Submitted:
08 January 2024
Posted:
09 January 2024
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Cell Culture
2.3. Cell Proliferation Assay
2.4. Alkaline Phosphatase (ALP) Activity and Staining
2.5. Mineralization Assay
2.6. Western Blot
2.7. Animal Experiment
2.7.1. Micro-Computed Tomography
2.7.2. Histomorphometry Analysis
2.8. Statistical Analysis
3. Results
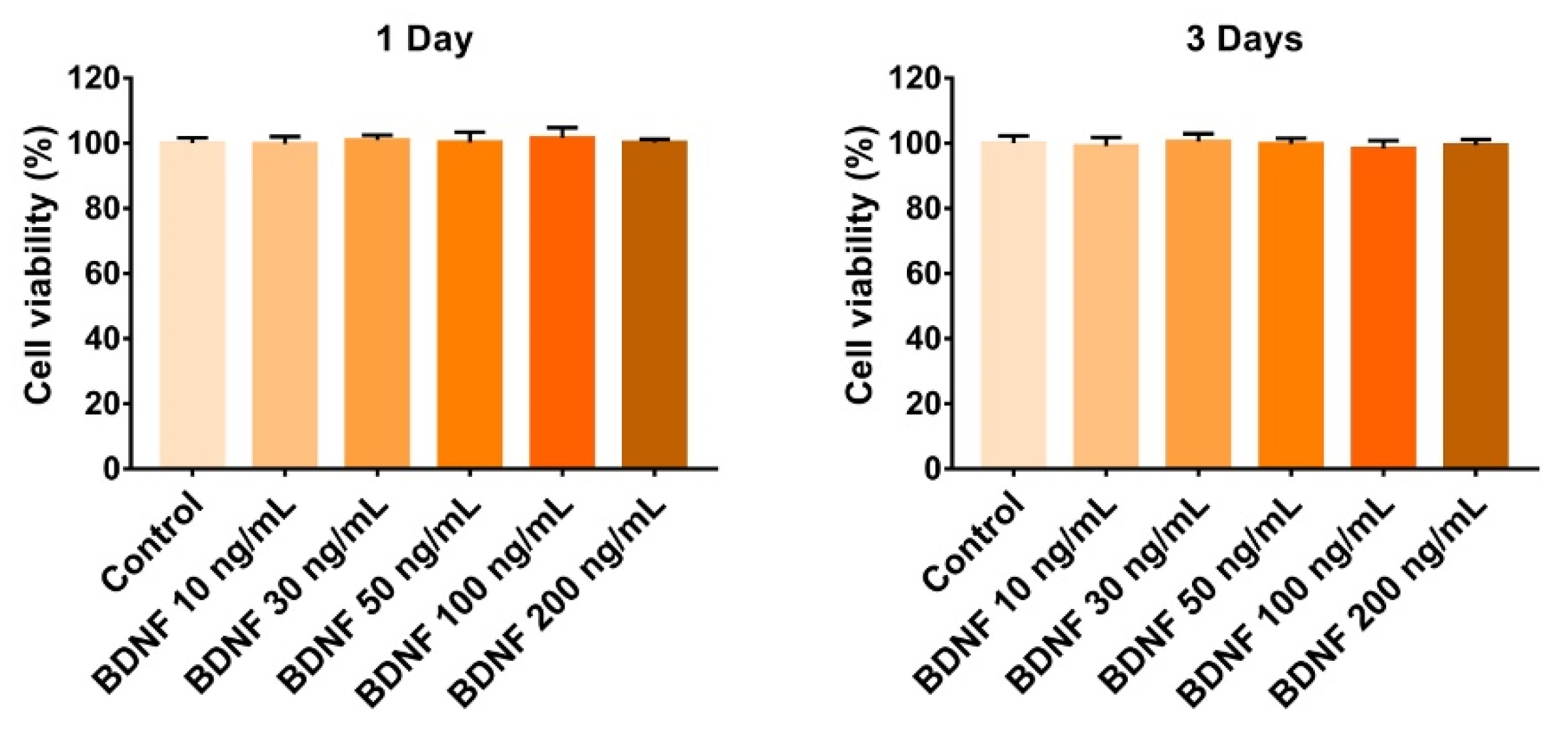
3.1. Effect of BDNF on the Proliferation of ST-2 Cells
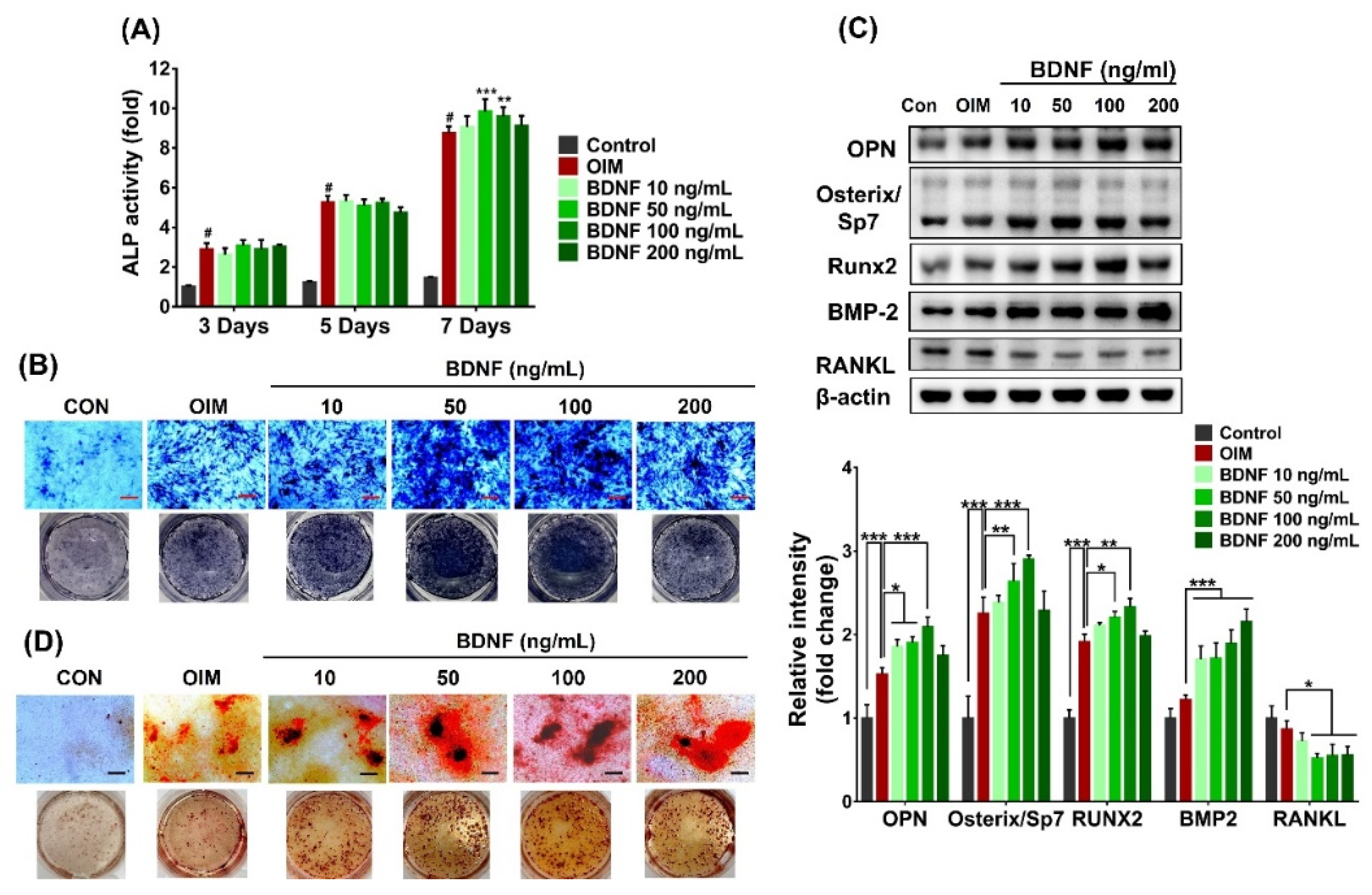
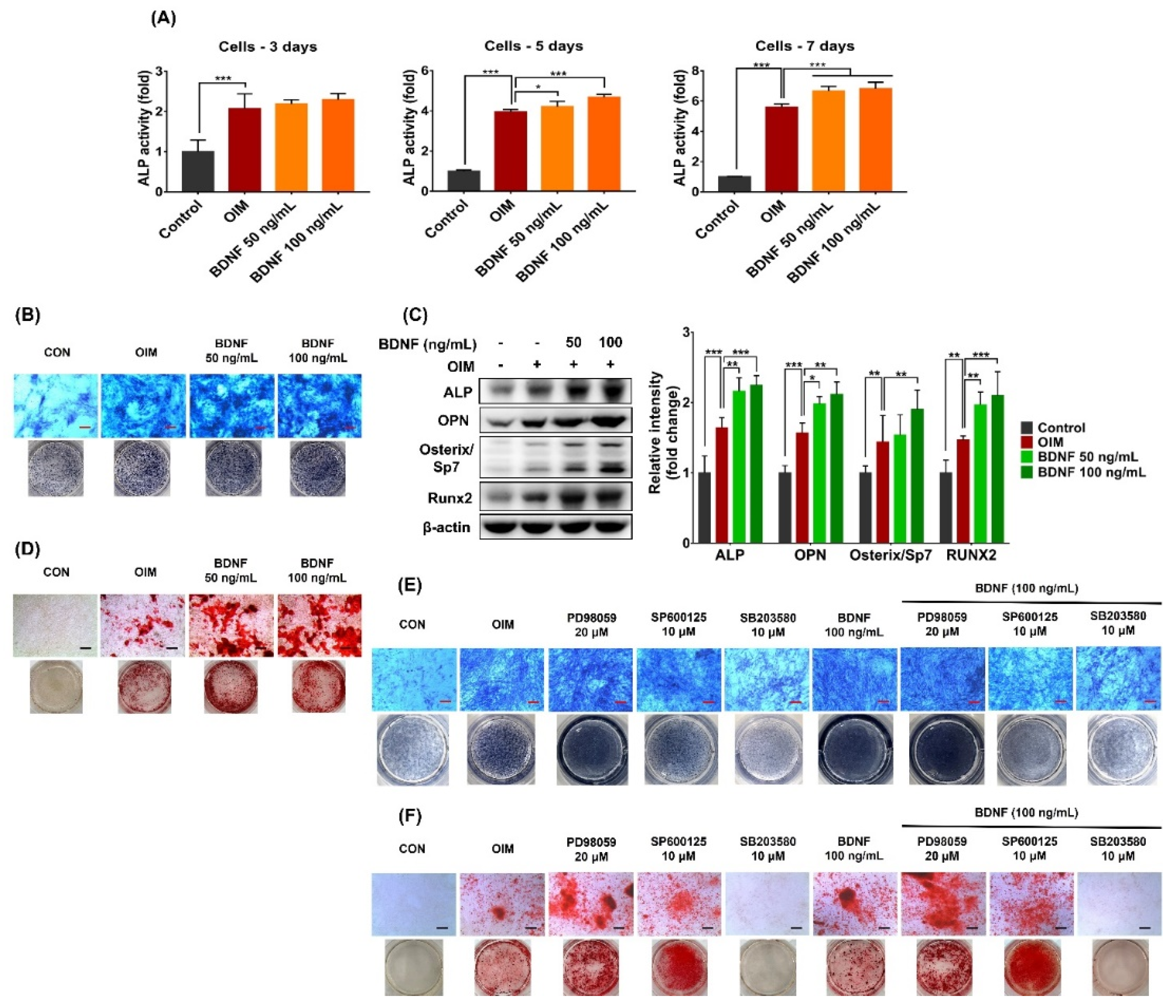
3.2. Effect of BDNF on the Osteoblast Differentiation of ST-2 Cells
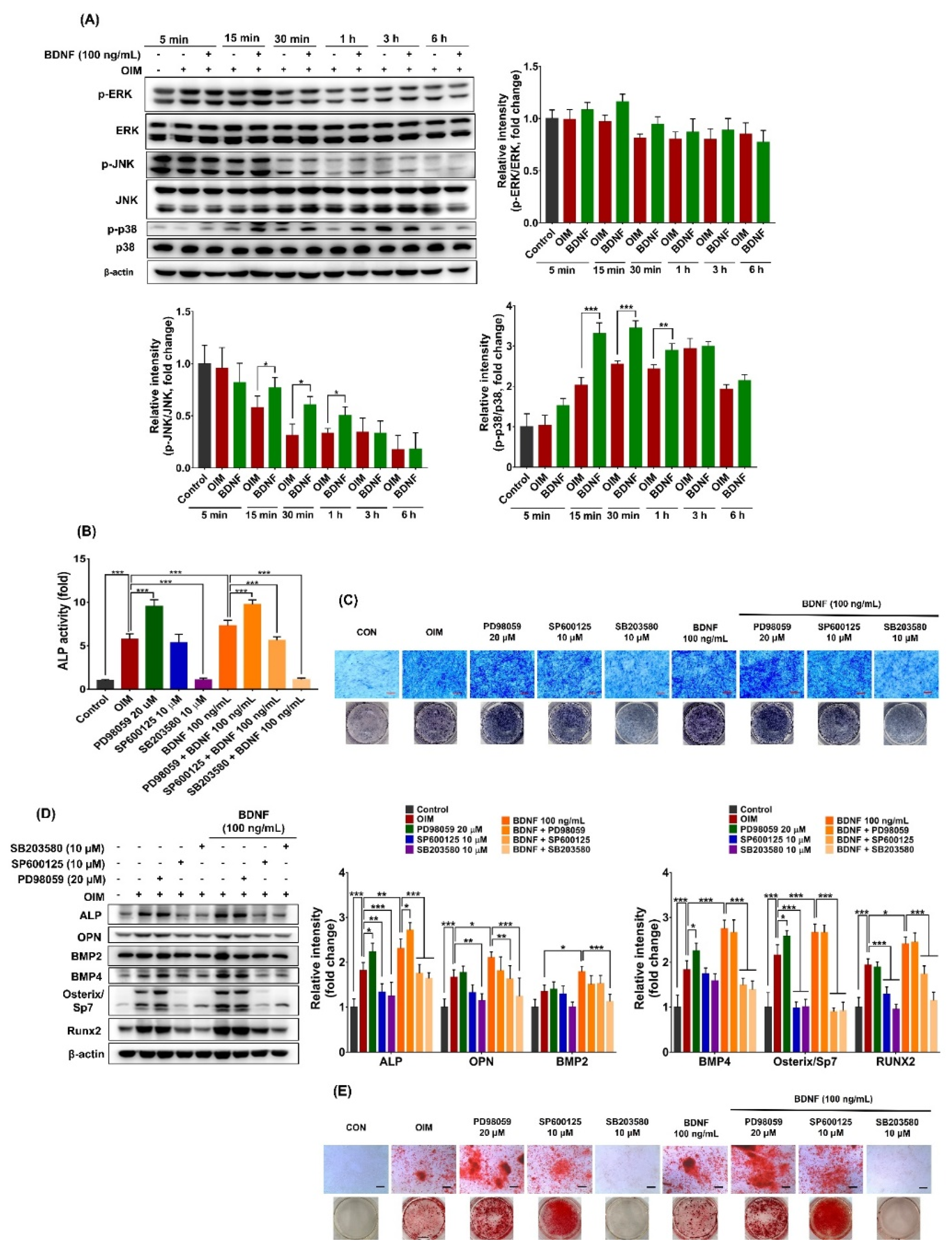
3.3. Effect of BDNF on the Mitogen-Activated Protein Kinase (MAPK) Signaling Pathways in ST-2 Cells
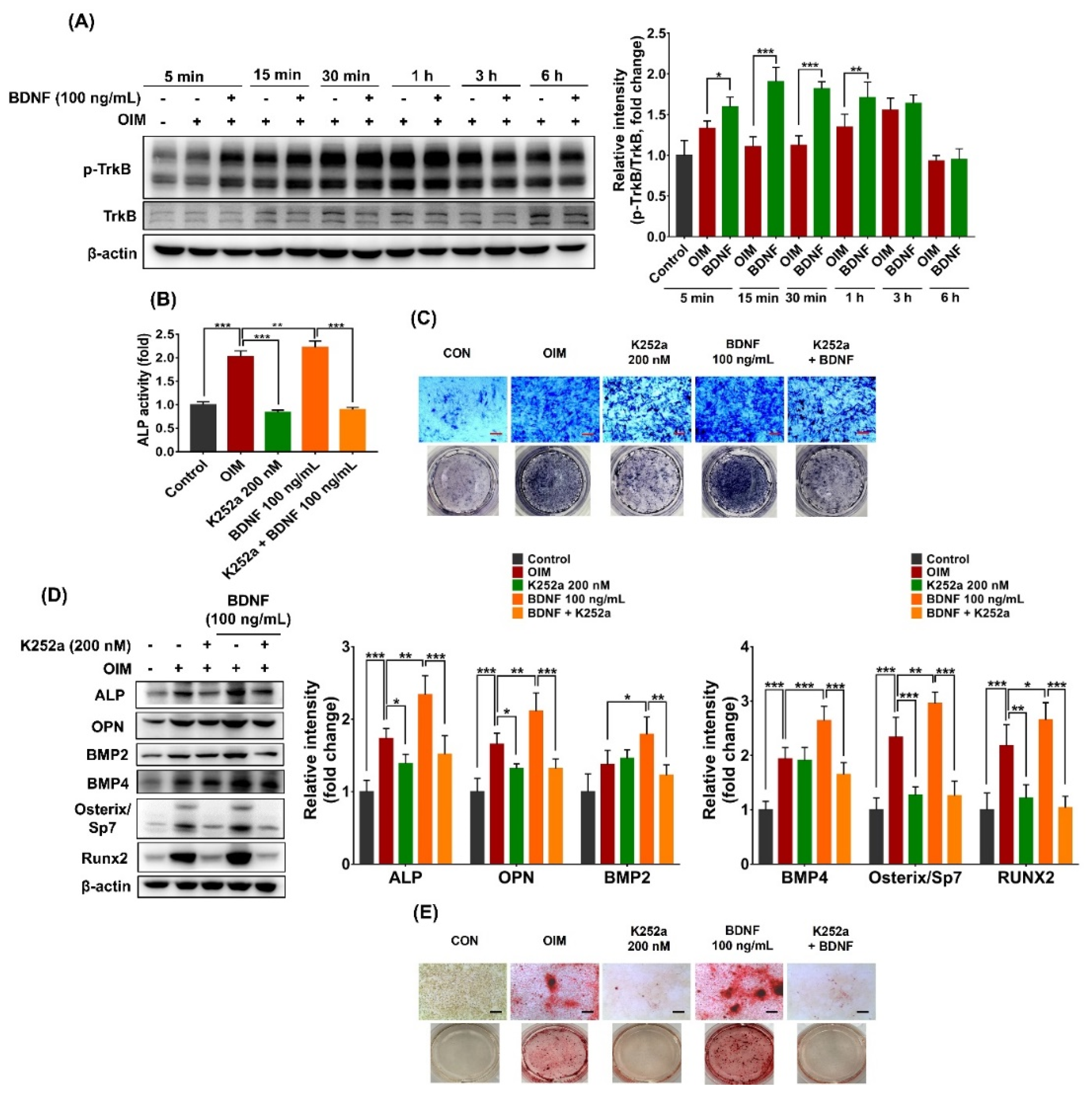
3.4. TrkB Suppression Reduces BDNF-Induced Osteoblast Differentiation
3.5. Effect of BDNF on the Osteogenic Differentiation in the Co-Culture System of Osteoblasts and Bone Marrow Stromal Cells
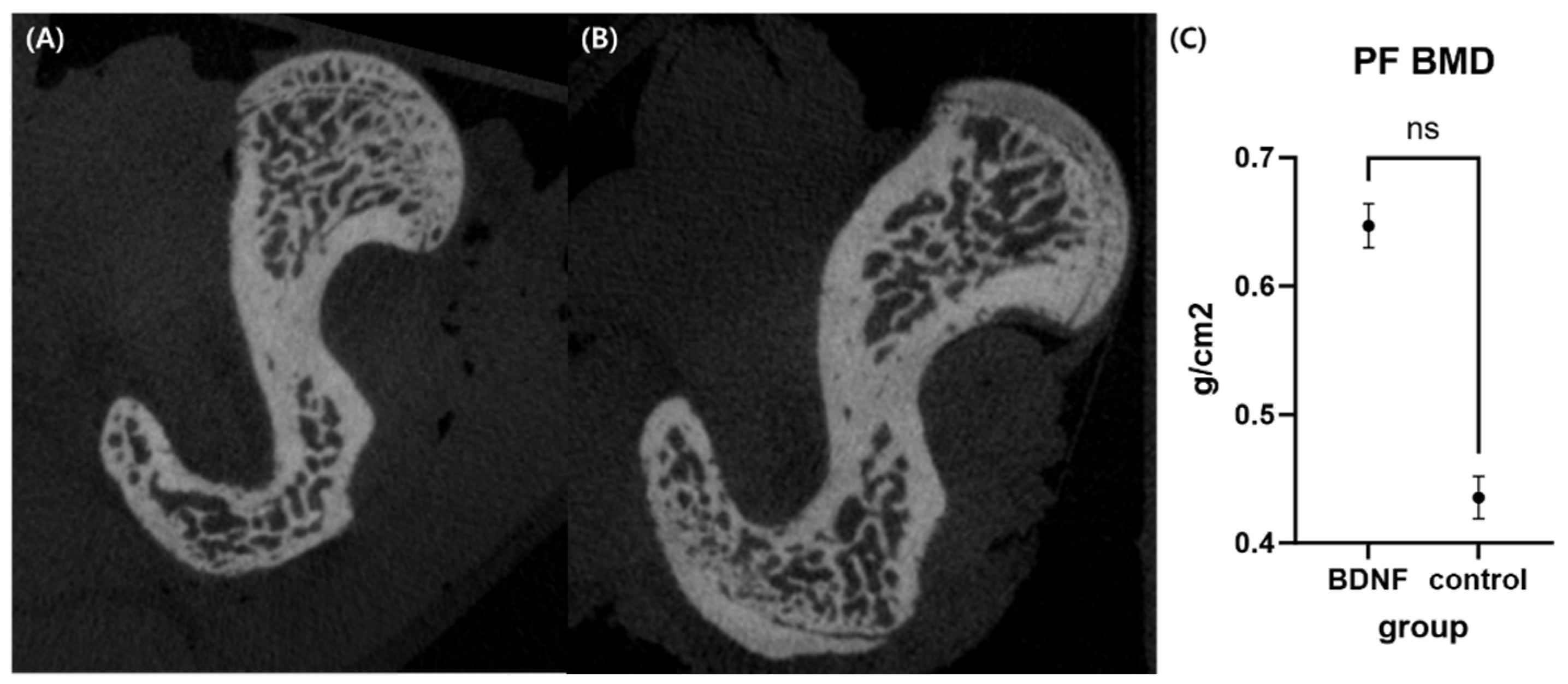
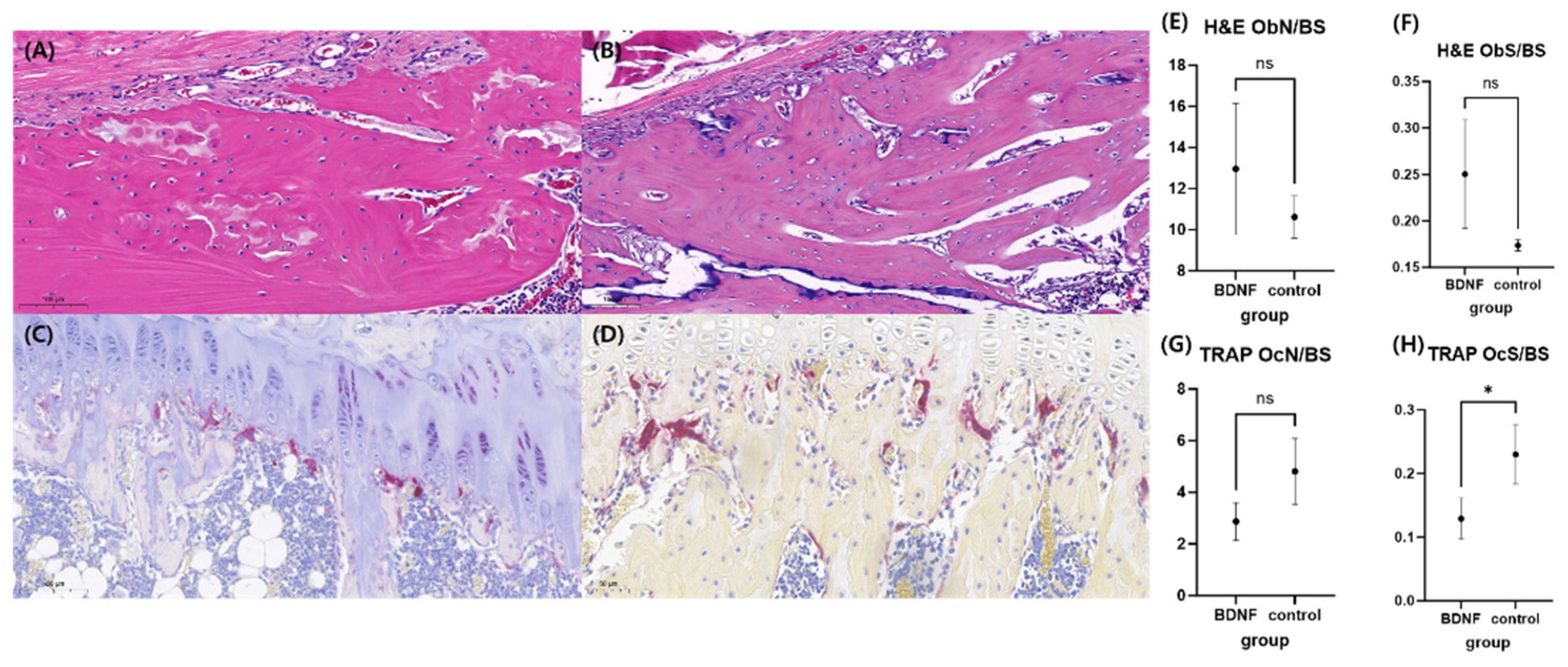
3.6. BDNF Prevents Bone Loss in OVX-Induced Rats
3.6.1. Micro-Computed Tomography
3.6.2. Histomorphometry Analysis
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Xue, F.; Zhao, Z.; Gu, Y.; Han, J.; Ye, K.; Zhang, Y. 7,8-Dihydroxyflavone modulates bone formation and resorption and ameliorates ovariectomy-induced osteoporosis. eLife 2021, 10, e64872. [Google Scholar] [CrossRef]
- Hou, H.; Peng, Q.; Wang, S.; Zhang, Y.; Cao, J.; Deng, Y.; Wang, Y.; Sun, W.C.; Wang, H.B. Anemonin Attenuates RANKL-Induced Osteoclastogenesis and Ameliorates LPS-Induced Inflammatory Bone Loss in Mice via Modulation of NFATc1. Front Pharmacol 2020, 10, 1696. [Google Scholar] [CrossRef]
- Liu, Y.; Wang, H.; Zhou, X.Z.; Li, N.; Guo, Y.C.; Chen, T.P. Pentraxin 3 promotes the osteoblastic differentiation of MC3T3-E1 cells through the PI3K/Akt signaling pathway. Biosci Rep 2020, 40, BSR20201165. [Google Scholar] [CrossRef]
- Tao, K.; Xiao, D.; Weng, J.; Xiong, A.; Kang, B.; Zeng, H. Berberine promotes bone marrow-derived mesenchymal stem cells osteogenic differentiation via canonical Wnt/β-catenin signaling pathway. Toxicol Lett 2016, 240, 68–80. [Google Scholar] [CrossRef]
- Sözen, T.; Özışık, L.; Başaran, N. An overview and management of osteoporosis. Eur J Rheumatol 2017, 4, 46–56. [Google Scholar] [CrossRef]
- Gerosa, L.; Lombardi, G. Bone-to-Brain: A Round Trip in the Adaptation to Mechanical Stimuli. Front Physiol 2021, 12, 623893. [Google Scholar] [CrossRef]
- Wan, Q.Q.; Qin, W.P.; Ma, Y.X.; Shen, M.J.; Li, J.; Zhang, Z.B.; Chen, J.H.; Tay, F.R.; Niu, L.N.; Jiao, K. Crosstalk between Bone and Nerves within Bone. Adv Sci 2021, 8, 2003390. [Google Scholar] [CrossRef]
- Elefteriou, F. Regulation of bone remodeling by the central and peripheral nervous system. Arch Biochem Biophys 2008, 473, 231–236. [Google Scholar] [CrossRef] [PubMed]
- Liu, S.; Jin, D.; Wu, J.Q.; Xu, Z.Y.; Fu, S.; Mei, G.; Zou, Z.L.; Ma, S.H. Neuropeptide Y stimulates osteoblastic differentiation and VEGF expression of bone marrow mesenchymal stem cells related to canonical Wnt signaling activating in vitro. Neuropeptides 2016, 56, 105–113. [Google Scholar] [CrossRef] [PubMed]
- Park, M.H.; Jin, H.K.; Min, W.K.; Lee, W.W.; Lee, J.E.; Akiyama, H.; Herzog, H.; Enikolopov, G.N.; Schuchman, E.H.; Bae, J.S. Neuropeptide Y regulates the hematopoietic stem cell microenvironment and prevents nerve injury in the bone marrow. EMBO J 2015, 34, 1648–1660. [Google Scholar] [CrossRef] [PubMed]
- He, H.; Chai, J.; Zhang, S.; Ding, L.; Yan, P.; Du, W.; Yang, Z. CGRP may regulate bone metabolism through stimulating osteoblast differentiation and inhibiting osteoclast formation. Mol Med Rep 2016, 13, 3977–3984. [Google Scholar] [CrossRef] [PubMed]
- Fu, S.; Mei, G.; Wang, Z.; Zou, Z.L.; Liu, S.; Pei, G.X.; Bi, L.; Jin, D. Neuropeptide Substance P Improves Osteoblastic and Angiogenic Differentiation Capacity of Bone Marrow Stem Cells In Vitro. Biomed Res Int 2014, 2014, 596023. [Google Scholar] [CrossRef] [PubMed]
- Appelt, J.; Baranowsky, A.; Jahn, D.; Yorgan, T.; Köhli, P.; Otto, E.; Farahani, S.K.; Graef, F.; Fuchs, M.; Herrera, A.; et al. The neuropeptide calcitonin gene-related peptide alpha is essential for bone healing. EBioMedicine 2020, 59, 102970. [Google Scholar] [CrossRef]
- Asaumi, K.; Nakanishi, T.; Asahara, H.; Inoue, H.; Takigawa, M. Expression of neurotrophins and their receptors (TRK) during fracture healing. Bone 2000, 26, 625–633. [Google Scholar] [CrossRef] [PubMed]
- Gustafsson, D.; Klang, A.; Thams, S.; Rostami, E. The Role of BDNF in Experimental and Clinical Traumatic Brain Injury. Int J Mol Sci 2021, 22, 3582. [Google Scholar] [CrossRef] [PubMed]
- Feliciano, D.P.; Sahbaie, P.; Shi, X.; Klukinov, M.; Clark, J.D.; Yeomans, D.C. Nociceptive sensitization and BDNF up-regulation in a rat model of traumatic brain injury. Neurosci Lett 2014, 583, 55–59. [Google Scholar] [CrossRef]
- Failla, M.D.; Conley, Y.P.; Wagner, A.K. Brain-Derived Neurotrophic Factor (BDNF) in Traumatic Brain Injury–Related Mortality: Interrelationships Between Genetics and Acute Systemic and Central Nervous System BDNF Profiles. Neurorehabilit Neural Repair 2015, 30, 83–93. [Google Scholar] [CrossRef] [PubMed]
- Ida-Yonemochi, H.; Yamada, Y.; Yoshikawa, H.; Seo, K. Locally Produced BDNF Promotes Sclerotic Change in Alveolar Bone after Nerve Injury. PLOS ONE 2017, 12, e0169201. [Google Scholar] [CrossRef]
- Kilian, O.; Hartmann, S.; Dongowski, N.; Karnati, S.; Baumgart-Vogt, E.; Härtel, F.V.; Noll, T.; Schnettler, R.; Lips, K.S. BDNF and its TrkB receptor in human fracture healing. Ann Anat 2014, 196, 286–295. [Google Scholar] [CrossRef]
- Li, Y.; Li, F.; Qin, D.; Chen, H.; Wang, J.; Wang, J.; Song, S.; Wang, C.; Wang, Y.; Liu, S.; et al. The role of brain derived neurotrophic factor in central nervous system. Front Aging Neurosci 2022, 14, 986443. [Google Scholar] [CrossRef]
- Miranda, M.; Morici, J.F.; Zanoni, M.B.; Bekinschtein, P. Brain-Derived Neurotrophic Factor: A Key Molecule for Memory in the Healthy and the Pathological Brain. Front Cell Neurosci 2019, 13, 363. [Google Scholar] [CrossRef]
- Kauschke, V.; Gebert, A.; Calin, M.; Eckert, J.; Scheich, S.; Heiss, C.; Lips, K.S. Effects of new beta-type Ti-40Nb implant materials, brain-derived neurotrophic factor, acetylcholine and nicotine on human mesenchymal stem cells of osteoporotic and non osteoporotic donors. PLOS ONE 2018, 13, e0193468. [Google Scholar] [CrossRef]
- Feng, X.; Lin, T.; Liu, X.; Yang, C.; Yang, S.; Fu, D. Long non-coding RNA BDNF-AS modulates osteogenic differentiation of bone marrow-derived mesenchymal stem cells. Mol Cell Biochem 2018, 445, 59–65. [Google Scholar] [CrossRef]
- Bouxsein, M.L.; Boyd, S.K.; Christiansen, B.A.; Guldberg, R.E.; Jepsen, K.J.; Muller, R. Guidelines for assessment of bone microstructure in rodents using micro-computed tomography. J Bone Miner Res 2010, 25, 1468–1486. [Google Scholar] [CrossRef] [PubMed]
- Piao, J.; Park, J.S.; Hwang, D.Y.; Son, Y.; Hong, H.S. Substance P blocks ovariectomy-induced bone loss by modulating inflammation and potentiating stem cell function. Aging 2020, 12, 20753–20777. [Google Scholar] [CrossRef]
- Lin, P.H.; Kuo, L.T.; Luh, H.T. The Roles of Neurotrophins in Traumatic Brain Injury. Life 2022, 12, 26. [Google Scholar] [CrossRef]
- Su, Y.W.; Zhou, X.F.; Foster, B.K.; Grills, B.L.; Xu, J.; Xian, C.J. Roles of neurotrophins in skeletal tissue formation and healing. J Cell Physiol 2018, 233, 2133–2145. [Google Scholar] [CrossRef] [PubMed]
- Liu, Q.; Lei, L.; Yu, T.; Jiang, T.; Kang, Y. Effect of Brain-Derived Neurotrophic Factor on the Neurogenesis and Osteogenesis in Bone Engineering. Tissue Eng Part A 2018, 24, 1283–1292. [Google Scholar] [CrossRef]
- Kajiya, M.; Shiba, H.; Fujita, T.; Ouhara, K.; Takeda, K.; Mizuno, N.; Kawaguchi, H.; Kitagawa, M.; Takata, T.; Tsuji, K.; Kurihara, H. Brain-derived Neurotrophic Factor Stimulates Bone/Cementum-related Protein Gene Expression in Cementoblasts. J Biol Chem 2008, 283, 16259–16267. [Google Scholar] [CrossRef]
- Xiong, J.; Liao, J.; Liu, X.; Zhang, Z.; Adams, J.; Pacifici, R.; Ye, K. A TrkB agonist prodrug prevents bone loss via inhibiting asparagine endopeptidase and increasing osteoprotegerin. Nat Commun 2022, 13, 4820. [Google Scholar] [CrossRef] [PubMed]
- Tencerova, M.; Kassem, M. The Bone Marrow-Derived Stromal Cells: Commitment and Regulation of Adipogenesis. Front Endocrinol 2016, 7, 127. [Google Scholar] [CrossRef] [PubMed]
- Tantikanlayaporn, D.; Wichit, P.; Suksen, K.; Suksamrarn, A.; Piyachaturawat, P. Andrographolide modulates OPG/RANKL axis to promote osteoblastic differentiation in MC3T3-E1 cells and protects bone loss during estrogen deficiency in rats. Biomed Pharmacother 2020, 131, 110763. [Google Scholar] [CrossRef] [PubMed]
- Loy, T.L.; Vehlow, D.; Kauschke, V.; Muller, M.; Heiss, C.; Lips, K.S. Effects of BDNF and PEC Nanoparticles on Osteocytes. Molecules 2020, 25, 4151. [Google Scholar] [CrossRef] [PubMed]
- Jensen, E.D.; Gopalakrishnan, R.; Westendorf, J.J. Regulation of gene expression in osteoblasts. BioFactors 2010, 36, 25–32. [Google Scholar] [CrossRef]
- Park, K.H.; Choi, Y.; Yoon, D.S.; Lee, K.M.; Kim, D.; Lee, J.W. Zinc Promotes Osteoblast Differentiation in Human Mesenchymal Stem Cells Via Activation of the cAMP-PKA-CREB Signaling Pathway. Stem Cells Dev 2018, 27, 1125–1135. [Google Scholar] [CrossRef]
- Liu, L.; Wang, D.; Qin, Y.; Xu, M.; Zhou, L.; Xu, W.; Liu, X.; Ye, L.; Yue, S.; Zheng, Q.; Li, D. Astragalin Promotes Osteoblastic Differentiation in MC3T3-E1 Cells and Bone Formation in vivo. Front Endocrinol 2019, 10, 228. [Google Scholar] [CrossRef]
- Boyce, B.F.; Xing, L. Functions of RANKL/RANK/OPG in bone modeling and remodeling. Arch Biochem Biophys 2008, 473, 139–146. [Google Scholar] [CrossRef] [PubMed]
- Marcadet, L.; Bouredji, Z.; Argaw, A.; Frenette, J. The Roles of RANK/RANKL/OPG in Cardiac, Skeletal, and Smooth Muscles in Health and Disease. Front Cell Dev Biol 2022, 10, 903657. [Google Scholar] [CrossRef]
- Greenblatt, M.B.; Shim, J.H.; Glimcher, L.H. Mitogen-Activated Protein Kinase Pathways in Osteoblasts. Annu Rev Cell Dev Biol 2013, 29, 63–79. [Google Scholar] [CrossRef]
- Kim, B.S.; Kang, H.J.; Park, J.Y.; Lee, J. Fucoidan promotes osteoblast differentiation via JNK- and ERK-dependent BMP2–Smad 1/5/8 signaling in human mesenchymal stem cells. Exp Mol Med 2015, 47, e128. [Google Scholar] [CrossRef]
- Rodríguez-Carballo, E.; Gámez, B.; Ventura, F. p38 MAPK Signaling in Osteoblast Differentiation. Front Cell Dev Biol 2016, 4, 40. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Z.; Zhang, Y.; Zhou, Z.; Shi, H.; Qiu, X.; Xiong, J.; Chen, Y. BDNF regulates the expression and secretion of VEGF from osteoblasts via the TrkB/ERK1/2 signaling pathway during fracture healing. Mol Med Rep 2017, 15, 1362–1367. [Google Scholar] [CrossRef] [PubMed]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).



