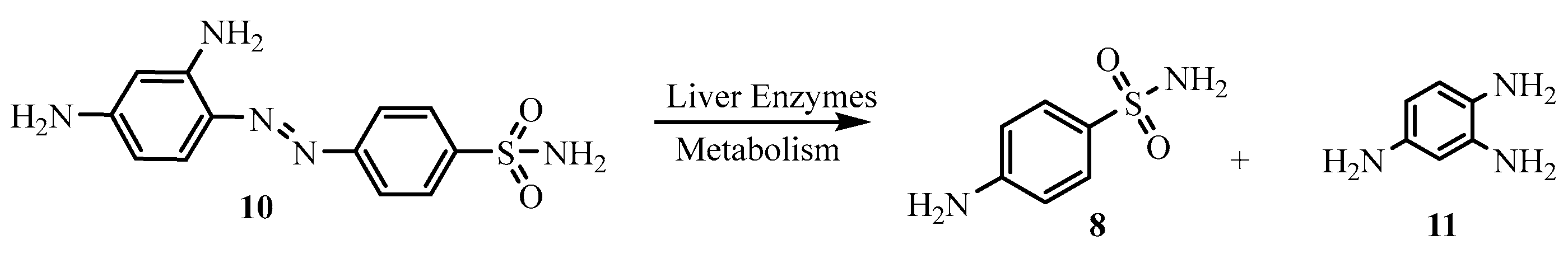
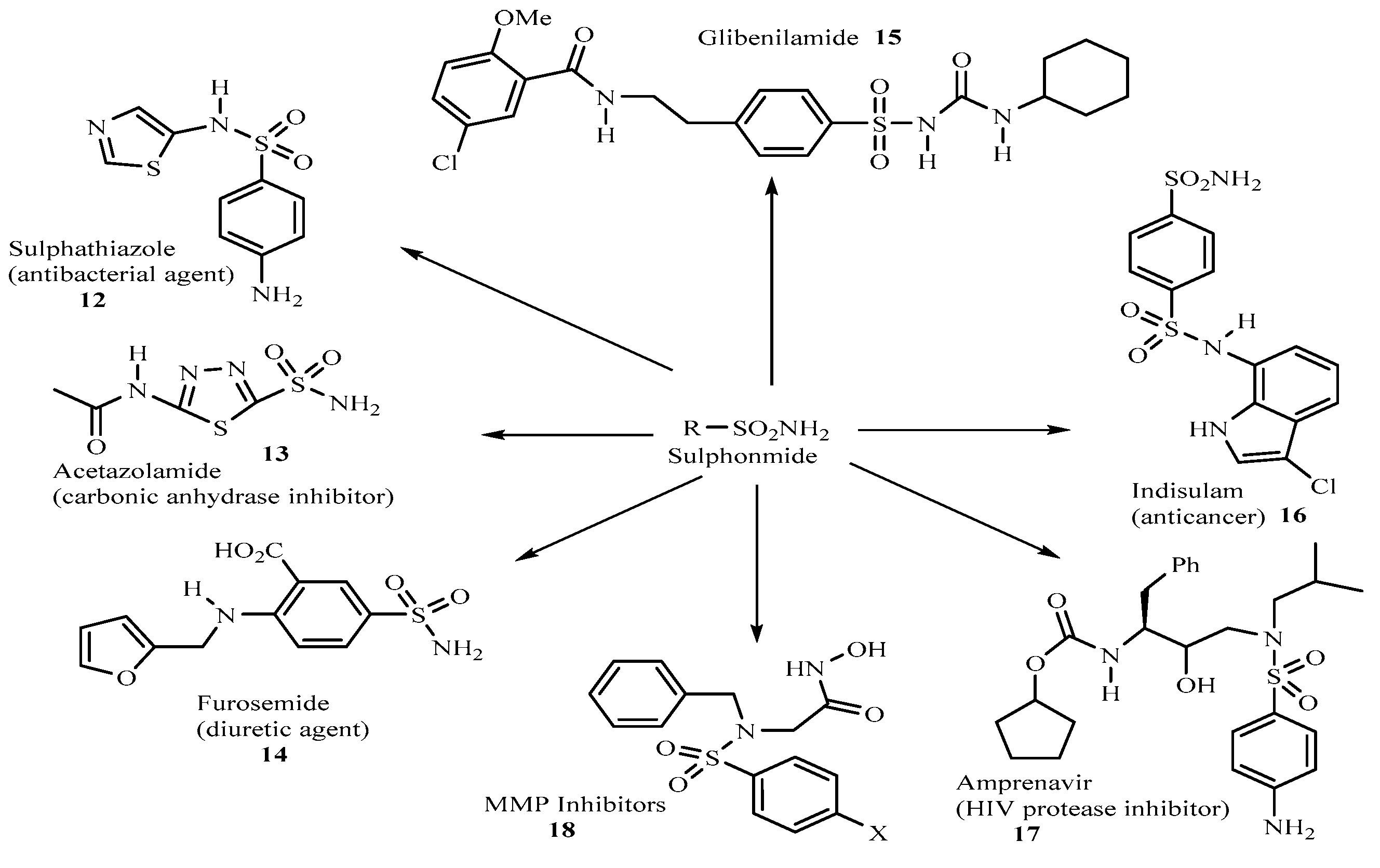
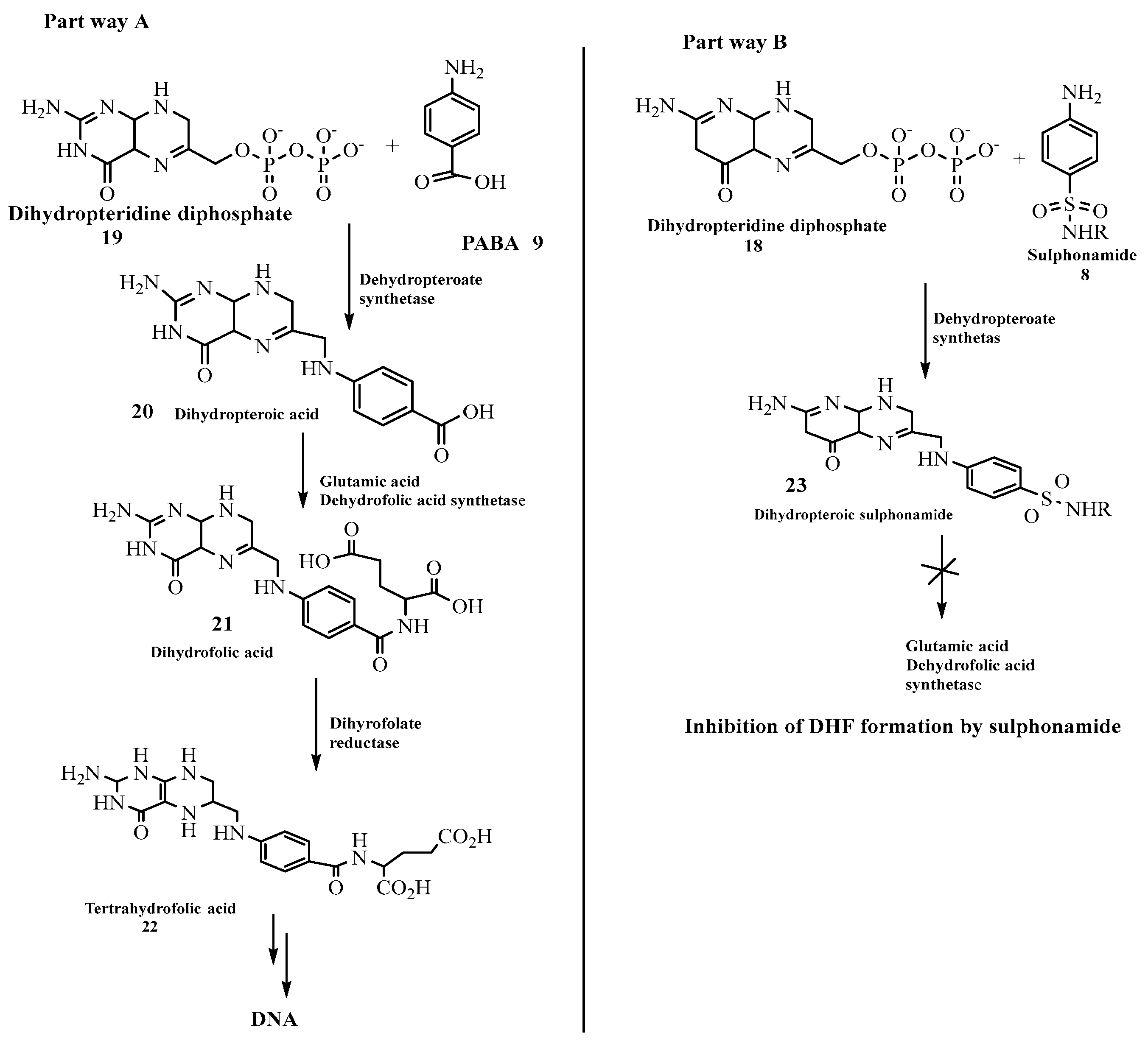
2. Results and Discussions
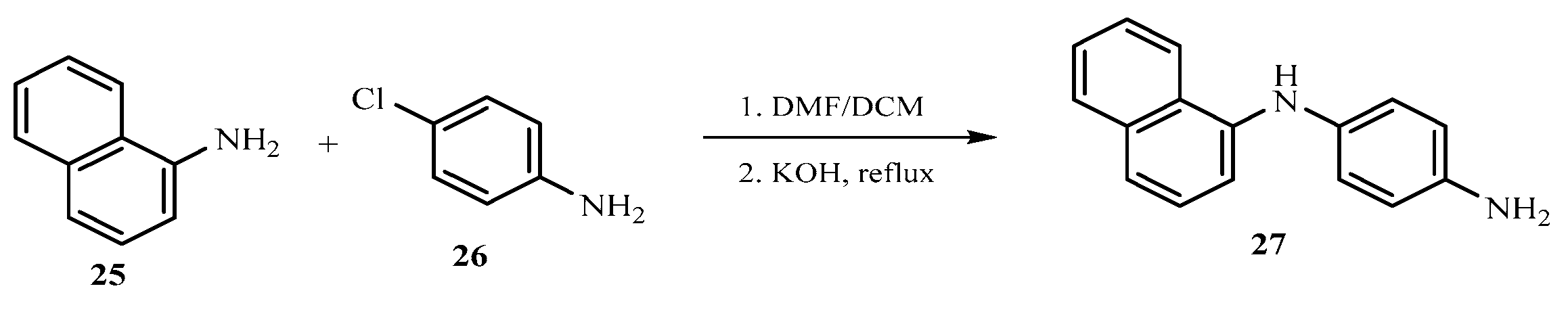
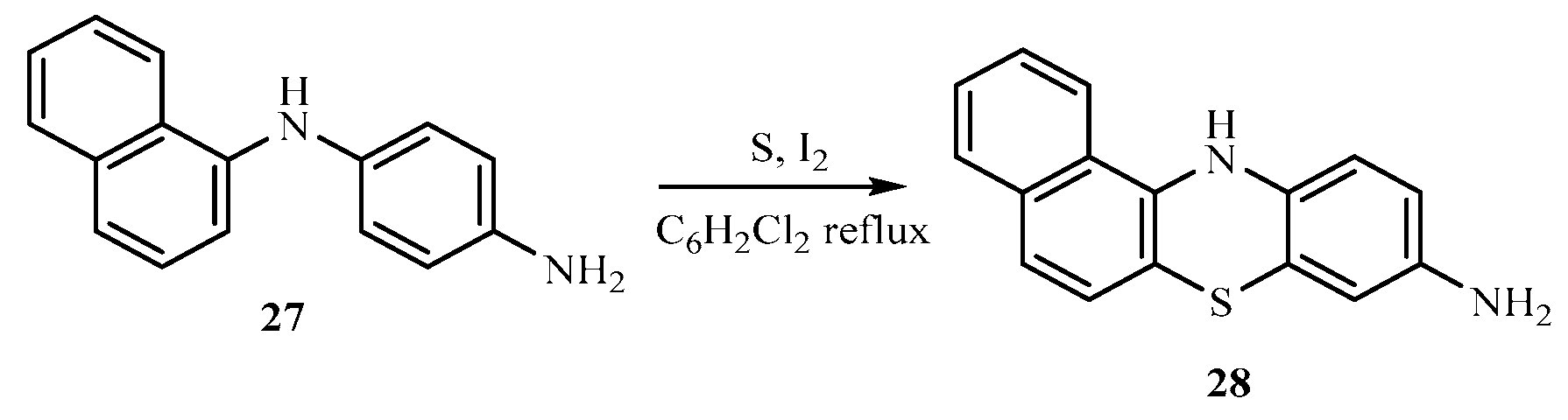
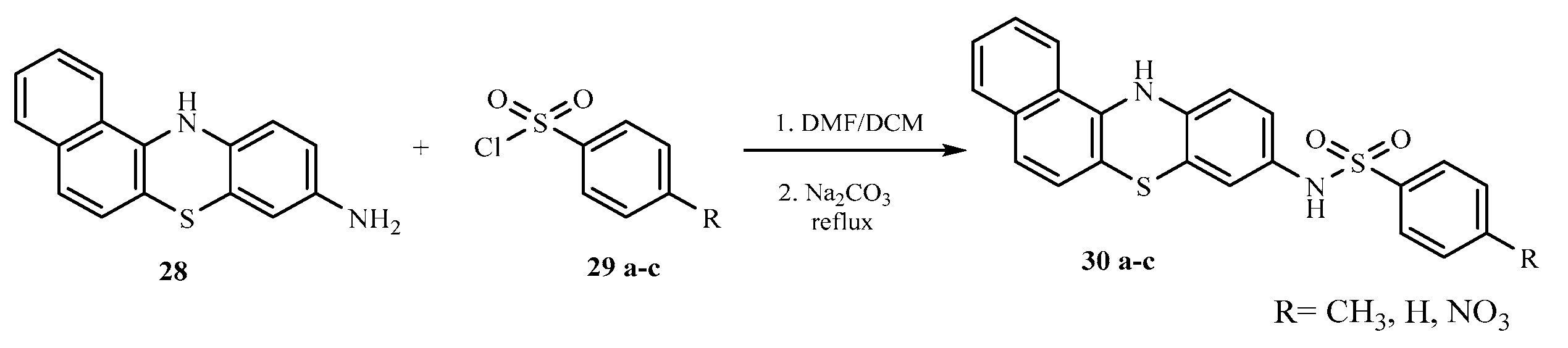
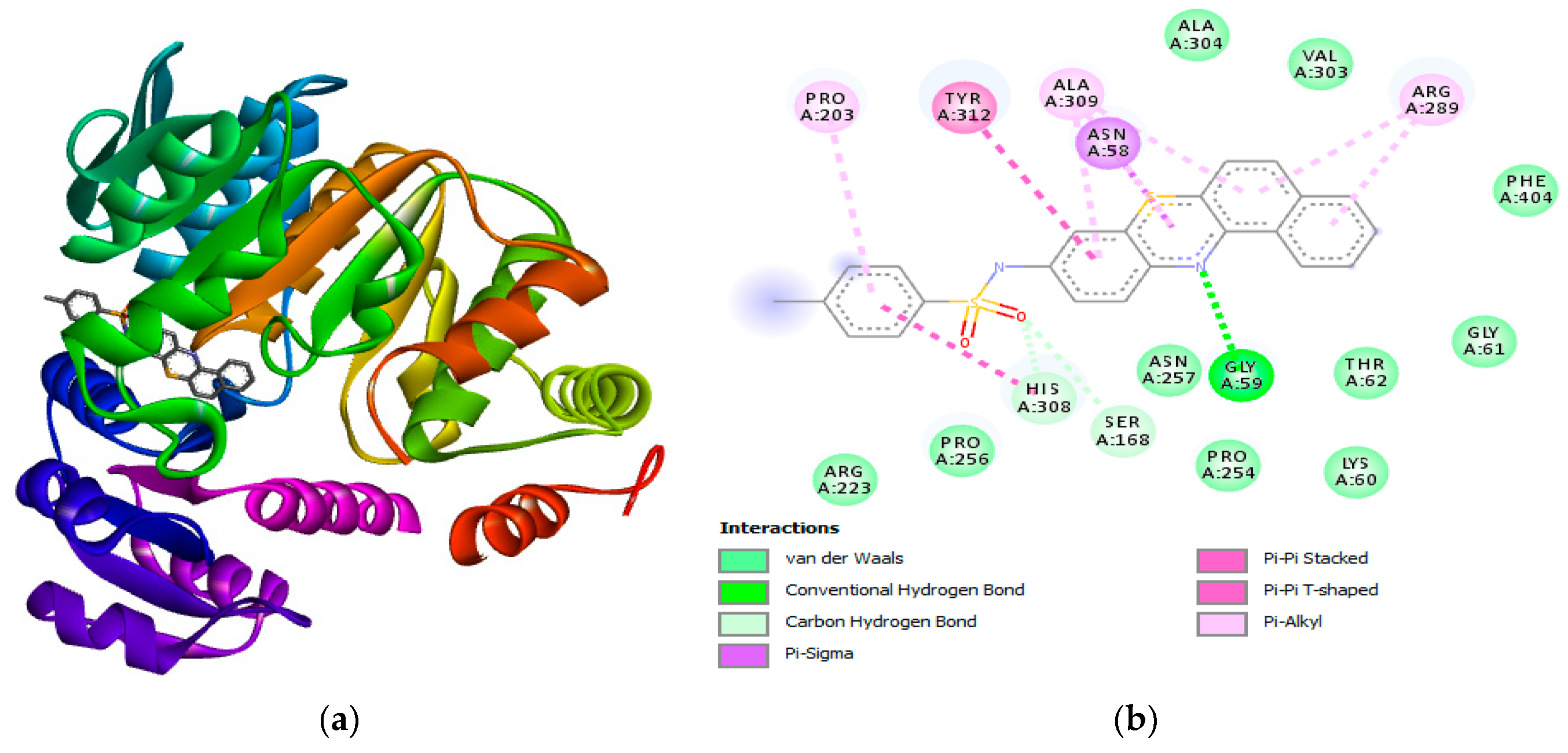
2.1. In silico antimicrobial studies
2.2. Comparative estimation of the physiochemical properties of synthesized compounds
| Compounds | MW* | miLogP | LogS# | TPSA@ | HBA | HBD | Nviolations | Nrotb |
|---|---|---|---|---|---|---|---|---|
| 28 | 264.07 | 4.22 | -4.63 | 41.81 | 1 | 3 | 0 | 0 |
| 30a | 418.08 | 6.36 | -7.11 | 61.96 | 3 | 2 | 1 | 3 |
| 30b | 404.07 | 5.73 | -7.18 | 61.96 | 3 | 2 | 1 | 3 |
| 30c | 449.05 | 5.91 | -5.20 | 107.78 | 5 | 2 | 1 | 4 |
| Acceptable threshold | ˂500 Da | ˂5 | 0 – (-6) | ≤140 A2 | ≤10 | ≤5 | 0 | 9 |
2.3. In vitro studies
3. Material and Methods
3.1. Synthesis of 1-(naphthalen-1-yl) benzene-1,4-diamine (27)
3.2. Synthesis of 12H-benzo[a]phenothiazin-9-amine (28)
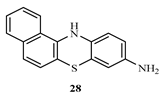
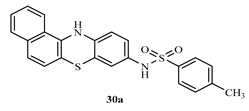
3.3. General method for the synthesis of phenothiazine-3-sulphonamides derivatives (30a-c)
N-(12H-benzo[a]phenothiazine-9-yl)-4-methylbenzenesulphonamide (30a)
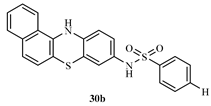
N-(12H-benzo[a]phenothiazine-9-yl)benzenesulphonamide (30b)
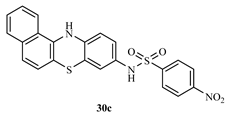
N-(12H-benzo[a]phenothiazine-9-yl)-4-nitrobenzenesulphonamide (30c)
3.4. Biological Studies
3.4.1. Preparation of selected derivatives concentrations
3.4.2. Nutrient agar preparation
3.4.3. The test microorganisms used:
3.4.4. Control test (standard):
3.4.5. Determination of antimicrobial activity
3.4.6. Preparation of nutrient agar for fungal strain
3.4.7. Preparation of Mueller-hinton agar for bacterial strains
3.6.8. Determination of Minimum Inhibition Concentration (MIC)
Molecular docking studies
4. Conclusion
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Efeturi, O.; Egu, A.; Ezeokonkwo, M.A.; Okoro, U.C. Highlights of molecular structures and applications of phenothiazine & phenoxazine polycycles. J. Mol. Struct. 2019, 1175, 956–962. [Google Scholar] [CrossRef]
- Sochacka, J.; Pacholczyk, M.; Jeleń, M.; Morak-Mloclawska, B.; Pluta, K. Interaction of new tri-, tetra-, and pentacyclic azaphenothiazine derivatives with calf thymus DNA: Spectroscopic and molecular docking studies. Spectrochim. Acta Part A: Mol. Bimolecular Spectrosc. 2021, 262, 120105. [Google Scholar] [CrossRef]
- Kidron, A.; Nguyen, H. Phenothiazine. Natl. Libr. Med. 2023. https://www.ncbi.nlm.nih.gov/books/NBK556113.
- Kenia, H.; Shivkumar, B.; Kotnal, R.B.; Ramesha, A.; Devadiga, P.; Simpi, C.C.; Chandrashekar, V.M. Synthesis and evaluation of phenothiazine derivatives. IOSR J. Pharm. 2020, 10, 54–62. [Google Scholar]
- Varga, B.; Csonka, A.; Csonka, A.; Molnár, J.; Amaral, L.; Spengler, G. Possible Biological and Clinical Applications of Phenothiazines. Anticancer. Res. 2017, 37, 5983–5993. [Google Scholar] [CrossRef]
- Gopi, C.; Dhanaraju, M.D. Recent progress in synthesis, structure and biological activities of phenothiazine derivatives, Rev. J. Chem. 2019, 9, 95–126. [Google Scholar] [CrossRef]
- Piotti, P.; Uccheddu, S.; Alliani, M.; Mariti, C.; Nuti, V.; Ogi, A.; Pierantoni, L.; Gazzano, A. Management of specific fears and anxiety in the behavioral medicine of companion animals: punctual use of psychoactive medications. Dog Behav. 2019, 5, 23–30. [Google Scholar] [CrossRef]
- Cunningham Owens, D.; Johnstone, E.C. The development of antipsychotic drugs. Brain Neurosci. Adv. 2018, 2, 1–6. [Google Scholar] [CrossRef] [PubMed]
- Seeman, M.V. History of the dopamine hypothesis of antipsychotic action. World J. Psychiatry 2021, 11, 355–364. [Google Scholar] [CrossRef] [PubMed]
- Chatterjee, S., and Chatterjee, R.P. Insights into the neuro-pharmacological treatment of schizophrenia: Past, present, and future. In cognizance of schizophrenia: A profound insight into the psyche. Springer Nature Singapore 2023, 113-125. [CrossRef]
- Howland, R.H. Methylene blue: The long and winding road from stain to brain: Part 2. J. Psychosoc. Nurs. Ment. Health Serv. 2016, 54, 21–26. [Google Scholar] [CrossRef] [PubMed]
- Posso, M.C. , Domingues, F.C., Ferreira, S., and Silvestre, S. Development of phenothiazine hybrids with potential medicinal interest: a review. Molecules 2022, 27, 276. [Google Scholar] [CrossRef] [PubMed]
- Lavanya, R. Sulphonamides: a pharmaceutical review. Int. J. Pharm. Sci. Invent. 2017, 6, 1–3. [Google Scholar]
- Gulçin, İ.; Taslimi, P. Sulfonamide inhibitors: a patent review 2013-present. Expert Opin. Ther. Pat. 2018, 28, 541–549. [Google Scholar] [CrossRef]
- Hutchings, M.I.; Truman, A.W.; Wilkinson, B. Antibiotics: past, present and future. Curr. Opin. Microbiol. 2019, 50, 72–80. [Google Scholar] [CrossRef] [PubMed]
- Al-Mudhafar, M.M.J.; Abdulkadir, M.Q.; Mohammed, A.A.; Al-hilli, F.A.; Mohammed hussian, A. . Synthesis and microbiological study of new sulphonamides. Int. J. Compr. Pharm. 2012, 3, 10–13. [Google Scholar]
- Büyükkıdan, N.; Bülbül, M.; Kasımoğulları, R.; Büyükkıdan, B. Synthesis and characterization of metal complexes of heterocyclic sulphonamide as carbonic anhydrase inhibitors. J. Enzym. Inhib. Med. Chem. 2013, 28, 311–315. [Google Scholar] [CrossRef] [PubMed]
- Krishna Kartheek Chinchilli, Andrea Angeli, Pavitra, S. Thacker, Laxman Naik Korra, Rashmita Biswas, Mohammed Arifuddin & Claudiu T. Supuran. Design, synthesis and biological evaluation of sulphonamide conjugates as potent carbonic. Metabolites 2020 10, 1–13. [CrossRef]
- El-Gaby, M.; Ammar, Y.A.; El-Qaliei, M.I.H.; Ali, A.M.; Farouk, M.; Abdelraheem, F. Sulfonamides: Synthesis and the recent applications in medicinal chemistry. Egptian J. Chem. 2020, 62, 5289–5327. [Google Scholar] [CrossRef]
- Lin Bei & Mok James. The Synthesis of functionalized sulphonamides. Dissertation Publishing UMI U591637, Microform Edition 2013, ProQuest LLC 789 East Eisenhower Parkway P.O. Box 1346, Ann Arbor, Ml 48106-1346, USA.
- Awad, S.H.; Sahib, S.A.; Hussein, F.A.; Al-Khfaji, H.A.H. Synthesis, characterization and study biological activity of new para-methoxy benzene sulfonamide derivatives and some amino acid. Mater. Sci. Eng. 2019, 571, 1–10. [Google Scholar] [CrossRef]
- Ghorab, M.M.; Soliman, A.M.; Alsaid, M.S.; Askar, A.A. Synthesis, antimicrobial activity and docking study of some novel 4-(4,4-dimethyl-2,6- dioxocyclohexylidene)methylamino derivatives carrying biologically active sulphonamide moiety. Arab. J. Chem. 2020, 13, 545–556. [Google Scholar] [CrossRef]
- Albalawi, M.A. Evaluation of antibacterial and antifungal effects of novel hydroxamic acids linked-natural amino acids. Egypt. J. Chem. 2017, 60, 613–618. [Google Scholar] [CrossRef]
- Tačić, A.; Nikolić, V.; Nikolić, L.; Savić, I. Antimicrobial sulphonamide drugs. Adv. Technol. 2017, 6, 58–71. [Google Scholar] [CrossRef]
- Ovung, A.; Bhattacharyya, J. Sulphonamide drugs: structure, antibacterial property, toxicity, and biophysical interactions. Biophys. Rev. 2021, 13, 259–272. [Google Scholar] [CrossRef] [PubMed]
- Masters, P.A.; O’Bryan, T.A.; Zurlo, J.; Miller, D.Q.; Joshi, N. Trimethoprim-sulphamethoxazole revisited. Arch International Medicine 2003, 163, 402–410. [Google Scholar] [CrossRef] [PubMed]
- Variya, H.H.; Panchal, V.; Patel, G.R. Synthesis, anti-tuberculosis and anti-bacterial activities of sulphonamide bearing 4-((2-(5-bromo-1H-pyrazolo-[3,4-b]pyridin-1-yl)-2-oxoethyl)amino)-N-(various substitutions)benzenesulphonamide, Indian J. Chem. 2020, 59, 682–689. [Google Scholar]
- Liu, J.; Seibold, S.A.; Rieke, C.J.; Song, I.; Cukier, R.I.; Smith, W.L. Prostaglandin endoperoxide H synthases: peroxidase hydroperoxide specificity and cyclooxygenase activation. J. Biol. Chem. 2007, 282, 18233–18244. [Google Scholar] [CrossRef]
- Rehman, H.; Qadir, A.; Ali, Z.; Nazir, S.; Zahra, A.; Shahzady, T.G. Synthesis and characterization of novel sulphonamides derivatives and their antimicrobial, antioxidant and cytotoxicity evaluation. Bull. Chem. Soc. Ethiop. 2017, 31, 491–498. [Google Scholar] [CrossRef]
- Akocak, S.; Lolak, N.; Bua, S.; Supuran, C.T. Discovery of novel 1,3-diaryltriazene sulphonamides as carbonic anhydrase I, II, VII, and IX inhibitors. J. Enzym. Inhib. Med. Chem. 2018, 33, 1575–1580. [Google Scholar] [CrossRef]
- Hussein, E.M.; Al-Rooqi, M.M.; Elkhawaga, A.A.; Ahmed, S.A. Tailoring of novel biologically active molecules based on N4-substituted sulphonamides bearing thiazole moiety exhibiting unique multi-addressable biological potentials. Arab. J. Chem. 2020, 13, 5345–5362. [Google Scholar] [CrossRef]
- Hasti. Kenia, B. Shivkumar, Ramaling. B. Kotnal, Ramesha. A. Ramakrishna, Pradeep. Devadiga, C. C. Simpi, Chandrashekar. V. M. Synthesis and evaluation of phenothiazine derivatives. IOSR J. Pharm. 2020, 10, 54–62. www.iosrphr.org.
- Eugene, L. Ayuk, Uchechukwu C. Okoro, Collins U. Ibeji, Comfort M. Nkambule. Synthesis, in silico studies and investigations on antimicrobial, antimalarial activities of p-toluenesulphonamoyl ‘Leu-Gly’ dipeptide carboxamide derivatives. J. Mol. Struct. 2023, 1285, 1–10. [Google Scholar]
- Lipinski, C.A. Lead-and drug-like compounds: the rule-of-five revolution. Drug Discovery Today. Technologies 2004, 1, 337–341. [Google Scholar]
- Lipinski, C.A.; Lombardo, F.; Dominy, B.W.; Feeney, P.J. Experimental and computational approaches to estimate solubility and permeability in drug discovery and development settings. Adv. Drug Deliv. Rev. 1997, 23, 3–25. [Google Scholar] [CrossRef]
- Omran, Z.; Rauch, C. Acid-mediated Lipinski’s second rule: application to drug design and targeting in cancer. Eur. Biophys. J. 2014, 43, 199–206. [Google Scholar] [CrossRef] [PubMed]
- Veber, D.F.; Johnson, S.R.; Cheng, H.-Y.; Smith, B.R.; Ward, K.W.; Kopple, K.D. Molecular properties that influence the oral bioavailability of drug candidates. J. Med. Chem. 2002, 45, 2615–2623. [Google Scholar] [CrossRef]
- Chapman, K.L.; Holzgrefe, H.; Black, L.E.; Brown, M.; Chellman, G.; Copeman, C.; Hoberman, A. Pharmaceutical toxicology: designing studies to reduce animal use, while maximizing human translation. Regul. Toxicol. Pharmacol. 2013, 66, 88–103. [Google Scholar] [CrossRef]
- Bennion, B.J.; Be, N.A.; McNerney, M.W.; Lao, V.; Carlson, E.M.; Valdez, C.A.; Lightstone, F.C. Predicting a drug’s membrane permeability: A computational model validated with in vitro permeability assay data. J. Phys. Chem. B 2017, 121, 5228–5237. [Google Scholar] [CrossRef]
- Ertl, P.; Rohde, B.; Selzer, P. Fast calculation of molecular polar surface area as a sum of fragment-based contributions and its application to the prediction of drug transport properties. J. Med. Chem. 2000, 43, 3714–3717. [Google Scholar] [CrossRef]
- Prasanna, S.; Doerksen, R. Topological polar surface area: a useful descriptor in 2D-QSAR. Curr. Med. Chem. 2009, 16, 21–41. [Google Scholar] [CrossRef] [PubMed]
- Daga, P.R.; Bolger, M.B.; Haworth, I.S.; Clark, R.D.; Martin, E.J. Physiologically based pharmacokinetic modeling in lead optimization. 2. Rational bioavailability design by global sensitivity analysis to identify properties affecting bioavailability. Mol. Pharm. 2018, 15, 831–839. [Google Scholar] [CrossRef] [PubMed]
- Doak, B.C.; Over, B.; Giordanetto, F.; Kihlberg, J. Oral druggable space beyond the rule of 5: insights from drugs and clinical candidates. Chem. Biol. 2014, 21, 1115–1142. [Google Scholar] [CrossRef]
- National Committee for Clinical Laboratory Standards NCCLS (2002). https://chempedia.info/info/national_committee_for_clinical_laboratory_standards_nccls/.
- B. Satyanarayana P. Muralikrishna, D. Ravi Kumar & D. Ramachandran. Preparation and biological evaluation of phenothiazine derivatives, Journal of Chemical and Pharmaceutical Research, 2013, 5, 262–266.
- C. O. Vincent, Pharmaceutical Microbiology: Principles of the Pharmaceutical Applications of Antimicrobial Agents, El ’Demak Publishers 2005, Enugu, Nigeria.
- Ayuk Eugene, L.; Oforji Chizoba, F.; Aronimo Samuel, B.; Ugwu Francis, C.; Njokunwaogbu Ambrose, N. Determination of secondary metabolites and biological potential of Gnetun africanum (okazi) leaves. Pharm. Chem. J. 2017, 4, 115–122. [Google Scholar]
- Song, L. and Ji, Q. The crystal structure of Staphylococcus aureus CntA in apo form. Protein Data Bank. Accession code 5YH5, 2018. [CrossRef]
- Buffalo, C.Z., Bahn-Suh, A.J. and Ghosh, P. Structure of human C4b-binding protein alpha cain CCP domains 1 and 2 in complex with the hypervariable region of group A Streptococcus M28 protein. Protein Data Bank. Accession code 5YHP, 2016. [CrossRef]
- Mima, M. and Ushiyama, F. Crystal structure of E.coli DNA gyrase B in complex with 2-oxo-1,2-dihydroquinoline derivative. Protein Data Bank. Accession code 6KZV, 2020. [CrossRef]
- Bergeron, J.R.C. and Strynadka, N.C.J. Crystal structure of PrgK 19-92. Protein Data Bank. Accession code 4W4M, 2015. [CrossRef]
- Wei, W. F. and Van Aalten, D. M. F. Aspergillus fumigatus Rho1 F25N. Protein Data Bank. Accession code 5ZVP, 2019. [CrossRef]
- Bax, B.D., Chan, P., Eggleston, D.S., Fosberry, A., Gentry, D.R., Gorrec, F., Giordano, I., Hann, M.M., Hennessy, A., Hibbs, M., Huang, J., Jones, E., Jones, J., Brown, K.K., Lewis, C.J., May, E., Singh, O., Spitzfaden, C., Shen, C., Shillings, A., Theobald, A., Wohlkonig, A., Pearson, N.D. and Gwynn, M.N. The twinned 3.35A structure of S-aureus Gyrase complex with Ciprofloxacin and DNA. Protein Data Bank. Accession code 2XCT, 2010. [CrossRef]
- Sjogren, T. and Ekroos, M. Crystal structure of human P403A4 in complex with ketoconazole. Protein Data Bank. Accession code 2VOM, 2007. [CrossRef]
- Trott, O.; Olson, A.J. AutoDock Vina: improving the speed and accuracy of docking with a new scoring function, efficient optimization and multithreading, Journal of Computational Chemistry 2010, 31, 455–461. [CrossRef]
| Compounds | Salmonella | S. aureus | E. coli | S. pyogenes | A. fumigatus | ||
|---|---|---|---|---|---|---|---|
| 27 | -5.7 | -5.1 | -6.3 | -5.3 | -6.4 | ||
| 28 | -6.1 | -7.3 | -6.3 | -7.4 | -7.1 | ||
| 30 a | -6.3 | -5.5 | -7.2 | -5.7 | -7.1 | ||
| 30 b | -7.1 | -5.9 | -7.2 | -6.1 | -7.5 | ||
| 30 c | -7.1 | -6.6 | -7.0 | -6.6 | -7.4 | ||
| Ciprofloxacin | -7.4 | -7.6 | -6.1 | -7.0 | - | ||
| Gentamycin | -7.0 | -7.0 | -5.7 | -7.3 | - | ||
| Ketoconazole | - | - | - | - | -7.6 | ||
| Compounds | Salmonella | S. aureus | E. coli | S. pyogenes | A. fumigatus |
|---|---|---|---|---|---|
| 28 | R | 1.0 | R | R | R |
| 30a | 1..5 | 1.0 | 2.0 | R | R |
| 30b | 2.0 | 0.5 | 3.5 | R | R |
| 30c | 1.0 | 0.1 | 0.5 | R | 0.5 |
| Ciprofloxacin | 0.5 | 0.5 | 0.5 | 0.5 | - |
| Gentamycin | 0.5 | 0.5 | 0.5 | 0.5 | - |
| Ketoconazole | - | - | - | - | 0.5 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




