Submitted:
01 January 2024
Posted:
03 January 2024
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Experimental Procedures
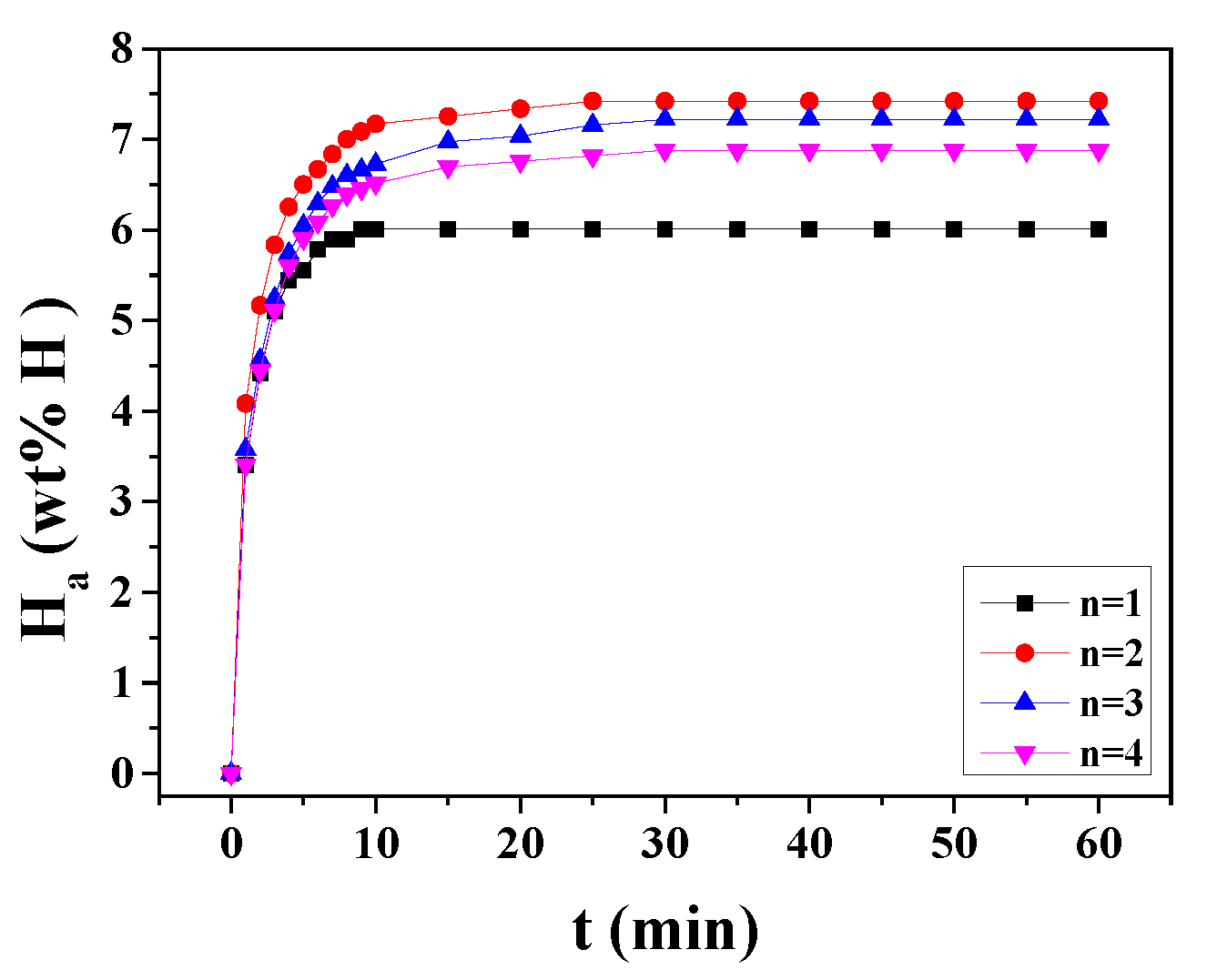
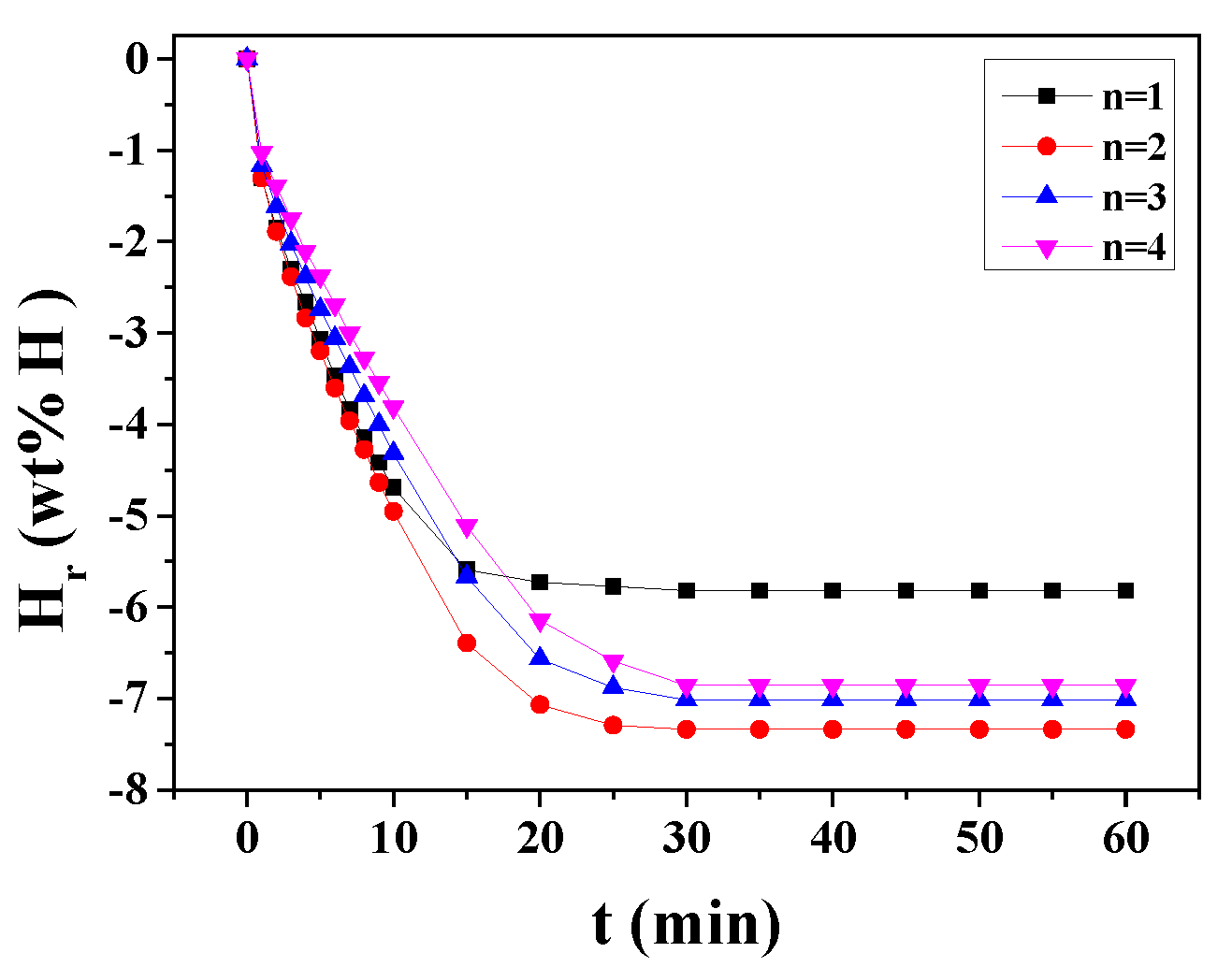
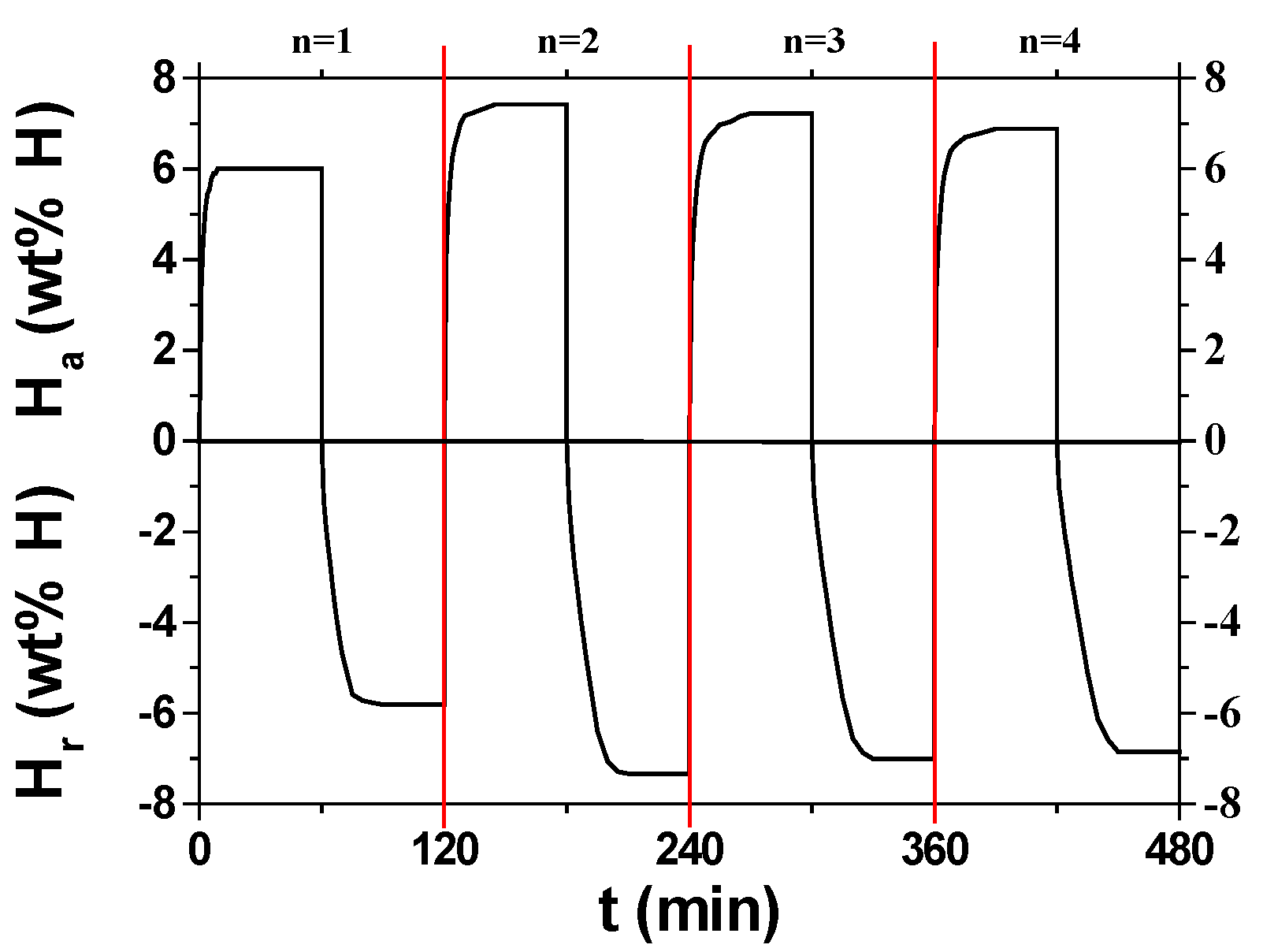
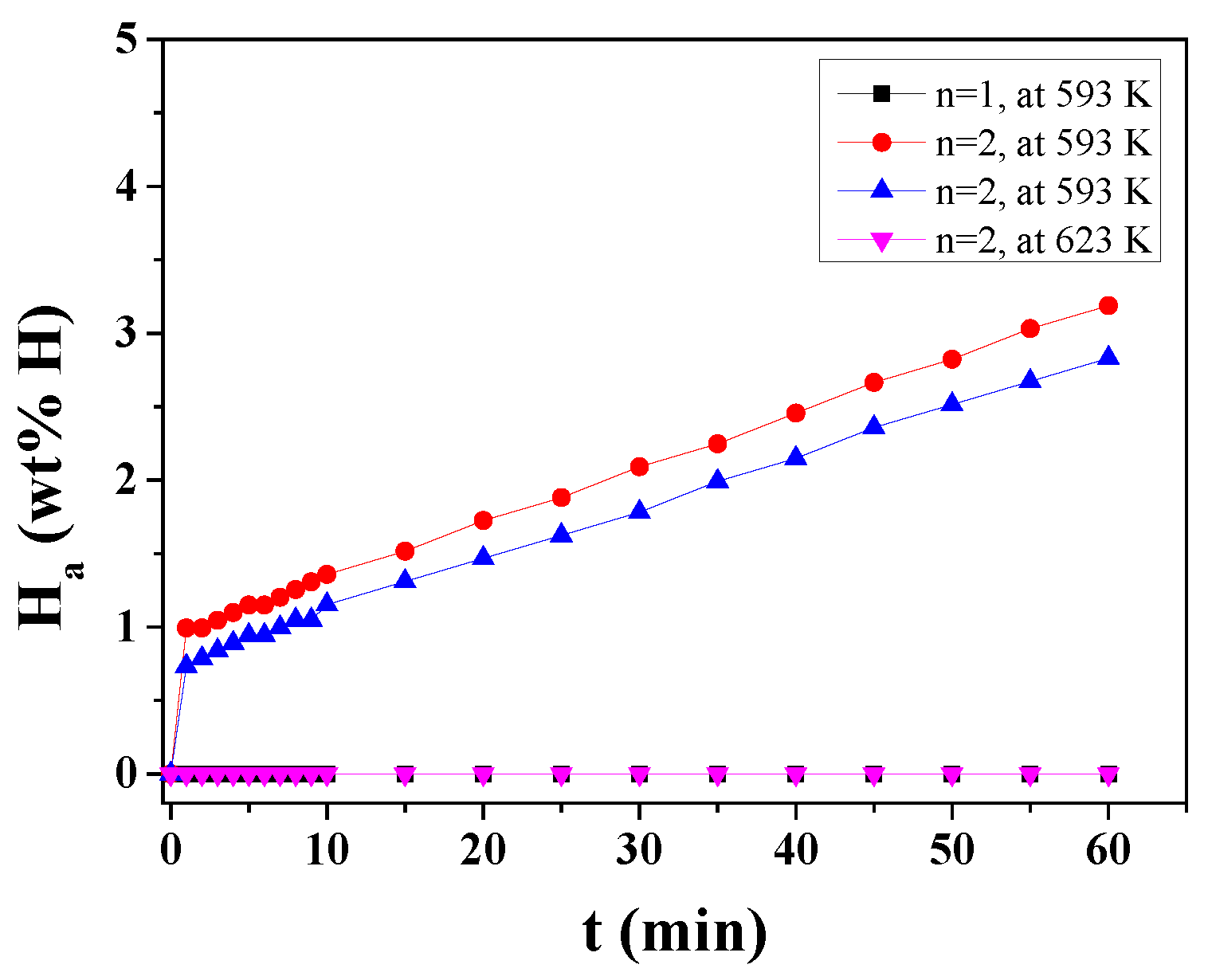
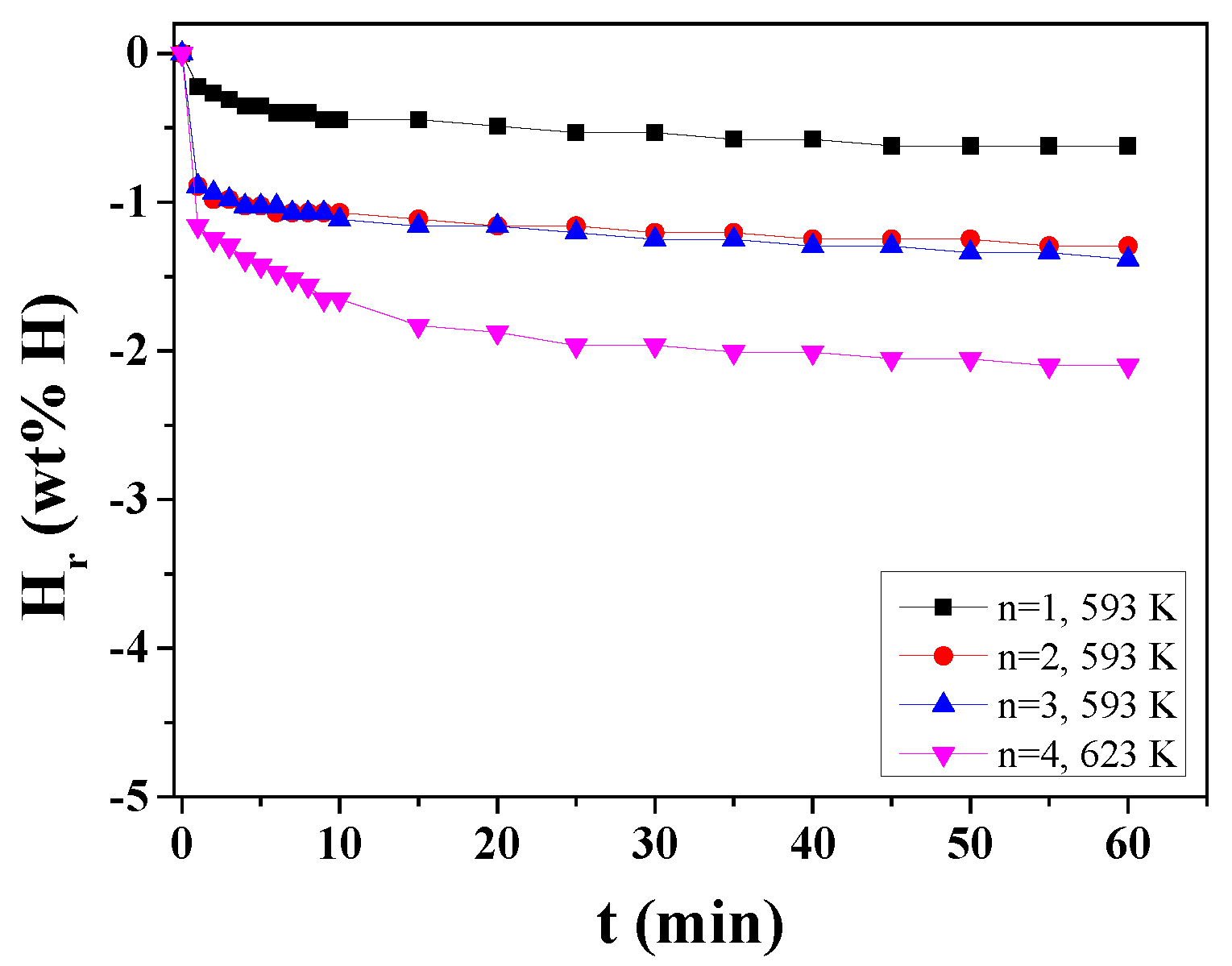
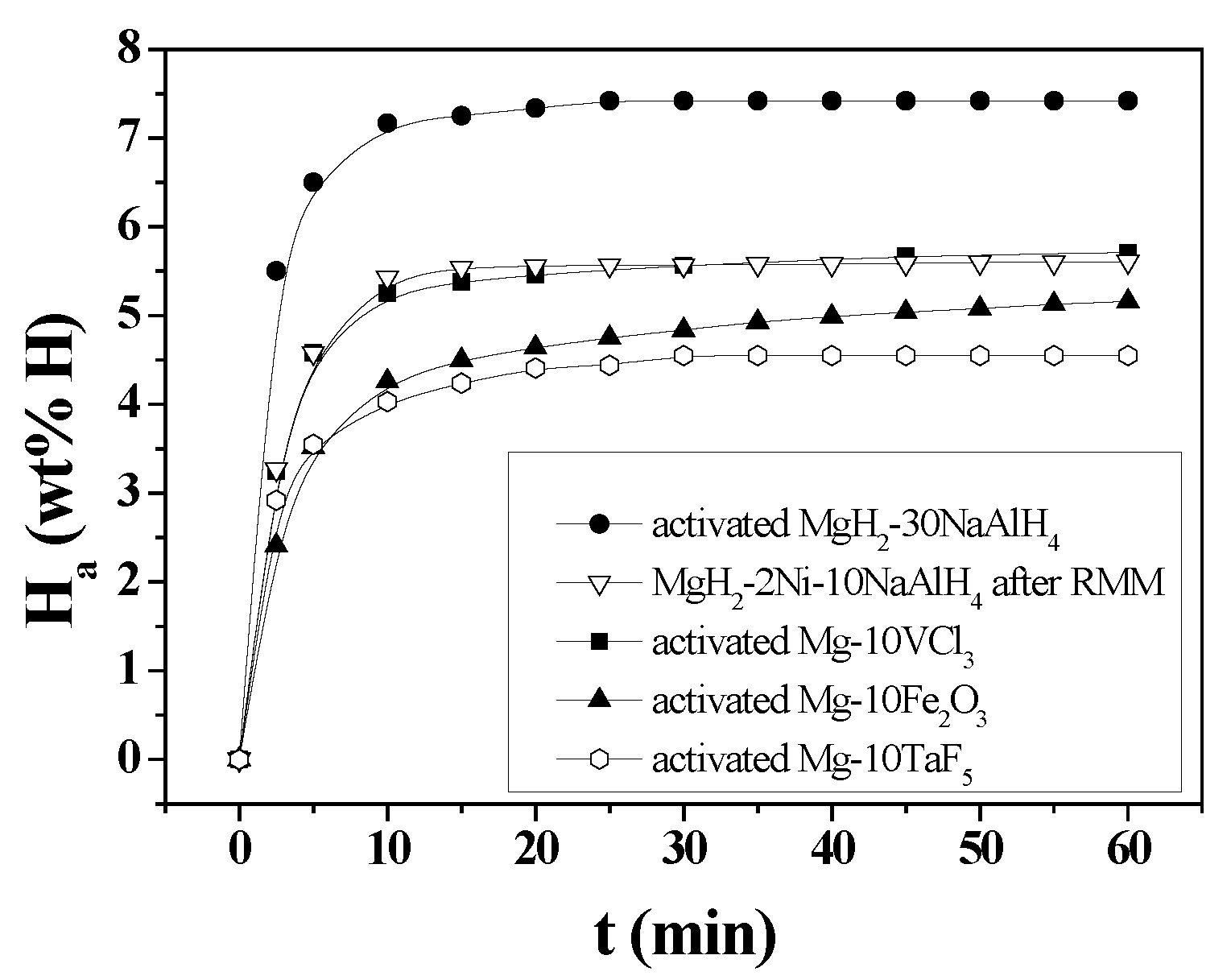
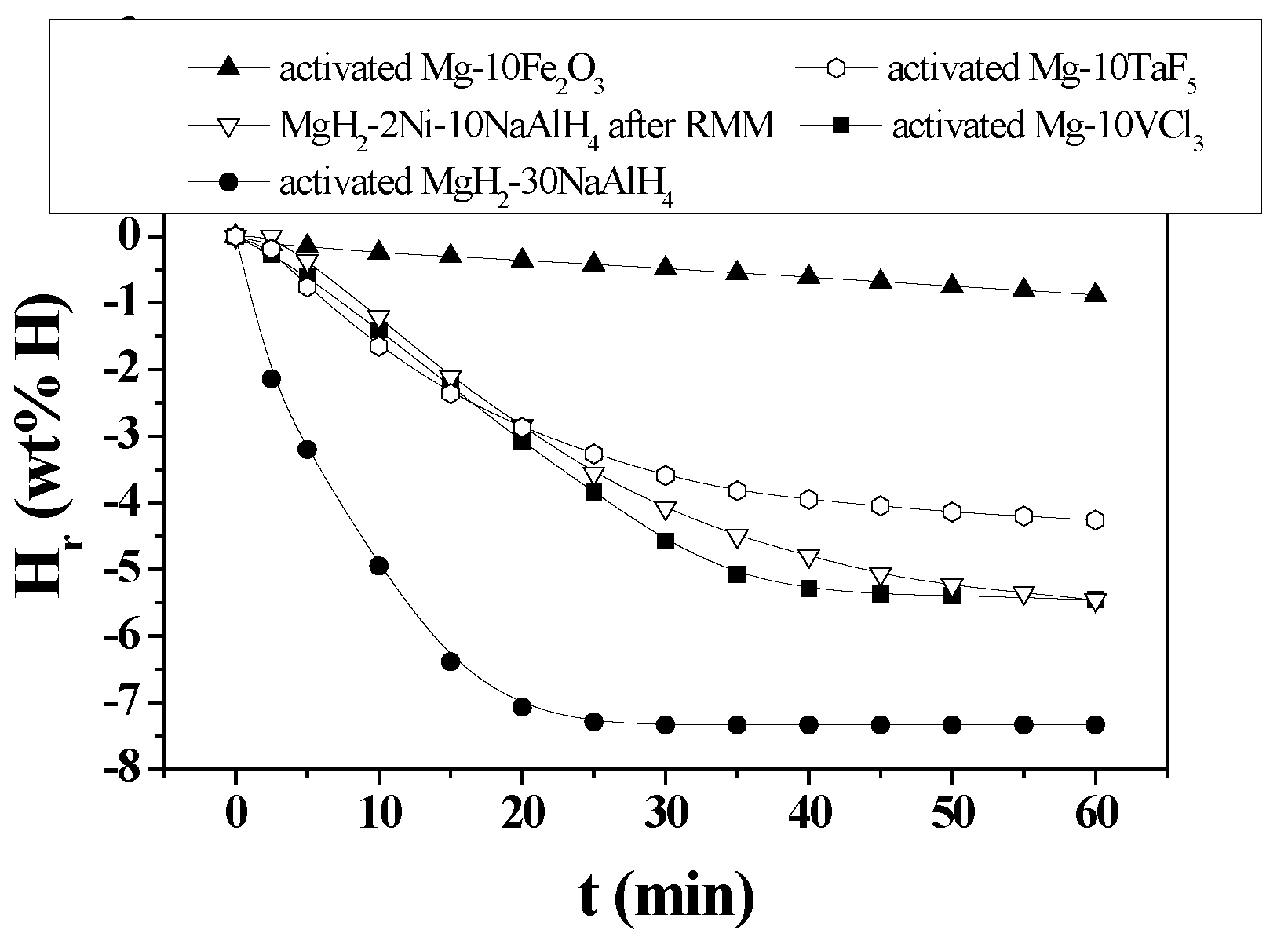
3. Results
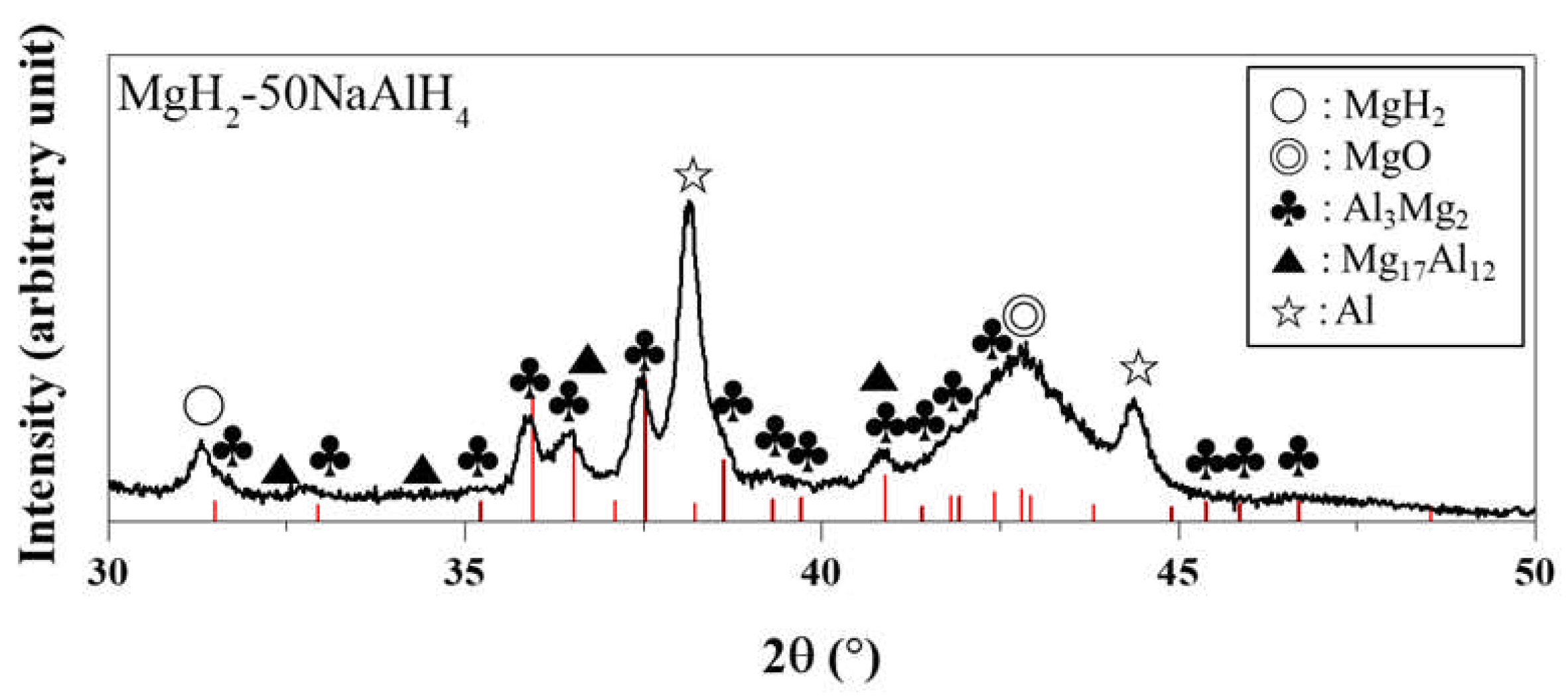
4. Discussion
5. Conclusions
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Reilly, J.J.; Wiswall, R.H. Reaction of hydrogen with alloys of magnesium and nickel and the formation of Mg2NiH4. Inorg. Chem. 1968, 7, 2254–2256. [Google Scholar] [CrossRef]
- Karty, A.; Genossar, J.G.; Rudman, P.S. Hydriding and dehydriding kinetics of Mg in a Mg/Mg2Cu eutectic alloy: Pressure sweep method. J. Appl. Physics 1979, 50, 7200–7209. [Google Scholar] [CrossRef]
- Akiba, E.; Nomura, K.; Ono, S.; Suda, S. Kinetics of the reaction between Mg-Ni alloys and H2. Int J. Hydrogen Energy 1982, 7, 787–791. [Google Scholar] [CrossRef]
- Bobet, J.-L.; Akiba, E.; Nakamura, Y.; Darriet, B. Study of Mg–M (M = Co, Ni and Fe) mixture elaborated by reactive mechanical alloying-hydrogen sorption properties. Int. J. Hydrogen Energy 2000, 25, 987–996. [Google Scholar] [CrossRef]
- Huot, J.; Ravnsbæk, D.B.; Zhang, J.; Cuevas, F.; Latroche, M.; Jensen, T.R. Mechanochemical synthesis of hydrogen storage materials. Prog. Mater. Sci. 2013, 58, 30–75. [Google Scholar] [CrossRef]
- Song, M.Y.; Ahn, D.S.; Kwon, I.H.; Ahn, H.J. Development of Hydrogen storage Alloys by Mechanical Alloying Mg with Fe and Co. Met. Mater. Int. 1999, 5, 485–490. [Google Scholar] [CrossRef]
- Ali, N.A.; Ismail, M. Advanced hydrogen storage of the Mg–Na–Al system: A review. J. Magnesium Alloys 2021, 9, 1111–1122. [Google Scholar] [CrossRef]
- Plerdsranoy, P.; Meethom, S.; Utke, R. Dehydrogenation kinetics, reversibility, and reaction mechanisms of reversible hydrogen storage material based on nanoconfined MgH2−NaAlH4. J. Phys. Chem. Solids 2015, 87, 16–22. [Google Scholar] [CrossRef]
- Rafi-ud-din; Xuanhui, Q.; Ping, L.; Zhang, L.; Ahmad, M.; Iqbal, M.Z.; Rafique, M.Y.; Farooq, M.H. Enhanced hydrogen storage performance for MgH2–NaAlH4 system—The effects of stoichiometry and Nb2O5 nanoparticles on cycling behaviour. RSC Adv. 2012, 2, 4891–4903. [Google Scholar] [CrossRef]
- Ismail, M.; Zhao, Y.; Yu, X.B.; Mao, J.F.; Dou, S.X. The hydrogen storage properties and reaction mechanism of the MgH2–NaAlH4 composite system. Int. J. Hydrogen Energy 2011, 36, 9045–9050. [Google Scholar] [CrossRef]
- Bendyna, J.K.; Dyjak, S.; Notten, P.H.L. The influence of ball-milling time on the dehydrogenation properties of the NaAlH4–MgH2 composite. Int. J. Hydrogen Energy 2015, 40, 4200–4206. [Google Scholar] [CrossRef]
- Liu, H.; Wang, X.; Liu, Y.; Dong, Z.; Ge, H.; Li, S.; Yan, M. Hydrogen Desorption Properties of the MgH2–AlH3 Composites. J. Phys. Chem.C 2014, 118, 37–45. [Google Scholar] [CrossRef]
- Stampfer, J.F.; Molley, C.E.; Suttle, J.F. The Magnesium-Hydrogen System1-3. J. Am. Chem. Soc. 1960, 82, 3504–3508. [Google Scholar] [CrossRef]
- Song, M.Y.; Kwon, I.H.; Kwon, S.N.; Park, C.G.; Hong, S.-H.; Bae, J.-S.; Mumm, D.R. Hydrogen-storage properties of Mg–oxide alloys prepared by reactive mechanical grinding. J. Alloys Compd. 2006, 415, 266–270. [Google Scholar] [CrossRef]
- Song, M.Y.; Kwon, S.N.; Park, H.R. Pressure-Composition Isotherms and Cycling Properties of Mg-xFe2O3-yNi Alloys. Korean J. Met. Mater. 2013, 51, 455–460. [Google Scholar] [CrossRef]
- Kwak, Y.J.; Lee, S.H.; Park, H.R.; Song, M.Y. Review Paper : Hydriding and Dehydriding Reactions of Mg-xTaF5 (x=0, 5, and 10) Prepared via Reactive Mechanical Grinding. Korean J. Met. Mater. 2014, 52, 957–962. [Google Scholar] [CrossRef]
- Kwak, Y.J.; Lee, S.H.; Park, H.R.; Song, M.Y. Hydrogen Storage Characteristics of Mg, Mg-5TaF5, and Mg-5NbF5 Prepared via Grinding in a Hydrogen Atmosphere. J. Nanoscience and Nanotechnology 2016, 16, 10508–10514. [Google Scholar] [CrossRef]
- Song, M.Y.; Lee, S.H.; Mumm, D.R. Fe2O3, MnO, and VCl3-added Mg composites by reactioninvolving grinding processing for hydrogen storage. J. Cer. Proc. Res 2018, 19(3), 211–217. [Google Scholar] [CrossRef]
- Hjort, P.; Krozer, A.; Kasemo, B. Hydrogen sorption kinetics in partly oxidized Mg films. J. Alloys Comp. 1996, 237(1-2), 74–80. [Google Scholar] [CrossRef]
- Zaluska, A.; Zaluski, L.; Ström-Olsen, J.O. Nanocrystalline magnesium for hydrogen storage. J. Alloys Comp. 1999, 288, 217–225. [Google Scholar] [CrossRef]
- Vigeholm, B.; Kjoller, J.; Larsen, B.; Pedersen, A.S. Formation and decomposition of magnesium hydride. J. Less-Common Met. 1983, 89(1), 135–144. [Google Scholar] [CrossRef]
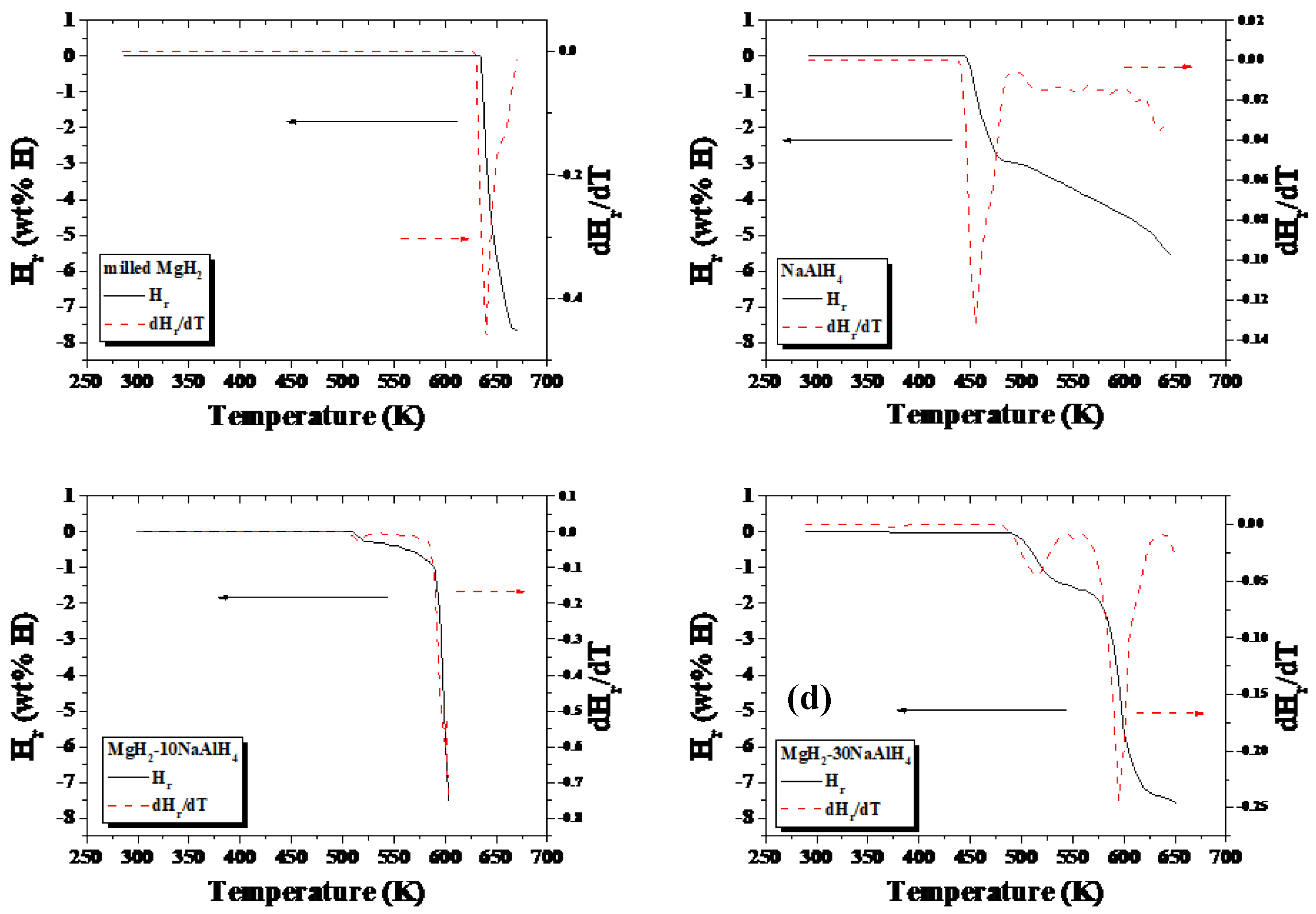
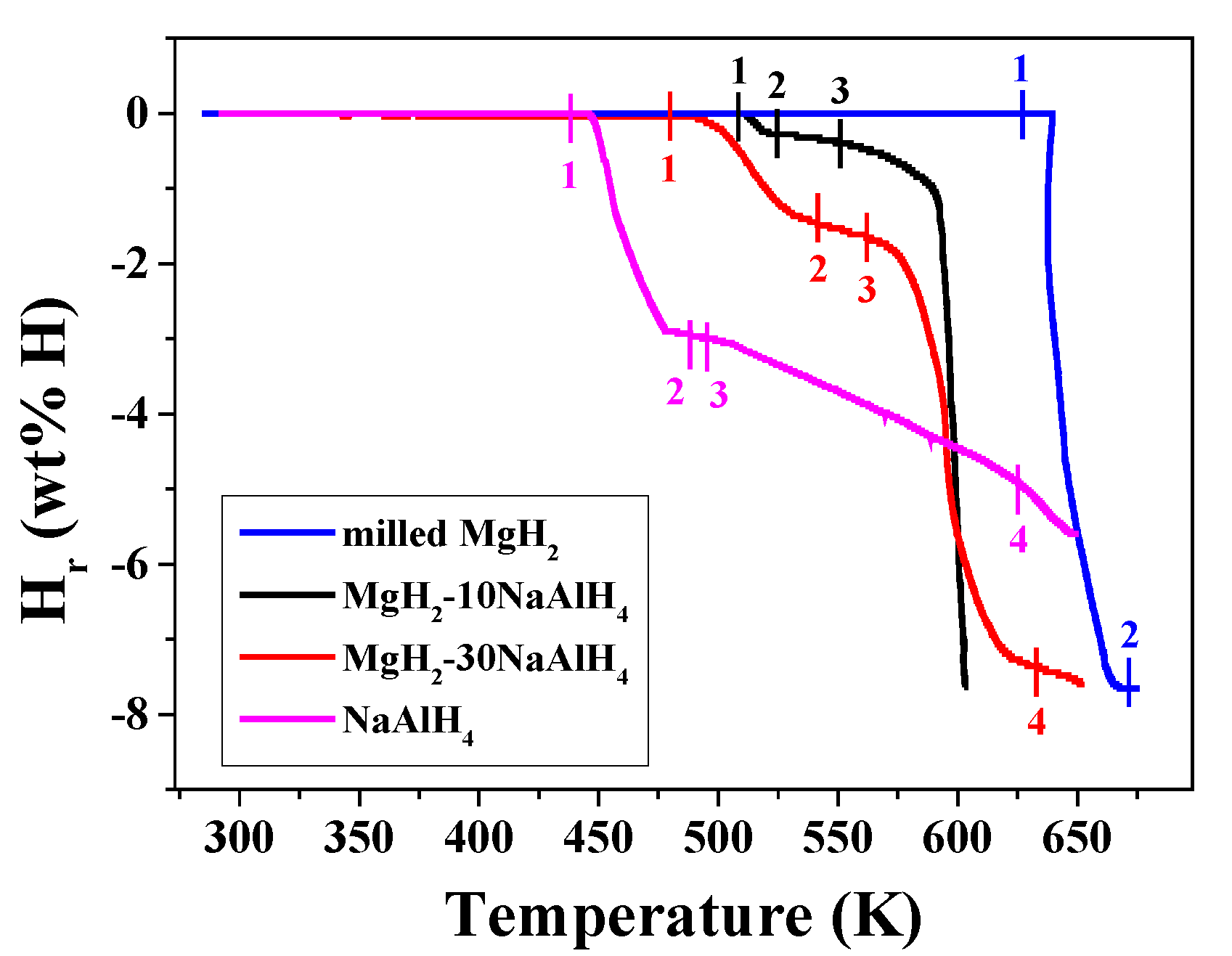
| Peak | Highest Peak | Peak | Peak | |
|---|---|---|---|---|
| milled MgH2 | 638 | |||
| MgH2-10NaAlH4 | 513 | 600 | ||
| MgH2-30NaAlH4 | 512 | 592 | ||
| NaAlH4 | 455 | 552 | 631 |
| Marked Points | 1 | 2 | 3 | 4 |
|---|---|---|---|---|
| milled MgH2 | 627 | 673 | ||
| MgH2-10NaAlH4 | 508 | 525 | 550 | |
| MgH2-30NaAlH4 | 480 | 541 | 562 | 633 |
| NaAlH4 | 438 | 488 | 495 | 625 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




