1. Introduction
Escherichia coli O26 comprises a group of diarrheagenic bacterial strains that exhibit diverse virulence mechanisms. These strains are categorized into enteropathogenic
E. coli (EPEC) and shiga toxin-producingc
E. coli (STEC) [
1]. The range of virulence mechanisms exhibited by these pathogens underlines their significance on public health. For instance, in developing countries, EPEC strains contribute to acute diarrhea and infant mortality, while globally, STEC strains are responsible for outbreaks of bloody diarrhea and hemolytic uremic syndrome (HUS) [2-4].
These bacteria are also considered emerging pathogens due to specific genetic sequences related to virulence present in their genomes. For instance, a pathogenicity island (PAI) almost identical to a PAI present in EPEC O15 was identified in the LEE (Locus of enterocyte effacement) region of O26 EHEC [
5]. Conversely a conserved high pathogenicity island (HPI) associated with enhanced virulence in enterohemorrhagic
E. coli (EHEC) O26 was detected in EPEC O26 strains [
6].
Additionally, in the case of EPEC O26 strains, they are shielded by a polysaccharide pseudo-capsule that shares the same composition as the O26 polysaccharides present in their LPS molecules [
7,
8]. This indicates that the pseudo-capsule in O26 EPEC strains potentially provides protection against the innate immune defenses of the host, as observed in O-antigen capsulated O55
E. coli [
9].
The presence of the capsule can also confer protection to the bacteria against various environmental factors, including antimicrobial molecules and phagocytes [10-12].
Nevertheless, the immune evasion mechanisms mediated by the capsule can be overcome by a humoral immune response that specifically targets the polysaccharides that compose the bacterial capsule or the polysaccharides that compose their LPS O-antigen [
9,
13,
14]. This humoral immune response has been shown to inhibit the adhesion of capsulated and non-capsulated O111 and O55
E. coli strains to epithelial cells and aid in the clearance of these pathogens by macrophages [
9,
14]. However, the efficacy of this response can be influenced by variations found in the ratio of the oligosaccharide units that constitute the O antigen of strains that belong to the same serogroup [
13].
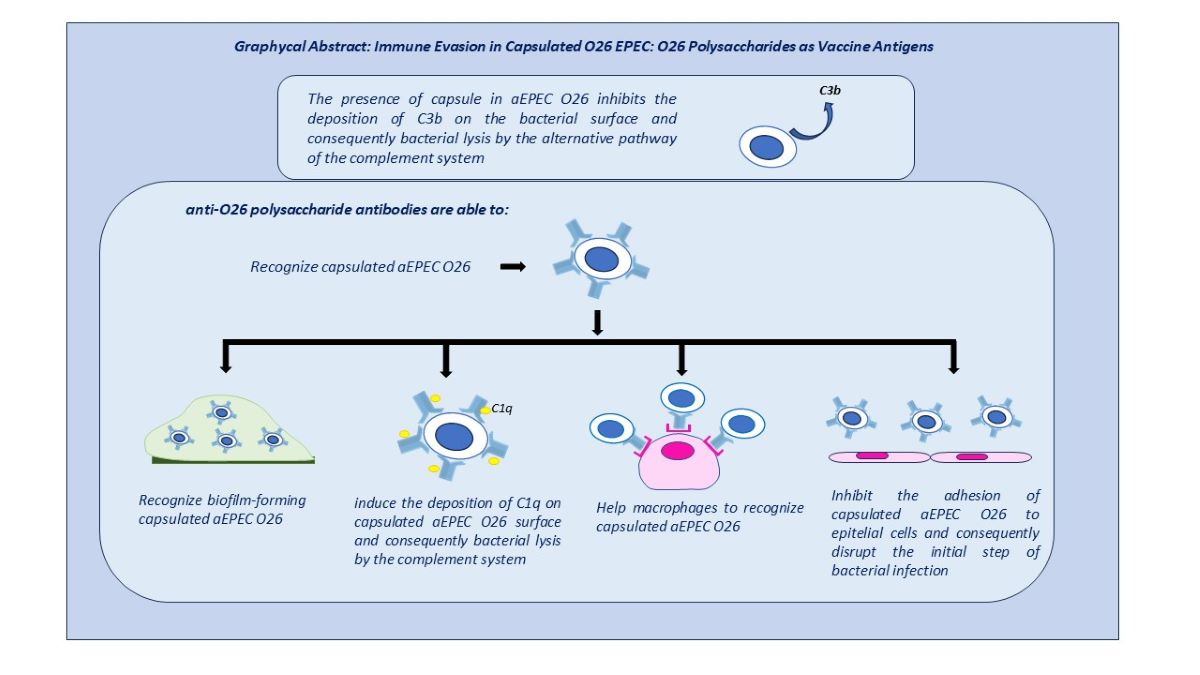
Therefore, considering that the capsule of O26 EPEC is composed of O26 polysaccharides, we investigated the influence of these polysaccharides on the immune evasion of capsulated O26 E. coli and assessed the impact of anti-O26 polysaccharide antibodies in overcoming their protection.
The results obtained indicated that the generation of a humoral response against the O26 polysaccharides can provide protection against capsulated O26 E. coli strains.
2. Results
3.1. Determination of the presence of pseudo-capsules on E. coli strains
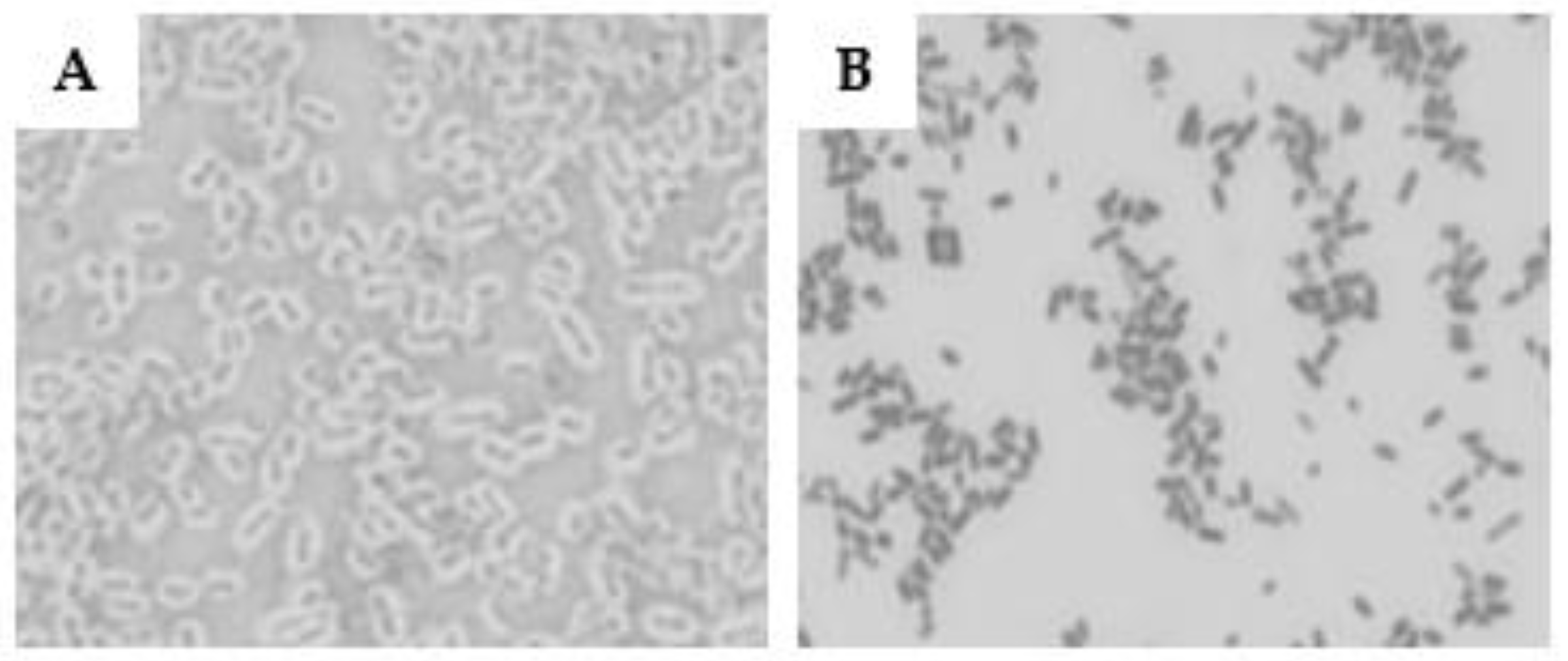
The utilization of the Maneval technique to evaluate the presence of a pseudo-capsule in O26 EPEC and STEC strains revealed that the EPEC strain displayed a thick capsule, while the STEC strain was non-capsulated (
Figure 1)
3.2. Gas chromatography analysis
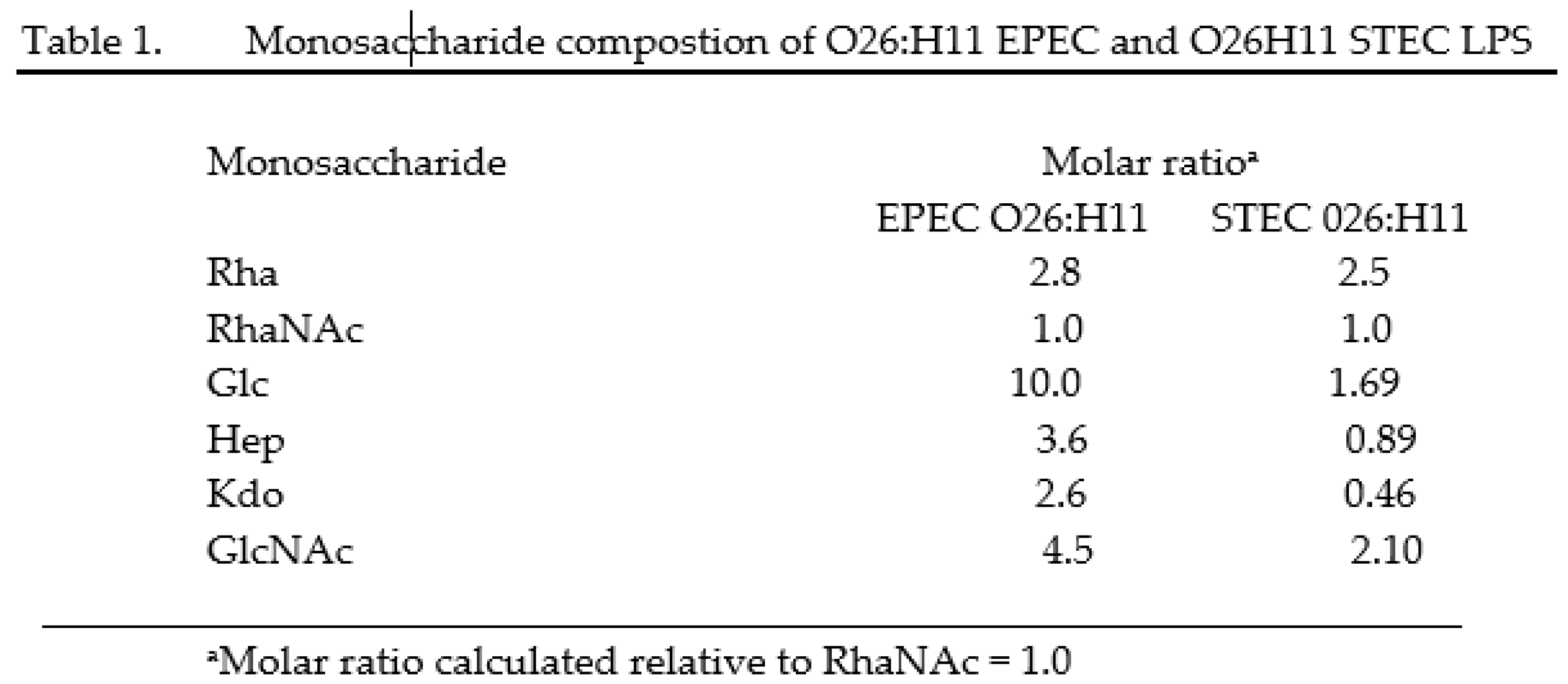
Gas chromatography analysis of the O26 LPS derived from EPEC and STEC has shown that the presence of rhamnose and N-acetyl-rhamnosamine was similar in both EPEC and STEC strains. However, the ratio of glucose, heptose and N-acetylglucosamine was found to be higher in EPEC compared to STEC, with a molar ratio of 10 to 1.69 for glucose (Glc), 3.6 to 0.89 for heptose (Hep), and 4.5 to 2.10 for N-acetylglucosamine (GlcNac) (Table 1).
Rha (Rhamnose); RhaNac (N-acetylrhaminosamine); Glc (Glucose), Hep (Heptose), Kdo (2-keto-3-deoxyoctonate), GlcNAc (N-acetylglucosamine)
3.3. Biofilm formation
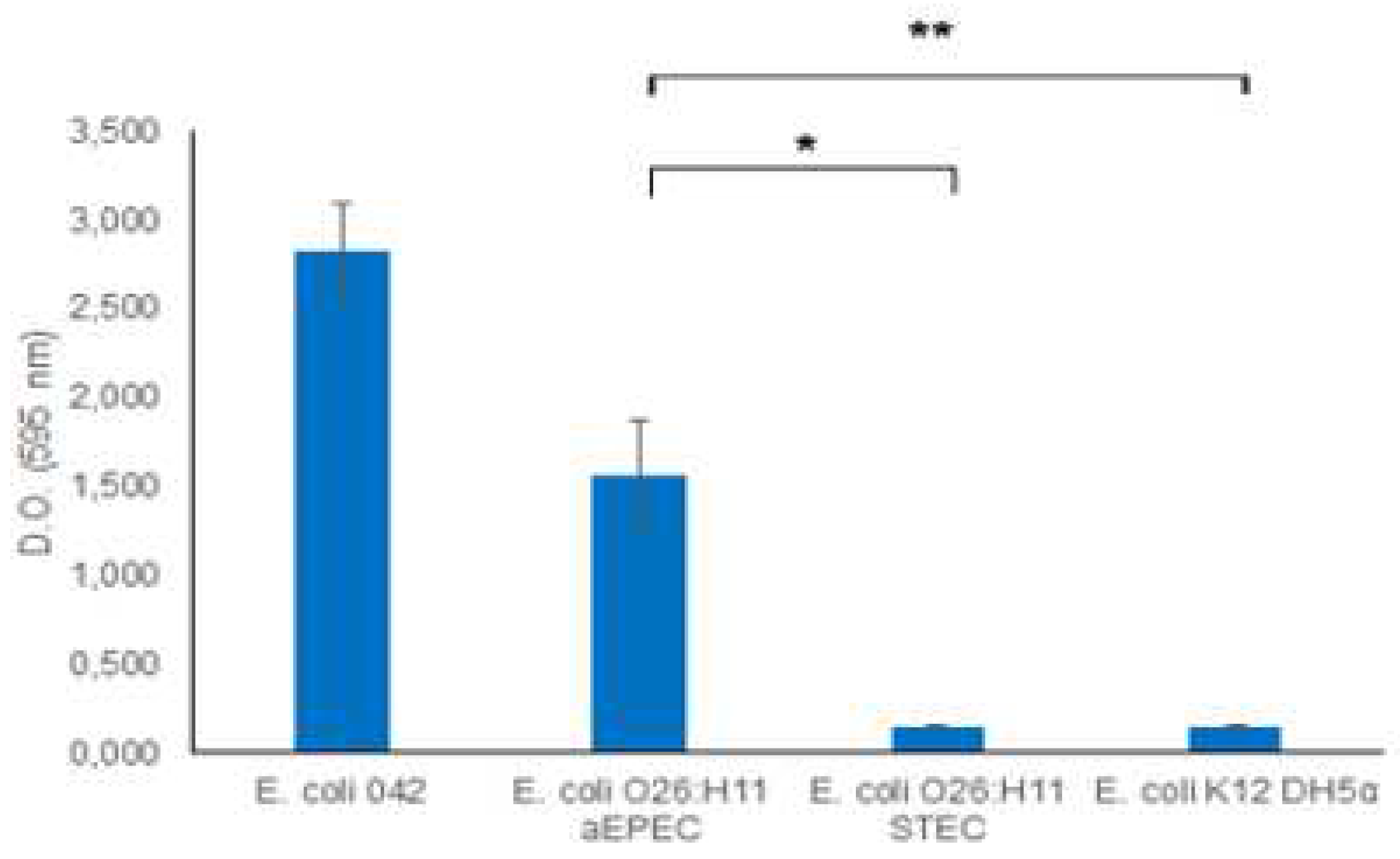
Recognizing the significance of biofilm as a virulence mechanism, we investigated the biofilm-forming capabilities of O26:H11 EPEC and STEC strains. The results demonstrated that the aEPEC strain exhibited a robust capacity to produce biofilm, while the STEC O26:H11 strain was unable to form a biofilm (
Figure 2).
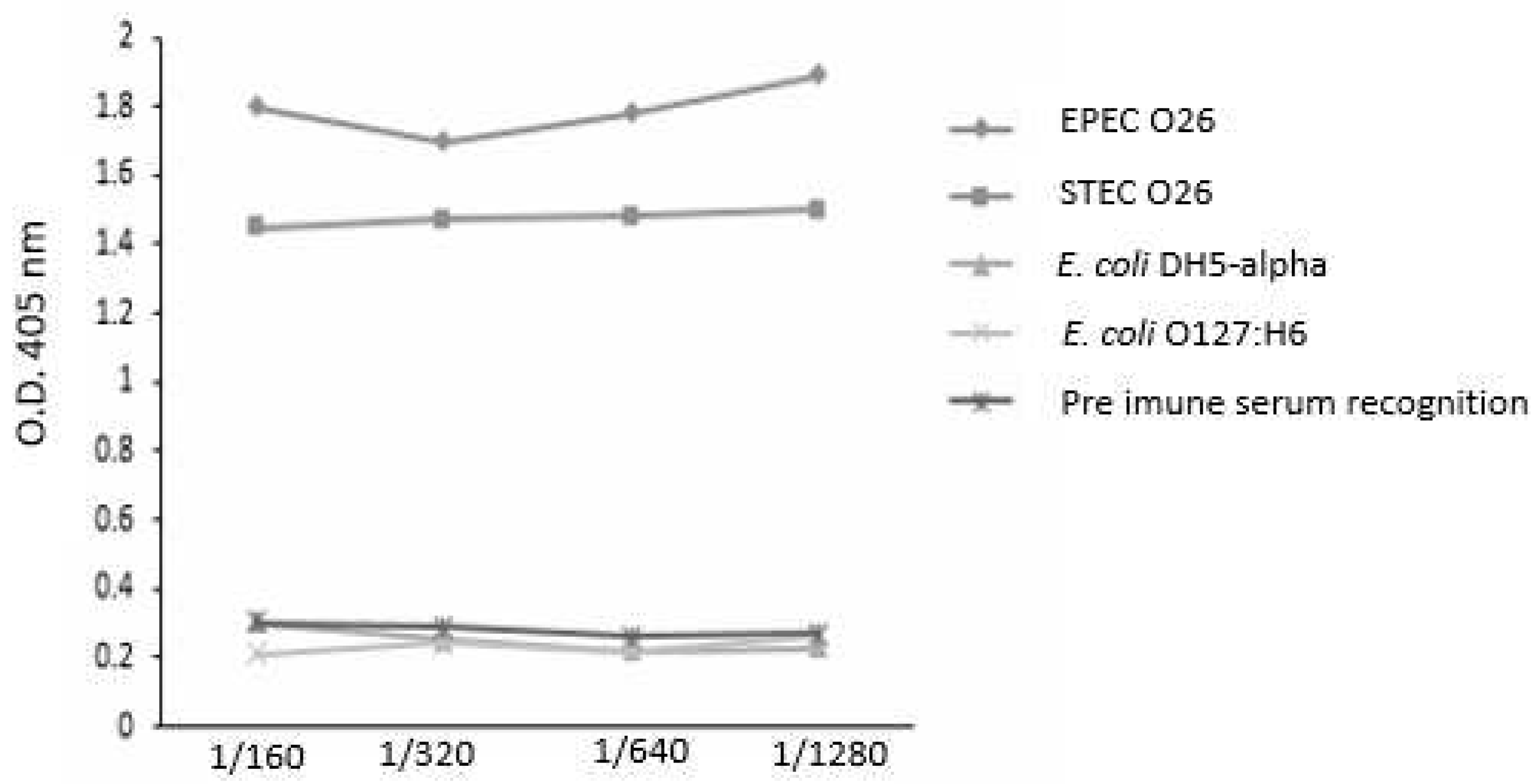
3.4–. Influence of anti-O26 polysaccharide antibodies on E. coli recognition
The capacity of the antibodies against O26 polysaccharides to recognize biofilm-forming and non-biofilm-forming
E. coli strains was analyzed by ELISA. The results demonstrated that the antibodies against O26 polysaccharides were able to recognize both biofilm-forming EPEC O26 and non-biofilm-forming STEC O26. They also did not recognize the heterologous
E. coli serotype O127:H6 and
E. coli DH5-alpha, which lack the O polysaccharide antigen (
Figure 3).
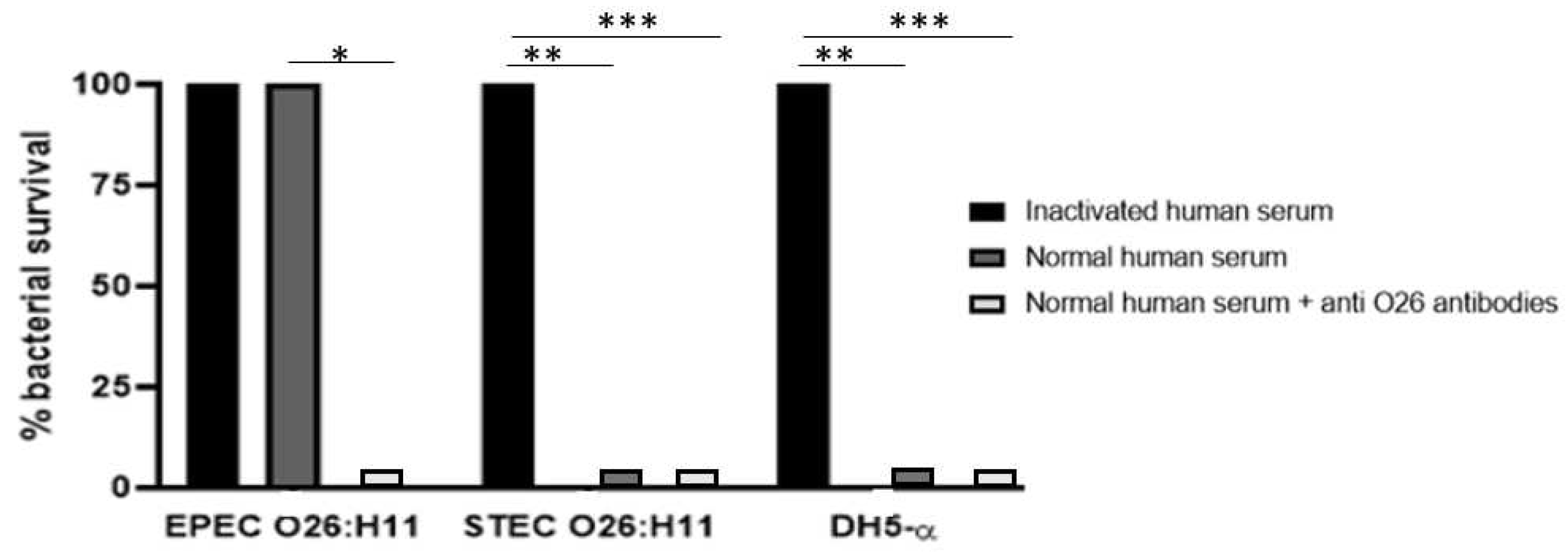
3.5-. Influence of Anti-O26 Antibodies on Complement Response
The ability of the O26 pseudocapsule of aEPEC to protect the pathogen against lysis by the complement system was assessed by incubating the bacteria with normal human serum (NHS) as a source of complement. The results demonstrated that non-capsulated O26:H11 STEC and non-capsulated
E. coli DH5α were lysed in the presence of NHS, however, lysis of the capsulated O26:H11 aEPEC strain was observed only when the pathogen was incubated in the presence of anti-O26 polysaccharide antibodies (
Figure 4).
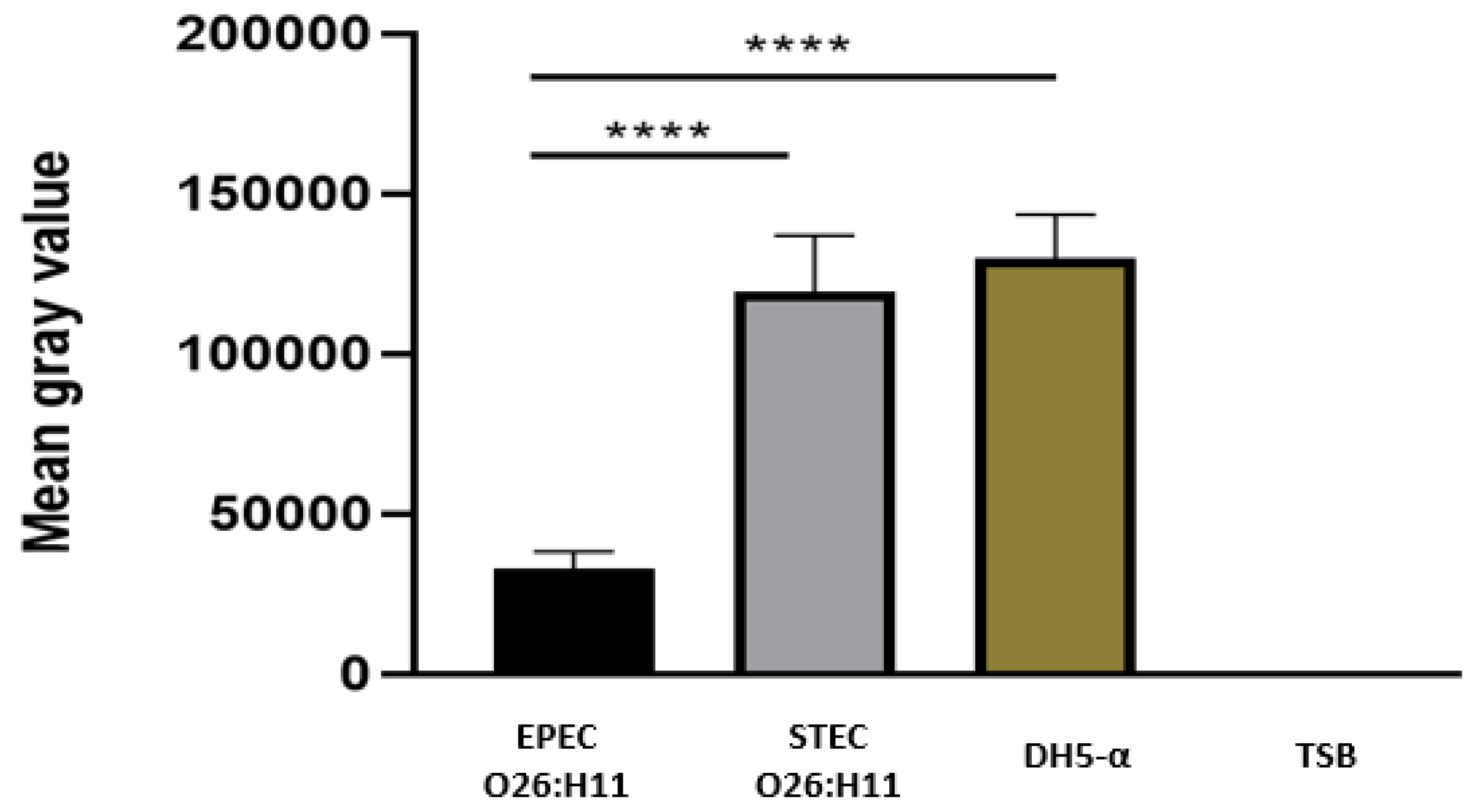
3.6. Deposition of C3b and C1q on the Bacterial Surface
The presence of C3b on capsulated and non-capsulated
E. coli was determine by the Dot Blot technique after bacterial incubation with normal human serum as a source of complement. The results revealed that the presence of a capsule in EPEC O26 impaired the deposition of C3b on the bacterial surface. Conversely, non-capsulated STEC O26:H11 and non-capsulated
E. coli DH5-α strains exhibited C3b deposition on their surfaces (
Figure 5).
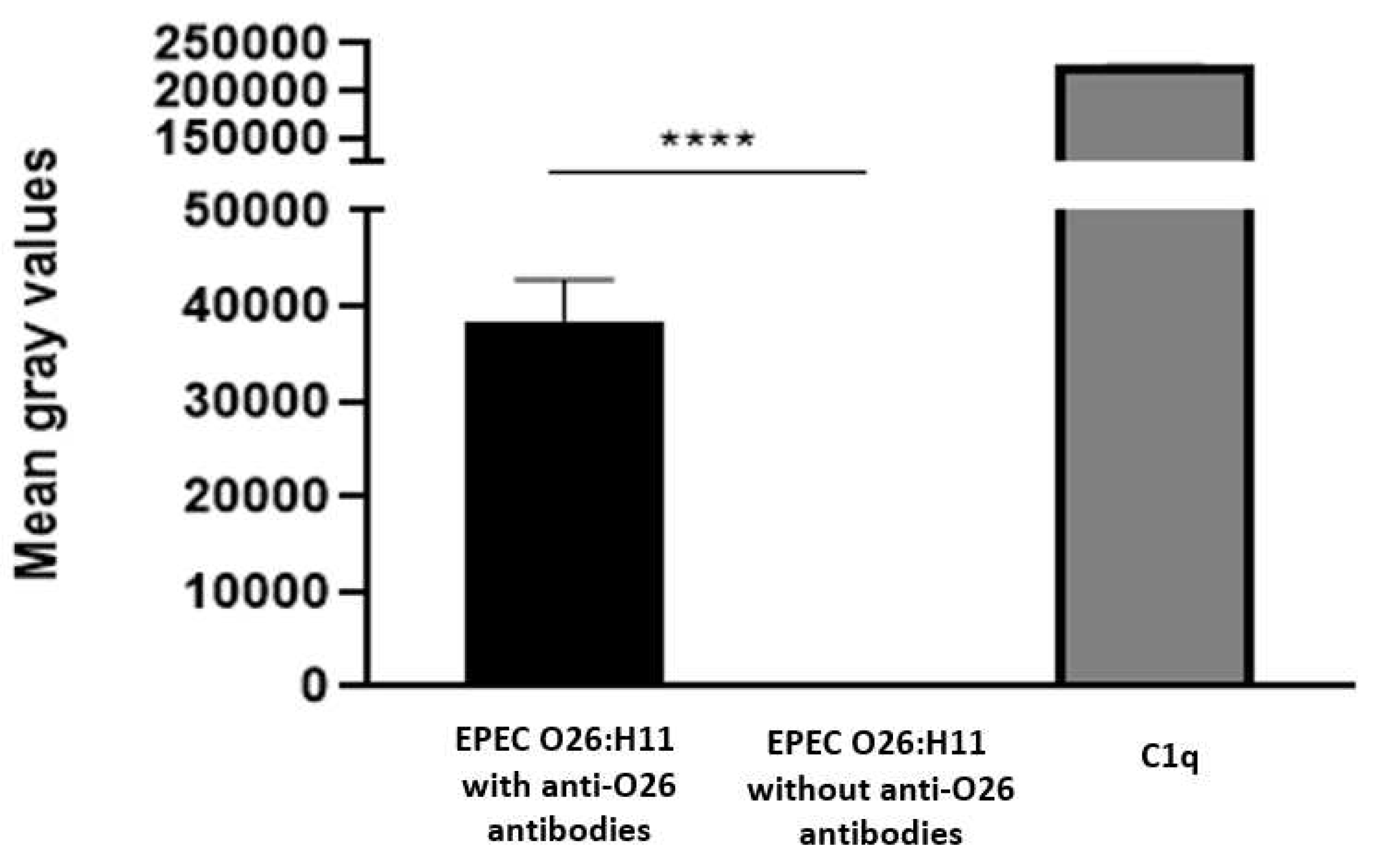
To investigate the influence of anti-O26 polysaccharide antibodies on complement lysis of capsulated EPEC O26, the presence of C1q on antibody opsonized and nonopsonized EPEC was determined using the Dot Blot technique. The results demonstrated that C1q deposition on the capsulated aEPEC O26:H11 surface only occurred when the bacteria were opsonized by anti-O26 polysaccharide antibodies (
Figure 6)
3.7. Influence of Anti-O26 Antibodies in Phagocytosis and Bacterial Adhesion
Taking into consideration that the presence of capsule can impair bacterial recognition by phagocytes [
12], the influence of anti-O26 polysaccharide antibodies in the phagocytosis of capsulated aEPEC O26:H11 was determined in macrophages. The results demonstrated that macrophages were not able to recognize capsulated O26:H11 aEPEC. However, this inability was overcome in the presence of anti-O26 polysaccharide antibodies (
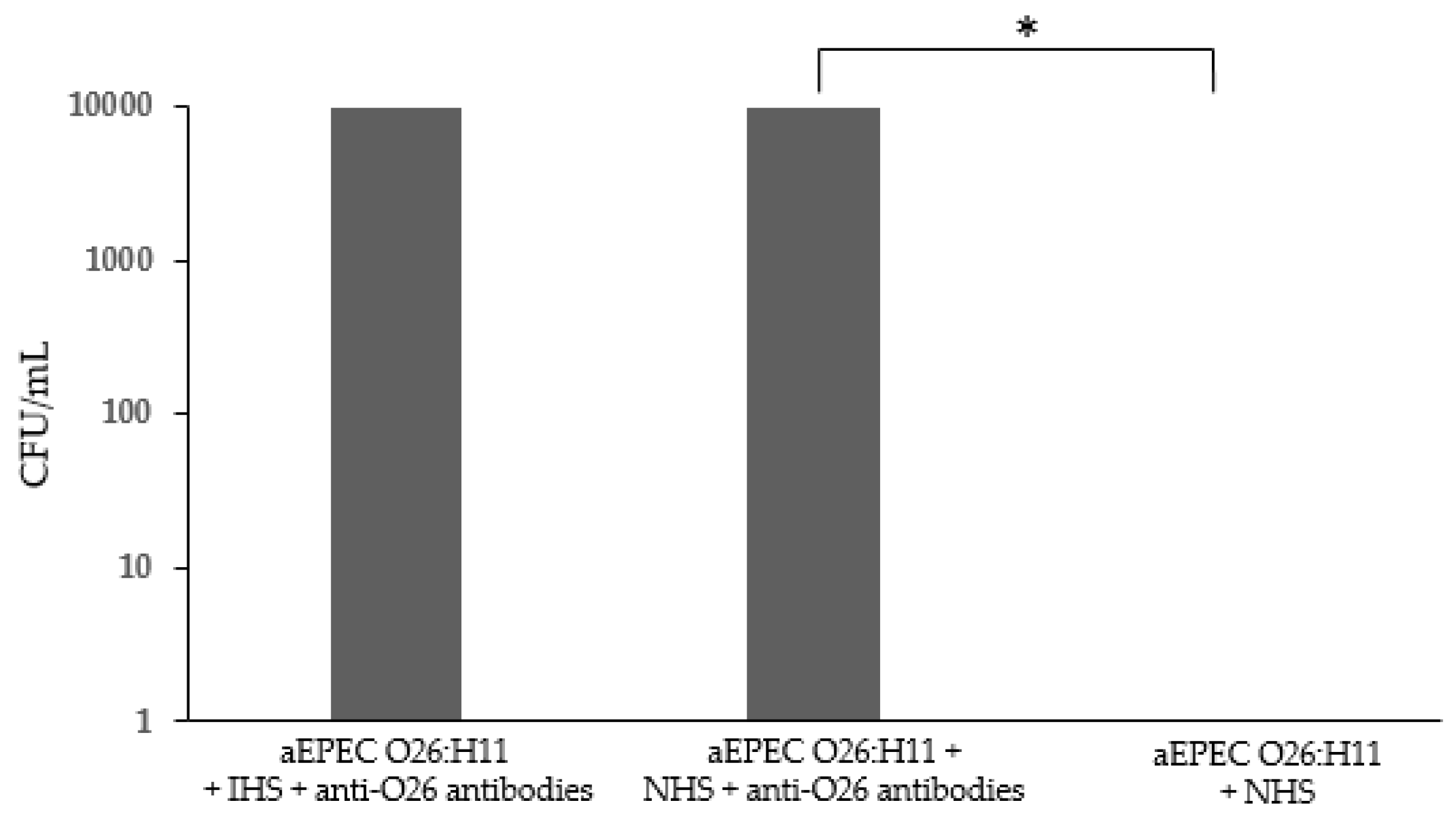
Figure 8)
3.8. Influence of anti-O26 Antibodies on the ahesion of capsulated aEPEC O26:H11 to epithelial cells.
Bearing in mind that the presence of capsule can impede bacterial adhesion to epithelial cells [
15], the influence of the anti-O26 polysaccharide antibodies on the adhesion of capsulated EPEC O26 to epithelial cells was investigated. The results demonstrated that the anti-O26 polysaccharide antibodies can effectively inhibit the adherance of the capsulated aEPEC O26:H11 strain to the epithelial cells (
Figure 8)
3. Discussion
EPEC strains belonging to the serogroup O26 produce a group 4 capsule composed of O26 polysaccharides, which confer upon them an O-antigen capsule status [
8]
However, concerns arise regarding the efficacy of this capsule in shielding the pathogen, given that the O26 polysaccharides contain sequences of N-acetyl glucosamine (GlcNac), which can be recognized by the MBL pathway of the complement system [
16]. Considering that GlcNAC is likely the immunodominant molecule of O26 polysaccharides, as it occurs in Group A Streptococcus [
17], an investigation was conducted to explore potential differences in the composition of O26 polysaccharides between capsulated EPEC and noncapsulated STEC strains.
The results revealed a significant decrease in the amount of GlcNAC present in O26 polysaccharides from EPEC compared to STEC, suggesting that EPEC can evade detection by the MBL pathway of the complement system by reducing the quantity of GlcNAC on their O26 polysaccharides.
However, despite the observed variations in the ratio of the O26 oligosaccharide units between EPEC and STEC, along with differences in their biofilm-forming capabilities, the anti-O26 polysaccharide antibodies were able to equally recognize both pathogens. Conversely, it was noted that the anti-O26 polysaccharide antibodies did not recognize an unrelated serogroup and an E. coli strain devoid of the O antigen, indicating that these antibodies would preferably target E. coli O26 strains without interfering with unrelated serogroups or commensal E. coli strains.
In addition, the anti-O26 polysaccharide antibodies were able to overcome the capsule’s protection against the innate immune system by mediating bacterial complement lysis through the classical pathway and assisting macrophages in recognizing capsulated strains ,which are crutial steps for pathogen clearance [
18]. Finally, by preventing bacterial attachment to the host cells, the anti-O26 polysaccharide antibodies were able to disrupt colonization and, as a consequence, the initial steps of bacterial infection.
Overall, these findings shed light on the complex interactions between O26 E. coli strains, their capsules, complement evasion, and the role of anti-O26 antibodies. They provide valuable insights for the development of vaccines targeting the O26 polysaccharide, which could effectively overcome immune evasion mechanisms and enhance protection against O26 E. coli infections.
Materials and Methods
4.1. Bacterial strains
The strains used in this study were O26:H11 (STEC), O26:H11 atypical-EPEC (a-EPEC), O127:H6 typical-EPEC (t-EPEC) and E.coli DH5α derived from the E. coli collection of the Laboratory of Bacteriology, Instituto Butantan, São Paulo, Brazil.
4.2. Cell lines
The HEp-2 and J774A.1 cell lines used in this study were obtained from the Instituto Adolfo Lutz, São Paulo, Brazil. The cell lines were previously acquired from the American Type Culture Collection (CCL 2). For maintenance, HEp-2 and J774A.1 cells were grown in DMEM (Dulbecco’s Modified Eagle Medium) and RPMI 1640 (Roswell Park Memorial Institute Medium) respectively supplemented with 10% calf serum, 1 mM L-glutamine and 50 IU/mL penicillin-streptomycin.
4.3. Antibodies against O26 polysaccharides
Rabbit serum against O26 polysaccharides was obtained commercially from PROBAC (São Paulo, SP, Brazil).
4.4. Pseudo-capsule visualization
In order to visualize the capsule, it was utilized the Maneval’s method. Briefly, bacterial cells, scraped from a fully grown agar plate, were transferred into a 10 mL drop of 1% aqueous Congo red solution (Sigma-Aldrich, St. Louis, MO, USA). This suspension was spread across a microscopic glass slide to form a thin film, which was air dried. Then, approximately, 10 mL solution of Maneval [3.33% fenol, 4.44% glacial acetic acid, 2.67% ferric chloride, 0.02% acid fuchsin (Sigma-Aldrich)] was distributed across the slide. After 2 minutes incubation the stain was discarded, the slides air-dried and the capsules were visualized in light microscopy (eyepiece, x10; objective, x100). Because the Maneval stain is a negative-stain method, the capsules appear as a transparent halo around the bacteria.
4.5. Extraction and purification of LPS
LPS (20 % of bacterial mass) derived either from O26:H11 STEC or O26:H11 EPEC was extracted using phenol-EDTA-TEA buffer (0.25 M EDTA and 5 % phenol adjusted to pH 6.9 with triethylamine as described by [
19]. The cell suspensions were incubated at 37 °C with constant agitation for 1 h and subsequently centrifuged at 10,000 g for 1 h. After centrifugation, the supernatants were collected and dialyzed in dialysis tubes with pore size of 2,000-molecular-weight (MW) against running water for 3 days and deionized water for 1 day. After dialysis, the samples were concentrated using a rotary evaporator, clarified by centrifugation at 5,000 g for 20 min, and lyophilized. For purification, the LPS extracts were solubilized in distilled water and ultracentrifuged at 105,000 x g for 16 h at 4°C. The pellets containing purified LPS were solubilized in distilled water and lyophilized. Contaminating nucleic acids were removed by treatment with nucleases (RNase and DNase, Sigma).
4.6. Sugar analysis
Monosaccharides from LPS (500 µg) were analyzed as their trimethylsilyl (TMS) methyl-glycosides after methanolysis with 0.5 M HCl in methanol at 80 °C for 18 h. The methanolyzed products were extracted with hexane. The methanolic phase was neutralized by addition of silver carbonate, re-N-acetylated with acetic anhydride (overnight at room temperature in dark), dried under a stream of nitrogen, and trimethylsilylated by addition of bis(trimethylsilyl)-trifluoracetamide (BSTFA)/pyridine (ratio, 1:1) at room temperature for 1 h. The TMS methyl-glycosides were identified by gas-liquid chromatography (GC) and GC-mass spectrometry (GC-MS) on DB-1 fused silica column (30m x 0.25 mm/internal diameter) using a temperature programme from 120
°C to 240
°C at 2
°C/min [
20,
21]. The TMS methyl-glycosides were initially characterized by comparison of their retention times to those of authentic standards and confirmed by GC-MS analysis on a Shimadzu GC 17 A gas chromatograph interfaced with a GC-MS-QP5050 quadruple mass spectrometer.
4.7. Biofilm formation
The capacity of a-EPEC O26:H11 and STEC O26:H11 strains to form biofilms on abiotic surfaces was determined. Briefly, 5 µL of bacterial culture previously grown in TSB (Tryptic Soy Broth) were added in triplicate to 96-well plates containing 200 µL of TSB medium per well. After 18 hours of incubation at 37 °C, the plates were washed three times with PBS. Then, the plate was incubated for 10 minutes at 37 °C with 75 % ethanol (200µL/well). After incubation, the plate were washed again, and subsequently, the bacterial biofilm complex was stained with 200 µL/well of 0.5 % Crystal Violet. After 5 minutes of incubation, the plate was washed again with PBS, and 200 µL/well of 95 % ethanol was added to solubilize the Cristal Violet. The optical density was measured at 595 nm in a microplate reader (Multiskan) [
22]. An optical density above 0.5 was considered positive for biofilm formation.
4.8. The Ability of anti-O26 polysaccharide antibodies to recognize biofilm-forming and non-biofilm-forming E. coli strains.
The ELISA assay was utilized to determine the capacity of the anti-O26 polysaccharide antibodies to recognize biofilm-forming EPEC O26:H11, non-biofilm-forming STEC O26:H11, E. coli O127:H6 and E. coli DH5α. In this experiment, 96-well culture plates were prepared following the procedure outlined in section 4.7 with slight modifications. Briefly, after the bacterial biofilm complex was fixed with 75 % ethanol as described above, the ethanol was discarded, and the plates were subsequently incubated with blocking buffer (3% Molico® milk in PBS) for 18 h at 4°C. Following incubation, the plates were washed three times with washing buffer (PBS + 0.05% Tween 20). Subsequently, rabbit anti-O26 polysaccharide serum, serially diluted (1/2), was added to the plates and incubated for 2 hours at 37°C in incubation buffer (PBS + 1% Molico® milk). After the incubation period, the plates were washed three times and incubated for 2 hours with the conjugate (goat anti-rabbit IgG-alkaline phosphate conjugate) diluted 1/5000 in incubation buffer. Subsequently, the plates were washed again and the p-nitrophenyl phosphate substrate was added for development. The test reading was performed using a microplate reader (Multiskan) at 405 nm.
4.9. Serum Resistance Assay
In order to determine the ability of the complement system to lyse the bacterial samples, 5 µL of aEPEC O26:H11 and STEC O26:H11 (1 × 10
13CFU/mL) were added to a 96 well culture plate. The plate was then incubated for 16 h at 37 °C with 15 µL of heat-inactivated serum (HIS ) or 15 µL of normal human serum (NHS) in the presence or absence of 50 µL of anti-O26 polysaccharide antibodies. Subsequently, the bacterial viability was determined by counting the number of colony-forming units (CFU) according to the methodology described by Baron and coworkers and Beck et al [
23,
24]. NHS was used as a source of complement.
4.10. C3b Deposition on the Bacterial Surface
The deposition of C3b on the surface of EPEC O26:H11 and STEC O26:H11 was assessed using the dot blot technique. Briefly, nitrocellulose membranes (0.42 µM) were coated with 2 µL of bacterial samples (1 × 1013 CFU/mL). Subsequently, the membranes were blocked for 18 h at room temperature with a solution of 3% BSA in PBS. After incubation, the membranes were washed three times with washing buffer (PBS containing 0.05% Tween 20) and incubated for 1 hour at 37°C with 10% normal human serum (NHS) in incubation buffer (1% BSA in PBS). Following that, the membranes were washed and incubated for 1 hour with goat anti-C3 antibodies (Complement Technology, Tyler, TX, USA) diluted 1:5000 in incubation buffer. After incubation, the membranes were washed again and incubated for 1 hour with rabbit anti-goat IgG peroxidase conjugate (Sigma-Aldrich) diluted 1:10,000 in incubation buffer. The membranes were then washed, and the deposition of C3b on the bacterial surface was detected by chemiluminescence using SuperSignal West Pico Enhanced Chemiluminescent Substrate (Pierce Biotechnology, Inc.—Thermo Fisher Scientific, Waltham, MA, USA). The intensity of the signals was determined in pixels using Image J software developed at the National Institutes of Health and the laboratory for Optical and Computational Institutes (LOCI, University of Wisconsin, USA) Version 1.5., and the results were presented as "Mean Gray Values" (average intensity units in selection).
4.11. C1q Deposition on the Bacterial Surface
The deposition of C1q on the surface of EPEC O26:H11 was also assessed using the dot blot technique. Nitrocellulose membranes (0.42 µM) were coated in triplicate with 2 µL of EPEC O26:H11 culture (1 × 1013 CFU/mL) previously incubated for one hour at 37°C in the presence or absence of anti-O26 polysaccharide antibodies diluted 1/10 in PBS. As a positive control, 2 µL of C1q (125 ng/µL) were coated on the membrane. The membrane was blocked and washed as decribed above, and subsequently, incubated for 1 hour with goat IgG anti-C1q (Complement Technology,Tyler, TX, USA) diluted 1:5000 in incubation buffer. After washing, the membranes were incubated for 1 hour at room temperature with rabbit anti-goat IgG conjugated with peroxidase (Sigma-Aldrich) diluted 1:10,000 in incubation buffer. Following another round of washing, the deposition of C1q on the bacterial surface was detected and quantified as described above.
4.12. Phagocytosis.
J774A.1 macrophages were seeded in 24-well cell culture plates at a concentration of 10
5 cells/mL in DMEM supplemented with 10% fetal bovine serum (1 mL/well). The plates were then incubated for 48 hours at 37°C in a 5% CO
2 incubator. In parallel, bacterial samples (40 µL) containing 10
7 cells/mL were incubated for 1 hour at 37°C with anti-O26 polysaccharide antibodies diluted 1/10 in 1 mL of DMEM without antibiotics, supplemented with either 10% normal human serum (NHS) or inactivated human serum (HIS). After incubation, the samples were added in triplicate to the macrophage cells and further incubated for 3 hours at 37°C in a 5% CO
2 incubator. Following incubation, the cells were washed 6 times with sterile PBS and then treated with 50 µg/mL of gentamicin for 30 minutes. Subsequently, the plates were washed 6 times with PBS, and the macrophages were lysed by incubating the cells with Triton X-100 (Merck) diluted 1/10 in PBS (500µL/well) for 10 minutes at room temperature. After incubation, 500 µL of PBS were added to each well to resuspend the lysate. Next, 100 µL of each lysate were serially diluted 10-fold in saline, starting with a dilution of 1 in 10. One hundred microliters of each dilution were then plated in triplicate on LB (Luria Broth) agar plates. The plates were incubated overnight at 37°C, and the number of colony-forming units (CFUs) was determined [
23,
24].
4.13. Inhibition of bacterial adhesion to epithelial cells
HEp-2 cells were grown to 70% confluence on circular coverslips in wells of 24-well tissue culture plates in the presence of DMEM without antibiotics. Forty microliters of bacterial culture (EPEC O26:H11) at a concentration of 107/mL previously incubated for 1 h at 37 °C with anti-O26 polysaccharide antibodies diluted 1/10 in DMEM containing 2% fetal bovine serum were added in triplicate to the wells (1 mL/well) and incubated for 3 h at 37 °C in 5% CO2. As a positive control for bacterial adhesion, the cells were incubated only with bacteria in the absence of antibodies. After incubation, the monolayers were washed 6 times with sterile PBS and then fixed with 100% methanol for 10 min, stained for 5 min with May–Grunwald stain diluted 1:2 in Sorensen buffer, and finally stained for 20 min with Giemsa stain diluted 1:3 in Sorensen buffer. The excess stain was discarded, and the coverslips with the stained cells were affixed to microscope slides for visualization by light microscopy (eyepiece, ×10; objective, ×100).
4.14. Statistical Analysis
Statistical analysis was conducted using GraphPad Prism version 8.0.2, GraphPad Software, San Diego, CA, USA) with the unpaired t-test. A p-value ≤ 0.05 (*) was considered statistically significant.
5. Conclusions
These findings strongly support the notion that O26 polysaccharides are promising antigen targets for developing a vaccine against capsulated E. coli O26 strains. By specifically targeting their O26 polysaccharides, such a vaccine has the potential to elicit a humoral immune response capable of neutralizing immune evasion, promoting bacterial clearance, and preventing initial adhesion, thereby offering protection against infections caused by these pathogenic strains
Author Contributions
T.J.L., H.G.S.S., J.O.P., E.O.F., M.R.F., B.S.M., G.F.S., R.H.N.G. were responsible for the methodology; L.F.S. was responsible for serotyping the bacterial strains; A.S.B. was responsible for the complement analysis; J.O.P, L.M.P. were responsible for conceptualization and review, M.O.D. was responsible for conceptualization, coordination, writing and editing the manuscript. All authors have read and agreed to the published version of the manuscript.
Funding
FAPESP: Grant number: 2012/11325-5.
Data Availability Statement
The data presented in this study are available on request from the corresponding author. The data are not available in the repository of the Butantan Institute (
https://repositorio. butantan.gov.br (accessed on 30 November 2023).
Conflicts of Interest
The authors declare no conflict of interest.
References
- Piazza R.M.F., Delannoy S., Fach P., Saridakis H.O., Pedroso M.Z., Rocha L.B., Gomes T.A.T., Vieira M.A.M., Beutin L., Guth B.E.C. Diarrheagenic E. coli O26 Strains Isolated in Brazil. Journal ASM. 2013. 79 (22): 6847-6854. Doi: 10.1128/AEM.01693-13. [CrossRef]
- Multistate Outbreaks of Shiga toxin-producing Escherichia coli O26 Infections Linked to chipote Mexican Grill Restaurants (final update). E. coli (Escherichia coli). Centers for Disease Control and Prevention. CDC – www.cdc.gov/ecoli/2015/o26-11-15. Posted: February 1, 2016. Accessed: March 2016.
- Outbreak of E. coli Infections Linked to Ground Beef. Centers for Disease. Control and Prevention. CDC. https://www.cdc.gov/ecoli/2018/o26-09-18/index.html#print. Last Reviewed: September, 20.2018. Accessed: December, 01.2022. Last Reviewed: September, 20.2018. Acessed: December, 01.2022.
- Minary K., Tanne C., Kwon T., Faudeux C., Clave S., Longevin L., Pietrment C., Enoch C., Parmentier C., Mariani-Kikdjian P., Well F.X., Jones G., Djouadi N., Morin D. Outbreak of hemolytic uremic syndrome with unusually severe clinical presentation caused by Shiga toxin-producing Escherichia coli O26:H11 in France. Archives de Pediatrie. 2022, 29:448-452. Doi: 10.1016/j.arcped.20. [CrossRef]
- Muniesa A., Schembri M.A., Hauf N., Chakraborty T. Active genetic elements present in the locus of enterocyte effacement in Escherichia coli O26 and their role in mobility. Infect. Immun. 2006, 74: 4190-4122. [CrossRef]
- Bielaszewska A., Sonntag A. Schmidt M.A. Karch H.. Presence of virulence and fitness gene modules of enterohemorrhagic Escherichia coli in atypical enteropathogenic Escherichia coli 026. Microbes and Infection. 2007, (9): 891-897. [CrossRef]
- Manca M.C.,Weintraub A., Widmalm G. Structural studies of the Escherichia coli O26 O-antigen polysaccharide. Carbohydrate Research. 1996, 281:155-160. [CrossRef]
- WHITFIELD, C. Biosynthesis and Assembly of Capsular Polysaccharides in Escherichia coli. Rev. Biochem. 2006, 75: 39-69. Doi: 10.1146/annurev.biochem.75.103004.142545. [CrossRef]
- Silva H.G.S., Franzolin M.R., Anjos G.F., Barbosa A.S., Santos L.F., Miranda K.F., Marques R.M., Souza M.C., Piazza R.M.F., Domingos M.O. O55 Polysaccharides Are Good Antigen Targets for the Formulation of Vaccines against O55 STEC and Capsulated aEPEC Strains. Pathogens. 2022, 11, 895: 1-11. Doi: 10.3390/pathogens11080895. [CrossRef]
- Campos M.A., Vargas M.A., Regueiro V., Hompart C.M., Alberti S., Bengoechea J.A. Capsule polysaccharide mediates bacterial resistance to antimicrobial peptides. Infect Immun. 2004, 72, 12: 7107-7114. Doi: 10.1128/IAI.72.12.7107-7114.2004. [CrossRef]
- Wen Z., Zang J.R. Bacterial capsules. Chapter 3. Molecular Medical Microbiology. 2015, 1: 33-35. Elsevier, Second edition. Doi: 10.1016/B978-0-12-39719-200003-2. [CrossRef]
- Kafmann S., Dorhoi A. Molecular determinants in phagocyte-bacteria interactions. Immunity. 2016, 44:476-491. Doi: 10.1016/j.immunji.2016.02.014. [CrossRef]
- Santos, M.F.; New, R.R.C.; Andrade, G.R.; Ozaki, C.Y.; Sant’Anna, O.A.; Mendonça-Previato, L.; Trabulsi, L.R.; Domingos, M.O. Lipopolysaccharide as an antigen Target for the Formulation of a Universal Vaccine against Escherichia coli O111 strains. Clin. Vaccine Immul. 2010, 11, 1172–1780. Doi: 10.1128/CVI.0032-10. [CrossRef]
- Andrade, G.R.; New, R.R.C.; Sant’Anna, O.A.; Williams, N.A.; Alves, R.C.B.; Pimenta, D.C.; Viferelli, H.; Melo, B.S.; Rocha, L.B.; Piazza, R.M.F.; et al. A universal polysaccharide conjugated vaccine against O111 E. coli. Hum. Vaccines Immunother. 2014, 10, 2864–2874. Doi: 10.4161/21645515.2014.972145. [CrossRef]
- Sahly H., Podschun R., Oelschlaeger T.A., Greiwe M., Parolis H., Hasty D., Kekow J., Ullmann U., Ofek I., Sela S. Capsule impedes adhesion to and invasion of epithelial cells by Klebsiella pneumoniae. Infection and Immunity. 2000, 68 (12): 6744-6749. [CrossRef]
- Matsushita M. Ficolins: Complement-Activating Lectins Involved in Innate Immunity. J. Innate Immun. 2010, 2: 4-32. Doi: 10.1159/000228160. [CrossRef]
- Sorge N.M., Cole J.N., Kuipers K., Henningham A., Aziz R.K., Kasirer-Friede A., Lin L et al. The classical Lancefield antigen of group A Straptococcus is a virulence determinant with implications for vaccine Design. Cell Host & Microbe. 2014.,15: 729=740. Doi: 10.1016/j.chom.2014.05.009. [CrossRef]
- Bournazos S., Gupta A., Raveth J.V. The role of IgG Fc receptors in antibody-dependent enhancement. Nature Reviews/Immunology. 2020. (20): 633-643. Doi: 10.1038/s41577-020-00410-0. [CrossRef]
- Ridley BL, Jeyaretnam BS, Carlson RW. The type and yield of lipopolysaccharide from symbiotically deficient Rhizobium lipopolysaccharide mutants vary depending on the extraction method. Glycobiology. 10:1013-1023, 2000. [CrossRef]
- Lopes Alves L, Travassos LR, Previato JO, Mendonça-Previato L. Novel antigenic determinants from peptidorhamonomannans of Sporothrix schenckii. Glycobiology. 1994, 4:281-288. [CrossRef]
- Heise N, Gutierrez AL, Mattos KA, Jones C, Wait R, Previato JO, Mendonça-Previato L. Molecular analysis of a novel family of complex glycoinositolphosphoryl ceramides from Cryptococcus neoformans: structural differences between encapsulated and acapsular yeast forms. Glycobiology. 2002, 12:409-420. [CrossRef]
- Sheikh J., Hicks S., Dall’Agnol M., Phillips A.D. and NataroJ.P . Roles for Fis and YafK in biofilm formation by enteroaggregative Escherichia coli. Molecular Microbiology. 2001, 41(5), 983–99. [CrossRef]
- Baron, F.; Cochet, M.F.; Ablain, W.; Grosset, N.; Madec, M.N.; Gonnet, F.; Jan, S.; Gautier, M. Rapid and cost-effective method for micro-organism enumeration based on miniaturization of conventional plate-couting technique. Lait INRA Ed. 2006, 86, 251–257. [CrossRef]
- Beck, N.K.; Callahan, K.; Nappier, S.P.; Kim, H.; Sobsey, M.D.; Meschke, J.S. Development of a Spot-Titer Culture assay for Quantifying bacteria and viral. J. Rapid Methods Autom. Microbiol. 2009, 17, 455–464. [CrossRef]
Figure 1.
Capsulated and non-capsulated O26 E. coli. The presence of a capsule in aEPEC O26:H11 (A) and STEC O26:H11 (B) was determined using the Maneval’s technique.
Figure 1.
Capsulated and non-capsulated O26 E. coli. The presence of a capsule in aEPEC O26:H11 (A) and STEC O26:H11 (B) was determined using the Maneval’s technique.
Figure 2.
– Biofilm formation. The capacity of O26:H11 aEPEC and O26:H11 STEC strains to form biofilm was evaluated using the Crystal Violet Assay and the production of biofilm was measured by determining the optical density at 595 nm. E. coli O42 was used as a positive control for biofilm production, and E. coli K12 DH5α was used as a negative control. An optical density above 0.5 was considered indicative of biofilme formation. Unpaired t-test: */** (p-value ≤ 0.05) was considered significant.
Figure 2.
– Biofilm formation. The capacity of O26:H11 aEPEC and O26:H11 STEC strains to form biofilm was evaluated using the Crystal Violet Assay and the production of biofilm was measured by determining the optical density at 595 nm. E. coli O42 was used as a positive control for biofilm production, and E. coli K12 DH5α was used as a negative control. An optical density above 0.5 was considered indicative of biofilme formation. Unpaired t-test: */** (p-value ≤ 0.05) was considered significant.
Figure 3.
– Recognition of E. coli strains in the presence or absence of biofilm by anti-O26 polysaccharide antibodies. Cell culture plates with 24 wells were incubated with live biofilm-forming capsulated O26:H11 EPEC and the non-biofilm forming E. coli strains (STEC O26:H11, E. coli O127:H6 and E. coli DH5α) for 18 hours at 37°C. Subsequently, the adhered bacteria in the presence or absence of biofilm were fixed using methanol. The ELISA technique was then employed to assess the ability of anti-O26 antibodies to recognize the bacterial samples.
Figure 3.
– Recognition of E. coli strains in the presence or absence of biofilm by anti-O26 polysaccharide antibodies. Cell culture plates with 24 wells were incubated with live biofilm-forming capsulated O26:H11 EPEC and the non-biofilm forming E. coli strains (STEC O26:H11, E. coli O127:H6 and E. coli DH5α) for 18 hours at 37°C. Subsequently, the adhered bacteria in the presence or absence of biofilm were fixed using methanol. The ELISA technique was then employed to assess the ability of anti-O26 antibodies to recognize the bacterial samples.
Figure 4.
- Influence of the anti-O26 polysaccharide antibodies on bacterial lysis by complement. Capsulated aEPEC O26:H11, non-capsulated STEC O26:H11 and non-capsulated E. coli DH5-α were incubated with inactivated normal human serum, normal human serum (as a complement source), or normal human serum with anti-O26 polysaccharide antibodies. After a 24-hour incubation period, bacterial viability was determined by counting the number of colony-forming units (CFUs). Unpaired t-test: */**/**** (p value ≤ 0.05) was considered significant.
Figure 4.
- Influence of the anti-O26 polysaccharide antibodies on bacterial lysis by complement. Capsulated aEPEC O26:H11, non-capsulated STEC O26:H11 and non-capsulated E. coli DH5-α were incubated with inactivated normal human serum, normal human serum (as a complement source), or normal human serum with anti-O26 polysaccharide antibodies. After a 24-hour incubation period, bacterial viability was determined by counting the number of colony-forming units (CFUs). Unpaired t-test: */**/**** (p value ≤ 0.05) was considered significant.
Figure 5.
- Determination of C3b deposition on O26 E. coli surface. The binding of C3b on bacterial surface was determined by the dot blot technique. Nitrocellulose membranes coated with aEPEC O26:H11, STEC O26:H11 and E. coli DH5-α were incubated with normal human serum as a source of complement. The deposition of C3b on the bacterial surface was detected by chemiluminescence (SuperSignalDensitometric) analyses, and the intensity of the signal was determined in pixels by Image J software. The results were plotted as “Mean Gray Values” (average of intensity units in selection). Unpaired t-test: **** (p value ≤ 0.05) was considered significant.
Figure 5.
- Determination of C3b deposition on O26 E. coli surface. The binding of C3b on bacterial surface was determined by the dot blot technique. Nitrocellulose membranes coated with aEPEC O26:H11, STEC O26:H11 and E. coli DH5-α were incubated with normal human serum as a source of complement. The deposition of C3b on the bacterial surface was detected by chemiluminescence (SuperSignalDensitometric) analyses, and the intensity of the signal was determined in pixels by Image J software. The results were plotted as “Mean Gray Values” (average of intensity units in selection). Unpaired t-test: **** (p value ≤ 0.05) was considered significant.
Figure 6.
- C1q Deposition on capsulated EPEC O26:H11 surface. The binding of C1q on bacterial surface was determined by the dot blot technique. Nitrocellulose membrane was coated with capsulated EPEC O26:H11, which was previously incubated for 1 hour at 37°C in the presence or absence of anti O26 polysaccharide antibodies. A positive control was established by coating the membrane with 125 ng of C1q. The deposition of C1q on the bacterial surface was detected by chemiluminescence analysis (SuperSignal Densitometric), and the signal intensity was quantified in pixels using Image J software. The results were presented as "Mean Gray Values," (average of intensity units in selection). Unpaired t-test: **** (p value ≤ 0.05) was considered significant.
Figure 6.
- C1q Deposition on capsulated EPEC O26:H11 surface. The binding of C1q on bacterial surface was determined by the dot blot technique. Nitrocellulose membrane was coated with capsulated EPEC O26:H11, which was previously incubated for 1 hour at 37°C in the presence or absence of anti O26 polysaccharide antibodies. A positive control was established by coating the membrane with 125 ng of C1q. The deposition of C1q on the bacterial surface was detected by chemiluminescence analysis (SuperSignal Densitometric), and the signal intensity was quantified in pixels using Image J software. The results were presented as "Mean Gray Values," (average of intensity units in selection). Unpaired t-test: **** (p value ≤ 0.05) was considered significant.
Figure 7.
Influence of anti-O26 antibodies on bacterial phagocytosis by macrophages. Macrophages were incubated with either capsulated aEPEC in the presence or absence of anti-O26 polysaccharide antibodies. Normal human serum (NHS) was used as a source of complement, and inactivated human serum (IHS) was used as a source of inactivated complement. After incubation, the phagocytes were lysed, and the amount of ingested bacteria was determined by counting the number of colony-forming units (CFU). Unpaired t-test: * (p-value ≤ 0.05) was considered statistically significant.
Figure 7.
Influence of anti-O26 antibodies on bacterial phagocytosis by macrophages. Macrophages were incubated with either capsulated aEPEC in the presence or absence of anti-O26 polysaccharide antibodies. Normal human serum (NHS) was used as a source of complement, and inactivated human serum (IHS) was used as a source of inactivated complement. After incubation, the phagocytes were lysed, and the amount of ingested bacteria was determined by counting the number of colony-forming units (CFU). Unpaired t-test: * (p-value ≤ 0.05) was considered statistically significant.
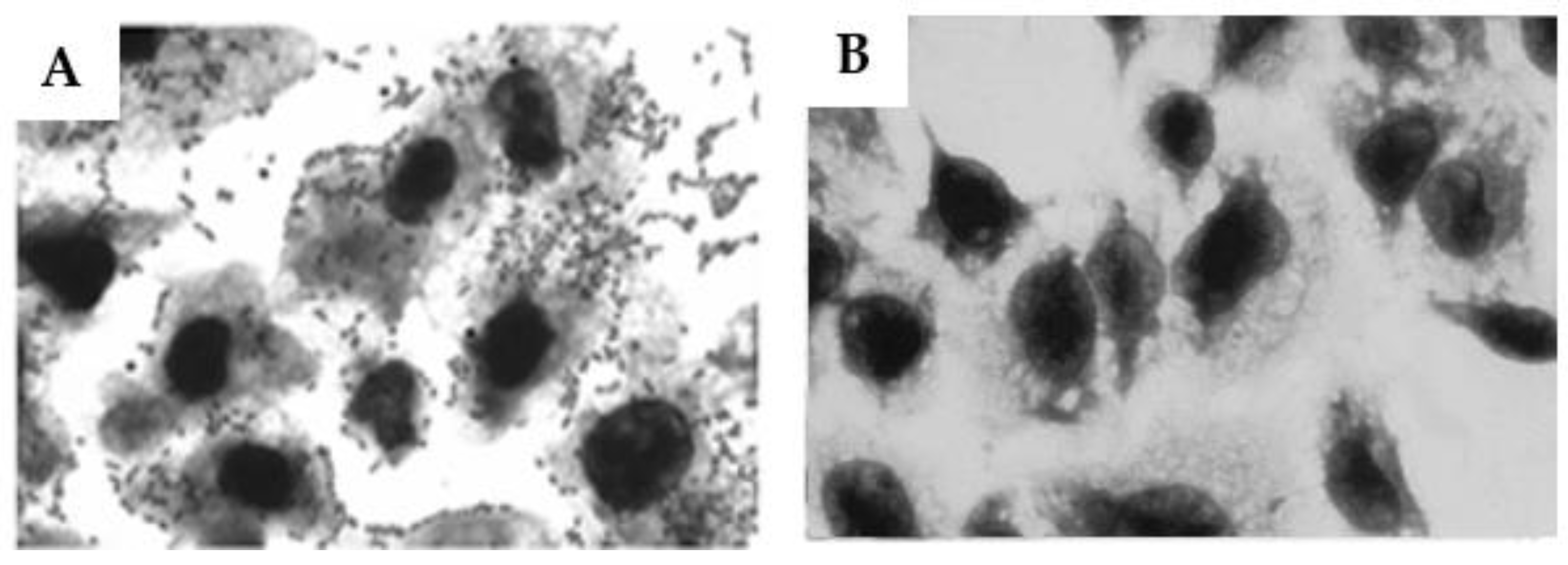
Figure 8.
– Determination of the capacity of the anti-O26 polysaccharide antibodies to inhibit the adhesion of O26:H11 EPEC to human epithelial cells. HEp-2 cells were incubated for 3 h with O26:H11 EPEC bacterial culture either alone (A), or in the presence of anti-O26 polysaccharide antibodies (B). After incubation the cells were washed and stained for visualization (Ocular x10, Objective x100).
Figure 8.
– Determination of the capacity of the anti-O26 polysaccharide antibodies to inhibit the adhesion of O26:H11 EPEC to human epithelial cells. HEp-2 cells were incubated for 3 h with O26:H11 EPEC bacterial culture either alone (A), or in the presence of anti-O26 polysaccharide antibodies (B). After incubation the cells were washed and stained for visualization (Ocular x10, Objective x100).
|
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).