Submitted:
16 December 2023
Posted:
18 December 2023
You are already at the latest version
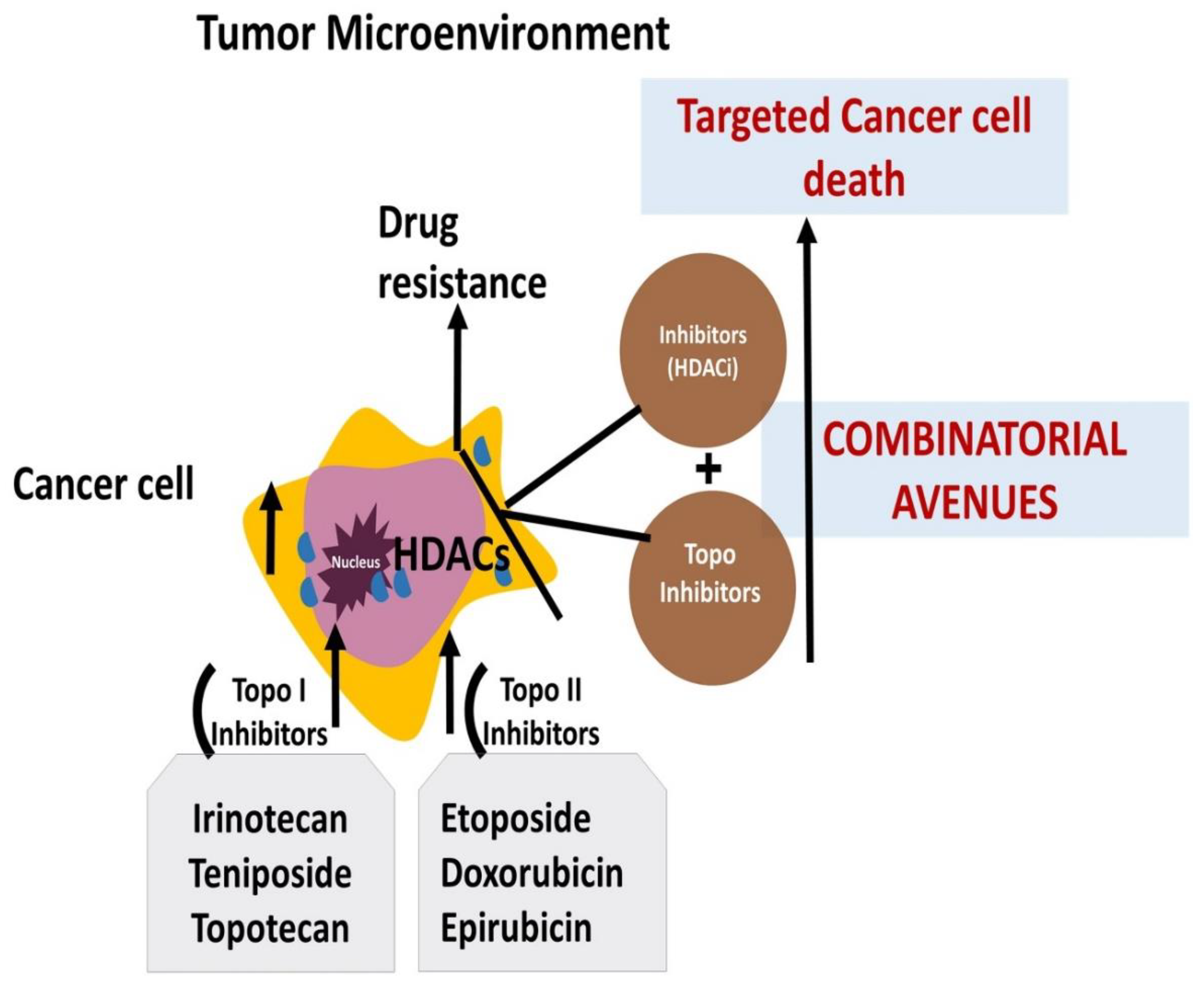
Abstract
Keywords:
1. Introduction
2. Topoisomerases
3. Mechanisms of action of Topo-active drugs
4. Tumor Heterogeneity and Topo active drugs
5. Mutation of target enzymes
6. Altered drug metabolisms and topo active drugs
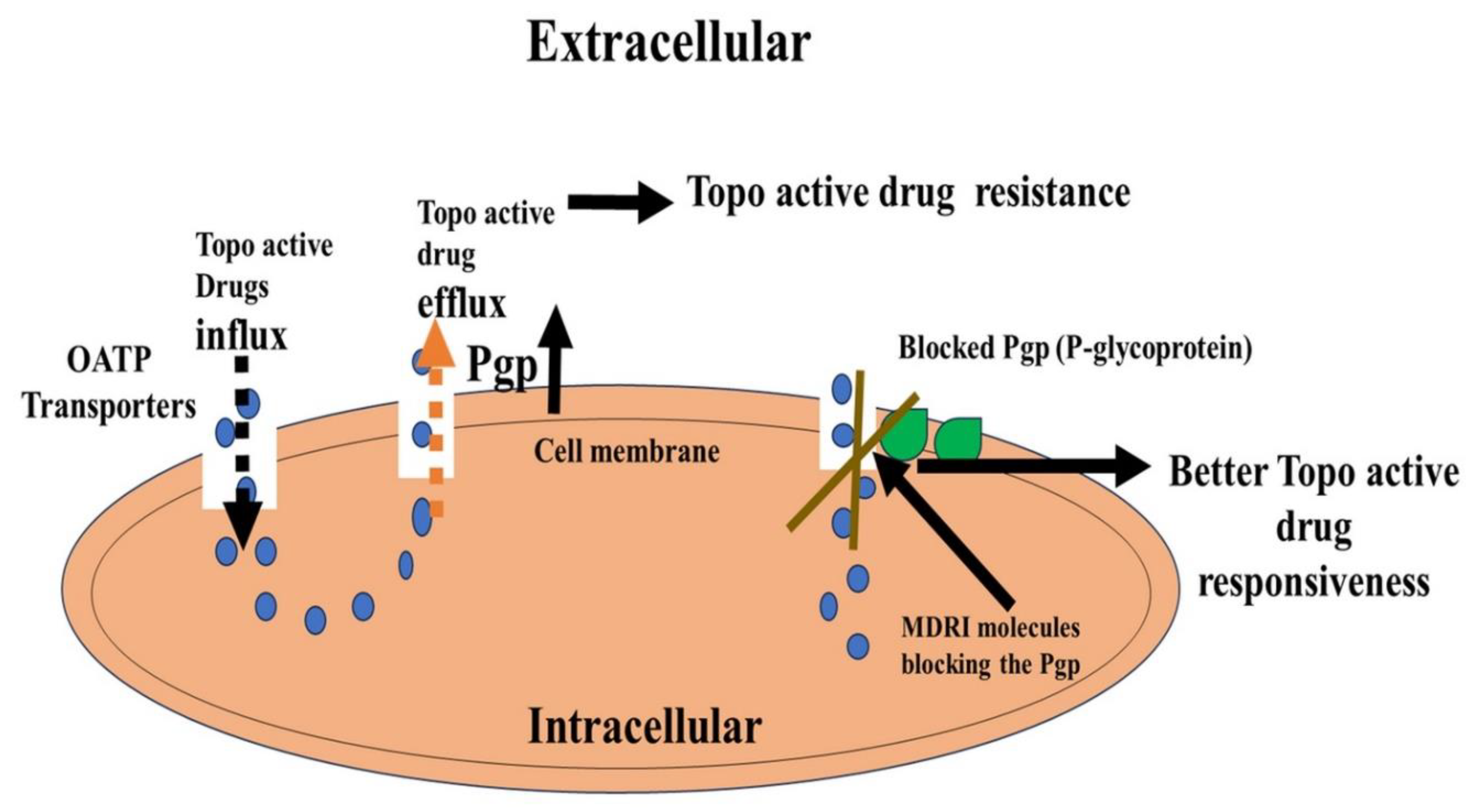
7. ABC transporters-mediated resistance of Topo drugs
8. GSH depletion and Topo-drug resistance
9. DNA damage response pathways and topo-active drug resistance
10. Metabolic reprogramming and topo active drug resistance
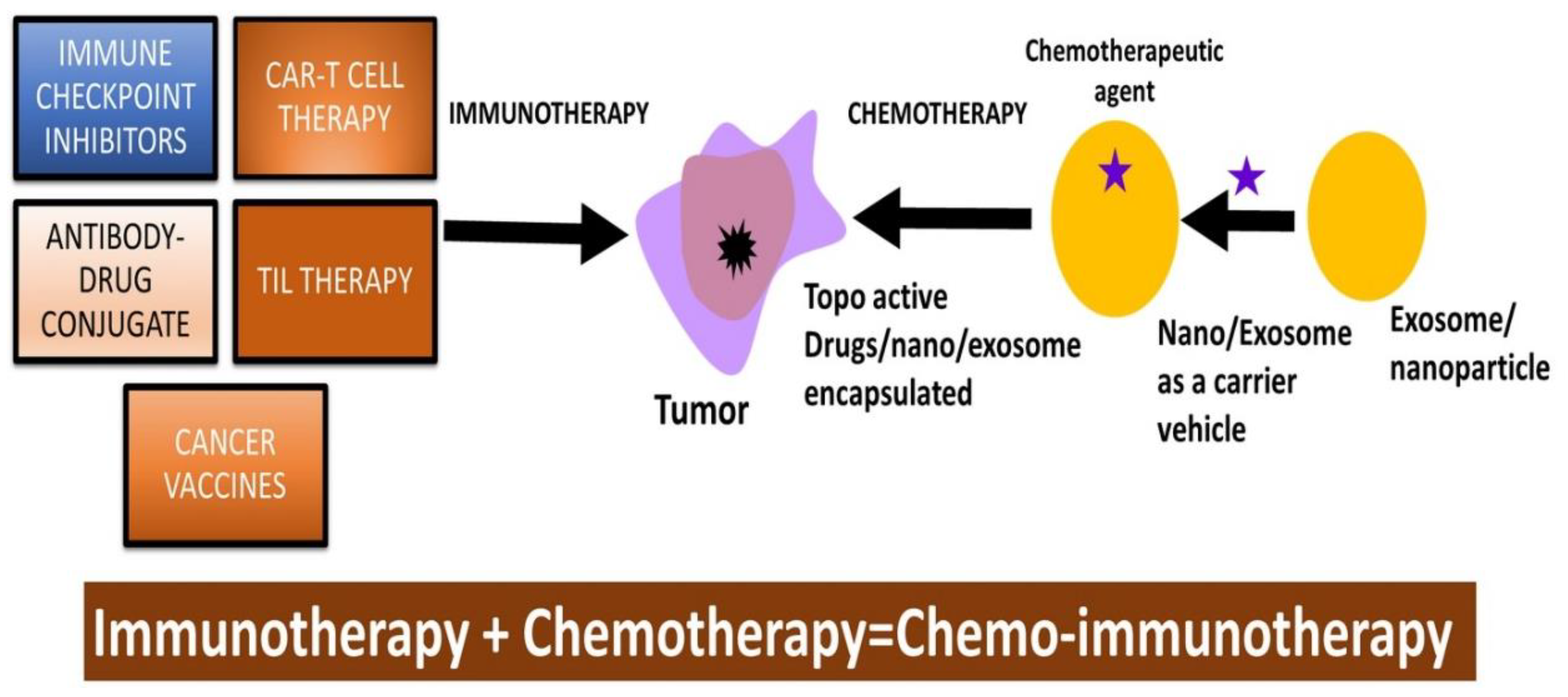
11. Immune checkpoint inhibitors and topo-active drugs
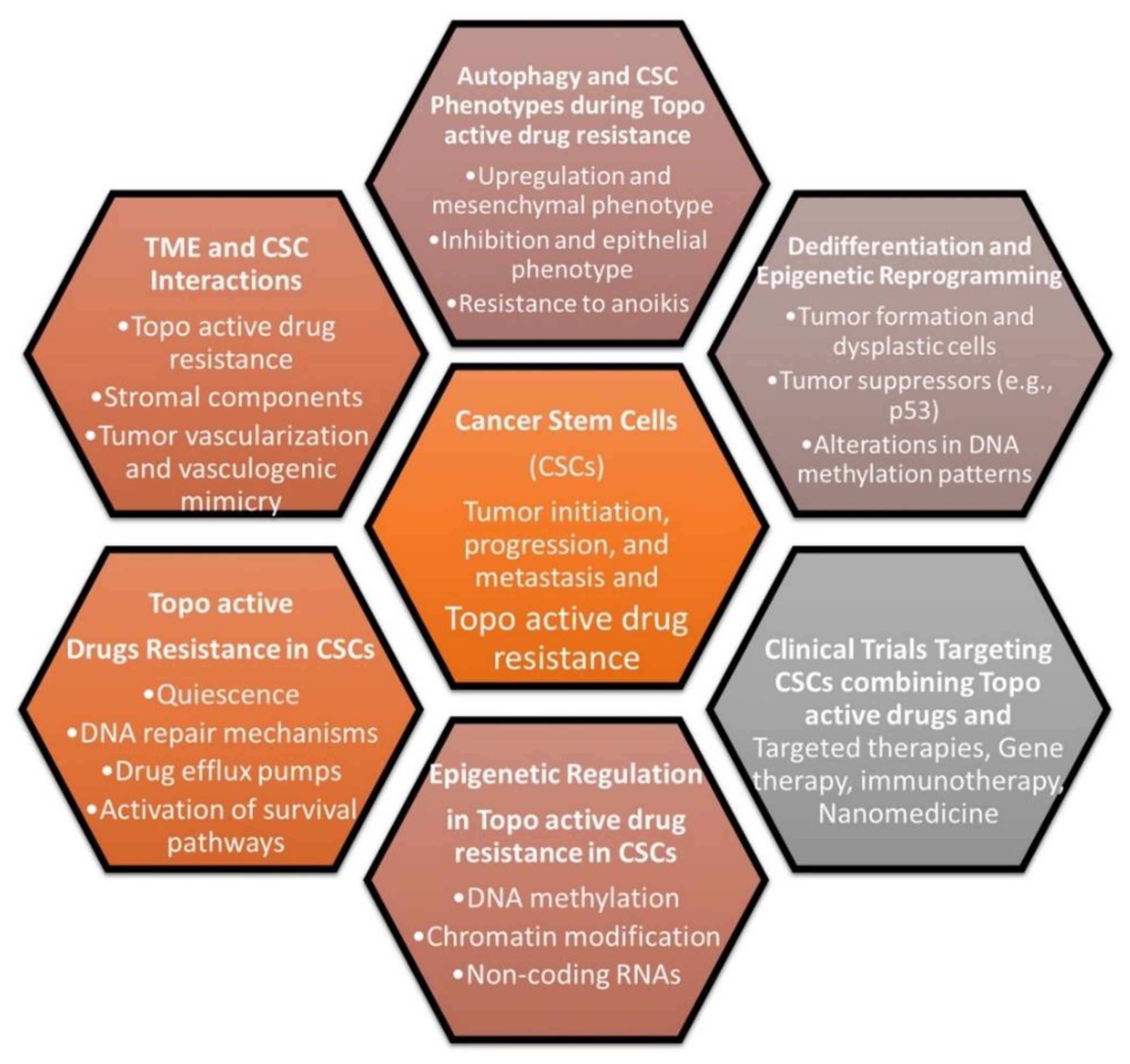
12. Topo-active drugs and cancer stem cells
13. Epigenetic changes and Topo-active drugs
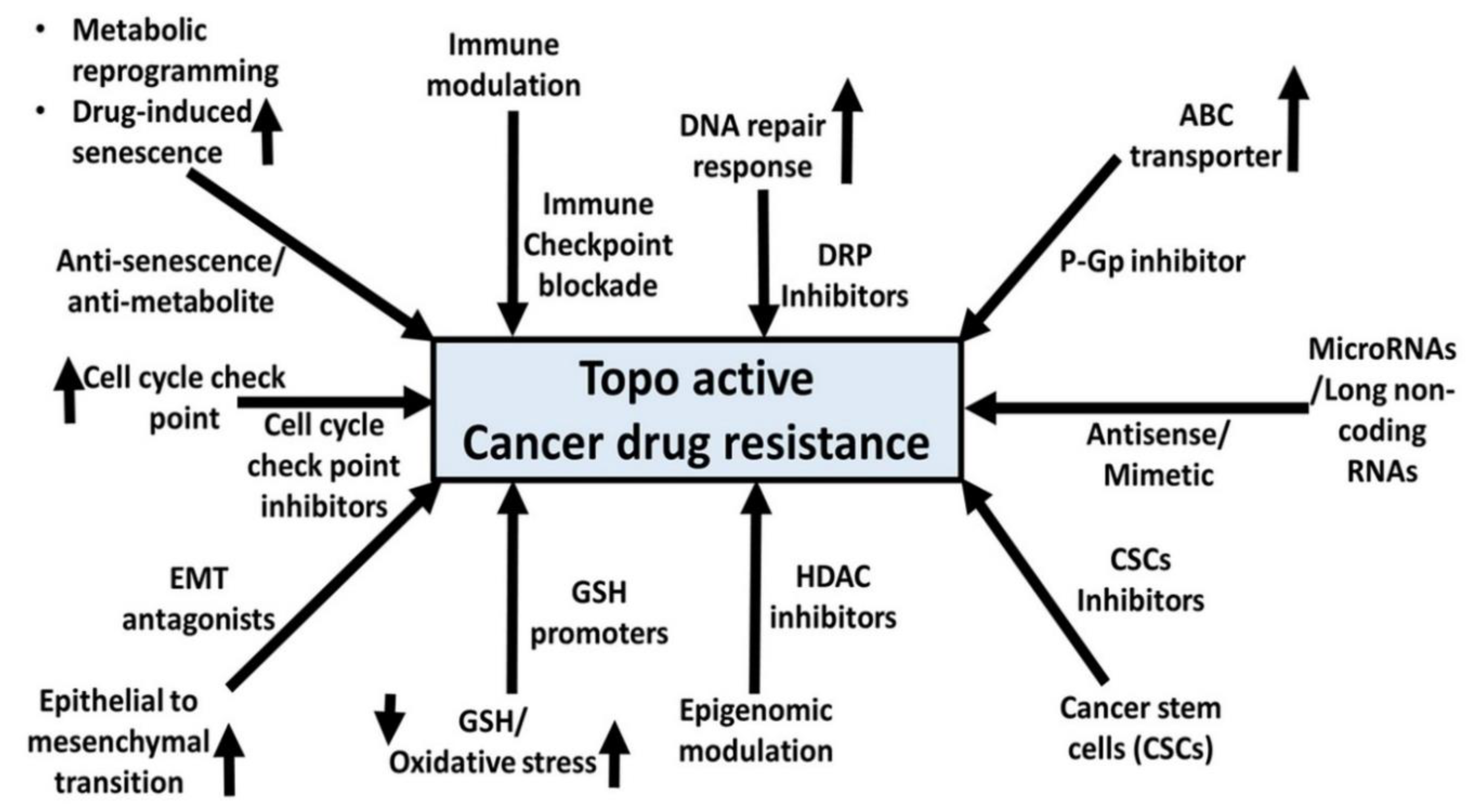
14. Miscellaneous mechanisms of resistance and Topo-active Drug-induced senescence and drug resistance
14.1. Regulatory RNAs and Topo active drugs
14.2. EMT and Topo-active drugs
15. Conclusion and Future Developments
Author Contributions
Funding
Conflicts of Interest
References
- Chhikara BS, Parang K. Global Cancer Statistics 2022: The Trends Projection Analysis. Chem Biol Lett. 2023;10(1):451.
- Morgan E, Arnold M, Gini A, Lorenzoni V, Cabasag CJ, Laversanne M, Vignat J, Ferlay J, Murphy N, Bray F. Global burden of colorectal cancer in 2020 and 2040: incidence and mortality estimates from GLOBOCAN. Gut. 2023;72(2):338-344. [CrossRef]
- Wang X, Zhang H, Chen X. Drug resistance and combating drug resistance in cancer. Cancer Drug Resist. 2019;2(2):141-160. [CrossRef]
- Emran TB, Shahriar A, Mahmud AR, Rahman T, Abir MH, Siddiquee MF, Ahmed H, Rahman N, Nainu F, Wahyudin E, Mitra S, Dhama K, Habiballah MM, Haque S, Islam A, Hassan MM. Multidrug Resistance in Cancer: Understanding Molecular Mechanisms, Immunoprevention and Therapeutic Approaches. Front Oncol. 2022;12:891652. [CrossRef]
- Vaidya FU, Sufiyan Chhipa A, Mishra V, Gupta VK, Rawat SG, Kumar A, Pathak C. Molecular and cellular paradigms of multidrug resistance in cancer. Cancer Rep (Hoboken). 2022;5(12):e1291. [CrossRef]
- Nikolaou M, Pavlopoulou A, Georgakilas AG, Kyrodimos E. The challenge of drug resistance in cancer treatment: a current overview. Clin Exp Metastasis. 2018;35(4):309-318. [CrossRef]
- Ramos A, Sadeghi S, Tabatabaeian H. Battling Chemoresistance in Cancer: Root Causes and Strategies to Uproot Them. Int J Mol Sci. 2021;22(17):9451. [CrossRef]
- You F, Gao C. Topoisomerase Inhibitors and Targeted Delivery in Cancer Therapy. Curr Top Med Chem. 2019;19(9):713-729. [CrossRef]
- Nitiss JL, Kiianitsa K, Sun Y, Nitiss KC, Maizels N. Topoisomerase Assays. Curr Protoc. 2021;1(10):e250. [CrossRef]
- Singh S, Pandey VP, Yadav K, Yadav A, Dwivedi UN. Natural Products as Anti-Cancerous Therapeutic Molecules Targeted towards Topoisomerases. Curr Protein Pept Sci. 2020;21(11):1103-1142. [CrossRef]
- Jang JY, Kim D, Kim ND. Recent Developments in Combination Chemotherapy for Colorectal and Breast Cancers with Topoisomerase Inhibitors. Int J Mol Sci. 2023;24(9):8457. [CrossRef]
- Madkour MM, Ramadan WS, Saleh E, El-Awady R. Epigenetic modulations in cancer: predictive biomarkers and potential targets for overcoming the resistance to topoisomerase I inhibitors. Ann Med. 2023;55(1):2203946. [CrossRef]
- Sun YL, Patel A, Kumar P, Chen ZS. Role of ABC transporters in cancer chemotherapy. Chin J Cancer. 2012;31(2):51-7. [CrossRef]
- Wtorek K, Długosz A, Janecka A. Drug resistance in topoisomerase-targeting therapy. Postępy Higieny i Medycyny Doświadczalnej. 2018;72:1073-1083. [CrossRef]
- Liu J, Qu L, Meng L, Shou C. Topoisomerase inhibitors promote cancer cell motility via ROS-mediated activation of JAK2-STAT1-CXCL1 pathway. J Exp Clin Cancer Res. 2019;38(1):370. [CrossRef]
- Nwabufo, CK. Relevance of ABC Transporters in Drug Development. Curr Drug Metab. 2022;23(6):434-446. [CrossRef]
- Simůnek T, Stérba M, Popelová O, Adamcová M, Hrdina R, Gersl V. Anthracycline-induced cardiotoxicity: overview of studies examining the roles of oxidative stress and free cellular iron. Pharmacol Rep. 2009;61(1):154-71. [CrossRef]
- Cai F, Luis MAF, Lin X, Wang M, Cai L, Cen C, Biskup E. Anthracycline-induced cardiotoxicity in the chemotherapy treatment of breast cancer: Preventive strategies and treatment. Mol Clin Oncol. 2019;11(1):15-23. [CrossRef]
- Kennedy L, Sandhu JK, Harper ME, Cuperlovic-Culf M. Role of Glutathione in Cancer: From Mechanisms to Therapies. Biomolecules. 2020;10(10):1429. [CrossRef]
- Sekhar, RV. GlyNAC Supplementation Improves Glutathione Deficiency, Oxidative Stress, Mitochondrial Dysfunction, Inflammation, Aging Hallmarks, Metabolic Defects, Muscle Strength, Cognitive Decline, and Body Composition: Implications for Healthy Aging. J Nutr. 2021;151(12):3606-3616. [CrossRef]
- Carrasco R, Castillo RL, Gormaz JG, Carrillo M, Thavendiranathan P. Role of Oxidative Stress in the Mechanisms of Anthracycline-Induced Cardiotoxicity: Effects of Preventive Strategies. Oxid Med Cell Longev. 2021;2021:8863789. [CrossRef]
- Hickman JA, Potten CS, Merritt AJ, Fisher TC. Apoptosis and cancer chemotherapy. Philos Trans R Soc Lond B Biol Sci. 1994;345(1313):319-25. [CrossRef]
- Dexheimer TS, Pommier Y. DNA cleavage assay for the identification of topoisomerase I inhibitors. Nat Protoc. 2008;3(11):1736-50. [CrossRef]
- Gokduman, K. Strategies Targeting DNA Topoisomerase I in Cancer Chemotherapy: Camptothecins, Nanocarriers for Camptothecins, Organic Non-Camptothecin Compounds and Metal Complexes. Curr Drug Targets. 2016;17(16):1928-1939. [CrossRef]
- Jurkovicova D, Neophytou CM, Gašparović AČ, Gonçalves AC. DNA Damage Response in Cancer Therapy and Resistance: Challenges and Opportunities. Int J Mol Sci. 2022;23(23):14672. [CrossRef]
- Sharma P, Hu-Lieskovan S, Wargo JA, Ribas A. Primary, Adaptive, and Acquired Resistance to Cancer Immunotherapy. Cell. 2017;168(4):707-723. [CrossRef]
- Jackson CM, Choi J, Lim M. Mechanisms of immunotherapy resistance: lessons from glioblastoma. Nat Immunol. 2019;20(9):1100-1109. [CrossRef]
- van Elsas MJ, van Hall T, van der Burg SH. Future Challenges in Cancer Resistance to Immunotherapy. Cancers (Basel). 2020;12(4):935. [CrossRef]
- Dragu DL, Necula LG, Bleotu C, Diaconu CC, Chivu-Economescu M. Therapies targeting cancer stem cells: Current trends and future challenges. World J Stem Cells. 2015;7(9):1185-201. [CrossRef]
- Harris KS, Shi L, Foster BM, Mobley ME, Elliott PL, Song CJ, Watabe K, Langefeld CD, Kerr BA. CD117/c-kit defines a prostate CSC-like subpopulation driving progression and TKI resistance. Sci Rep. 2021;11(1):1465. [CrossRef]
- Lei X, He Q, Li Z, Zou Q, Xu P, Yu H, Ding Y, Zhu W. Cancer stem cells in colorectal cancer and the association with chemotherapy resistance. Med Oncol. 2021;38(4):43. [CrossRef]
- Zhou G, Qin M, Zhang X, Yang J, Yu H. Topotecan induces hepatocellular injury via ASCT2 mediated oxidative stress. Gastroenterol Hepatol. 2021;44(1):1-12. [CrossRef]
- El-Far AH, Tantawy MA, Al Jaouni SK, Mousa SA. Thymoquinone-chemotherapeutic combinations: new regimen to combat cancer and cancer stem cells. Naunyn Schmiedebergs Arch Pharmacol. 2020;393(9):1581-1598. [CrossRef]
- Garcia-Mayea Y, Mir C, Masson F, Paciucci R, LLeonart ME. Insights into new mechanisms and models of cancer stem cell multidrug resistance. Semin Cancer Biol. 2020;60:166-180. [CrossRef]
- Phan LM, Yeung SC, Lee MH. Cancer metabolic reprogramming: importance, main features, and potentials for precise targeted anti-cancer therapies. Cancer Biol Med. 2014;11(1):1-19. [CrossRef]
- Szwed A, Kim E, Jacinto E. Regulation and metabolic functions of mTORC1 and mTORC2. Physiol Rev. 2021;101(3):1371-1426. [CrossRef]
- Faubert B, Solmonson A, DeBerardinis RJ. Metabolic reprogramming and cancer progression. Science. 2020;368(6487):eaaw5473. [CrossRef]
- Wagner A, Kosnacova H, Chovanec M, Jurkovicova D. Mitochondrial Genetic and Epigenetic Regulations in Cancer: Therapeutic Potential. Int J Mol Sci. 2022;23(14):7897. [CrossRef]
- Karlstaedt A, Moslehi J, de Boer RA. Cardio-onco-metabolism: metabolic remodelling in cardiovascular disease and cancer. Nat Rev Cardiol. 2022;19(6):414-425. [CrossRef]
- Pommier Y, Orr A, Kohn KW, Riou JF. Differential effects of amsacrine and epipodophyllotoxins on topoisomerase II cleavage in the human c-myc protooncogene. Cancer Res. 1992;52(11):3125-30.
- Champoux, JJ. DNA topoisomerases: structure, function, and mechanism. Annu Rev Biochem. 2001; 70:369-413. [CrossRef]
- Martínez-Reyes I, Chandel NS. Cancer metabolism: looking forward. Nat Rev Cancer. 2021;21(10):669-680. [CrossRef]
- Cuvier O, Stanojcic S, Lemaitre JM, Mechali M. A topoisomerase II-dependent mechanism for resetting replicons at the S-M-phase transition. Genes Dev. 2008;22(7):860-5. [CrossRef]
- Sakurai K, Muguruma K, Murata A, Toyokawa T, Amano R, Kubo N, Tanaka H, Yashiro M, Maeda K, Ohira M, Hirakawa K. Early gastric cancer with suspected brain metastasis arising eight years after curative resection: a case report. BMC Res Notes. 2014;7:818. [CrossRef]
- Oliveira-Costa JP, de Carvalho AF, da Silveira da GG, Amaya P, Wu Y, Park KJ, Gigliola MP, Lustberg M, Buim ME, Ferreira EN, Kowalski LP, Chalmers JJ, Soares FA, Carraro DM, Ribeiro-Silva A. Gene expression patterns through oral squamous cell carcinoma development: PD-L1 expression in primary tumor and circulating tumor cells. Oncotarget. 2015;6(25):20902-20. [CrossRef]
- McClendon AK, Osheroff N. DNA topoisomerase II, genotoxicity, and cancer. Mutat Res. 2007; 623(1-2):83-97. [CrossRef]
- Nitiss, JL. DNA topoisomerase II and its growing repertoire of biological functions. Nat Rev Cancer. 2009;9(5):327-37. [CrossRef]
- Delgado-Benito V, Rosen DB, Wang Q, Gazumyan A, Pai JA, Oliveira TY, Sundaravinayagam D, Zhang W, Andreani M, Keller L, Kieffer-Kwon KR, Pękowska A, Jung S, Driesner M, Subbotin RI, Casellas R, Chait BT, Nussenzweig MC, Di Virgilio M. The Chromatin Reader ZMYND8 Regulates Igh Enhancers to Promote Immunoglobulin Class Switch Recombination. Mol Cell, 2018;72(4):636-649.e8. [CrossRef]
- Burden DA, Kingma PS, Froelich-Ammon SJ, Bjornsti MA, Patchan MW, Thompson RB, Osheroff N. Topoisomerase II.etoposide interactions direct the formation of drug-induced enzyme-DNA cleavage complexes. J Biol Chem. 1996;271(46):29238-44. [CrossRef]
- Zhang C, Xu C, Gao X, Yao Q. Platinum-based drugs for cancer therapy and anti-tumor strategies. Theranostics, 2013;12(5):2115-2132. [CrossRef]
- Ishikawa T, Ikegami Y, Sano K, Nakagawa H, Sawada S. Transport mechanism-based drug molecular design: novel camptothecin analogues to circumvent ABCG2-associated drug resistance of human tumor cells. Curr Pharm Des. 2006;12(3):313-25. [CrossRef]
- Takagi T, Saotome T. Chemotherapy with irinotecan (CPT-11), a topoisomerase-I inhibitor, for refractory and relapsed non-Hodgkin's lymphoma. Leuk Lymphoma. 2001;42(4):577-86. [CrossRef]
- Quintieri L, Geroni C, Fantin M, Battaglia R, Rosato A, Speed WC, Zanovello P, Floreani M. Formation and Antitumor Activity of PNU-159682, A Major Metabolite of Nemorubicin in Human Liver Microsomes. American Association for Cancer Research; 2005;11(4):1608–1617. [CrossRef]
- Danesi R, Agen C, Grandi M, Nardini V, Bevilacqua G, Del Tacca M. 3'-Deamino-3'-(2-methoxy-4-morpholinyl)-doxorubicin (FCE 23762): a new anthracycline derivative with enhanced cytotoxicity and reduced cardiotoxicity. Eur J Cancer. 1993;29A(11):1560-5. [CrossRef]
- Scalabrin M, Quintieri L, Palumbo M, Riccardi Sirtori F, Gatto B. Virtual Cross-Linking of the Active Nemorubicin Metabolite PNU-159682 to Double-Stranded DNA. Chem Res Toxicol. 2017;30(2):614-624. [CrossRef]
- Mastrangelo S, Attinà G, Triarico S, Romano A, Maurizi P, Ruggiero A. The DNA-topoisomerase inhibitors in cancer therapy. Biomedical and Pharmacology Journal Oriental Scientific Publishing Company; 2022;15(2):553–562. [CrossRef]
- Sinha, BK. Topoisomerase inhibitors. A review of their therapeutic potential in cancer. Drugs. 1995;49(1):11-9. [CrossRef]
- Selas A, Martin-Encinas E, Fuertes M, Masdeu C, Rubiales G, Palacios F, Alonso C. A patent review of topoisomerase I inhibitors (2016-present). Expert Opin Ther Pat. 2021;31(6):473-508. [CrossRef]
- Zander SA, Sol W, Greenberger L, Zhang Y, van Tellingen O, Jonkers J, Borst P, Rottenberg S. EZN-2208 (PEG-SN38) overcomes ABCG2-mediated topotecan resistance in BRCA1-deficient mouse mammary tumors. PLoS One. 2012;7(9):e45248. [CrossRef]
- Dao NV, Ercole F, Urquhart MC, Kaminskas LM, Nowell CJ, Davis TP, Sloan EK, Whittaker MR, Quinn JF. Trisulfide linked cholesteryl PEG conjugate attenuates intracellular ROS and collagen-1 production in a breast cancer co-culture model. Biomater Sci. 2021;9(3):835-846. [CrossRef]
- Okoro CO, Fatoki TH. A Mini Review of Novel Topoisomerase II Inhibitors as Future Anticancer Agents. International Journal of Molecular Sciences. 2023;24(3):2532. [CrossRef]
- Khalil WKB, Zarouk WA, Eldeen GN, Ramadan A, Fayez A, Esmaiel NN, Foda BM, Hamed KH, Kassem SM, El-Bassyouni HT. Apoptosis, reactive oxygen species and DNA damage in Familial Mediterranean Fever patients.Gene Reports. 2019;14:76–80. [CrossRef]
- Sinha BK, Kumar A, Bhattacharjee S, Espey MG, Mason RP. Effect of nitric oxide on the anticancer activity of the topoisomerase-active drugs etoposide and adriamycin in human melanoma cells. J Pharmacol Exp Ther. 2013;347(3):607-14. [CrossRef]
- Sharma NK, Kumar A, Kumari A, Tokar EJ, Waalkes MP, Bortner CD, Williams J, Ehrenshaft M, Mason RP, Sinha BK. Nitric Oxide Down-Regulates Topoisomerase I and Induces Camptothecin Resistance in Human Breast MCF-7 Tumor Cells. PLoS One. 2015;10(11):e0141897. [CrossRef]
- Muhammad N, Usmani D, Tarique M, Naz H, Ashraf M, Raliya R, Tabrez S, Zughaibi TA, Alsaieedi A, Hakeem IJ, Suhail M. The Role of Natural Products and Their Multitargeted Approach to Treat Solid Cancer. Cells. 2022;11(14):2209. [CrossRef]
- Chen L, Zhu X, Zou Y, Xing J, Gilson E, Lu Y, Ye J. The topoisomerase II catalytic inhibitor ICRF-193 preferentially targets telomeres that are capped by TRF2. Am J Physiol Cell Physiol. 2015;308(5):C372-7. [CrossRef]
- Baikar S, Malpathak N. Secondary metabolites as DNA topoisomerase inhibitors: A new era towards designing of anticancer drugs. Pharmacogn Rev. 2010;4(7):12 26. [CrossRef]
- Sinicropi MS, Lappano R, Caruso A, Santolla MF, Pisano A, Rosano C, Capasso A, Panno A, Lancelot JC, Rault S, Saturnino C, Maggiolini M. (6-bromo-1,4-dimethyl-9H-carbazol-3-yl-methylene)-hydrazine (carbhydraz) acts as a GPER agonist in breast cancer cells. Curr Top Med Chem. 2015;15(11):1035-42. [CrossRef]
- Sordet O, Goldman A, Pommier Y. Topoisomerase II and tubulin inhibitors both induce the formation of apoptotic topoisomerase I cleavage complexes. Mol Cancer Ther. 2006;(12):3139-44. [CrossRef]
- Kumar S, Kushwaha PP, Gupta S. Emerging targets in cancer drug resistance. Cancer Drug Resist. 2019;2(2):161-177. [CrossRef]
- Vasan N, Baselga J, Hyman DM. A view on drug resistance in cancer. Nature. 2019;575(7782):299-309. [CrossRef]
- Lim ZF, Ma PC. Emerging insights of tumor heterogeneity and drug resistance mechanisms in lung cancer targeted therapy. J Hematol Oncol. 2019;12(1):134. [CrossRef]
- Galmarini CM, Galmarini FC. Multidrug resistance in cancer therapy: role of the microenvironment. Curr Opin Investig Drugs. 2003;4(12):1416-21.
- Liu Y, Zhang J, Li L, Yin G, Zhang J, Zheng S, Cheung H, Wu N, Lu N, Mao X, Yang L, Zhang J, Zhang L, Seth S, Chen H, Song X, Liu K, Xie Y, Zhou L, Zhao C, Han N, Chen W, Zhang S, Chen L, Cai W, Li L, Shen M, Xu N, Cheng S, Yang H, Lee JJ, Correa A, Fujimoto J, Behrens C, Chow CW, William WN, Heymach JV, Hong WK, Swisher S, Wistuba II, Wang J, Lin D, Liu X, Futreal PA, Gao Y. Genomic heterogeneity of multiple synchronous lung cancer. Nat Commun. 2016;7:13200. [CrossRef]
- Haider T, Pandey V, Banjare N, Gupta PN, Soni V. Drug resistance in cancer: mechanisms and tackling strategies. Pharmacol Rep. 2020;72(5):1125-1151. [CrossRef]
- Maleki EH, Bahrami AR, Matin MM. Cancer cell cycle heterogeneity as a critical determinant of therapeutic resistance. Genes Dis. 2023;11(1):189-204. [CrossRef]
- Asif PJ, Longobardi C, Hahne M, Medema JP. The Role of Cancer-Associated Fibroblasts in Cancer Invasion and Metastasis. Cancers (Basel). 2021;13(18):4720. [CrossRef]
- Wright K, Ly T, Kriet M, Czirok A, Thomas SM. Cancer-Associated Fibroblasts: Master Tumor Microenvironment Modifiers. Cancers (Basel). 2023;15(6):1899. [CrossRef]
- Li M, Li M, Yin T, Shi H, Wen Y, Zhang B, Chen M, Xu G, Ren K, Wei Y. Targeting of cancer-associated fibroblasts enhances the efficacy of cancer chemotherapy by regulating the tumor microenvironment. Mol Med Rep. 2016;13(3):2476-84. [CrossRef]
- Mao X, Xu J, Wang W, Liang C, Hua J, Liu J, Zhang B, Meng Q, Yu X, Shi S. Crosstalk between cancer-associated fibroblasts and immune cells in the tumor microenvironment: new findings and future perspectives. Mol Cancer. 2021;20(1):131. [CrossRef]
- Rimal R, Desai P, Daware R, Hosseinnejad A, Prakash J, Lammers T, Singh S. Cancer-associated fibroblasts: Origin, function, imaging, and therapeutic targeting. Adv Drug Deliv Rev. 2022;189:114504. [CrossRef]
- Tie Y, Tang F, Peng D, Zhang Y, Shi H. TGF-beta signal transduction: biology, function and therapy for diseases. Mol Biomed. 2022;3(1):45. [CrossRef]
- Zhang C, Fei Y, Wang H, Hu S, Liu C, Hu R, Du Q. CAFs orchestrates tumor immune microenvironment-A new target in cancer therapy? Front Pharmacol. 2023;14:1113378.
- Beca F, Polyak K. Intratumor Heterogeneity in Breast Cancer. Adv Exp Med Biol. 2016;882:169-89. [CrossRef]
- Tong M, Deng Z, Zhang X, He B, Yang M, Cheng W, Liu Q. New insights from the widening homogeneity perspective to target intratumor heterogeneity. Cancer Commun (Lond). 2018;38(1):17. [CrossRef]
- Turajlic S, Sottoriva A, Graham T, Swanton C. Resolving genetic heterogeneity in cancer. Nat Rev Genet. 2019;20(7):404-416. [CrossRef]
- Marusyk A, Janiszewska M, Polyak K. Intratumor Heterogeneity: The Rosetta Stone of Therapy Resistance. Cancer Cell. 2020;37(4):471-484. [CrossRef]
- Hyman DM, Piha-Paul SA, Won H, Rodon J, Saura C, Shapiro GI, Juric D, Quinn DI, Moreno V, Doger B, Mayer IA, Boni V, Calvo E, Loi S, Lockhart AC, Erinjeri JP, Scaltriti M, Ulaner GA, Patel J, Tang J, Beer H, Selcuklu SD, Hanrahan AJ, Bouvier N, Melcer M, Murali R, Schram AM, Smyth LM, Jhaveri K, Li BT, Drilon A, Harding JJ, Iyer G, Taylor BS, Berger MF, Cutler RE Jr, Xu F, Butturini A, Eli LD, Mann G, Farrell C, Lalani AS, Bryce RP, Arteaga CL, Meric-Bernstam F, Baselga J, Solit DB. HER kinase inhibition in patients with HER2- and HER3-mutant cancers. Nature. 2018;554(7691):189-194. [CrossRef]
- Guo M, Peng Y, Gao A, Du C, Herman JG. Epigenetic heterogeneity in cancer. Biomark Res. 2019;7:23. [CrossRef]
- Mattos EC, Silva LP, Valero C, de Castro PA, Dos Reis TF, Ribeiro LFC, Marten MR, Silva-Rocha R, Westmann C, da Silva CHTP, Taft CA, Al-Furaiji N, Bromley M, Mortensen UH, Benz JP, Brown NA, Goldman GH. The Aspergillus fumigatus Phosphoproteome Reveals Roles of High-Osmolarity Glycerol Mitogen-Activated Protein Kinases in Promoting Cell Wall Damage and Caspofungin Tolerance. mBio. 2020;11(1):e02962-19. [CrossRef]
- Beyes S, Bediaga NG, Zippo A. An Epigenetic Perspective on Intra-Tumour Heterogeneity: Novel Insights and New Challenges from Multiple Fields. Cancers (Basel). 2021;13(19):4969. [CrossRef]
- Vessoni AT, Filippi-Chiela EC, Lenz G, Batista LFZ. Tumor propagating cells: drivers of tumor plasticity, heterogeneity, and recurrence. Oncogene. 2020;39(10):2055-2068. [CrossRef]
- Biswas A, De S. Drivers of dynamic intratumor heterogeneity and phenotypic plasticity. Am J Physiol Cell Physiol. 2021;320(5):C750-C760. [CrossRef]
- Jones PA, Baylin SB. The fundamental role of epigenetic events in cancer. Nat Rev Genet. 2002; 3(6):415-28. [CrossRef]
- Garinis GA, Patrinos GP, Spanakis NE, Menounos PG. DNA hypermethylation: when tumour suppressor genes go silent. Hum Genet. 2002;111(2):115-27. [CrossRef]
- Vaissière T, Sawan C, Herceg Z. Epigenetic interplay between histone modifications and DNA methylation in gene silencing. Mutat Res. 2008;659(1-2):40-8. [CrossRef]
- Zhao S, Allis CD, Wang GG. The language of chromatin modification in human cancers. Nat Rev Cancer. 2021;21(7):413-430. [CrossRef]
- Jiang YZ, Liu Y, Xiao Y, Hu X, Jiang L, Zuo WJ, Ma D, Ding J, Zhu X, Zou J, Verschraegen C, Stover DG, Kaklamani V, Wang ZH, Shao ZM. Molecular subtyping and genomic profiling expand precision medicine in refractory metastatic triple-negative breast cancer: the FUTURE trial. Cell Res. 2021;31(2):178-186. [CrossRef]
- Yang C, Mai Z, Liu C, Yin S, Cai Y, Xia C. Natural Products in Preventing Tumor Drug Resistance and Related Signaling Pathways. Molecules. 2022;27(11):3513. [CrossRef]
- Gongora C, Vezzio-Vie N, Tuduri S, Denis V, Causse A, Auzanneau C, Collod-Beroud G, Coquelle A, Pasero P, Pourquier P, Martineau P, Del Rio M. New Topoisomerase I mutations are associated with resistance to camptothecin. Mol Cancer. 2011;10:64. [CrossRef]
- Sinha BK, Haim N, Dusre L, Kerrigan D, Pommier Y. DNA strand breaks produced by etoposide (VP-16,213) in sensitive and resistant human breast tumor cells: implications for the mechanism of action. Cancer Res. 1988;48(18):5096-100.
- Beck WT, Danks MK. Mechanisms of resistance to drugs that inhibit DNA topoisomerases. Semin Cancer Biol. 1991;2:235–44.
- Pommier Y, Leo E, Zhang H, Marchand C. DNA topoisomerases and their poisoning by anticancer and antibacterial drugs. Chem Biol. 2010;17(5):421-33. [CrossRef]
- Vos SM, Tretter EM, Schmidt BH, Berger JM. All tangled up: how cells direct, manage and exploit topoisomerase function. Nat Rev Mol Cell Biol. 2011;12(12):827-41. [CrossRef]
- Pommier Y, Sun Y, Huang SN, Nitiss JL. Roles of eukaryotic topoisomerases in transcription, replication and genomic stability. Nat Rev Mol Cell Biol. 2016;17(11):703-721. [CrossRef]
- Boot A, Liu M, Stantial N, Shah V, Yu W, Nitiss KC, Nitiss JL, Jinks-Robertson S, Rozen SG.Recurrent mutations in topoisomerase IIα cause a previously undescribed mutator phenotype in human cancers. Proc Natl Acad Sci U S A. 2022;119(4):e2114024119. [CrossRef]
- Bandak AF, Blower TR, Nitiss KC, Shah V, Nitiss JL, Berger JM. Using energy to go downhill - a genoprotective role for ATPase activity in DNA topoisomerase II. bioRxiv [Preprint]. 2023:2023.06.27.546777. [CrossRef]
- Hinds M, Deisseroth K, Mayes J, Altschuler E, Jansen R, Ledley FD, Zwelling LA. Identification of a point mutation in the topoisomerase II gene from a human leukemia cell line containing an amsacrine-resistant form of topoisomerase II. Cancer Res. 1991;51(17):4729-31.
- Campain JA, Gottesman MM, Pastan I. A novel mutant topoisomerase II alpha present in VP-16-resistant human melanoma cell lines has a deletion of alanine 429. Biochemistry. 1994; 33(37):11327-32. [CrossRef]
- Robert J, Larsen AK. Drug resistance to topoisomerase II inhibitors. Biochimie. 1998;80(3):247-54. [CrossRef]
- Borst, P. Genetic mechanisms of drug resistance. A review. Acta Oncol. 1991;30(1):87-105. [CrossRef]
- Ganapathi RN, Ganapathi MK. Mechanisms regulating resistance to inhibitors of topoisomerase II. Front Pharmacol. 2013;4:89. [CrossRef]
- Licata S, Saponiero A, Mordente A, Minotti G. Doxorubicin metabolism and toxicity in human myocardium: role of cytoplasmic deglycosidation and carbonyl reduction. Chem Res Toxicol. 2000;13(5):414-20. [CrossRef]
- Chen AY, Chou R, Shih SJ, Lau D, Gandara D. Enhancement of radiotherapy with DNA topoisomerase I-targeted drugs. Crit Rev Oncol Hematol. 2004;50(2):111-9. [CrossRef]
- Kumar A, Patel S, Bhatkar D, Sarode SC, Sharma NK. A novel method to detect intracellular metabolite alterations in MCF-7 cells by doxorubicin induced cell death. Metabolomics. 2021;17(1):3. [CrossRef]
- Potęga, A. Glutathione-Mediated Conjugation of Anticancer Drugs: An Overview of Reaction Mechanisms and Biological Significance for Drug Detoxification and Bioactivation. Molecules. 2022;27(16):5252. [CrossRef]
- Sinha BK, Mimnaugh EG. Free radicals and anticancer drug resistance: oxygen free radicals in the mechanisms of drug cytotoxicity and resistance by certain tumors. Free Radic Biol Med. 1990;8(6):567-81. [CrossRef]
- Bartoszek A, Wolf CR. Enhancement of doxorubicin toxicity following activation by NADPH cytochrome P450 reductase. Biochem Pharmacol. 1992;43(7):1449-57. [CrossRef]
- Laroche-Clary A, Le Morvan V, Yamori T, Robert J. Cytochrome P450 1B1 gene polymorphisms as predictors of anticancer drug activity: studies with in vitro models. Mol Cancer Ther. 2010;9(12):3315-21. [CrossRef]
- Pathania S, Bhatia R, Baldi A, Singh R, Rawal RK. Drug metabolizing enzymes and their inhibitors' role in cancer resistance. Biomed Pharmacother. 2018;105:53-65. [CrossRef]
- Mathijssen RH, de Jong FA, van Schaik RH, Lepper ER, Friberg LE, Rietveld T, de Bruijn P, Graveland WJ, Figg WD, Verweij J, Sparreboom A. Prediction of irinotecan pharmacokinetics by use of cytochrome P450 3A4 phenotyping probes. J Natl Cancer Inst. 2004;96(21):1585-92. [CrossRef]
- de Man FM, Goey AKL, van Schaik RHN, Mathijssen RHJ, Bins S. Individualization of Irinotecan Treatment: A Review of Pharmacokinetics, Pharmacodynamics, and Pharmacogenetics. Clin Pharmacokinet. 2018;57(10):1229-1254. [CrossRef]
- Riordan JR, Ling V. Purification of P-glycoprotein from plasma membrane vesicles of Chinese hamster ovary cell mutants with reduced colchicine permeability. J Biol Chem. 1979;254(24):12701-5.
- Alpsoy A, Yasa S, Gündüz U. Etoposide resistance in MCF-7 breast cancer cell line is marked by multiple mechanisms. Biomed Pharmacother. 2014;68(3):351-5. [CrossRef]
- Goldwirt L, Beccaria K, Carpentier A, Farinotti R, Fernandez C. Irinotecan and temozolomide brain distribution: a focus on ABCB1. Cancer Chemother Pharmacol. 2014;74(1):185-93. [CrossRef]
- Choi YH, Yu AM. ABC transporters in multidrug resistance and pharmacokinetics, and strategies for drug development. Curr Pharm Des. 2014;20(5):793-807. [CrossRef]
- Thomas C, Tampé R. Structural and Mechanistic Principles of ABC Transporters. Annu Rev Biochem. 2020;89:605-636. [CrossRef]
- Wei T, Bi G, Bian Y, Ruan S, Yuan G, Xie H, Zhao M, Shen R, Zhu Y, Wang Q, Yang Y, Zhu D. The Significance of Secreted Phosphoprotein 1 in Multiple Human Cancers. Front Mol Biosci. 2020; 7:565383. [CrossRef]
- Długosz A, Gach-Janczak K, Szymański J, Deredas D, Janecki T, Janecka A. Involvement of α-methylene-γ- and δ-lactones in the suppression of multidrug resistance in MCF-7 cells. Pharmacol Rep. 2018;70(4):631-638. [CrossRef]
- Zander SA, Kersbergen A, Sol W, Gonggrijp M, van de Wetering K, Jonkers J, Borst P, Rottenberg S. Lack of ABCG2 shortens latency of BRCA1-deficient mammary tumors and this is not affected by genistein or resveratrol. Cancer Prev Res (Phila). 2012;5(8):1053-60. [CrossRef]
- Natarajan K, Xie Y, Baer MR, Ross DD. Role of breast cancer resistance protein (BCRP/ABCG2) in cancer drug resistance. Biochem Pharmacol. 2012;83(8):1084-103. [CrossRef]
- Omori M, Noro R, Seike M, Matsuda K, Hirao M, Fukuizumi A, Takano N, Miyanaga A, Gemma A. Inhibitors of ABCB1 and ABCG2 overcame resistance to topoisomerase inhibitors in small cell lung cancer. Thorac Cancer. 2022;13(15):2142-2151. [CrossRef]
- Kawabata S, Oka M, Shiozawa K, Tsukamoto K, Nakatomi K, Soda H, Fukuda M, Ikegami Y, Sugahara K, Yamada Y, Kamihira S, Doyle LA, Ross DD, Kohno S. Breast cancer resistance protein directly confers SN-38 resistance of lung cancer cells. Biochem Biophys Res Commun. 20012;80(5):1216-23. [CrossRef]
- Nakanishi T, Ross DD. Breast cancer resistance protein (BCRP/ABCG2): its role in multidrug resistance and regulation of its gene expression. Chin J Cancer. 2012;31(2):73-99. [CrossRef]
- Bukowski K, Kciuk M, Kontek R. Mechanisms of Multidrug Resistance in Cancer Chemotherapy. Int J Mol Sci. 2020;21(9):3233. [CrossRef]
- Robey RW, Pluchino KM, Hall MD, Fojo AT, Bates SE, Gottesman MM. Revisiting the role of ABC transporters in multidrug-resistant cancer. Nat Rev Cancer. 2018;18(7):452-464. [CrossRef]
- Bonitto EP, McKeown BT, Goralski KB. Jadomycins: A potential chemotherapy for multi-drug resistant metastatic breast cancer. Pharmacol Res Perspect. 2021;9(6):e00886. [CrossRef]
- Alferiev IS, Guerrero DT, Guan P, Nguyen F, Kolla V, Soberman D, Pressly BB, Fishbein I, Brodeur GM, Chorny M. Poloxamer-linked prodrug of a topoisomerase I inhibitor SN22 shows efficacy in models of high-risk neuroblastoma with primary and acquired chemoresistance. FASEB J. 2022;36(3):e22213. [CrossRef]
- Sies, H. Glutathione and its role in cellular functions. Free Radic Biol Med. 1999;27(9-10):916-21. [CrossRef]
- Lee J, Roh JL. Unleashing Ferroptosis in Human Cancers: Targeting Ferroptosis Suppressor Protein 1 for Overcoming Therapy Resistance. Antioxidants (Basel). 2023;12(6):1218. [CrossRef]
- Traverso N, Ricciarelli R, Nitti M, Marengo B, Furfaro AL, Pronzato MA, Marinari UM, Domenicotti C. Role of glutathione in cancer progression and chemoresistance. Oxid Med Cell Longev. 2013;2013:972913. [CrossRef]
- Daga M, Ullio C, Argenziano M, Dianzani C, Cavalli R, Trotta F, Ferretti C, Zara GP, Gigliotti CL, Ciamporcero ES, Pettazzoni P, Corti D, Pizzimenti S, Barrera G. GSH-targeted nanosponges increase doxorubicin-induced toxicity "in vitro" and "in vivo" in cancer cells with high antioxidant defenses. Free Radic Biol Med. 2016;97:24-37. [CrossRef]
- Colla R, Izzotti A, De Ciucis C, Fenoglio D, Ravera S, Speciale A, Ricciarelli R, Furfaro AL, Pulliero A, Passalacqua M, Traverso N, Pronzato MA, Domenicotti C, Marengo B. Glutathione-mediated antioxidant response and aerobic metabolism: two crucial factors involved in determining the multi-drug resistance of high-risk neuroblastoma. Oncotarget. 2016;7(43):70715-70737. [CrossRef]
- Cai Y, Zhao B, Liang Q, Zhang Y, Cai J, Li G. The selective effect of glycyrrhizin and glycyrrhetinic acid on topoisomerase IIα and apoptosis in combination with etoposide on triple negative breast cancer MDA-MB-231 cells. Eur J Pharmacol. 2017;809:87-97. [CrossRef]
- Deshpande NU, Jayakannan M. Cisplatin-Stitched Polysaccharide Vesicles for Synergistic Cancer Therapy of Triple Antagonistic Drugs. Biomacromolecules. 2017;18(1):113-126. [CrossRef]
- Rashmi R, Nedungadi D, Podder A, Mishra N, Bhuniya S. Monitoring of topoisomerase (I) inhibitor camptothecin release from endogenous redox-stimulated GO-polymer hybrid carrier. J Photochem Photobiol B. 2018;189:14-20. [CrossRef]
- Narayanan S, Gupta P, Nazim U, Ali M, Karadkhelkar N, Ahmad M, Chen ZS. Anti-cancer effect of Indanone-based thiazolyl hydrazone derivative on colon cancer cell lines. Int J Biochem Cell Biol. 2019;110:21-28. [CrossRef]
- Dilshara MG, Jayasooriya RGPT, Choi YH, Kim GY. Camptothecin induces c-Myc- and Sp1-mediated hTERT expression in LNCaP cells: Involvement of reactive oxygen species and PI3K/Akt. Food Chem Toxicol. 2019;127:53-60. [CrossRef]
- Sinha BK, van 't Erve TJ, Kumar A, Bortner CD, Motten AG, Mason RP. Synergistic enhancement of topotecan-induced cell death by ascorbic acid in human breast MCF-7 tumor cells. Free Radic Biol Med. 2017;113:406-412. [CrossRef]
- Sinha BK, Tokar EJ, Bushel PR. Elucidation of Mechanisms of Topotecan-Induced Cell Death in Human Breast MCF-7 Cancer Cells by Gene Expression Analysis. Front Genet. 2020;11:775. [CrossRef]
- Maszczyk M, Rzepka Z, Rok J, Beberok A, Wrześniok D. Neobavaisoflavone May Modulate the Activity of Topoisomerase Inhibitors towards U-87 MG Cells: An In Vitro Study. Molecules. 2021;26(15):4516. [CrossRef]
- Shi C, Zhang Z, Xu R, Zhang Y, Wang Z. Contribution of HIF-1α/BNIP3-mediated autophagy to lipid accumulation during irinotecan-induced liver injury. Sci Rep. 2023;13(1):6528. [CrossRef]
- Al-Karmalawy AA, Rashed M, Sharaky M, Abulkhair HS, Hammouda MM, Tawfik HO, Shaldam MA. Novel fused imidazotriazines acting as promising top. II inhibitors and apoptotic inducers with greater selectivity against head and neck tumors: Design, synthesis, and biological assessments. Eur J Med Chem. 2023;259:115661. [CrossRef]
- Nayak J, P SV, Sahoo SK, Kumar M, Vashistha VK, Kumar R. Computational insight of antioxidant and doxorubicin combination for effective cancer therapy. J Biomol Struct Dyn. 2023:1-9. [CrossRef]
- Liu S, Costa M. The role of NUPR1 in response to stress and cancer development. Toxicol Appl Pharmacol. 2022;454:116244. [CrossRef]
- Balaji S, Neupane R, Malla S, Khupse R, Amawi H, Kumari S, Tukaramrao DB, Chattopadhyay S, Ashby CR Jr, Boddu SHS, Karthikeyan C, Trivedi P, Raman D, Tiwari AK. IND-2, a Quinoline Derivative, Inhibits the Proliferation of Prostate Cancer Cells by Inducing Oxidative Stress, Apoptosis and Inhibiting Topoisomerase II. Life (Basel). 2022;12(11):1879. [CrossRef]
- Chen S, Chen J, Du W, Mickelsen DM, Shi H, Yu H, Kumar S, Yan C. PDE10A Inactivation Prevents Doxorubicin-Induced Cardiotoxicity and Tumor Growth. Circ Res. 2023;133(2):138-157. [CrossRef]
- Zhang W, Fan Z, Wang F, Yin L, Wu J, Li D, Song S, Wang X, Tang Y, Huang C. Tubeimoside I Ameliorates Doxorubicin-Induced Cardiotoxicity by Upregulating SIRT3. Oxid Med Cell Longev. 2023;2023:9966355. [CrossRef]
- Selvakumaran M, Pisarcik DA, Bao R, Yeung AT, Hamilton TC. Enhanced cisplatin cytotoxicity by disturbing the nucleotide excision repair pathway in ovarian cancer cell lines. Cancer Res. 2003;63(6):1311-6.
- Mountzios G, Dimopoulos MA, Papadimitriou C. Excision Repair Cross-Complementation Group 1 Enzyme as a Molecular Determinant of Responsiveness to Platinum-Based Chemotherapy for non Small-Cell Lung Cancer. Biomark Insights. 2008;3:219-226. [CrossRef]
- Curtin, NJ. DNA repair dysregulation from cancer driver to therapeutic target. Nat Rev Cancer. 2012;12(12):801-17. [CrossRef]
- Awwad, S. W., Serrano-Benitez, A., Thomas, J. C., Gupta, V., & Jackson, S. P. Revolutionizing DNA repair research and cancer therapy with CRISPR-Cas screens. Nat Rev Mol Cell Biol. 2023;24(7):477-494. [CrossRef]
- Rocha JC, Busatto FF, Guecheva TN, Saffi J. Role of nucleotide excision repair proteins in response to DNA damage induced by topoisomerase II inhibitors. Mutat Res Rev Mutat Res. 2016;768:68-77. [CrossRef]
- Carlsen L, El-Deiry WS. Anti-cancer immune responses to DNA damage response inhibitors: Molecular mechanisms and progress toward clinical translation. Front Oncol. 2022;12:998388. [CrossRef]
- Foo TK, Xia B. BRCA1-Dependent and Independent Recruitment of PALB2-BRCA2-RAD51 in the DNA Damage Response and Cancer. Cancer Res. 2022;82(18):3191-3197. [CrossRef]
- da Costa AABA, Chowdhury D, Shapiro GI, D'Andrea AD, Konstantinopoulos PA. Targeting replication stress in cancer therapy. Nat Rev Drug Discov. 2023;22(1):38-58. [CrossRef]
- Groelly FJ, Fawkes M, Dagg RA, Blackford AN, Tarsounas M. Targeting DNA damage response pathways in cancer. Nat Rev Cancer. 2023;23(2):78-94. [CrossRef]
- Saha LK, Saha S, Yang X, Huang SN, Sun Y, Jo U, Pommier Y. Replication-associated formation and repair of human topoisomerase IIIα cleavage complexes. Nat Commun. 2023;14(1):1925. [CrossRef]
- Dexheimer TS, Antony S, Marchand C, Pommier Y. Tyrosyl-DNA phosphodiesterase as a target for anticancer therapy. Anticancer Agents Med Chem. 2008;8(4):381-9. [CrossRef]
- Caldecott, KW. DNA single-strand break repair and human genetic disease. Trends Cell Biol. 2022;32(9):733-745. [CrossRef]
- Okamoto R, Takano H, Okamura T, Park JS, Tanimoto K, Sekikawa T, Yamamoto W, Sparreboom A, Verweij J, Nishiyama M. O(6)-methylguanine-DNA methyltransferase (MGMT) as a determinant of resistance to camptothecin derivatives. Jpn J Cancer Res. 2002;93(1):93-102. [CrossRef]
- Xu Y, Villalona-Calero MA. Irinotecan: mechanisms of tumor resistance and novel strategies for modulating its activity. Ann Oncol. 2002;13(12):1841-51. [CrossRef]
- Huang X, Okafuji M, Traganos F, Luther E, Holden E, Darzynkiewicz Z. Assessment of histone H2AX phosphorylation induced by DNA topoisomerase I and II inhibitors topotecan and mitoxantrone and by the DNA cross-linking agent cisplatin. Cytometry A. 2004;58(2):99-110. [CrossRef]
- Di Leo A, Claudino WM, Pestrin M, Licitra S, Biganzoli L. Using specific cytotoxics with a targeted mind. Breast. 2007;16 Suppl 2:S120-6. [CrossRef]
- Liu Y, Zhang J, Feng S, Zhao T, Li Z, Wang L, Wang P, Du H, Yuan S, Sun L. A Novel Camptothecin Derivative 3j Inhibits Nsclc Proliferation Via Induction of Cell Cycle Arrest By Topo I-Mediated DNA Damage. Anticancer Agents Med Chem. 2019;19(3):365-374. [CrossRef]
- Huang X, Kurose A, Tanaka T, Traganos F, Dai W, Darzynkiewicz Z. Activation of ATM and histone H2AX phosphorylation induced by mitoxantrone but not by topotecan is prevented by the antioxidant N-acetyl-L-cysteine. Cancer Biol Ther. 2006;5(8):959-64. [CrossRef]
- Zhao H, Traganos F, Darzynkiewicz Z. Phosphorylation of p53 on Ser15 during cell cycle caused by Topo I and Topo II inhibitors in relation to ATM and Chk2 activation. Cell Cycle. 2008;7(19):3048-55. [CrossRef]
- Peleg R, Bobilev D, Priel E. Topoisomerase I as a target of erlotinib and gefitinib: efficacy of combined treatments with camptothecin. Int J Oncol. 2014;44(3):934-42. [CrossRef]
- Ranganathan P, Kashyap T, Yu X, Meng X, Lai TH, McNeil B, Bhatnagar B, Shacham S, Kauffman M, Dorrance AM, Blum W, Sampath D, Landesman Y, Garzon R. XPO1 Inhibition using Selinexor Synergizes with Chemotherapy in Acute Myeloid Leukemia by Targeting DNA Repair and Restoring Topoisomerase IIα to the Nucleus. Clin Cancer Res. 2016;22(24):6142-6152. [CrossRef]
- Nateewattana J, Dutta S, Reabroi S, Saeeng R, Kasemsook S, Chairoungdua A, Weerachayaphorn J, Wongkham S, Piyachaturawat P. Induction of apoptosis in cholangiocarcinoma by an andrographolide analogue is mediated through topoisomerase II alpha inhibition. Eur J Pharmacol. 2014;723:148-55. [CrossRef]
- Yuan Z, Chen S, Chen C, Chen J, Chen C, Dai Q, Gao C, Jiang Y. Design, synthesis and biological evaluation of 4-amidobenzimidazole acridine derivatives as dual PARP and Topo inhibitors for cancer therapy. Eur J Med Chem. 2017;138:1135-1146. [CrossRef]
- de Camargo MS, De Grandis RA, da Silva MM, da Silva PB, Santoni MM, Eismann CE, Menegário AA, Cominetti MR, Zanelli CF, Pavan FR, Batista AA. Determination of in vitro absorption in Caco-2 monolayers of anticancer Ru(II)-based complexes acting as dual human topoisomerase and PARP inhibitors. Biometals. 2019;32(1):89-100. [CrossRef]
- Baglini E, Salerno S, Barresi E, Robello M, Da Settimo F, Taliani S, Marini AM. Multiple Topoisomerase I (TopoI), Topoisomerase II (TopoII) and Tyrosyl-DNA Phosphodiesterase (TDP) inhibitors in the development of anticancer drugs. Eur J Pharm Sci. 2021;156:105594. [CrossRef]
- Zhang H, Wang Y, Wang Y, Han Q, Yan H, Yang T, Guo Z, Wang X. Platinum complexes as inhibitors of DNA repair protein Ku70 and topoisomerase IIα in cancer cells. Dalton Trans. 2022;51(8):3188-3197. [CrossRef]
- Boichuk S, Bikinieva F, Mustafin I, Zykova S, Ryzkin S, Galembikova A. 2-Amino-Pyrrole-Carboxylate Attenuates Homology-Mediated DNA Repair and Sensitizes Cancer Cells to Doxorubicin. Biochemistry (Mosc). 2022. 87(5):391-399. [CrossRef]
- Collins A, Møller P, Gajski G, Vodenková S, Abdulwahed A, Anderson D, Bankoglu EE,Bonassi S, Boutet-Robinet E, Brunborg G, Chao C, Cooke MS et al. Measuring DNA modifications with the comet assay: a compendium of protocols. Nat Protoc. 2023 18(3):929-989. [CrossRef]
- Petrella G, Corsi F, Ciufolini G, Germini S, Capradossi F, Pelliccia A, Torino F, Ghibelli L, Cicero DO. Metabolic Reprogramming of Castration-Resistant Prostate Cancer Cells as a Response to Chemotherapy. Metabolites. 2022;13(1):65. [CrossRef]
- Chakraborty S, Balan M, Sabarwal A, Choueiri TK, Pal S. Metabolic reprogramming in renal cancer: Events of a metabolic disease. Biochim Biophys Acta Rev Cancer. 2021;1876(1):188559. [CrossRef]
- Chen M, Lan H, Yao S, Jin K, Chen Y. Metabolic Interventions in Tumor Immunity: Focus on Dual Pathway Inhibitors. Cancers (Basel). 2023;15(7):2043. [CrossRef]
- Xiang H, Yang R, Tu J, Xi Y, Yang S, Lv L, Zhai X, Zhu Y, Dong D, Tao X. Metabolic reprogramming of immune cells in pancreatic cancer progression. Biomed Pharmacother. 2023;157:113992. [CrossRef]
- Kang H, Kim H, Lee S, Youn H, Youn B. Role of Metabolic Reprogramming in Epithelial⁻Mesenchymal Transition (EMT). Int J Mol Sci. 2019;20(8):2042. [CrossRef]
- Naidoo J, Li BT, Schindler K, Page DB. What does the future hold fo immunotherapy in cancer? Ann Transl Med. 2016;4(9):177. [CrossRef]
- Vermeulen JF, Van Hecke W, Adriaansen EJM, Jansen MK, Bouma RG, Villacorta Hidalgo J, Fisch P, Broekhuizen R, Spliet WGM, Kool M, Bovenschen N. Prognostic relevance of tumor-infiltrating lymphocytes and immune checkpoints in pediatric medulloblastoma. Oncoimmunology. 2017;7(3):e1398877. [CrossRef]
- Toor SM, Nair VS, Decock J, Elkord E. Immune checkpoints in the tum microenvironment. Seminars in Cancer Biology. 2020;65(1-12). [CrossRef]
- Byun DJ, Wolchok JD, Rosenberg LM, Girotra M. Cancer immunotherapy – immune checkpoint blockade and associated endocrinopathies. Nat Rev Endocrinol. 2017;13(4):195-207. [CrossRef]
- Beyersdorf N, Kerkau T, Hünig T. CD28 co-stimulation in T-cell homeostasis: recent perspective. Immunotargets Ther. 2015;4:111-22. [CrossRef]
- Topalian SL, Sznol M, McDermott DF, Kluger HM, Carvajal RD, Sharfman WH, Brahmer JR, Lawrence DP, Atkins MB, Powderly JD, Leming PD, Lipson EJ, Puzanov I, Smith DC, Taube JM, Wigginton JM, Kollia GD, Gupta A, Pardoll DM, Sosman JA, Hodi FS. Survival, durable tumor remission, and long-term safety in patients with advanced melanoma receiving nivolumab. J Clin Oncol. 2014;32(10):1020-30. [CrossRef]
- Latchman Y, Wood CR, Chernova T, Chaudhary D, Borde M, Chernova I, Iwai Y, Long AJ, Brown JA, Nunes R, Greenfield EA, Bourque K, Boussiotis VA, Carter LL, Carreno BM, Malenkovich N, Nishimura H, Okazaki T, Honjo T, Sharpe AH, Freeman GJ. PD-L2 is a second ligand for PD-1 and inhibits T cell activation. Nat Immunol. 2001;2(3):261-8. [CrossRef]
- Topalian SL, Drake CG, Pardoll DM. Targeting the PD-1/B7-H1(PD-L1) pathway to activate anti-tumor immunity. Curr Opin Immunol. 2012;24(2):207-12. [CrossRef]
- Ramsay, AG. Immune checkpoint blockade immunotherapy to activate anti-tumou T-cell immunity. Br J Haematol. 2013;162(3):313-25. [CrossRef]
- Han Y, Liu D, Li L. PD-1/PD-L1 pathway: current researches in cancer. Am Cancer Res. 2020;10(3):727-742.
- Mishima S, Shitara K. Trastuzumab deruxtecan for the treatment of HER2-positiv gastric cancer. Expert Opin Biol Ther. 2021;21(7):825-830.
- Cai S, Chen Z, Wang Y, Wang M, Wu J, Tong Y, Chen L, Lu C, Yang H. Reducing PD-L1 expression with a self-assembled nanodrug: an alternative to PD-L1 antibody for enhanced chemo-immunotherapy. Theranostics. 2021;11(4):1970-1981. [CrossRef]
- Chung YM, Khan PP, Wang H, Tsai WB, Qiao Y, Yu B, Larrick JW, Hu MC. Sensitizing tumors to anti-PD-1 therapy by promoting NK and CD8+ T cells vi pharmacological activation of FOXO3. J Immunother Cancer. 2021;9(12):e002772. [CrossRef]
- Liu Q, Hua S, Wang X, Chen F, Gou S. The introduction of immunosuppresso (TDO inhibitor) significantly improved the efficacy of irinotecan in treating hepatocellular carcinoma. Cancer Immunol Immunother. 2021;70(2):497-508. [CrossRef]
- McKenzie JA, Mbofung RM, Malu S, Zhang M, Ashkin. Et al. The Effect of Topoisomerase I Inhibitors on the Efficacy of T-Cell-Based Cancer Immunotherapy. J Natl Cancer Inst. 2018;110(7):777-786. [CrossRef]
- Goldenberg DM, Sharkey RM. Sacituzumab govitecan, a novel, third-generation, antibody-drug conjugate (ADC) for cancer therapy. Expert Opin Biol The. 2020;20(8):871-885. [CrossRef]
- Bardia A, Hurvitz SA, Tolaney SM, Loirat D, Punie K et al. ASCENT Clinica Trial Investigators. Sacituzumab Govitecan in Metastatic Triple-Negative Breast Cancer. N Engl J Med. 2021;384(16):1529-1541. [CrossRef]
- Zhou Y, Bastian IN, Long MD, Dow M, Li W, Liu T. et al. Activation of NF-κ and p300/CBP potentiates cancer chemoimmunotherapy through induction of MHC-I antige presentation. Proc Natl Acad Sci U S A. 2021;118(8):e2025840118. [CrossRef]
- Hassan M, Trung V, Bedi D, Shaddox S, Gunturu D, Yates C, Datta P, Samuel T. Interference with pathways activated by topoisomerase inhibition alters the surface expression of PD-L1 and MHC I in colon cancer cells. Oncol Lett. 2022;25(1):41. [CrossRef]
- Hao X, Zhao B, Zhou W, Liu H, Fukumoto T, Gabrilovich D, Zhang R Sensitization of ovarian tumor to immune checkpoint blockade by boosting senescence-associated secretory phenotype. iScience. 2020;24(1):102016. [CrossRef]
- Vitiello A, Ferrara F, Lasala R, Zovi A. Precision Medicine in the Treatment of Locally Advanced or Metastatic Urothelial Cancer: New Molecular Targets and Pharmacological Therapies. Cancers (Basel). 2022;14(20):5167. [CrossRef]
- Bedi D, Henderson HJ, Manne U, Samuel T. Camptothecin Induces PD-L1 and Immunomodulatory Cytokines in Colon Cancer Cells. Medicines (Basel). 2019;6(2):51. [CrossRef]
- Wang Z, Chen J, Hu J, Zhang H, Xu F, He W, Wang X, Li M, Lu W, Zeng G, Zho P, Huang P, Chen S, Li W, Xia LP, Xia X. cGAS/STING axis mediates a topoisomerase II inhibitor-induced tumor immunogenicity. J Clin Invest. 2019;129(11):4850-4862. [CrossRef]
- Li X, Li Y, Zhao Z, Miao N, Liu G, Deng L, Wei S, Hou J. Immunogenicity o small-cell lung cancer associates with STING pathway activation and is enhanced by AT and TOP1 inhibition. Cancer Med. 2023;12(4):4864-4881.
- Mohan N, Jiang J, Dokmanovic M, Wu WJ. Trastuzumab-mediated cardiotoxicity current understanding, challenges, and frontiers. Antib Ther. 2018;1(1):13-17.
- Tan X, Fang P, Li K, You M, Cao Y, Xu H, Zhu X, Wang L, Wei X, Wen H, Li W Shi L, Sun X, Yu D, Zhu H, Wang Z, Liu D, Shen H, Zhou W, An M. A HER2-targeted antibody- novel DNA topoisomerase I inhibitor conjugate induces durable adaptive antitumor immunity by activating dendritic cells. MAbs. 2023;15(1):2220466.
- Liu L, Kshirsagar PG, Gautam SK, Gulati M, Wafa EI, Christiansen JC, White BM, Mallapragada SK, Wannemuehler MJ, Kumar S, Solheim JC, Batra SK, Salem AK, Narasimhan B, Jain M. Nanocarriers for pancreatic cancer imaging, treatments, and immunotherapies. Theranostics. 2022;12(3):1030-1060. [CrossRef]
- Cao J, Huang D, Peppas NA. Advanced engineered nanoparticulate platforms to address key biological barriers for delivering chemotherapeutic agents to target sites. Adv Drug Deliv Rev. 2020;167:170–188. [CrossRef]
- Bahmani B, Uehara M, Ordikhani F, Li XF, Jiang LW, Banouni N. et al. Ectopic high endothelial venules in pancreatic ductal adenocarcinoma: A unique site for targeted delivery. Ebiomedicine. 2018;38:79–88. [CrossRef]
- Zhao X, Li F, Li Y, Wang H, Ren H, Chen J. et al. Co-delivery of HIF1α siRN and Gemcitabine via biocompatible lipid-polymer hybrid nanoparticles for effective treatment of pancreatic cancer. Biomaterials. 2015;46:13–25.
- Mittal A, Chitkara D, Behrman SW, Mahato RI. Efficacy of Gemcitabine conjugated and miRNA-205 complexed micelles for treatment of advanced pancreatic cancer. Biomaterials. 2014;35:7077–87. [CrossRef]
- Li Y, Wang Z, Ajani JA, Song S. Drug resistance and Cancer stem cells. Cell Commun Signal. 2021;19(1):19. [CrossRef]
- Hsu JM, Xia W, Hsu YH, Chan LC, Yu WH, Cha JH, Chen CT, Liao HW, Kuo CW, Khoo KH, Hsu JL, Li CW, Lim SO, Chang SS, Chen YC, Ren GX, Hung MC. STT3-dependent PD-L1 accumulation on cancer stem cells promotes immune evasion. Na Commun. 2018;9(1):1908. [CrossRef]
- Schulten HJ, Hussein D. Array expression meta-analysis of cancer stem cell gene identifies upregulation of PODXL especially in DCC low expression meningiomas. PLoS One. 2019 May 13;14(5):e0215452. [CrossRef]
- Lee PJ, Ho CC, Ho H, Chen WJ, Lin CH, Lai YH, Juan YC, Chu WC, Lee JH, Su SF, hen HY, Chen JJW, Chang GC, Li KC, Yang PC, Chen HW. Tumor microenvironment- based screening repurposes drugs targeting cancer stem cells and cancer-associated fibroblasts. Theranostics. 2021;11(19):9667-9686. [CrossRef]
- Maroufi NF, Amiri M, Dizaji BF, Vahedian V, Akbarzadeh M, Roshanravan N, Haiaty S, Nouri M, Rashidi MR. Inhibitory effect of melatonin on hypoxia-induced vasculogenic mimicry via suppressing epithelial-mesenchymal transition (EMT) in breast cancer stem cells. Eur J Pharmacol. 2020;15;881:173282. [CrossRef]
- Nicolay NH, Rühle A, Perez RL, Trinh T, Sisombath S, Weber KJ, Schmezer P, Ho AD, Debus J, Saffrich R, Huber PE. Mesenchymal stem cells exhibit resistance to topoisomerase inhibition. Cancer Lett. 2016;374(1):75-84. [CrossRef]
- Hong Y, Sang M, Shang C, Xue YX, Liu YH. Quantitative analysis of topoisomerase II alpha and evaluation of its effects on cell proliferation and apoptosis in glioblastoma cancer stem cells. Neurosci Lett. 2012;518(2):138-43. [CrossRef]
- Peleg R, Romzova M, Kogan-Zviagin I, Apte RN, Priel E. Modification of topoisomerases in mammospheres derived from breast cancer cell line: clinical implications for combined treatments with tyrosine kinase inhibitors. BMC Cancer. 2014;3;14:910. [CrossRef]
- Conley SJ, Baker TL, Burnett JP, Theisen RL, Lazarus D, Peters CG, Clouthier SG, Eliasof S, Wicha MS. CRLX101, an investigational camptothecin-containing nanoparticle- drug conjugate, targets cancer stem cells and impedes resistance to antiangiogenic therapy in mouse models of breast cancer. Breast Cancer Res Treat. 2015;150(3):559-67. [CrossRef]
- Boichuk S, Dunaev P, Mustafin I, Mani S, Syuzov K, Valeeva E, Bikinieva F, Galembikova A. Infigratinib (BGJ 398), a Pan-FGFR Inhibitor, Targets P-Glycoprotein and Increases Chemotherapeutic-Induced Mortality of Multidrug-Resistant Tumor Cells. Biomedicines. 2022;10(3):601. [CrossRef]
- Wang H, Agarwal P, Zhao S, Xu RX, Yu J, Lu X, He X. Hyaluronic acid-decorated dual responsive nanoparticles of Pluronic F127, PLGA, and chitosan for targeted co-delivery of doxorubicin and irinotecan to eliminate cancer stem-like cells. Biomaterials. 2015;72:74-89. [CrossRef]
- Ngo SNT, Williams DB. Protective Effect of Isothiocyanates from Cruciferou Vegetables on Breast Cancer: Epidemiological and Preclinical Perspectives. Anticancer Agents Med Chem. 2021;21(11):1413-1430. [CrossRef]
- Capelôa T, Benyahia Z, Zampieri LX, Blackman MCNM, Sonveaux P. Metabolic and non-metabolic pathways that control cancer resistance to anthracyclines. Semin Cell Dev Biol. 2020;98:181-191. [CrossRef]
- Perez-Plasencia C, Duenas-Gonzalez A. Can the state of cancer chemotherapy resistance be reverted by epigenetic therapy? Mol Cancer. 2006;10;5:27.
- Guo L, Lee YT, Zhou Y, Huang Y. Targeting epigenetic regulatory machinery to overcome cancer therapy resistance. Semin Cancer Biol. 2022;83:487-502. [CrossRef]
- Xu X, Peng Q, Jiang X, Tan S, Yang Y, Yang W, Han Y, Chen Y, Oyang L, Lin J, Xia L, Peng M, Wu N, Tang Y, Li J, Liao Q, Zhou Y. Metabolic reprogramming and epigenetic modifications in cancer: from the impacts and mechanisms to the treatment potential. Exp Mol Med. 2023; 55(7):1357-1370. [CrossRef]
- Das C, Adhikari S, Bhattacharya A, Chakraborty S, Mondal P, Yadav SS, Adhikary S, Hunt CR, Yadav KK, Pandita S, Roy S, Tainer JA, Ahmed Z, Pandita TK. Epigenetic-Metabolic Interplay in the DNA Damage Response and Therapeutic Resistance of BreastCancer. Cancer Res. 2023;83(5):657-666.
- Jang JY, Sung B, Kim ND. Role of Induced Programmed Cell Death in the Chemopreventive Potential of Apigenin. Int J Mol Sci. 2022;23(7):3757. [CrossRef]
- Arts J, de Schepper S, Van Emelen K. Histone deacetylase inhibitors: from chromatin remodeling to experimental cancer therapeutics. Curr Med Chem. 2003;10(22):2343-50. [CrossRef]
- Seto E, Yoshida M. Erasers of histone acetylation: the histone deacetylase enzymes Cold Spring Harb Perspect Biol. 2014;6(4):a018713.
- Kim HJ, Bae SC. Histone deacetylase inhibitors: molecular mechanisms of action and clinical trials as anti-cancer drugs. Am J Transl Res. 2011;3(2):166-79.
- Tiffon C, Adams J, van der Fits L, Wen S, Townsend P, Ganesan A, Hodges E, Vermeer M, Packham G. The histone deacetylase inhibitors vorinostat and romidepsin downmodulate IL-10 expression in cutaneous T-cell lymphoma cells. Br J Pharmacol. 2011;162(7):1590-602. [CrossRef]
- Kaewpiboon C, Srisuttee R, Malilas W, Moon J, Oh S, Jeong HG, Johnston RN, Assavalapsakul W, Chung YH. Upregulation of Stat1-HDAC4 confers resistance to etoposide through enhanced multidrug resistance 1 expression in human A549 lung cancer cells. Mol Med Rep. 2015;11(3):2315-21. [CrossRef]
- Wasim L, Chopra M. Panobinostat induces apoptosis via production of reactive oxygen species and synergizes with topoisomerase inhibitors in cervical cancer cells. Biomed Pharmacother. 2016;84:1393-1405. [CrossRef]
- Sampson VB, Vetter NS, Kamara DF, Collier AB, Gresh RC, Kolb EA. Vorinostat Enhances Cytotoxicity of SN-38 and Temozolomide in Ewing Sarcoma Cells and Activates STAT3/AKT/MAPK Pathways. PLoS One. 2015;10(11):e0142704. [CrossRef]
- Meisenberg C, Ashour ME, El-Shafie L, Liao C, Hodgson A, Pilborough A, Khurram SA, Downs JA, Ward SE, El-Khamisy SF. Epigenetic changes in histone acetylation underpin resistance to the topoisomerase I inhibitor irinotecan. Nucleic Acids Res. 2017;45(3):1159-1176. [CrossRef]
- He S, Dong G, Wang Z, Chen W, Huang Y, Li Z, Jiang Y, Liu N, Yao J, Miao Z, Zhang W, Sheng C. Discovery of Novel Multiacting Topoisomerase I/II and Histone Deacetylase Inhibitors. ACS Med Chem Lett. 2015;6(3):239-43. [CrossRef]
- Cincinelli R, Musso L, Artali R, Guglielmi M, Bianchino E, Cardile F, Colelli F, Pisano C, Dallavalle S. Camptothecin-psammaplin A hybrids as topoisomerase I and HDAC dual-action inhibitors. Eur J Med Chem. 2018;143:2005-2014. [CrossRef]
- Yamashita M, Tahara T, Hayakawa S, Matsumoto H, Wada SI, Tomioka K, Iida A. Synthesis and biological evaluation of histone deacetylase and DNA topoisomerase II- Targeted inhibitors. Bioorg Med Chem. 2018;26(8):1920-1928. [CrossRef]
- Chen J, Li D, Li W, Yin J, Zhang Y, Yuan Z, Gao C, Liu F, Jiang Y. Design synthesis and anticancer evaluation of acridine hydroxamic acid derivatives as dual Topo and HDAC inhibitors. Bioorg Med Chem. 2018;26(14):3958-3966. [CrossRef]
- Liu T, Wan Y, Xiao Y, Xia C, Duan G. Dual-Target Inhibitors Based on HDACs: Novel Antitumor Agents for Cancer Therapy. J Med Chem. 2020;63(17):8977-9002. [CrossRef]
- Marx C, Sonnemann J, Beyer M, Maddocks ODK, Lilla S, Hauzenberger I, Piée-Staffa A, Siniuk K, Nunna S, Marx-Blümel L, Westermann M, Wagner T, Meyer FB, Thierbach R,Mullins CS, Kdimati S, Linnebacher M, Neri F, Heinzel T, Wang ZQ, Krämer OH. Mechanistic insights into p53-regulated cytotoxicity of combined entinostat and irinotecan against colorectal cancer cells. Mol Oncol. 2021;15(12):3404-3429.
- Wu S, Huang Y, Wang T, Li K, Lu J, Huang M, Dong G, Sheng C. Evodiamine-Inspired Topoisomerase-Histone Deacetylase Dual Inhibitors: Novel Orally Active Antitumor Agents for Leukemia Therapy. J Med Chem. 2022;65(6):4818-4831. [CrossRef]
- Zhu F, Meng X, Liang H, Sheng C, Dong G, Liu D, Wu S. Design, Synthesis, and Structure-Activity relationships of Evodiamine-Based topoisomerase (Top)/Histone deacetylase (HDAC) dual inhibitors. Bioorg Chem. 2022;122:105702.
- El-Kalyoubi S, Elbaramawi SS, Eissa AG, Al-Ageeli E, Hobani YH, El-Sharkawy AA, Mohamed HT, Al-Karmalawy AA, Abulkhair HS. Design and synthesis of novel uracil- linked Schiff bases as dual histone deacetylase type II/topoisomerase type I inhibitors with apoptotic potential. Future Med Chem. 2023;15(11):937-958. [CrossRef]
- Kim HY, Choi SA, Koh EJ, Kim KH, Phi JH, Lee JY, Kim SK. Combination Treatment of CI-994 With Etoposide Potentiates Anticancer Effects Through a Topoisomerase II- Dependent Mechanism in Atypical Teratoid/Rhabdoid Tumor (AT/RT). Front Oncol. 2021;11:648023. [CrossRef]
- Zhao M, Yang K, Zhu X, Gao T, Yu W, Liu H, You Z, Liu Z, Qiao X, Song Y. Design, synthesis and biological evaluation of dual Topo II/HDAC inhibitors bearing pyrimido[5,4-b]indole and pyrazolo[3,4-d]pyrimidine motifs. Eur J Med Chem 2023;252:115303. [CrossRef]
- Flor AC, Doshi AP, Kron SJ. Modulation of therapy-induced senescence by reactive lipid aldehydes. Cell Death Discov. 2016;2:16045. [CrossRef]
- Ogiso Y, Tomida A, Lei S, Omura S, Tsuruo T. Proteasome inhibition circumvents olid tumor resistance to topoisomerase II-directed drugs. Cancer Res. 2000;60(9):242 34.
- Drummond CJ, Finlay GJ, Broome L, Marshall ES, Richardson E, Baguley BC. Action of SN 28049, a new DNA binding topoisomerase II-directed antitumour drug: comparison with doxorubicin and etoposide. Invest New Drugs. 2011;29(5):1102-10. [CrossRef]
- Polewska J, Skwarska A, Augustin E, Konaopa J. DNA-damaging imidazoacridinone C- 1311 induces autophagy followed by irreversible growth arrest and senescence in human lung cancer cells. J Pharmacol Exp Ther. 2013;346(3):393-405. [CrossRef]
- Huang RX, Zhou PK. DNA damage response signaling pathways and targets for radiotherapy sensitization in cancer. Signal Transduct Target Ther. 2020 May 1;5(1):60. [CrossRef]
- Courapied S, Cherier J, Vigneron A, Troadec MB, Giraud S, Valo I, Prigent C, Gamelin E, Coqueret O, Barré B. Regulation of the Aurora-A gene following topoisomerase I inhibition: implication of the Myc transcription factor. Mol Cancer., 2010;9:205. [CrossRef]
- Taschner-Mandl S, Schwarz M, Blaha J, Kauer M, Kromp F, Frank N, Rifatbegovic F, Weiss T, Ladenstein R, Hohenegger M, Ambros IM, Ambros PF. Metronomic topotecan impedes tumor growth of MYCN-amplified neuroblastoma cells in vitro and in vivo by therapy induced senescence. Oncotarget. 2016;7(3):3571-86.
- Hao L, Rohani N, Zhao RT, Pulver EM, Mak H, Kelada OJ, Ko H, Fleming HE, Gertler FB, Bhatia SN. Microenvironment-triggered multimodal precision diagnostics. Nat Mater. 2021;20(10):1440-1448. [CrossRef]
- Marx OM, Mankarious MM, Eshelman MA, Ding W, Koltun WA, Yochum GS. Transcriptome Analyses Identify Deregulated MYC in Early Onset Colorectal Cancer. Biomolecules, 2022;12(9):1223. [CrossRef]
- Saleh T, Carpenter VJ, Tyutyunyk-Massey L, Murray G, Leverson JD, Souers AJ, Alotaibi MR, Faber AC, Reed J, Harada H, Gewirtz DA. Clearance of therapy-induced senescent tumor cells by the senolytic ABT-263 via interference with BCL-XL –BAX interaction. Mol Oncol. 2023;14(10):2504-2519.
- Meredith AM, Dass CR. Increasing role of the cancer chemotherapeutic doxorubicin in cellular metabolism. J Pharm Pharmacol, 2016;68(6):729-41. [CrossRef]
- Lee KA, Flores RR, Jang IH, Saathoff A, Robbins PD. Immune Senescence, Immunosenescence and Aging. Front Aging. 2022;3:900028. [CrossRef]
- Vulpis E, Cuollo L, Borrelli C, Antonangeli F, Masuelli L, Cippitelli M, Fionda C, Caracciolo G, Petrucci MT, Santoni A, Zingoni A, Soriani A. Doxorubicin-Mediated miR-433 Expression on Exosomes Promotes Bystander Senescence in Multiple Myeloma Cells in a DDR-Independent Manner. Int J Mol Sci. 2023;24(7):6862. [CrossRef]
- Takigawa N, Vaziri SA, Grabowski DR, Chikamori K, Rybicki LR, Bukowski RM, Ganapathi MK, Ganapathi R, Mekhail T. Proteasome inhibition with bortezomib enhances activity of topoisomerase I-targeting drugs by NF-kappaB-independent mechanisms. Anticancer Res. 2006;26(3A):1869-76.
- Rudolf E, John S, Cervinka M. Irinotecan induces senescence and apoptosis in colonic cells in vitro. Toxicol Lett. 2012. 214(1):1-8. [CrossRef]
- Gewirtz, DA. Growth arrest and cell death in the breast tumor cell in response to ionizing radiation and chemotherapeutic agents which induce DNA damage. Breast Cancer Res Treat. 2000. 62(3):223-35. [CrossRef]
- Han H, Bearss DJ, Browne LW, Calaluce R, Nagle RB, Von Hoff DD. Identification of differentially expressed genes in pancreatic cancer cells using cDNA microarray. Cancer Res. 2002 ;62(10):2890-6. 15 May.
- Elmore JG, Armstrong K, Lehman CD, Fletcher SW. Screening for breast cancer JAMA. 2005;293(10):1245-56.
- Guichard G, Larré S, Gallina A, Lazar A, Faucon H, Chemama S, Allory Y, Patar J, Vordos D, Hoznek A, Yiou R, Salomon L, Abbou CC, De La Taille A. Extended 21- Sample Needle Biopsy Protocol for diagnosis of prostate cancer in 1000 consecutive patients. Elsevier BV; 2007;52(2):430–435. [CrossRef]
- Gleyzer N, Scarpulla RC. Activation of a PGC-1-related coactivator (PRC)-dependent inflammatory stress program linked to apoptosis and premature senescence. J Biol Chem. 2013;288(12):8004-8015. [CrossRef]
- Yeo SK, Wen J, Chen S, Guan J. Autophagy differentially regulates distinct breast cancer stem-like cells in murine models via EGFR/STAT3 and TGFΒ/SMAD signaling. American Association for Cancer Research; 2016;76(11):3397–3410. [CrossRef]
- Chen S, Zhao Y, Liu S, Zhang J, Assaraf YG, Cui W, Wang L. Epigenetic enzyme mutations as mediators of anti-cancer drug resistance. Drug Resist Updat. 2022;61:100821. [CrossRef]
- Zhang X, Dang CV. Time to hit pause on mitochondria-targeting cancer therapies Nat Med. 2023;29(1):29-30.
- Çalışkan M, Güler H, Bozok Çetintaş V. Current updates on microRNAs a regulators of chemoresistance. Biomed Pharmacother. 2017;95:1000-1012.
- Torki Z, Ghavi D, Hashemi S, Rahmati Y, Rahmanpour D, Pornour M, Alivand MR. The related miRNAs involved in doxorubicin resistance or sensitivity of various cancers: an update. Cancer Chemother Pharmacol. 2021;88(5):771-793. [CrossRef]
- Taheri M, Mahmud Hussen B, Tondro Anamag F, Shoorei H, Dinger ME, Ghafouri-Fard S. The role of miRNAs and lncRNAs in conferring resistance to doxorubicin. J Drug Target. 2022;30(1):1-21. [CrossRef]
- Sritharan S, Guha S, Hazarika S, Sivalingam N. Meta analysis of bioactive compounds, miRNA, siRNA and cell death regulators as sensitizers to doxorubicin induced chemoresistance. Apoptosis. 2022;27(9-10):622-646. [CrossRef]
- Sun M, Zhang Q, Yang X, Qian SY, Guo B. Vitamin D Enhances the Efficacy of Irinotecan through miR-627-Mediated Inhibition of Intratumoral Drug Metabolism. Mol Cancer Ther. 2016;15(9):2086-95. [CrossRef]
- Carvajal-Moreno J, Hernandez VA, Wang X, Li J, Yalowich JC, Elton TS. Effects of hsa- miR-9-3p and hsa-miR-9-5p on Topoisomerase IIβ Expression in Human Leukemia K562 Cells with Acquired Resistance to Etoposide. J Pharmacol Exp Ther. 2023;384(2):265-276.
- Kubiliūtė R, Šulskytė I, Daniūnaitė K, Daugelavičius R, Jarmalaitė S. Molecular features of doxorubicin-resistance development in colorectal cancer CX-1 cell line. Medicina (Kaunas). 2016;52(5):298-306. [CrossRef]
- Mirzaei S, Abadi AJ, Gholami MH, Hashemi F, Zabolian A, Hushmandi K, Zarrabi A, Entezari M, Aref AR, Khan H, Ashrafizadeh M, Samarghandian S. The involvement of epithelial-to-mesenchymal transition in doxorubicin resistance: Possible molecular targets. Eur J Pharmacol. 2021;908:174344. [CrossRef]
- Zhou Q, Abraham AD, Li L, Babalmorad A, Bagby S, Arcaroli JJ, Hansen RJ, Valeriote FA, Gustafson DL, Schaack J, Messersmith WA, LaBarbera DV. Topoisomerase Iiα mediates TCF-dependent epithelial-mesenchymal transition in colon cancer. Oncogene. 2016;35(38):4990-9. [CrossRef]
- Abraham AD, Esquer H, Zhou Q, Tomlinson N, Hamill BD, Abbott JM, Li L, Pike LA, Rinaldetti S, Ramirez DA, Lunghofer PJ, Gomez JD, Schaack J, Nemkov T, D'Alessandro A, Hansen KC, Gustafson DL, Messersmith WA, LaBarbera DV. Drug Design Targeting T-Cell Factor-Driven Epithelial-Mesenchymal Transition as a Therapeutic Strategy for Colorectal Cancer. J Med Chem. 2019;62(22):10182-10203. [CrossRef]
- Davis JT, Ghosh TM, Mazumder S, Mitra A, Bird RC, Arnold RD. Extended Exposure Topotecan Significantly Improves Long-Term Drug Sensitivity by Decreasing Malignant Cell Heterogeneity and by Preventing Epithelial-Mesenchymal Transition. Int J Mol Sci. 2023;24(10):8490. [CrossRef]
- Ohtsuka K, Ohnìshì H, Morii T, Fujiwara M, Kishino T, Ogura W, Chiba M, Matsushima S, Goya T, Watanabe T. Downregulated ABCG2 enhances sensitivity to topoisomerase I inhibitor in epidermal growth factor receptor tyrosine kinase Inhibitor-Resistant non- small cell lung cancer. Elsevier BV; 2010;5(11):1726–1733. [CrossRef]
- Goswami S, Sharma-Walia N. Osteoprotegerin secreted by inflammatory and invasive breast cancer cells induces aneuploidy, cell proliferation and angiogenesis. BMC Cancer. 2015;15:935. [CrossRef]
- Bentires-Alj M, Barbu V, Fillet M, Chariot A, Relic B, Jacobs N, Gielen J, Merville MP, Bours V. NF-kappaB transcription factor induces drug resistance through MDR1 expression in cancer cells. Oncogene. 2003 Jan 9;22(1):90-7.
- Zhang X, Linder S, Bazzaro M. Drug Development Targeting the Ubiquitin-Proteasome System (UPS) for the Treatment of Human Cancers. Cancers (Basel). 2020 Apr 7;12(4):902. [CrossRef]
- Żabka A, Winnicki K, Polit JT, Maszewski J. The effects of anti-DNA topoisomerase II drugs, etoposide and ellipticine, are modified in root meristem cells of Allium cepa by MG132, an inhibitor of 26S proteasomes. Plant Physiol Biochem. 2015;96:72-82. [CrossRef]
- Sinha BK, Perera L, Cannon RE. Reversal of drug resistance by JS-K and nitric oxide in ABCB1- and ABCG2-expressing multi-drug resistant human tumor cells. Biomed Pharmacother. 2019. 120:109468. [CrossRef]
- Sinha BK, Perera L, Cannon RE. NCX-4040, a Unique Nitric Oxide Donor, Induces Reversal of Drug-Resistance in Both ABCB1- and ABCG2-Expressing Multidrug Human Cancer Cells. Cancers (Basel). 2021. 13(7):1680. [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).



