Submitted:
19 January 2024
Posted:
22 January 2024
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Pathogenesis
2.1. Theory of Invagination of Endometrial Basalis in the Myometrium
2.2. De Novo Development Theories
3. Endocrine Pathogenic Mediators and Molecular Mechanisms
3.1. Intrinsic Hormonal Dysregulation
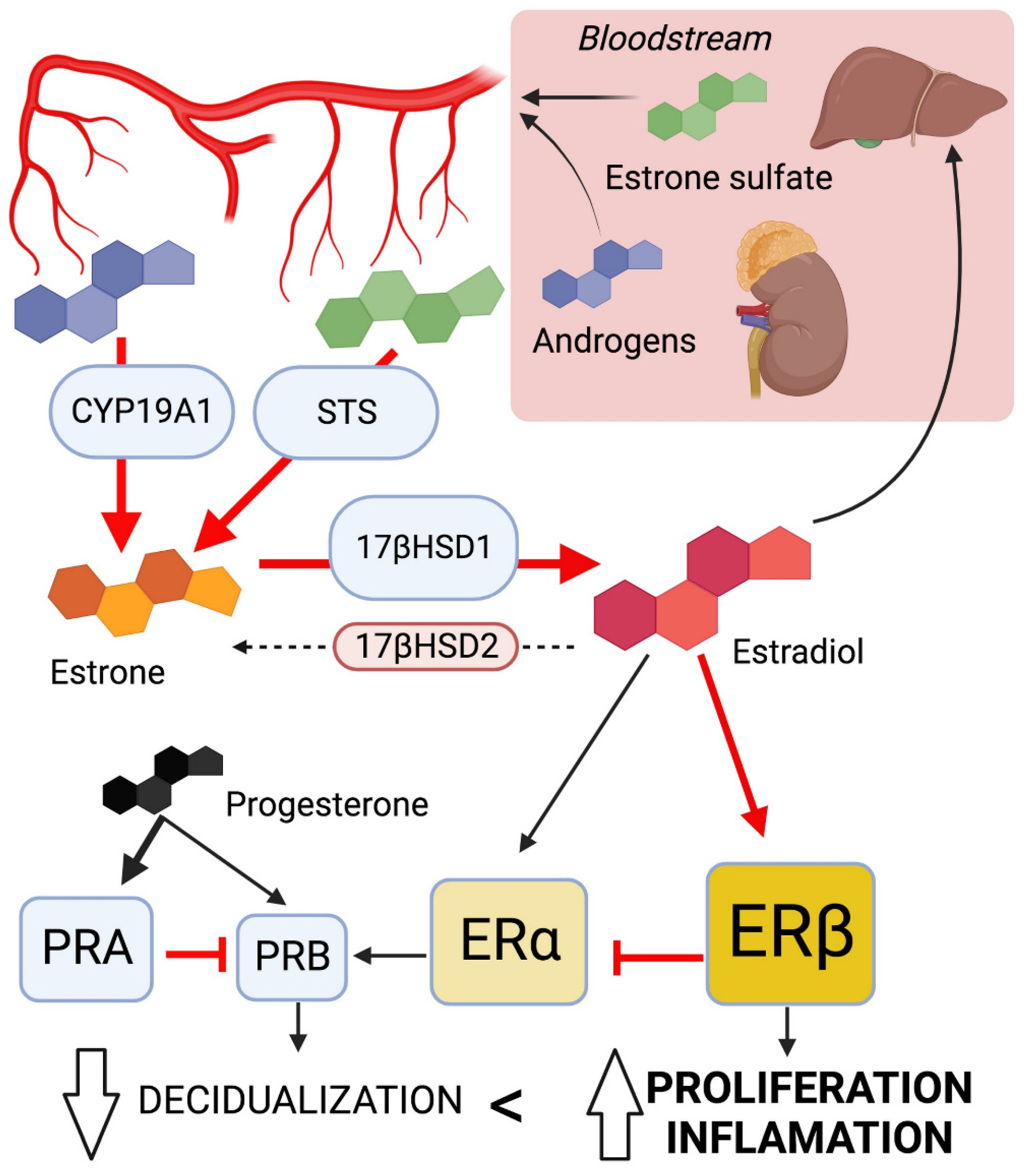
3.1.1. The Imbalance of Sex Steroid Hormones
Effects of High Estrogen Concentration
Hypersensitivity to Estrogen
Progesterone Resistance
Summary on Sex Steroid Dysregulation
3.1.2. The Pituitary Gland Influence
Prolactin
Oxytocin
Insulin-Like Growth Factor 1
3.1.3. Genetics and Epigenetics Alteration of Endocrine Signaling
3.2. Extrinsic Factors
3.2.1. Medical Therapies
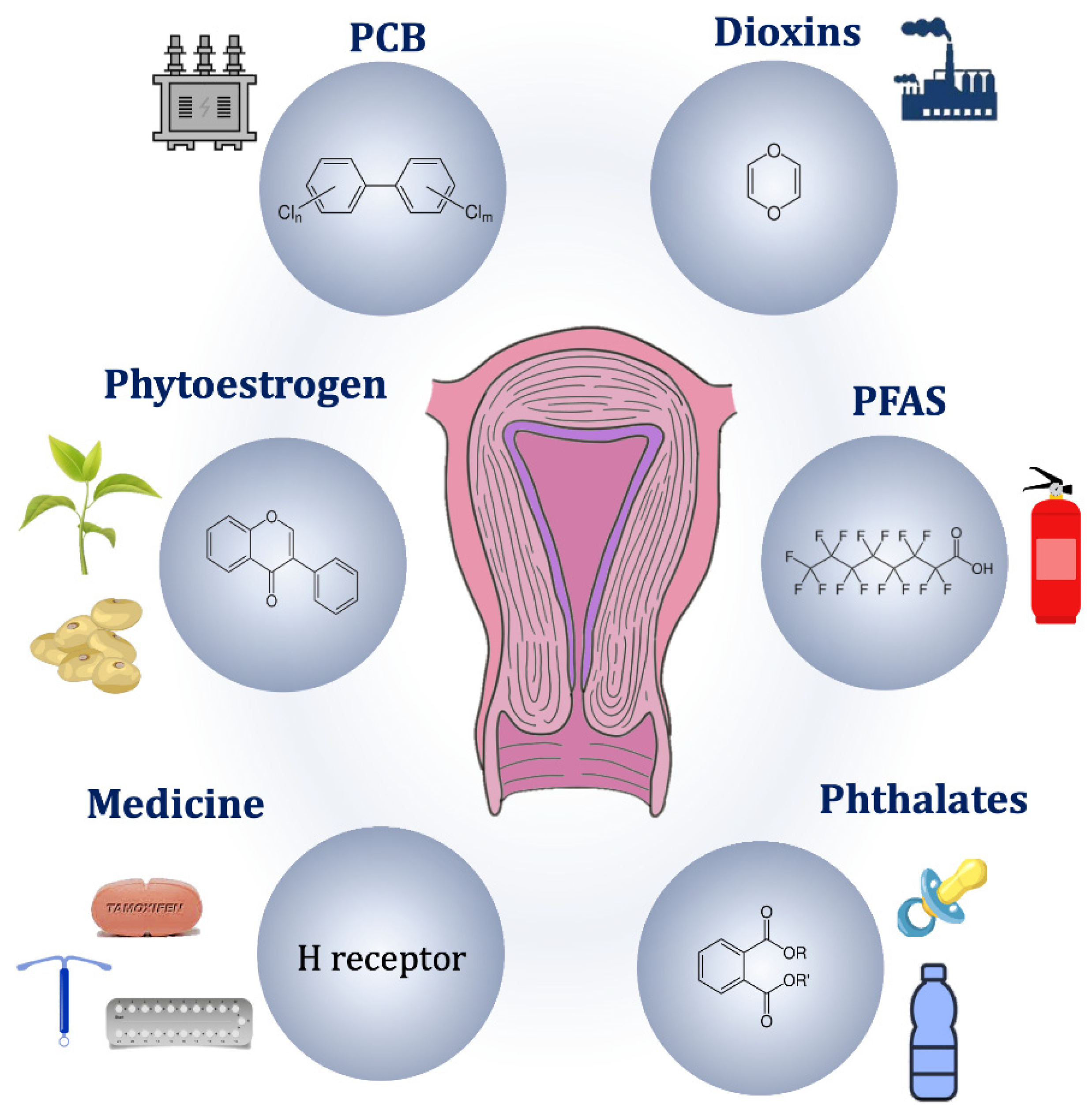
3.2.2. Endocrine Disrupting Chemicals
Persistent Organic Pollutants
Non-Persistent Organic Pollutants
3.2.3. Natural Endocrine Disruptors
3.2.4. Mode of Action of Endocrine Disruption
4. Infertility in ADM
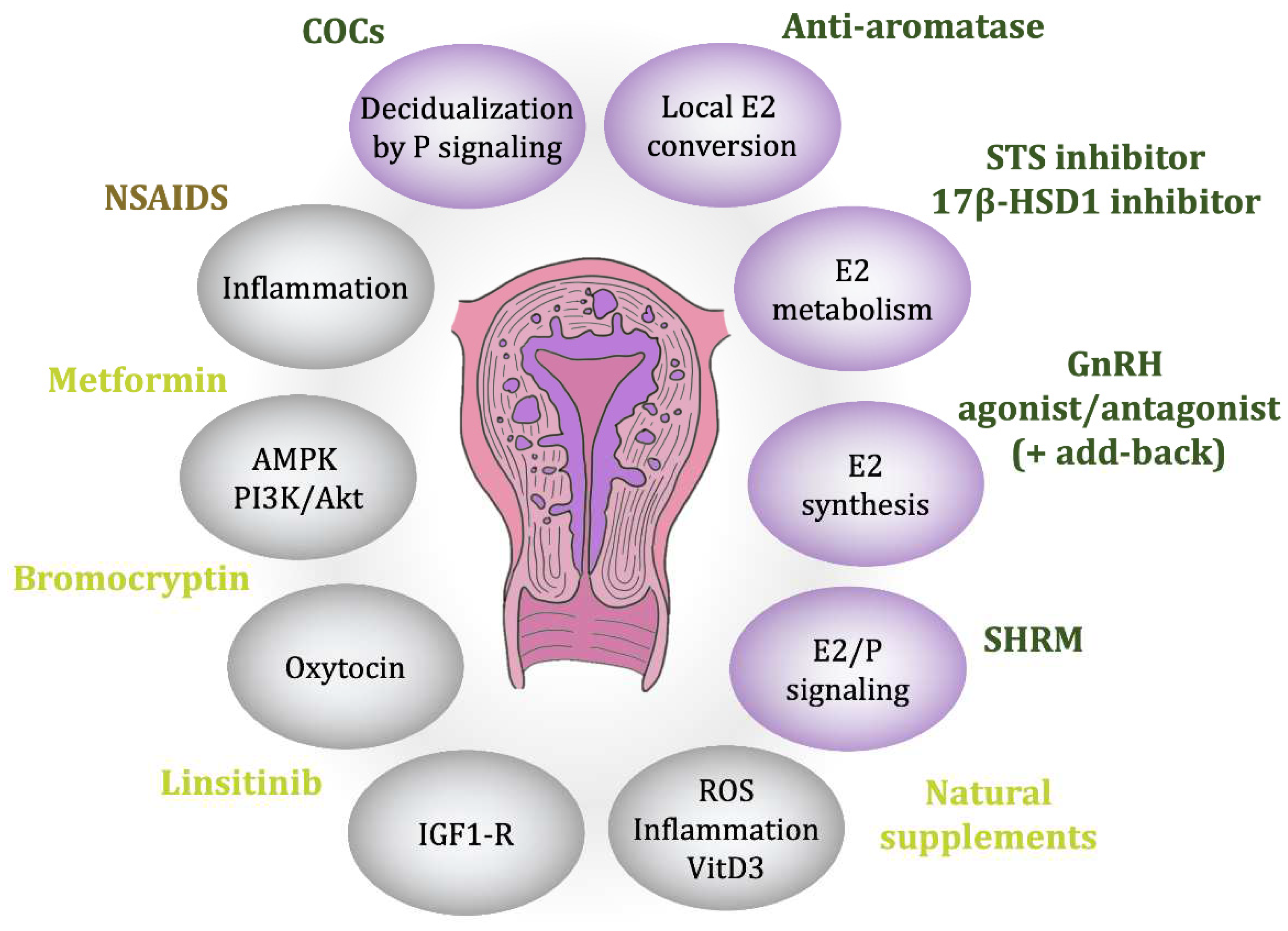
5. Therapeutics Options
5.1. Conventional Hormonal Treatments
5.1.1. Non-Steroidal Anti-Inflammatory Drugs
5.1.2. Combined Oral Contraceptives and Progestin
5.1.3. Gonadotropin-Releasing Hormone Agonists and Antagonists
5.1.4. Hormonal Targeting Therapies
Selective Hormonal Receptor Modulators
Aromatase Inhibitors
Sulfatase Inhibitors
17β-Hydroxysteroid Dehydrogenase Type 1 Inhibitors
5.2. Unconventional Treatments and Ongoing Research
5.2.1. Bromocriptine
5.2.2. Oxytocin Antagonists
5.2.3. Metformin
5.2.4. Linsitinib
5.2.5. Nutritional Supplements
Epigallactocatechin-3-Gallate
Vitamin D
Curcumin
Quercetin
6. Conclusion and Perspectives
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Upson, K.; Missmer, S.A. Epidemiology of Adenomyosis. Semin Reprod Med. 2020, 38, 089–107. [Google Scholar] [CrossRef]
- Taran, F.A.; Stewart, E.A.; Brucker, S. Adenomyosis: Epidemiology, Risk Factors, Clinical Phenotype and Surgical and Interventional Alternatives to Hysterectomy. Geburtshilfe Frauenheilkd. 2013, 73, 924–931. [Google Scholar] [CrossRef]
- Harada, T.; Taniguchi, F.; Harada, T. Increased Risk of Obstetric Complications in Patients with Adenomyosis: A Narrative Literature Review. Reprod Med Biol. 2022, 21. [Google Scholar] [CrossRef] [PubMed]
- Benagiano, G.; Brosens, I.; Habiba, M. Structural and Molecular Features of the Endomyometrium in Endometriosis and Adenomyosis. Hum. Reprod. Update. 2014, 20, 386–402. [Google Scholar] [CrossRef] [PubMed]
- Leyendecker, G.; Wildt, L.; Laschke, M.W.; Mall, G. Archimetrosis: The Evolution of a Disease and Its Extant Presentation: Pathogenesis and Pathophysiology of Archimetrosis (Uterine Adenomyosis and Endometriosis). Arch Gynecol Obstet. 2023, 307, 93–112. [Google Scholar] [CrossRef] [PubMed]
- Benagiano, G.; Brosens, I. History of Adenomyosis. Best Pract Res Clin Obstet Gynaecol. 2006, 20, 449–463. [Google Scholar] [CrossRef] [PubMed]
- Harada, T.; Khine, YM.; Kaponis, A.; Nikellis, T.; Decavalas, G.; Taniguchi, F.; Professor, A.; Doctor, T. The Impact of Adenomyosis on Women’s Fertility. Obstet Gynecol Surv. 2016, 71, 557–568. [Google Scholar] [CrossRef]
- Sam, M.; Raubenheimer, M.; Manolea, F.; Aguilar, H.; Mathew, R.P.; Patel, V.H.; Low, G. Accuracy of Findings in the Diagnosis of Uterine Adenomyosis on Ultrasound. Abdom. Radiol. 2020, 45, 842–850. [Google Scholar] [CrossRef]
- Van Den Bosch, T.; Dueholm, M.; Leone, F.P.G.; Valentin, L.; Rasmussen, C.K.; Votino, A.; Van Schoubroeck, D.; Landolfo, C.; Installé, A.J.F.; Guerriero, S.; Exacoustos, C.; Gordts, S.; Benacerraf, B.; D'Hooghe, T.; De Moor, B.; Brölmann, H.; Goldstein, S.; Epstein, E.; Bourne, T.; Timmerman, D. Terms, Definitions and Measurements to Describe Sonographic Features of Myometrium and Uterine Masses: A Consensus Opinion from the Morphological Uterus Sonographic Assessment (MUSA) Group. Ultrasound Obstet Gynecol. 2015, 46, 284–298. [Google Scholar] [CrossRef]
- Exacoustos, C.; Morosetti, G.; Conway, F.; Camilli, S.; Martire, F.G.; Lazzeri, L.; Piccione, E.; Zupi, E. New Sonographic Classification of Adenomyosis: Do Type and Degree of Adenomyosis Correlate to Severity of Symptoms? J Minim Invasive Gynecol. 2020, 27, 1308–1315. [Google Scholar] [CrossRef]
- Chapron, C.; Vannuccini, S.; Santulli, P.; Abrão, M.S.; Carmona, F.; Fraser, I.S.; Gordts, S.; Guo, S.W.; Just, P.A.; Noël, J.C.; Pistofidis, G.; Van den Bosch, T.; Petraglia, F. Diagnosing Adenomyosis: An Integrated Clinical and Imaging Approach. Hum. Reprod. Update. 2020, 26, 392–411. [Google Scholar] [CrossRef]
- Antero, M.F.; Ayhan, A.; Segars, J.; Shih, I.M. Pathology and Pathogenesis of Adenomyosis. Semin Reprod Med. 2020, 38, 108–118. [Google Scholar] [CrossRef]
- Kishi, Y.; Suginami, H.; Kuramori, R.; Yabuta, M.; Suginami, R.; Taniguchi, F. Four Subtypes of Adenomyosis Assessed by Magnetic Resonance Imaging and Their Specification. Am J Obstet Gynecol. 2012, 207, 114.e1–114.e7. [Google Scholar] [CrossRef]
- Moawad, G.; Fruscalzo, A.; Youssef, Y.; Kheil, M.; Tawil, T.; Nehme, J.; Pirtea, P.; Guani, B.; Afaneh, H.; Ayoubi, J.M.; Feki, A. Adenomyosis: An Updated Review on Diagnosis and Classification. J Clin Med. 2023, 12, 4828. [Google Scholar] [CrossRef]
- Vannuccini, S.; Luisi, S.; Tosti, C.; Sorbi, F.; Petraglia, F. Role of Medical Therapy in the Management of Uterine Adenomyosis. Fertil Steril. 2018, 109, 398–405. [Google Scholar] [CrossRef] [PubMed]
- Habiba, M.; Benagiano, G.; Guo, S.W. An Appraisal of the Tissue Injury and Repair (TIAR) Theory on the Pathogenesis of Endometriosis and Adenomyosis. Biomolecules. 2023, 13, 975. [Google Scholar] [CrossRef] [PubMed]
- Zhai, J.; Vannuccini, S.; Petraglia, F.; Giudice, L.C. Adenomyosis: Mechanisms and Pathogenesis. Semin Reprod Med. 2020, 38, 129–143. [Google Scholar] [CrossRef] [PubMed]
- Bruner-Tran, K.L.; Duleba, A.J.; Taylor, H.S.; Osteen, K.G. Developmental Toxicant Exposure Is Associated with Transgenerational Adenomyosis in a Murine Model. Biol Reprod. 2016, 95, 73. [Google Scholar] [CrossRef]
- Donnez, J.; Donnez, O.; Dolmans, M.M. Introduction: Uterine Adenomyosis, Another Enigmatic Disease of Our Time. Fertil Steril. 2018, 109, 369–370. [Google Scholar] [CrossRef]
- García-Solares, J.; Donnez, J.; Donnez, O.; Dolmans, M.M. Pathogenesis of Uterine Adenomyosis: Invagination or Metaplasia? Fertil Steril. 2018, 109, 371–379. [Google Scholar] [CrossRef]
- Huang, J.H.; Duan, H.; Wang, S.; Wang, Y.Y. Estrogen 17βestradiol Accelerates the Proliferation of Uterine Junctional Zone Smooth Muscle Cells via the Let7a/Lin28B Axis in Adenomyosis. Mol Med Rep. 2021, 23, 337. [Google Scholar] [CrossRef] [PubMed]
- Leyendecker, G.; Wildt, L.; Leyendecker, G. A New Concept of Endometriosis and Adenomyosis: Tissue Injury and Repair (TIAR). Horm Mol Biol Clin Investig. 2011, 5, 125–142. [Google Scholar] [CrossRef] [PubMed]
- Niu, W.; Zhang, Y.; Liu, H.; Liang, N.; Xu, L.; Li, Y.; Yao, W.; Shi, W.; Liu, Z. Single-Cell Profiling Uncovers the Roles of Endometrial Fibrosis and Microenvironmental Changes in Adenomyosis. J Inflamm Res. 2023, 16, 1949–1965. [Google Scholar] [CrossRef] [PubMed]
- Hao, M.; Liu, X.; Guo, S.W. Adenomyosis in Mice Resulting from Mechanically or Thermally Induced Endometrial–Myometrial Interface Disruption and Its Possible Prevention. Reprod Biomed Online. 2020, 41, 925–942. [Google Scholar] [CrossRef]
- Rossi, M.; Vannuccini, S.; Capezzuoli, T.; Fambrini, M.; Vannuzzi, V.; Donati, C.; Petraglia, F. Mechanisms and Pathogenesis of Adenomyosis. Curr Obstet Gynecol Rep. 2022, 11, 95–102. [Google Scholar] [CrossRef]
- Khan, K.N.; Fujishita, A.; Mori, T. Pathogenesis of Human Adenomyosis: Current Understanding and Its Association with Infertility. J Clin Med. 2022, 11, 4057. [Google Scholar] [CrossRef]
- Khan, K.N.; Kitajima, M.; Inoue, T.; Tateishi, S.; Fujishita, A.; Nakashima, M.; Masuzaki, H. Additive Effects of Inflammation and Stress Reaction on Toll-like Receptor 4-Mediated Growth of Endometriotic Stromal Cells. Hum. Reprod. 2013, 28, 2794–2803. [Google Scholar] [CrossRef]
- Khan, K.N.; Kitajima, M.; Imamura, T.; Hiraki, K.; Fujishita, A.; Sekine, I.; Ishimaru, T.; Masuzaki, H. Toll-like Receptor 4-Mediated Growth of Endometriosis by Human Heat-Shock Protein 70. Hum. Reprod. 2008, 23, 2210–2219. [Google Scholar] [CrossRef]
- Guo, S.W. The Pathogenesis of Adenomyosis Vis-à-Vis Endometriosis. J Clin Med. 2020, 9, 485. [Google Scholar] [CrossRef]
- Bergeron, C.; Amant, F.; Ferenczy, A. Pathology and Physiopathology of Adenomyosis. Best Pract Res Clin Obstet Gynaecol. 2006, 20, 511–521. [Google Scholar] [CrossRef]
- Spencer, T.E.; Hayashi, K.; Hu, J.; Carpenter, K.D. Comparative Developmental Biology of the Mammalian Uterus. Curr Top Develop Biol. 2005, 68, 85–122. [Google Scholar] [CrossRef]
- Spencer, T.E.; Dunlap, K.A.; Filant, J. Comparative Developmental Biology of the Uterus: Insights into Mechanisms and Developmental Disruption. Mol. Cell. Endocrinol. 2012, 354, 34–53. [Google Scholar] [CrossRef]
- Gargett, C.E.; Nguyen, H.P.T.; Ye, L. Endometrial Regeneration and Endometrial Stem/Progenitor Cells. Rev Endocr Metab Disord. 2012, 13, 235–251. [Google Scholar] [CrossRef]
- Gargett, C.E. Uterine Stem Cells: What Is the Evidence? Hum. Reprod. Update. 2007, 13, 87–101. [Google Scholar] [CrossRef]
- Gurung, S.; Deane, J.A.; Masuda, H.; Maruyama, T.; Gargett, C.E. Stem Cells in Endometrial Physiology. Semin Reprod Med. 2015, 33, 326–332. [Google Scholar] [CrossRef]
- Hufnagel, D.; Li, F.; Cosar, E.; Krikun, G.; Taylor, H.S. The Role of Stem Cells in the Etiology and Pathophysiology of Endometriosis. Semin Reprod Med. 2015, 33, 333–340. [Google Scholar] [CrossRef]
- Vannuccini, S.; Tosti, C.; Carmona, F.; Huang, S.J.; Chapron, C.; Guo, S.W.; Petraglia, F. Pathogenesis of Adenomyosis: An Update on Molecular Mechanisms. Reprod BioMed Online. 2017, 35, 592–601. [Google Scholar] [CrossRef] [PubMed]
- Chapron, C.; Tosti, C.; Marcellin, L.; Bourdon, M.; Lafay-Pillet, M.C.; Millischer, A.E.; Streuli, I.; Borghese, B.; Petraglia, F.; Santulli, P. Relationship between the Magnetic Resonance Imaging Appearance of Adenomyosis and Endometriosis Phenotypes. Hum. Reprod. 2017, 32, 1393–1401. [Google Scholar] [CrossRef] [PubMed]
- Khan, K.N.; Fujishita, A.; Koshiba, A.; Kuroboshi, H.; Mori, T.; Ogi, H.; Itoh, K.; Nakashima, M.; Kitawaki, J. Biological Differences between Intrinsic and Extrinsic Adenomyosis with Coexisting Deep Infiltrating Endometriosis. Reprod Biomed Online. 2019, 39, 343–353. [Google Scholar] [CrossRef] [PubMed]
- Takahashi, K.; Nagata, H.; Kitao, M. Clinical Usefulness of Determination of Estradiol Level in the Menstrual Blood for Patients with Endometriosis. Nihon Sanka Fujinka Gakkai Zasshi. 1989, 41, 1849–1850. [Google Scholar]
- Yamamoto, T.; Noguchi, T.; Tamura, T.; Kitawaki, J.; Okada, H. Evidence for Estrogen Synthesis in Adenomyotic Tissues. Am J Obstet Gynecol. 1993, 169, 734–738. [Google Scholar] [CrossRef] [PubMed]
- Bulun, S.E.; Lin, Z.; Imir, G.; Amin, S.; Demura, M.; Yilmaz, B.; Martin, R.; Utsunomiya, H.; Thung, S.; Gurates, B.; Tamura, M.; Langoi, D.; Deb, S. Regulation of Aromatase Expression in Estrogen-Responsive Breast and Uterine Disease: From Bench to Treatment. Pharmacological Reviews. 2005, 57, 359–383. [Google Scholar] [CrossRef] [PubMed]
- Purohit, A.; Reed, M.J. Regulation of Estrogen Synthesis in Postmenopausal Women. Steroids. 2002, 67, 979–983. [Google Scholar] [CrossRef]
- Kitawaki, J. Adenomyosis: The Pathophysiology of an Oestrogen-Dependent Disease. Best Pract Res Clin Obstet Gynaecol. 2006, 20, 493–502. [Google Scholar] [CrossRef]
- Kitawaki, J.; Noguchi, T.; Amatsu, T.; Maeda, K.; Tsukamoto, K.; Yamamoto, T.; Fushiki, S.; Osawa, Y.; Honjo, H. Expression of Aromatase Cytochrome P450 Protein and Messenger Ribonucleic Acid in Human Endometriotic and Adenomyotic Tissues but Not in Normal Endometrium. Biol Reprod. 1997, 57, 514–519. [Google Scholar] [CrossRef]
- Colette, S.; Lousse, J.C.; Defrère, S.; Curaba, M.; Heilier, J.F.; Van Langendonckt, A.; Mestdagt, M.; Foidart, J.M.; Loumaye, E.; Donnez, J. Absence of Aromatase Protein and MRNA Expression in Endometriosis. Hum. Reprod. 2009, 24, 2133–2141. [Google Scholar] [CrossRef]
- Sharma, S.; Roychoudhury, S.; Padmaja Bhattacharya, M.; Hazra, S.; Majhi, A.K.; Oswal, K.C.; Chattopadhyay, R. Low-Dose Letrozole À an Effective Option for Women with Symptomatic Adenomyosis Awaiting IVF: A Pilot Randomized Controlled Trial. Reprod Biomed Online. 2023, 47, 84–93. [Google Scholar] [CrossRef]
- Heinosalo, T.; Rytkönen, K.T.; Saarinen, N.; Järvensivu, P.; Damdimopoulou, P.; Strauss, L.; Orasniemi, S.; Horshauge, P.; Gabriel, M.; Koskimies, P.; Ohlsson, C.; Kronqvist, P.; Poutanen, M. Overexpression of Human Estrogen Biosynthetic Enzyme Hydroxysteroid (17beta) Dehydrogenase Type 1 Induces Adenomyosis-like Phenotype in Transgenic Mice. Int J Mol Sci. 2022, 23. [Google Scholar] [CrossRef]
- Kitawaki, J.O.; Koshiba, H.; Ishihara, H.; Kusuki, I.; Tsukamoto, K.; Honjo, H. Progesterone Induction of 17-Hydroxysteroid Dehydrogenase Type 2 during the Secretory Phase Occurs in the Endometrium of Estrogen-Dependent Benign Diseases But Not in Normal Endometrium. J Clin Endocrinol Metab. 2000, 85, 3292–3296. [Google Scholar] [CrossRef]
- Urabe, M.; Yamamoto, T.; Kitawaki, J.; Honjo, H.; Okada, H. Estrogen Biosynthesis in Human Uterine Adenomyosis. Acta Endocrinol (Copenh). 1989, 121, 259–264. [Google Scholar] [CrossRef] [PubMed]
- Hewitt, S.C.; Winuthayanon, W.; Korach, K.S. What’s New in Estrogen Receptor Action in the Female Reproductive Tract. J Mol Endocrinol. 2016, 56, 55–71. [Google Scholar] [CrossRef]
- Oehler, M.K.; Greschik, H.; Fischer, D.C.; Tong, X.; Schuele, R.; Kieback, D.G. Functional Characterization of Somatic Point Mutations of the Human Estrogen Receptor α (HERα) in Adenomyosis Uteri. Mol. Hum. Reprod. 2004, 10, 853–860. [Google Scholar] [CrossRef]
- Yu, K.; Huang, Z.Y.; Xu, X.L.; Li, J.; Fu, X.W.; Deng, S.L. Estrogen Receptor Function: Impact on the Human Endometrium. Front Endocrinol (Lausanne). 2022, 13. [Google Scholar] [CrossRef]
- Kitawaki, J.; Obayashi, H.; Ishihara, H.; Koshiba, H.; Kusuki, I.; Kado, N.; Tsukamoto, K.; Hasegawa, G.; Nakamura, N.; Honjo, H. Oestrogen Receptor-Alpha Gene Polymorphism Is Associated with Endometriosis, Adenomyosis and Leiomyomata. Hum. Reprod. 2001, 16, 51–55. [Google Scholar] [CrossRef] [PubMed]
- Sztachelska, M.; Ponikwicka-Tyszko, D.; Martínez-Rodrigo, L.; Bernaczyk, P.; Palak, E.; Półchłopek, W.; Bielawski, T.; Wołczyński, S. Functional Implications of Estrogen and Progesterone Receptors Expression in Adenomyosis, Potential Targets for Endocrinological Therapy. J Clin Med. 2022, 11. [Google Scholar] [CrossRef]
- Hong, D.G.; Park, J.Y.; Chong, G.O.; Lee, Y.H.; Lee, H.J.; Shinn, J.U.; Lee, Y.S.; Seong, W.J. Transmembrane G Protein-Coupled Receptor 30 Gene Polymorphisms and Uterine Adenomyosis in Korean Women. Gynecol. Endocrinol. 2019, 35, 498–501. [Google Scholar] [CrossRef] [PubMed]
- Wallach, E.E.; Editor, A.; Nisolle, M.; Donnez, J. Modern Trends Peritoneal Endometriosis, Ovarian Endometriosis, and Adenomyotic Nodules of the Rectovaginal Septum Are Three Different Entities; American Society for Reproductive Medicine. 1997, 68.
- Mehasseb, M.K.; Panchal, R.; Taylor, A.H.; Brown, L.; Bell, S.C.; Habiba, M. Estrogen and Progesterone Receptor Isoform Distribution through the Menstrual Cycle in Uteri with and without Adenomyosis. Fertil Steril. 2011, 95. [Google Scholar] [CrossRef] [PubMed]
- Mori, T.; Nagasawa, H.; Takahashi, S. The induction of adenomyosis in mice by intrauterine pituitary isografts. Life Sci. 1981, 29, 1277–1282. [Google Scholar] [CrossRef] [PubMed]
- Singtripop, T.; Mori, T.; Park, MK.; Sakamoto, S.; Kawashima, S. Development of Uterine Adenomyosis after Treatment with Dopamine Antagonists in Mice. Life Sci. 1991, 49, 201–206. [Google Scholar] [CrossRef] [PubMed]
- Łupicka, M.; Socha, B.M.; Szczepańska, A.A.; Korzekwa, A.J. Prolactin Role in the Bovine Uterus during Adenomyosis. Domest Anim Endocrinol 2017, 58, 1–13. [Google Scholar] [CrossRef]
- Nagasawa, H.; Mori, T. Stimulation of mammary tumorigenesis and suppression of uterine adenomyosis by temporary inhibition of pituitary prolactin secretion during youth in mice (41492). Proc Soc Exp Biol Med. 1982, 171, 164–167. [Google Scholar] [CrossRef]
- Sengupta, P.; Sharma, A.; Mazumdar, G.; Banerjee, I.; Tripathi, S.K.; Bagchi, C.; Das, N. The Possible Role of Fluoxetine in Adenomyosis: An Animal Experiment with Clinical Correlations. J. Clin. Diagnostic Res. 2013, 7, 1530–1534. [Google Scholar] [CrossRef] [PubMed]
- Mori, T.; Ohta, Y.; Nagasawa, H. Ultrastructural changes in uterine myometrium of mice with experimentally-induced adenomyosis. Experientia. 1984, 40, 1385–1387. [Google Scholar] [CrossRef] [PubMed]
- Marquardt, R.M.; Jeong, J.W.; Fazleabas, A.T. Animal Models of Adenomyosis. Semin Reprod Med 2020, 38, 168–178. [Google Scholar] [CrossRef] [PubMed]
- Andersson, J.K.; Khan, Z.; Weaver, A.L.; Vaughan, L.E.; Gemzell-Danielsson, K.; Stewart, E.A. Vaginal Bromocriptine Improves Pain, Menstrual Bleeding and Quality of Life in Women with Adenomyosis: A Pilot Study. Acta Obstet Gynecol Scand. 2019, 98, 1341–1350. [Google Scholar] [CrossRef] [PubMed]
- Takemura, M.; Nomura, S.; Kimura, T.; Inoue, T.; Onoue, H.; Azuma, C.; Saji, F.; Kitamura, Y.; Tanizawa, O. Expression and localization of oxytocin receptor gene in human uterine endometrium in relation to the menstrual cycle. Endocrinology. 1993, 132, 1830–1835. [Google Scholar] [CrossRef]
- Mechsner, S.; Grum, B.; Gericke, C.; Loddenkemper, C.; Dudenhausen, J.W.; Ebert, A.D. Possible Roles of Oxytocin Receptor and Vasopressin-1α Receptor in the Pathomechanism of Dysperistalsis and Dysmenorrhea in Patients with Adenomyosis Uteri. Fertil Steril. 2010, 94, 2541–2546. [Google Scholar] [CrossRef]
- Zhang, Y.; Yu, P.; Sun, F.; Li, T.C.; Cheng, J.M.; Duan, H. Expression of Oxytocin Receptors in the Uterine Junctional Zone in Women with Adenomyosis. Acta Obstet Gynecol Scand. 2015, 94, 412–418. [Google Scholar] [CrossRef] [PubMed]
- Guo, S.W.; Mao, X.; Ma, Q.; Liu, X. Dysmenorrhea and Its Severity Are Associated with Increased Uterine Contractility and Overexpression of Oxytocin Receptor (OTR) in Women with Symptomatic Adenomyosis. Fertil Steril. 2013, 99, 231–240. [Google Scholar] [CrossRef]
- Sekulovski, N.; Whorton, A.E.; Shi, M.; Hayashi, K.; MacLean, J.A. Insulin Signaling Is an Essential Regulator of Endometrial Proliferation and Implantation in Mice. FASEB Journal. 2021, 35. [Google Scholar] [CrossRef]
- Konopka, B.; Skasko, E.; Kluska, A.; Goluda, M.; Janiec-Jankowska, A.; Paszko, Z.; Ujec, M. Changes in the concentrations of receptors of insulin-like growth factor-I, epithelial growth factor, oestrogens and progestagens in adenomyosis foci, endometrium and myometrium of women during menstrual cycle. Eur J Gynaecol Oncol. 1998, 19, 93–97. [Google Scholar]
- Artymuk, N.; Zotova, O.; Gulyaeva, L. Adenomyosis: Genetics of Estrogen Metabolism. Horm Mol Biol Clin Investig. 2019, 37. [Google Scholar] [CrossRef]
- Inoue, S.; Hirota, Y.; Ueno, T.; Fukui, Y.; Yoshida, E.; Hayashi, T.; Kojima, S.; Takeyama, R.; Hashimoto, T.; Kiyono, T. Uterine Adenomyosis Is an Oligoclonal Disorder Associated with KRAS Mutations. Nat Commun. 2019, 10. [Google Scholar] [CrossRef]
- Bulun, S.E.; Yildiz, S.; Adli, M.; Wei, J.J. Adenomyosis Pathogenesis: Insights from next-Generation Sequencing. Hum. Reprod. Update. 2021, 27, 1086–1097. [Google Scholar] [CrossRef]
- Liu, X.; Guo, S.W. Aberrant Immunoreactivity of Deoxyribonucleic Acid Methyltransferases in Adenomyosis. Gynecol Obstet Invest. 2012, 74, 100–108. [Google Scholar] [CrossRef]
- Cedar, H.; Bergman, Y. Linking DNA Methylation and Histone Modification: Patterns and Paradigms. Nat Rev Genet. 2009, 10, 295–304. [Google Scholar] [CrossRef] [PubMed]
- Nie, J.; Liu, X.; Guo, S.W. Promoter Hypermethylation of Progesterone Receptor Isoform B (PR-B) in Adenomyosis and Its Rectification by a Histone Deacetylase Inhibitor and a Demethylation Agent. Reprod Sci. 2010, 17, 995–1005. [Google Scholar] [CrossRef]
- Liu, X.; Nie, J.; Guo, S.W. Elevated Immunoreactivity against Class i Histone Deacetylases in Adenomyosis. Gynecol Obstet Invest. 2012, 74, 50–55. [Google Scholar] [CrossRef] [PubMed]
- Liu, X.; Guo, S.W. Valproic Acid Alleviates Generalized Hyperalgesia in Mice with Induced Adenomyosis. J. Obstet. Gynaecol. Res. 2011, 37, 696–708. [Google Scholar] [CrossRef] [PubMed]
- Zhai, J.; Li, S.; Sen, S.; Opoku-Anane, J.; Du, Y.; Chen, Z.J.; Giudice, L.C. M6A RNA Methylation Regulators Contribute to Eutopic Endometrium and Myometrium Dysfunction in Adenomyosis. Front. Genet. 2020, 11. [Google Scholar] [CrossRef]
- Templeman, C.; Marshall, S.F.; Ursin, G.; Horn-Ross, P.L.; Clarke, C.A.; Allen, M.; Deapen, D.; Ziogas, A.; Reynolds, P.; Cress, R.; Anton-Culver, H.; West, D.; Ross, R.K.; Bernstein, L. Adenomyosis and Endometriosis in the California Teachers Study. Fertil Steril. 2008, 90, 415–424. [Google Scholar] [CrossRef]
- Parazzini, F.; Vercellini, P.; Panazza, S.; Chatenoud, L.; Oldani, S. Crosignani PG. Risk factors for adenomyosis. Hum. Reprod. 1997, 12, 1275–1279. [Google Scholar] [CrossRef]
- Parazzini, F.; Mais, V.; Cipriani, S.; Busacca, M.; Venturini, P. Determinants of Adenomyosis in Women Who Underwent Hysterectomy for Benign Gynecological Conditions: Results from a Prospective Multicentric Study in Italy. Eur. J. Obstet. Gynecol. Reprod. Biol. 2009, 143, 103–106. [Google Scholar] [CrossRef]
- Cohen, I.; Beyth, Y.; Shapira, J.; Tepper, R.; Fishman, A.; Cordoba, M.; Bernheim, J.; Yigael, D.; Altaras, MM. High frequency of adenomyosis in postmenopausal breast cancer patients treated with tamoxifen. Gynecol Obstet Invest. 1997 44, 200–205. [CrossRef]
- Jin, Z.; Wu, X.; Liu, H.; Xu, C. Celecoxib, a Selective COX2 Inhibitor, Markedly Reduced the Severity of Tamoxifeninduced Adenomyosis in a Murine Model. Exp Ther Med. 2020. [Google Scholar] [CrossRef] [PubMed]
- Shen, M.; Liu, X.; Zhang, H.; Guo, S.W. Transforming Growth Factor Β1 Signaling Coincides with Epithelial-Mesenchymal Transition and Fibroblast-to-Myofibroblast Transdifferentiation in the Development of Adenomyosis in Mice. Hum. Reprod. 2016, 31, 355–369. [Google Scholar] [CrossRef]
- Rumph, J.T.; Stephens, V.R.; Archibong, A.E.; Osteen, K.G.; Bruner-Tran, K.L. Environmental Endocrine Disruptors and Endometriosis. In Advances in Anatomy Embryology and Cell Biology; Springer Science and Business Media Deutschland GmbH. 2020; Vol. 232, pp 57–78. [CrossRef]
- Cho, Y.J.; Yun, J.H.; Kim, S.J.; Kwon, H.Y. Nonpersistent Endocrine Disrupting Chemicals and Reproductive Health of Women. Obstet Gynecol Sci. 2020, 63, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Stephens, V.R.; Rumph, J.T.; Ameli, S.; Bruner-Tran, K.L.; Osteen, K.G. The Potential Relationship Between Environmental Endocrine Disruptor Exposure and the Development of Endometriosis and Adenomyosis. Front. Physiol. 2022, 28. [Google Scholar] [CrossRef]
- Schug, T.T.; Johnson, A.F.; Birnbaum, L.S.; Colborn, T.; Guillette, L.J.; Crews, D.P.; Collins, T.; Soto, A.M.; Vom Saal, F.S.; McLachlan, J. A Sonnenschein, C.; Heindel, JJ.; Minireview: Endocrine Disruptors: Past Lessons and Future Directions. Mol Endocrinol 2016, 30, 833–847. [Google Scholar] [CrossRef]
- Signorile, P.G.; Spugnini, E.P.; Mita, L.; Mellone, P.; D’Avino, A.; Bianco, M.; Diano, N.; Caputo, L.; Rea, F.; Viceconte, R.; Portaccio, M.; Viggiano, E.; Citro, G.; Pierantoni, R.; Sica, V.; Vincenzi, B.; Mita, D.G.; Baldi, F.; Baldi, A. Pre-Natal Exposure of Mice to Bisphenol A Elicits an Endometriosis-like Phenotype in Female Offspring. Gen Comp Endocrinol. 2010, 168, 318–325. [Google Scholar] [CrossRef] [PubMed]
- Jefferson, W.N.; Karimi Kinyamu, H.; Wang, T.; Miranda, A.X.; Padilla-Banks, E.; Suen, A.A.; Williams, C.J. Widespread Enhancer Activation via ERα Mediates Estrogen Response in Vivo during Uterine Development. Nucleic Acids Res. 2018, 46, 5487–5503. [Google Scholar] [CrossRef]
- Liu, X.; Ding, D.; Shen, M.; Yan, D.; Guo, S.W. Shorter Anogenital Distance in Women with Ovarian Endometriomas and Adenomyosis, but Not Uterine Leiomyomas. Biomedicines. 2023, 11. [Google Scholar] [CrossRef] [PubMed]
- Louis, G.M.; Peterson, C.M.; Chen, Z.; Hediger, ML.; Croughan, M.S.; Sundaram, R.; Stanford, J.B.; Fujimoto, V.Y.; Varner, M.W.; Giudice, L.C.; Kennedy, A.; Sun, L.; Wu, Q.; Kannan, K. Perfluorochemicals and endometriosis: The ENDO study. Epidemiology. 2012, 23, 799–805. [Google Scholar] [CrossRef] [PubMed]
- Matta, K.; Lefebvre, T.; Vigneau, E.; Cariou, V.; Marchand, P.; Guitton, Y.; Royer, A.L.; Ploteau, S.; Le Bizec, B.; Antignac, J.P.; Cano-Sancho, G. Associations between Persistent Organic Pollutants and Endometriosis: A Multiblock Approach Integrating Metabolic and Cytokine Profiling. Environ Int. 2022, 158. [Google Scholar] [CrossRef]
- Rier, S.E.; Martin, D.C.; Bowman, R.E.; Dmowski, W.P.; Becker, J.L. Endometriosis in rhesus monkeys (Macaca mulatta) following chronic exposure to 2,3,7,8-tetrachlorodibenzo-p-dioxin. Fundam Appl Toxicol. 1993 21, 433–441. [CrossRef]
- Szántó, M.; Gupte, R.; Kraus, W.L.; Pacher, P.; Bai, P. PARPs in lipid metabolism and related diseases. Prog Lipid Res. 2021, 84, 101117. [Google Scholar] [CrossRef] [PubMed]
- Bruner-Tran, K.L.; Gnecco, J.; Ding, T.; Glore, D.R.; Pensabene, V.; Osteen, K.G. Exposure to the Environmental Endocrine Disruptor TCDD and Human Reproductive Dysfunction: Translating Lessons from Murine Models. Reprod. Toxicol. 2017, 68, 59–71. [Google Scholar] [CrossRef]
- Huang, P.C.; Tsai, E.M.; Li, W.F.; Liao, P.C.; Chung, M.C.; Wang, Y.H.; Wang, S.L. Association between Phthalate Exposure and Glutathione S-Transferase M1 Polymorphism in Adenomyosis, Hum. Reprod. 2010 Apr;25:986-94. [CrossRef]
- Skinner, M.K.; Manikkam, M.; Guerrero-Bosagna, C. Epigenetic Transgenerational Actions of Environmental Factors in Disease Etiology. Trends Endocrinol. Metab. 2010, 21, 214–222. [Google Scholar] [CrossRef]
- Alavian-Ghavanini, A.; Rüegg, J. Understanding Epigenetic Effects of Endocrine Disrupting Chemicals: From Mechanisms to Novel Test Methods. Basic Clin Pharmacol Toxicol. 2018 122, 38–45. [CrossRef]
- Czernych, R.; Chraniuk, M.; Zagożdżon, P.; Wolska, L. Characterization of Estrogenic and Androgenic Activity of Phthalates by the XenoScreen YES/YAS in Vitro Assay. Environ Toxicol Pharmacol. 2017, 53, 95–104. [Google Scholar] [CrossRef]
- Reddy, B.S.; Rozati, R.; Reddy, S.; Kodampur, S.; Reddy, P.; Reddy, R. High Plasma Concentrations of Polychlorinated Biphenyls and Phthalate Esters in Women with Endometriosis: A Prospective Case Control Study. Fertil Steril. 2006, 85, 775–779. [Google Scholar] [CrossRef]
- Reddy, B.S.; Rozati, R.; Reddy, B.V.R.; Raman, N.V.V.S.S. Association of Phthalate Esters with Endometriosis in Indian Women. BJOG 2006, 113, 515–520. [Google Scholar] [CrossRef]
- Upson, K.; Sathyanarayana, S.; De Roos, A.J.; Thompson, M. Lou; Scholes, D.; Dills, R.; Holt, V.L. Phthalates and Risk of Endometriosis. Environ Res. 2013, 126, 91–97. [Google Scholar] [CrossRef] [PubMed]
- Huang, P.C.; Li, W.F.; Liao, P.C.; Sun, C.W.; Tsai, E.M.; Wang, S.L. Risk for Estrogen-Dependent Diseases in Relation to Phthalate Exposure and Polymorphisms of CYP17A1 and Estrogen Receptor Genes. Environ. Sci. Pollut. Res. 2014, 21, 13964–13973. [Google Scholar] [CrossRef] [PubMed]
- Aldad, T.S.; Rahmani, N.; Leranth, C.; Taylor, H.S. Bisphenol-A Exposure Alters Endometrial Progesterone Receptor Expression in the Nonhuman Primate. Fertil Steril. 2011, 96, 175–179. [Google Scholar] [CrossRef] [PubMed]
- Newbold, R.R.; Jefferson, W.N.; Padilla-Banks, E. Long-Term Adverse Effects of Neonatal Exposure to Bisphenol A on the Murine Female Reproductive Tract. Reprod. Toxicol. 2007, 24, 253–258. [Google Scholar] [CrossRef] [PubMed]
- Giusti, R.M.; Iwamoto, K.; Hatch, E.E. Diethylstilbestrol revisited: A review of the long-term health effects. Ann Intern Med. 1995, 122, 778–788. [Google Scholar] [CrossRef]
- Upson, K.; Sathyanarayana, S.; Scholes, D.; Holt, V.L. Early-Life Factors and Endometriosis Risk. Fertil Steril. 2015, 104, 964–971. [Google Scholar] [CrossRef] [PubMed]
- Huseby, R.A.; Thurlow, S. Effects of Prenatal Exposure of Mice to “Low-Dose” Diethylstilbestrol and the Development of Adenomyosis Associated with Evidence of Hyperprolactinemia. Am J Obstet Gynecol. 1982, 144, 939–949. [Google Scholar] [CrossRef] [PubMed]
- McLachlan, J.A.; Newbold, R.R.; Bullock, B.C. Long-term effects on the female mouse genital tract associated with prenatal exposure to diethylstilbestrol. Cancer Res. 1980, 40, 3988–3999. [Google Scholar] [PubMed]
- Domínguez-López, I.; Yago-Aragón, M.; Salas-Huetos, A.; Tresserra-Rimbau, A.; Hurtado-Barroso, S. Effects of Dietary Phytoestrogens on Hormones throughout a Human Lifespan: A Review. Nutrients. 2020, 12, 1–25. [Google Scholar] [CrossRef] [PubMed]
- Youseflu, S.; Sadatmahalleh, S.J.; Mottaghi, A.; Kazemnejad, A. Dietary Phytoestrogen Intake and the Risk of Endometriosis in Iranian Women: A Case-Control Study. Int J Fertil Steril. 2020, 13, 296–300. [Google Scholar] [CrossRef] [PubMed]
- Kouzmenko, A.; Ohtake, F.; Fujiki, R.; Kato, S. Hormonal Gene Regulation through DNA Methylation and Demethylation. Epigenomics. 2010 2, 765–774. [CrossRef]
- Bruner-Tran, K.L.; Resuehr, D.; Ding, T.; Lucas, J.A.; Osteen, K.G. The Role of Endocrine Disruptors in the Epigenetics of Reproductive Disease and Dysfunction: Potential Relevance to Humans. Curr Obstet Gynecol Rep. 2012, 1, 116–123. [Google Scholar] [CrossRef] [PubMed]
- Derghal, A.; Djelloul, M.; Trouslard, J.; Mounien, L. An Emerging Role of Micro-RNA in the Effect of the Endocrine Disruptors. Front. Neurosci. 2016, 10, 318. [Google Scholar] [CrossRef] [PubMed]
- Liu, J.; Zhang, L.; Winterroth, L.C.; Garcia, M.; Weiman, S.; Wong, J.W.; Sunwoo, J.B.; Nadeau, K.C. Epigenetically Mediated Pathogenic Effects of Phenanthrene on Regulatory T Cells. J Toxicol. 2013, 2013. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Zhang, Y.; Tao, S.; Guan, Y.; Zhang, T.; Wang, Z. Global DNA Methylation in Gonads of Adult Zebrafish Danio Rerio under Bisphenol A Exposure. Ecotoxicol Environ Saf. 2016, 130, 124–132. [Google Scholar] [CrossRef]
- Jefferson, W.N.; Chevalier, D.M.; Phelps, J.Y.; Cantor, A.M.; Padilla-Banks, E.; Newbold, R.R.; Archer, T.K.; Karimi Kinyamu, H.; Williams, C.J. Persistently Altered Epigenetic Marks in the Mouse Uterus after Neonatal Estrogen Exposure. Mol. Endocrinol. 2013, 27, 1666–1677. [Google Scholar] [CrossRef]
- Nayyar, T.; Bruner-Tran, K.L.; Piestrzeniewicz-Ulanska, D.; Osteen, K.G. Developmental Exposure of Mice to TCDD Elicits a Similar Uterine Phenotype in Adult Animals as Observed in Women with Endometriosis. Reprod. Toxicol. 2007, 23, 326–336. [Google Scholar] [CrossRef]
- La Merrill, M.A.; Vandenberg, L.N.; Smith, M.T.; Goodson, W.; Browne, P.; Patisaul, H.B.; Guyton, K.Z.; Kortenkamp, A.; Cogliano, V.J.; Woodruff, T.J.; Rieswijk, L.; Sone, H.; Korach, K.S.; Gore, A.C.; Zeise, L.; Zoeller, RT. Consensus on the Key Characteristics of Endocrine-Disrupting Chemicals as a Basis for Hazard Identification. Nat Rev Endocrinol. 2020, 16, 45–57. [Google Scholar] [CrossRef]
- Routledge, E.J.; White, R.; Parker, M.G.; Sumpter, J.P. Differential Effects of Xenoestrogens on Coactivator Recruitment by Estrogen Receptor (ER) α and ERβ. J Biol Chem. 2000, 275, 35986–35993. [Google Scholar] [CrossRef]
- Clinical trial: Adenomyosis and ART (ADENOFERT). Available online: https://clinicaltrials.gov/study/NCT05937490 (accessed on 18 January 2024).
- Moawad, G.; Kheil, M.H.; Ayoubi, J.M.; Klebanoff, J.S.; Rahman, S.; Sharara, F.I. Adenomyosis and Infertility. Journal of Assisted Reproduction and Genetics. 2022, 39, 1027–1031. [Google Scholar] [CrossRef]
- Tan, J.; Moriarty, S.; Taskin, O.; Allaire, C.; Williams, C.; Yong, P.; Bedaiwy, M.A. Reproductive Outcomes after Fertility-Sparing Surgery for Focal and Diffuse Adenomyosis: A Systematic Review. J Minim Invasive Gyneco. 2018, 25, 608–621. [Google Scholar] [CrossRef]
- Vercellini, P.; Viganò, P.; Bandini, V.; Buggio, L.; Berlanda, N.; Somigliana, E. Association of Endometriosis and Adenomyosis with Pregnancy and Infertility. Fertil Steril. 2023, 119, 727–740. [Google Scholar] [CrossRef] [PubMed]
- Szubert, M.; Koziróg, E.; Olszak, O.; Krygier-Kurz, K.; Kazmierczak, J.; Wilczynski, J. Adenomyosis and Infertility—Review of Medical and Surgical Approaches. Int. J. Environ. Res. Public Health 2021, 18, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Iacovides, S.; Avidon, I.; Baker, F.C. What We Know about Primary Dysmenorrhea Today: A Critical Review. Hum. Reprod. Update. 2015, 21, 762–778. [Google Scholar] [CrossRef] [PubMed]
- Dawood, M.Y. Primary dysmenorrhea: Advances in pathogenesis and management. Obstet Gynecol. 2006, 108, 428–441. [Google Scholar] [CrossRef]
- Rivera, R.; Yacobson, I.; Grimes, D. The mechanism of action of hormonal contraceptives and intrauterine contraceptive devices. Am J Obstet Gynecol. 1999, 181 Pt 1, 1263–1269. [Google Scholar] [CrossRef]
- Osayande, A.S.; Mehulic, S. Diagnosis and initial management of dysmenorrhea. Am Fam Physician. 2014, 89, 341–346. [Google Scholar]
- Wong, C.L.; Farquhar, C.; Roberts, H.; Proctor, M. Oral Contraceptive Pill as Treatment for Primary Dysmenorrhoea. Cochrane Database Syst. Rev. 2009, 2. [Google Scholar] [CrossRef]
- Hassanin, A.I.; Youssef, A.A.; Yousef, A.M.; Ali, M.K. Comparison of Dienogest versus Combined Oral Contraceptive Pills in the Treatment of Women with Adenomyosis: A Randomized Clinical Trial. Int J Gynaecol Obstet. 2021, 154, 263–269. [Google Scholar] [CrossRef]
- Shaaban, O.M.; Ali, M.K.; Sabra, A.M.A.; Abd El Aal, D.E.M. Levonorgestrel-Releasing Intrauterine System versus a Low-Dose Combined Oral Contraceptive for Treatment of Adenomyotic Uteri: A Randomized Clinical Trial. Contraception. 2015, 92, 301–307. [Google Scholar] [CrossRef]
- Abbas, A.M.; Samy, A.; Atwa, K.; Ghoneim, H.M.; Lotfy, M.; Saber Mohammed, H.; Abdellah, A.M.; El Bahie, A.M.; Aboelroose, A.A.; El Gedawy, A.M.; Mostafa, M.; Elsenity, M.A.; Samy, M.; Safwat, S.; Abdelrahman, R.M.; Salah El Din, A.S.; Badran, E.; Abdelkader, A.M.; Abdallah, A. The role of levonorgestrel intra-uterine system in the management of adenomyosis: A systematic review and meta-analysis of prospective studies. Acta Obstet Gynecol Scand. 2020, 99, 571–581. [Google Scholar] [CrossRef] [PubMed]
- Beatty, M.N.; Blumenthal, P.D. The levonorgestrel-releasing intrauterine system: Safety, efficacy, and patient acceptability. Ther Clin Risk Manag. 2009, 5, 561–574. [Google Scholar] [CrossRef] [PubMed]
- Stratopoulou, C.A.; Donnez, J.; Dolmans, M.M. Conservative Management of Uterine Adenomyosis: Medical vs. Surgical Approach. J Clin Med. 2021, 10, 4878. [Google Scholar] [CrossRef]
- Donnez, J.; Dolmans, M.M. Endometriosis and Medical Therapy: From Progestogens to Progesterone Resistance to GNRH Antagonists: A Review. J Clin Med. 2021, 10, 1–21. [Google Scholar] [CrossRef] [PubMed]
- Nelson, J.R.; Corson, S.L. Long-Term Management of Adenomyosis with a Gonadotropin-Releasing Hormone Agonist: A Case Report. Fertil Steril. 1993, 59, 441–443. [Google Scholar] [CrossRef] [PubMed]
- Imaoka, I.; Ascher, S.M.; Sugimura, K.; Takahashi, K.; Li, H.; Cuomo, F.; Simon, J.; Arnold, L.L. MR Imaging of Diffuse Adenomyosis Changes after GnRH Analog Therapy. J Magn Reson Imaging. 2002, 15, 285–290. [Google Scholar] [CrossRef]
- Sauerbrun-Cutler, M.T.; Alvero, R. Short- and Long-Term Impact of Gonadotropin-Releasing Hormone Analogue Treatment on Bone Loss and Fracture. Fertil Steril. 2019, 112, 799–803. [Google Scholar] [CrossRef]
- Donnez, J.; Donnez, O.; Dolmans, M.M. Evolution of Uterine Adenomyosis Volume during and after GnRH Antagonist (Linzagolix) Treatment: Lessons for Further Clinical Trials. Fertil Steril. 2023, 120, 1071–1073. [Google Scholar] [CrossRef] [PubMed]
- Tsui, K.H.; Lee, W.L.; Chen, C.Y.; Sheu, B.C.; Yen, M.S.; Chang, T.C.; Wang, P.H. Medical Treatment for Adenomyosis and/or Adenomyoma. Taiwanese Journal of Obstetrics and Gynecology. 2014, 53, 459–465. [Google Scholar] [CrossRef] [PubMed]
- Guo, S.W.; Groothuis, P.G. Is It Time for a Paradigm Shift in Drug Research and Development in Endometriosis/Adenomyosis? Hum. Reprod. Update. 2018, 24, 577–598. [Google Scholar] [CrossRef] [PubMed]
- Zhou, Y.F.; Matsuda, M.; Mori, T.; Sakamoto, S.; Mitamura, T. Effects of mifepristone (RU486) treatment on the development of uterine adenomyosis induced by pituitary grafting in mice. Life Sci. 2000, 67, 2713–2720. [Google Scholar] [CrossRef] [PubMed]
- Che, X.; Wang, J.; He, J.; Yu, Q.; Sun, W.; Chen, S.; Zou, G.; Li, T.; Guo, X.; Zhang, X. A New Trick for an Old Dog: The Application of Mifepristone in the Treatment of Adenomyosis. J. Cell. Mol. Med. 2020, 24, 1724–1737. [Google Scholar] [CrossRef]
- Che, X.; Wang, J.; Sun, W.; He, J.; Wang, Q.; Zhu, D.; Zhu, W.; Zhang, J.; Dong, J.; Xu, J.; Zheng, F.; Zhou; Zhao, W.; Lin, Q.; Ye, L.; Zhao, X.; Xu, Z.; Chen, Y.; Wang, J.; Wu, W.; Zhai, L.; Zhou, Y.; Zheng, J.; Zhang, X. Effect of Mifepristone vs Placebo for Treatment of Adenomyosis With Pain Symptoms: A Randomized Clinical Trial. JAMA Netw Open. 2023, 6, e2317860. [CrossRef]
- Rosato, E.; Farris, M.; Bastianelli, C. Mechanism of Action of Ulipristal Acetate for Emergency Contraception: A Systematic Review. Front. Pharmacol. 2016, 6, 315. [Google Scholar] [CrossRef]
- Whitaker, L.H.R.; Middleton, L.J.; Daniels, J.P.; Williams, A.R.W.; Priest, L.; Odedra, S.; Cheed, V.; Stubbs, C.E.; Clark, T.J.; Lumsden, M.A.; Hapangama, D.K.; Bhattacharya, S.; Smith, P.P.; Nicholls, E.P.; Roberts, N.; Semple, S.I.; Saraswat, L.; Walker, J.; Chodankar, R.R.; Critchley, H.O.D. UCON Collaborative. Ulipristal acetate versus levonorgestrel-releasing intrauterine system for heavy menstrual bleeding (UCON): A randomised controlled phase III trial. EClinicalMedicine 2023, 60, 101995. [Google Scholar] [CrossRef]
- Ferrero, S.; Scala, C.; Racca, A.; Tafi, E.; Venturini, P.; Leone Roberti Maggiore, U. Changes in Adenomyosis after Treatment with Ulipristal Acetate. Fertil Steril. 2016, 106, e281–e282. [Google Scholar] [CrossRef]
- Gonçalves-Henriques, M.; de Pinho, A.; Freixo, M.; Liz-Coelho, M.; Castro, F.; Ceschin, N.; Brandão, P. Ulipristal Acetate in Adenomyosis. Gynecol Minim Invasive Ther. 2022, 11, 198. [Google Scholar] [CrossRef]
- Kimura, F.; Takahashi, K.; Takebayashi, K.; Fujiwara, M.; Kita, N.; Noda, Y.; Harada, N. Concomitant Treatment of Severe Uterine Adenomyosis in a Premenopausal Woman with an Aromatase Inhibitor and a Gonadotropin-Releasing Hormone Agonist. Fertil Steril. 2007, 87, 1468.e9–1468.e12. [Google Scholar] [CrossRef]
- Badawy, A.M.; Elnashar, A.M.; Mosbah, A.A. Aromatase Inhibitors or Gonadotropin-Releasing Hormone Agonists for the Management of Uterine Adenomyosis: A Randomized Controlled Trial. Acta Obstetricia et Gynecologica Scandinavica. 2012, 91, 489–495. [Google Scholar] [CrossRef]
- Rižner, T.L. The Important Roles of Steroid Sulfatase and Sulfotransferases in Gynecological Diseases. Front. Pharmacol. 2016, 7. [Google Scholar] [CrossRef]
- Messinger, J.; Husen, B.; Koskimies, P.; Hirvelä, L.; Kallio, L.; Saarenketo, P.; Thole, H. Estrone C15 Derivatives-A New Class of 17β-Hydroxysteroid Dehydrogenase Type 1 Inhibitors. Mol. Cell. Endocrinol. 2009, 301, 216–224. [Google Scholar] [CrossRef]
- Clinical trial: A Study to Investigate Efficacy and Safety of OG-6219 BID in 3 Dose Levels Compared With Placebo in Participants Aged 18 to 49 With Moderate to Severe Endometriosis-related Pain (ELENA). Available online: https://clinicaltrials.gov/study/NCT05560646 (accessed on 18 January 2024).
- Poirier, D.; Nyachieo, A.; Romano, A.; Roy, J.; Maltais, R.; Chai, D.; Delvoux, B.; Tomassetti, C.; Vanhie, A. An Irreversible Inhibitor of 17β-Hydroxysteroid Dehydrogenase Type 1 Inhibits Estradiol Synthesis in Human Endometriosis Lesions and Induces Regression of the Non-Human Primate Endometriosis. J. Steroid Biochem. Mol. Biol. 2022, 222. [Google Scholar] [CrossRef]
- Poirier, D.; Nyachieo, A.; Romano, A.; Roy, J.; Maltais, R.; Chai, D.; Delvoux, B.; Tomassetti, C.; Vanhie, A. An Irreversible Inhibitor of 17β-Hydroxysteroid Dehydrogenase Type 1 Inhibits Estradiol Synthesis in Human Endometriosis Lesions and Induces Regression of the Non-Human Primate Endometriosis. J. Steroid Biochem. Mol. Biol. 2022, 222. [Google Scholar] [CrossRef] [PubMed]
- Sahin, C.; Mamillapalli, R.; Yi, K.W.; Taylor, H.S. MicroRNA Let-7b: A Novel Treatment for Endometriosis. J. Cell. Mol. Med. 2018, 22, 5346–5353. [Google Scholar] [CrossRef] [PubMed]
- Andersson, J.K.; Pozzi Mucelli, R.; Epstein, E.; Stewart, E.A.; Gemzell-Danielsson, K. Vaginal Bromocriptine for Treatment of Adenomyosis: Impact on Magnetic Resonance Imaging and Transvaginal Ultrasound. Eur. J. Obstet. Gynecol. Reprod. Biol. 2020, 254, 38–43. [Google Scholar] [CrossRef] [PubMed]
- Auriemma, R.S.; Del Vecchio, G.; Scairati, R.; Pirchio, R.; Liccardi, A.; Verde, N.; de Angelis, C.; Menafra, D.; Pivonello, C.; Conforti, A.; Alviggi, C.; Pivonello, R.; Colao, A. The Interplay Between Prolactin and Reproductive System: Focus on Uterine Pathophysiology. Front Endocrinol (Lausanne). 2020, 11. [Google Scholar] [CrossRef] [PubMed]
- Tang, Y.; Ponandai-srinivasan, S.; Frisendahl, C.; Andersson, J.K.; Pavone, D.; Stewart, E.A.; Lalitkumar, P.G.L.; Korsching, E.; Bogavarappu, N.R.; Gemzell-Danielsson, K. Bromocriptine Inhibits Proliferation in the Endometrium from Women with Adenomyosis. Front Endocrinol (Lausanne). 2023, 14. [Google Scholar] [CrossRef] [PubMed]
- Mahar, K.M.; Enslin, M.B.; Gress, A.; Amrine-Madsen, H.; Cooper, M. Single- and Multiple-Day Dosing Studies to Investigate High-Dose Pharmacokinetics of Epelsiban and Its Metabolite, GSK2395448, in Healthy Female Volunteers. Clin Pharmacol Drug Dev. 2018, 7, 33–43. [Google Scholar] [CrossRef]
- Clinical trial: Placebo-controlled Proof of Concept Study of Epelsiban in Women With Adenomyosis. Available online: https://clinicaltrials.gov/study/NCT02794467 (accessed on 18 January 2024).
- Barberis, C. L372662; Elseviers Ebooks, 2007.
- Xue, J.; Zhang, H.; Liu, W.; Liu, M.; Shi, M.; Wen, Z.; Li, C. Metformin Inhibits Growth of Eutopic Stromal Cells from Adenomyotic Endometrium via AMPK Activation and Subsequent Inhibition of AKT Phosphorylation: A Possible Role in the Treatment of Adenomyosis. Reproduction. 2013, 146, 397–406. [Google Scholar] [CrossRef]
- Forster, R.; Sarginson, A.; Velichkova, A.; Hogg, C.; Dorning, A.; Horne, A.W.; Saunders, P.T.K.; Greaves, E. Macrophage-Derived Insulin-like Growth Factor-1 Is a Key Neurotrophic and Nerve-Sensitizing Factor in Pain Associated with Endometriosis. FASEB Journal. 2019, 33, 11210–11222. [Google Scholar] [CrossRef]
- Yalçın Bahat, P.; Ayhan, I.; Üreyen Özdemir, E.; İnceboz, Ü.; Oral, E. Dietary supplements for treatment of endometriosis: A review. Acta Biomed. 2022, 93, e2022159. [Google Scholar] [CrossRef]
- Chen, Y.; Zhu, B.; Zhang, H.; Liu, X.; Guo, S.W. Epigallocatechin-3-Gallate Reduces Myometrial Infiltration, Uterine Hyperactivity, and Stress Levels and Alleviates Generalized Hyperalgesia in Mice Induced with Adenomyosis. Reprod Sci. 2013, 20, 1478–1491. [Google Scholar] [CrossRef]
- Chen, Y.; Zhu, B.; Zhang, H.; Ding, D.; Liu, X.; Guo, S.W. Possible Loss of GABAergic Inhibition in Mice with Induced Adenomyosis and Treatment with Epigallocatechin-3-Gallate Attenuates the Loss with Improved Hyperalgesia. Reprod Sci. 2014, 21, 869–882. [Google Scholar] [CrossRef]
- Moldassarina, R.S.; Manabayeva, G.K.; Akylzhanova, Z.Y.; Rashidova, A.M. The Importance of Vitamin D in the Diagnosis and Treatment of Adenomyosis. Mol Cell Biochem. 2023, 478, 571–579. [Google Scholar] [CrossRef]
- Singh, A.; Dasgupta, S.; Bhattacharya, A.; Mukherjee, G.; Chaudhury, K. Therapeutic Potential of Curcumin in Endometrial Disorders: Current Status and Future Perspectives. Drug Discovery Today. 2022, 27, 900–911. [Google Scholar] [CrossRef]
- Cheng, W.X.; Wei, S. Bin; Zhou, Y.; Shao, Y.; Li, M.Y. Exosomes: Potential Diagnostic Markers and Drug Carriers for Adenomyosis. Front. Pharmacol. 2023, 14. [Google Scholar] [CrossRef]
- Shahzad, H.; Giribabu, N.; Muniandy, S.; Salleh, N. Quercetin induces morphological and proliferative changes of rat's uteri under estrogen and progesterone influences. Int J Clin Exp Pathol. 2014, 7, 5484–5494. [Google Scholar]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).



