Submitted:
26 November 2023
Posted:
12 December 2023
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Results
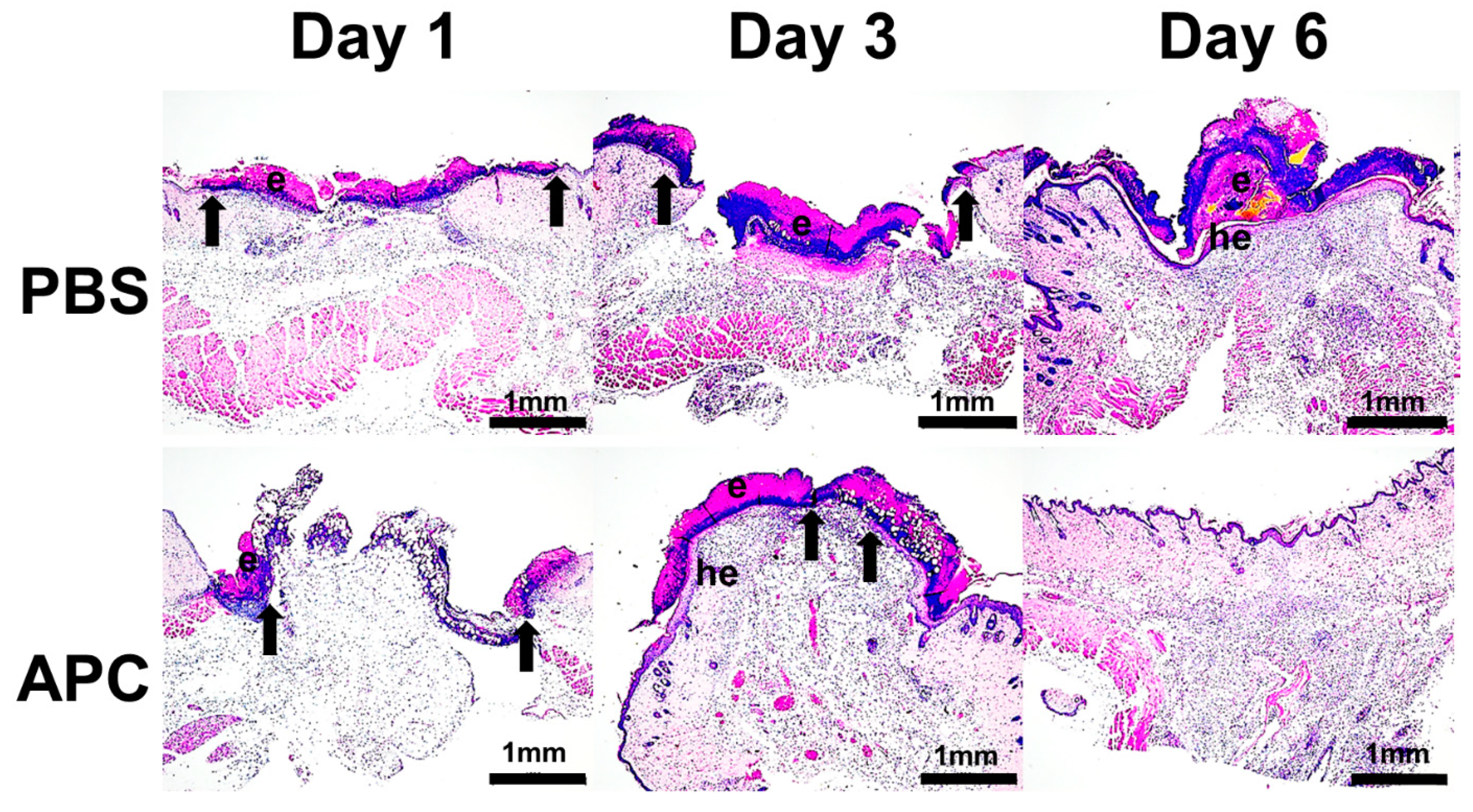
- Activated protein C accelerates murine cutaneous wound healing
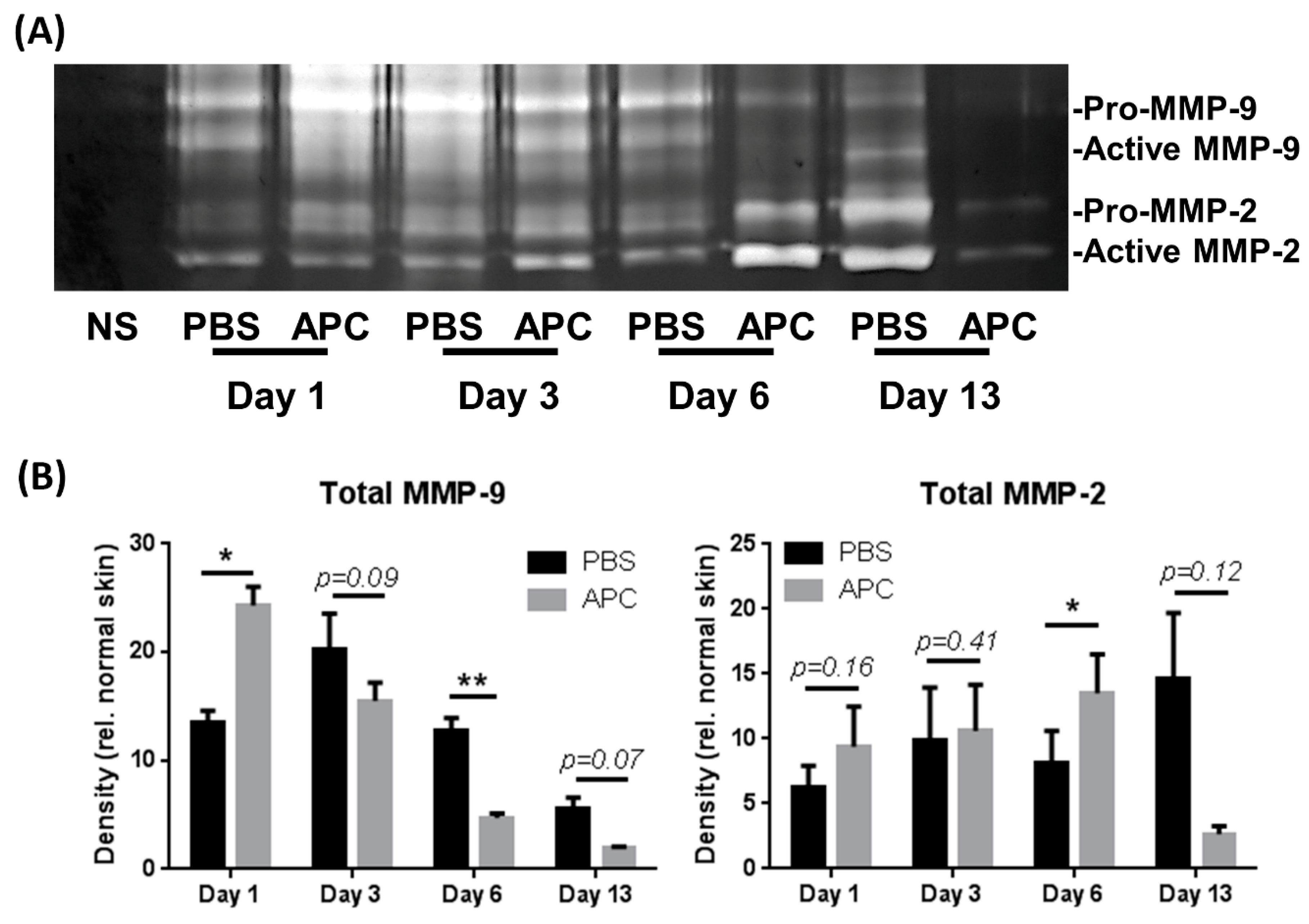
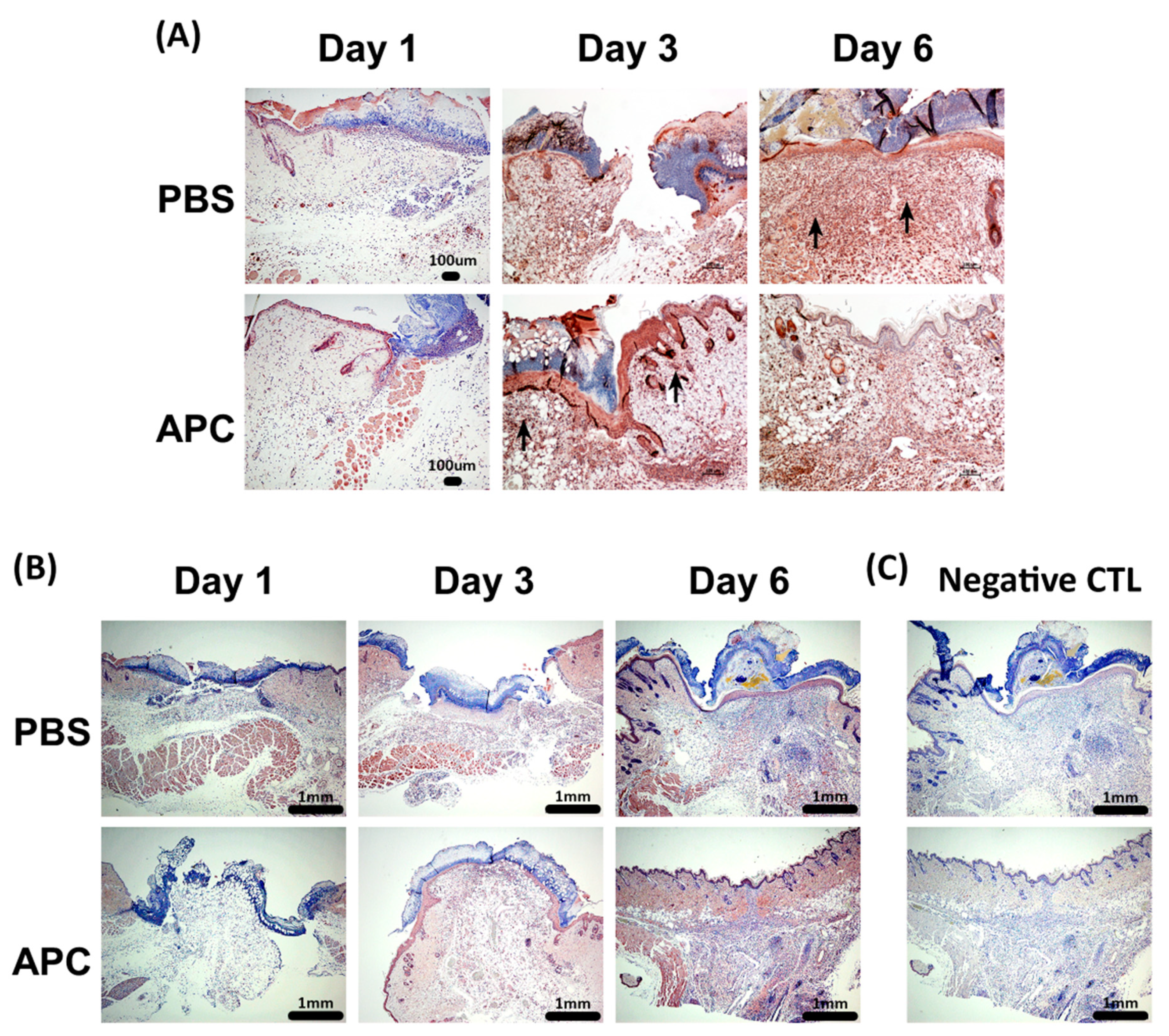
- APC-treated wounds induce activity of MMP-2 and MMP-9 earlier than control wounds
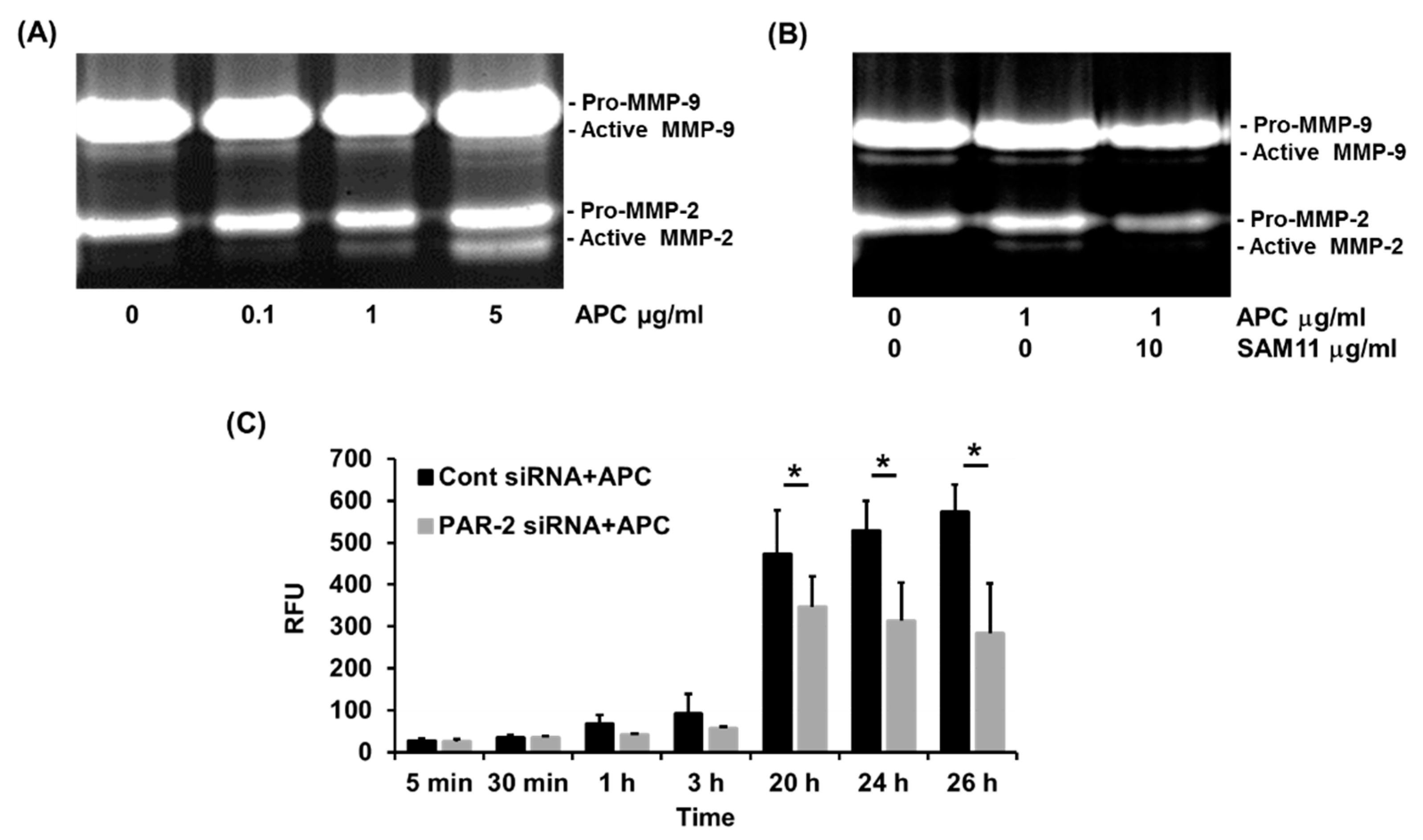
- APC-induced MMP-2 by human primary keratinocytes requires PAR-2
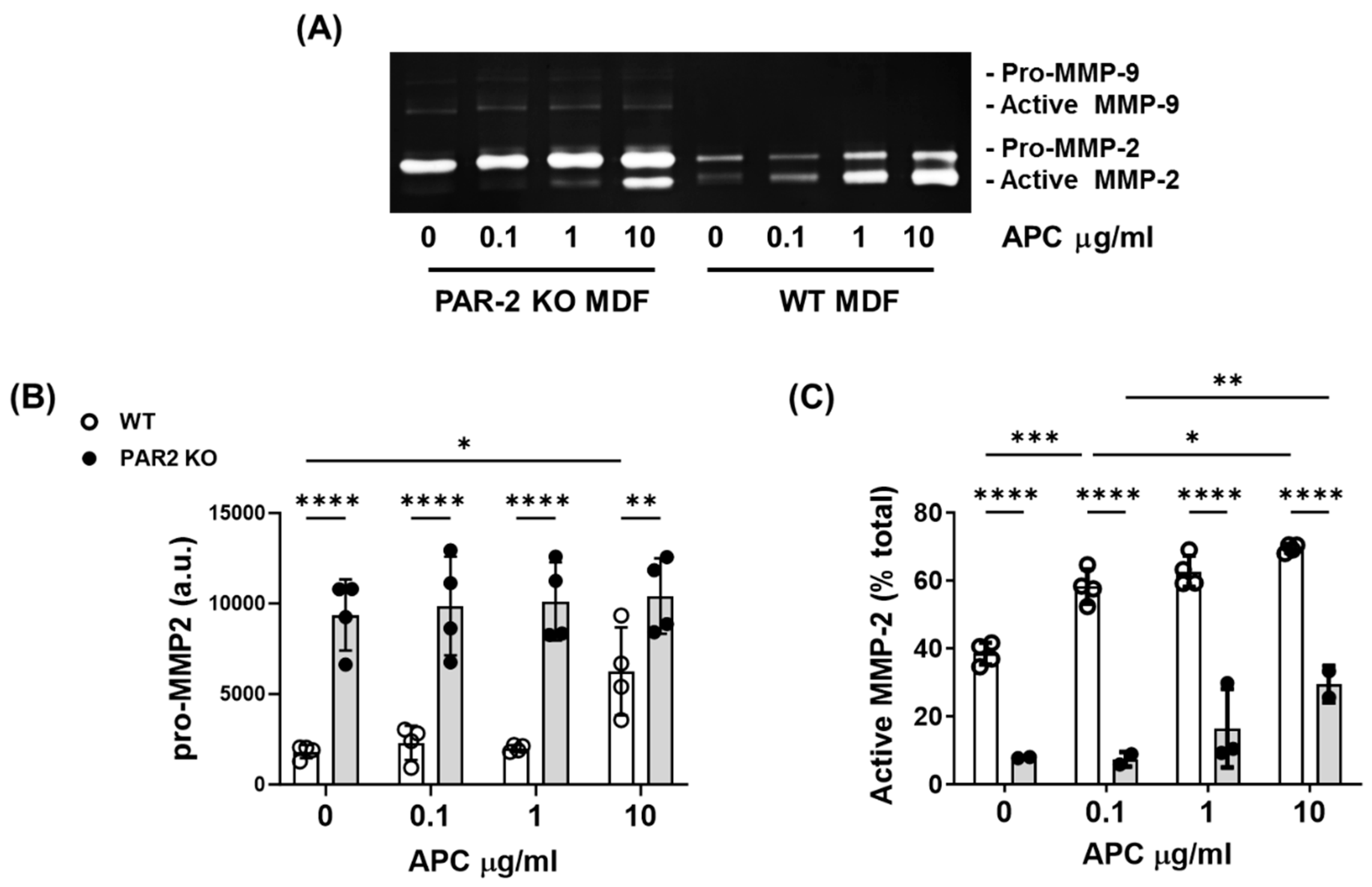
- PAR-2 deficiency reduces MMP-2 activation by murine dermal fibroblasts
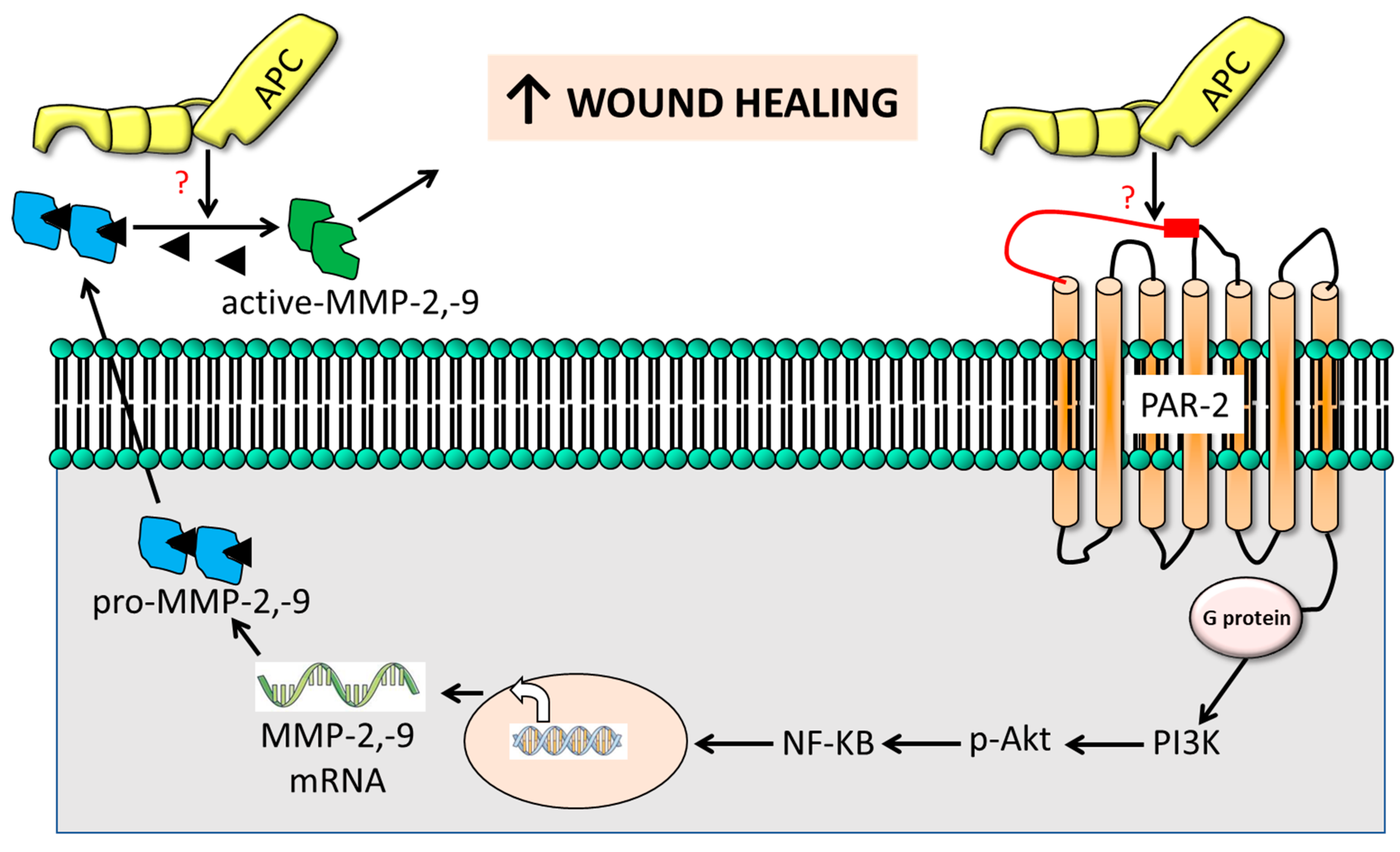
3. Discussion
4. Materials and Methods
- Animals
- Wound Healing Model
- Histology and Immunohistochemistry
- Cell Culture and Reagents
- Knockdown of PAR-2 through siRNA
- Zymography
- Fluorometric assay
- Statistical Analysis
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Gurtner, G.C., et al., Wound repair and regeneration. Nature, 2008. 453(7193): p. 314-21.
- Xue, M. and C.J. Jackson, Extracellular Matrix Reorganization During Wound Healing and Its Impact on Abnormal Scarring. Adv Wound Care (New Rochelle), 2015. 4(3): p. 119-136.
- Zhao, R., et al., Activated Protein C in Cutaneous Wound Healing: From Bench to Bedside. Int J Mol Sci, 2019. 20(4).
- Larouche, J., et al., Immune Regulation of Skin Wound Healing: Mechanisms and Novel Therapeutic Targets. Adv Wound Care (New Rochelle), 2018. 7(7): p. 209-231.
- Lechapt-Zalcman, E., et al., Transforming growth factor-beta1 increases airway wound repair via MMP-2 upregulation: a new pathway for epithelial wound repair? Am J Physiol Lung Cell Mol Physiol, 2006. 290(6): p. L1277-82.
- McCawley, L.J., P. O'Brien, and L.G. Hudson, Epidermal growth factor (EGF)- and scatter factor/hepatocyte growth factor (SF/HGF)- mediated keratinocyte migration is coincident with induction of matrix metalloproteinase (MMP)-9. J Cell Physiol, 1998. 176(2): p. 255-65.
- Rohani, M.G. and W.C. Parks, Matrix remodeling by MMPs during wound repair. Matrix Biol, 2015. 44-46: p. 113-21.
- Frøssing, S., et al., Skin wound healing in MMP2-deficient and MMP2 / Plasminogen double-deficient mice. Exp. Dermatol., 2010. 19(8): p. e234-e240.
- Kyriakides, T.R., et al., Mice that lack matrix metalloproteinase-9 display delayed wound healing associated with delayed reepithelization and disordered collagen fibrillogenesis. Matrix Biol., 2009. 28(2): p. 65-73.
- Hingorani, D.V., et al., Impact of MMP-2 and MMP-9 enzyme activity on wound healing, tumor growth and RACPP cleavage. PLoS One, 2018. 13(9): p. e0198464.
- Jackson, C.J., et al., Activated protein C prevents inflammation yet stimulates angiogenesis to promote cutaneous wound healing. Wound Repair Regen., 2005. 13(3): p. 284-294.
- Xue, M., et al., Activated protein C stimulates proliferation, migration and wound closure, inhibits apoptosis and upregulates MMP-2 activity in cultured human keratinocytes. Exp. Cell Res., 2004. 299(1): p. 119-127.
- Xue, M., et al., Activated Protein C Enhances Human Keratinocyte Barrier Integrity via Sequential Activation of Epidermal Growth Factor Receptor and Tie2. J. Biol. Chem., 2011. 286(8): p. 6742-6750.
- McKelvey, K., C.J. Jackson, and M. Xue, Activated protein C: A regulator of human skin epidermal keratinocyte function. World J Biol Chem, 2014. 5(2): p. 169-79.
- Peach, C.J., et al., Protease-activated receptors in health and disease. Physiol Rev, 2023. 103(1): p. 717-785.
- Vergnolle, N., et al., Protease-activated receptors in inflammation, neuronal signaling and pain. Trends Pharmacol. Sci., 2001. 22(3): p. 146-152.
- Julovi, S.M., et al., Protease activated receptor-2 mediates activated protein c–induced cutaneous wound healing via inhibition of p38. Am. J.Pathol., 2011. 179(5): p. 2233-2242.
- Xue, M., et al., Activated Protein C Protects against Murine Contact Dermatitis by Suppressing Protease-Activated Receptor 2. Int J Mol Sci, 2022. 23(1).
- Lan, R.S., et al., Role of PGE2 in protease-activated receptor-1, −2 and −4 mediated relaxation in the mouse isolated trachea. Br. J. Pharmacol., 2001. 132(1): p. 93-100.
- Cocks, T.M. and J.D. Moffatt, Protease-activated Receptor-2 (PAR2) in the Airways. Pulm.Pharmacol. Ther., 2001. 14(3): p. 183-191.
- Julovi, S.M., et al., Disease-modifying interactions between chronic kidney disease and osteoarthritis: a new comorbid mouse model. RMD Open, 2023. 9(3).
- Xue, M., et al., Endothelial protein c receptor and protease-activated receptor-1 mediate induction of a wound-healing phenotype in human keratinocytes by activated protein c. J. Invest. Dermatol., 2005. 125(6): p. 1279-1285.
- Julovi, S.M., et al., Activated Protein C Inhibits Proliferation and Tumor Necrosis Factor α–Stimulated Activation of p38, c-Jun NH2-Terminal Kinase (JNK) and Akt in Rheumatoid Synovial Fibroblasts. Molecular Medicine, 2013. 19(1): p. 324-331.
- Babiarz-Magee, L., et al., The Expression and Activation of Protease-Activated Receptor-2 Correlate with Skin Color. Pigment Cell Research, 2004. 17(3): p. 241-251.
- Jackson, M.T., et al., Activation of cartilage matrix metalloproteinases by activated protein C. Arthritis & Rheumatism, 2009. 60(3): p. 780-791.
- Page-McCaw, A., A.J. Ewald, and Z. Werb, Matrix metalloproteinases and the regulation of tissue remodelling. Nature reviews. Molecular cell biology, 2007. 8(3): p. 221-233.
- Whitmont, K., et al., Treatment of chronic leg ulcers with topical activated protein c. Arch. Dermatol., 2008. 144(11): p. 1479-1483.
- Whitmont, K., et al., Treatment of chronic diabetic lower leg ulcers with activated protein C: a randomised placebo-controlled, double-blind pilot clinical trial. Int. Wound J., 2013. [CrossRef]
- Wijewardena, A., et al., Combination of activated protein c and topical negative pressure rapidly regenerates granulation tissue over exposed bone to heal recalcitrant orthopedic wounds. Int. J. Low. Extrem. Wounds, 2011. 10(3): p. 146-151.
- Rattenholl, A. and S. M., Proteinase-activated receptor-2 in the skin: receptor expression, activation and function during health and disease. Drug News Perspect., 2008. 21(7): p. 369-381.
- Moormann, C., et al., Functional Characterization and Expression Analysis of the Proteinase-Activated Receptor-2 in Human Cutaneous Mast Cells. J Invest Dermatol, 2006. 126(4): p. 746-755.
- Howells, G.L., et al., Proteinase-activated receptor-2: expression by human neutrophils. Journal of Cell Science, 1997. 110(7): p. 881-887.
- Hou, L., et al., Immunolocalization of protease-activated receptor-2 in skin: receptor activation stimulates interleukin-8 secretion by keratinocytes in vitro. Immunology, 1998. 94(3): p. 356-362.
- Johansson, U., et al., Human peripheral blood monocytes express protease receptor-2 and respond to receptor activation by production of IL-6, IL-8, and IL-1β. Journal of Leukocyte Biology, 2005. 78(4): p. 967-975.
- Das, K., et al., Matrix metalloproteinase-2: A key regulator in coagulation proteases mediated human breast cancer progression through autocrine signaling. Biomed Pharmacother, 2018. 105: p. 395-406.
- Falconer, A.M.D., et al., Collagenolytic matrix metalloproteinases antagonize proteinase-activated receptor-2 activation, providing insights into extracellular matrix turnover. J Biol Chem, 2019. 294(26): p. 10266-10277.
- Wilson, S.R., et al., Amplification of MMP-2 and MMP-9 production by prostate cancer cell lines via activation of protease-activated receptors. Prostate, 2004. 60(2): p. 168-174.
- Kawao, N., et al., Signal transduction for proteinase-activated receptor-2-triggered prostaglandin E2 formation in human lung epithelial cells. J Pharmacol Exp Ther, 2005. 315(2): p. 576-89.
- Heuberger, D.M. and R.A. Schuepbach, Protease-activated receptors (PARs): mechanisms of action and potential therapeutic modulators in PAR-driven inflammatory diseases. Thromb J, 2019. 17: p. 4.
- Mendes, O., et al., MMP2 role in breast cancer brain metastasis development and its regulation by TIMP2 and ERK1/2. Clin Exp Metastasis, 2007. 24(5): p. 341-51.
- Cabral-Pacheco, G.A., et al., The Roles of Matrix Metalloproteinases and Their Inhibitors in Human Diseases. Int J Mol Sci, 2020. 21(24).
- van Hinsbergh, V.W., M.A. Engelse, and P.H. Quax, Pericellular proteases in angiogenesis and vasculogenesis. Arterioscler Thromb Vasc Biol, 2006. 26(4): p. 716-28.
- Nguyen, M., J. Arkell, and C.J. Jackson, Activated Protein C Directly Activates Human Endothelial Gelatinase A. Journal of Biological Chemistry, 2000. 275(13): p. 9095-9098.
- Jackson, C.J. and M. Xue, Activated protein C (APC) as a novel agent to promote wound healing. Primary Intention, 2006. 14(1): p. 31-34.
- Xue, M., et al., Differential regulation of matrix metalloproteinase 2 and matrix metalloproteinase 9 by activated protein C: Relevance to inflammation in rheumatoid arthritis. Arthritis & Rheumatism, 2007. 56(9): p. 2864-2874.
- Minhas, N., et al., Activated protein C utilizes the angiopoietin/Tie2 axis to promote endothelial barrier function. FASEB J., 2010. 24(3): p. 873-881.
- Xue, M. and C.J. Jackson, Autocrine Actions of Matrix Metalloproteinase (MMP)-2 Counter the Effects of MMP-9 to Promote Survival and Prevent Terminal Differentiation of Cultured Human Keratinocytes. J Invest Dermatol, 2008. 128(11): p. 2676-2685.
- Van Den Steen, P.E., et al., Gelatinase B/MMP-9 and neutrophil collagenase/MMP-8 process the chemokines human GCP-2/CXCL6, ENA-78/CXCL5 and mouse GCP-2/LIX and modulate their physiological activities. Eur J Biochem, 2003. 270(18): p. 3739-49.
- Jackson, M.T., et al., Activation of Matrix Metalloproteinases 2, 9, and 13 by Activated Protein C in Human Osteoarthritic Cartilage Chondrocytes. Arthritis & Rheumatology, 2014. 66(6): p. 1525-1536.
- McQuibban, G.A., et al., Matrix metalloproteinase processing of monocyte chemoattractant proteins generates CC chemokine receptor antagonists with anti-inflammatory properties in vivo. Blood, 2002. 100(4): p. 1160-7.
- Madhusudhan, T., B.A. Kerlin, and B. Isermann, The emerging role of coagulation proteases in kidney disease. Nat Rev Nephrol, 2016. 12(2): p. 94-109.
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).



