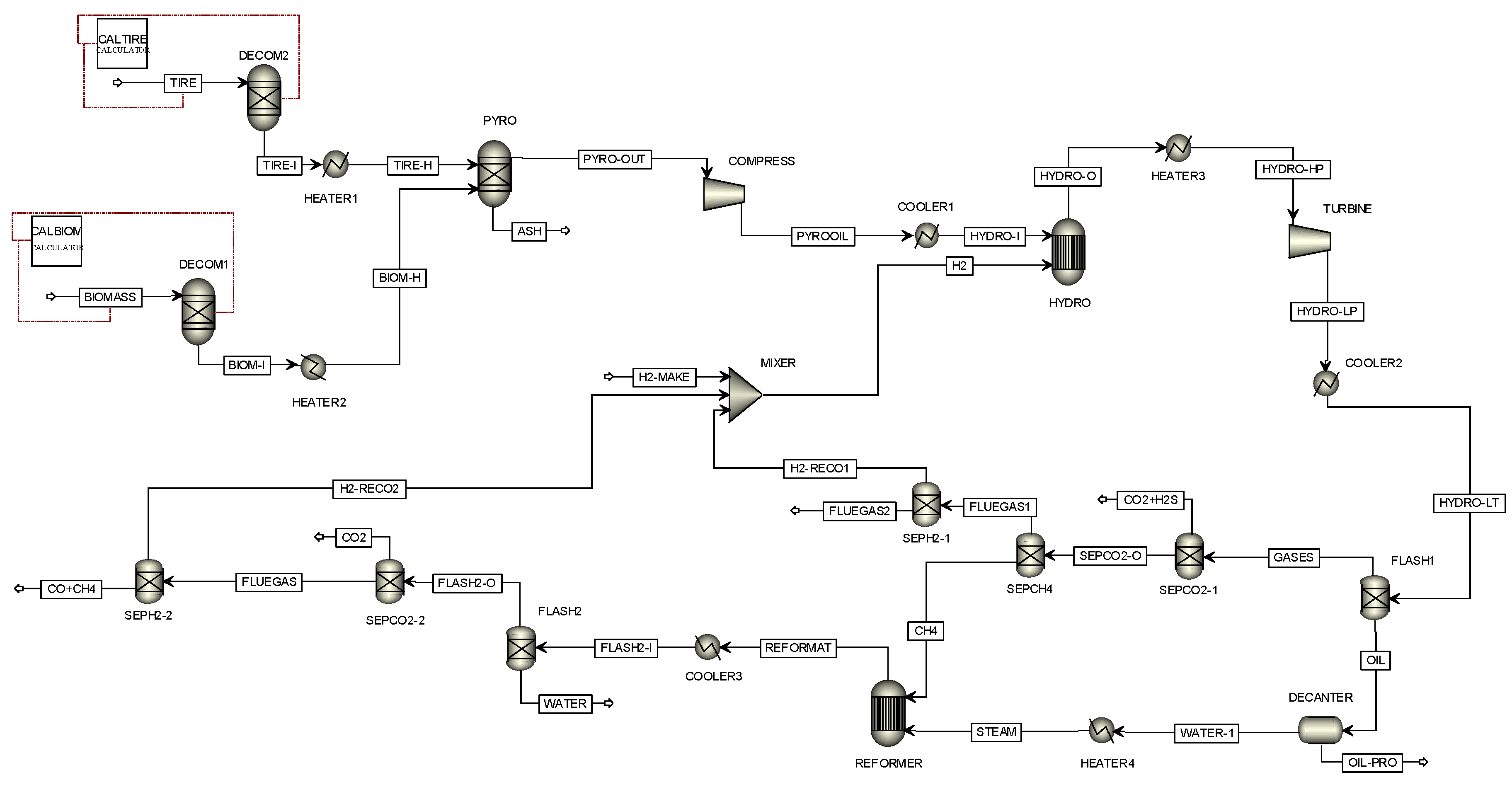
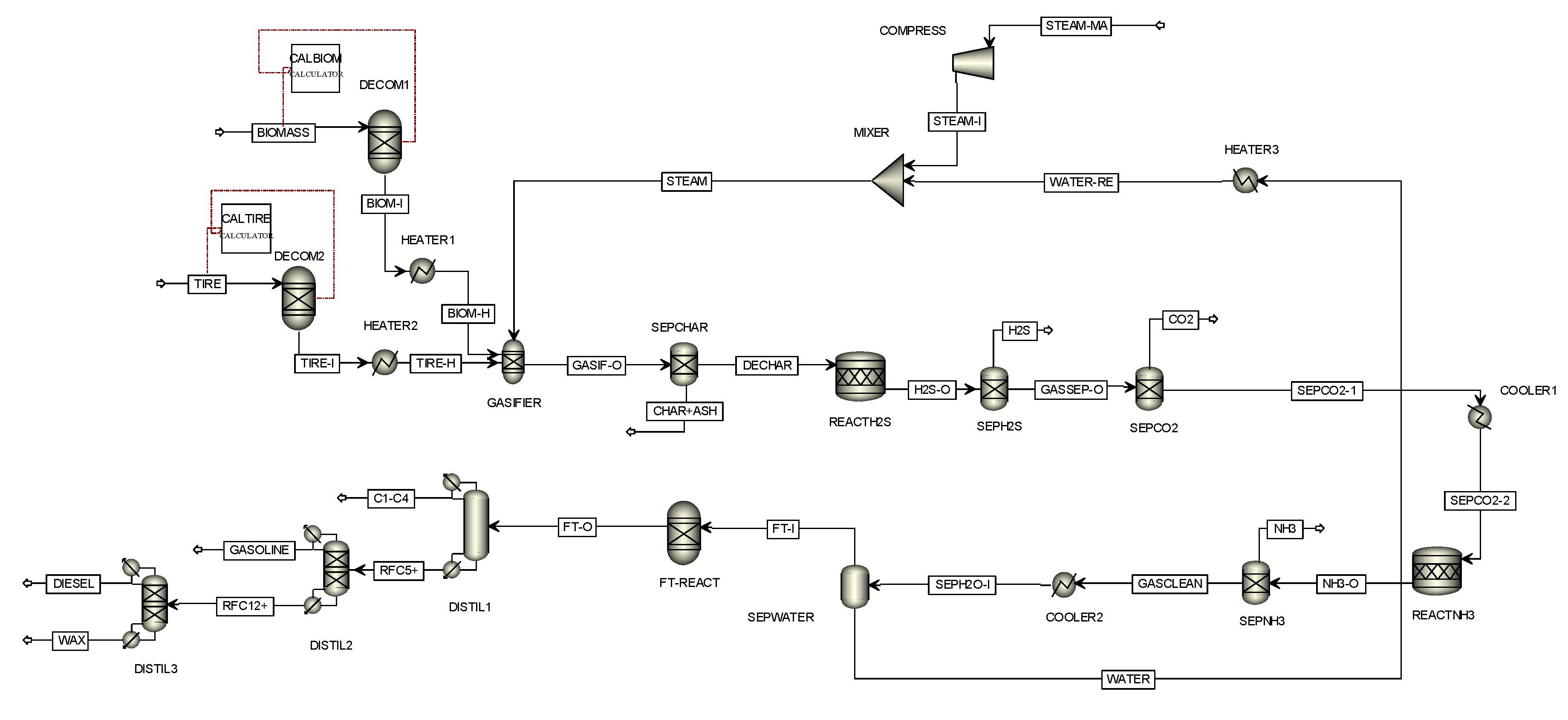
4.1. Model Validation
To ensure that the proposed model can predict realistic results, gases, oils, and other products obtained from both models are compared with the experimental data extracted from the literature. In case of co-pyrolysis model, the simulated results are compared with the experimental data of D. Pradhan et al. [
41]. Their experiment used mahua seed and waste thermocol as a feedstock in which the proximate and ultimate analysis results are listed in
Table 7. In their work, the co-pyrolysis process operated at 550 °C and 1 bar with the blending weight ratio 1:1. The experimental results showed that the products are composed of gases, oil, char and aqueous liquid. Under the same feed-stock and operating conditions, the model validation, as shown in
Table 8, indicates that the simulated results agree with the experimental data. The root means square error (RMS) is 11.90, which is an acceptable value for this study. The gas products obtained from the model have more proportion than the experimental data while the oil, char and aqueous liquid products obtained from the model have less proportion than one. The deviation between simulation and experiment can be explained by two reasons: (1) The model uses simplified assumptions about the reaction mechanism which may not reflect the complex reality of the reaction. The product obtained from the simulation is calculated by minimizing the Gibbs free energy. These gas products have lower Gibbs free energy compared to oil, char, and aqueous liquid products. This means that they are more stable and energetically favorable to form during the reaction and (2) this model does not consider the effect of the catalyst to specify the desired products. Although the validation results deviate, this model is acceptable.
Next, the simulated results obtained from the gasification model are compared with the experimental data of W. Doherty et al. [
42]. Feedstock used in their experiment was wood chips in which proximate and ultimate analysis are listed in
Table 7. In their work, the gasification process operated at 850
°C and 1 bar with the mass flow rate of biomass and a S/F ratio as 1508.64 kg/h and 0.75, respectively. The conditions of steam were at 450
°C and 1 bar. The experimental results showed that the products are composed of gases (H
2, CH
4, CO, and CO
2), char, and others (H
2S, NH
3, and H
2O). The comparison of product gas composition between simulation results and experimental data under the same feedstock and operating conditions are shown in
Table 9. It is found that the simulation results show similar trends with the experimental data. RMS is 9.26, which is an acceptable value for this study. However, it can be observed that higher hydrogen, lower carbon dioxide, and methane concentrations can be provided compared with the experimental data. The deviation between simulation and experiment can be explained by two reasons: (1) the calculation of the product gas composition based on the equilibrium which neglects the effect of a catalyst and (2) tar formation is not considered in this study.
4.2. Liquid Fuel Production from the Co-Pyrolysis Process
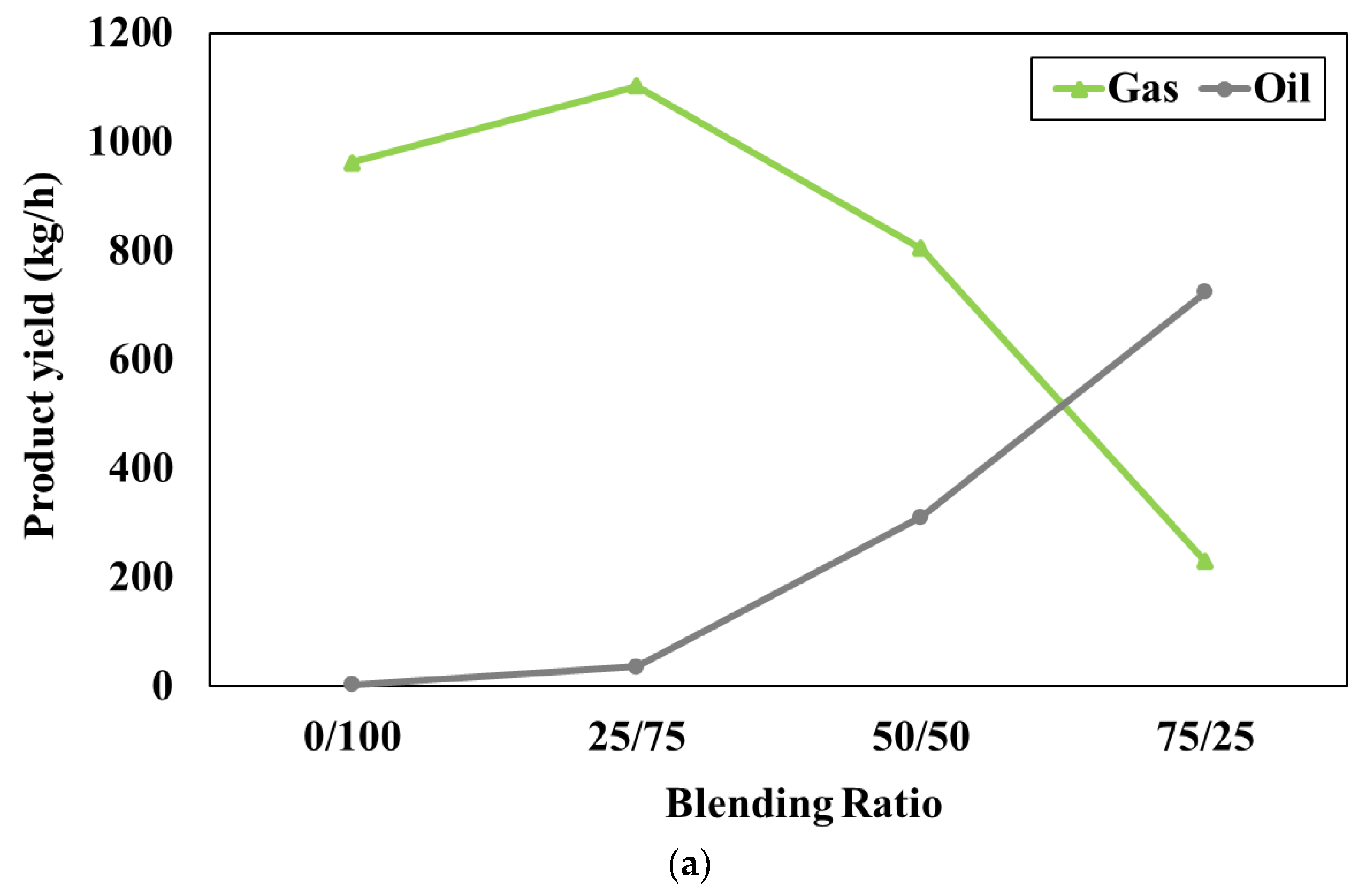
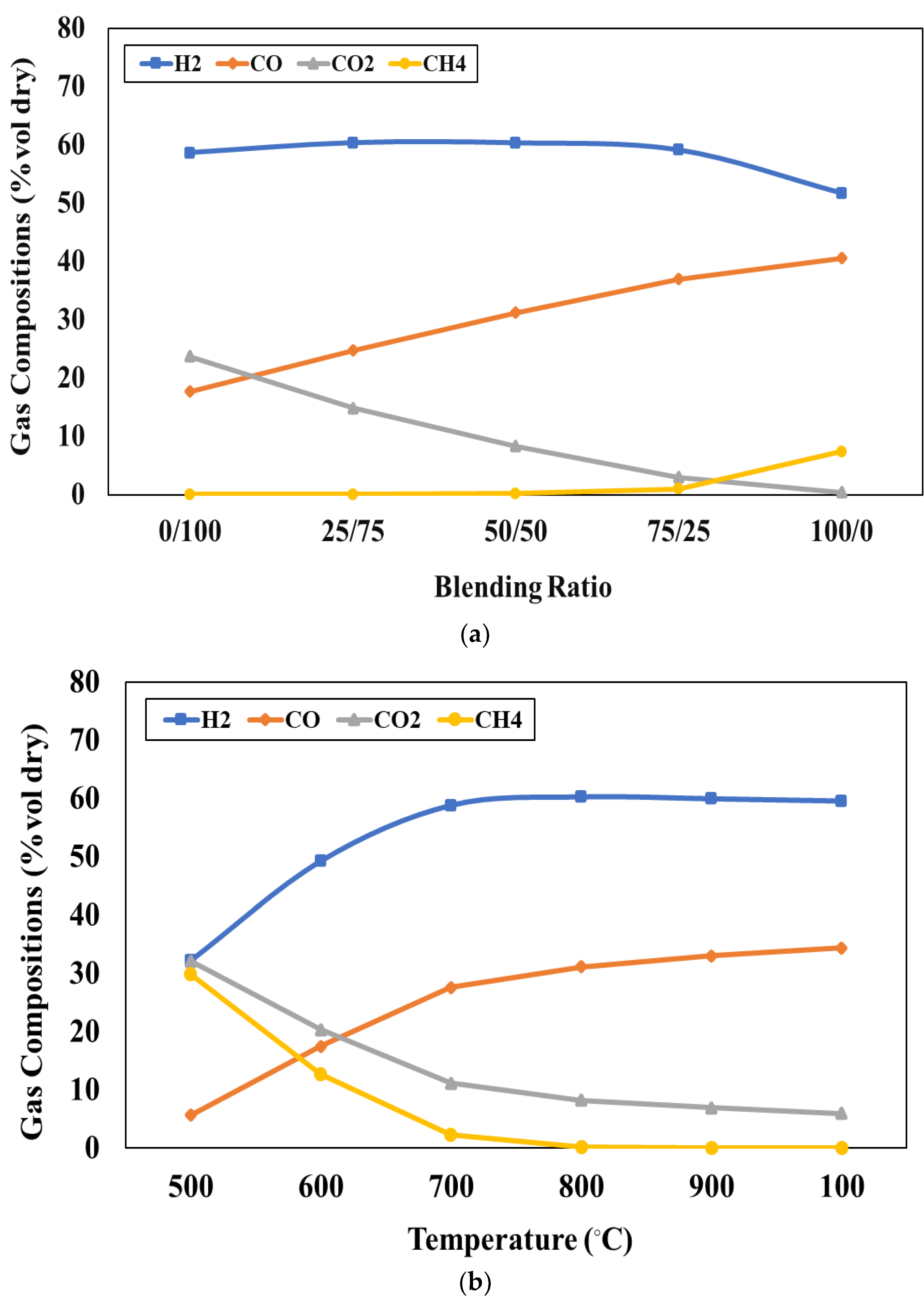
Figure 3a represents the effect of the blending weight ratio on the product compositions when the pyrolyser temperature is fixed at 600°C. In this study, the blending weight ratio of feedstocks (W/B) varied as 0:100, 25:75, 50:50, and 75:25, respectively. The simulation results show that gas product is lower while oil product is higher with increasing blending weight ratio (waste tires increases while water hyacinth decreases). According to the ultimate analysis, the waste tires have carbon as the main component whereas water hyacinth has oxygen as a main component and both feedstocks have similar amounts of hydrogen. It should be noted that the oil product consists of mainly hydrocarbon (hydrogen and carbon) content whereas the gas product is mainly composed of carbon dioxide which is referred to the oxygenated compound. Therefore, when the ratio of waste tires increases, carbon and hydrogen can easily combine to form hydrocarbon compounds in the oil products. On the other hand, when the ratio of water hyacinth increases, oxygen can easily combine with other components to form the oxygenated compound in the gas products. However, there is no result reported in
Figure 3a at the blending weight ratio of 100:0. It can be implied that the use of feedstock as pure waste tires (no water hyacinth) cannot generate oil products consisting of C
10H
12O
4. This is because the small amount of oxygen in waste tires is not sufficient to form the oxygenated compound in oil products.
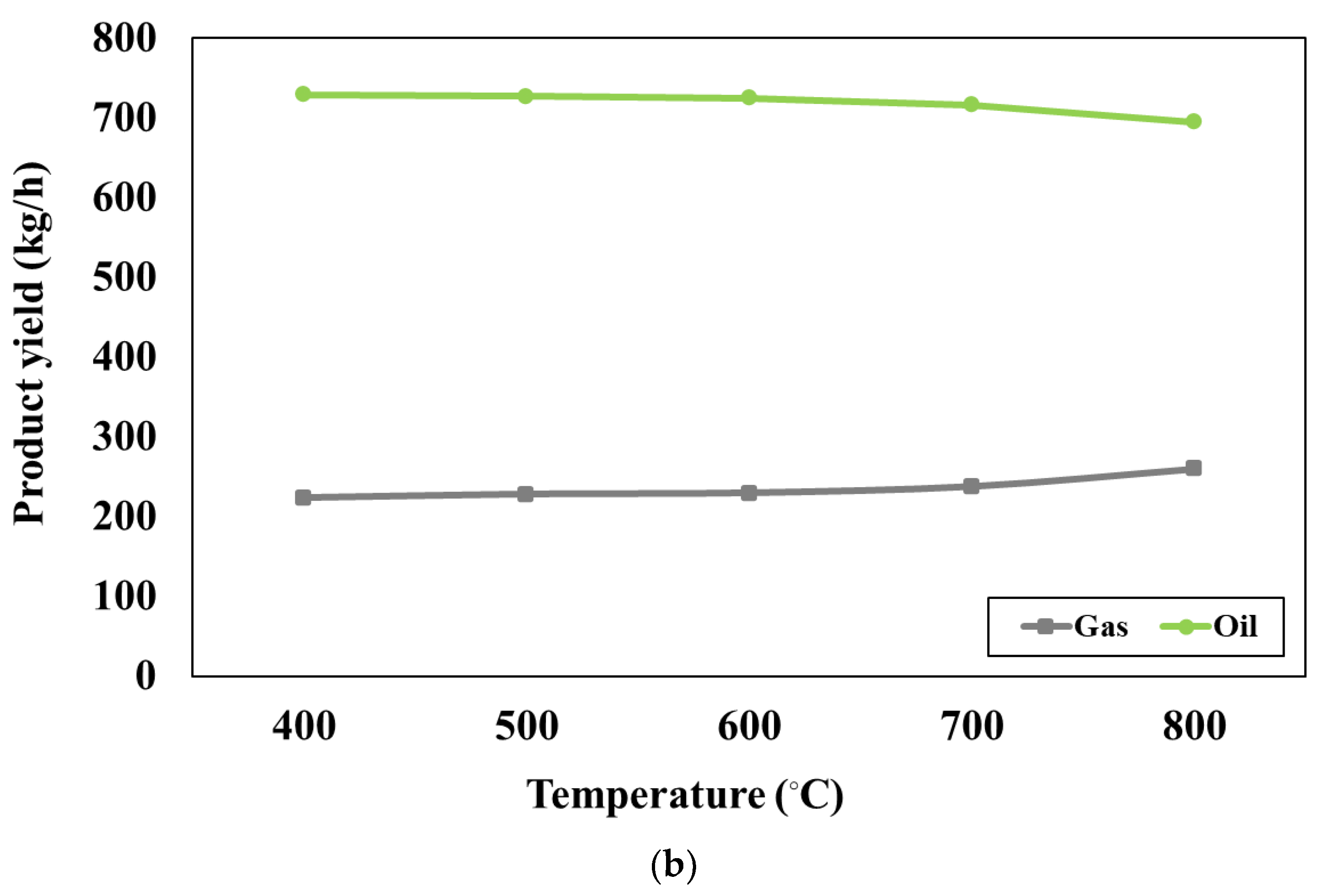
Figure 3b presents the effect of pyrolyser temperature varying between 400°C and 800°C on the product at the blending weight ratio of 75:25. The simulation results show that increasing pyrolyser temperature can slightly increase the gas products whereas the oil products may slightly decrease. When the pyrolyser temperature is higher, the larger hydrocarbon chains will be cracked into the smaller ones, according to the study conducted by H.Y. Ismail et al [
43]. At the pyrolyser temperature of 400 °C, the desired product (oil yield) has the largest quantity and the undesired product (gas yield) has the smallest amount. Therefore, the pyrolyser temperature of 400 °C is the most suitable for the pyrolysis process.
4.3. Liquid Fuel Production through the Co-Gasification Integrated with Fischer-Tropsch Process
Figure 4a presents the effect of blending weight ratio on the product gas compositions at the gasifier temperature of 800 °C and S/F ratio of 1. It is found that the concentrations of H
2 little change at the beginning and decrease after that when the blending weight ratio increases. The concentrations of CO
2 decrease while the concentrations of CO and CH
4 increase when the blending weight ratio increases. At higher blending weight ratios of solid waste, adding solid waste to the feedstock can increase the production of H
2 and CO in the syngas due to the high volatile matter content of solid waste. In addition, it also increases the reactivity of the feedstock, leading to a higher conversion of carbon to syngas. As a result, the CH
4 concentration in the syngas may increase. However, at high blending weight ratios of biomass, the higher oxygen content of biomass promotes the oxidation of syngas due to the presence of oxygen-containing functional groups in the biomass. As a result, H
2 and CO
2 concentrations may decrease due to loss of oxidation whereas the CO
2 content in the syngas may increase. In this study, the target of H
2/CO ratio is around 2 to further use in the FT process according to Y.D. Kim et al. [
4]. The results reveal that the H
2/CO ratio as 3.32, 2.44, 1.93, 1.60, and 1.28 can be provided when the blending weight ratio is 0:100, 25:75, 50:50, 75:25, and 100:0, respectively. Although the blending weight ratio at 75:25 provides a higher CO content than the blending weight ratio at 50:50, the H
2/CO ratio close to 2 can be provided when the blending ratio is 50:50. Therefore, the blending weight ratio of 50:50 is suitable for liquid fuel production through co-gasification integrated with the FT process.
Figure 4b presents the effect of gasifier temperature, varying between 500 and 1000 °C, on the product gas compositions at the blending weight ratio of 50:50 and S/F ratio of 1. It can be observed that the concentrations of CO
2 and CH
4 decrease whereas the concentrations of H
2 and CO increase with increasing temperature. This is because the Bouduard and water gas reactions as endothermic reactions favor high gasifier temperature operation and thus, the forward reaction can occur. On the other hand, the methanation reaction as the exothermic reaction is shifted to the reactant side when the gasifier temperature increases. In addition, increasing gasifier temperature causes the reverse water gas shift (WGS) reaction, leading to a decrease in the concentration of CO2. The drop in CH
4 concentration is caused by the consumption in the steam methane reforming (SMR) reaction. From the simulation results, the gasifier temperature of 800°C is suitable for the gasification process because the H
2/CO ratio is mostly close to 2 and provides the high concentration of H
2 and CO for the following FT process.
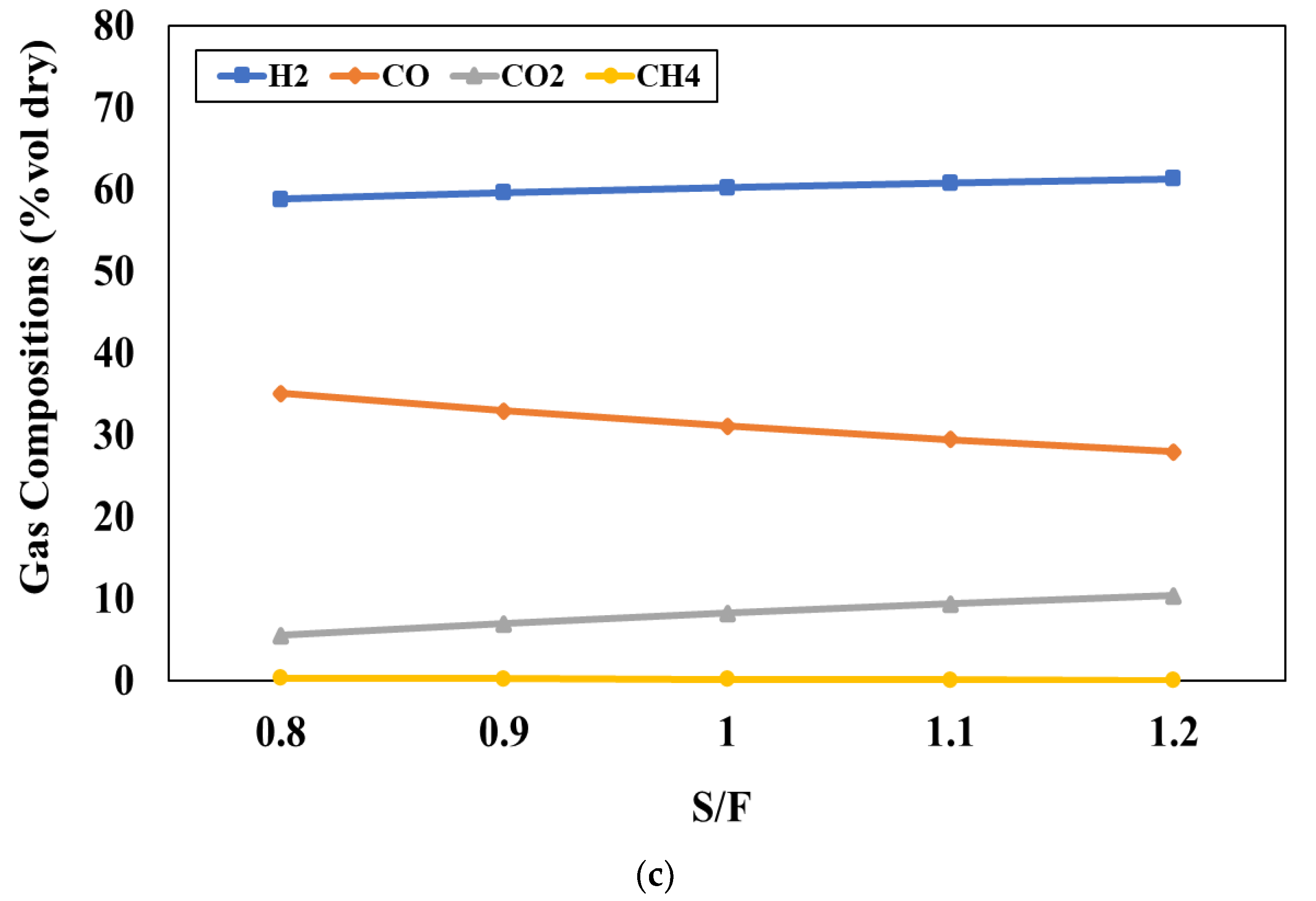
Figure 4c shows the influence of S/F ratio, varying between 0.8 and 1.2, on the product gas compositions at the blending weight ratio of 50:50 and the gasifier temperature of 800 °C. It can be observed that increasing S/F ratio leads to an increase in the H
2 and CO
2 concentrations and a decrease in CO and CH
4 concentrations. Adding steam into the process will support the water gas and SMR reactions and this results in the increase of H
2 and CO concentrations while the CH
4 concentration decreases due to the SMR reaction. However, the CO concentration decreases with an increase in the S/F ratio because the WGS reaction will occur continuously and leads to H
2 and CO
2 concentrations being increased. The simulation results indicate that S/F ratio of 1 is suitable for the gasification because it can provide the suitable H
2/CO ratio and product yield for the FT process.
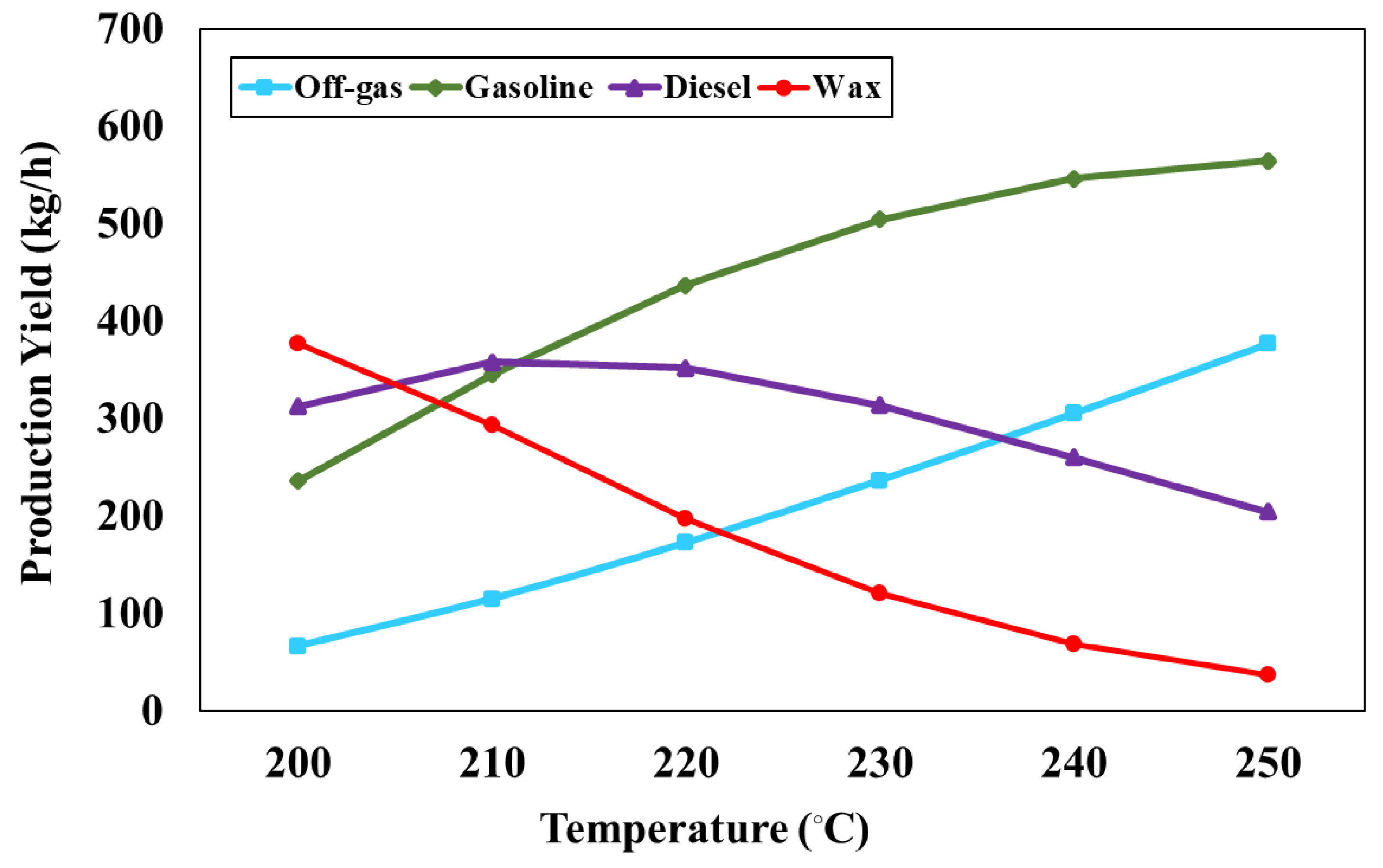
Figure 5 represents the impact of FT reactor temperature, varying between 200 and 250 °C, on the production rate of FT products at the reactor pressure of 1 bar and H
2/CO ratio of 2. From the simulation results, it is found that the production rate of off-gas and gasoline increases with increasing temperature, whereas the production rate of diesel and wax decreases. Increasing FT temperature causes the decomposition of larger hydrocarbon chains into smaller ones but increases the lower hydrocarbon chains. In addition, the product rate of the FT process was estimated with the ASF distribution that depends on the FT temperature and syngas composition (H
2/CO ratio). The simulation results show that the production rate of liquid fuel (gasoline and diesel) as 549.15, 704.17, 789.90, 819.04, 807.62, and 769.67 kg/h can be obtained when FT is operated at 200, 210, 220, 230, 240 and 250°C, respectively. Therefore, the FT reactor temperature at 230 °C is suitable for the FT process as it provides the highest production rate of liquid fuel.