Submitted:
18 January 2024
Posted:
23 January 2024
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
2.1. Ethical approval
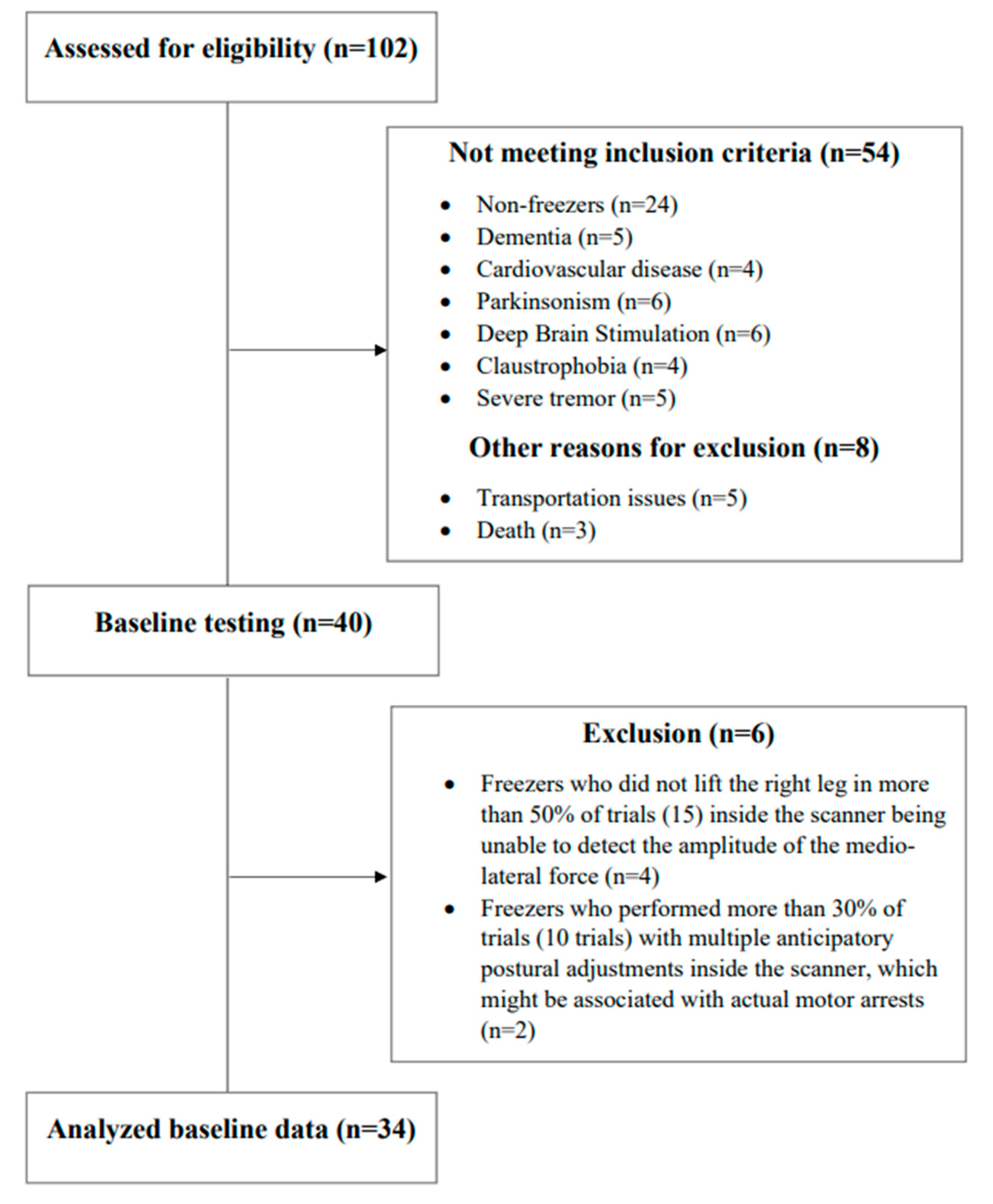
2.2. Participants
2.3. Study procedures
2.4. Outcome assessments
2.4.1. Clinical assessments
2.4.2. Behavioral assessments
2.4.3. Test and conditioned H-reflexes
2.4.4. Assessment of PSI of the soleus muscle during APA for step initiation
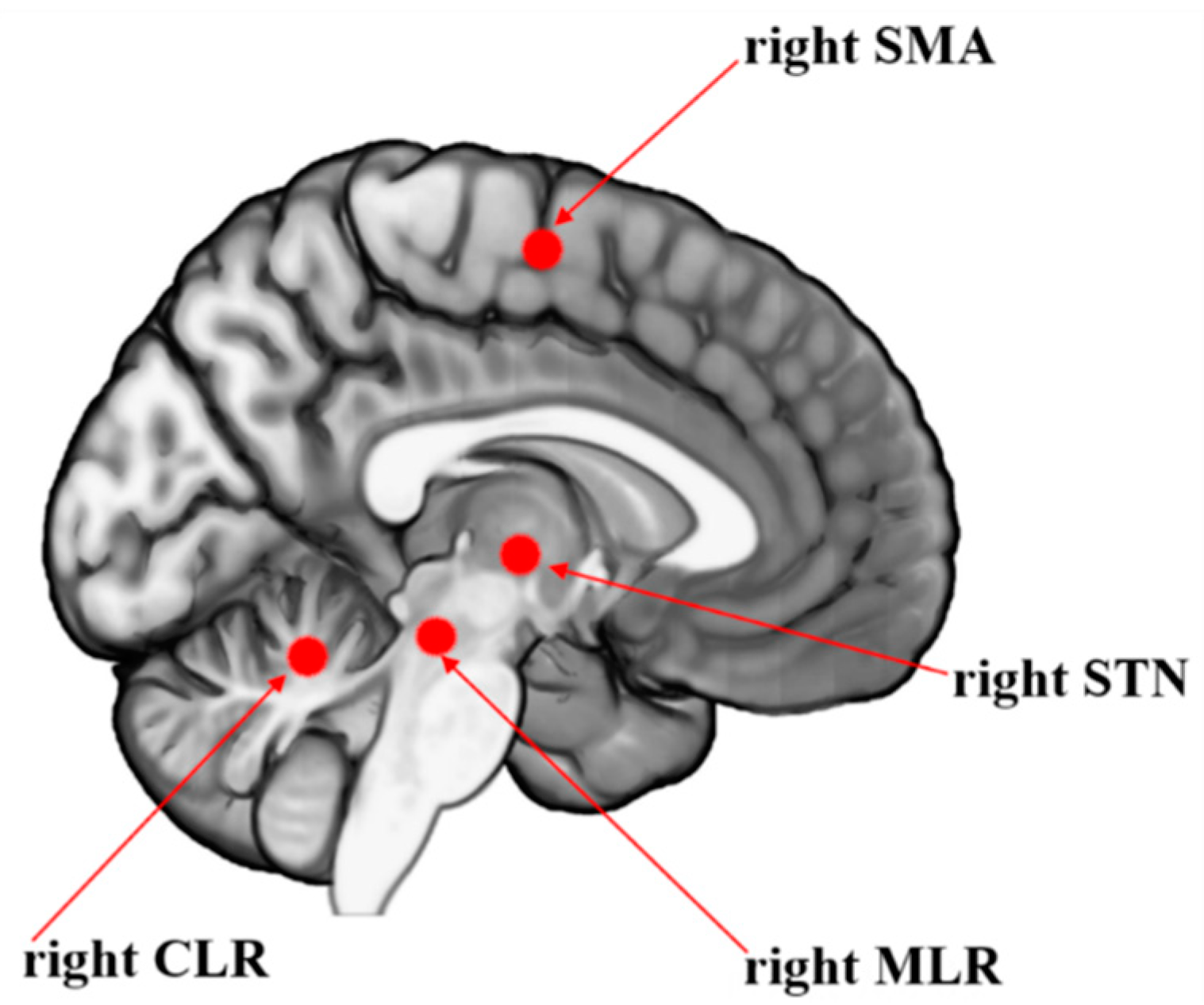
2.4.5. Beta of the BOLD signal change of locomotor regions during step initiation
2.5. Statistical Analyses
3. Results
3.1. Participants
3.2. MLR activity and APA amplitude during step initiation explain the loss of PSI of the soleus muscle for step initiation
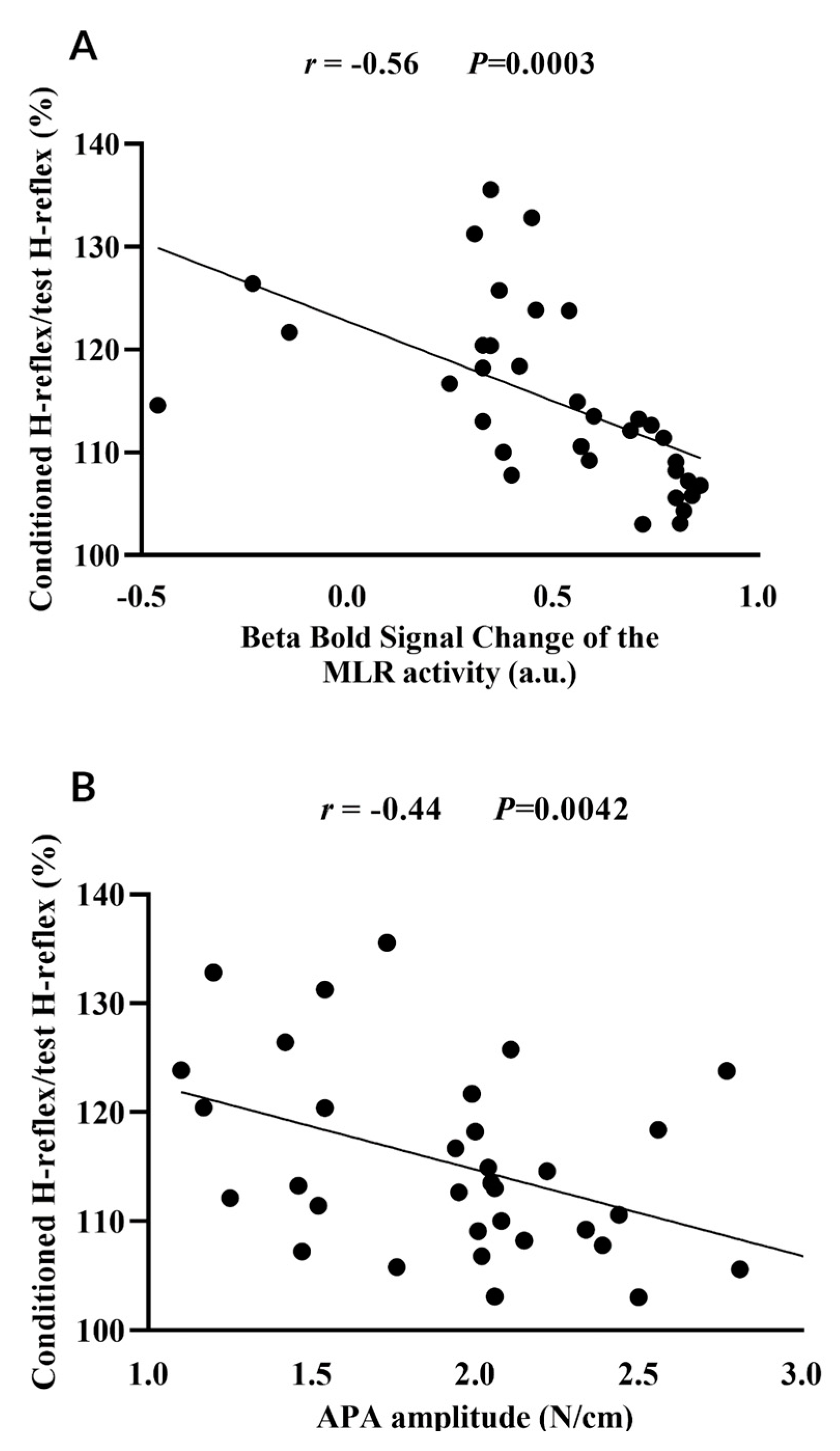
4. Discussion
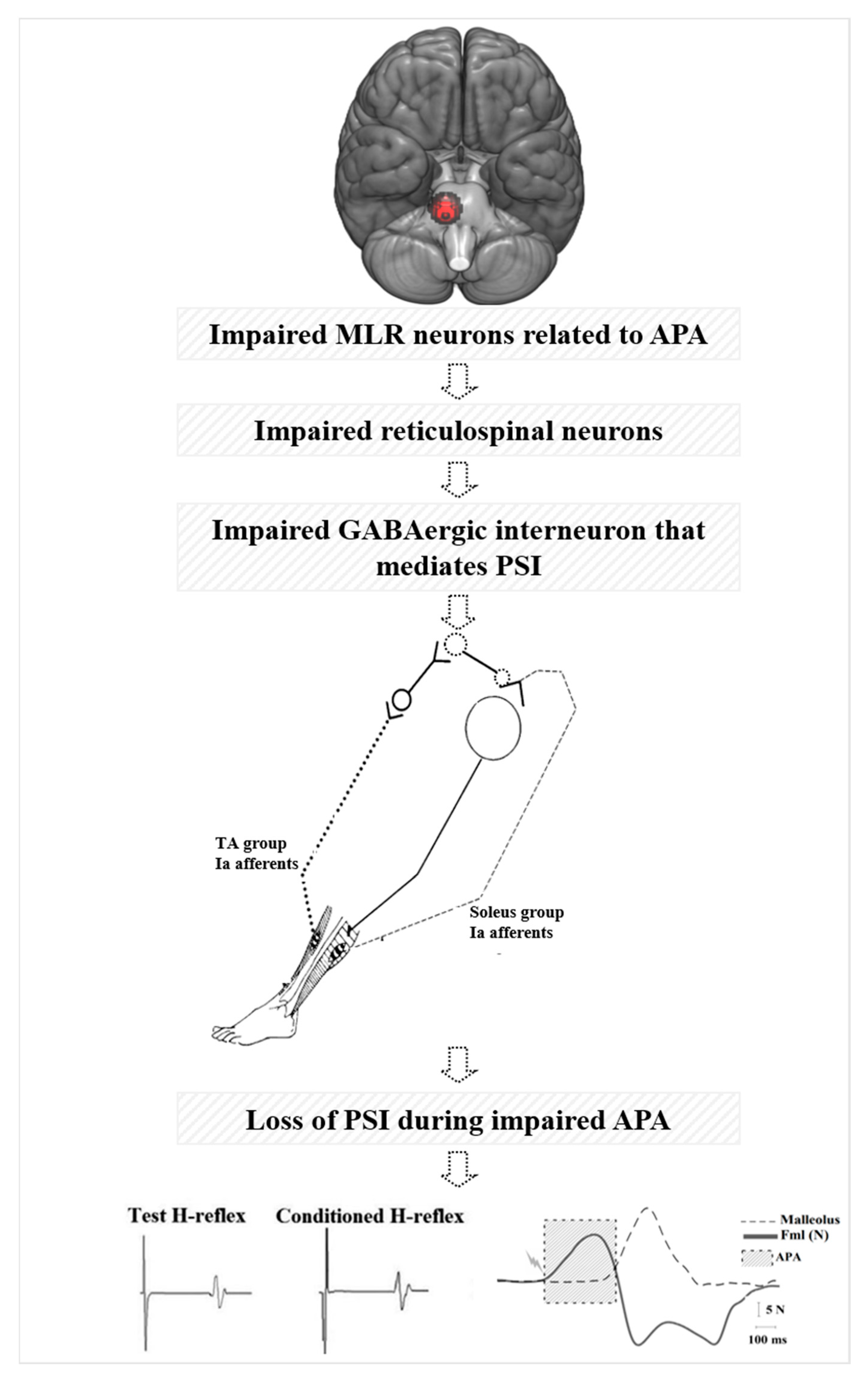
4.1. Why do MLR activity and APA amplitudes explain the loss of PSI of the soleus muscle during step initiation in freezers?
4.2. Future directions for treatment strategies
4.3. Limitations
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Amboni, M.; Stocchi, F.; Abbruzzese, G.; Morgante, L.; Onofrj, M.; Ruggieri, S.; Tinazzi, M.; Zappia, M.; Attar, M.; Colombo, D.; et al. Prevalence and associated features of self-reported freezing of gait in Parkinson disease: The DEEP FOG study. Parkinsonism & related disorders. 2015, 21, 644–649. [Google Scholar] [CrossRef]
- Bloem, B.R.; Hausdorff, J.M.; Visser, J.E.; Giladi, N. Falls and freezing of gait in Parkinson's disease: a review of two interconnected, episodic phenomena. Movement disorders : official journal of the Movement Disorder Society. 2004, 19, 871–884. [Google Scholar] [CrossRef]
- Moore, O.; Peretz, C.; Giladi, N. Freezing of gait affects quality of life of peoples with Parkinson's disease beyond its relationships with mobility and gait. Movement disorders : official journal of the Movement Disorder Society. 2007, 22, 2192–2195. [Google Scholar] [CrossRef] [PubMed]
- Nutt, J.G.; Bloem, B.R.; Giladi, N.; Hallett, M.; Horak, F.B.; Nieuwboer, A. Freezing of gait: moving forward on a mysterious clinical phenomenon. The Lancet. Neurology. 2011, 10, 734–744. [Google Scholar] [CrossRef]
- Yiou, E.; Caderby, T.; Delafontaine, A.; Fourcade, P.; Honeine, J.L. Balance control during gait initiation: State-of-the-art and research perspectives. World journal of orthopedics. 2017, 8, 815–828. [Google Scholar] [CrossRef] [PubMed]
- Schlenstedt, C.; Mancini, M.; Nutt, J.; Hiller, A.P.; Maetzler, W.; Deuschl, G.; Horak, F. Are Hypometric Anticipatory Postural Adjustments Contributing to Freezing of Gait in Parkinson's Disease? Front Aging Neurosci. 2018, 10, 36. [Google Scholar] [CrossRef]
- Lira, J.L.O.; Ugrinowitsch, C.; Coelho, D.B.; Teixeira, L.A.; de Lima-Pardini, A.C.; Magalhaes, F.H.; Barbosa, E.R.; Horak, F.B.; Silva-Batista, C. Loss of presynaptic inhibition for step initiation in parkinsonian individuals with freezing of gait. J Physiol. 2020, 598, 1611–1624. [Google Scholar] [CrossRef] [PubMed]
- Jacobs, J.V.; Horak, F.B. External postural perturbations induce multiple anticipatory postural adjustments when subjects cannot pre-select their stepping foot. Experimental brain research. 2007, 179, 29–42. [Google Scholar] [CrossRef]
- Cohen, R.G.; Nutt, J.G.; Horak, F.B. Recovery from Multiple APAs Delays Gait Initiation in Parkinson's Disease. Front Hum Neurosci. 2017, 11, 60. [Google Scholar] [CrossRef]
- Lira, J.L.O.; Ugrinowitsch, C.; Coelho, D.B.; Teixeira, L.A.; de Lima-Pardini, A.C.; Magalhaes, F.H.; Barbosa, E.R.; Horak, F.B.; Silva-Batista, C. Reply from Jumes Leopoldino Oliveira Lira, Carlos Ugrinowitsch, Daniel Boari Coelho, Luis Augusto Teixeira, Andrea Cristina de Lima-Pardini, Fernando Henrique Magalhaes, Egberto Reis Barbosa, Fay B. Horak, and Carla Silva-Batista. J Physiol. 2022, 600, 421–422. [Google Scholar] [CrossRef]
- Fling, B.W.; Cohen, R.G.; Mancini, M.; Nutt, J.G.; Fair, D.A.; Horak, F.B. Asymmetric pedunculopontine network connectivity in parkinsonian patients with freezing of gait. Brain. 2013, 136, 2405–2418. [Google Scholar] [CrossRef] [PubMed]
- Filho, S.S.; Coelho, D.B.; Ugrinowitsch, C.; de Souza, C.R.; Magalhães, F.H.; de Lima-Pardini, A.C.; de Oliveira É, M.B.; Mattos, E.; Teixeira, L.A.; Silva-Batista, C. Age-Related Changes in Presynaptic Inhibition During Gait Initiation. The journals of gerontology. Series A, Biological sciences and medical sciences. 2021, 76, 568–575. [Google Scholar] [CrossRef] [PubMed]
- Capaday, C.; Lavoie, B.A.; Comeau, F. Differential effects of a flexor nerve input on the human soleus H-reflex during standing versus walking. Canadian journal of physiology and pharmacology. 1995, 73, 436–449. [Google Scholar] [CrossRef] [PubMed]
- Katz, R.; Meunier, S.; Pierrot-Deseilligny, E. Changes in presynaptic inhibition of Ia fibres in man while standing. Brain. 1988, 111, 417–437. [Google Scholar] [CrossRef] [PubMed]
- Rudomin, P.; Schmidt, R.F. Presynaptic inhibition in the vertebrate spinal cord revisited. Experimental brain research. 1999, 129, 1–37. [Google Scholar] [CrossRef] [PubMed]
- Hultborn, H.; Meunier, S.; Morin, C.; Pierrot-Deseilligny, E. Assessing changes in presynaptic inhibition of I a fibres: a study in man and the cat. The Journal of physiology. 1987, 389, 729–756. [Google Scholar] [CrossRef] [PubMed]
- Sirois, J.; Frigon, A.; Gossard, J.P. Independent control of presynaptic inhibition by reticulospinal and sensory inputs at rest and during rhythmic activities in the cat. J Neurosci. 2013, 33, 8055–8067. [Google Scholar] [CrossRef] [PubMed]
- Fink, A.J.; Croce, K.R.; Huang, Z.J.; Abbott, L.F.; Jessell, T.M.; Azim, E. Presynaptic inhibition of spinal sensory feedback ensures smooth movement. Nature. 2014, 509, 43–48. [Google Scholar] [CrossRef]
- Baudry, S.; Duchateau, J. Age-related influence of vision and proprioception on Ia presynaptic inhibition in soleus muscle during upright stance. The Journal of physiology. 2012, 590, 5541–5554. [Google Scholar] [CrossRef]
- Fling, B.W.; Cohen, R.G.; Mancini, M.; Carpenter, S.D.; Fair, D.A.; Nutt, J.G.; Horak, F.B. Functional reorganization of the locomotor network in Parkinson patients with freezing of gait. PLoS One. 2014, 9, e100291. [Google Scholar] [CrossRef]
- Sinnamon, H.M.; Jassen, A.K.; Vita, L.A. Brainstem regions with neuronal activity patterns correlated with priming of locomotor stepping in the anesthetized rat. Neuroscience. 2000, 99, 77–91. [Google Scholar] [CrossRef]
- Gurfinkel, V.S.; Lipshits, M.I.; Lestienne, F.G. Anticipatory neck muscle activity associated with rapid arm movements. Neurosci Lett. 1988, 94, 94,104–108. [Google Scholar] [CrossRef]
- Viallet, F.; Massion, J.; Massarino, R.; Khalil, R. Coordination between posture and movement in a bimanual load lifting task: putative role of a medial frontal region including the supplementary motor area. Exp Brain Res. 1992, 88, 674–684. [Google Scholar] [CrossRef] [PubMed]
- Garcia-Rill, E.; Skinner, R.D. The mesencephalic locomotor region. II. Projections to reticulospinal neurons. Brain research. 1987, 411, 13–20. [Google Scholar] [CrossRef] [PubMed]
- Hirabayashi, R.; Kojima, S.; Edama, M.; Onishi, H. Activation of the Supplementary Motor Areas Enhances Spinal Reciprocal Inhibition in Healthy Individuals. Brain Sci. 2020, 10, 587. [Google Scholar] [CrossRef] [PubMed]
- Schepens, B.; Drew, T. Independent and convergent signals from the pontomedullary reticular formation contribute to the control of posture and movement during reaching in the cat. J Neurophysiol. 2004, 92, 2217–2238. [Google Scholar] [CrossRef] [PubMed]
- de Lima-Pardini, A.C.; Coelho, D.B.; Nucci, M.P.; Boffino, C.C.; Batista, A.X.; de Azevedo Neto, R.M.; Silva-Batista, C.; Barbosa, E.R.; Cohen, R.G.; Horak, F.B.; et al. Brain networks associated with anticipatory postural adjustments in Parkinson's disease patients with freezing of gait. Neuroimage Clin. 2020, 28, 102461. [Google Scholar] [CrossRef]
- de Lima-Pardini, A.C.; de Azevedo Neto, R.M.; Coelho, D.B.; Boffino, C.C.; Shergill, S.S.; de Oliveira Souza, C.; Brant, R.; Barbosa, E.R.; Cardoso, E.F.; Teixeira, L.A.; et al. An fMRI-compatible force measurement system for the evaluation of the neural correlates of step initiation. Scientific reports. 2017, 7, 43088. [Google Scholar] [CrossRef] [PubMed]
- Vieira-Yano, B.; Martini, D.N.; Horak, F.B.; de Lima-Pardini, A.; Almeida, F.; Santana, V.P.; Lima, D.; Batista, A.X.; Marquesini, R.; Lira, J.; et al. The Adapted Resistance Training with Instability Randomized Controlled Trial for Gait Automaticity. Movement disorders : official journal of the Movement Disorder Society. 2021, 36, 152–163. [Google Scholar] [CrossRef]
- Silva-Batista, C.; de Lima-Pardini, A.C.; Nucci, M.P.; Coelho, D.B.; Batista, A.; Piemonte, M.E.P.; Barbosa, E.R.; Teixeira, L.A.; Corcos, D.M.; Amaro, E., Jr.; et al. A Randomized, Controlled Trial of Exercise for Parkinsonian Individuals With Freezing of Gait. Movement disorders : official journal of the Movement Disorder Society. 2020, 35, 1607–1617. [Google Scholar] [CrossRef]
- Moreira-Neto, A.; Ugrinowitsch, C.; Coelho, D.B.; de Lima-Pardini, A.C.; Barbosa, E.R.; Teixeira, L.A.; Amaro, E., Jr.; Horak, F.B.; Mancini, M.; Nucci, M.P.; et al. Freezing of gait, gait initiation, and gait automaticity share a similar neural substrate in Parkinson's disease. Hum Mov Sci. 2022, 86, 103018. [Google Scholar] [CrossRef] [PubMed]
- Shine, J.M.; Matar, E.; Ward, P.B.; Bolitho, S.J.; Gilat, M.; Pearson, M.; Naismith, S.L.; Lewis, S.J. Exploring the cortical and subcortical functional magnetic resonance imaging changes associated with freezing in Parkinson's disease. Brain : a journal of neurology. 2013, 136, 1204–1215. [Google Scholar] [CrossRef]
- Hughes, A.J.; Daniel, S.E.; Kilford, L.; Lees, A.J. Accuracy of clinical diagnosis of idiopathic Parkinson's disease: a clinico-pathological study of 100 cases. J Neurol Neurosurg Psychiatry. 1992, 55, 181–184. [Google Scholar] [CrossRef] [PubMed]
- Nieuwboer, A.; Rochester, L.; Herman, T.; Vandenberghe, W.; Emil, G.E.; Thomaes, T.; Giladi, N. Reliability of the new freezing of gait questionnaire: agreement between patients with Parkinson's disease and their carers. Gait & posture. 2009, 30, 459–463. [Google Scholar] [CrossRef]
- Folstein, M.F.; Folstein, S.E.; McHugh, P.R. "Mini-mental state". A practical method for grading the cognitive state of patients for the clinician. J Psychiatr Res. 1975, 12, 189–198. [Google Scholar] [CrossRef]
- Seto, E.; Sela, G.; McIlroy, W.E.; Black, S.E.; Staines, W.R.; Bronskill, M.J.; McIntosh, A.R.; Graham, S.J. Quantifying head motion associated with motor tasks used in fMRI. NeuroImage. 2001, 14, 284–297. [Google Scholar] [CrossRef] [PubMed]
- Fahn, S.; Elton, R. UPDRS Program Members. Unified Parkinson’s disease rating scale. In: Fahn S, Marsden CD, Goldstein M. Calne DB, editors. Recent developments in Parkinson’s disease, Vol. 2. Florham Park, NJ: Macmillan Healthcare Information. 1987, p.153–163, 293–304.
- Tomlinson, C.L.; Stowe, R.; Patel, S.; Rick, C.; Gray, R.; Clarke, C.E. Systematic review of levodopa dose equivalency reporting in Parkinson's disease. Movement disorders official journal of the Movement Disorder Society. 2010, 25, 2649–2653. [Google Scholar] [CrossRef]
- Troyer, A.K.; Leach, L.; Strauss, E. Aging and response inhibition: Normative data for the Victoria Stroop Test. Neuropsychol Dev Cogn B Aging Neuropsychol Cogn. 2006, 13, 20–35. [Google Scholar] [CrossRef]
- Mancini, M.; Smulders, K.; Cohen, R.G.; Horak, F.B.; Giladi, N.; Nutt, J.G. The clinical significance of freezing while turning in Parkinson's disease. Neuroscience. 2017, 343, 222–228. [Google Scholar] [CrossRef]
- Crone, C.; Hultborn, H.; Mazieres, L.; Morin, C.; Nielsen, J.; Pierrot-Deseilligny, E. Sensitivity of monosynaptic test reflexes to facilitation and inhibition as a function of the test reflex size: a study in man and the cat. Experimental brain research. 1990, 81, 35–45. [Google Scholar] [CrossRef]
- Crone, C.; Hultborn, H.; Jespersen, B.; Nielsen, J. Reciprocal Ia inhibition between ankle flexors and extensors in man. The Journal of physiology. 1987, 389, 163–185. [Google Scholar] [CrossRef] [PubMed]
- Patikas, D.A.; Kotzamanidis, C.; Robertson, C.T.; Koceja, D.M. The effect of the ankle joint angle in the level of soleus Ia afferent presynaptic inhibition. Electromyography and clinical neurophysiology. 2004, 44, 503–511. [Google Scholar] [PubMed]
- Magalhaes, F.H.; Elias, L.A.; da Silva, C.R.; de Lima, F.F.; de Toledo, D.R.; Kohn, A.F. D1 and D2 Inhibitions of the Soleus H-Reflex Are Differentially Modulated during Plantarflexion Force and Position Tasks. PloS one. 2015, 10, e0143862. [Google Scholar] [CrossRef] [PubMed]
- Silva-Batista, C.; Mattos, E.C.; Corcos, D.M.; Wilson, J.M.; Heckman, C.J.; Kanegusuku, H.; Piemonte, M.E.; Tulio de Mello, M.; Forjaz, C.; Roschel, H.; et al. Resistance training with instability is more effective than resistance training in improving spinal inhibitory mechanisms in Parkinson's disease. J Appl Physiol (1985). 2017, 122, 1–10. [Google Scholar] [CrossRef]
- Iles, J.F. Evidence for cutaneous and corticospinal modulation of presynaptic inhibition of Ia afferents from the human lower limb. The Journal of physiology. 1996, 491, 197–207. [Google Scholar] [CrossRef] [PubMed]
- Earles, D.; Vardaxis, V.; Koceja, D. Regulation of motor output between young and elderly subjects. Clinical neurophysiology : official journal of the International Federation of Clinical Neurophysiology. 2001, 112, 1273–1279. [Google Scholar] [CrossRef]
- Geertsen, S.S.; Lundbye-Jensen, J.; Nielsen, J.B. Increased central facilitation of antagonist reciprocal inhibition at the onset of dorsiflexion following explosive strength training. Journal of applied physiology. 2008, 105, 915–922. [Google Scholar] [CrossRef]
- Knikou, M. The H-reflex as a probe: pathways and pitfalls. J Neurosci Methods. 2008, 171, 1–12. [Google Scholar] [CrossRef]
- Baudry, S.; Lecoeuvre, G.; Duchateau, J. Age-related changes in the behavior of the muscle-tendon unit of the gastrocnemius medialis during upright stance. Journal of applied physiology. 2012, 112, 296–304. [Google Scholar] [CrossRef]
- Wang, M.; Jiang, S.; Yuan, Y.; Zhang, L.; Ding, J.; Wang, J.; Zhang, J.; Zhang, K.; Wang, J. Alterations of functional and structural connectivity of freezing of gait in Parkinson's disease. Journal of neurology. 2016, 263, 1583–1592. [Google Scholar] [CrossRef]
- Smith, S.M.; Jenkinson, M.; Woolrich, M.W.; Beckmann, C.F.; Behrens, T.E.; Johansen-Berg, H.; Bannister, P.R.; De Luca, M.; Drobnjak, I.; Flitney, D.E.; et al. Advances in functional and structural MR image analysis and implementation as FSL. NeuroImage. 2004, 23, S208–S219. [Google Scholar] [CrossRef] [PubMed]
- Poldrack, R.A.; Fletcher, P.C.; Henson, R.N.; Worsley, K.J.; Brett, M.; Nichols, T.E. Guidelines for reporting an fMRI study. NeuroImage. 2008, 40, 409–414. [Google Scholar] [CrossRef] [PubMed]
- Jenkinson, M.; Bannister, P.; Brady, M.; Smith, S. Improved optimization for the robust and accurate linear registration and motion correction of brain images. NeuroImage. 2002, 17, 825–841. [Google Scholar] [CrossRef] [PubMed]
- Smith, S.M. Fast robust automated brain extraction. Human brain mapping. 2002, 17, 143–155. [Google Scholar] [CrossRef] [PubMed]
- Jenkinson, M.; Smith, S. A global optimisation method for robust affine registration of brain images. Medical image analysis. 2001, 5, 143–156. [Google Scholar] [CrossRef]
- Buschbacher, R.M. Normal range for H-reflex recording from the calf muscles. American journal of physical medicine & rehabilitation. 1999, 78, S75–S79. [Google Scholar] [CrossRef]
- Morita, H.; Shindo, M.; Yanagawa, S.; Yoshida, T.; Momoi, H.; Yanagisawa, N. Progressive decrease in heteronymous monosynaptic Ia facilitation with human ageing. Experimental brain research. 1995, 104, 167–170. [Google Scholar] [CrossRef]
- Dormann, C.F.; Elith, J.; Bacher, S.; Carré, G.C.G.; García Márquez, J.R.; Gruber, B.; Lafourcade, B.; Leitao, P.J.; Münkemüller, T.; McClean, C.J.; et al. Collinearity: a review of methods to deal with it and a simulation study evaluating their performance. Ecography. 2012, 36, 27–46. [Google Scholar] [CrossRef]
- Lewis, S.J.; Barker, R.A. A pathophysiological model of freezing of gait in Parkinson's disease. Parkinsonism & related disorders. 2009, 15, 333–338. [Google Scholar] [CrossRef]
- Sherman, D.; Fuller, P.M.; Marcus, J.; Yu, J.; Zhang, P.; Chamberlin, N.L.; Saper, C.B.; Lu, J. Anatomical Location of the Mesencephalic Locomotor Region and Its Possible Role in Locomotion, Posture, Cataplexy, and Parkinsonism. Frontiers in neurology. 2015, 6, 140. [Google Scholar] [CrossRef]
- Snijders, A.H.; Leunissen, I.; Bakker, M.; Overeem, S.; Helmich, R.C.; Bloem, B.R.; Toni, I. Gait-related cerebral alterations in patients with Parkinson's disease with freezing of gait. Brain a journal of neurology. 2011, 134, 59–72. [Google Scholar] [CrossRef] [PubMed]
- Shik, M.L.J.B. Control of walking and running by means of electrical stimulation of the midbrain. Biophys 1966, 11, 659–666. [Google Scholar]
- Shik, M.L.; Orlovsky, G.N. Neurophysiology of locomotor automatism. Physiological reviews. 1976, 56, 465–501. [Google Scholar] [CrossRef] [PubMed]
- Takakusaki, K.; Chiba, R.; Nozu, T.; Okumura, T. Brainstem control of locomotion and muscle tone with special reference to the role of the mesopontine tegmentum and medullary reticulospinal systems. Journal of neural transmission 2016, 123, 695–729. [Google Scholar] [CrossRef] [PubMed]
- Takakusaki, K.; Habaguchi, T.; Ohtinata-Sugimoto, J.; Saitoh, K.; Sakamoto, T. Basal ganglia efferents to the brainstem centers controlling postural muscle tone and locomotion: a new concept for understanding motor disorders in basal ganglia dysfunction. Neuroscience. 2003, 119, 293–308. [Google Scholar] [CrossRef] [PubMed]
- Rossignol, S.; Dubuc, R.; Gossard, J.P. Dynamic sensorimotor interactions in locomotion. Physiological reviews. 2006, 86, 89–154. [Google Scholar] [CrossRef] [PubMed]
- Jordan, L.M.; Liu, J.; Hedlund, P.B.; Akay, T.; Pearson, K.G. Descending command systems for the initiation of locomotion in mammals. Brain research reviews. 2008, 57, 183–191. [Google Scholar] [CrossRef] [PubMed]
- Takakusaki, K.; Kohyama, J.; Matsuyama, K.; Mori, S. Medullary reticulospinal tract mediating the generalized motor inhibition in cats: parallel inhibitory mechanisms acting on motoneurons and on interneuronal transmission in reflex pathways. Neuroscience. 2001, 103, 511–527. [Google Scholar] [CrossRef] [PubMed]
- Carpenter, D.; Engberg, I.; Lundberg, A. Primary afferent depolarization evoked from the brain stem and the cerebellum. Archives italiennes de biologie. 1966, 104, 73–85. [Google Scholar]
- Jankowska, E.; Lund, S.; Lundberg, A.; Pompeiano, O. Inhibitory effects evoked through ventral reticulospinal pathways. Archives italiennes de biologie. 1968, 106, 124–140. [Google Scholar]
- Takakusaki, K.; Ohta, Y.; Mori, S. Single medullary reticulospinal neurons exert postsynaptic inhibitory effects via inhibitory interneurons upon alpha-motoneurons innervating cat hindlimb muscles. Experimental brain research 1989, 74, 11–23. [Google Scholar] [CrossRef] [PubMed]
- Sakai, S.T.; Davidson, A.G.; Buford, J.A. Reticulospinal neurons in the pontomedullary reticular formation of the monkey (Macaca fascicularis). Neuroscience 2009, 163, 1158–1170. [Google Scholar] [CrossRef] [PubMed]
- Riddle, C.N.; Edgley, S.A.; Baker, S.N. Direct and indirect connections with upper limb motoneurons from the primate reticulospinal tract. The Journal of neuroscience the official journal of the Society for Neuroscience. 2009, 29, 4993–4999. [Google Scholar] [CrossRef]
- Jacobs, J.V.; Nutt, J.G.; Carlson-Kuhta, P.; Stephens, M.; Horak, F.B. Knee trembling during freezing of gait represents multiple anticipatory postural adjustments. Experimental neurology. 2009, 215, 334–341. [Google Scholar] [CrossRef] [PubMed]
- Takakusaki, K. Functional Neuroanatomy for Posture and Gait Control. J Mov Disord. 2017, 10, 1–17. [Google Scholar] [CrossRef] [PubMed]
- Takakusaki, K.; Kohyama, J.; Matsuyama, K. Medullary reticulospinal tract mediating a generalized motor inhibition in cats: III. Functional organization of spinal interneurons in the lower lumbar segments. Neuroscience. 2003, 121, 731–746. [Google Scholar] [CrossRef]
- Nachev, P.; Kennard, C.; Husain, M. Functional role of the supplementary and pre-supplementary motor areas. Nat Rev Neurosci. 2008, 9, 856–869. [Google Scholar] [CrossRef] [PubMed]
- Takakusaki, K. Neurophysiology of gait: from the spinal cord to the frontal lobe. Movement disorders official journal of the Movement Disorder Society 2013, 28, 1483–1491. [Google Scholar] [CrossRef]
- Lundberg, A.; Voorhoeve, P. Effects from the pyramidal tract on spinal reflex arcs. Acta physiologica Scandinavica 1962, 56, 201–219. [Google Scholar] [CrossRef]
- Iles, J.F. Evidence for cutaneous and corticospinal modulation of presynaptic inhibition of Ia afferents from the human lower limb. The Journal of physiology 1996, 491, 197–207. [Google Scholar] [CrossRef]
- Meunier, S.; Pierrot-Deseilligny, E.J.E. Cortical control of presynaptic inhibition of Ia afferents in humans. Experimental brain research 1998, 119, 415–426. [Google Scholar] [CrossRef] [PubMed]
- Vercruysse, S.; Spildooren, J.; Heremans, E.; Wenderoth, N.; Swinnen, S.P.; Vandenberghe, W.; Nieuwboer, A. The neural correlates of upper limb motor blocks in Parkinson's disease and their relation to freezing of gait. Cerebral cortex 2014, 24, 3154–3166. [Google Scholar] [CrossRef] [PubMed]
- Takakusaki, K.; Takahashi, M.; Noguchi, T.; Chiba, R.J.N.; Neuroscience, C. Neurophysiological mechanisms of gait disturbance in advanced Parkinson's disease patients. Neurol. Clin. Neurosci. 2022. [Google Scholar] [CrossRef]
- Lewis, S.J.; Shine, J.M. The Next Step: A Common Neural Mechanism for Freezing of Gait. The Neuroscientist: a review journal bringing neurobiology, neurology and psychiatry 2016, 22, 72–82. [Google Scholar] [CrossRef] [PubMed]
- Fling, B.W.; Cohen, R.G.; Mancini, M.; Carpenter, S.D.; Fair, D.A.; Nutt, J.G.; Horak, F.B. Functional reorganization of the locomotor network in Parkinson patients with freezing of gait. PloS ONE 2014, 9, e100291. [Google Scholar] [CrossRef] [PubMed]
- Pahapill, P.A.; Lozano, A.M. The pedunculopontine nucleus and Parkinson's disease. Brain : a journal of neurology. 2000, 123, 1767–1783. [Google Scholar] [CrossRef]
- Zweig, R.M.; Jankel, W.R.; Hedreen, J.C.; Mayeux, R.; Price, D.L. The pedunculopontine nucleus in Parkinson's disease. Annals of neurology. 1989, 26, 41–46. [Google Scholar] [CrossRef] [PubMed]
- Curtze, C.; Nutt, J.G.; Carlson-Kuhta, P.; Mancini, M.; Horak, F.B. Levodopa Is a Double-Edged Sword for Balance and Gait in People With Parkinson's Disease. Movement disorders : official journal of the Movement Disorder Society. 2015, 30, 1361–1370. [Google Scholar] [CrossRef] [PubMed]
- Lin, F.; Wu, D.; Lin, C.; Cai, H.; Chen, L.; Cai, G.; Ye, Q.; Cai, G. Pedunculopontine Nucleus Deep Brain Stimulation Improves Gait Disorder in Parkinson's Disease: A Systematic Review and Meta-analysis. Neurochemical research. 2020, 45, 709–719. [Google Scholar] [CrossRef]
- Pierantozzi, M.; Palmieri, M.G.; Galati, S.; Stanzione, P.; Peppe, A.; Tropepi, D.; Brusa, L.; Pisani, A.; Moschella, V.; Marciani, M.G.; et al. Pedunculopontine nucleus deep brain stimulation changes spinal cord excitability in Parkinson's disease patients. Journal of neural transmission (Vienna, Austria : 1996). 2008, 115, 731–735. [Google Scholar] [CrossRef]
- Streumer, J.; Selvaraj, A.K.; Kurt, E.; Bloem, B.R.; Esselink, R.A.J.; Bartels, R.; Georgiev, D.; Vinke, R.S. Does spinal cord stimulation improve gait in Parkinson's disease: A comprehensive review. Parkinsonism & related disorders. [CrossRef]
- Fonoff, E.T.; de Lima-Pardini, A.C.; Coelho, D.B.; Monaco, B.A.; Machado, B.; Pinto de Souza, C.; Dos Santos Ghilardi, M.G.; Hamani, C. Spinal Cord Stimulation for Freezing of Gait: From Bench to Bedside. Frontiers in neurology. 2019, 10, 905. [Google Scholar] [CrossRef] [PubMed]
- de Lima-Pardini, A.C.; Coelho, D.B.; Souza, C.P.; Souza, C.O.; Ghilardi, M.; Garcia, T.; Voos, M.; Milosevic, M.; Hamani, C.; Teixeira, L.A.; et al. Effects of spinal cord stimulation on postural control in Parkinson's disease patients with freezing of gait. eLife. 2018, 7, 2–10. [Google Scholar] [CrossRef]
- Samotus, O.; Parrent, A.; Jog, M. Spinal Cord Stimulation Therapy for Gait Dysfunction in Advanced Parkinson's Disease Patients. Movement disorders : official journal of the Movement Disorder Society. 2018, 33, 783–792. [Google Scholar] [CrossRef] [PubMed]
- Fuentes, R.; Petersson, P.; Siesser, W.B.; Caron, M.G.; Nicolelis, M.A. Spinal cord stimulation restores locomotion in animal models of Parkinson's disease. Science. 2009, 323, 1578–1582. [Google Scholar] [CrossRef]
- Yadav, A.P.; Nicolelis, M.A.L. Electrical stimulation of the dorsal columns of the spinal cord for Parkinson's disease. Movement disorders : official journal of the Movement Disorder Society. 2017, 32, 820–832. [Google Scholar] [CrossRef]
- Fanselow, E.E.; Reid, A.P.; Nicolelis, M.A. Reduction of pentylenetetrazole-induced seizure activity in awake rats by seizure-triggered trigeminal nerve stimulation. The Journal of neuroscience : the official journal of the Society for Neuroscience. 2000, 20, 8160–8168. [Google Scholar] [CrossRef] [PubMed]
- Jasmin, L.; Wu, M.V.; Ohara, P.T. GABA puts a stop to pain. Current drug targets. CNS and neurological disorders. 2004, 3, 487–505. [Google Scholar] [CrossRef] [PubMed]
- Magoul, R.; Onteniente, B.; Geffard, M.; Calas, A. Anatomical distribution and ultrastructural organization of the GABAergic system in the rat spinal cord. An immunocytochemical study using anti-GABA antibodies. Neuroscience. 1987, 20, 1001–1009. [Google Scholar] [CrossRef]
- Emborg, M.E.; Carbon, M.; Holden, J.E.; During, M.J.; Ma, Y.; Tang, C.; Moirano, J.; Fitzsimons, H.; Roitberg, B.Z.; Tuccar, E.; et al. Subthalamic glutamic acid decarboxylase gene therapy: changes in motor function and cortical metabolism. Journal of cerebral blood flow and metabolism : official journal of the International Society of Cerebral Blood Flow and Metabolism. 2007, 27, 501–509. [Google Scholar] [CrossRef]
- Kaplitt, M.G.; Feigin, A.; Tang, C.; Fitzsimons, H.L.; Mattis, P.; Lawlor, P.A.; Bland, R.J.; Young, D.; Strybing, K.; Eidelberg, D.; et al. Safety and tolerability of gene therapy with an adeno-associated virus (AAV) borne GAD gene for Parkinson's disease: an open label, phase I trial. Lancet 2007, 369, 2097–2105. [Google Scholar] [CrossRef]
- Mackie, M.; Hughes, D.I.; Maxwell, D.J.; Tillakaratne, N.J.; Todd, A.J. Distribution and colocalisation of glutamate decarboxylase isoforms in the rat spinal cord. Neuroscience 2003, 119, 461–472. [Google Scholar] [CrossRef]
- Luo, J.; Kaplitt, M.G.; Fitzsimons, H.L.; Zuzga, D.S.; Liu, Y.; Oshinsky, M.L.; During, M.J. Subthalamic GAD gene therapy in a Parkinson's disease rat model. Science 2002, 298, 425–429. [Google Scholar] [CrossRef] [PubMed]
- Silva-Batista, C.; Corcos, D.M.; Roschel, H.; Kanegusuku, H.; Gobbi, L.T.; Piemonte, M.E.; Mattos, E.C.; MT, D.E.M.; Forjaz, C.L.; Tricoli, V.; et al. Resistance Training with Instability for Patients with Parkinson's Disease. Med Sci Sports Exerc. 2016, 48, 1678–1687. [Google Scholar] [CrossRef] [PubMed]
- Harrington, W.; Greenberg, A.; King, E.; McNames, J.; Holmstrom, L.; Horak, F.B.; Mancini, M. Alleviating Freezing of Gait using phase-dependent tactile biofeedback. Annual International Conference of the IEEE Engineering in Medicine and Biology Society. IEEE Engineering in Medicine and Biology Society. Annual International Conference. 2016, 2016, 5841–5844. [Google Scholar] [CrossRef] [PubMed]
- Novak, P.; Novak, V. Effect of step-synchronized vibration stimulation of soles on gait in Parkinson's disease: a pilot study. Journal of neuroengineering and rehabilitation. 2006, 3, 9. [Google Scholar] [CrossRef] [PubMed]
- Mancini, M.; Smulders, K.; Harker, G.; Stuart, S.; Nutt, J.G. Assessment of the ability of open- and closed-loop cueing to improve turning and freezing in people with Parkinson's disease. Scientific reports. 2018, 8, 12773. [Google Scholar] [CrossRef]
- Phuenpathom, W.; Panyakaew, P.; Vateekul, P.; Surangsrirat, D.; Hiransuthikul, A.; Bhidayasiri, R. Vibratory and plantar pressure stimulation: Steps to improve freezing of gait in Parkinson's disease. Parkinsonism & related disorders 2022, 105, 43–51. [Google Scholar] [CrossRef]
- Klaver, E.C.; van Vugt, J.P.P.; Bloem, B.R.; van Wezel, R.J.A.; Nonnekes, J.; Tjepkema-Cloostermans, M.C. Good vibrations: tactile cueing for freezing of gait in Parkinson's disease. Journal of neurology 2023. [Google Scholar] [CrossRef] [PubMed]
- Nolano, M.; Provitera, V.; Estraneo, A.; Selim, M.M.; Caporaso, G.; Stancanelli, A.; Saltalamacchia, A.M.; Lanzillo, B.; Santoro, L. Sensory deficit in Parkinson's disease: evidence of a cutaneous denervation. Brain: a journal of neurology. 2008, 131, 1903–1911. [Google Scholar] [CrossRef]
- Souron, R.; Baudry, S.; Millet, G.Y.; Lapole, T. Vibration-induced depression in spinal loop excitability revisited. The Journal of physiology. 2019, 597, 5179–5193. [Google Scholar] [CrossRef]
- Gillies, J.D.; Lance, J.W.; Neilson, P.D.; Tassinari, C.A. Presynaptic inhibition of the monosynaptic reflex by vibration. The Journal of physiology. 1969, 205, 329–339. [Google Scholar] [CrossRef]
- Desmedt, J.E.; Godaux, E. Mechanism of the vibration paradox: excitatory and inhibitory effects of tendon vibration on single soleus muscle motor units in man. The Journal of physiology. 1978, 285, 197–207. [Google Scholar] [CrossRef] [PubMed]
- Lapole, T.; Deroussen, F.; Pérot, C.; Petitjean, M. Acute effects of Achilles tendon vibration on soleus and tibialis anterior spinal and cortical excitability. Applied physiology, nutrition, and metabolism = Physiologie appliquee, nutrition et metabolisme. 2012, 37, 657–663. [Google Scholar] [CrossRef] [PubMed]
- Lapole, T.; Canon, F.; Pérot, C. Acute postural modulation of the soleus H-reflex after Achilles tendon vibration. Neuroscience letters. 2012, 523, 154–157. [Google Scholar] [CrossRef]
- Cui, X.; Bray, S.; Bryant, D.M.; Glover, G.H.; Reiss, A.L. A quantitative comparison of NIRS and fMRI across multiple cognitive tasks. NeuroImage. 2011, 54, 2808–2821. [Google Scholar] [CrossRef]
- de Lima-Pardini, A.C.; de Azevedo Neto, R.M.; Coelho, D.B.; Boffino, C.C.; Shergill, S.S.; de Oliveira Souza, C.; Brant, R.; Barbosa, E.R.; Cardoso, E.F.; Teixeira, L.A.; et al. An fMRI-compatible force measurement system for the evaluation of the neural correlates of step initiation. Sci. Rep. 2017, 7, 43088. [Google Scholar] [CrossRef] [PubMed]
| Freezers (n=34) | Range | |
| Demographics | ||
| Men/women (number) | 26/8 | - |
| Age (years) | 63.7(9.0) | 49 to 80 |
| Educational level (years) | 10.1(5.2) | 4 to 24 |
| Body mass (kg) | 69.6(11.2) | 49 to 94 |
| Height (cm) | 1.5(0.2) | 1 to 2 |
| Body mass index (kg/m2) | 25.7(3.1) | 20 to 32 |
| MMSE (score) | 26.0(1.6) | 24 to 29 |
| Clinical variables | ||
| Years since diagnosis (years) | 8.8(5.1) | 2 to 25 |
| Hoehn and Yahr staging scale (a.u) | 3.2(0.4) | 3 to 4 |
| Number of participants in stage 3 | 28 | - |
| Number of participants in stage 4 | 6 | - |
| UPDRS-III (score) | 49.9(11.2) | 23 to 67 |
| PIGD (score) | 8.6(2.3) | 4 to 13 |
| NFOGQ (score) | 22.2(5.4) | 12 to 28 |
| Stroop Color-Word Test (a.u) | 69.5(42.9) | 20.8 to 160.2 |
| L-Dopa equivalent units (mg•day-1) | 802.6(270.7) | 300 to 1300 |
| Behavioral variables | ||
| The ratio of the conditioned H-reflex relative to the test H-reflex (%) | 115.1(8.6) | 103.0 to 135.6 |
| FOG-ratio (a.u) | 12.3(10.8) | 2.1 to 54.6 |
| APA amplitude (N/cm) | 1.9(0.4) | 1.1 to 3.0 |
| APA duration (ms) | 483.1(59.9) | 402.6 to 676.7 |
| raEMG of the tibial anterior muscle (mV) | 0.06(0.03) | 0.1 to 0.10 |
| raEMG of the soleus muscle (mV) | 0.08(0.04) | 0.02 to 0.19 |
| Co-contraction ratio (%) | 89.8(58.9) | 0 to 219.9 |
| Beta of the BOLD signal change | ||
| Beta of BOLD signal change of the right SMA (a.u.) | 0.3(0.4) | -0.5 to 1.1 |
| Beta of BOLD signal change of the right STN (a.u.) | 0.3(0.5) | -1.0 to 0.9 |
| Beta of BOLD signal change of the right MLR (a.u.) | 0.4(0.3) | -0.4 to 0.8 |
| Beta of BOLD signal change of the right CLR (a.u.) | 0.3(0.5) | -1.1 to 1.2 |
| Independent factors | Partial R² | Model R² Change |
F Value | P value | Variance Explained (Adjusted Model R2 Change) |
|---|---|---|---|---|---|
| Beta of BOLD signal change of the MLR during step initiation (a.u) | 0.3152 | 0.3152 | 14.73 | 0.0006 | 0.49 |
| APA amplitude (N/cm) | 0.1347 | 0.4500 | 7.59 | 0.0097 | |
| FOG-ratio (a.u.) Disease duration (years) |
0.0541 0.0549 |
0.5041 0.5590 |
3.28 3.61 |
0.0804 0.0674 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




