Submitted:
21 December 2023
Posted:
25 December 2023
You are already at the latest version
Abstract
Keywords:
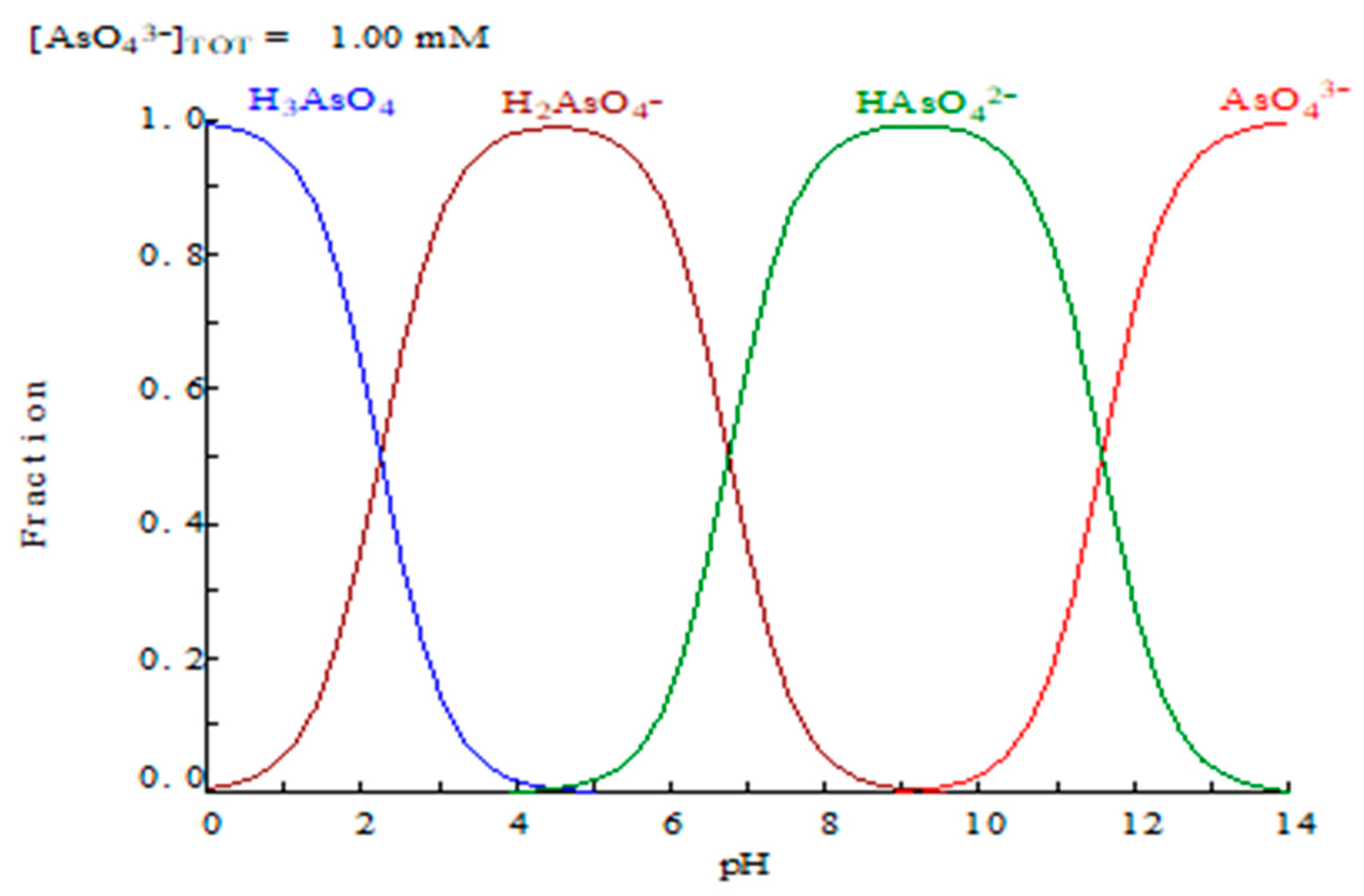
1. Introduction
2. Results and Discussion
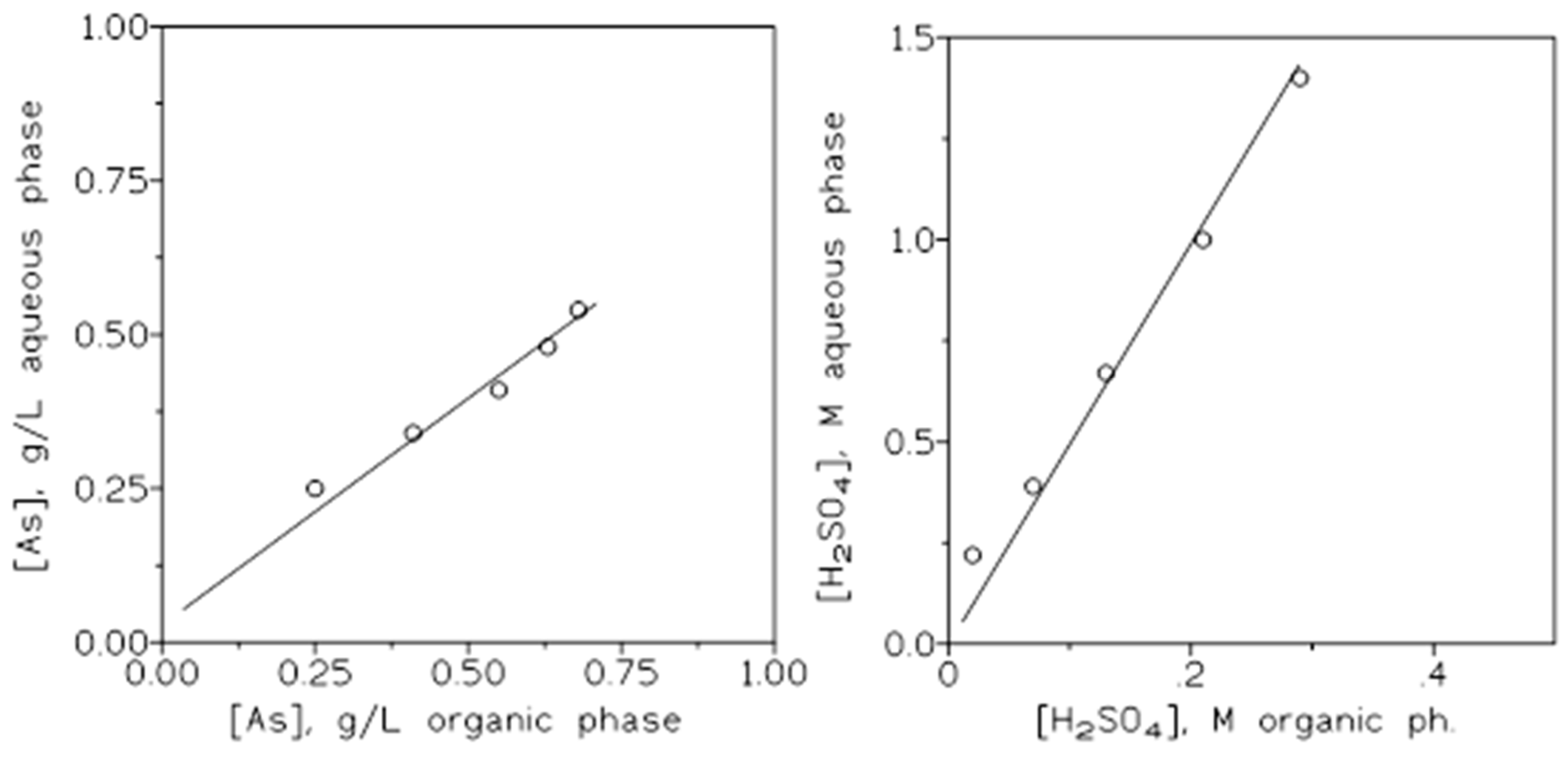
2.1. Arsenic (V) and Sulphuric Acid Extraction
2.1.1. Influence of the Equilibration Time
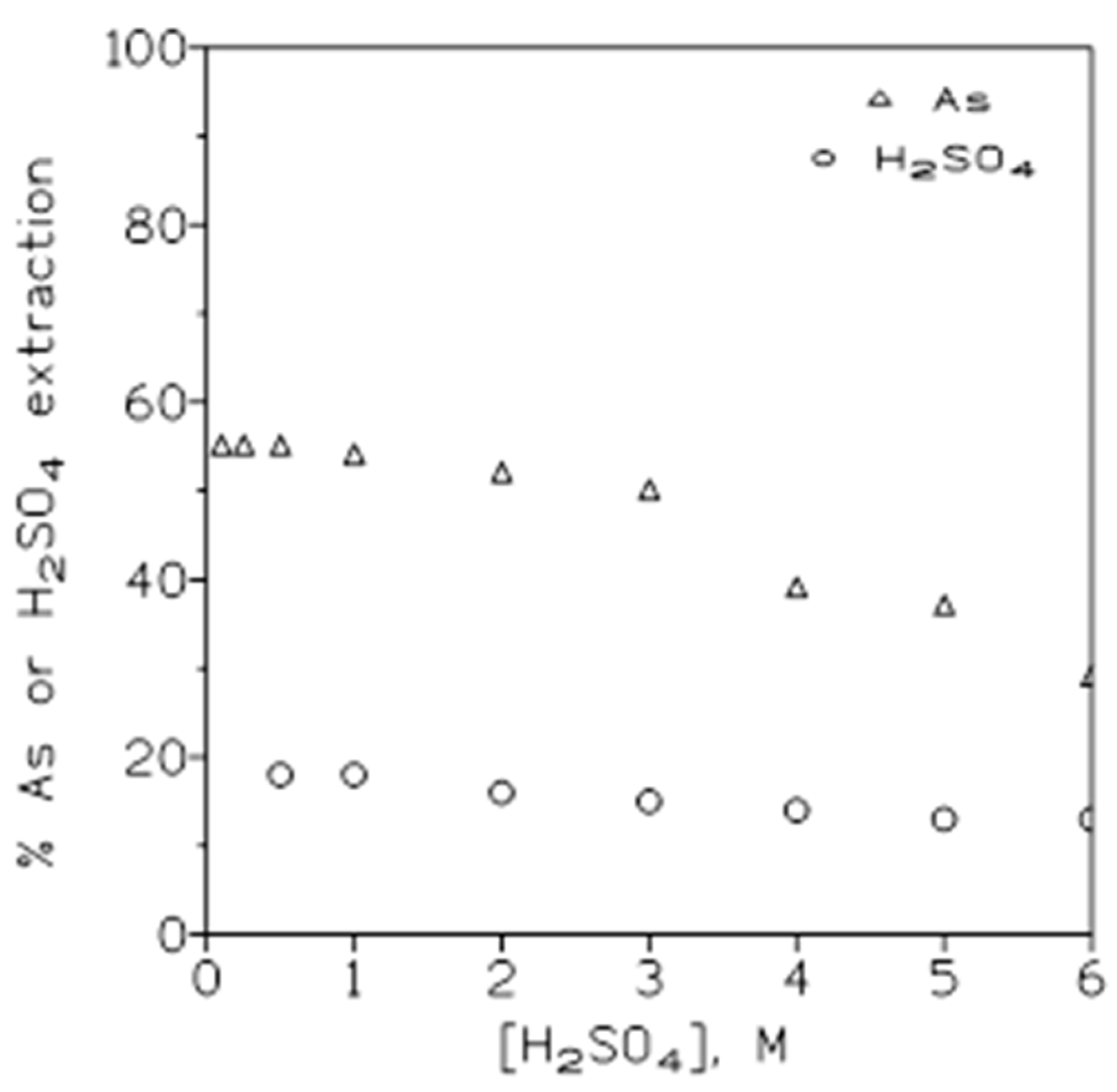
2.1.2. Influence of Acidity of the Aqueous Phase
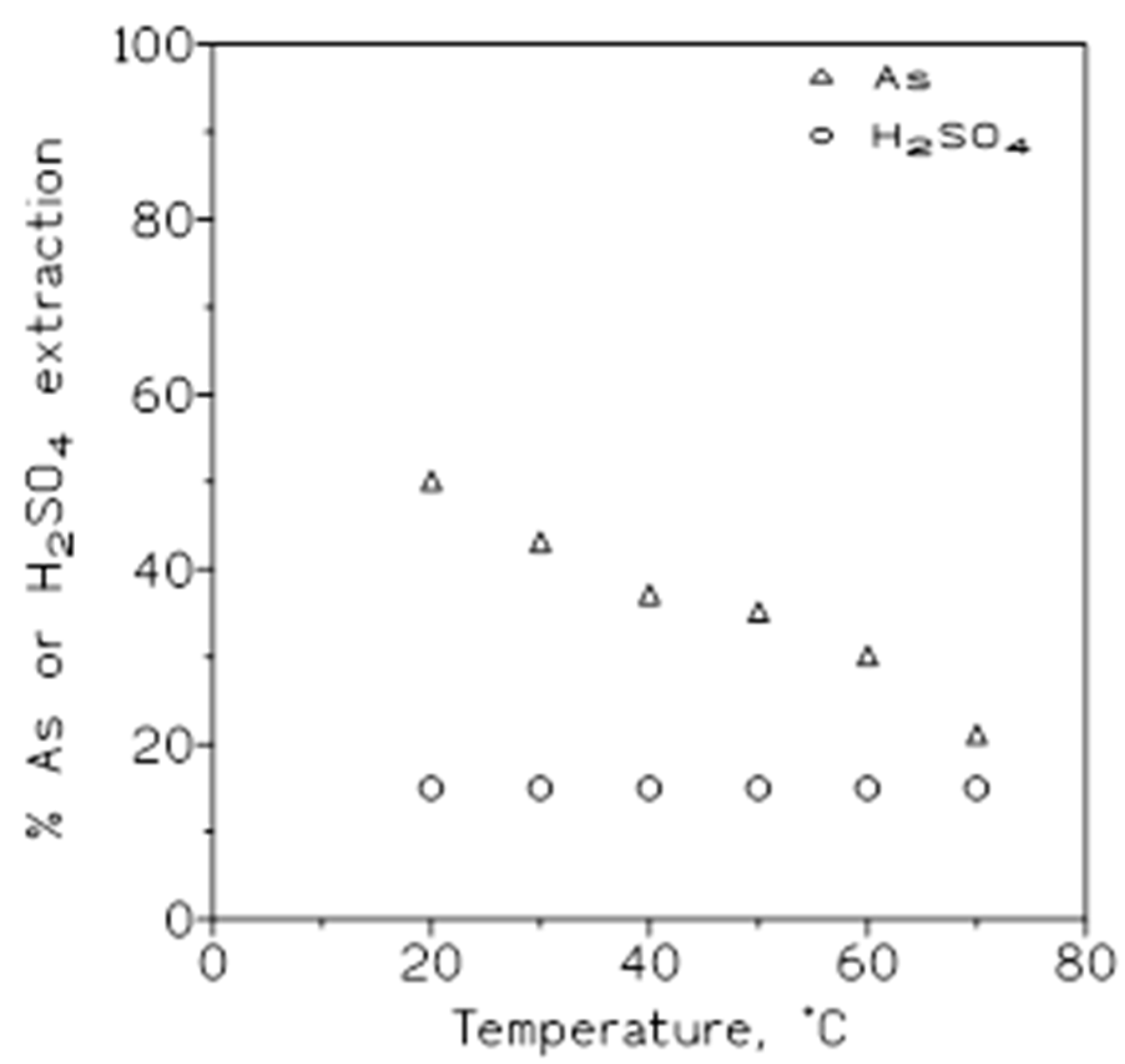
2.1.3. Influence of the Temperature
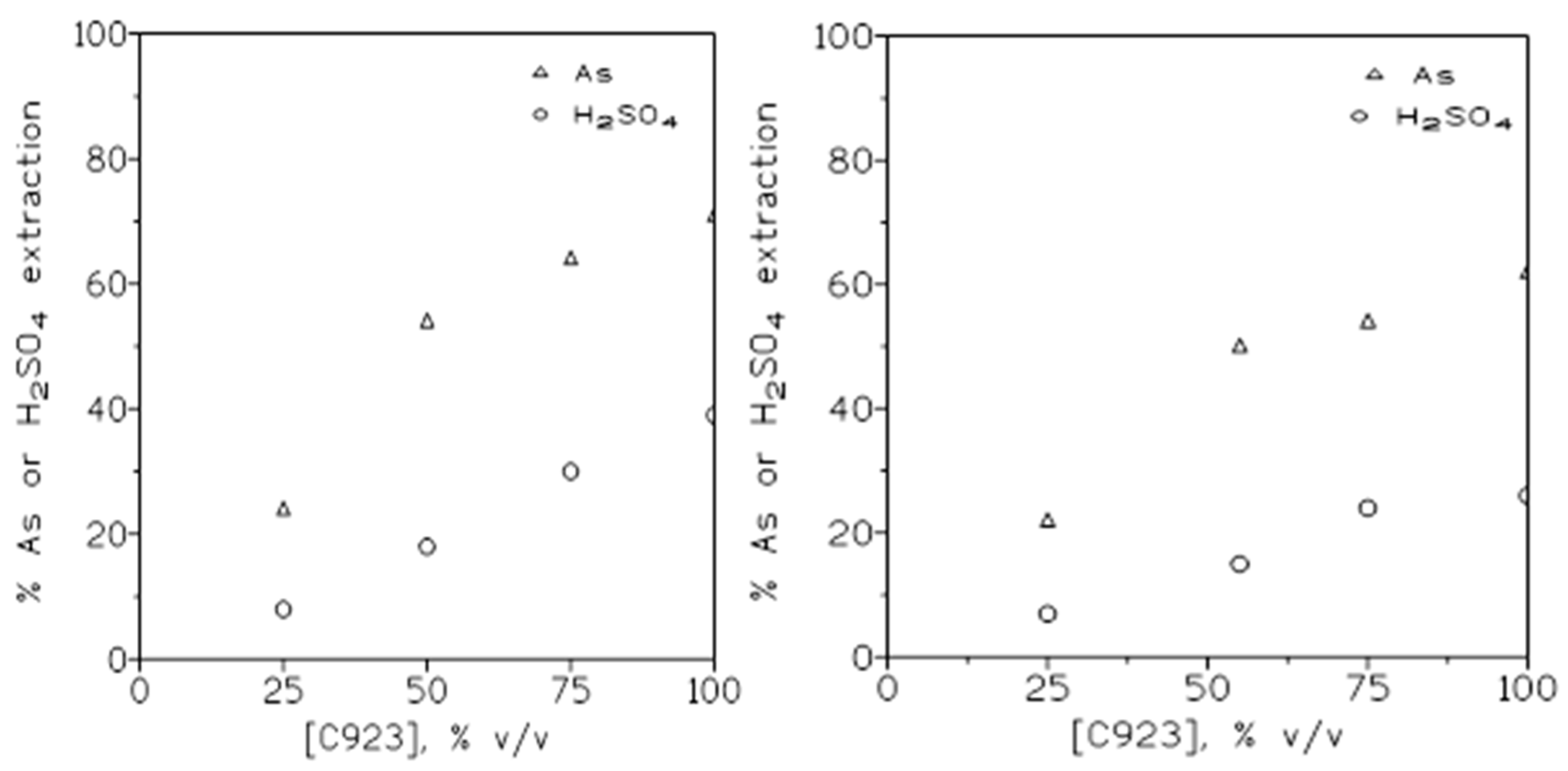
2.1.4. Influence of the Extractant Concentration
| Sulphuric acid, M | Species | Kext | U |
|---|---|---|---|
| 1.5 3 |
H3AsO4·L H3AsO4·2L H3AsO4·L |
0.36 0.67 0.80 |
0.012 0.011 |
2.1.5. Influence of the Initial Arsenic Concentration in the Aqueous Phase
2.1.6. Influence of the O/A Relationship
2.2. Arsenic(V) and Sulphuric Acid Stripping
2.2.1. Influence of the Temperature
2.2.2. Influence of the O/A Relationship
2.3. Selectivity of the System
2.4. Comparison with Other Solvation Extractants
3. Materials and Methods
3.1. Materials
3.2. Solvent Extraction Procedure
4. Conclusions
Author Contributions
Acknowledgements
Conflicts of Interest
References
- Emsley, J. The Elements of Murder, Oxford University Press, Oxford, 2005.
- Hackethal, C.; Pabel, U.; Jung, C.; Schwerdtle, T.; Lindtner, O. Chronic dietary exposure to total arsenic, inorganic arsenic and water-soluble organic arsenic species based on results of the first German total diet study. Sci. Tot. Environ. 2023, 859, 160261. [Google Scholar] [CrossRef] [PubMed]
- Monteiro De Oliveira, E.C; Siqueira Caixeta, E.; Santana Vieira Santos, V.; Barbosa Pereira, B. Arsenic exposure from groundwater: environmental contamination, human health effects, and sustainable solutions. J. Toxicol. Environ. Health, Part B. 2021, 24, 119–135. [Google Scholar] [CrossRef] [PubMed]
- Reyes, D.; Ganesan, N.; Boffetta, P.; Labgaa, I. Arsenic-contaminated drinking water and cholangiocarcinoma. Eur. J. Cancer Prev. 2023, 32, 10–17. [Google Scholar] [CrossRef] [PubMed]
- Glade, S.; Bandaru, S.R.S.; Nahata, M.; Majmudar, J.; Gadgil, A. Adapting a drinking water treatment technology for arsenic removal to the context of a small, low-income California community. Water Res. 2021, 204, 117595. [Google Scholar] [CrossRef] [PubMed]
- Ayub, A.; Srithilat, K.; Fatima, I.; Panduro-Tenazoa, N.M.; Ahmed, I.; Akhtar, M.U.; Shabbir, W.; Ahmad, K.; Muhammad, A. Arsenic in drinking water: overview of removal strategies and role of chitosan biosorbent for its remediation. Environ, Sci. Poll. Res. 2022, 29, 64312–64344. [Google Scholar] [CrossRef] [PubMed]
- Singh, A.; Chauhan, S.; Varjani, S.; Pandey, A.; Bhargava, P. C. Integrated approaches to mitigate threats from emerging potentially toxic elements: A way forward for sustainable environmental management. Environ. Res. 2022, 209, 112844. [Google Scholar] [CrossRef] [PubMed]
- Somayaji, A.; Sarkar, S.; Balasubramaniam, S.; Raval, R. Synthetic biology techniques to tackle heavy metal pollution and poisoning. Synth. Syst. Biotechnol. 2022, 7, 841–846. [Google Scholar] [CrossRef] [PubMed]
- Alguacil, F.J.; Escudero, E. The removal of toxic metals from liquid effluents by ion exchange resins. Part XVII: arsenic(V))/H+/Dowex 1x8. Rev. Metal. 2022, 58, e221. [Google Scholar] [CrossRef]
- Benabdallah, N.; Luo, D.; Hadj, Y.M.; Lopez, J.; Fernández de Labastida, M.; Sastre, A.M.; Valderrama, C.A.; Cortina, J.L. Increasing the circularity of the copper metallurgical industry: Recovery of Sb(III) and Bi(III) from hydrochloric solutions by integration of solvating organophosphorous extractants and selective precipitation. Chem. Eng. J. 2023, 453, 139811. [Google Scholar] [CrossRef]
- Li, G.; Zhang, A.; Qi, X.; Yan, G.; Zhi, G. Arsenic removal from aqueous solution by chitosan loaded with Al/Ti elements. Ser. Sci. Technol. 2023, 58, 2298–2306. [Google Scholar] [CrossRef]
- Puigdomenech, I. Chemical equilibrium software Hydra/Medusa. 2023. https://sites.google.com/site/chemdiagr/home.
- Alguacil, F.J.; Lopez, F.A. The extraction of mineral acids by the phosphine oxide Cyanex 923. Hydrometallurgy 1996, 42, 245–255. [Google Scholar] [CrossRef]
- Bogacki, M.B.; Wiśniewski, M.; Szymanowski, J. Effect of extractant on arsenic(V) recovery from sulfuric acid solutions. J. Radioanal. Nucl. Chem. 1998, 228, 57–61. [Google Scholar] [CrossRef]
- Iberhan, L.; Wisniewski, M. Removal of arsenic(III) and arsenic(V) from sulfuric acid solution by liquid-liquid extraction. J Chem Technol Biotechnol. 2003, 78, 659–665. [Google Scholar] [CrossRef]
- Wisniewski, M. Extraction of arsenic from sulphuric acid solutions by Cyanex 923. Hydrometallurgy 1997, 46, 235–241. [Google Scholar] [CrossRef]
- Wisniewski, M. Removal of arsenic from sulphuric acid solutions. J Radioanal Nucl Chem. 1998, 228, 105–109. [Google Scholar] [CrossRef]
- Jantunen, N.; Virolainen, S.; Latostenmaa, P.; Salminen, J.; Haapalainen, M.; Sainio, T. Removal and recovery of arsenic from concentrated sulfuric acid by solvent extraction. Hydrometallurgy 2019, 187, 101–112. [Google Scholar] [CrossRef]
- El Wakil, A.F.; Zaki, S.A.; Ismaiel, D.A.; Salem, H.M.; Orabi, A.H. Extraction and separation of gallium by solvent extraction with 5-nonyl-2-hydroxyacetophenone oxime: Fundamentals and a case study. Hydrometallurgy 2023, 216, 106022. [Google Scholar] [CrossRef]
- Prusty, S.; Pradhan, S.; Mishra, S. Extraction and separation studies of Nd/Fe and Sm/Co by deep eutectic solvent containing Aliquat 336 and glicerol. J. Chem. Technol. Biotechnol. 2023, 98, 1631–1641. [Google Scholar] [CrossRef]
- Shadbad, M.R.A.; Zaheri, P.; Abolghasemi, H.; Fazel Zahakifar, F. The performance evaluation of Alamine336 in solvent extraction and polymer inclusion membrane methods for valuable ions extraction: A case study of Te(IV) separation intensification. Chem. Eng. Proc. 2023, 184, 109268. [Google Scholar] [CrossRef]
- Zhang, Q.; Sang, Z.; Li, Q.; Gong, J.; Shi, R.; Zhang, B.; Zhang, Z.; Li, S.; Yang, X. Palladium(II) extraction from acidic chloride media using an ionic liquid-based aqueous two-phase system (IL-ATPS) in the presence of dipotassium hydrogen phosphate salting-out agent and reductive stripping with hydrazine hydrate to recover palladium metal. Hydrometallurgy 2023, 216, 106017. [Google Scholar] [CrossRef]
- Distler, P.; Stamberg, K.; John, J.; Harwood, L.M.; Lewis, F.W. Thermodynamic parameters of Am(III), Cm(III) and Eu(III) extraction by CyMe4-BTPhen in cyclohexanone from HNO3 solutions. J. Chem. Thermodyn. 2020, 141, 105955. [Google Scholar] [CrossRef]
- Wang, W.; Wang, X.; Wang, Y.; Li, D.; Liao, W. Physicochemical properties, surface active species and formation of reverse micelles in the Cyanex 923-n-heptane/cerium(IV)-H2SO4 extraction system. J. Chem. Technol. Biotechnol. 2008, 83, 1056–1063. [Google Scholar] [CrossRef]
- Dreisinger, D.B.; Scholey, B.J.Y. Ion exchange removal of antimony and bismuth from copper refinery electrolytes, in: Proceedings of the Copper 95 International Conference, Santiago (Chile), pp. 26–29, 1995.
- Navarro, P.; Simpson, J.; Alguacil, F. J. Removal of antimony (III) from copper in sulphuric acid solutions by solvent extraction with LIX 1104SM. Hydrometallurgy 1999, 53, 121–131. [Google Scholar] [CrossRef]
- Navarro, P.; Alguacil, F.J. Adsorption of antimony and arsenic from a copperelectrorefining solution onto activated carbon. Hydrometallurgy 2002, 66, 101–105. [Google Scholar] [CrossRef]
- Beauchemin, S.; Chen, T.T.; Dutrizac, J.E. Behaviour of antimony and bismuth in copper electrorefining circuits, Can. Metall. Q. 2008, 47, 9–25. [Google Scholar] [CrossRef]
- Wang, X.; Chen, Q.; Yin, Z.; Wang, M.; Xiao, B.; Zhang, F. Homogeneous precipitation of As, Sb and Bi impurities in copper electrolyte during electrorefining. Hydrometallurgy 2010, 105, 355–358. [Google Scholar] [CrossRef]
- Arroyo-Torralvo, F.; Rodríguez-Almansa, A.; Ruiz, I.; Gonzalez, A.I.; Ríos, G.; Ferńandez-Pereira, C.; Vilches-Arenas, L.F. Optimizing operating conditions in an ion-exchange column treatment applied to the removal of Sb and Bi impurities from an electrolyte of a copper electro-refining plant. Hydrometallurgy 2027, 171, 285–297. [Google Scholar] [CrossRef]
- Artzer, A.; Moats, M.; Bender, J. Removal of antimony and bismuth from copper electrorefining electrolyte: Part II—An investigation of two proprietary solvent extraction extractants. JOM 2018, 70, 2856–2863. [Google Scholar] [CrossRef]
- Gonzalez de las Torres, A.I.; Moats, M.S.; Ríos, G.; Rodríguez Almansa, A.D.; Śanchez-Rodas, D. Removal of Sb impurities in copper electrolyte and evaluation of As and Fe species in an electrorefining plant. Metals 2021, 11, 902. [Google Scholar] [CrossRef]
- Gonzalez de las Torres, A.I.; Ríos, G.; Rodriguez Almansa, A.; Sánchez-Rodas, D.; Moats, M.S. . Solubility of bismuth, antimony and arsenic in synthetic and industrial copper electrorefining electrolyte. Hydrometallurgy 2022, 208, 105807. [Google Scholar] [CrossRef]
- Monhemius, A.J.; Swash, P.M. Removing and stabilizing As from copper refining circuits by hydrothermal processing. JOM 1999, 51, 30–33. [Google Scholar] [CrossRef]
- Navarro, P.; Vargas, C.; Araya, E.; Martín, I.; Alguacil, F.J. Arsenic precipitation from metallurgical effluents. Rev. Metal. 2004, 40, 409–412. [Google Scholar] [CrossRef]
- De Klerk, R.J.; Feldmann, T.; Daenzer, R.; Demopoulos, G.P. Continuous circuit coprecipitation of arsenic(V) with ferric iron by lime neutralization: The effect of circuit staging, co-ions and equilibration pH on long-term arsenic retention. Hydrometallurgy 2015, 151, 42–50. [Google Scholar] [CrossRef]
- Doerfelt, C.; Feldmann, T.; Roy, R.; Demopoulos, G.P. Stability of arsenate-bearing Fe(III)/Al(III) co-precipitates in the presence of sulfide as reducing agent under anoxic conditions. Chemosphere 2026, 151, 318–323. [Google Scholar] [CrossRef] [PubMed]
- Stoyanova, V.; Stefanov, E.; Planska, B.; Dimitrova, R.; Valtcheva, I.; Kadiyski, M.; Stoyanova, V. Arsenic removal in a stable form from industrial wastewater in the copper industry. J. Chem. Technol. Metall. 2022, 57, 1038–1050. [Google Scholar]
- Dziwinski, E.; Szymanowski, J. Composition of CYANEX 923, CYANEX 925, CYANEX 921 and TOPO. Solvent Extr. Ion Exch. 1998, 16, 1515–1525. [Google Scholar] [CrossRef]
| [H2SO4], M | [H2SO4]org/[As]org |
|---|---|
| 0.5 1 2 3 4 5 6 |
8 16 32 46 75 89 129 |
| Cyanex 923, % v/v | 1.5 M H2SO4 | 3 M H2SO4 |
|---|---|---|
| 25 50 75 100 |
25 25 35 39 |
50 46 65 69 |
| Cyanex 923, % v/v | Arsenic | Sulphuric acid |
|---|---|---|
| 25 50 75 100 |
28 (24) 52 (54) 64 (64) 69 (71) |
8 (8) 18 (18) 30 (30) 39 (39) |
| Cyanex 923, % v/v | [H2SO4]org/[As]org |
|---|---|
| 25 50 75 100 |
6 8 10 13 |
| O/A ratio | 1.5 M sulphuric acid | 3 M sulphuric acid |
|---|---|---|
| 0.25 0.5 1 2 4 |
5.74 5.78 5.32 5.46 5.39 |
5.55 5.55 5.55 4.70 4.48 |
| Temperature, ºC | DAs | DH2SO4 | βH2SO4/As |
|---|---|---|---|
| 20 30 40 50 60 |
0.83 1.08 1.59 2.40 3.17 |
5.57 6.70 8.20 10.5 14.3 |
6.7 6.2 5.2 4.4 4.5 |
| O/A ratio | DAs | DH2SO4 | βH2SO4/As |
|---|---|---|---|
| 0.5 1 2 4 8 |
1 0.83 0.75 0.76 0.79 |
11 5.6 5.3 4.8 4.8 |
11 6.7 7.1 6.3 6.1 |
| Element | Concentration, g/L | H2SO4, M | % extraction | % H2SO4 extraction |
|---|---|---|---|---|
| Cu(II) Cu(II) Bi(III) Bi(III) Sb(III) Sb(III) Ni(II) Ni(II) As(V) As(V) |
0.1 10 0.1 1 0.07 0.07 7 7 1.5 1.5 |
3 1.5 3 1.5 3 1.5 3 1.5 3 1.5 |
2 5 nil nil 25 26 nil nil 50 54 |
16 18 16 18 16 17 16 17 15 18 |
| Extractant | O/A ratio | % As extraction | % H2SO4 extraction |
|---|---|---|---|
| TBP DBBP Cyanex 923 |
1 2 1 2 1 2 |
18 26 46 54 73 88 |
2 4 10 14 39 50 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




