Submitted:
07 December 2023
Posted:
08 December 2023
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
2.1. Study patients
2.2. Data collection and analyses of AQP4-IgG and MOG-IgG serostatus
2.3. Statistical analyses
3. Results
3.1. Patient characteristics
3.2. Rituximab treatment
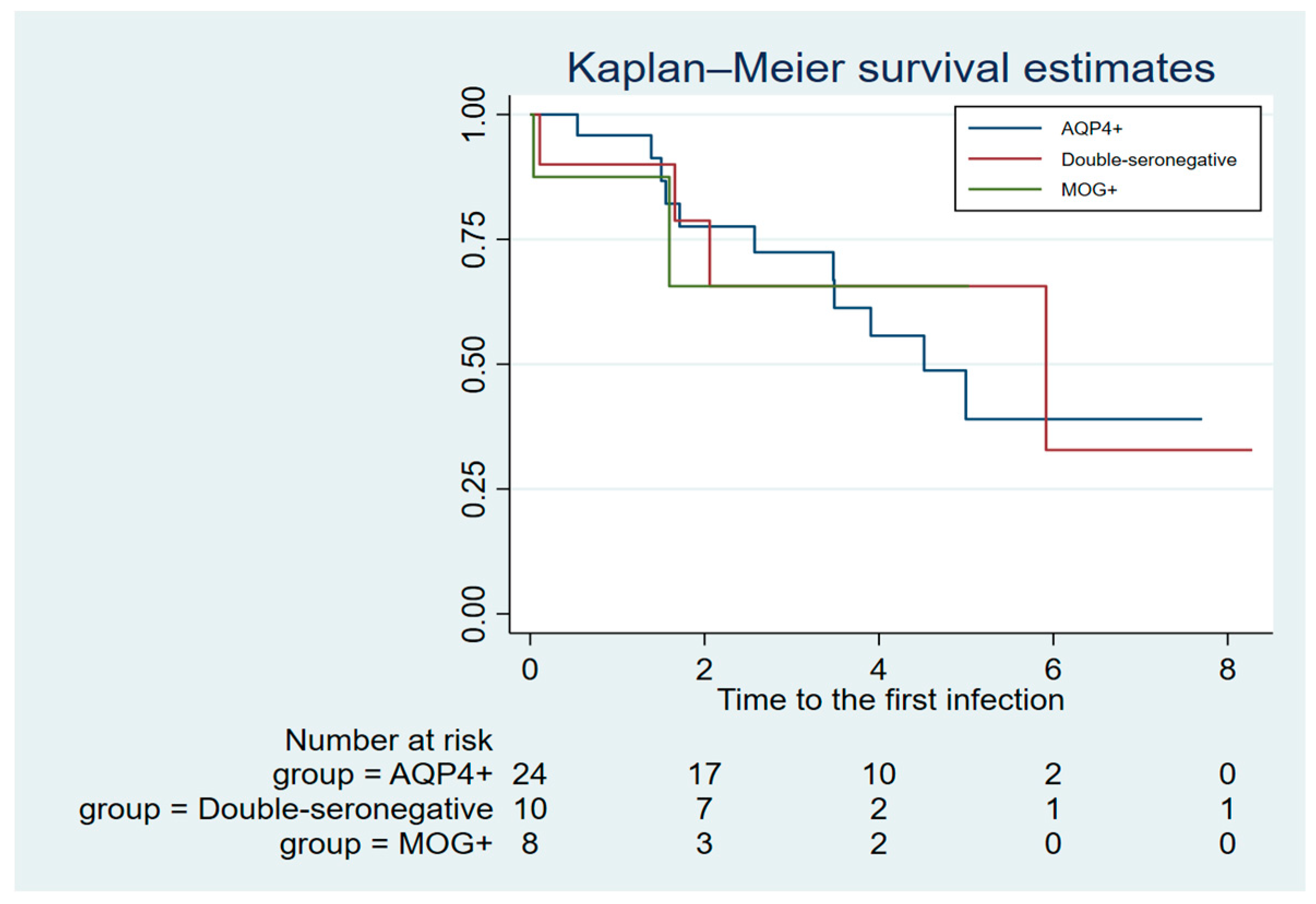
3.3. Relapse activity
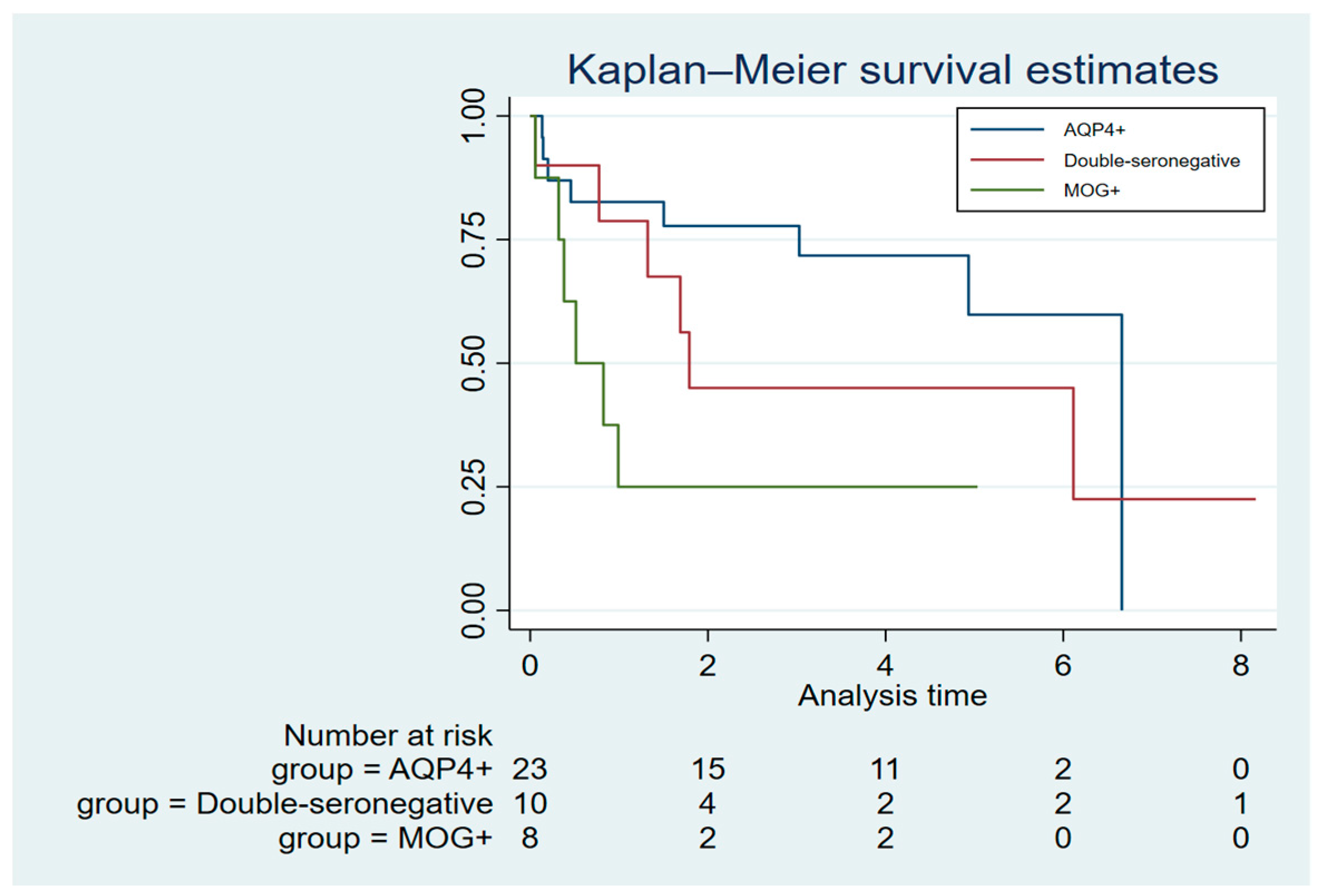
3.4. Severe infectious events and malignancy
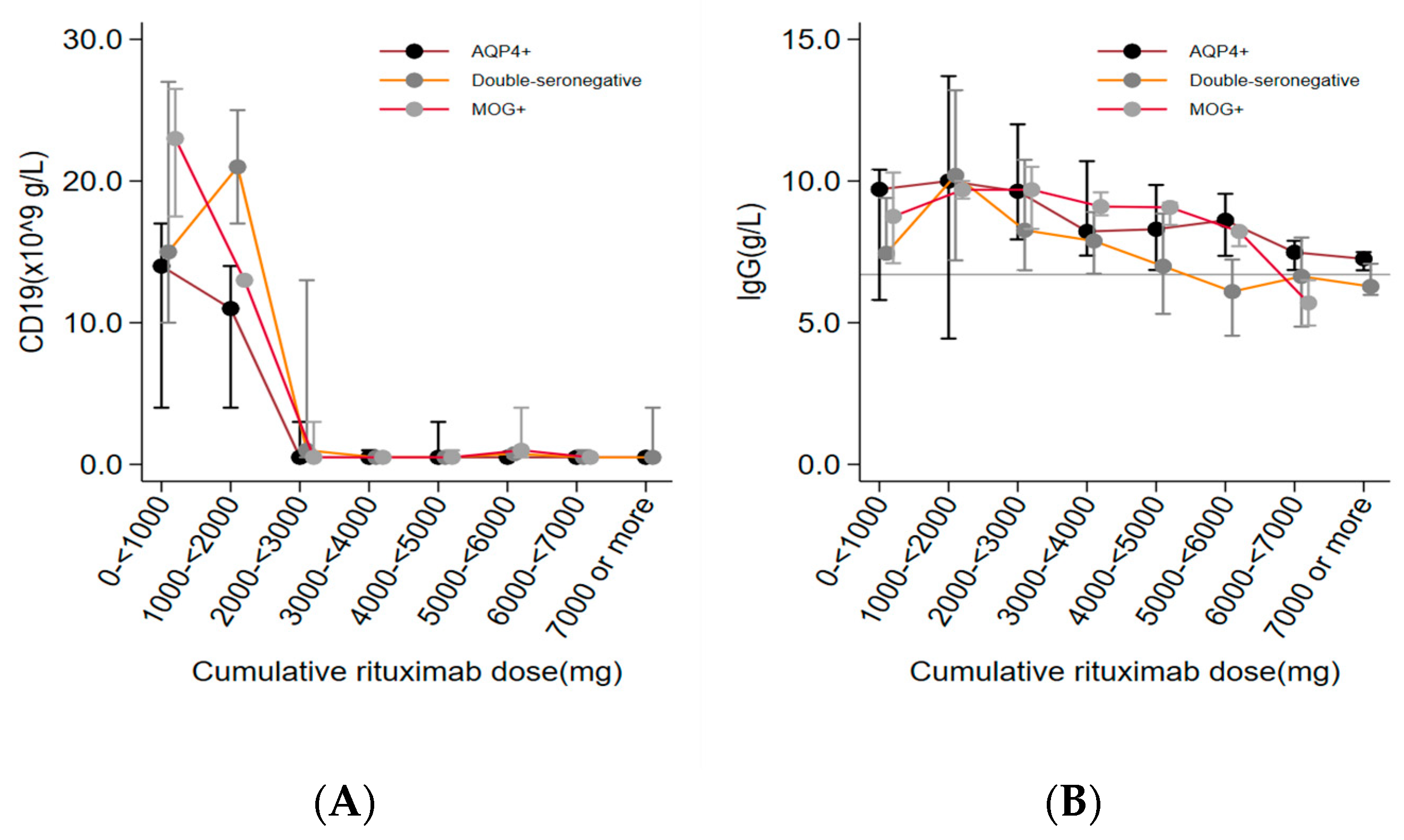
CD19+ lymphocyte counts and IgG levels
4. Discussion
5. Conclusion
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Wingerchuk DM, Lennon VA, Lucchinetti CF, Pittock SJ, Weinshenker BG. The spectrum of neuromyelitis optica. Lancet Neurol. 2007;6(9):805-15.
- Wingerchuk DM, Hogancamp WF, O’Brien PC, Weinshenker BG. The clinical course of neuromyelitis optica (Devic’s syndrome). Neurology. 1999;53(5):1107-14. [CrossRef]
- Lennon VA, Wingerchuk DM, Kryzer TJ, Pittock SJ, Lucchinetti CF, Fujihara K, et al. A serum autoantibody marker of neuromyelitis optica: distinction from multiple sclerosis. Lancet. 2004;364(9451):2106-12. [CrossRef]
- Wingerchuk DM, Banwell B, Bennett JL, Cabre P, Carroll W, Chitnis T, et al. International consensus diagnostic criteria for neuromyelitis optica spectrum disorders. Neurology. 2015;85(2):177-89. [CrossRef]
- Hyun JW, Jeong IH, Joung A, Kim SH, Kim HJ. Evaluation of the 2015 diagnostic criteria for neuromyelitis optica spectrum disorder. Neurology. 2016;86(19):1772-9. [CrossRef]
- Hamid SH, Elsone L, Mutch K, Solomon T, Jacob A. The impact of 2015 neuromyelitis optica spectrum disorders criteria on diagnostic rates. Mult Scler. 2017;23(2):228-33. [CrossRef]
- Reindl M, Waters P. Myelin oligodendrocyte glycoprotein antibodies in neurological disease. Nat Rev Neurol. 2019;15(2):89-102. [CrossRef]
- Sechi E, Cacciaguerra L, Chen JJ, Mariotto S, Fadda G, Dinoto A, et al. Myelin Oligodendrocyte Glycoprotein Antibody-Associated Disease (MOGAD): A Review of Clinical and MRI Features, Diagnosis, and Management. Front Neurol. 2022;13:885218. [CrossRef]
- Wu Y, Geraldes R, Jurynczyk M, Palace J. Double-negative neuromyelitis optica spectrum disorder. Mult Scler. 2023;29(11-12):1353-62. [CrossRef]
- Jonsson DI, Sveinsson O, Hakim R, Brundin L. Epidemiology of NMOSD in Sweden from 1987 to 2013: A nationwide population-based study. Neurology. 2019;93(2):e181-e9. [CrossRef]
- Flanagan EP, Cabre P, Weinshenker BG, Sauver JS, Jacobson DJ, Majed M, et al. Epidemiology of aquaporin-4 autoimmunity and neuromyelitis optica spectrum. Ann Neurol. 2016;79(5):775-83. [CrossRef]
- Cabrera-Gomez JA, Bonnan M, Gonzalez-Quevedo A, Saiz-Hinarejos A, Marignier R, Olindo S, et al. Neuromyelitis optica positive antibodies confer a worse course in relapsing-neuromyelitis optica in Cuba and French West Indies. Mult Scler. 2009;15(7):828-33. [CrossRef]
- Yamamura T, Kleiter I, Fujihara K, Palace J, Greenberg B, Zakrzewska-Pniewska B, et al. Trial of Satralizumab in Neuromyelitis Optica Spectrum Disorder. N Engl J Med. 2019;381(22):2114-24. [CrossRef]
- Traboulsee A, Greenberg BM, Bennett JL, Szczechowski L, Fox E, Shkrobot S, et al. Safety and efficacy of satralizumab monotherapy in neuromyelitis optica spectrum disorder: a randomised, double-blind, multicentre, placebo-controlled phase 3 trial. Lancet Neurol. 2020;19(5):402-12. [CrossRef]
- Cree BAC, Bennett JL, Kim HJ, Weinshenker BG, Pittock SJ, Wingerchuk DM, et al. Inebilizumab for the treatment of neuromyelitis optica spectrum disorder (N-MOmentum): a double-blind, randomised placebo-controlled phase 2/3 trial. Lancet. 2019;394(10206):1352-63. [CrossRef]
- Pittock SJ, Fujihara K, Palace J, Berthele A, Kim HJ, Oreja-Guevara C, et al. Eculizumab monotherapy for NMOSD: Data from PREVENT and its open-label extension. Mult Scler. 2022;28(3):480-6.
- Costanzi C, Matiello M, Lucchinetti CF, Weinshenker BG, Pittock SJ, Mandrekar J, et al. Azathioprine: tolerability, efficacy, and predictors of benefit in neuromyelitis optica. Neurology. 2011;77(7):659-66.
- Moog TM, Smith AD, Burgess KW, McCreary M, Okuda DT. High-efficacy therapies reduce clinical and radiological events more effectively than traditional treatments in neuromyelitis optica spectrum disorder. J Neurol. 2023;270(7):3595-602. [CrossRef]
- Chen H, Qiu W, Zhang Q, Wang J, Shi Z, Liu J, et al. Comparisons of the efficacy and tolerability of mycophenolate mofetil and azathioprine as treatments for neuromyelitis optica and neuromyelitis optica spectrum disorder. Eur J Neurol. 2017;24(1):219-26. [CrossRef]
- Nikoo Z, Badihian S, Shaygannejad V, Asgari N, Ashtari F. Comparison of the efficacy of azathioprine and rituximab in neuromyelitis optica spectrum disorder: a randomized clinical trial. J Neurol. 2017;264(9):2003-9. [CrossRef]
- Yang Y, Chen L, Wu L, Yao J, Wang N, Su X, et al. Effective Rituximab Treatment in Patients with Neuromyelitis Optica Spectrum Disorders Compared with Azathioprine and Mycophenolate. Neurol Ther. 2022;11(1):137-149. [CrossRef]
- Montcuquet A, Collongues N, Papeix C, Zephir H, Audoin B, Laplaud D, et al. Effectiveness of mycophenolate mofetil as first-line therapy in AQP4-IgG, MOG-IgG, and seronegative neuromyelitis optica spectrum disorders. Mult Scler. 2017;23(10):1377-84. [CrossRef]
- Tahara M, Oeda T, Okada K, Kiriyama T, Ochi K, Maruyama H, et al. Safety and efficacy of rituximab in neuromyelitis optica spectrum disorders (RIN-1 study): a multicentre, randomised, double-blind, placebo-controlled trial. Lancet Neurol. 2020;19(4):298-306. [CrossRef]
- Poupart J, Giovannelli J, Deschamps R, Audoin B, Ciron J, Maillart E, et al. Evaluation of efficacy and tolerability of first-line therapies in NMOSD. Neurology. 2020;94(15):e1645-e56. [CrossRef]
- Novi G, Bovis F, Capobianco M, Frau J, Mataluni G, Curti E, et al. Efficacy of different rituximab therapeutic strategies in patients with neuromyelitis optica spectrum disorders. Mult Scler Relat Disord. 2019;36:101430. [CrossRef]
- Damato V, Evoli A, Iorio R. Efficacy and Safety of Rituximab Therapy in Neuromyelitis Optica Spectrum Disorders: A Systematic Review and Meta-analysis. JAMA Neurol. 2016;73(11):1342-8.
- Trebst C, Jarius S, Berthele A, Paul F, Schippling S, Wildemann B, et al. Update on the diagnosis and treatment of neuromyelitis optica: recommendations of the Neuromyelitis Optica Study Group (NEMOS). J Neurol. 2014;261(1):1-16. [CrossRef]
- Jarius S, Aktas O, Ayzenberg I, Bellmann-Strobl J, Berthele A, Giglhuber K, et al. Update on the diagnosis and treatment of neuromyelits optica spectrum disorders (NMOSD) - revised recommendations of the Neuromyelitis Optica Study Group (NEMOS). Part I: Diagnosis and differential diagnosis. J Neurol. 2023;270(7):3341-68.
- https://www.svenskamssallskapet.se/nmosd/.
- Barreras P, Vasileiou ES, Filippatou AG, Fitzgerald KC, Levy M, Pardo CA, et al. Long-term Effectiveness and Safety of Rituximab in Neuromyelitis Optica Spectrum Disorder and MOG Antibody Disease. Neurology. 2022;99(22):e2504-e16. [CrossRef]
- Nepal G, Kharel S, Coghlan MA, Rayamajhi P, Ojha R. Safety and efficacy of rituximab for relapse prevention in myelin oligodendrocyte glycoprotein immunoglobulin G (MOG-IgG)-associated disorders (MOGAD): A systematic review and meta-analysis. J Neuroimmunol. 2022;364:577812.
- Rico A, Ninove L, Maarouf A, Boutiere C, Durozard P, Demortiere S, et al. Determining the best window for BNT162b2 mRNA vaccination for SARS-CoV-2 in patients with multiple sclerosis receiving anti-CD20 therapy. Mult Scler J Exp Transl Clin. 2021;7(4):20552173211062142.
- Avouac A, Maarouf A, Stellmann JP, Rico A, Boutiere C, Demortiere S, et al. Rituximab-Induced Hypogammaglobulinemia and Infections in AQP4 and MOG Antibody-Associated Diseases. Neurol Neuroimmunol Neuroinflamm. 2021;8(3). [CrossRef]
- Banwell B, Bennett JL, Marignier R, Kim HJ, Brilot F, Flanagan EP, et al. Diagnosis of myelin oligodendrocyte glycoprotein antibody-associated disease: International MOGAD Panel proposed criteria. Lancet Neurol. 2023;22(3):268-82. [CrossRef]
- Kurtzke JF. Rating neurologic impairment in multiple sclerosis: an expanded disability status scale (EDSS). Neurology. 1983;33(11):1444-52.
- Gao F, Chai B, Gu C, Wu R, Dong T, Yao Y, et al. Effectiveness of rituximab in neuromyelitis optica: a meta-analysis. BMC Neurol. 2019;19(1):36. [CrossRef]
- Durozard P, Rico A, Boutiere C, Maarouf A, Lacroix R, Cointe S, et al. Comparison of the Response to Rituximab between Myelin Oligodendrocyte Glycoprotein and Aquaporin-4 Antibody Diseases. Ann Neurol. 2020;87(2):256-66. [CrossRef]
- Chen JJ, Flanagan EP, Bhatti MT, Jitprapaikulsan J, Dubey D, Lopez Chiriboga ASS, et al. Steroid-sparing maintenance immunotherapy for MOG-IgG associated disorder. Neurology. 2020;95(2):e111-e20.
- Kim SH, Park NY, Kim KH, Hyun JW, Kim HJ. Rituximab-Induced Hypogammaglobulinemia and Risk of Infection in Neuromyelitis Optica Spectrum Disorders: A 14-Year Real-Life Experience. Neurol Neuroimmunol Neuroinflamm. 2022;9(5).
- Sellner J, Hemmer B, Muhlau M. The clinical spectrum and immunobiology of parainfectious neuromyelitis optica (Devic) syndromes. J Autoimmun. 2010;34(4):371-9. [CrossRef]
- Wang L, Zhou L, ZhangBao J, Huang W, Tan H, Fan Y, et al. Causal associations between prodromal infection and neuromyelitis optica spectrum disorder: A Mendelian randomization study. Eur J Neurol. 2023. [CrossRef]
- Sellner J, Cepok S, Kalluri SR, Nestler A, Kleiter I, Kumpfel T, et al. Aquaporin 4 antibody positive central nervous system autoimmunity and multiple sclerosis are characterized by a distinct profile of antibodies to herpes viruses. Neurochem Int. 2010;57(6):662-7.
- Md Yusof MY, Vital EM, McElvenny DM, Hensor EMA, Das S, Dass S, et al. Predicting Severe Infection and Effects of Hypogammaglobulinemia During Therapy With Rituximab in Rheumatic and Musculoskeletal Diseases. Arthritis Rheumatol. 2019;71(11):1812-23.
- Fujihara K. MOG-antibody-associated disease is different from MS and NMOSD and should be classified as a distinct disease entity - Commentary. Mult Scler. 2020;26(3):276-8. [CrossRef]
- Jarius S, Ruprecht K, Wildemann B, Kuempfel T, Ringelstein M, Geis C, et al. Contrasting disease patterns in seropositive and seronegative neuromyelitis optica: A multicentre study of 175 patients. J Neuroinflammation. 2012;9:14. [CrossRef]
| Total | AQP4+ | MOG+ | Double- seronegative |
|
|---|---|---|---|---|
| No. of patients | 42 | 24 | 8 | 10 |
| Age at disease onset, mean (range) years | 40 (15-72) | 42 (21-72) | 35 (15-59) | 37 (18-71) |
| Disease duration*, median (IQR) months | 15 (3-72) | 48 (2.0-96) | 16 (2.5-48) | 8 (5-36) |
| Age*, mean (SD) | 45 (16.0) | 50 (15.6) | 37 (16.7) | 41(13.9) |
| EDSS* (range) | 4.0 (2-7) | 4.0 (2-7) | 3.5 (2-6.5) | 4.0 (2-6.5) |
| Sex (female) n (%) | 33 (82.5 %) | 22 (91.7 %) | 4 (50 %) | 7 (70 %) |
| Co-morbid rheumatic disease, n (%) | 6 (14.2 %) | 6 (25 %) | 0 (0 %) | 0 (0 %) |
| Malignancy, n (%) | 3 (2.4 %) | 1 (4.2 %) | 0 (0 %) | 2 (20 %) |
| Prior immunosuppressive therapy | 12 (29%) | 9 (37.5%) | 1 (12.5%) | 2 (20%) |
| Total | AQP4+ | MOG+ | Double- seronegative |
|
|---|---|---|---|---|
| Induction dose rituximab, range, mg | 250-2000 | 250 – 2000 | 500 – 1000 | 500 – 1000 |
| Maintenance dose rituximab, range, mg | 100- 1000 | 100 – 1000 | 500 – 1000 | 500 – 1000 |
| Cumulative dose rituximab, mean (median), mg |
4640 (4100) | 4946 (4650) | 3281 (3625) | 4990 (3850) |
| Withdrawal of rituximab, n (%) | 16 (38%) | 7 (29%) | 5 (63%) | 4 (40%) |
| Follow-up time, mean (min.- max.) years* |
4 (0.5-8.25) | 4.4 (0.9-7.7) | 2.6 (0.5-4.8) | 4.2 (0.8-8.25) |
| Patients (n) with a relapse during rituximab treatment |
21(50%) | 9 (37%) | 6 (75%) | 6 (60%) |
| Annual relapse rate; mean (median) | 0.55 (0.05) | 0.21 (0) | 1.7 (1.6) | 0.48 (0.25) |
| Total | AQP4+ | MOG+ | Double seronegative |
|
|---|---|---|---|---|
| Patients with ≥ SIE n (%)* | 17 (40%) | 11/24 (46%) | 2/8 (25%) | 4/10 (40%) |
| SIE/year | 0.24 | 0.32 | 0.1 | 0.18 |
| Death during SIE, n (%) | 3 (7%) | 3 (7%) | 0 (0%) | 0 (0%) |
| Type of SIE | ||||
| Urinary tract infection | 21 (50%) | 13 (54.2%) | 4 (50%) | 4 (40%) |
| Upper respiratory tract infection | 21 (50%) | 10 (41.7%) | 5 (50%) | 6 (60%) |
| Pneumonia | 11 (26.2%) | 7 (29.2%) | 2 (25%) | 2 (20%) |
| Bacterial skin infection | 11 (26.2%) | 7 (29.2%) | 0 (0%) | 4 (40%) |
| Sepsis | 7 (16.7%) | 5 (20.8%) | 0 (0%) | 2 (20%) |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




