Submitted:
08 December 2023
Posted:
13 December 2023
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Precapillary cardiovascular disturbances and Capillary disturbances in ME/CFS and PCS - The causes and effect of hypovolemia and low stroke volume
2.1. The disturbed vasoconstrictor/vasodilator balance causes excessive vasoconstriction
3. Microcirculatory disturbances in ME/CFS and PCS
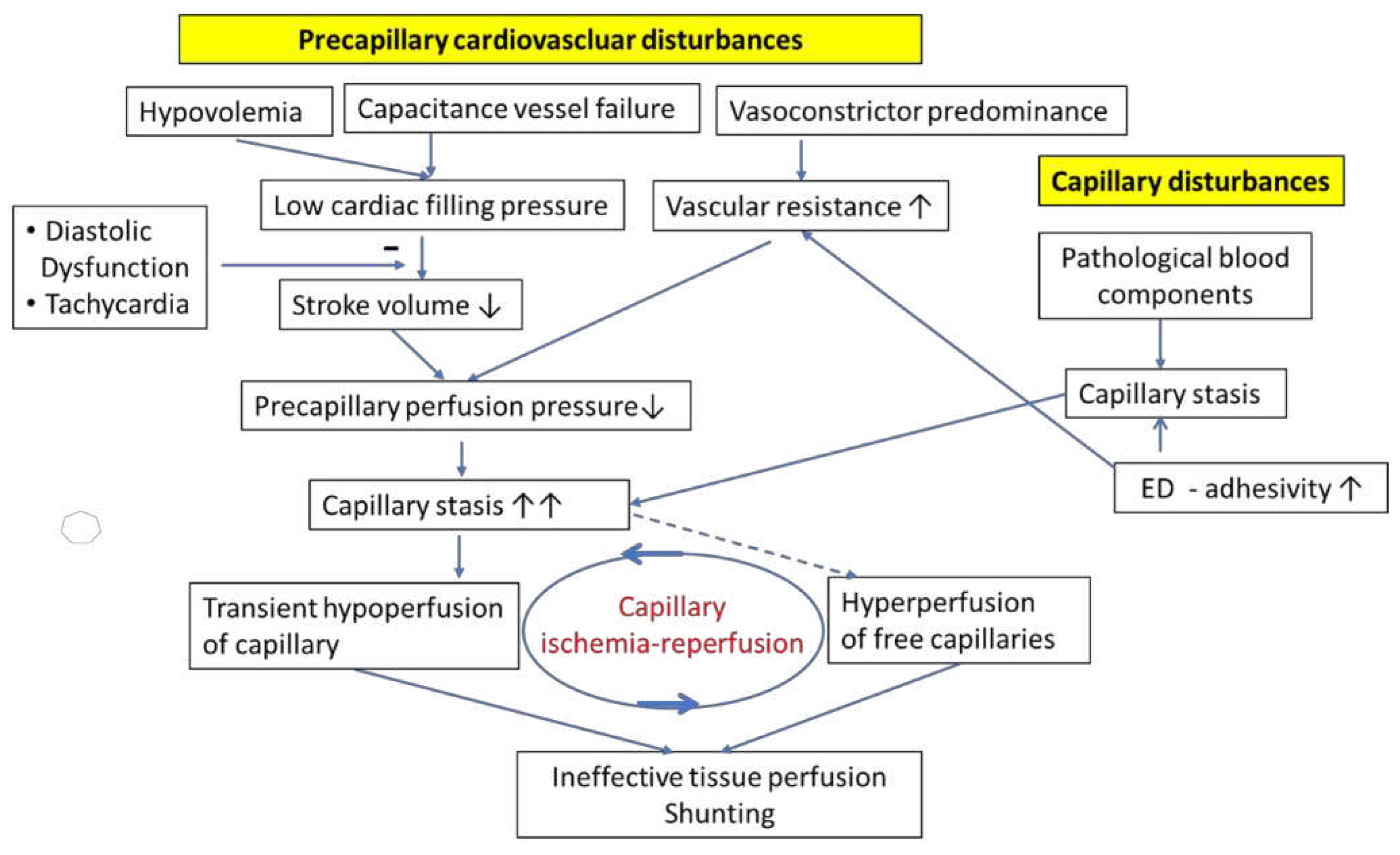
4. The Interaction of the precapillary cardiovascular disturbances with the capillary disturbances – a highly unfavorable synergistic interaction in ME/CFS and PCS
5. Mitochondrial dysfunction
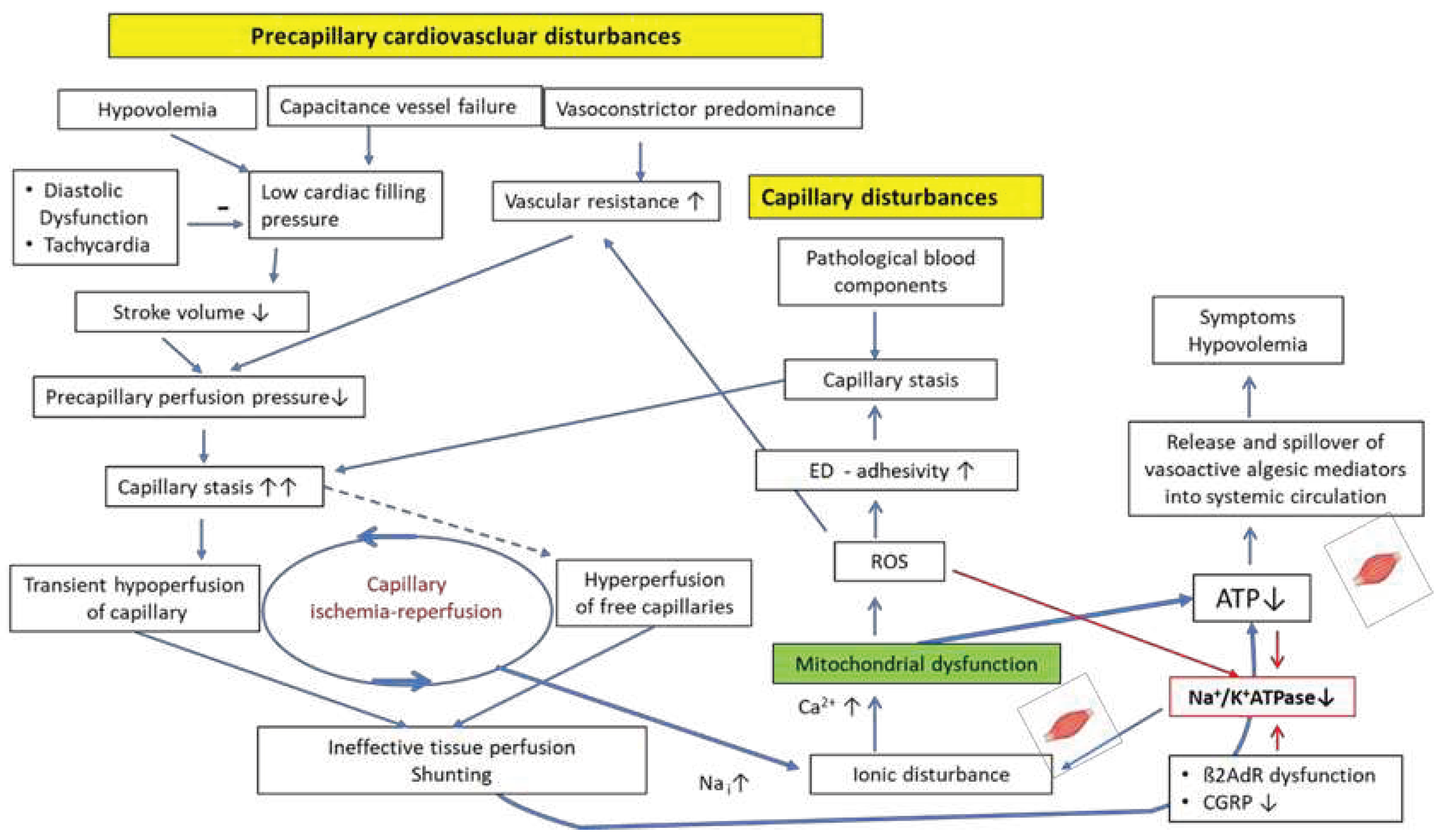
6. The consequences of critical energetic situation in skeletal muscle
Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Dixit, N.M.; Churchill, A.; Nsair, A.; Hsu, J.J. Post-Acute COVID-19 Syndrome and the cardiovascular system: What is known? Am Heart J Plus 2021, 5, 100025. [Google Scholar] [CrossRef] [PubMed]
- Yong, S.J.; Liu, S. Proposed subtypes of post-COVID-19 syndrome (or long-COVID) and their respective potential therapies. Rev Med Virol 2022, 32, e2315. [Google Scholar] [CrossRef] [PubMed]
- Wirth, K.J.; Lohn, M. Orthostatic Intolerance after COVID-19 Infection: Is Disturbed Microcirculation of the Vasa Vasorum of Capacitance Vessels the Primary Defect? Medicina (Kaunas) 2022, 58. [Google Scholar] [CrossRef] [PubMed]
- Legler, F.; Meyer-Arndt, L.; Modl, L.; Kedor, C.; Freitag, H.; Stein, E.; Hoppmann, U.; Rust, R.; Wittke, K.; Siebert, N.; et al. Long-term symptom severity and clinical biomarkers in post-COVID-19/chronic fatigue syndrome: results from a prospective observational cohort. EClinicalMedicine 2023, 63, 102146. [Google Scholar] [CrossRef]
- Kubankova, M.; Hohberger, B.; Hoffmanns, J.; Furst, J.; Herrmann, M.; Guck, J.; Krater, M. Physical phenotype of blood cells is altered in COVID-19. Biophys J 2021, 120, 2838–2847. [Google Scholar] [CrossRef] [PubMed]
- Morris, G.; Berk, M.; Galecki, P.; Maes, M. The emerging role of autoimmunity in myalgic encephalomyelitis/chronic fatigue syndrome (ME/cfs). Mol Neurobiol 2014, 49, 741–756. [Google Scholar] [CrossRef]
- Saha, A.K.; Schmidt, B.R.; Wilhelmy, J.; Nguyen, V.; Abugherir, A.; Do, J.K.; Nemat-Gorgani, M.; Davis, R.W.; Ramasubramanian, A.K. Red blood cell deformability is diminished in patients with Chronic Fatigue Syndrome. Clin Hemorheol Microcirc 2019, 71, 113–116. [Google Scholar] [CrossRef]
- Eaton-Fitch, N.; du Preez, S.; Cabanas, H.; Staines, D.; Marshall-Gradisnik, S. A systematic review of natural killer cells profile and cytotoxic function in myalgic encephalomyelitis/chronic fatigue syndrome. Syst Rev 2019, 8, 279. [Google Scholar] [CrossRef]
- Turner, S.; Khan, M.A.; Putrino, D.; Woodcock, A.; Kell, D.B.; Pretorius, E. Long COVID: pathophysiological factors and abnormalities of coagulation. Trends Endocrinol Metab 2023, 34, 321–344. [Google Scholar] [CrossRef]
- Nunes, J.M.; Kruger, A.; Proal, A.; Kell, D.B.; Pretorius, E. The Occurrence of Hyperactivated Platelets and Fibrinaloid Microclots in Myalgic Encephalomyelitis/Chronic Fatigue Syndrome (ME/CFS). Pharmaceuticals (Basel) 2022, 15. [Google Scholar] [CrossRef]
- Natelson, B.H.; Brunjes, D.L.; Mancini, D. Chronic Fatigue Syndrome and Cardiovascular Disease: JACC State-of-the-Art Review. J Am Coll Cardiol 2021, 78, 1056–1067. [Google Scholar] [CrossRef]
- Chilazi, M.; Duffy, E.Y.; Thakkar, A.; Michos, E.D. COVID and Cardiovascular Disease: What We Know in 2021. Curr Atheroscler Rep 2021, 23, 37. [Google Scholar] [CrossRef]
- Duffy, E.; Chilazi, M.; Cainzos-Achirica, M.; Michos, E.D. Cardiovascular Disease Prevention During the COVID-19 Pandemic: Lessons Learned and Future Opportunities. Methodist Debakey Cardiovasc J 2021, 17, 68–78. [Google Scholar] [CrossRef]
- Wirth, K.; Scheibenbogen, C. A Unifying Hypothesis of the Pathophysiology of Myalgic Encephalomyelitis/Chronic Fatigue Syndrome (ME/CFS): Recognitions from the finding of autoantibodies against ss2-adrenergic receptors. Autoimmun Rev 2020, 19, 102527. [Google Scholar] [CrossRef] [PubMed]
- Wirth, K.J.; Lohn, M. Myalgic Encephalomyelitis/Chronic Fatigue Syndrome (ME/CFS) and Comorbidities: Linked by Vascular Pathomechanisms and Vasoactive Mediators? Medicina (Kaunas) 2023, 59. [Google Scholar] [CrossRef]
- Wirth, K.J.; Scheibenbogen, C.; Paul, F. An attempt to explain the neurological symptoms of Myalgic Encephalomyelitis/Chronic Fatigue Syndrome. J Transl Med 2021, 19, 471. [Google Scholar] [CrossRef] [PubMed]
- McLaughlin, M.; Sanal-Hayes, N.E.M.; Hayes, L.D.; Berry, E.C.; Sculthorpe, N.F. People With Long COVID and Myalgic Encephalomyelitis/Chronic Fatigue Syndrome (ME/CFS) Exhibit Similarly Impaired Vascular Function. LID - S0002-9343(23)00609-5 [pii] LID -. [CrossRef]
- Kuorilehto, T.; Poyhonen, M.; Keski-Nisula, L.; Laurikka, J.; Salenius, J.P. [Vascular malformations associated with Marfan syndrome, Ehlers-Danlos syndrome and neurofibromatosis 1 and their surgical treatment]. Duodecim 2011, 127, 2280–2286. [Google Scholar]
- Meester, J.A.N.; Verstraeten, A.; Schepers, D.; Alaerts, M.; Van Laer, L.; Loeys, B.L. Differences in manifestations of Marfan syndrome, Ehlers-Danlos syndrome, and Loeys-Dietz syndrome. Ann Cardiothorac Surg 2017, 6, 582–594. [Google Scholar] [CrossRef] [PubMed]
- Islam, M.; Chang, C.; Gershwin, M.E. Ehlers-Danlos Syndrome: Immunologic contrasts and connective tissue comparisons. J Transl Autoimmun 2021, 4, 100077. [Google Scholar] [CrossRef]
- Roma, M.; Marden, C.L.; De Wandele, I.; Francomano, C.A.; Rowe, P.C. Postural tachycardia syndrome and other forms of orthostatic intolerance in Ehlers-Danlos syndrome. Auton Neurosci 2018, 215, 89–96. [Google Scholar] [CrossRef]
- Rowe, P.C.; Barron, D.F.; Calkins, H.; Maumenee, I.H.; Tong, P.Y.; Geraghty, M.T. Orthostatic intolerance and chronic fatigue syndrome associated with Ehlers-Danlos syndrome. J Pediatr 1999, 135, 494–499. [Google Scholar] [CrossRef]
- Campen, C.; Rowe, P.C.; Visser, F.C. Orthostatic Symptoms and Reductions in Cerebral Blood Flow in Long-Haul COVID-19 Patients: Similarities with Myalgic Encephalomyelitis/Chronic Fatigue Syndrome. Medicina (Kaunas) 2021, 58. [Google Scholar] [CrossRef] [PubMed]
- van Campen, C.; Rowe, P.C.; Visser, F.C. Deconditioning does not explain orthostatic intolerance in ME/CFS (myalgic encephalomyelitis/chronic fatigue syndrome). J Transl Med 2021, 19, 193. [Google Scholar] [CrossRef] [PubMed]
- van Campen, C.; Verheugt, F.W.A.; Rowe, P.C.; Visser, F.C. Orthostatic chronotropic incompetence in patients with myalgic encephalomyelitis/chronic fatigue syndrome (ME/CFS). IBRO Neurosci Rep 2023, 15, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Muller-Ruchholtz, E.R.; Losch, H.M.; Grund, E.; Lochner, W. Effect of alpha adrenergic receptor stimulation on integrated systemic venous bed. Pflugers Arch 1977, 370, 241–246. [Google Scholar] [CrossRef] [PubMed]
- Coupar, I.M. The effect of isoprenaline on adrenoceptors in human saphenous vein. Br J Pharmacol 1970, 39, 465–475. [Google Scholar] [CrossRef] [PubMed]
- Dachman, W.D.; Bedarida, G.; Blaschke, T.F.; Hoffman, B.B. Histamine-induced venodilation in human beings involves both H1 and H2 receptor subtypes. J Allergy Clin Immunol 1994, 93, 606–614. [Google Scholar] [CrossRef] [PubMed]
- Muller-Schweinitzer, E. On the pharmacology of venous smooth muscle from dog and man. Folia Haematol Int Mag Klin Morphol Blutforsch 1979, 106, 690–704. [Google Scholar] [PubMed]
- Weinstock, L.B.; Brook, J.B.; Walters, A.S.; Goris, A.; Afrin, L.B.; Molderings, G.J. Mast cell activation symptoms are prevalent in Long-COVID. Int J Infect Dis 2021, 112, 217–226. [Google Scholar] [CrossRef]
- Glynne, P.; Tahmasebi, N.; Gant, V.; Gupta, R. Long COVID following mild SARS-CoV-2 infection: characteristic T cell alterations and response to antihistamines. J Investig Med 2022, 70, 61–67. [Google Scholar] [CrossRef]
- Mashauri, H.L. Covid-19 Histamine theory: Why antihistamines should be incorporated as the basic component in Covid-19 management? Health Sci Rep 2023, 6, e1109. [Google Scholar] [CrossRef] [PubMed]
- Qu, C.; Fuhler, G.M.; Pan, Y. Could Histamine H1 Receptor Antagonists Be Used for Treating COVID-19? Int J Mol Sci 2021, 22. [Google Scholar] [CrossRef] [PubMed]
- Pinto, M.D.; Lambert, N.; Downs, C.A.; Abrahim, H.; Hughes, T.D.; Rahmani, A.M.; Burton, C.W.; Chakraborty, R. Antihistamines for Postacute Sequelae of SARS-CoV-2 Infection. J Nurse Pract 2022, 18, 335–338. [Google Scholar] [CrossRef] [PubMed]
- Salvucci, F.; Codella, R.; Coppola, A.; Zacchei, I.; Grassi, G.; Anti, M.L.; Nitisoara, N.; Luzi, L.; Gazzaruso, C. Antihistamines improve cardiovascular manifestations and other symptoms of long-COVID attributed to mast cell activation. Front Cardiovasc Med 2023, 10, 1202696. [Google Scholar] [CrossRef] [PubMed]
- Sivri, F.; Turkoz, I.; Sencan, M.; Icen, Y.K.; Aksoy, F.; Ceyhan, B.O. Does COVID-19 Cause Non-Dipper Hypertension? Angiology 2023, 33197231209584. [Google Scholar] [CrossRef] [PubMed]
- Blitshteyn, S.; Brinth, L.; Hendrickson, J.E.; Martinez-Lavin, M. Autonomic dysfunction and HPV immunization: an overview. Immunol Res 2018, 66, 744–754. [Google Scholar] [CrossRef] [PubMed]
- Pretorius, E.; Vlok, M.; Venter, C.; Bezuidenhout, J.A.; Laubscher, G.J.; Steenkamp, J.; Kell, D.B. Persistent clotting protein pathology in Long COVID/Post-Acute Sequelae of COVID-19 (PASC) is accompanied by increased levels of antiplasmin. Cardiovasc Diabetol 2021, 20, 172. [Google Scholar] [CrossRef] [PubMed]
- Pretorius, E.; Venter, C.; Laubscher, G.J.; Lourens, P.J.; Steenkamp, J.; Kell, D.B. Prevalence of readily detected amyloid blood clots in 'unclotted' Type 2 Diabetes Mellitus and COVID-19 plasma: a preliminary report. Cardiovasc Diabetol 2020, 19, 193. [Google Scholar] [CrossRef] [PubMed]
- Pretorius, E.; Venter, C.; Laubscher, G.J.; Kotze, M.J.; Oladejo, S.O.; Watson, L.R.; Rajaratnam, K.; Watson, B.W.; Kell, D.B. Prevalence of symptoms, comorbidities, fibrin amyloid microclots and platelet pathology in individuals with Long COVID/Post-Acute Sequelae of COVID-19 (PASC). Cardiovasc Diabetol 2022, 21, 148. [Google Scholar] [CrossRef]
- Schlick, S.; Lucio, M.; Wallukat, G.; Bartsch, A.; Skornia, A.; Hoffmanns, J.; Szewczykowski, C.; Schroder, T.; Raith, F.; Rogge, L.; et al. Post-COVID-19 Syndrome: Retinal Microcirculation as a Potential Marker for Chronic Fatigue. Int J Mol Sci 2022, 23. [Google Scholar] [CrossRef]
- Iba, T.; Connors, J.M.; Levy, J.H. What Role Does Microthrombosis Play in Long COVID? Semin Thromb Hemost 2023. [Google Scholar] [CrossRef]
- Nusz, D.J.; White, D.C.; Dai, Q.; Pippen, A.M.; Thompson, M.A.; Walton, G.B.; Parsa, C.J.; Koch, W.J.; Annex, B.H. Vascular rarefaction in peripheral skeletal muscle after experimental heart failure. Am J Physiol Heart Circ Physiol 2003, 285, H1554–1562. [Google Scholar] [CrossRef] [PubMed]
- Joseph, P.; Arevalo, C.; Oliveira, R.K.F.; Faria-Urbina, M.; Felsenstein, D.; Oaklander, A.L.; Systrom, D.M. Insights From Invasive Cardiopulmonary Exercise Testing of Patients With Myalgic Encephalomyelitis/Chronic Fatigue Syndrome. CHEST 2021, 160, 642–651. [Google Scholar] [CrossRef] [PubMed]
- Greene, A.S.; Tonellato, P.J.; Lui, J.; Lombard, J.H.; Cowley, A.W., Jr. Microvascular rarefaction and tissue vascular resistance in hypertension. Am J Physiol 1989, 256, H126–131. [Google Scholar] [CrossRef] [PubMed]
- Shan, Z.Y.; Barnden, L.R.; Kwiatek, R.A.; Bhuta, S.; Hermens, D.F.; Lagopoulos, J. Neuroimaging characteristics of myalgic encephalomyelitis/chronic fatigue syndrome (ME/CFS): a systematic review. J Transl Med 2020, 18, 335. [Google Scholar] [CrossRef]
- Shan, Z.Y.; Mohamed, A.Z.; Andersen, T.; Rendall, S.; Kwiatek, R.A.; Fante, P.D.; Calhoun, V.D.; Bhuta, S.; Lagopoulos, J. Multimodal MRI of myalgic encephalomyelitis/chronic fatigue syndrome: A cross-sectional neuroimaging study toward its neuropathophysiology and diagnosis. Front Neurol 2022, 13, 954142. [Google Scholar] [CrossRef] [PubMed]
- Adingupu, D.D.; Soroush, A.; Hansen, A.; Twomey, R.; Dunn, J.F. Reduced Cerebrovascular Oxygenation in Individuals with Post-Acute COVID-19 Syndrome (PACS) ("long COVID"). Adv Exp Med Biol 2023, 1438, 211–216. [Google Scholar] [CrossRef] [PubMed]
- van Campen, C.; Rowe, P.C.; Visser, F.C. Blood Volume Status in ME/CFS Correlates With the Presence or Absence of Orthostatic Symptoms: Preliminary Results. Front Pediatr 2018, 6, 352. [Google Scholar] [CrossRef] [PubMed]
- Guven, G.; Hilty, M.P.; Ince, C. Microcirculation: Physiology, Pathophysiology, and Clinical Application. Blood Purif 2020, 49, 143–150. [Google Scholar] [CrossRef]
- Charfeddine, S.; Ibn Hadj Amor, H.; Jdidi, J.; Torjmen, S.; Kraiem, S.; Hammami, R.; Bahloul, A.; Kallel, N.; Moussa, N.; Touil, I.; et al. Long COVID 19 Syndrome: Is It Related to Microcirculation and Endothelial Dysfunction? Insights From TUN-EndCOV Study. Front Cardiovasc Med 2021, 8, 745758. [Google Scholar] [CrossRef]
- Jannasz, I.; Pruc, M.; Rahnama-Hezavah, M.; Targowski, T.; Olszewski, R.; Feduniw, S.; Petryka, K.; Szarpak, L. The Impact of COVID-19 on Carotid-Femoral Pulse Wave Velocity: A Systematic Review and Meta-Analysis. J Clin Med 2023, 12. [Google Scholar] [CrossRef] [PubMed]
- Mali, W.; Doevendans, P.A. Form and function, both matter. Neth Heart J 2015, 23, 312–313. [Google Scholar] [CrossRef] [PubMed]
- O'Rourke, M. Arterial stiffness, systolic blood pressure, and logical treatment of arterial hypertension. Hypertension 1990, 15, 339–347. [Google Scholar] [CrossRef] [PubMed]
- Day, H.; Yellman, B.; Hammer, S.; Rond, C.; Bell, J.; Abbaszadeh, S.; Stoddard, G.; Unutmaz, D.; Bateman, L.; Vernon, S.D. Cognitive impairment in post-acute sequelae of COVID-19 and short duration myalgic encephalomyelitis patients is mediated by orthostatic hemodynamic changes. Front Neurosci 2023, 17, 1203514. [Google Scholar] [CrossRef] [PubMed]
- Tas, S.; Tas, U. Effects of COVID-19 on the Autonomic Cardiovascular System: Heart Rate Variability and Turbulence in Recovered Patients. Tex Heart Inst J 2023, 50. [Google Scholar] [CrossRef]
- Delle Donne, G.; Roses Noguer, F.; Till, J.; Salukhe, T.; Prasad, S.K.; Daubeney, P.E.F. Ivabradine in Postural Orthostatic Tachycardia Syndrome: Preliminary Experience in Children. Am J Cardiovasc Drugs 2018, 18, 59–63. [Google Scholar] [CrossRef] [PubMed]
- Tahir, F.; Bin Arif, T.; Majid, Z.; Ahmed, J.; Khalid, M. Ivabradine in Postural Orthostatic Tachycardia Syndrome: A Review of the Literature. Cureus 2020, 12, e7868. [Google Scholar] [CrossRef] [PubMed]
- Ruzieh, M.; Sirianni, N.; Ammari, Z.; Dasa, O.; Alhazmi, L.; Karabin, B.; Grubb, B. Ivabradine in the treatment of postural tachycardia syndrome (POTS), a single center experience. Pacing Clin Electrophysiol 2017, 40, 1242–1245. [Google Scholar] [CrossRef] [PubMed]
- Barzilai, M.; Jacob, G. The Effect of Ivabradine on the Heart Rate and Sympathovagal Balance in Postural Tachycardia Syndrome Patients. Rambam Maimonides Med J 2015, 6. [Google Scholar] [CrossRef]
- Lacasa, M.; Launois, P.; Prados, F.; Alegre, J.; Casas-Roma, J. Unsupervised cluster analysis reveals distinct subtypes of ME/CFS patients based on peak oxygen consumption and SF-36 scores. Clinical Therapeutics. [CrossRef]
- Wirth, K.J.; Scheibenbogen, C. Pathophysiology of skeletal muscle disturbances in Myalgic Encephalomyelitis/Chronic Fatigue Syndrome (ME/CFS). J Transl Med 2021, 19, 162. [Google Scholar] [CrossRef]
- Avkiran, M.; Gross, G.; Karmazyn, M.; Klein, H.; Murphy, E.; Ytrehus, K. Na+/H+ exchange in ischemia, reperfusion and preconditioning. Cardiovasc Res 2001, 50, 162–166. [Google Scholar] [CrossRef]
- Karmazyn, M.; Sawyer, M.; Fliegel, L. The Na(+)/H(+) exchanger: a target for cardiac therapeutic intervention. Curr Drug Targets Cardiovasc Haematol Disord 2005, 5, 323–335. [Google Scholar] [CrossRef] [PubMed]
- Clausen, T. Na+-K+ Pump Regulation and Skeletal Muscle Contractility. Physiological Reviews 2003, 83, 1269–1324. [Google Scholar] [CrossRef] [PubMed]
- Petter, E.; Scheibenbogen, C.; Linz, P.; Stehning, C.; Wirth, K.; Kuehne, T.; Kelm, M. Muscle sodium content in patients with Myalgic Encephalomyelitis/Chronic Fatigue Syndrome. J Transl Med 2022, 20, 580. [Google Scholar] [CrossRef] [PubMed]
- Pirkmajer, S.; Chibalin, A.V. Na,K-ATPase regulation in skeletal muscle. Am J Physiol Endocrinol Metab 2016, 311, E1–E31. [Google Scholar] [CrossRef] [PubMed]
- Clausen, T. Quantification of Na+,K+ pumps and their transport rate in skeletal muscle: Functional significance. Journal of General Physiology 2013, 142, 327–345. [Google Scholar] [CrossRef] [PubMed]
- Cotecchia, S.; Stanasila, L.; Diviani, D. Protein-protein interactions at the adrenergic receptors. Curr Drug Targets 2012, 13, 15–27. [Google Scholar] [CrossRef] [PubMed]
- Oaklander, A.L.; Nolano, M. Scientific Advances in and Clinical Approaches to Small-Fiber Polyneuropathy: A Review. JAMA Neurol 2019. [Google Scholar] [CrossRef]
- Abrams, R.M.C.; Simpson, D.M.; Navis, A.; Jette, N.; Zhou, L.; Shin, S.C. Small fiber neuropathy associated with SARS-CoV-2 infection. Muscle & Nerve 2022, 65, 440–443. [Google Scholar] [CrossRef]
- Oaklander, A.L.; Mills, A.J.; Kelley, M.; Toran, L.S.; Smith, B.; Dalakas, M.C.; Nath, A. Peripheral Neuropathy Evaluations of Patients With Prolonged Long COVID. Neurol Neuroimmunol Neuroinflamm 2022, 9. [Google Scholar] [CrossRef]
- Brain, S.D.; Cox, H.M. Neuropeptides and their receptors: innovative science providing novel therapeutic targets.
- Nielsen, O.B.; Hilsted L Fau - Clausen, T.; Clausen, T. Excitation-induced force recovery in potassium-inhibited rat soleus muscle.
- Jacobs, L.M.C.; Wintjens, M.; Nagy, M.; Willems, L.; Ten Cate, H.; Spronk, H.M.H.; van Kuijk, S.M.J.; Ghossein-Doha, C.; Netea, M.G.; Groh, L.A.; et al. Biomarkers of sustained systemic inflammation and microvascular dysfunction associated with post-COVID-19 condition symptoms at 24 months after SARS-CoV-2-infection. Front Immunol 2023, 14, 1182182. [Google Scholar] [CrossRef] [PubMed]
- Gunter, T.E.; Yule, D.I.; Gunter, K.K.; Eliseev, R.A.; Salter, J.D. Calcium and mitochondria. FEBS Lett 2004, 567, 96–102. [Google Scholar] [CrossRef] [PubMed]
- Jammes, Y.; Adjriou, N.; Kipson, N.; Criado, C.; Charpin, C.; Rebaudet, S.; Stavris, C.; Guieu, R.; Fenouillet, E.; Retornaz, F. Altered muscle membrane potential and redox status differentiates two subgroups of patients with chronic fatigue syndrome. J Transl Med 2020, 18, 173. [Google Scholar] [CrossRef] [PubMed]
- Klein, J.A.-O.; Wood, J.; Jaycox, J.R.; Dhodapkar, R.A.-O.; Lu, P.A.-O.X.; Gehlhausen, J.R.; Tabachnikova, A.; Greene, K.; Tabacof, L.; Malik, A.A.; et al. Distinguishing features of long COVID identified through immune profiling.
- Miwa, K. Down-regulation of renin–aldosterone and antidiuretic hormone systems in patients with myalgic encephalomyelitis/chronic fatigue syndrome. Journal of cardiology 2017, 69, 684–688. [Google Scholar] [CrossRef]
- Su, Y.; Yuan, D.; Chen, D.G.; Ng, R.H.; Wang, K.; Choi, J.; Li, S.; Hong, S.; Zhang, R.; Xie, J.; et al. Multiple early factors anticipate post-acute COVID-19 sequelae. Cell 2022, 185, 881–895.e820. [Google Scholar] [CrossRef]
- Joffre, J.; Hellman, J. Oxidative Stress and Endothelial Dysfunction in Sepsis and Acute Inflammation. Antioxid Redox Signal 2021, 35, 1291–1307. [Google Scholar] [CrossRef] [PubMed]
- Liu, J.; Kennedy, D.J.; Yan, Y.; Shapiro, J.I. Reactive Oxygen Species Modulation of Na/K-ATPase Regulates Fibrosis and Renal Proximal Tubular Sodium Handling. Int J Nephrol 2012, 2012, 381320. [Google Scholar] [CrossRef]
- Myhill, S.; Booth, N.E.; McLaren-Howard, J. Chronic fatigue syndrome and mitochondrial dysfunction. Int J Clin Exp Med 2009, 2, 1–16. [Google Scholar] [PubMed]
- Rutherford, G.; Manning, P.; Newton, J.L. Understanding Muscle Dysfunction in Chronic Fatigue Syndrome. J Aging Res 2016, 2016, 2497348. [Google Scholar] [CrossRef]
- Fluge, O.; Mella, O.; Bruland, O.; Risa, K.; Dyrstad, S.E.; Alme, K.; Rekeland, I.G.; Sapkota, D.; Rosland, G.V.; Fossa, A.; et al. Metabolic profiling indicates impaired pyruvate dehydrogenase function in myalgic encephalopathy/chronic fatigue syndrome. JCI Insight 2016, 1, e89376. [Google Scholar] [CrossRef] [PubMed]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).



