Submitted:
02 January 2024
Posted:
04 January 2024
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
2.1. Collection of Plant Samples
2.2. Isolation of Endophytic Fungi
2.3. Identification of the Isolates
2.4. Antimicrobial Activity Test
2.5. Phosphate and Zinc oxide Solubilization Assay
2.6. IAA Production Assay
3. Results
3.1. Isolation and Identification of Endophytic Fungi
3.2. Antimicrobial Activity
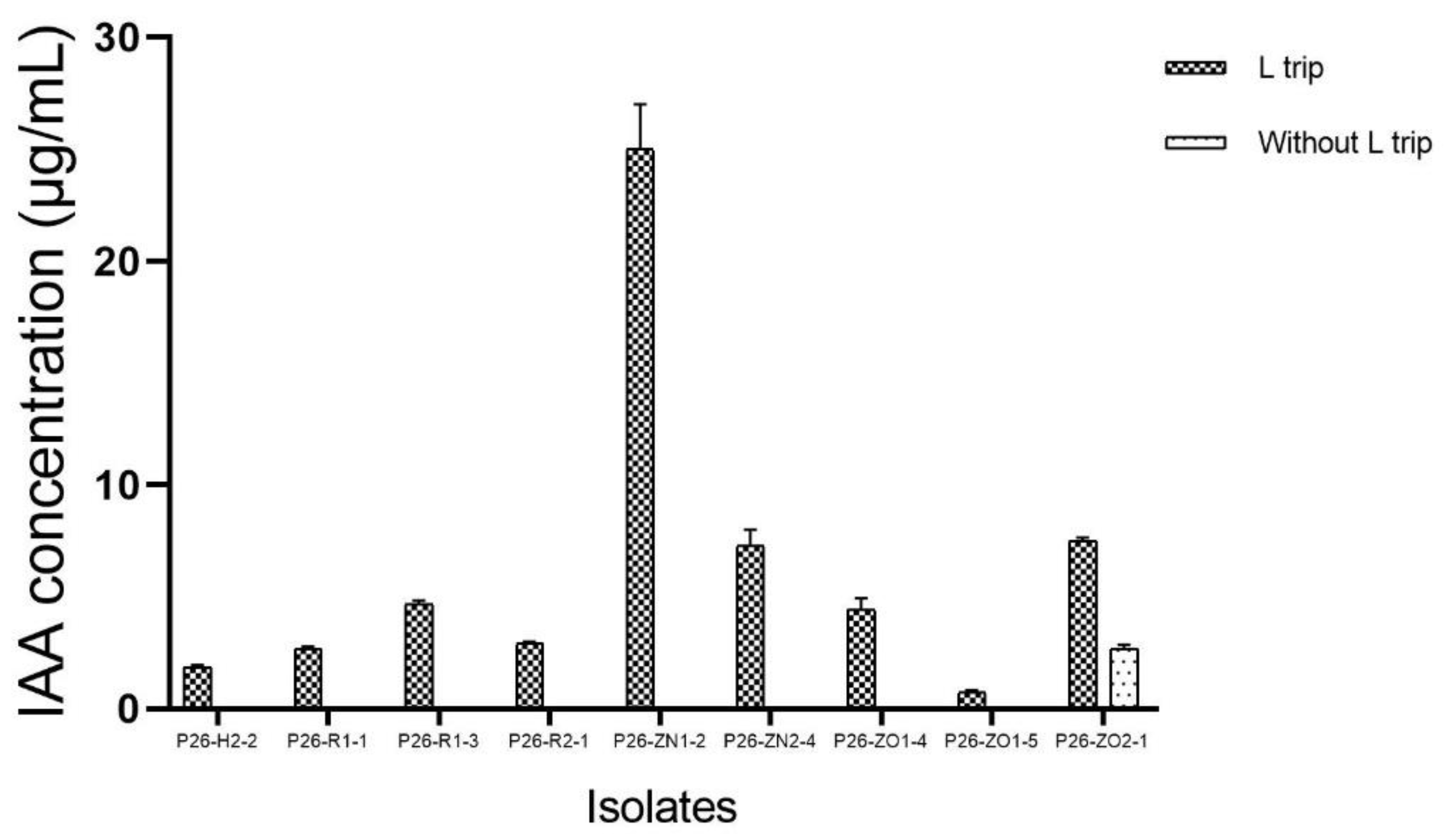
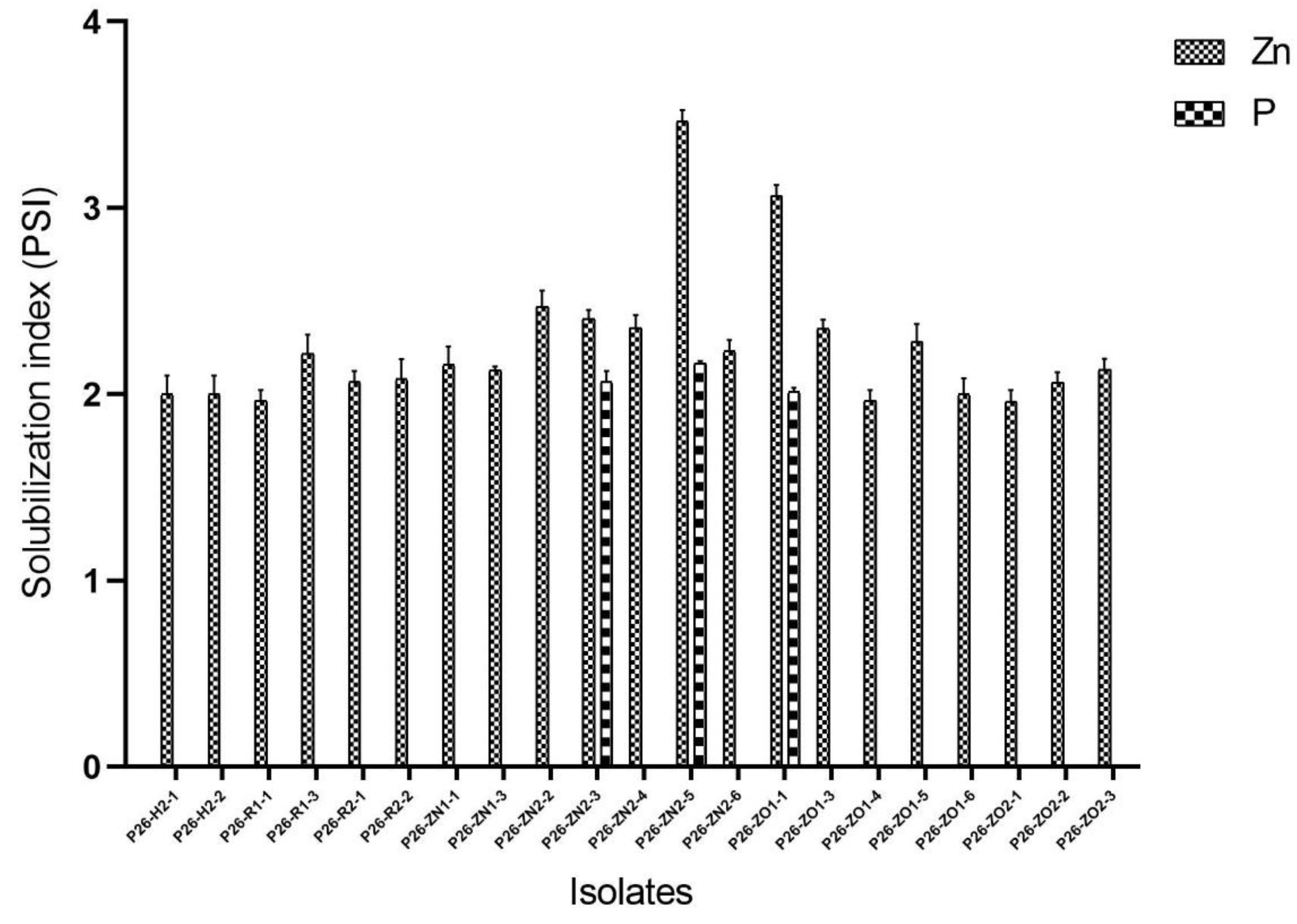
3.3. In Vitro Test for Plant Growth Promoting Traits of Endophytes
4. Discussion
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- CBD Convention on Biological Diversity. United Nations, 1992. Available online: https://www.cbd.int/doc/legal/cbd-en.pdf (accessed on 11 March 2020).
- Law on Genetic Resources of Mongolia. 2021. Available online: https://legalinfo.mn/mn/detail?lawId=16390399395691.
- Climate Change in Mongolia. Outputs from GCM. M. Available online: https://www.env.go.jp/content/900448010.pdf.
- Le Cocq, K.; Gurr, S.J.; Hirsch, P.R.; Mauchline, T.H. Exploitation of endophytes for sustainable agricultural intensification. Mol Plant Pathol. 2017, 18, 469–473. [Google Scholar] [CrossRef]
- Baron, N.C.; Rigobelo, E.C. Endophytic fungi: a tool for plant growth promotion and sustainable agriculture. Mycology 2022, 13, 39–55. [Google Scholar] [CrossRef]
- Bamisile, B.S.; Dash, C.K.; Akutse, K.S.; Keppanan, R.; Wang, L. Fungal Endophytes: Beyond Herbivore Management. Front Microbiol. 2018, 9, 544. [Google Scholar] [CrossRef]
- Rana, K.L.; Kour, D.; Kaur, T.; Devi, R.; Yadav, A.N.; Yadav, N.; Dhaliwal, H.S.; Saxena, A.K. Endophytic microbes: biodiversity, plant growth-promoting mechanisms and potential applications for agricultural sustainability. Anton Leeuw Int J G. 2020, 113, 1075–1107. [Google Scholar] [CrossRef]
- Khan, M.S. , Zaidi, A., Ahemad, M., Oves, M., Wani, P.A. Plant growth promotion by phosphate solubilizing fungi–current perspective. Arch Agron Soil Sci. 2010, 56, 73–98. [Google Scholar] [CrossRef]
- Upadhayay, V.K.; Singh, A.V.; Khan, A.; Sharma, A. Contemplating the role of zinc-solubilizing bacteria in crop biofortification: an approach for sustainable bioeconomy. Front Agron. 2022, 72, 1–13. [Google Scholar] [CrossRef]
- Gouda, S.; Das, G.; Sen, S.K.; Shin, H.S.; Patra, J.K. Endophytes: A Treasure House of Bioactive Compounds of Medicinal Importance. Front Microbiol. 2016, 7, 1538. [Google Scholar] [CrossRef] [PubMed]
- Strobel, G.A.; Daisy, B. Bioprospecting for microbial endophytes and their natural products. Microbiol. Mol. Biol. Rev. 2003, 2003. 67, 491–502. [Google Scholar] [CrossRef]
- Alam, B.; Lǐ, J.; Gě, Q.; Khan, M.A.; Gōng, J.; Mehmood, S.; Yuán, Y.; Gǒng, W. Endophytic Fungi: From Symbiosis to Secondary Metabolite Communications or Vice Versa? Front. Plant Sci. 2021, 12, 791033. [Google Scholar] [CrossRef] [PubMed]
- Caruso, D.J.; Palombo, E.A.; Moulton, S.E.; Zaferanloo, B. Exploring the Promise of Endophytic Fungi: A Review of Novel Antimicrobial Compounds. Microorganisms 2022, 10, 1990. [Google Scholar] [CrossRef] [PubMed]
- Jha, P.; Kaur, T.; Chhabra, I.; Panja, A.; Paul, S.; Kumar, V.; Malik, T. Endophytic fungi: hidden treasure chest of antimicrobial metabolites interrelationship of endophytes and metabolites. Front Microbiol. 2023, 14, 1227830. [Google Scholar] [CrossRef] [PubMed]
- Strobel, G. The emergence of endophytic microbes and their biological promise. J. Fungi 2018, 4, 57. [Google Scholar] [CrossRef] [PubMed]
- Tuvaanjav, S.; Shuqin, H.; Komata, M.; Ma, C.J.; Kanamoto, T.; Nakashima, H.; Yoshida, T. Isolation and antiviral activity of water-soluble Cynomorium songaricum Rupr. polysaccharides. J Asian Nat Prod Res. 2016, 18(2), 159–171. [Google Scholar] [CrossRef] [PubMed]
- 17. Cui, J-L.; Gong, Y.; Xue, X-Z.; Zhang, Y-Y; Wang, M-L.; Wang, J-H. A Phytochemical and Pharmacological Review on Cynomorium songaricum as Functional and Medicinal Food. Nat. Prod. Commun. 2018. 13. [CrossRef]
- Ligaa, U.; Davaasuren, B.; Ninjin, N. Medicinal plants of Mongolia used in Western and Eastern medicine (in Mongolian language), JKC printing: Ulaanbaatar, Mongolia, 2006; pp. 228–229.
- Cui, J.L.; Vijayakumar, V.; Zhang, G. Partitioning of Fungal Endophyte Assemblages in Root-Parasitic Plant Cynomorium songaricum and Its Host Nitraria tangutorum. Front Microbiol. 2018, 9, 666. [Google Scholar] [CrossRef]
- Cui, J.L.; Gong, Y.; Vijayakumar, V.; Zhang, G.; Wang, M.L.; Wang, J.H.; Xue, X.Z. Correlation in chemical metabolome and endophytic mycobiome in Cynomorium songaricum from different desert locations in China. J Agr Food Chem. 2019, 67, 3554–3564. [Google Scholar] [CrossRef] [PubMed]
- Miao, S.M.; Zhang, Y.Y.; Cui, J.L.; Zhang, G. Species and geographic specificity between endophytic fungi and host supported by parasitic Cynomorium songaricum and its host Nitraria tangutorum distributed in desert. Arch Microbiol. 2021, 203, 2511–2519. [Google Scholar] [CrossRef]
- Thi Minh Le, T.; Thi Hong Hoang, A.; Thi Bich Le, T.; Thi Bich Vo, T.; Van Quyen, D.; Hoang Chu, H. Isolation of endophytic fungi and screening of Huperzine A–producing fungus from Huperzia serrata in Vietnam. Sci Rep. 2019, 9, 16152. [Google Scholar] [CrossRef] [PubMed]
- White, T.J.; Bruns, T.; Lee, S.; Taylor, J. Amplification and direct sequencing of fungal ribosomal RNA genes for phylogenetics. In PCR Protocols: A Guide to Methods and Applications; Innis, M.A., Gelfand, D.H., Sninsky, J.J., White, T.J., Eds.; Academic Press, Inc.: New York, NY, USA, 1990; pp. 315–322. ISBN 978-0-12-372180-8. [Google Scholar]
- Lv, Y.L.; Zhang, F.S.; Chen, J.; Cui, J.L.; Xing, Y.M.; Li, X.D.; Guo, S.X. Diversity and antimicrobial activity of endophytic fungi associated with the alpine plant Saussurea involucrata. Biol Pharm Bull. 2010, 33, 1300–1306. [Google Scholar] [CrossRef]
- Balouiri, M.; Sadiki, M.; Ibnsouda, S.K. Methods for in vitro evaluating antimicrobial activity: A review. J Pharm Anal. 2016, 6, 71–79. [Google Scholar] [CrossRef]
- Tan, Xm.; Zhou, Yq.; Zhou, Xl. Diversity and bioactive potential of culturable fungal endophytes of Dysosma versipellis; a rare medicinal plant endemic to China. Sci Rep. 2018, 8, 5929. [Google Scholar] [CrossRef]
- Ibrahim, D.; Lee, C.C.; Sheh-Hong, L. Antimicrobial Activity of Endophytic Fungi Isolated from Swietenia macrophylla Leaves. Nat. Prod. Commun. 2014, 9, 1934578X1400900. [Google Scholar] [CrossRef]
- Senthil Kumar, C.M.; Jacob, T.K.; Devasahayam, S.; Thomas, S.; Geethu, C. Multifarious plant growth promotion by an entomopathogenic fungus Lecanicillium psalliotae. Microbiol. Res. 2018, 207, 153–160. [Google Scholar] [CrossRef] [PubMed]
- Rajini, S.B.; Nandhini, M.; Udayashankar, A.C.; Niranjana, S.R.; Lund, O.S.; Prakash, H.S. Diversity, plant growth-promoting traits, and biocontrol potential of fungal endophytes of Sorghum bicolor. Plant Pathol. [CrossRef]
- Saikkonen, K. Forest structure and fungal endophytes. Fungal Biol. Rev. 2007, 21, pp–67. [Google Scholar] [CrossRef]
- Mohammad Golam Dastogeer, K.; Oshita, Y.; Yasuda, M.; Kanasugi, M.; Matsuura, E.; Xu, Q.; Okazaki, S. Host Specificity of Endophytic Fungi from Stem Tissue of Nature Farming Tomato (Solanum lycopersicum Mill.) in Japan. Agronomy 2020, 10, 1019. [Google Scholar] [CrossRef]
- Dastogeer, K.M.G.; Li, H.; Sivasithamparam, K.; Jones, M.G.K.; Wylie, S.J. Host Specificity of Endophytic Mycobiota of Wild Nicotiana Plants from Arid Regions of Northern Australia. Microbial ecology 2018, 75, 74–87. [Google Scholar] [CrossRef]
- Fan, S., Miao, L., Li, H., Lin, A., Song, F., Zhang, P. Illumina-based analysis yields new insights into the diversity and composition of endophytic fungi in cultivated Huperzia serrata. PLoS ONE 2020, 15 (11), e0242258. [CrossRef]
- Lucking, R.; Aime, M.C. , Robbertse, B. et al. Unambiguous identification of fungi: where do we stand and how accurate and precise is fungal DNA barcoding? IMA fungus 2020, 11, 14. [Google Scholar] [CrossRef]
- Paul, N.C.; Yu, S.H. Endophytic fungi from medicinal plants in Korea. Lap Lambert Academic Publishing; Saarbrücken, Germany, 2011.
- Guevara-Araya, M.J.; Vilo, C.; Urzúa, A. Differences in community composition of endophytic fungi between above- and below-ground tissues of Aristolochia chilensis in an arid ecosystem. Rev. Chil. de Hist. Nat. 2020, 93, 3. [Google Scholar] [CrossRef]
- Gams, W.; Diederich, P.; Poldmaa, K. Fungicolous fungi. In: Biodiversity of Fungi: Inventory and Monitoring Methods, eds Mueller, G.M.; Bills, G.F.; Foster, M.S. Elsevier Academic Press: Burlington, MA, pp 343–392. [CrossRef]
- Dan, F.J.; Dubey, Mukesh, D.; Birgit, J., Magnus, K. Clonostachys rosea to control plant diseases. Burleigh Dodds Science Publishing: UK. [CrossRef]
- Han, P.; Zhang, X.; Xu, D.; Zhang, B.; Lai, D.; Zhou, L. Metabolites from Clonostachys Fungi and Their Biological Activities. Journal of fungi (Basel, Switzerland) 2020, 6, 229. [Google Scholar] [CrossRef]
- Qiao, H.; Sun, X. R.; Wu, X.Q.; Li, G.E.; Wang, Z.; Li, D.W. The phosphate-solubilizing ability of Penicillium guanacastense and its effects on the growth of Pinus massoniana in phosphate-limiting conditions. Biology open 2019, 8, bio046797. [Google Scholar] [CrossRef]
- Wakelin, S.A.; Warren, R.A.; Harvey, P.R. Phosphate solubilization by Penicillium spp. closely associated with wheat roots. Biol Fertil Soils 2004, 40, 36–43. [Google Scholar] [CrossRef]
- Qiao, H.; Sun, X.R.; Wu, X. Q.; Li, G.E.; Wang, Z.; Li, D.W. The phosphate-solubilizing ability of Penicillium guanacastense and its effects on the growth of Pinus massoniana in phosphate-limiting conditions. Biology open 2019, 8, bio046797. [Google Scholar] [CrossRef]
- Doilom, M.; Guo, J.-W.; Phookamsak, R.; Mortimer, P.E.; Karunarathna, S.C.; Dong, W.; Liao, C.-F.; Yan, K.; Pem, D.; Suwannarach, N.; et al. Screening of Phosphate-Solubilizing Fungi from Air and Soil in Yunnan, China: Four Novel Species in Aspergillus, Gongronella, Penicillium, and Talaromyces. Front. Microbiol 2020, 11(585215). [Google Scholar] [CrossRef] [PubMed]
- Devi, D.; Gupta, S.B.; Mishra, B.K.; Verma, N.P. Isolation and identification of zinc solubilizing fungal isolates from cumin of semi-arid region of Rajasthan. The Pharma Innovation 2022, 11, 1036–1040. [Google Scholar]
- de Hoog, G.S.; van Diepeningen, A.D; Mahgoub, E-S.; van de Sande, W.W.J. New Species of Madurella, Causative Agents of Black-Grain Mycetoma. J Clin Microbiol. 2012, 50, 988 – 994. [CrossRef]
- Pushpa, H.; Kavya, S.; Pooja, K.; Sneha, L.; Arer, V.O. An isolation, identification and diversity of endophytic fungi from Catharanthus roseus and screening for their L-asparaginase activity. Int J Env Ecol Fam Urban Stud 2018, 8(7). Available online: https://issuu.com/tjprc/docs/2.ijeefusdec20182.
- Hapida, Y. ; Elfita.; Widjajanti, H.; Salni. Biodiversity and antibacterial activity of endophytic fungi isolated from jambu bol (Syzygium malaccense). Biodiversitas J Biol Diversity, 5677. [Google Scholar] [CrossRef]
- Orole, O.O.; Adejumo, T.O.; Link, T.; Voegele, R.T. Molecular identification of endophytes from maize roots and their biocontrol potential against toxigenic fungi of Nigerian maize. Science progress 2023, 106, 368504231186514. [Google Scholar] [CrossRef]


| Strain | Accession No | Plant part | Species with most homologous sequence (Accession No) | Similarity % |
|---|---|---|---|---|
| P26-H1-1 | LC769420 | Aboveground |
Fusarium equiseti CB33-4 (MT558601) |
99.81 |
| P26-H1-3 | LC769421 | Aboveground |
Clonostachys rosea MR44 (KY320599) |
99.65 |
| P26-H2-1 | LC769422 | Aboveground |
Fusarium solani GBC-Fungus 27 (MN077430) |
100 |
| P26-H2-2 | LC769423 | Aboveground |
Fusarium solani N-49-1 (MT560378) |
100 |
| P26-R1-1 | LC769424 | Underground |
Fusarium equiseti NL-374-D (OQ561206) |
99.63 |
| P26-R1-3 | LC769425 | Underground |
Alternaria sp. INM5 (KY781740) |
99.30 |
| P26-R2-1 | LC769426 | Underground |
Fusarium solani N-13-2 (MT560338) |
100 |
| P26-R2-2 | LC769427 | Underground |
Clonostachys rosea MR44 (KY320599) |
99.13 |
| P26-ZN1-1 | LC769428 | Fresh rhizome |
Penicillium chrysogenum MZC-0 (MN069559) |
99.66 |
| P26-ZN1-2 | LC769429 | Fresh rhizome |
Madurella fahalii 332- pus (OQ421454) |
98.96 |
| P26-ZN1-3 | LC769430 | Fresh rhizome |
Aspergillus tabacinus fung8 (MT635280) |
100 |
| P26-ZN2-2 | LC769431 | Fresh rhizome | Penicillium chrysogenum MZC-0 (MN069559) | 99.83 |
| P26-ZN2-3 | LC769432 | Fresh rhizome | Penicillium roseopurpureum IHEM:28005 (OU989457) | 99.83 |
| P26-ZN2-4 | LC769433 | Fresh rhizome |
Fusarium sp. GFR18 (MT447523) |
100 |
| P26-ZN2-5 | LC769434 | Fresh rhizome |
Penicillium vinaceum 533 (DQ681340) |
100 |
| P26-ZN2-6 | LC769435 | Fresh rhizome | Penicillium roseopurpureum G5-2 (MN206951) | 100 |
| P26-ZO1-1 | LC769436 | Old rhizome |
Penicillium sp. FP-027-A7 (MH102087) |
99.49 |
| P26-ZO1-3 | LC769437 | Old rhizome |
Clonostachys rosea daef27 (MH550497) |
99.12 |
| P26-ZO1-4 | LC663164 | Old rhizome |
Clonostachys rosea Potato root (MT448899) |
100 |
| P26-ZO1-5 | LC769438 | Old rhizome |
Clonostachys sp. 1R1D (OR365747) |
99.82 |
| P26-ZO1-6 | LC769439 | Old rhizome |
Fusarium sp. GFR18 (MT447523) |
99.82 |
| P26-ZO2-1 | LC769440 | Old rhizome |
Fusarium proliferatum CBB-6 (MT560216) |
100 |
| P26-ZO2-2 | LC769441 | Old rhizome |
Clonostachys rosea MR44 (KY320599) |
99.83 |
| P26-ZO2-3 | LC769442 | Old rhizome |
Clonostachys rosea N25 (MH259861) |
100 |
| Strain | Taxa | Escherichia coli | Bacillus subtilis | Staphylococcus aureus | Candida albicans | Aspergillus niger |
|---|---|---|---|---|---|---|
| P26-H1-1 | Fusarium equiseti | - | 15.5±0.7 | 12±0 | - | - |
| P26-H1-3 | Clonostachys rosea | - | 10.5±0.7 | - | - | 15.5±0.7 |
| P26-H2-1 | Fusarium proliferatum | - | 11.5±1.4 | 13±0.7 | - | - |
| P26-H2-2 | Fusarium solani | - | - | 10.5±0.7 | - | 23±2.8 |
| P26-R1-1 | Fusarium equiseti | - | 15.5±0.7 | 13.5±0.7 | - | - |
| P26-R2-1 | Fusarium solani | - | - | - | - | 22.5±0.7 |
| P26-R2-2 | Clonostachys rosea | - | - | 7.5±2.1 | - | - |
| P26-ZN1-2 | Madurella fahalii | 7.5±0.7 | 9.5±2.1 | 15.5±0.7 | - | - |
| P26-ZN1-3 | Aspergillus amoenus | - | 8±1.4 | 7±0 | - | - |
| P26-ZO1-3 | Clonostachys rosea | - | 15±0 | 18.5±0.7 | - | 13.5±2.1 |
| P26-ZO1-4 | Clonostachys rosea | - | 9±4.2 | 9.5±0.7 | - | 12.5±2.1 |
| P26-ZO1-5 | Clonostachys rosea | - | 13.5±0.7 | 16.5±0.7 | - | 14±1.4 |
| P26-ZO1-6 | Fusarium tonkinense | - | 11.5±0.7 | 17.5±0.7 | - | 12±0 |
| P26-ZO2-2 | Clonostachys rosea | - | 12±0 | 15.5±2.1 | - | - |
| P26-ZO2-3 | Clonostachys rosea | - | 13.5±0.7 | 16±0 | - | 13±1.4 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




