Submitted:
21 December 2023
Posted:
21 December 2023
Read the latest preprint version here
Abstract
Keywords:
1. Introduction
2. Results
2.1. In Silico Study
2.1.1. Ligand preparation
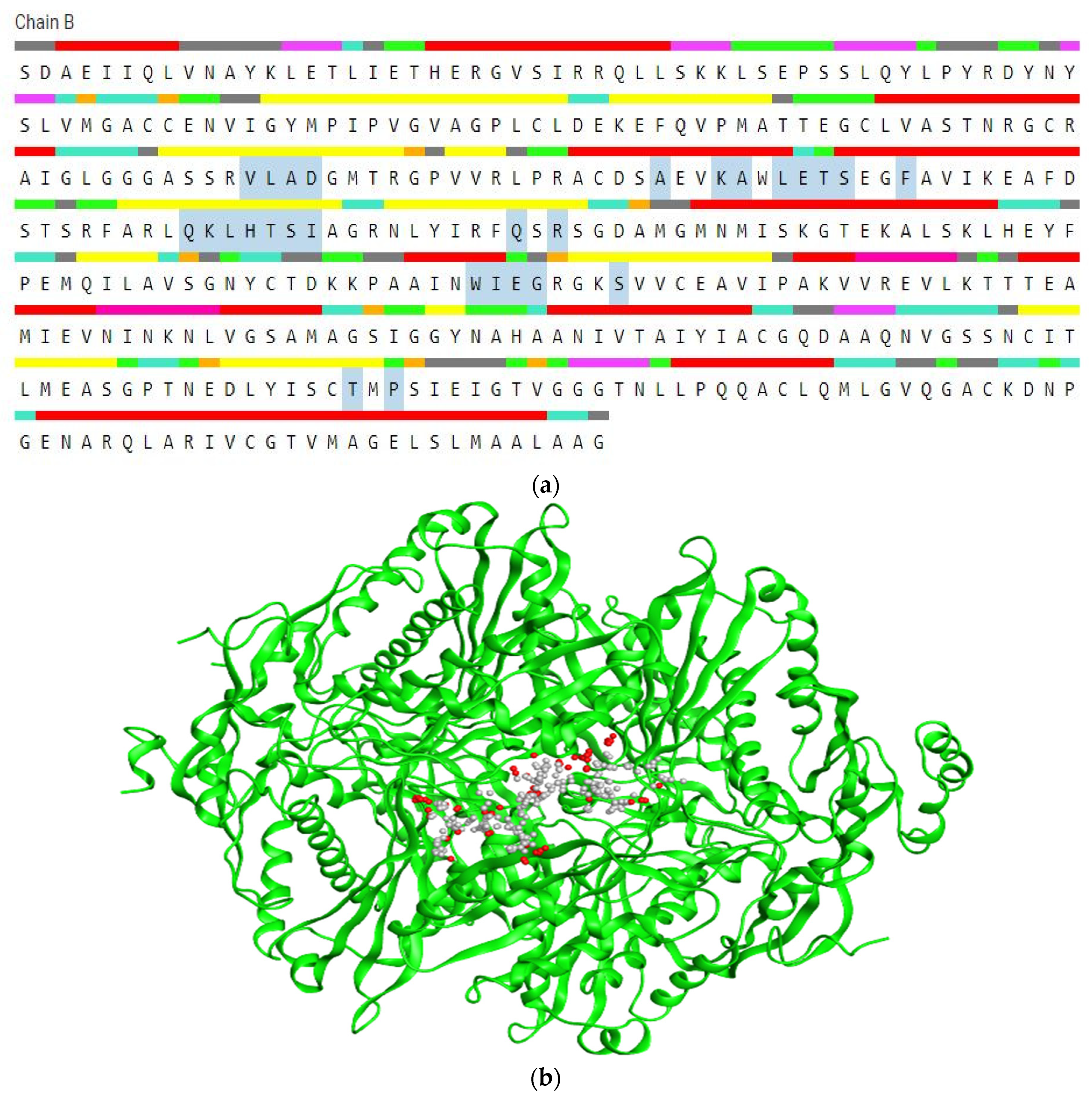
2.1.2. Receptor protein Refinement
2.1.3. Active site determination
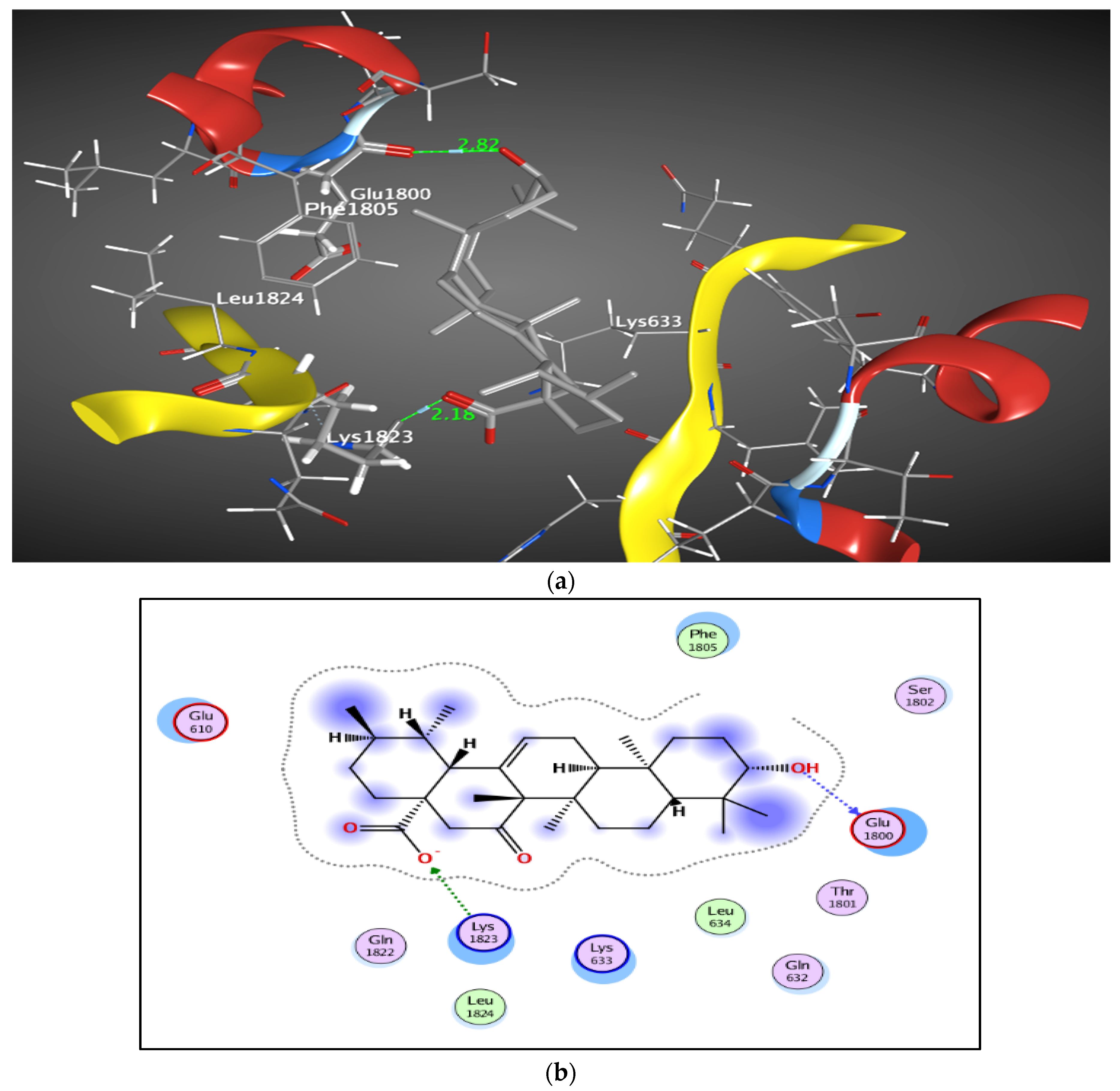
2.1.4. Molecular docking
3. Discussion
4. Materials and methods
4.1. Plant materials
4.2. Extraction and isolation
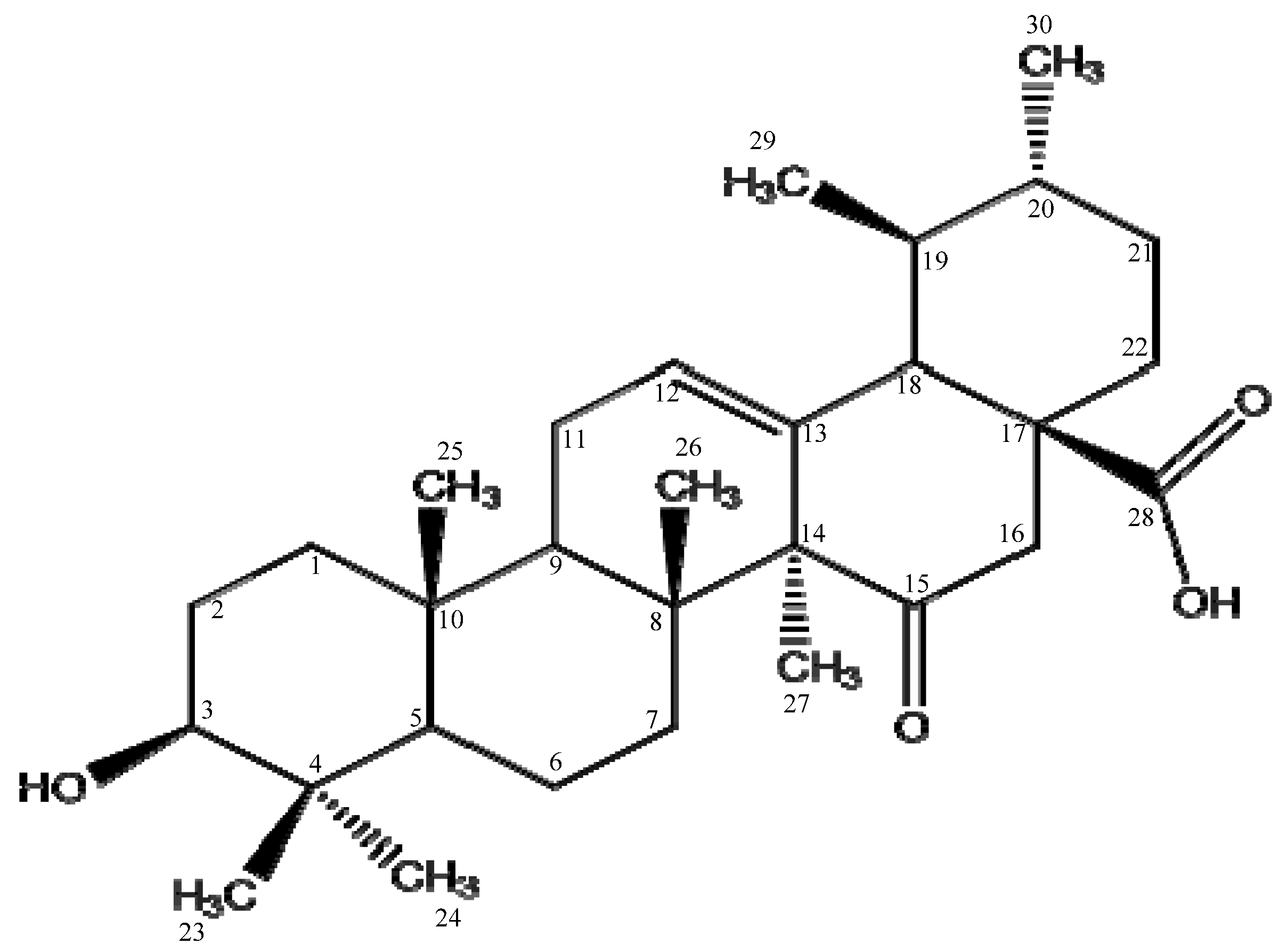
4.3. Physical data of 15-Oxoursolic acid
4.4. In Vivo Study
4.4.1. Animals
4.4.2. Anti-hyperlipidemic activity
- 1)
- NC: Normal control diet and vehicle throughout the study
- 2)
- HFD: High-fat diet and vehicle throughout the study
- 3)
- Test drug: High fat diet + test drug (30mg/kg)
- 4)
- Lovastatin: High fat diet + Lovastatin (5 mg/kg)
4.4.3. Biochemical analysis/Screening
4.5. Molecular Docking
4.5.1. Ligand Preparation
4.5.2. Preparation of protein
4.5.3. Active Site Prediction
4.5.4. Molecular docking Process
4.6. Statistical analysis
5. Conclusion
Author Contributions
References
- Thayyil, A.H.; Surulivel, M.K.M.; Ahmed, M.F.; Ahamed, G.S.S.; Sidheeq, A.; Rasheed, A.; Ibrahim, M. Hypolipidemic Activity of Luffa Aegiptiaca Fruits in Cholesterol Fed Hypercholesterolemic Rabbits. Int. J. Pharm. Appl. 2011, 2, 81–88. [Google Scholar]
- Jacobson, T.A.; Miller, M.; Schaefer, E.J. Hypertriglyceridemia and Cardiovascular Risk Reduction. Clin. Ther. 2007, 29, 763–777. [Google Scholar] [CrossRef]
- Adisakwattana, S.; Moonrat, J.; Srichairat, S.; Chanasit, C.; Tirapongporn, H.; Chanathong, B.; Ngamukote, S.; Mäkynen, K.; Sapwarobol, S. Lipid-Lowering Mechanisms of Grape Seed Extract (Vitis Vinifera L) and Its Antihyperlidemic Activity. J. Med. Plants Res. 2010, 4, 2113–2120. [Google Scholar]
- Stancu, C.; Sima, A. Statins: Mechanism of Action and Effects. J. Cell. Mol. Med. 2001, 5, 378–387. [Google Scholar] [CrossRef] [PubMed]
- Thompson, P.D.; Clarkson, P.; Karas, R.H. Statin-Associated Myopathy. Jama 2003, 289, 1681–1690. [Google Scholar] [CrossRef] [PubMed]
- Hor, S.Y.; Farsi, E.; Yam, M.F.; Nuyah, N.M.; Asmawi, M.Z. Lipid-Lowering Effects of Coriolus Versicolor Extract in Poloxamer 407-Induced Hypercholesterolaemic Rats and High Cholesterol-Fed Rats. J. Med. Plants Res. 2011, 5, 2261–2266. [Google Scholar]
- Zhang, X.; Wu, C.; Wu, H.; Sheng, L.; Su, Y.; Zhang, X.; Luan, H.; Sun, G.; Sun, X.; Tian, Y.; et al. Anti-Hyperlipidemic Effects and Potential Mechanisms of Action of the Caffeoylquinic Acid-Rich Pandanus Tectorius Fruit Extract in Hamsters Fed a High Fat-Diet. PLoS One 2013, 8. [Google Scholar] [CrossRef] [PubMed]
- Ferreira, E.A.; Gris, E.F.; Rebello, J.M.; Correia, J.F.G.; De Oliveira, L.F.S.; Filho, D.W.; Pedrosa, R.C. The 2′,4′,6′-Trihydroxyacetophenone Isolated from Myrcia Multiflora Has Antiobesity and Mixed Hypolipidemic Effects with the Reduction of Lipid Intestinal Absorption. Planta Med. 2011, 77, 1569–1574. [Google Scholar] [CrossRef] [PubMed]
- Mayaux, J.F.; Bousseau, A.; Pauwels, R.; Huet, T.; Hénin, Y.; Dereu, N.; Evers, M.; Soler, F.; Poujade, C.; De Clercq, E.; et al. Triterpene Derivatives That Block Entry of Human Immunodeficiency Virus Type 1 into Cells. Proc. Natl. Acad. Sci. USA 1994, 91, 3564–3568. [Google Scholar] [CrossRef] [PubMed]
- Kashiwada, Y.; Nagao, T.; Hashimoto, A.; Ikeshiro, Y.; Okabe, H.; Cosentino, L.M.; Lee, K.H. Anti-AIDS Agents 38. Anti-HIV Activity of 3-O-Acyl Ursolic Acid Derivatives. J. Nat. Prod. 2000, 63, 1619–1622. [Google Scholar] [CrossRef]
- Zhu, Y.M.; Shen, J.K.; Wang, H.K.; Cosentino, L.M.; Lee, K.H. Synthesis and Anti-HIV Activity of Oleanolic Acid Derivatives. Bioorganic Med. Chem. Lett. 2001, 11, 3115–3118. [Google Scholar] [CrossRef]
- Ma, C.; Nakamura, N.; Miyashiro, H.; Hattori, M.; Shimotohno, K. Inhibitory Effects of Constituents from Cynomorium Songaricum and Related Triterpene Derivatives on HIV-1 Protease. Chem. Pharm. Bull. 1999, 47, 141–145. [Google Scholar] [CrossRef] [PubMed]
- Wolska, K.I.; Grudniak, A.M.; Fiecek, B.; Kraczkiewicz-Dowjat, A.; Kurek, A. Antibacterial Activity of Oleanolic and Ursolic Acids and Their Derivatives. Cent. Eur. J. Biol. 2010, 5, 543–553. [Google Scholar] [CrossRef]
- Lee, K.H.; Lin, Y.M.; Wu, T.S.; Zhang, D.C.; Yamagishi, T.; Hayashi, T.; Hall, I.H.; Chang, J.J.; Wu, R.Y.; Yang, T.H. The Cytotoxic Principles of Prunella Vulgaris, Psychotria Serpens, and Hyptis Capitata: Ursolic Acid and Related Derivatives. Planta Med. 1988, 54, 308–311. [Google Scholar] [CrossRef] [PubMed]
- Lin, C.N.; Lu, C.M.; Cheng, M.K.; Gan, K.H.; Won, S.J. The Cytotoxic Principles of Solanum Incanum. J. Nat. Prod. 1990, 53, 513–516. [Google Scholar] [CrossRef]
- Liu, J. Pharmacology of Oleanolic Acid and Ursolic Acid. J. Ethnopharmacol. 1995, 49, 57–68. [Google Scholar] [CrossRef]
- Machaba, K.E.; Cobongela, S.Z.Z.; Mosa, R.A.; Oladipupo, L.A.; Djarova, T.G.; Opoku, A.R. In Vivo Anti-Hyperlipidemic Activity of the Triterpene from the Stem Bark of Protorhus Longifolia (Benrh) Engl. Lipids Health Dis. 2014, 13. [Google Scholar] [CrossRef]
- Ali, S.; Nisar, M.; Qaisar, M.; Khan, A.; Khan, A.A. Evaluation of the Cytotoxic Potential of a New Pentacyclic Triterpene from Rhododendron Arboreum Stem Bark. Pharm. Biol. 2017, 55, 1927–1930. [Google Scholar] [CrossRef]
- Chaudhari, H.S.; Bhandari, U.; Khanna, G. Preventive Effect of Embelin from Embelia Ribes on Lipid Metabolism and Oxidative Stress in High-Fat Diet-Induced Obesity in Rats. Planta Med. 2012, 78, 651–657. [Google Scholar] [CrossRef] [PubMed]
- Derosa, G.; Salvadeo, S.; Cicero, A.F.G. Prospects for the Development of Novel Anti-Hyperlipidemic Drugs. Curr. Opin. Investig. Drugs 2006, 7, 826–833. [Google Scholar] [PubMed]
- Karalis, D.G.; Victor, B.; Ahedor, L.; Liu, L. Use of Lipid-Lowering Medications and the Likelihood of Achieving Optimal LDL-Cholesterol Goals in Coronary Artery Disease Patients. Cholesterol 2012, 2012. [Google Scholar] [CrossRef]
- Santos, F.A.; Frota, J.T.; Arruda, B.R.; De Melo, T.S.; Da Silva, A.A.D.C.A.; Brito, G.A.D.C.; Chaves, M.H.; Rao, V.S. Antihyperglycemic and Hypolipidemic Effects of A,β-Amyrin, a Triterpenoid Mixture from Protium Heptaphyllum in Mice. Lipids Health Dis. 2012, 11. [Google Scholar] [CrossRef]
- Gutierrez, R.M.P. Evaluation of the Hypoglycemic and Hypolipidemic Effects of Triterpenoids from Prosthechea Michuacana in Streptozotocin-Induced Type 2 Diabetic Mice. Pharmacologia 2013, 4, 170–179. [Google Scholar] [CrossRef]
- Sudhahar, V.; Kumar, S.A.; Sudharsan, P.T.; Varalakshmi, P. Protective Effect of Lupeol and Its Ester on Cardiac Abnormalities in Experimental Hypercholesterolemia. Vascul. Pharmacol. 2007, 46, 412–418. [Google Scholar] [CrossRef]
- Brown, A.W.; Hang, J.; Dussault, P.H.; Carr, T.P. Plant Sterol and Stanol Substrate Specificity of Pancreatic Cholesterol Esterase. J. Nutr. Biochem. 2010, 21, 736–740. [Google Scholar] [CrossRef]
- Ansell, B.J.; Watson, K.E.; Fogelman, A.M.; Navab, M.; Fonarow, G.C. High-Density Lipoprotein Function: Recent Advances. J. Am. Coll. Cardiol. 2005, 46, 1792–1798. [Google Scholar] [CrossRef]
- Pires, D.E.V.; Blundell, T.L.; Ascher, D.B. pkCSM: Predicting Small-Molecule Pharmacokinetic and Toxicity Properties Using Graph-Based Signatures. J. Med. Chem. 2015, 58, 4066–4072. [Google Scholar] [CrossRef]
- Banerjee, P.; Eckert, A.O.; Schrey, A.K.; Preissner, R. ProTox-II: A Webserver for the Prediction of Toxicity of Chemicals. Nucleic Acids Res. 2018, 46, W257–W263. [Google Scholar] [CrossRef] [PubMed]
- Lin, S.H.; Huang, K.J.; Weng, C.F.; Shiuan, D. Exploration of Natural Product Ingredients as Inhibitors of Human HMG-CoA Reductase through Structure-Based Virtual Screening. Drug Des. Devel. Ther. 2015, 9, 3313–3324. [Google Scholar] [CrossRef] [PubMed]
- Fatriansyah, J.F.; Rizqillah, R.K.; Yandi, M.Y.; Fadilah; Sahlan, M. Molecular Docking and Dynamics Studies on Propolis Sulabiroin-A as a Potential Inhibitor of SARS-CoV-2. J. King Saud Univ. - Sci. 2022, 34. [Google Scholar] [CrossRef]
- Dolgonosov, A.M. The Universal Relationship between the Energy and Length of a Covalent Bond Derived from the Theory of Generalized Charges. Russ. J. Inorg. Chem. 2017, 62, 344–350. [Google Scholar] [CrossRef]
- Luo, Y.; Li, G.; Li, G.; Yan, J.; Yi, J.; Zhang, G. Discovery and Identification of 2-Phenylethyl 2,6-Dihydroxybenzoate as a Natural Lipid-Lowering Lead. Planta Med. 2011, 77, 2047–2049. [Google Scholar] [CrossRef] [PubMed]
- Durendić-Brenesel, M.; Popović, T.; Pilija, V.; Arsić, A.; Milić, M.; Kojić, D.; Jojić, N.; Milić, N. Hypolipidemic and Antioxidant Effects of Buckwheat Leaf and Flower Mixture in Hyperlipidemic Rats. Bosn. J. Basic Med. Sci. 2013, 13, 100–108. [Google Scholar] [CrossRef]
- Imafidon, K.E.; Okunrobo, L.O. Study on Biochemical Indices of Liver Function Tests of Albino Rats Supplemented with Three Sources of Vegetable Oils. Niger. J. Basic Appl. Sci. 2012, 19, 105–110. [Google Scholar]
- Rasekh, H.R.; Khoshnood-Mansourkhani, M.J.; Kamalinejad, M. Hypolipidemic Effects of Teucrium Polium in Rats. Fitoterapia 2001, 72, 937–939. [Google Scholar] [CrossRef] [PubMed]
- Friedewald, W.T.; Levy, R.I.; Fredrickson, D.S. Estimation of the Concentration of Low-Density Lipoprotein Cholesterol in Plasma, without Use of the Preparative Ultracentrifuge. Clin. Chem. 1972, 18, 499–502. [Google Scholar] [CrossRef]
- Mir, A.; Ismatullah, H.; Rauf, S.; Niazi, U.H.K. Identification of Bioflavonoid as Fusion Inhibitor of Dengue Virus Using Molecular Docking Approach. Informatics Med. Unlocked 2016, 3, 1–6. [Google Scholar] [CrossRef]
- Iqbal, D.; Khan, M.S.; Khan, M.S.; Ahmad, S.; Hussain, M.S.; Ali, M. Bioactivity Guided Fractionation and Hypolipidemic Property of a Novel HMG-CoA Reductase Inhibitor from Ficus Virens Ait. Lipids Health Dis. 2015, 14. [Google Scholar] [CrossRef]
- Marahatha, R.; Basnet, S.; Bhattarai, B.R.; Budhathoki, P.; Aryal, B.; Adhikari, B.; Lamichhane, G.; Poudel, D.K.; Parajuli, N. In-Silico Analysis of Secondary Metabolites That Modulates Enzymes of Cholesterol Target. bioRxiv, 2020; 2020.07.24.219998. [Google Scholar]
- Punetha, A.; Muthukumaran, J.; Hemrom, A.J.; Arumugam, N.; Jayakanthan, M. Sundar Durai Towards Understanding the Regulation of Rubber Biosynthesis: Insights into the Initiator and Elongator Enzymes. J. Bioinforma. Seq. Anal. 2010, 2, 001–010. [Google Scholar]
- Tian, W.; Chen, C.; Lei, X.; Zhao, J.; Liang, J. CASTp 3.0: Computed Atlas of Surface Topography of Proteins. Nucleic Acids Res. 2018, 46, W363–W367. [Google Scholar] [CrossRef]
- Rabbi, M.F.; Akter, S.A.; Hasan, M.J.; Amin, A. In Silico Characterization of a Hypothetical Protein from Shigella Dysenteriae ATCC 12039 Reveals a Pathogenesis-Related Protein of the Type-VI Secretion System. Bioinform. Biol. Insights 2021, 15. [Google Scholar] [CrossRef] [PubMed]
- Tomar, N.R.; Singh, V.; Marla, S.S.; Chandra, R.; Kumar, R.; Kumar, A. Molecular Docking Studies with Rabies Virus Glycoprotein to Design Viral Therapeutics. Indian J. Pharm. Sci. 2010, 72, 486–490. [Google Scholar] [CrossRef] [PubMed]
- Alhumaydhi, F.A.; Rauf, A.; Rashid, U.; Bawazeer, S.; Khan, K.; Mubarak, M.S.; Aljohani, A.S.M.; Khan, H.; El-Saber Batiha, G.; El-Esawi, M.A.; et al. In Vivo and in Silico Studies of Flavonoids Isolated from Pistacia Integerrima as Potential Antidiarrheal Agents. ACS Omega 2021, 6, 15617–15624. [Google Scholar] [CrossRef]
- Alhumaydhi, F.A.; Aljohani, A.S.M.; Rashid, U.; Shah, Z.A.; Rauf, A.; Muhammad, N.; Al-Awthan, Y.S.; Bahattab, O.S. In Vivo Antinociceptive, Muscle Relaxant, Sedative, and Molecular Docking Studies of Peshawaraquinone Isolated from Fernandoa Adenophylla (Wall. Ex G. Don) Steenis. ACS Omega 2021, 6, 996–1002. [Google Scholar] [CrossRef]
- Iftikhar, F.; Yaqoob, F.; Tabassum, N.; Jan, M.S.; Sadiq, A.; Tahir, S.; Batool, T.; Niaz, B.; Ansari, F.L.; Choudhary, M.I.; et al. Design, Synthesis, in-Vitro Thymidine Phosphorylase Inhibition, in-Vivo Antiangiogenic and in-Silico Studies of C-6 Substituted Dihydropyrimidines. Bioorg. Chem. 2018, 80, 99–111. [Google Scholar] [CrossRef] [PubMed]
| Parameter (mg/dl) | Normal | Positive Control (HFD) |
15-oxoursolic acid (30 mg/kg) | Lovastatin (5 mg/kg) |
|---|---|---|---|---|
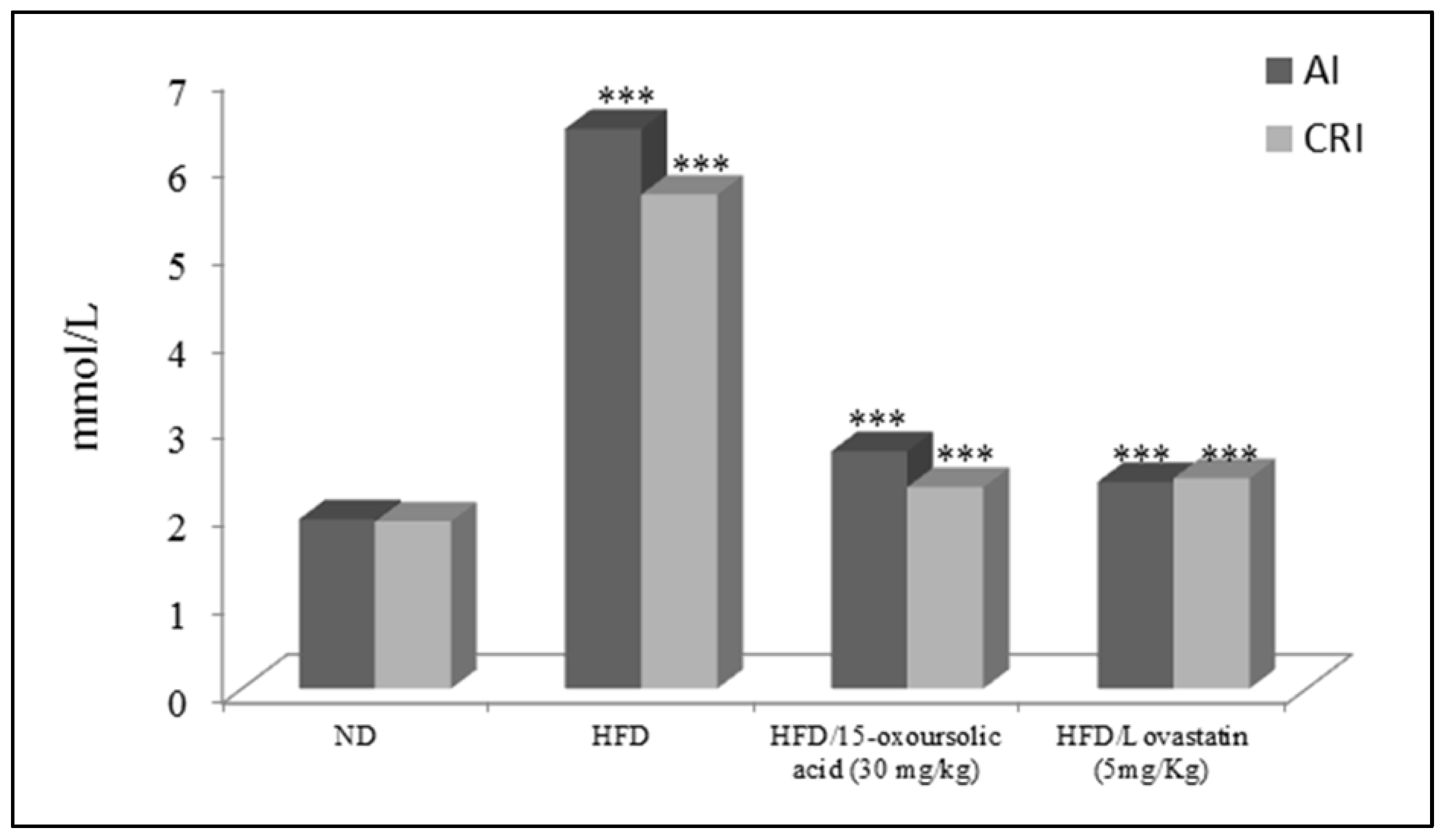
| Total cholesterol | 37.90 ± 0.45 | 58.34 ± 0.21 | 42.65 ± 0.54 * | 43.17 ± 0.12 * |
| Triglycerides | 28.56 ± 0.15 | 45.45 ± 0.39 | 32.33 ± 0.16 * | 32.16 ± 0.16 * |
| HDLc | 19.78 ± 0.34 | 10.34 ± 0.18 | 18.56 ± 0.34 * | 18.24 ± 0.59 * |
| LDLc | 37.11 ± 0.11 | 66.12 ± 0.10 | 50.34 ± 0.15 * | 43.10 ± 0.13 * |
| VLDLc | 12.45 ± 0.22 | 19.34 ± 0.16 | 13.15 ± 0.45 * | 14.32 ± 0.12 * |
| Group | AST(U/L) | ALT(U/L) | ALP(U/L) |
|---|---|---|---|
| ND | 215.34±12.34 | 40.96±10.01 | 70.00±12.19 |
| HFD | 295.00±23.12** | 82.93±32.24 | 215.23±10.72 |
| HFD/Test drug (30mg/kg, b.w) | 155.13±20.71 | 39.90±12.34 | 167.51±12.45 |
| HFD/ Lovastatin (5 mg/kg) | 185.21±17.10 | 42.25±13.34 | 105.12±53.34 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




