Submitted:
19 December 2023
Posted:
19 December 2023
You are already at the latest version
Abstract
Keywords:
Introduction:
| Antifungal Class | Drug Name | Specific Target |
| Allylamines | Naftifine | Squalene epoxidase |
| Terbinafine | ||
| Azoles |
Imidazole: clotrimazole, miconazole, bifonazole, ketoconazole, luliconazole, lanoconazole . |
Cytochrome P450, 14α- Lanosterol Demethylase. |
| Triazole: itraconazole, fluconazole, efinaconazole, Albaconazole, posaconazole, ravuconazole, fosravuconazole, isavuconazole | ||
| Tetrazole: Oteseconazole | ||
| Polyenes | Amphotericin B | Membrane barrier function. |
| Nystatin | ||
| Morpholines | Amorolfine | Sterol reductase and Isomerase. |
| Thiocarbamate | Tolnaftate | Squalene epoxidase |
| Others | Griseofulvin | Sliding of Microtubules. |
Antifungal resistance mechanisms:
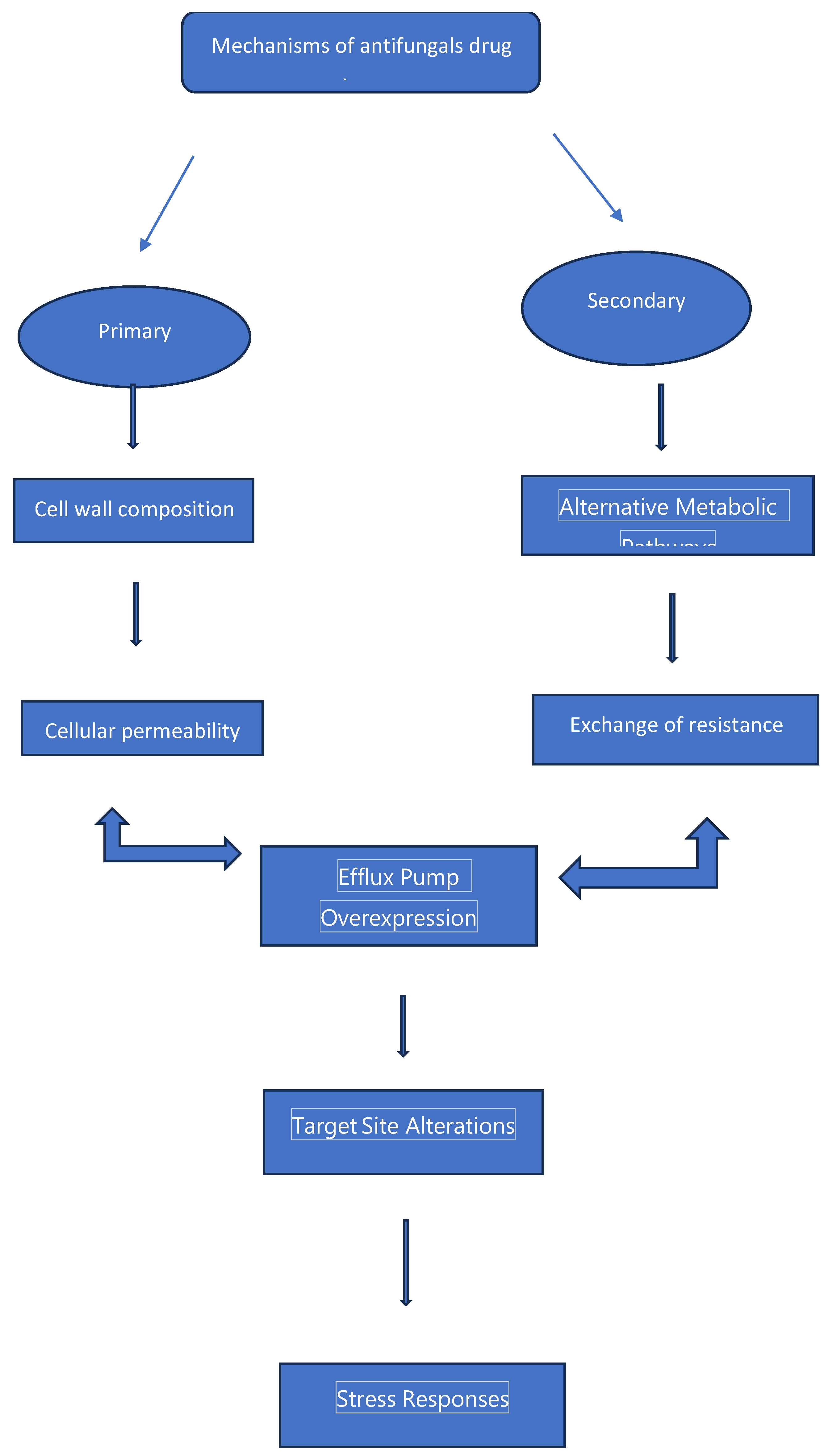
Assessment of antifungal resistance:
Terbinafine resistance:
Azole resistance:
Griseofulvin:
Development of Resistance to recently approved Triazoles antifungals:
Resistance to topical antifungal agents:
Antifungals cross resistance:
Emerging Fungal Pathogens with Multiple Drug Resistance:
How to overcome antifungal resistance:
Conclusion:
References
- Singh, A., Masih, A., Khurana, A., et al., 2018. High terbinafine resistance in Trichophyton interdigitale isolates in Delhi, India harbouring mutations in the squalene epoxidase gene. Mycoses 61, 477–484.
- Saunte, D.M.; Arendrup, M.C. Månedsbladet Rationel Farmakoterapi 2015, nr. 3. Svampeinfektioner i Hud, Hår og Negle.
- White TC, Marr KA, Bowden RA. 1998. Clinical, cellular, and molecular factors that contribute to antifungal drug resistance. Clin Microbiol Rev 11: 382–402. [CrossRef]
- Jabet A, Brun S, Normand AC, Imbert S, Akhoundi M, Dannaoui E, Audiffred L, Chasset F, Izri A, Laroche L, Piarroux R, Bachmeyer C, Hennequin C, Sabater AM. Extensive Dermatophytosis Caused by Terbinafine-Resistant Trichophyton indotineae, France. Emerg Infect Dis. 2022 Jan;28(1):229-233. [CrossRef]
- Bishnoi, A., Vinay, K., Dogra, S., 2018. Emergence of recalcitrant dermatophytosis in India. Lancet Infect. Dis. 18, 250–251. [CrossRef]
- Gupta AK, Renaud HJ, Quinlan EM, et al. The growing problem of antifungal resistance in onychomycosis and other superficial mycoses. Am J Clin Dermatol. 2021;22(2): 149–157. [CrossRef]
- Antifungal resistance j fungal diseases j CDC [Internet]. 2020. [cited 2021 Apr 19].
- Mukherjee PK, Leidich SD, Isham N, et al. Clinical Trichophyton rubrum strain exhibiting primary resistance to terbinafine. Antimicrob Agents Chemother. 2003;47(1): 82–86. [CrossRef]
- Ghannoum M. Azole resistance in dermatophytes: prevalence and mechanism of action. J Am Podiatr Med Assoc. 2016;106(1):79–86. [CrossRef]
- Jiang Y, Luo W, Verweij PE, et al. regional differences in antifungal susceptibility of the prevalent dermatophyte Trichophyton rubrum. Mycopathologia. 2021;186(1):53–70. [CrossRef]
- Marichal P, Koymans L, Willemsens S, et al. Contribution of mutations in the cytochrome P450 14alpha-demethylase (Erg11p, Cyp51p) to azole resistance in Candida albicans.
- Ghannoum M, Isham N. Fungal nail infections (onychomycosis): a never-ending story? PLoS Pathog. 2014;10(6): e1004105.
- Yadav A., Jain K., Wang Y., Pawar K., Kaur H., Sharma H.K., Tripathy V., Singh A., Xu J., Chowdhary A. Candida auris on Apples: Diversity and Clinical Significance. mBio. 2022;13:e0051822.
- Arendrup MC, Cuenca-Estrella M, Lass-Florl C, Hope WW. Breakpoints for antifungal agents: an update from EUCAST focussing on echinocandins against Candida spp and triazoles against Aspergillus spp. Drug Resist Updat 2013; 16: 81–95. [CrossRef]
- Pfaller MA, Andes D, Arendrup MC, et al. Clinical breakpoints for voriconazole and Candida spp revisited: review of microbiologic, molecular, pharmacodynamic, and clinical data as they pertain to the development of species-specific interpretive criteria. Diagn Microbiol Infect Dis 2011; 70: 330–43. [CrossRef]
- Pfaller MA, Diekema DJ, Andes D, et al. Clinical breakpoints for the echinocandins and candida revisited: integration of molecular, clinical, and microbiological data to arrive at species-specific interpretive criteria. Drug Resist Updat 2011; 14: 164–76. [CrossRef]
- Espinel-Ingroff A, Pfaller MA, Bustamante B, et al. Multilaboratory study of epidemiological cutoff values for detection of resistance in eight Candida species to fluconazole, posaconazole, and voriconazole. Antimicrob Agents Chemother 2014; 58: 2006–12. [CrossRef]
- Turnidge J, Kahlmeter G, Kronvall G. Statistical characterisation of bacterial wild-type MIC value distributions and the determination of epidemiological cut-off values. Clin Microbiol Infect 2006; 12: 418–25. [CrossRef]
- Espinel-Ingroff A, Arendrup MC, Pfaller MA, et al. Interlaboratory variability of caspofungin MICs for Candida spp using CLSI and EUCAST methods: should the clinical laboratory be testing this agent? Antimicrob Agents Chemother 2013; 57: 5836–42.
- Perlin DS, Rautemaa-Richardson R, Alastruey-Izquierdo A. The global problem of antifungal resistance: prevalence, mechanisms, and management. Lancet Infect Dis. 2017 Dec;17(12): e383-e392. Epub 2017 Jul 31. PMID: 28774698. [CrossRef]
- Crawford F, Young P, Godfrey C, et al. Oral treatments for toenail onychomycosis: a systematic review. Arch Dermatol. 2002; 138:811–816.
- Singh A, Masih A, Khurana A, et al. High terbinafine resistance in Trichophyton interdigitale isolates in Delhi, India harbouring mutations in the squalene epoxidase gene. Mycoses. 2018;61(7):477–484.
- Santos HL, Lang EAS, Segato F, et al. Terbinafine resistance conferred by multiple copies of the salicylate 1- monooxygenase gene in Trichophyton rubrum. Med Mycol. 2018;56(3):378–381. [CrossRef]
- Singh A, Masih A, Monroy-Nieto J, et al. A unique multidrug-resistant clonal Trichophyton population distinct from Trichophyton mentagrophytes/Trichophyton interdigitale complex causing an ongoing alarming dermatophytosis outbreak in India: genomic insights and resistance profile. Fungal Genet Biol. 2019;133(103266): 103266. [CrossRef]
- Kano R, Kimura U, Kakurai M, et al. Trichophyton indotineae sp. nov.: a new highly terbinafine-resistant anthropophilic dermatophyte species. Mycopathologia. 2020;185(6): 947–958. [CrossRef]
- Ebert A, Monod M, Salamin K, et al. Alarming India-wide phenomenon of antifungal resistance in dermatophytes: a multicentre study. Mycoses. 2020;63(7):717–728. [CrossRef]
- Salehi Z, Shams-Ghahfarokhi M, Razzaghi-Abyaneh M. Antifungal drug susceptibility profile of clinically important dermatophytes and determination of point mutations in terbinafine-resistant isolates. Eur J Clin Microbiol Infect Dis. 2018;37(10):1841–1846. [CrossRef]
- Nenoff P, Verma SB, Ebert A, Süß A, Fischer E, Auerswald E, et al. Spread of terbinafine-resistant Trichophyton mentagrophytes type VIII (India) in Germany—“the tip of the iceberg? J Fungi (Basel). 2020; 6:207.
- Saunte D, Pereiro-Ferreiros M, Rodrıguez-Cerdeira C, et al. Emerging antifungal treatment failure of dermatophytosis in Europe: take care or it may become endemic. J Eur Acad Dermatol Venereol. 2021;35(7):1582–1586. [CrossRef]
- Gaurav V, Bhattacharya SN, Sharma N, et al. Terbinafine resistance in dermatophytes: time to revisit alternate antifungal therapy. J Mycol Med. 2021;31(1):101087. [CrossRef]
- Mast, N., Zheng, W., Stout, C.D., et al., 2013. Antifungal azoles: structural insights into undesired tight binding to cholesterol metabolizing CYP46A1. Mol. Pharmacol. 84, 86–94. [CrossRef]
- Pai V, Ganavalli A, Kikkeri NN. Antifungal resistance in dermatology. Indian J Dermatol. 2018;63(5):361–368. [CrossRef]
- Coleman, J.J1., Mylonakis, E., 2009. Efflux in fungi : la pièce de résistance. PLoSPathog. 5, e1000486.
- Martinez-Rossi, N.M., Bitencourt, T.A., Peres, N.T.A., Lang, E.A.S., Gomes, E.V., Quaresemin, N.R., Martins, M.P., Lopes, L., Rossi, A., 2018. Dermatophyte resistance to antifungal drugs: mechanisms and prospectus. Front. Microbiol. 9, 1108. [CrossRef]
- Wilkens, S., 2015. Structure and mechanism of ABC transporters. F1000Prime Rep. 7, 14. [CrossRef]
- Fachin, A.L., Ferreira-Nozawa, M.S., Maccheroni Jr, W., et al., 2006. Role of the ABC transporter TruMDR2 in terbinafine, 4- nitroquinoline N-oxide and ethidium bromide susceptibility in Trichophyton rubrum. J. Med. Microbiol. 55 (Pt 8), 1093–1099.
- Feng, W., Yang, J., Xi, Z., et al., 2017. Mutations and/or overexpression of ERG4 and ERG11 genes in clinical azoles-resistant isolates of Candida albicans. Microb. Drug Resist. 23, 563–570.
- Xiang, M.J., Liu, J.Y., Ni, P.H., et al., 2013. Erg11 mutations associated with azole resistance in clinical isolates of Candida albicans. FEMS Yeast Res. 13 (4), 386–393. [CrossRef]
- Peres, N.T., Sanches, P.R., Falcão, J.P., et al., 2010. Transcriptional profiling reveals the expression of novel genes in response to various stimuli in the human dermatophyte Trichophyton rubrum. BMC Microbiol. 10, 39. [CrossRef]
- Paião, F.G., Segato, F., Cursino-Santos, J.R., et al., 2007. Analysis of Trichophyton rubrum gene expression in response to cytotoxic drugs. FEMS Microbiol. Lett. 271, 180–186 Epub 2007 Apr 10.
- Martinez-Rossi, N.M., Peres, N.T., Rossi, A., 2017. Pathogenesis of dermatophytosis: sensing the host tissue. Mycopathologia 182, 215–227. [CrossRef]
- Perlin, D.S., Shor, E., Zhao, Y., 2015. Update on antifungal drug resistance. Curr. Clin. Microbiol. Rep. 2, 84–85. [CrossRef]
- Martinez-Rossi, Nilce M., Tiago, R., et al., 2016. Heat shock proteins in dermatophytes: current advances and perspectives. Curr. Genom. 17, 99–111.
- Hsieh A, Quenan S, Riat A, et al. A new mutation in the SQLE gene of Trichophyton mentagrophytes associated to terbinafine resistance in a couple with disseminated tinea corporis. J Mycol Med. 2019;29(4):352–355. [CrossRef]
- Gaurav V, Bhattacharya SN, Sharma N, et al. Terbinafine resistance in dermatophytes: time to revisit alternate antifungal therapy. J Mycol Med. 2021;31(1):101087. [CrossRef]
- Nofal A, Fawzy MM, El-Hawary EE. Successful treatment of resistant onychomycosis with voriconazole in a liver transplant patient. Dermatol Ther. 2020;33: e14014. [CrossRef]
- Gu D, Hatch M, Ghannoum M, et al. Treatment-resistant dermatophytosis: a representative case highlighting an emerging public health threat. JAAD Case Rep. 2020;6(11): 1153–1155. [CrossRef]
- Farowski F, Vehreschild JJ, Cornely OA. Posaconazole: a next generation triazole antifungal. Future Microbiol. 2007; 2(3):231–243. [CrossRef]
- Epstein, W.L., Shah, V.P., Riegelman, S., 1972. Griseofulvin levels in stratum corneum. Study after oral administration in man. Arch. Dermatol. 106, 344–348. [CrossRef]
- Yenişehirli, G1., Tunçoğlu, E., Yenişehirli, A., Bulut, Y., 2013. In vitro activities of antifungal drugs against dermatophytes isolated in Tokat, Turkey. Int. J. Dermatol. 52, 1557–1560. [CrossRef]
- Chadeganipour, M1., Nilipour, S., Havaei, A., 2004. In vitro evaluation of griseofulvin against clinical isolates of dermatophytes from Isfahan. Mycoses 47, 503–507. [CrossRef]
- Ghannoum, M.A., Chaturvedi, V., Espinel-Ingroff, A., et al., 2004. Intra- and interlaboratory study of a method for testing the antifungal susceptibilities of dermatophytes. J. Clin. Microbiol. 7, 2977–2979. [CrossRef]
- Mistik, S., Ferahbas, A., Koc, A.N., Ayangil, D., Ozturk, A., 2006. What defines the quality of patient care in tinea pedis? J. Eur. Acad. Dermatol. Venereol. 20 (2), 158–165.
- Rudramurthy, S.M., Shankarnarayan, S.A., Dogra, S., et al., 2018. Mutation in the squalene epoxidase gene of Trichophyton interdigitale and Trichophyton rubrum associated with Allylamine resistance. Antimicrob. Agents Chemother. 62, e02522–e2617. [CrossRef]
- Korting, H.C., Rosenkranz, S., 1990. In vitro susceptibility of dermatophytes from Munich to griseofulvin, miconazole and ketoconazole. Mycoses 33, 136–139. [CrossRef]
- Martins, M.P., Franceschini, A.C.C., Jacob, T.R., et al., 2016. Compensatory expression of multidrug-resistance genes encoding ABC transporters in dermatophytes. 65, 605–610. [CrossRef]
- Cervelatti, E.P., Fachin, A.L., Ferreira-Nozawa, M.S., et al., 2006. Molecular cloning and characterization of a novel ABC transporter gene in the human pathogen Trichophyton rubrum. Med. Mycol. 44, 141–147. [CrossRef]
- Osborne, C.S., Hofbauer, B., Favre, B., et al., 2003. In vitro analysis of the ability of Trichophyton rubrum to become resistant to terbinafine. Antimicrob. Agents Chemother. 47, 3634–3636. [CrossRef]
- Favre, B., Ryder, N.S., 1996. Characterization of squalene epoxidase activity from the dermatophyte Trichophyton rubrum and its inhibition by terbinafine and other antimycotic agents. Antimicrob. Agents Chemother. 40, 443–447. [CrossRef]
- Ghelardi, E., Celandroni, F., Gueye, S.A., et al., 2014. Potential of Ergosterol synthesis inhibitors to cause resistance or cross-resistance in Trichophyton rubrum. Antimicrob. Agents Chemother. 58, 2825–2829. [CrossRef]
- Hryncewicz-Gwóźdź, A., Kalinowska, K., Plomer-Niezgoda, E., et al., 2013. Increase in resistance to fluconazole and itraconazole in Trichophyton rubrum clinical isolates by sequential passages in vitro under drug pressure. Mycopathologia 176, 49–55. [CrossRef]
- Singh, A., Kalinowska, K., Plomer-Niezgoda, E., et al., 2013. Increase in resistance to fluconazole and itraconazole in Trichophyton rubrum clinical isolates by sequential passages in vitro under drug pressure. Mycopathologia 176, 49–55. [CrossRef]
- McTaggart LR, Cabrera A, Cronin K, Kus JV. Antifungal Susceptibility of Clinical Yeast Isolates from a Large Canadian Reference Laboratory and Application of Whole-Genome Sequence Analysis to Elucidate Mechanisms of Acquired Resistance. Antimicrob Agents Chemother. 2020 Aug 20;64(9):e00402-20. [CrossRef]
- Fattahi A, Shirvani F, Ayatollahi A, et al. Multidrug-resistant Trichophyton mentagrophytes genotype VIII in an Iranian family with generalized dermatophytosis: report of four cases and review of literature. Int J Dermatol. 2021;60(6): 686–692. [CrossRef]
- Suß A, Uhrlaß S, Ludes A, et al. [Extensive tinea corporis € due to a terbinafine-resistant Trichophyton mentagrophytes isolate of the Indian genotype in a young infant from Bahrain in Germany]. Hautarzt. 2019;70(11):888–896.
- Das S, De A, Saha R, et al. The current Indian epidemic of dermatophytosis: a study on causative agents and sensitivity patterns. Indian J Dermatol. 2020;65(2):118–122. [CrossRef]
- Marquez L, Quave CL. Prevalence, and therapeutic challenges of fungal drug resistance: role for plants in drug discovery. Antibiotics (Basel). 2020;9(4):150. [cited 2021 Apr 19] Available from: pmc/articles/PMC7235788. [CrossRef]
- Frías-De-León MG, Hernández-Castro R, Conde-Cuevas E, García-Coronel IH, Vázquez-Aceituno VA, Soriano-Ursúa MA, Farfán-García ED, Ocharán-Hernández E, Rodríguez-Cerdeira C, Arenas R, Robledo-Cayetano M, Ramírez-Lozada T, Meza-Meneses P, Pinto-Almazán R, Martínez-Herrera E. Candida glabrata Antifungal Resistance and Virulence Factors, a Perfect Pathogenic Combination. Pharmaceutics. 2021 Sep 22;13(10):1529.
- Forsberg K, Woodworth K, Walters M, et al. Candida auris: the recent emergence of a multidrug-resistant fungal pathogen. Med Mycol. 2019;57(1):1–12. [CrossRef]
- Dong J, Liang G, Zheng H, et al. In Vitro activity of ravuconazole against Candida auris and vaginal candida isolates. Mycoses. 2021; 64:651–655. [CrossRef]
- Alexander BD, Perfect JR. Antifungal resistance trends towards the year 2000. Implications for therapy and new approaches. Drugs 1997; 54: 657–78. [CrossRef]
- Andes D, Stamsted T, Conklin R. Pharmacodynamics of amphotericin B in a neutropenic-mouse disseminated-candidiasis model. Antimicrob Agents Chemother 2001; 45: 922–26. [CrossRef]
- Louie A, Drusano GL, Banerjee P, et al. Pharmacodynamics of fluconazole in a murine model of systemic candidiasis. Antimicrob Agents Chemother 1998; 42: 1105–09. [CrossRef]
- Polak A. The past, present and future of antimycotic combination therapy. Mycoses 1999; 42: 355–70. [CrossRef]
- Francis P, Walsh TJ. Evolving role of flucytosine in immunocompromised patients: new insights into safety, pharmacokinetics, and antifungal therapy. Clin Infect Dis 1992; 15: 1003–18. [CrossRef]
- Groll AH, Piscitelli SC, Walsh TJ. Clinical pharmacology of systemic antifungal agents: a comprehensive review of agents in clinical use, current investigational compounds, and putative targets for antifungal drug development. Adv Pharmacol 1998; 44: 343–500.
- Sheehan DJ, Hitchcock CA, Sibley CM. Current and emerging azole antifungal agents. Clin Microbiol Rev 1999; 12: 40–79. [CrossRef]
- Neely MN, Ghannoum MA. The exciting future of antifungal therapy. Eur J Clin Microbiol Infect Dis 2000; 19: 897–914.
- Kontoyiannis DP. A clinical perspective for the management of invasive fungal infections: focus on IDSA guidelines. Infectious Diseases Society of America. Pharmacotherapy 2001; 21: 175S–87S. [CrossRef]
- Gupta AK, Venkataraman M, Renaud Hj Summerbell R, et al. The increasing problem of treatment-resistant fungal infections: a call for antifungal stewardship programs. Int J Dermatol. 2021. [CrossRef]
- Chen E, Ghannoum M, Elewski BE. Treatment-resistant tinea corporis, a potential public health issue. Br J Dermatol. 2021;184(1):164–165.
- Kumar S, Kaur A, Kaur S. Autoimplantation therapy in extensive and recalcitrant dermatophytosis: a case series. J Clin Aesthet Dermatol. 2021;14(1):34–37.
- Abastabar M, Hosseini T, Valadan R, et al. Novel point mutations in cyp51A and cyp51B genes associated with itraconazole and posaconazole resistance in Aspergillus clavatus isolates. Microb Drug Resist. 2019;25(5):652–662. [CrossRef]
- Hivary S, Fatahinia M, Halvaeezadeh M, et al. Luliconazole, a new antifungal, vs. amphotericin B, voriconazole, posaconazole and caspofungin against clinical and environmental Aspergillus nigri complex. bioRxiv. 2019.
- Beardsley J, Halliday CL, Chen SC-A, et al. Responding to the emergence of antifungal drug resistance: perspectives from the bench and the bedside. Future Microbiol. 2018; 13:1175–1191. [CrossRef]
- Yamaguchi H. Potential of ravuconazole and its prodrugs as the new oral therapeutics for onychomycosis. Med Mycol J. 2016;57(4): E93– E110. [CrossRef]
- Kano R, Hiruma J, Yokota M, et al. In Vitro ravuconazole susceptibility of anthropophilic dermatophyte strains isolated from Japanese patients. Jpn J Infect Dis. 2020;73(3): 250–252. [CrossRef]
- Mazzantini D, Celandroni F, Calvigioni M, et al. In Vitro resistance and evolution of resistance to tavaborole in Trichophyton rubrum. Antimicrob Agents Chemother. 2021; 65(4): e02324–20. [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




