Submitted:
04 January 2024
Posted:
04 January 2024
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
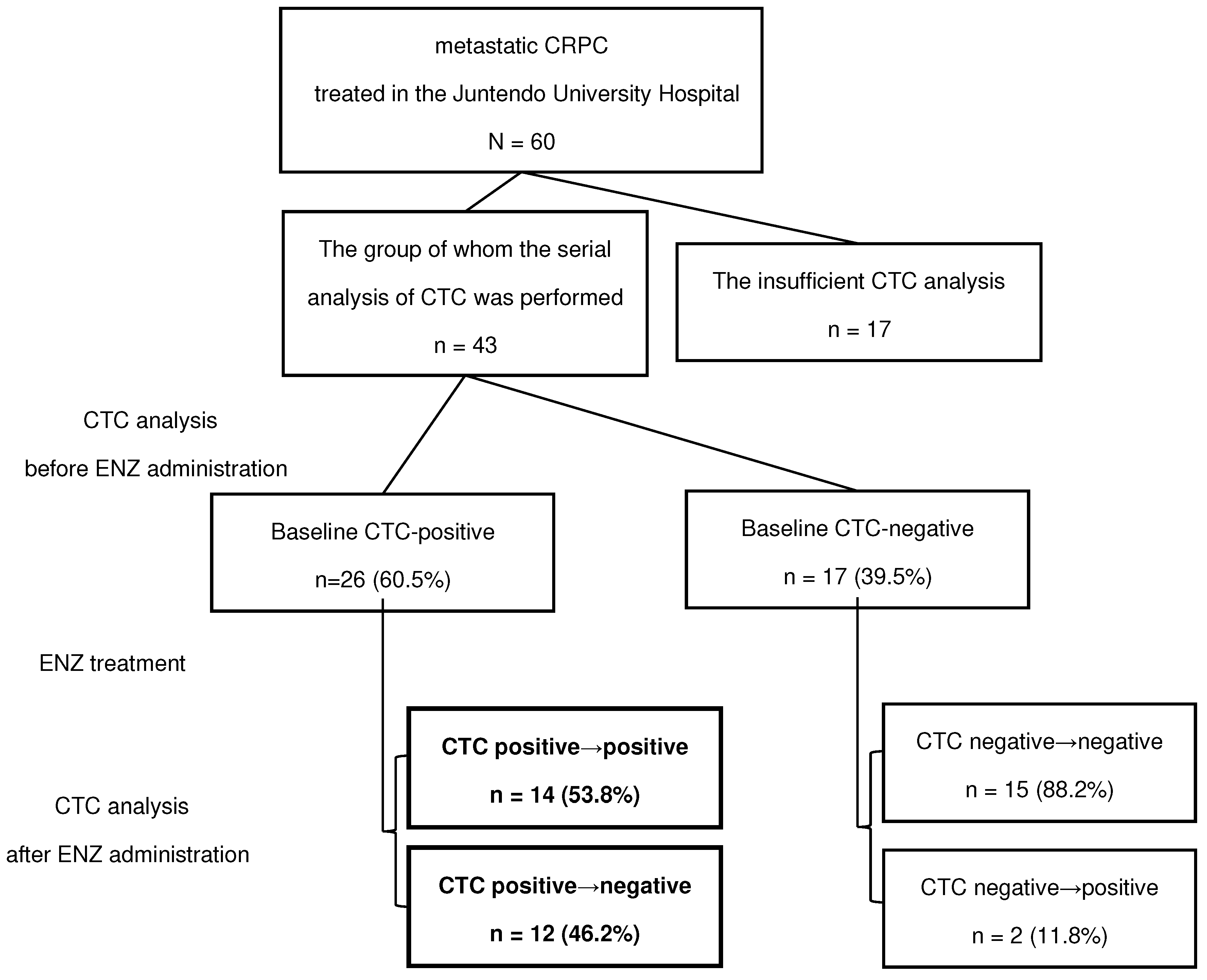
2.1. Patients and Study Design
2.2. CTC Analyses
2.3. Statistical Analyses
2.4. Ethics STATEMENT
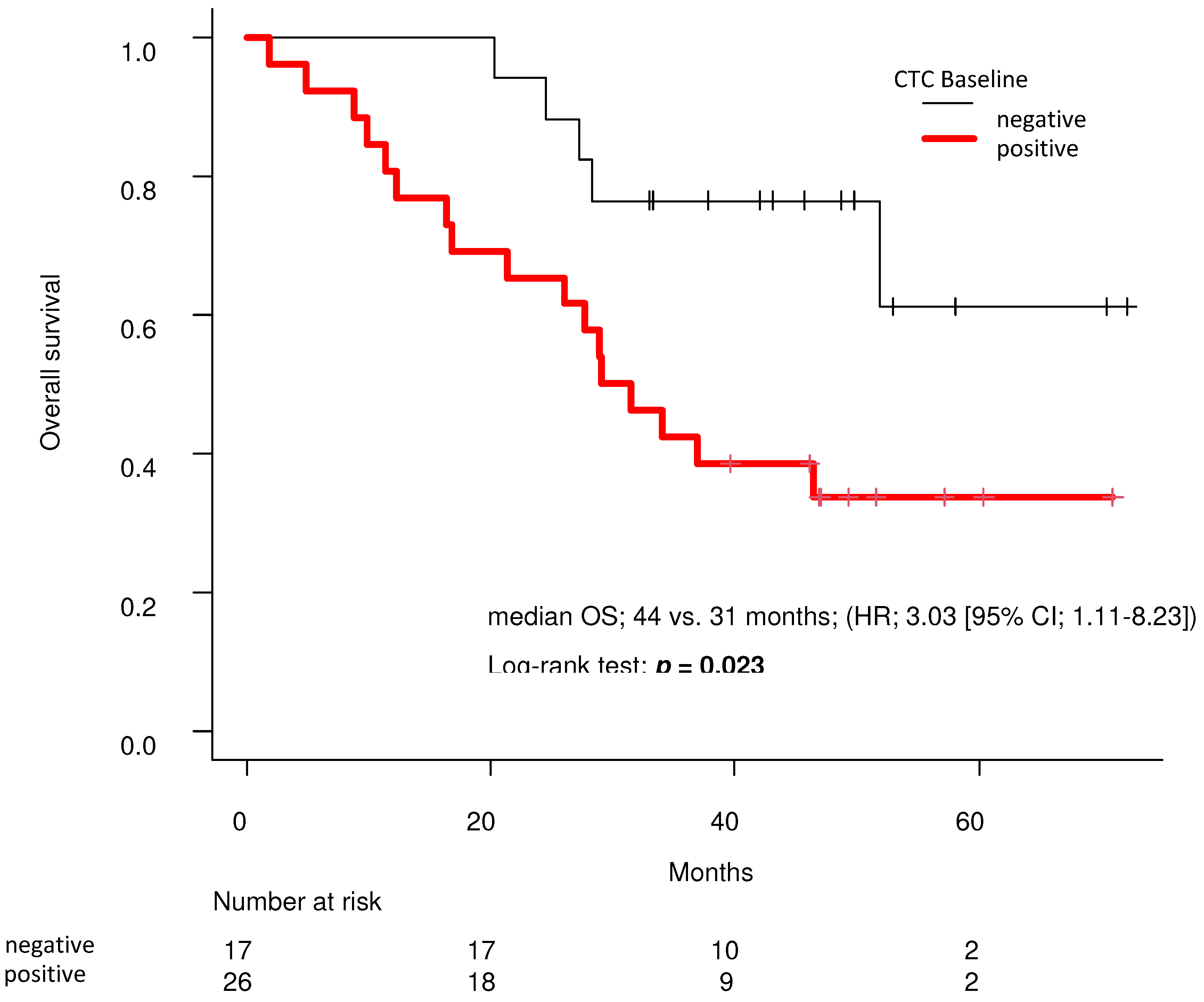
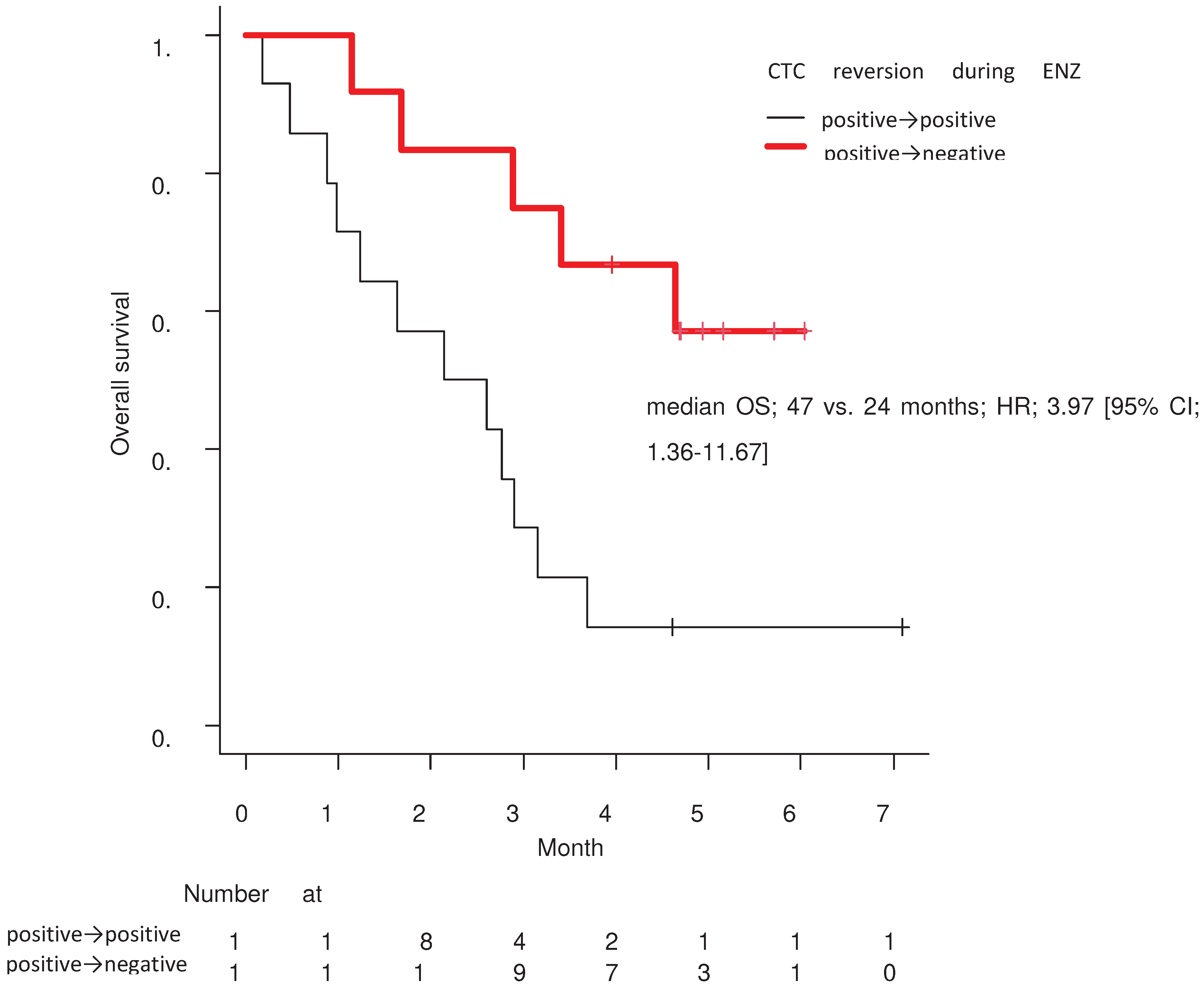
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Tran, C.; Ouk, S.; Clegg, N.J.; Chen, Y.; Watson, P.A.; Arora, V.; et al. Development of a second-generation antiandrogen for treatment of advanced prostate cancer. Science. 2009, 324, 787–90. [Google Scholar] [CrossRef]
- Scher, H.I.; Fizazi, K.; Saad, F.; Taplin, M.E.; Sternberg, C.N.; Miller, K.; et al. Increased survival with enzalutamide in prostate cancer after chemotherapy. N Engl J Med. 2012, 367, 1187–97. [Google Scholar] [CrossRef]
- Beer, T.M.; Armstrong, A.J.; Rathkopf, D.E.; Loriot, Y.; Sternberg, C.N.; Higano, C.S.; et al. Enzalutamide in metastatic prostate cancer before chemotherapy. N Engl J Med. 2014, 371, 424–33. [Google Scholar] [CrossRef]
- Antonarakis, E.S.; Lu, C.; Luber, B.; Wang, H.; Chen, Y.; Zhu, Y.; et al. Clinical significance of androgen receptor splice variant-7 mRNA detection in circulating tumor cells of men with metastatic castration-resistant prostate cancer treated with first- and second-line Abiraterone and Enzalutamide. J Clin Oncol. 2017, 35, 2149–2156. [Google Scholar] [CrossRef]
- Armstrong, A.J.; Garrett-Mayer, E.S.; Yang, Y.C.; de Wit, R.; Tannock, I.F.; Eisenberger, M. A contemporary prognostic nomogram for men with hormone-refractory metastatic prostate cancer: a TAX327 study analysis. Clin Cancer Res. 2007, 13, 6396–403. [Google Scholar] [CrossRef]
- Armstrong, A.J.; Lin, P.; Higano, C.S.; Sternberg, C.N.; Sonpavde, G.; Tombal, B.; et al. Development and validation of a prognostic model for overall survival in chemotherapy-naive men with metastatic castration-resistant prostate cancer. Ann Oncol. 2018, 29, 2200–7. [Google Scholar] [CrossRef] [PubMed]
- Halabi, S.; Kelly, W.K.; Ma, H.; Zhou, H.; Solomon, N.C.; Fizazi, K.; et al. Meta-analysis evaluating the impact of site of metastasis on overall survival in men with castration resistant prostate cancer. J Clin Oncol. 2016, 34, 1652–9. [Google Scholar] [CrossRef] [PubMed]
- Marrugo-Ramirez, J.; Mir, M.; Samitier, J. Blood-based cancer biomarkers in liquid biopsy: A promising non-invasive alternative to tissue biopsy. Int J Mol Sci. 2018, 19, 2877. [Google Scholar] [CrossRef]
- Antonarakis, E.S.; Lu, C.; Wang, H.; Luber, B.; Nakazawa, M.; Roeser, J.C.; et al. AR-V7 and resistance to enzalutamide and abiraterone in prostate cancer. N Engl J Med. 2014, 371, 1028–38. [Google Scholar] [CrossRef] [PubMed]
- Onstenk, W.; Sieuwerts, A.M.; Kraan, J.; Van, M.; Nieuweboer, A.J.; Mathijssen, R.H.; et al. Efficacy of cabazitaxel in castration-resistant prostate cancer is independent of the presence of AR-V7 in circulating tumor cells. Eur Urol. 2015, 68, 939–45. [Google Scholar] [CrossRef] [PubMed]
- Antonarakis, E.S.; Lu, C.; Luber, B.; Wang, H.; Chen, Y.; Nakazawa, M.; et al. Androgen receptor splice variant 7 and efficacy of taxane chemotherapy in patients with metastatic castration-resistant prostate cancer. JAMA Oncol. 2015, 1, 582–91. [Google Scholar] [CrossRef] [PubMed]
- Ashizawa, T.; Nagata, M.; Nakamura, S.; Hirano, H.; Nagaya, N.; Lu, Y.; et al. Efficacy of cabazitaxel and androgen splicing variant-7 status in circulating tumor cells in Asian patients with metastatic castration-resistant prostate cancer. Sci Rep. 2022, 12, 18016. [Google Scholar] [CrossRef] [PubMed]
- Hirano, H.; Nagata, M.; Nagaya, N.; Nakamura, S.; Ashizawa, T.; Lu, Y. Bone Scan Index (BSI) scoring by using bone scintigraphy and circulating tumor cells (CTCs): predictive factors for enzalutamide effectiveness in patients with castration-resistant prostate cancer and bone metastases. Sci Rep. 2023, 13, 8704. [Google Scholar] [CrossRef] [PubMed]
- Nakazawa, M.; Lu, C.; Chen, Y. Serial blood-based analysis of AR-V7 in men with advanced prostate cancer. Ann Oncol. 2015, 26, 1859–1865. [Google Scholar] [CrossRef] [PubMed]
- Nagaya, N.; Kanayama, M.; Nagata, M.; Horie, S. Abiraterone rechallenge based on sequential testing of androgen receptor splice variant 7 expression in circulating tumor cells: A case report. Front Oncol. 2020, 10, 495-8. [Google Scholar] [CrossRef]
- Nagaya, N.; Nagata, M.; Lu, Y.; Kanayama, M.; Hou, Q.; Hotta, Z.; et al. Prostate-specific membrane antigen in circulating tumor cells is a new poor prognostic marker for castration-resistant prostate cancer. PLOS One. 2020, 15, e0226219. [Google Scholar] [CrossRef] [PubMed]
- Jiang, S.; Tang, W.; Huang, B. Longitudinal change of circulating tumour cell count and its relation to prognosis in advanced intrahepatic cholangiocarcinoma patients. Scand J Clin Lab Invest. 2023, 83, 234–240. [Google Scholar] [CrossRef]
- Hristozova, T.; Konschak, R.; Stromberger, C.; Fusi, A.; Liu, Z.; Weichert, W.; et al. The presence of circulating tumor cells (CTCs) correlates with lymph node metastasis in nonresectable squamous cell carcinoma of the head and neck region (SCCHN). Ann Oncol 2011, 22, 1878–1885. [Google Scholar] [CrossRef]
- Armstrong, A.J.; Tannock, I.F.; de Wit, R.; George, D.J.; Eisenberger, M.; Halabi, S.; et al. The development of risk groups in men with metastatic castration-resistant prostate cancer based on risk factors for PSA decline and survival. Eur J Cancer. 2010, 46, 517–25. [Google Scholar] [CrossRef]
- Danila, D.C.; Heller, G.; Gignac, G.A.; Gonzalez-Espinoza, R.; Anand, A.; Tanaka, E.; et al. Circulating tumor cell number and prognosis in progressive castration-resistant prostate cancer. Clin Cancer Res. 2007, 13, 7053–8. [Google Scholar] [CrossRef] [PubMed]
- Cattrini, C.; Rubagotti, A.; Zinoli, L.; Cerbone, L.; Zanardi, E.; Capaia, M.; et al. Role of circulating tumor cells (CTC), androgen receptor full length (AR-FL) and androgen receptor splice variant 7 (AR-V7) in a prospective cohort of castration-resistant metastatic prostate cancer patients. Cancers (Basel). 2019, 11, 1365. [Google Scholar] [CrossRef] [PubMed]
- Heller, G.; McCormack, R.; Kheoh, T.; Molina, A.; Smith, M.R.; Dreicer, R.; et al. Circulating tumor cell number as a response measure of prolonged survival for metastatic castration-resistant prostate cancer: a comparison with prostate-specific antigen across five randomized phase III clinical trials. J Clin Oncol. 2018, 36, 572–580. [Google Scholar] [CrossRef]
- Armstrong, A.J.; Lin, P.; Tombal, B.; Saad, F.; Higano, C.S.; Joshua, A.M.; et al. Five-year survival prediction and safety outcomes with enzalutamide in men with chemotherapy-naïve metastatic castration-resistant prostate cancer from the PREVAIL Trial. Eur Urol. 2020, 78, 347–357. [Google Scholar] [CrossRef] [PubMed]
- Sharp, A.; Welti, J.C.; Lambros, M.B.; Dolling, D.; Rodriguez, D.N.; Pope, L.; et al. Clinical utility of circulating tumour cell androgen receptor splice Variant-7 status in metastatic castration-resistant prostate cancer. Eur Urol. 2019, 76, 676–685. [Google Scholar] [CrossRef] [PubMed]
| All patients | CTC Positive at baseline | CTC Negative at baseline | P-value | |
|---|---|---|---|---|
| Number of patients (n) | 43 | 26 | 17 | |
| Age median (IQR) | 73.0 (69.0 - 68.0) | 74.0 (70.3 - 77.8) | 73.0 (68.0 - 78.0) | 0.524 |
| Gleason sum at diagnosis (6-7/8-10) | 6-7:10 (23%) / 8-10:33 (77%) | 6-7:5 (19%) / 8-10:21 (81%) | 6-7:5 (29%) / 8-10:12 (71%) | 0.481 |
| Body Mass Index median (IQR) | 22.8 (21.0 - 24.5) | 23.0 (21.0 - 25.0) | 22.0 (21.0 - 23.0) | 0.175 |
| Initial PSA median (IQR) (ng/ml) | 119.2 (36.3 - 658.4) | 335.2 (41.5 - 1981.6) | 98.0 (18.3 - 332.4) | 0.162 |
| Baseline Bone Scan Index median (IQR) (%) | 0.44 (0.11 - 1.31) | 0.97 (0.36-1.65) | 0.12 (0.10 - 0.29) | 0.002 |
| Presence of bone metastases at diagnosis (y/n) | Yes 37 (86%) / No 6 (14%) | Yes 22 (85%) / No 4 (15%) | Yes 15 (88%) / No 2 (12%) | >0.999 |
| Presence of lung metastases (y/n) | Yes 4 (9%) / No 39 (91%) | Yes 2 (8%) / No 24 (92%) | Yes 2 (12%) / No 15 (88%) | >0.999 |
| Presence of lymph node metastases (y/n) | Yes 10 (23%)/ No 33 (77%) | Yes 6 (23%) / No 20 (77%) | Yes 4 (24%) / No 13 (76%) | >0.999 |
| Radiation treatment (y/n) | Yes 6 (14%) / No 37 (86%) | Yes 2 (8%) / No 24 (92%) | Yes 4 (24%) / No 13 (76%) | 0.193 |
| Docetaxel before ENZ (y/n) | Yes 4 (9%) / No 39 (91%) | Yes 3 (12%) / No 23 (88%) | Yes 1 (6%) / No 16 (94%) | >0.999 |
| Abiraterone before ENZ (y/n) | Yes 3 (7%) / No 40 (93%) | Yes 2 (8%) / No 24 (92%) | Yes 1 (6%) / No 16 (94%) | >0.999 |
| Baseline alkaline phosphatase median (IQR) (U/L) | 238.0 (158.5 - 354.0) | 264.5 (178.5 - 387.5) | 179.0 (148.0 - 243.0) | 0.068 |
| Baseline PSA median (IQR) (ng/mL) | 9.6 (4.1 - 36.6) | 26.2 (4.7- 80.2) | 4.4 (2.6 - 8.2) | 0.002 |
| Baseline serum calcium median (IQR) (mg/dL) | 9.3 (9.0 - 9.5) | 9.3 (9.0 - 9.7) | 9.3 (9.0 - 9.4) | 0.717 |
| Baseline hemoglobin median (IQR) (mg/dL) | 13.1 (11.6 - 13.7) | 12.6 (11.3 - 13.6) | 13.4 (12.7 - 13.8) | 0.189 |
| Time to CRPC median (IQR) (month) | 16.0 (10.0 - 39.0) | 15.5 (8.3 - 32.5) | 16.0 (13.0 - 42.0) | 0.363 |
| CTC-negative reversion (N=12) | CTC-positive continuation (N=14) | P-value | |
|---|---|---|---|
| Number of patients (n) | 12 | 14 | |
| Age median (IQR) | 72.5 (69.8 - 77.0) | 74.0 (71.0 - 78.8) | 0.174 |
| Gleason sum at diagnosis (6-7/8-10) | 6-7:2 (17%) / 8-10:10 (83%) | 6-7:3 (21%) / 8-10:11 (79%) | >0.999 |
| Body Mass Index median (IQR) | 23.0 (21.5 - 27.2) | 23.0 (21.0 - 23.8) | 0.341 |
| Initial PSA median (IQR) (ng/ml) | 471.5 (37.8 - 2765.1) | 335.2 (50.5 - 1015.0) | 0.354 |
| Baseline Bone Scan Index median (IQR) (%) | 0.50 (0.1 - 1.1) | 1.46 (0.7 - 2.7) | 0.517 |
| Presence of lung metastases (y/n) | Yes 1 (8%) / No 11 (92%) | Yes 1 (7%) / No 13 (93%) | >0.999 |
| Presence of lymph node metastases (y/n) | Yes 0 (0%) / No 12 (100%) | Yes 6 (43%) / No 8 (57%) | 0.017 |
| Radiation treatment (y/n) | Yes 1 (8%) / No 11 (92%) | Yes 0 (0%) / No 14 (100%) | >0.999 |
| Docetaxel before ENZ (y/n) | Yes 1 (8%) / No 11 (92%) | Yes 2 (14%) / No 12 (86%) | >0.999 |
| Abiraterone before ENZ (y/n) | Yes 2 (17%) / No 10 (83%) | Yes 0 (0%) / No 14 (100%) | 0.483 |
| Baseline alkaline phosphatase median (IQR) (U/L) | 267.0 (188.8 - 359.8) | 264.5 (178.5 - 628.3) | 0.132 |
| Baseline PSA median (IQR) (ng/mL) | 18.1 (4.3 - 48.7) | 35.1 (9.6 - 139.2) | 0.318 |
| Baseline serum calcium median (IQR) (mg/dL) | 9.3 (9.1 - 9.7) | 9.2 (8.8 - 9.4) | 0.162 |
| Baseline hemoglobin median (IQR) (mg/dL) | 13.5 (12.2 - 14.4) | 11.9 (11.0 - 12.9) | 0.069 |
| Time to CRPC median (IQR) (month) | 19.0 (11.5 - 38.3) | 10.5 (6.0 - 27.8) | 0.490 |
| Clinical background factors in the group with CTC positive continuation and CTC-negative reversion during ENZ treatment. Fisher’s exact test was used for categorical variables, and the t-test was used for normally distributed continuous variables. The t-test with the natural logarithm of the variables was used for non-normally distributed variables. IQR, Interquartile range; CRPC, castration-resistant prostate cancer; CTC, circulation tumor cell; ENZ, enzalutamide; PSA, prostate-specific antigen. | |||
| Variable | P-value |
|---|---|
| Age (> 74y) | 0.201 |
| Initial PSA (> 335.2ng/mL) | 0.165 |
| Gleason sum at diagnosis (8-10 / 6-7) | 0.306 |
| Time to CRPC (< 15.5month) | 0.702 |
| Presence of bone metastasis at diagnosis (y/n) | 0.478 |
| Presence of lung metastasis (y/n) | 0.749 |
| Presence of lymph node metastases (y/n) | 0.410 |
| Use of denosumab (y/n) | 0.286 |
| Baseline PSA (> 26.2ng/mL) | 0.532 |
| Baseline Bone Scan Index (> 0.97%) | 0.560 |
| Baseline alkaline phosphatase (> 264.5U/L) | 0.235 |
| Baseline serum calcium (> 9.3mg/dL) | 0.268 |
| Baseline hemoglobin (< 12.6mg/dL) | 0.026 |
| PSA at 3 months after ENZ (> 5.1) | 0.511 |
| Bone Scan Index at 3 months after ENZ (> 0.85%) | 0.502 |
| Alkaline phosphatase at 3 months after ENZ (> 219.5U/L) | 0.405 |
| Serum calcium at 3months after ENZ (> 9.3mg/dL) | 0.780 |
| Hemoglobin at 3months after ENZ (< 12.9mg/dL) | 0.085 |
| Positive-continuation / CTC-negative reversion | 0.007 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




