Submitted:
13 December 2023
Posted:
14 December 2023
You are already at the latest version
Abstract
Keywords:
Introduction
Overview of the guidance development process
Aims, scope and target of the guidance
Need for updated guidance on the handling of GTMPs
Review of existing guidance
General operating procedures for handling GTMPs in hospital pharmacy
Handling of GTMPs and patient specimens
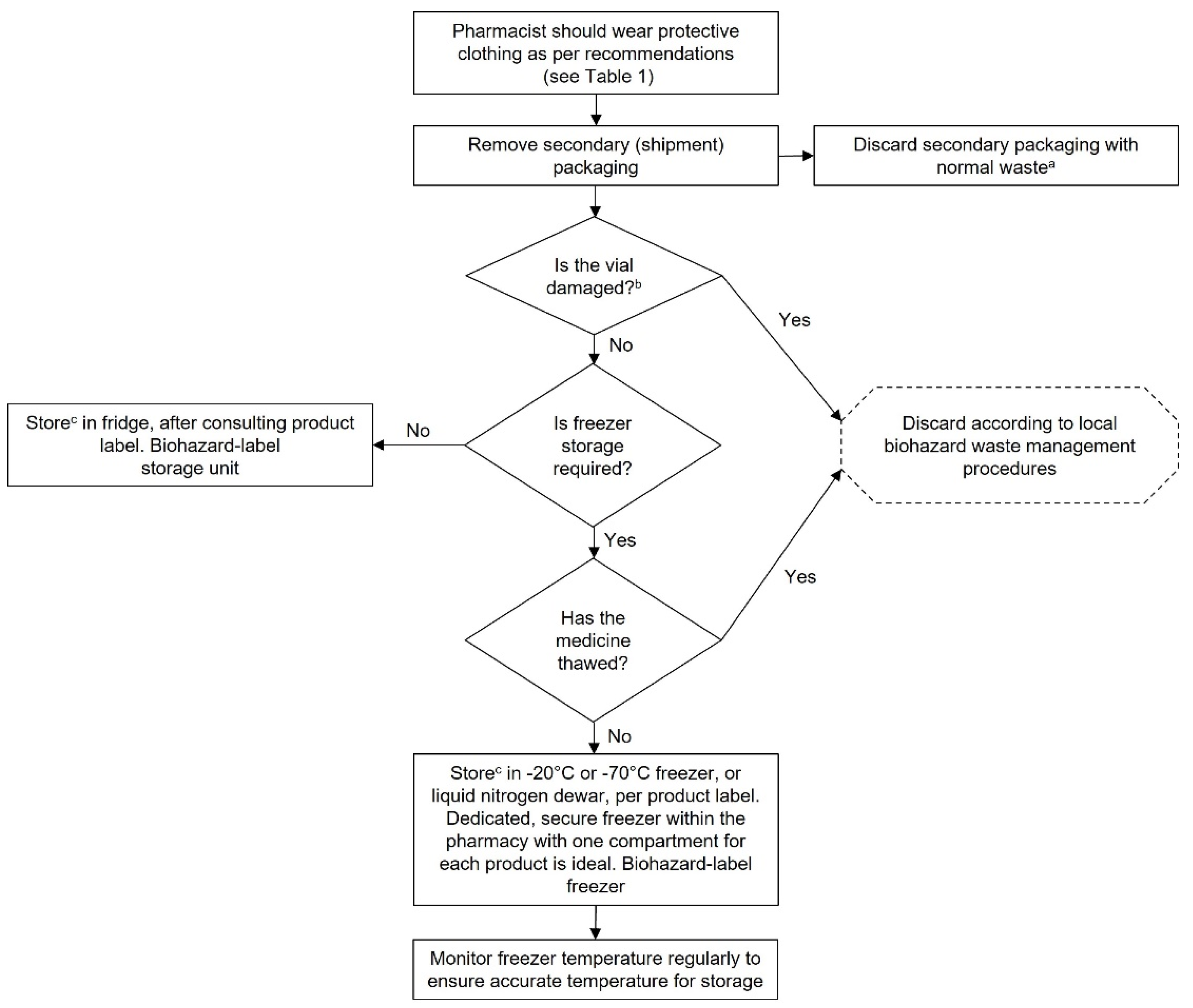
Receipt and Storage of GTMPs
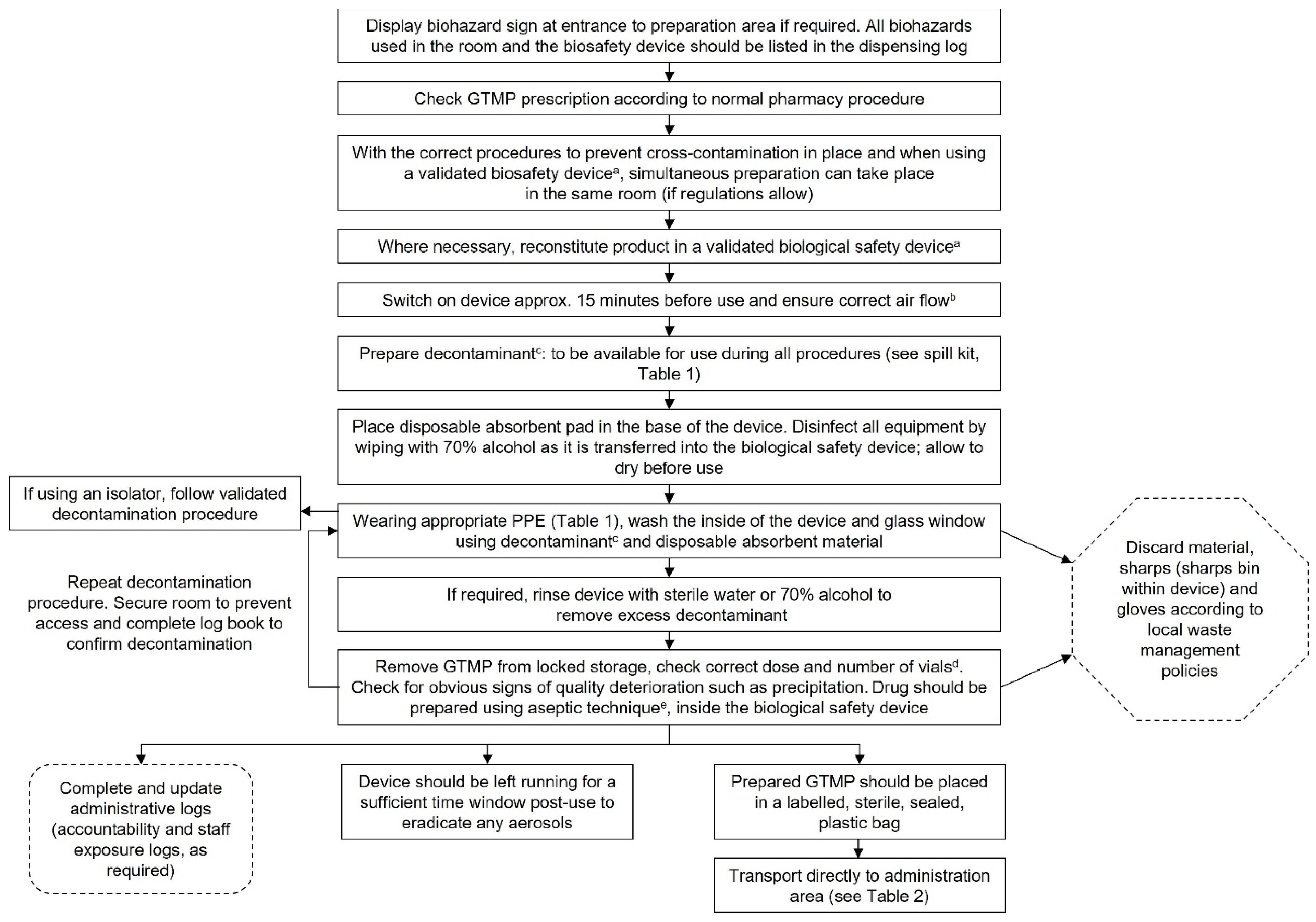
Reconstitution and dispensing of GTMPs
Transportation of GTMPs
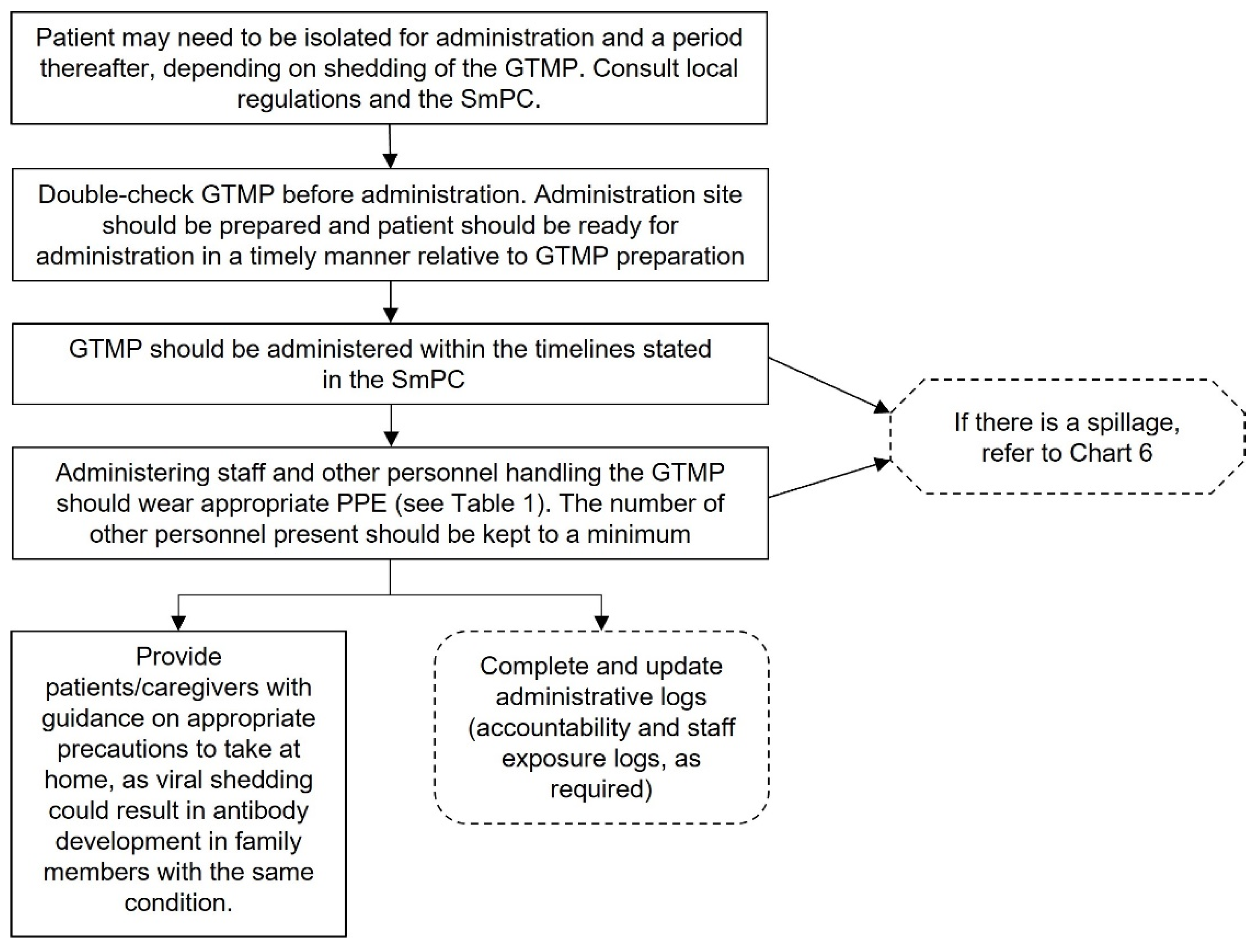
Administering GTMPs
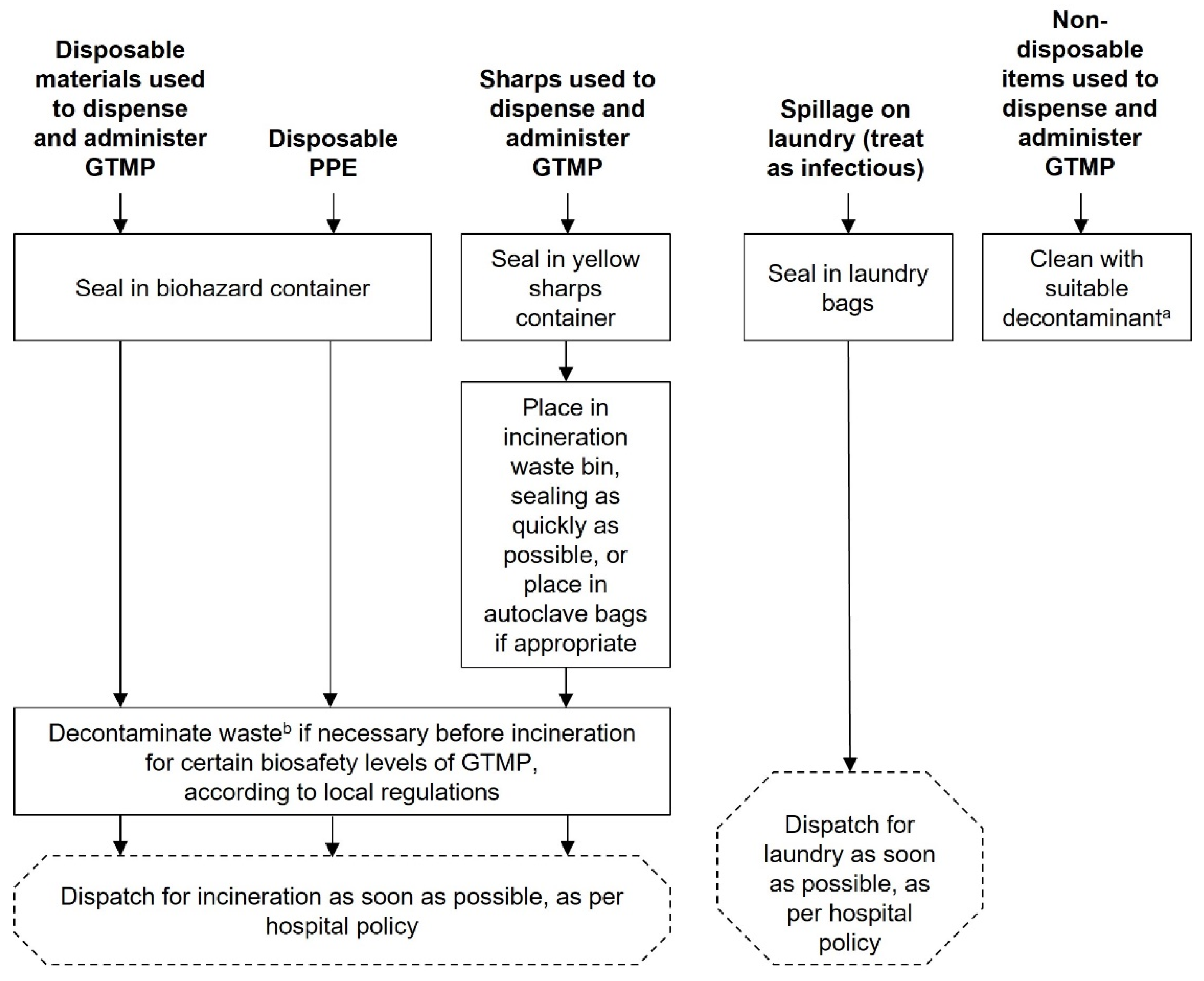
GTMP waste disposal
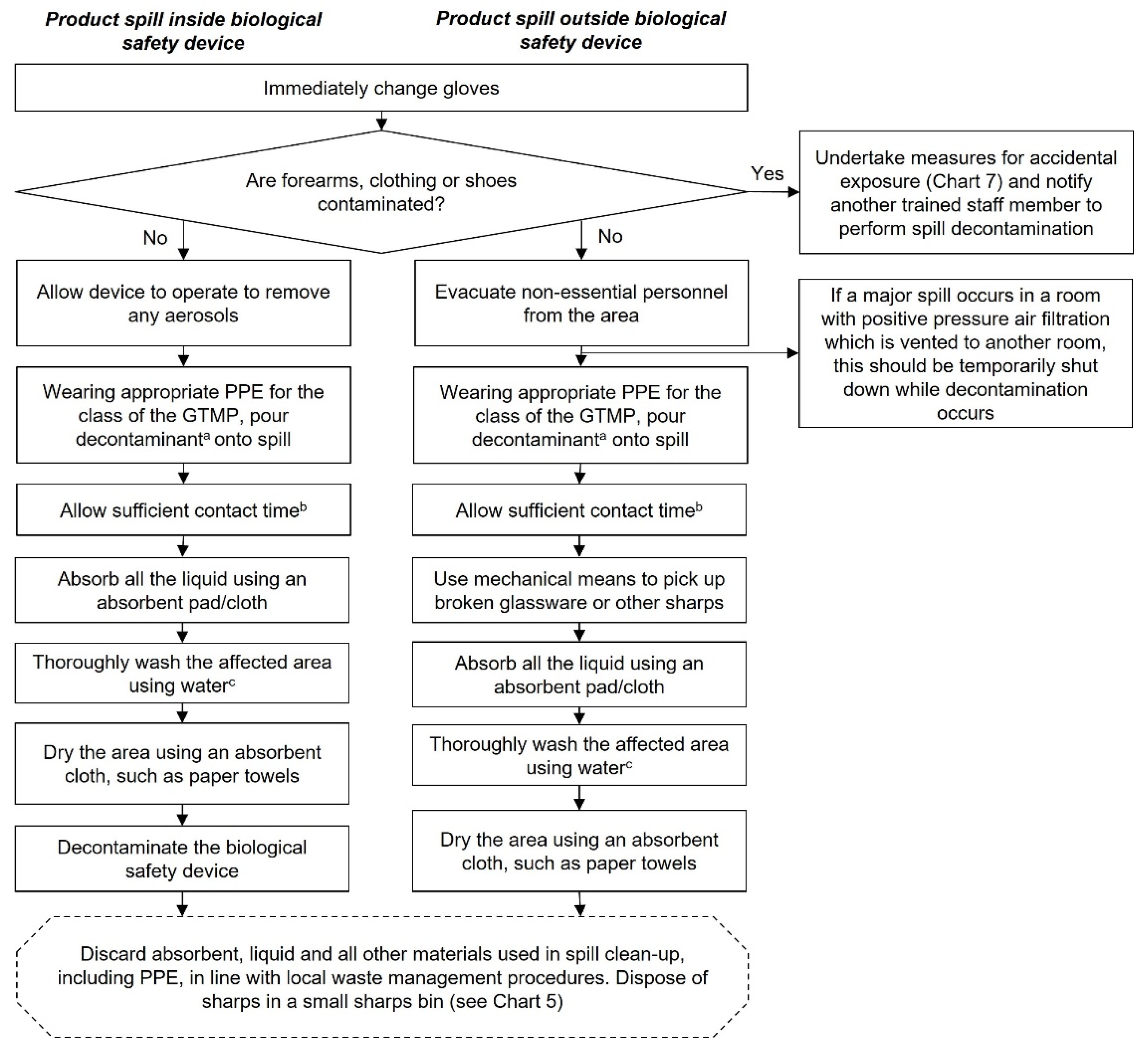
Decontamination of GTMP spills
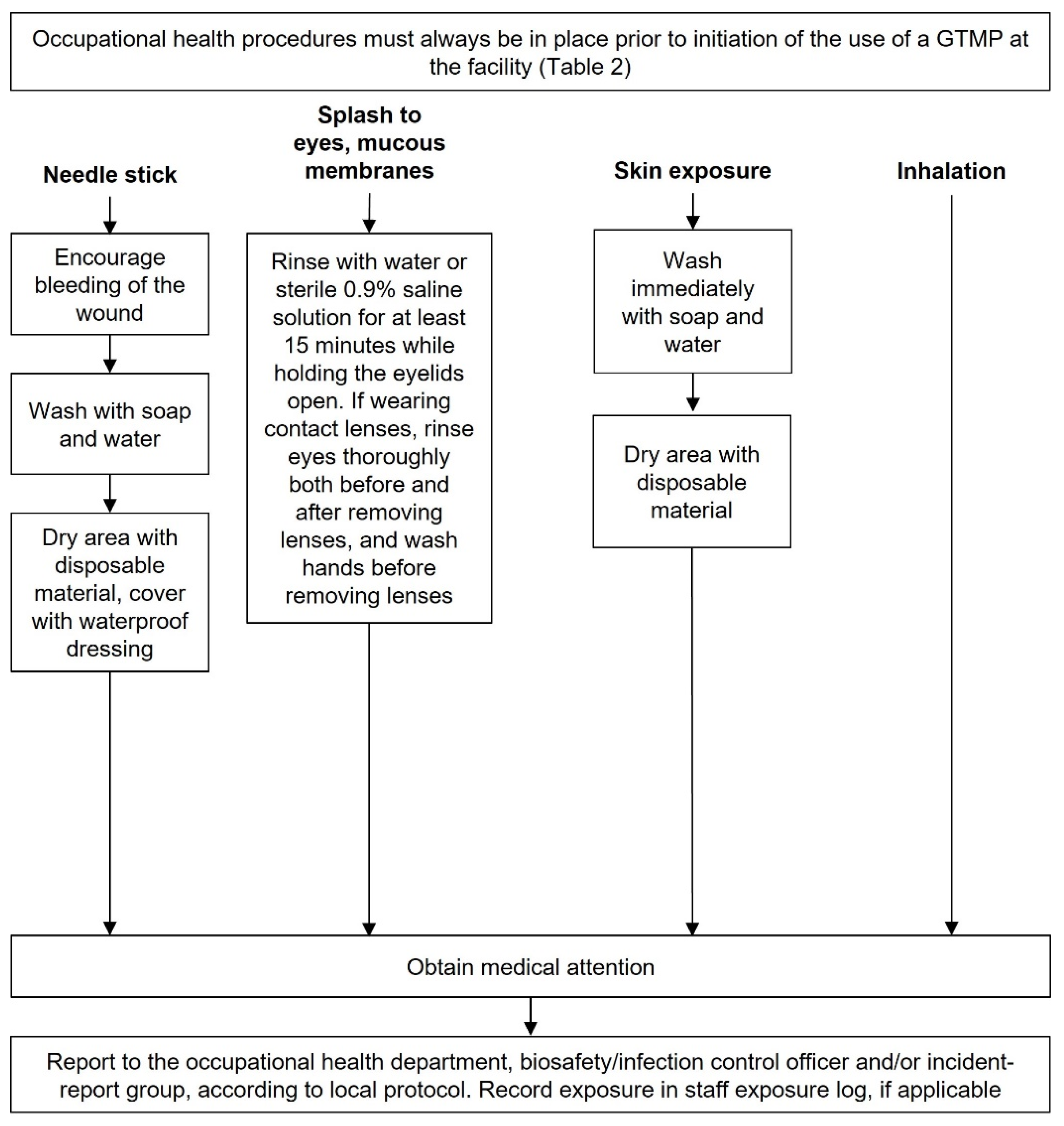
Accidental exposure to GTMPs
Discussion
Acknowledgements and funding support
Competing interest statement
Copyright statement
Appendices
| Term | Definition |
| Accidental exposure | Accidental release of the GTMP from containment, resulting in unintended exposure of staff or the public to the agent. |
| Administration | The process of treating the patient with the GTMP, from receipt of the GTMP by clinical staff up to the point where the patient leaves the hospital and all waste has been disposed of safely. |
| Advanced therapy medicinal product (ATMP) | A GTMP, somatic cell therapy medicinal product, or a tissue engineered product (5). |
| Biosafety | The safe working practices required for the handling of biological materials, especially infectious agents. |
| Biosafety level 1 | The containment level required for a microorganism that is unlikely to cause human or animal disease (no or low individual and community risk; risk group 1). Often no biosafety device is required for this level; check local regulations (8). |
| Biosafety level 2 | The containment level required for a pathogen that can cause human or animal disease, but is unlikely to be a serious hazard. Laboratory exposure could cause serious infection, but effective treatment and prevention measures are available and the risk of spread is limited (moderate individual risk, low community risk; risk group 2). Biosafety device, PPE and biohazard sign usually required; check local regulations (8). |
| Biosafety level 3 | The containment level required for a pathogen that causes serious human or animal disease but does not usually spread between individuals. Effective treatment and preventative measures are available (high individual risk, low community risk; risk group 3). Biosafety device, PPE, biohazard sign, controlled access and directional airflow usually required; check local regulations (8). |
| Biosafety level 4 | The containment level required for a pathogen that usually causes serious human or animal disease and is easily transmissible. Effective treatment and preventative measures are not usually available (high individual and community risk; risk group 4). Highly restricted access with airlock entry, shower exit and specialist waste disposal usually required. Specialised PPE and/or class 3 biosafety devices may be required, as well as additional safety measures; check local requirements (8). |
| Containment | The culturing, storage, transport, destruction, disposal, or any other use of a genetically modified organism within the bounds of physical, chemical and/or biological barriers to limit their contact with people and the environment (17) . |
| Decontamination | Any process for removing/killing microorganisms (8). |
| Disinfection | A physical or chemical means of killing microorganisms, but not necessarily spores (8). |
| Genetically modified organism (GMO) | An organism that has undergone genetic modification. |
| Genetic modification | Occurs when the genetic material of an organism has been altered in a way that does not occur naturally (either by mating or natural recombination), and uses recombinant nucleic acid techniques to form new combinations of genetic material (17). |
| Gene therapy | Treatment or prophylaxis of disease by the deliberate introduction of genetic material into the isolated cells of a patient (ex vivo) or directly into the patient (in vivo) (6). |
| Gene therapy medicinal product (GTMP) | A GTMP must fulfil two conditions: 1) the product must be a biological medicinal product and contain recombinant nucleic acid(s) and 2) the recombinant nucleic acid(s) should be directly involved in the mechanism of action and hence therapeutic action of the product (5). |
| Handling | Includes storage, dispensing and reconstitution, transportation, administration, waste disposal, spills and accidental exposure, and any other process where the GTMP is in use. |
| Naked DNA | DNA that is free in solution, not packaged in a vector. |
| Spill, majora | More than 5 mL or 5 g of a substance. |
| Spill, minora | Less than 5 mL or 5 g of a substance. |
| Storage | Containment of a GTMP when it is not in use. |
| Reconstitution | The process of making the GTMP ready-to-administer. Also referred to as preparation (6). |
| Transportation | Movement of the GTMP around and/or between hospitals using hospital transport. NOT courier transport. |
| GTMP | Manufacturer | Indication |
in vivo/ ex vivo |
Viral vector | Biosafety level of vectorb (28) |
| Glybera (alipogene tiparvovec)a (32) | uniQure biopharma | Familial lipoprotein lipase deficiency | in vivo | AAV-1 | 1 |
| Imlygic (talimogene laherparepvec) (33) | Amgen/IDT Biologika | Unresectable, metastatic melanoma | in vivo | HSV-1 | 1/2d |
| Strimvelisc (34) | AGC Biologics | ADA-SCID | ex vivo | Retroviral | 2 |
| Yescarta (axicabtagene ciloleucel)c (35) | Kite Pharma | DLBCL, HGBL, PMBCL, FL | ex vivo | Retroviral | 2 |
| Kymriah (tisagenlecleucel)c (36) | Novartis | ALL, DLBCL, FL | ex vivo | Lentiviral | 2 |
| Luxturna (voretigene neparvovec) (37) | Spark Therapeutics/Novartis | Inherited retinal dystrophy | in vivo | AAV-2 | 1 |
| Zynteglo (betibeglogene autotemcel)a,c (38) | Minaris Regenerative Medicine | TDT | ex vivo | Lentiviral | 2 |
| Zolgensma (onasemnogene abeparvovec) (39) | Novartis/Almac Pharma Services | 5q SMA | in vivo | AAV-9 | 1 |
| Libmeldy (atidarsagene autotemcel)c (40) | AGC Biologics | ARSA-mutant MLD | ex vivo | Lentiviral | 2 |
| Tecartus (brexucabtagene autoleucel)c (41) | Kite Pharma | MCL, ALL | ex vivo | Retroviral | 2 |
| Skysona (elivaldogene autotemcel)a,c (42) | Minaris Regenerative Medicine | ABCD1-mutant early cerebral adrenoleukodystrophy | ex vivo | Lentiviral | 2 |
| Abecma (idecabtagene vicleucel)c (43) | Celgene | Multiple myeloma | ex vivo | Lentiviral | 2 |
| Breyanzi (lisocabtagene maraleucel)c (44) | Juno Therapeutics/Celgene | DLBCL, PMBCL, FL | ex vivo | Lentiviral | 2 |
| Carvykti (ciltacaptagene autoleucel)c (45) | Janssen | Multiple myeloma | ex vivo | Lentiviral | 2 |
| Upstaza (eladocagene exuparvovec) (46) | MassBiologics South Coast/Almac Pharma Services | AADC deficiency | in vivo | AAV-2 | 1 |
| Roctavian (valoctocogene roxaparvovec) (47) | BioMarin | Haemophilia A | in vivo | AAV-5 | 1 |
| Hemgenix (etranacogene dezaparvovec) (48) | CSL Behring | Haemophilia B | in vivo | AAV-5 | 1 |
| Containment measure | Level 1 | Level 2 |
| Autoclave | Required on site | Required in the building |
| Access restricted to authorised personnel | Not required | Required |
| Measures to control aerosol dissemination | Not required | Required |
| PPE | Required | Required |
| Specified disinfection procedures | Required | Required |
| Safe storage of GTMP | Required | Required |
| Inactivation of GTMP in contaminated material and waste | Required by validated means | Required by validated means |
| Handling stage | Chief pharmacist | Hospital pharmacy staff | Physician | Theatre/ward nurse | Biosafety officera/hygiene services/infection control | Occupational health | Cleaners | Porters | Waste services |
| Initiation of GTMP treatment and setting up conditions, including environmental considerations | CI | CI | R | CI | CI | CI | CI | I | CI |
| Assessment: ability to handle, staff trainingb | R | S | RA | RS | S | ||||
| Screening GTMP prescriptions (patient basis) | R | RS | AC | CI | Ic | Ic | |||
| Receipt of GTMP from the manufacturer and inspection | R | S | CI | CI | Ic | Ic | |||
| Transportation | RA | S | CI | CI | SC | C | I | ||
| Storage | R | S | SC | 1 | |||||
| Preparation and decontamination of biological safety device | R | SI | SC | ||||||
| Dispensing | RA | S | |||||||
| Administration (product-dependent) | R | RS | |||||||
| Waste disposal | R | S | R | R | SC | S | S | S | |
| Decontamination of GTMP spills | Rd | RdS | Rd | Rd | ACI | CI | SI | ||
| Accidental exposure | Rd | Rd | RdI | RdI | ACI | CI |
References
- Hernandez, JM. Biosafety considerations for viral vector gene therapy: An explanation and guide for the average everyday-hero pharmacist. J Pharm Pract. 2022:8971900221104250.
- Vulto AG, Stoner N, Balásová H, Cercos A-C, Hoppe-Tichy T, Genestar JLV, et al. European Association of Hospital Pharmacists (EAHP) Guidance on the Pharmacy Handling of Gene Medicines. European Journal of Hospital Pharmacy. 2007, 13:29-39.
- European Association of Hospital Pharmacists. Special Interest Group Report: Hospital pharmacist’s preparedness for in-vivo gene therapy medicinal products 2023 [Available from: https://www.eahp.eu/sites/default/files/eahp_sig_gene_therapy_report.pdf.
- Appraisal of Guidelines for Research and Evaluation II 2017 [Available from: https://www.agreetrust.org/wp-content/uploads/2017/12/AGREE-II-Users-Manual-and-23-item-Instrument-2009-Update-2017.pdf.
- European Medicines Agency. Reflection paper on classification of advanced therapy medicinal products 2015 [updated 21 May 2015. Available from: https://www.ema.europa.eu/en/documents/scientific-guideline/reflection-paper-classification-advanced-therapy-medicinal-products_en-0.pdf.
- Pan UK Pharmacy Working Group for ATMPs. Gene therapy medicinal products: Governance and preparation requirements 2019 [updated October 2019. Available from: https://www.sps.nhs.uk/wp-content/uploads/2019/09/PAN-UK-PWG-for-ATMPs-Gene-Therapy-Guidance-issue-2.pdf.
- Sung YK, Kim SW. Recent advances in the development of gene delivery systems. Biomater Res. 2019, 23:8.
- World Health Organization. Laboratory biosafety manual. 3 ed 2004.
- European Medicines Agency. CAT quarterly highlights and approved ATMPs 2023 [updated 31 January 2023. Available from: https://www.ema.europa.eu/en/documents/report/cat-quarterly-highlights-approved-atmps-january-2023_en.pdf.
- Stoner, N. Are UK hospital pharmacy departments ready for the rise of gene therapy medicinal products? Expert Opin Biol Ther. 2018, 18, 837–40. [Google Scholar] [CrossRef] [PubMed]
- McNulty HBO, Duckert LR. Good Practice Initiative: Development of method for mobile aseptic preparation of advanced therapy medicinal products. 26th Congress of the European Association of Hospital Pharmacists; 23-25 March 2022, Vienna, Austria2022. p. PC11229.
- Société Française de Pharmacie Oncologique. Recommandations S.F.P.O. sur le circuit hospitalier des Médicaments de Thérapie Innovante (MTI) 2023 [updated August 2023. Available from: https://sfpo.com/wp-content/uploads/2023/08/Recommandations-MTI-V2-Aout-2023.pdf.
- Advanced Therapy Treatment Centres. Advanced therapies NHS readiness toolkit 2023 [Available from: https://www.theattcnetwork.co.uk/advanced-therapies-nhs-readiness-toolkit.
- National Health Service. Advanced Therapy Medicinal Products programme 2023 [Available from: https://www.e-lfh.org.uk/programmes/advanced-therapy-medicinal-products/.
- Health and Safety Executive. Containment and control of activities involving genetically modified microorganisms 2014 [Available from: https://www.hse.gov.uk/biosafety/gmo/acgm/acgmcomp/part3.pdf.
- Health and Safety Executive. Risk assessment of genetically modified microorganisms (other than those associated with plants) 2014 [Available from: https://www.hse.gov.uk/biosafety/gmo/acgm/acgmcomp/part2.pdf.
- Health and Safety Executive. Guidance on the use of genetically modified microorganisms in a clinical setting 2014 [updated 2014. Available from: https://www.hse.gov.uk/biosafety/gmo/acgm/acgmcomp/part6.pdf.
- Haut Conseil des Biotechnologies. Manuel du HCB pour l’utilisation confinée d’organismes génétiquement modifiés 2019 [updated 4 July 2019. Available from: https://www.ecologie.gouv.fr/sites/default/files/manuelduconfine2019.pdf.
- Ellison SL, Hunt DL. Perceived versus real risks of handling gene transfer agents in the pharmacy environment. Am J Health Syst Pharm. 2010, 67, 838–48.
- Webb TL, Hong E. GMO medicines and hospital pharmacy practice: a review. Journal of Pharmacy Practice and Research. 2021, 51, 203–10.
- Petrich J, Marchese D, Jenkins C, Storey M, Blind J. Gene replacement therapy: A primer for the health-system pharmacist. J Pharm Pract. 2020, 33, 846–55. [CrossRef] [PubMed]
- Blind JE, McLeod EN, Brown A, Patel H, Ghosh S. Biosafety practices for in vivo viral-mediated gene therapy in the health care setting. Appl Biosaf. 2020, 25, 194–200. [CrossRef] [PubMed]
- Blind JE, McLeod EN, Campbell KJ. Viral-mediated gene therapy and genetically modified therapeutics: A primer on biosafety handling for the health-system pharmacist. American Journal of Health-System Pharmacy. 2019, 76, 795–802.
- Armitstead JA, Zilich AJ, Williams KL, Sitzlar SC, Wermeling D. Hospital and pharmacy departmental policies and procedures for gene therapy at a teaching institution. Hosp Pharm. 2001, 36:56-66.
- Myers, C.J. Preparing pharmacists to manage gene therapies. J Am Pharm Assoc (2003). 2021, 61, e78–e82. [Google Scholar] [CrossRef] [PubMed]
- Gene Therapy Network 2020 [Available from: https://genetherapynetwork.com/emea.
- Danish Arbejdstilsynet. Risikovurdering af genteknologiske forskningsprojekter mv. 2001.
- Baldo A, van den Akker E, Bergmans HE, Lim F, Pauwels K. General considerations on the biosafety of virus-derived vectors used in gene therapy and vaccination. Current Gene Therapy. 2013, 13:385-94.
- What is RASCI/RACI: A comprehensive guide on how and when to use them 2023 [Available from: https://www.interfacing.com/what-is-rasci-raci.
- Sinclair A, Islam S, Jones S. Gene therapy: An overview of approved and pipeline technologies. Canadian Agency for Drugs and Technologies in Health. 2018(171).
- University of British Columbia. Cytotoxic spill clean up 2021 [updated January 2021. Available from: https://riskmanagement.sites.olt.ubc.ca/files/2021/01/CHEM-SWP-004-Cytotoxic-Spill-Clean-Up.pdf.
- GLYBERA Summary of Product Characteristics 2017 [updated 10 July 2017. Available from: https://www.ema.europa.eu/en/documents/product-information/glybera-epar-product-information_en.pdf.
- IMLYGIC Summary of Product Characteristics 2022 [updated 22 November 2022. Available from: https://www.ema.europa.eu/en/documents/product-information/imlygic-epar-product-information_en.pdf.
- STRIMVELIS Summary of Product Characteristics 2022 [updated 08 July 2022. Available from: https://www.ema.europa.eu/en/documents/product-information/strimvelis-epar-product-information_en.pdf.
- YESCARTA Summary of Product Characteristics 2023 [updated 30 January 2023. Available from: https://www.ema.europa.eu/en/documents/product-information/yescarta-epar-product-information_en.pdf.
- KYMRIAH Summary of Product Characteristics 2022 [updated 07 November 2022. Available from: https://www.ema.europa.eu/en/documents/product-information/kymriah-epar-product-information_en.pdf.
- LUXTURNA Summary of Product Characteristics 2023 [updated 04 May 2023. Available from: https://www.ema.europa.eu/en/documents/product-information/luxturna-epar-product-information_en.pdf.
- ZYNTEGLO Summary of Product Characteristics 2022 [updated 20 January 2022. Available from: https://www.ema.europa.eu/en/documents/product-information/zynteglo-epar-product-information_en-1.pdf.
- ZOLGENSMA Summary of Product Characteristics 2023 [updated 13 March 2023. Available from: https://www.ema.europa.eu/en/documents/product-information/zolgensma-epar-product-information_en.pdf.
- LIBMELDY Summary of Product Characteristics 2023 [updated 17 April 2023. Available from: https://www.ema.europa.eu/en/documents/product-information/libmeldy-epar-product-information_en.pdf.
- TECARTUS Summary of Product Characteristics 2023 [updated 30 January 2023. Available from: https://www.ema.europa.eu/en/documents/product-information/tecartus-epar-product-information_en.pdf.
- SKYSONA Summary of Product Characteristics 2022 [updated 04 April 2022. Available from: https://www.ema.europa.eu/en/documents/product-information/skysona-epar-product-information_en.pdf.
- ABECMA Summary of Product Characteristics 2022 [updated 28 October 2021. Available from: https://www.ema.europa.eu/en/documents/product-information/abecma-epar-product-information_en.pdf.
- BREYANZI Summary of Product Characteristics 2022 [updated 08 April 2022. Available from: https://www.ema.europa.eu/en/documents/product-information/breyanzi-epar-product-information_en.pdf.
- CARVYKTI Summary of Product Characteristics 2023 [updated 14 April 2023. Available from: https://www.ema.europa.eu/en/documents/product-information/carvykti-epar-product-information_en.pdf.
- UPSTAZA Summary of Product Characteristics 2023 [updated 04 April 2023. Available from: https://www.ema.europa.eu/en/documents/product-information/upstaza-epar-product-information_en-0.pdf.
- ROCTAVIAN Summary of Product Characteristics 2022 [updated 09 November 2022. Available from: https://www.ema.europa.eu/en/documents/product-information/roctavian-epar-product-information_en.pdf.
- HEMGENIX Summary of Product Characteristics 2023 [updated 20 June 2023. Available from: https://www.ema.europa.eu/en/documents/product-information/hemgenix-epar-product-information_en.pdf.
- National Institutes of Health. NIH Guidelines for Research Involving Recombinant or Synthetic Nucleic Acid Molecules (NIH Guidelines) 2019 [Available from: https://osp.od.nih.gov/wp-content/uploads/NIH_Guidelines.pdf.

| Precaution | Details |
|---|---|
| Protective clothinga | Disposable apron, gown, or lab coatbSafety glasses or gogglesGlovesMucous membrane splash protector (face mask) (26) |
| Biological safety cabinet (minimum class 2, type B) or pharmaceutical grade isolator (compliant with European standard EN12469:2000) | See Chart 2 |
| Care with use and disposal of needles and sharps | See Chart 5 |
| Decontamination of work surface areas | See Chart 2 |
| Decontaminate patient bedding according to procedures for blood- or body-fluid-soiled laundryb | See Chart 5 |
| No special precautions for patient elimination of stools or urine, unless specified in the Summary of Product Characteristics for product-specific information | Patients may use normal bathroom facilities unless advised otherwise |
| All transport and storage of patient specimens, GTMPs and associated waste must be in a double-enclosed, biohazard-labelled, leak-proof container (20, 26) | See Table 2 |
| Disposal of GTMPs, contaminated waste and patient specimens must follow institutional decontamination procedures | See Chart 5 |
| A spill kit must always be on hand (6, 20, 26) |
Suggested spill kit contents: 2× disposable gowns or arm covers 4× gloves 2× masks 2× aprons 2× goggles 4× disposable shoe covers 2× disinfectant sachets (or other, pre-prepared, decontaminant)Absorbent paper towels 2× disposable forceps 2× biohazard incineration bags Emergency contact numberCopy of spillage procedure |
| Institutional procedures and policies should be put in place for all steps along the GTMP handling pipeline (21) | Steps include storage, transport, reconstitution, dispensing, administration, disposal and decontamination, accidental exposure and spills and personnel training (21) |
| Hazardous signage must be used (20, 21, 26) | On all containers, storage cabinets/fridges/freezers and the door to the reconstitution room (20, 26) |
| All GTMPs should be added to the institutional hazardous drugs list (22) | As required by local legislation |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




