Submitted:
29 December 2023
Posted:
29 December 2023
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
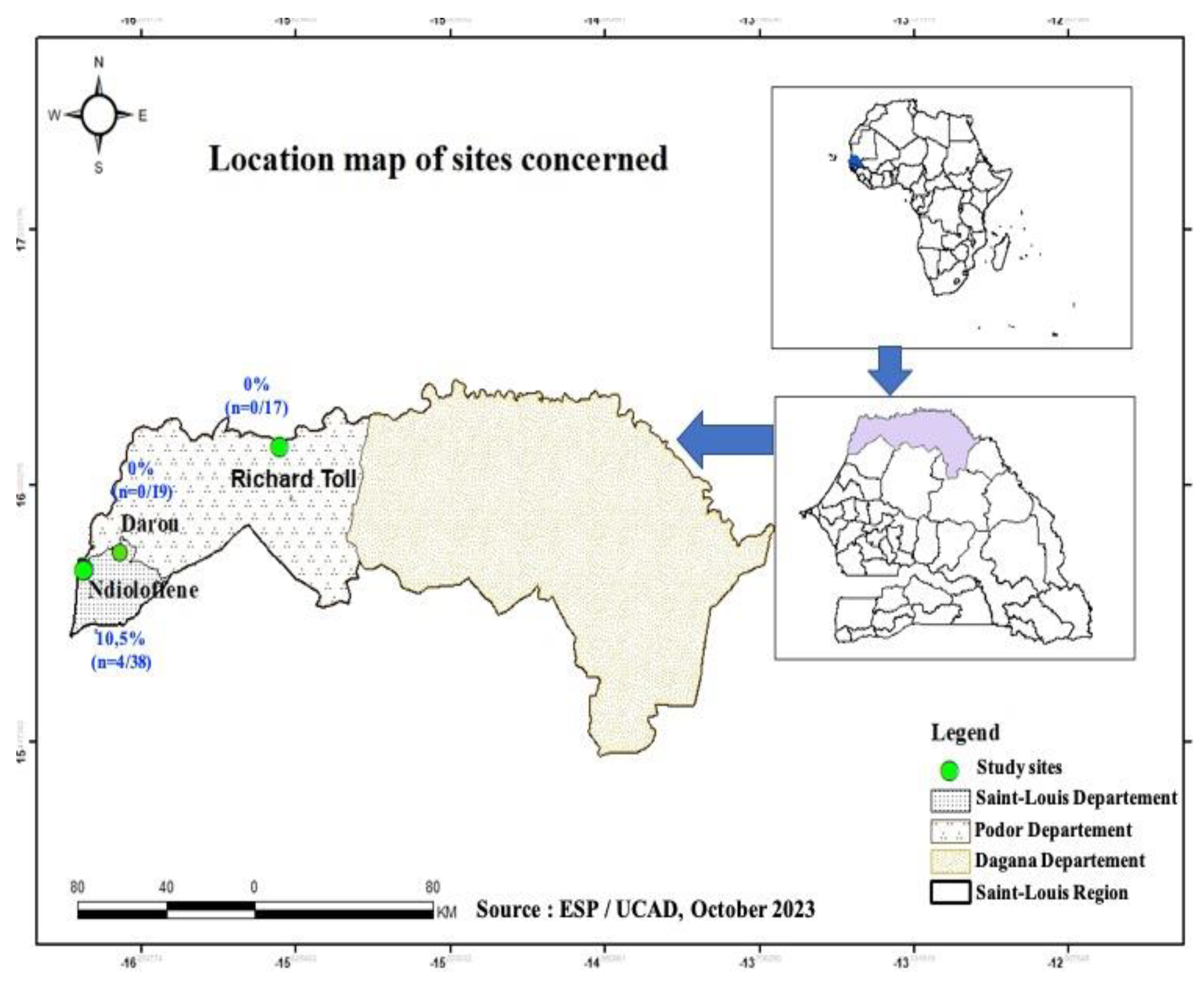
2.1. Sampling
2.2. Extraction of Total Nucleic Acid (TNA)
2.3. RT-PCR
2.4. Sequencing and phylogenetic analysis
2.5. Statistical analysis
3. Results
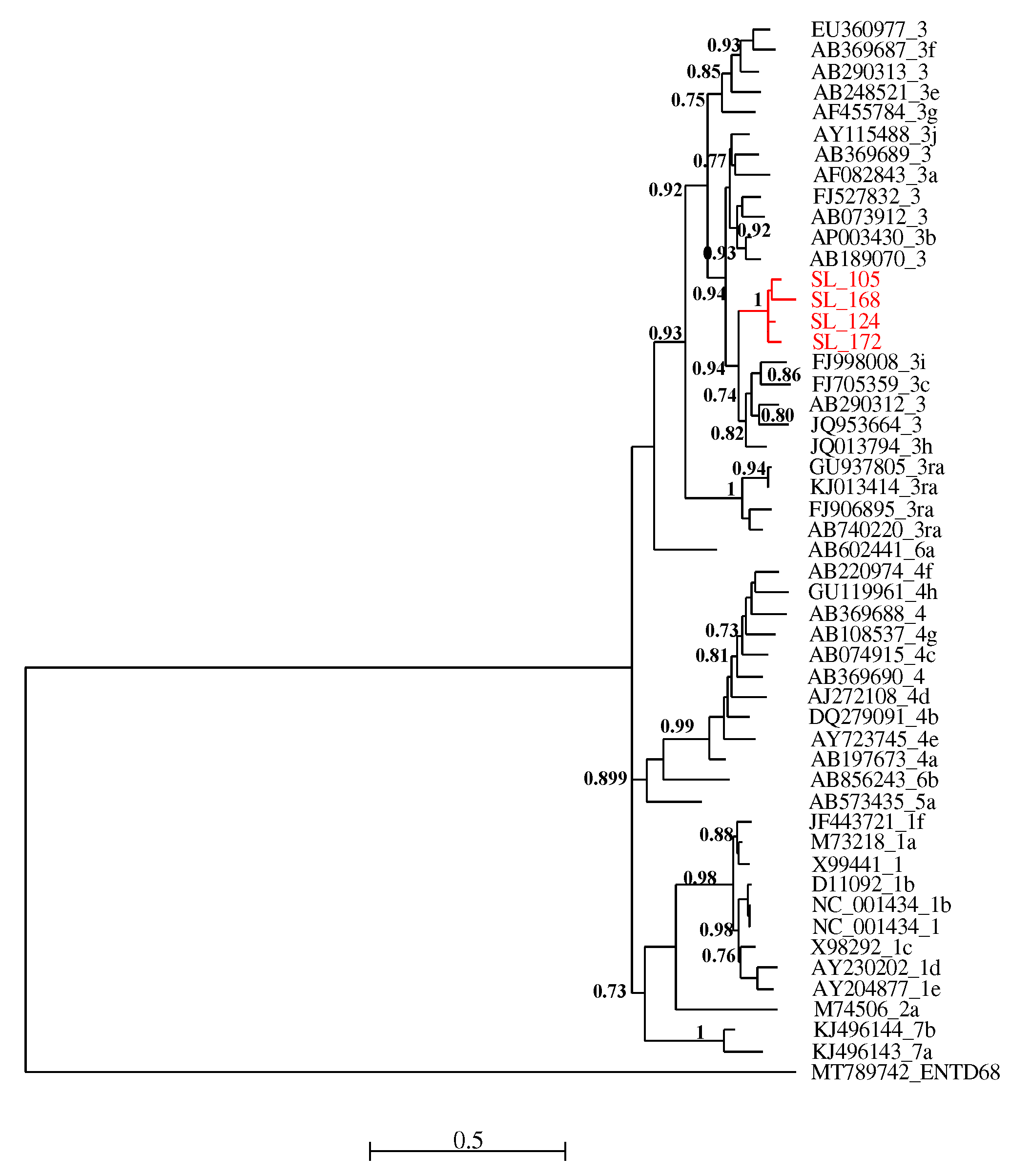
4. Discussion
5. Conclusion
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- A. A. M. Diouara et al., « Hepatitis E Virus Seroprevalence and Associated Risk Factors in Pregnant Women Attending Antenatal Consultations in Senegal », Viruses, vol. 14, no 8, p. 1742, août 2022. [CrossRef]
- Y. Debing, D. Moradpour, J. Neyts, et J. Gouttenoire, « Update on hepatitis E virology: Implications for clinical practice », J. Hepatol., vol. 65, no 1, p. 200-212, juill. 2016. [CrossRef]
- J. Wang, N. Li, H. Zhang, F. Li, S. Fanning, et T. Jiang, « Detection of Hepatitis E Virus in the Pig Livers and Retail Pork Samples Collected in Selected Cities in China », Foodborne Pathog. Dis., vol. 18, no 2, p. 97-103, févr. 2021. [CrossRef]
- WHO, « Hepatitis E ». Consulté le: 11 août 2023. [En ligne]. Disponible sur: https://www.who.int/news-room/fact-sheets/detail/hepatitis-e.
- G. R. Takuissu et al., « Hepatitis E Virus in Water Environments: A Systematic Review and Meta-analysis », Food Environ. Virol., vol. 14, no 3, p. 223-235, sept. 2022. [CrossRef]
- D. B. Smith et al., « Update: proposed reference sequences for subtypes of hepatitis E virus (species Orthohepevirus A) », J. Gen. Virol., vol. 101, no 7, p. 692-698, juill. 2020. [CrossRef]
- M. A. Fatawou, M. G. Chavely, M. Y. M. Henri, K. N. Daniel, E. Z. M. Claire, et N. Richard, « First Detection and Characterization of Hepatitis E Virus in Sewage Samples in Cameroon », Food Environ. Virol., vol. 15, no 3, p. 255-261, sept. 2023. [CrossRef]
- I. Nimgaonkar, Q. Ding, R. E. Schwartz, et A. Ploss, « Hepatitis E virus: advances and challenges », Nat. Rev. Gastroenterol. Hepatol., vol. 15, no 2, Art. no 2, févr. 2018. [CrossRef]
- W. H. Van der Poel, « Food and environmental routes of Hepatitis E virus transmission », Curr. Opin. Virol., vol. 4, p. 91-96, févr. 2014. [CrossRef]
- N. Pavio, P. Kooh, V. Cadavez, U. Gonzales-Barron, et A. Thébault, « Risk factors for sporadic hepatitis E infection: a systematic review and meta-analysis », Microb. Risk Anal., vol. 17, p. 100129, avr. 2021. [CrossRef]
- A. Shrestha et al., « Prevalence and risk of hepatitis E virus infection in the HIV population of Nepal », Virol. J., vol. 14, no 1, p. 228, nov. 2017. [CrossRef]
- M. A. Purdy et al., « ICTV Virus Taxonomy Profile: Hepeviridae 2022 », J. Gen. Virol., vol. 103, no 9, sept. 2022. [CrossRef]
- J. E. Forero et al., « Phylogenetic analysis of Hepatitis E virus strains isolated from slaughter-age pigs in Colombia », Infect. Genet. Evol., vol. 49, p. 138-145, avr. 2017. [CrossRef]
- N. Thippornchai et al., « Survey of hepatitis E virus in pork products and pig stools in Nakhon Pathom Province, Thailand », Vet. Med. Sci., vol. 8, no 5, p. 1975-1981, mai 2022. [CrossRef]
- L. Milojević et al., « Screening and Molecular Characterization of Hepatitis E Virus in Slaughter Pigs in Serbia », Food Environ. Virol., vol. 11, no 4, p. 410-419, déc. 2019. [CrossRef]
- X. J. Meng, « Hepatitis E virus: Animal reservoirs and zoonotic risk », Vet. Microbiol., vol. 140, no 3, p. 256-265, janv. 2010. [CrossRef]
- M. Andraud et al., « Direct contact and environmental contaminations are responsible for HEV transmission in pigs », Vet. Res., vol. 44, no 1, p. 102, oct. 2013. [CrossRef]
- P. Colson et al., « Pig liver sausage as a source of hepatitis E virus transmission to humans », J. Infect. Dis., vol. 202, no 6, p. 825-834, sept. 2010. [CrossRef]
- A. M. I. Montone et al., « Occurrence of HEV-RNA in Italian Regional Pork and Wild Boar Food Products », Food Environ. Virol., vol. 11, no 4, p. 420-426, déc. 2019. [CrossRef]
- M. Bouwknegt, F. Lodder-Verschoor, W. H. M. Van Der Poel, S. A. Rutjes, et A. M. De Roda Husman, « Hepatitis E Virus RNA in Commercial Porcine Livers in The Netherlands », J. Food Prot., vol. 70, no 12, p. 2889-2895, déc. 2007. [CrossRef]
- A. R. Feagins, T. Opriessnig, D. K. Guenette, P. G. Halbur, et X.-J. Meng, « Detection and characterization of infectious Hepatitis E virus from commercial pig livers sold in local grocery stores in the USA », J. Gen. Virol., vol. 88, no 3, p. 912-917, 2007. [CrossRef]
- C. Feurer et al., « High load of hepatitis E viral RNA in pork livers but absence in pork muscle at French slaughterhouses », Int. J. Food Microbiol., vol. 264, p. 25-30, janv. 2018. [CrossRef]
- Y. Yazaki et al., « Sporadic acute or fulminant hepatitis E in Hokkaido, Japan, may be food-borne, as suggested by the presence of hepatitis E virus in pig liver as food », J. Gen. Virol., vol. 84, no 9, p. 2351-2357, 2003. [CrossRef]
- T.-C. Li et al., « Hepatitis E Virus Transmission from Wild Boar Meat », Emerg. Infect. Dis., vol. 11, no 12, p. 1958-1960, déc. 2005. [CrossRef]
- C. Renou, A.-M. R. Afonso, et N. Pavio, « Foodborne Transmission of Hepatitis E Virus from Raw Pork Liver Sausage, France », Emerg. Infect. Dis., vol. 20, no 11, p. 1945-1947, nov. 2014. [CrossRef]
- M. Riveiro-Barciela, B. Mínguez, R. Gironés, F. Rodriguez-Frías, J. Quer, et M. Buti, « Phylogenetic demonstration of hepatitis E infection transmitted by pork meat ingestion », J. Clin. Gastroenterol., vol. 49, no 2, p. 165-168, févr. 2015. [CrossRef]
- A. Rivero-Juarez et al., « Familial Hepatitis E Outbreak Linked to Wild Boar Meat Consumption », Zoonoses Public Health, vol. 64, no 7, p. 561-565, 2017. [CrossRef]
- ISO 17604:2015, « Microbiology of the food chain — Carcass sampling for microbiological analysis ». Consulté le: 11 décembre 2023. [En ligne]. Disponible sur: https://www.iso.org/obp/ui#iso:std:iso:17604:ed-2:v1:en.
- A. A. M. Diouara et al., « Antiretroviral treatment outcome in HIV-1-infected patients routinely followed up in capital cities and remote areas of Senegal, Mali and Guinea-Conakry », J. Int. AIDS Soc., vol. 17, no 1, p. 19315, 2014. [CrossRef]
- H. Mizuo et al., « Polyphyletic Strains of Hepatitis E Virus Are Responsible for Sporadic Cases of Acute Hepatitis in Japan », J. Clin. Microbiol., vol. 40, no 9, p. 3209-3218, sept. 2002. [CrossRef]
- M. Gouy, S. Guindon, et O. Gascuel, « SeaView Version 4: A Multiplatform Graphical User Interface for Sequence Alignment and Phylogenetic Tree Building », Mol. Biol. Evol., vol. 27, no 2, p. 221-224, févr. 2010. [CrossRef]
- S. Guindon, J.-F. Dufayard, V. Lefort, M. Anisimova, W. Hordijk, et O. Gascuel, « New Algorithms and Methods to Estimate Maximum-Likelihood Phylogenies: Assessing the Performance of PhyML 3.0 », Syst. Biol., vol. 59, no 3, p. 307-321, mars 2010. [CrossRef]
- S. Kumar, G. Stecher, M. Li, C. Knyaz, et K. Tamura, « MEGA X: Molecular Evolutionary Genetics Analysis across Computing Platforms », Mol. Biol. Evol., vol. 35, no 6, p. 1547-1549, juin 2018. [CrossRef]
- M. Anisimova, M. Gil, J.-F. Dufayard, C. Dessimoz, et O. Gascuel, « Survey of Branch Support Methods Demonstrates Accuracy, Power, and Robustness of Fast Likelihood-based Approximation Schemes », Syst. Biol., vol. 60, no 5, p. 685-699, oct. 2011. [CrossRef]
- Rambaut, « FigTree v. 1.4.4. », ResearchGate. Consulté le: 11 décembre 2023. [En ligne]. Disponible sur: : http://tree. bio. ed. ac. uk/software/figtree/.
- A. F. Modiyinji et al., « Epidemiology of hepatitis E virus infection in animals in Africa: a systematic review and meta-analysis », BMC Vet. Res., vol. 17, no 1, p. 50, janv. 2021. [CrossRef]
- N. Pavio, T. Merbah, et A. Thébault, « Frequent Hepatitis E Virus Contamination in Food Containing Raw Pork Liver, France », Emerg. Infect. Dis., vol. 20, no 11, p. 1925-1927, nov. 2014. [CrossRef]
- M. Salines, M. Andraud, et N. Rose, « From the epidemiology of hepatitis E virus (HEV) within the swine reservoir to public health risk mitigation strategies: a comprehensive review », Vet. Res., vol. 48, no 1, p. 31, mai 2017. [CrossRef]
- E. Carella et al., « Molecular and serological investigation of Hepatitis E virus in pigs slaughtered in Northwestern Italy », BMC Vet. Res., vol. 19, no 1, p. 21, janv. 2023. [CrossRef]
- I. L. A. Boxman, C. C. C. Jansen, G. Hägele, A. Zwartkruis-Nahuis, A. S. L. Tijsma, et H. Vennema, « Monitoring of pork liver and meat products on the Dutch market for the presence of HEV RNA », Int. J. Food Microbiol., vol. 296, p. 58-64, mai 2019. [CrossRef]
- Di Bartolo et al., « Hepatitis E Virus in Pork Production Chain in Czech Republic, Italy, and Spain, 2010 - PMC ». Consulté le: 29 septembre 2023. [En ligne]. Disponible sur: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3414029/.
- M. Bouwknegt et al., « The course of hepatitis E virus infection in pigs after contact-infection and intravenous inoculation », BMC Vet. Res., vol. 5, no 1, p. 7, févr. 2009. [CrossRef]
- A. F. Modiyinji, G. M. A. M. Sanding, M. A. Atsama, C. G. Monamele, M. Nola, et R. Njouom, « Serological and molecular investigation of hepatitis E virus in pigs reservoirs from Cameroon reveals elevated seroprevalence and presence of genotype 3 », PLOS ONE, vol. 15, no 2, p. e0229073, févr. 2020. [CrossRef]
- P. El-Duah et al., « Detection and genomic characterization of hepatitis E virus genotype 3 from pigs in Ghana, Africa », One Health Outlook, vol. 2, no 1, p. 10, juill. 2020. [CrossRef]
- K. A. Traoré et al., « Hepatitis E Virus Exposure is Increased in Pork Butchers from Burkina Faso », Am. J. Trop. Med. Hyg., vol. 93, no 6, p. 1356-1359, déc. 2015. [CrossRef]
- C. Spahr, T. Knauf-Witzens, T. Vahlenkamp, R. G. Ulrich, et R. Johne, « Hepatitis E virus and related viruses in wild, domestic and zoo animals: A review », Zoonoses Public Health, vol. 65, no 1, p. 11-29, 2018. [CrossRef]
- P. P. Primadharsini, S. Nagashima, et H. Okamoto, « Genetic Variability and Evolution of Hepatitis E Virus », Viruses, vol. 11, no 5, p. 456, mai 2019. [CrossRef]
- B. D. Sadio et al., « First hepatitis E outbreak in Southeastern Senegal », Sci. Rep., vol. 12, no 1, Art. no 1, oct. 2022. [CrossRef]
- Lalèyê et al., « La filière porcine au Sénégal : commercialisation et consommation des viandes de porc et de phacochère dans les départements de Dakar, Fatick, Ziguinchor et Kolda », 2007.
- W. Ossebi et al., « Analyse zootechnique et économique des systèmes d’élevage de porcs en Casamance (Sénégal) », Rev. D’élevage Médecine Vét. Pays Trop., vol. 72, p. 13, mai 2019. [CrossRef]
| Sites/Samples | Pork meat | Pork liver | TOTAL |
|---|---|---|---|
| Ndioloffène | 29 | 9 | 38 |
| Richard-Toll | 17 | 0 | 17 |
| Darou | 19 | 0 | 19 |
| TOTAL | 65 | 9 | 74 |
| Sample | Sample sizes |
Positive samples | Prevalence of HEV | 95% CI | p-value |
|---|---|---|---|---|---|
| Pork meat | 65 | 2 | 3.1% | [0.53%-11.6%] | 0.0699 |
| Pork liver | 9 | 2 | 22.2% | [3.9%-59.8%] |
| Sites | Positive samples | Prevalence of HEV | 95% CI | p-value |
|---|---|---|---|---|
| Ndioloffène | 4 | 10.50% | [3.4%-25.7%] | 0.1698 |
| Darou | 0 | 0% | [0%-20.9%] | |
| Richard-Toll | 0 | 0% | [0%-22.9%] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




