Submitted:
04 December 2023
Posted:
06 December 2023
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
2.1. Study Design
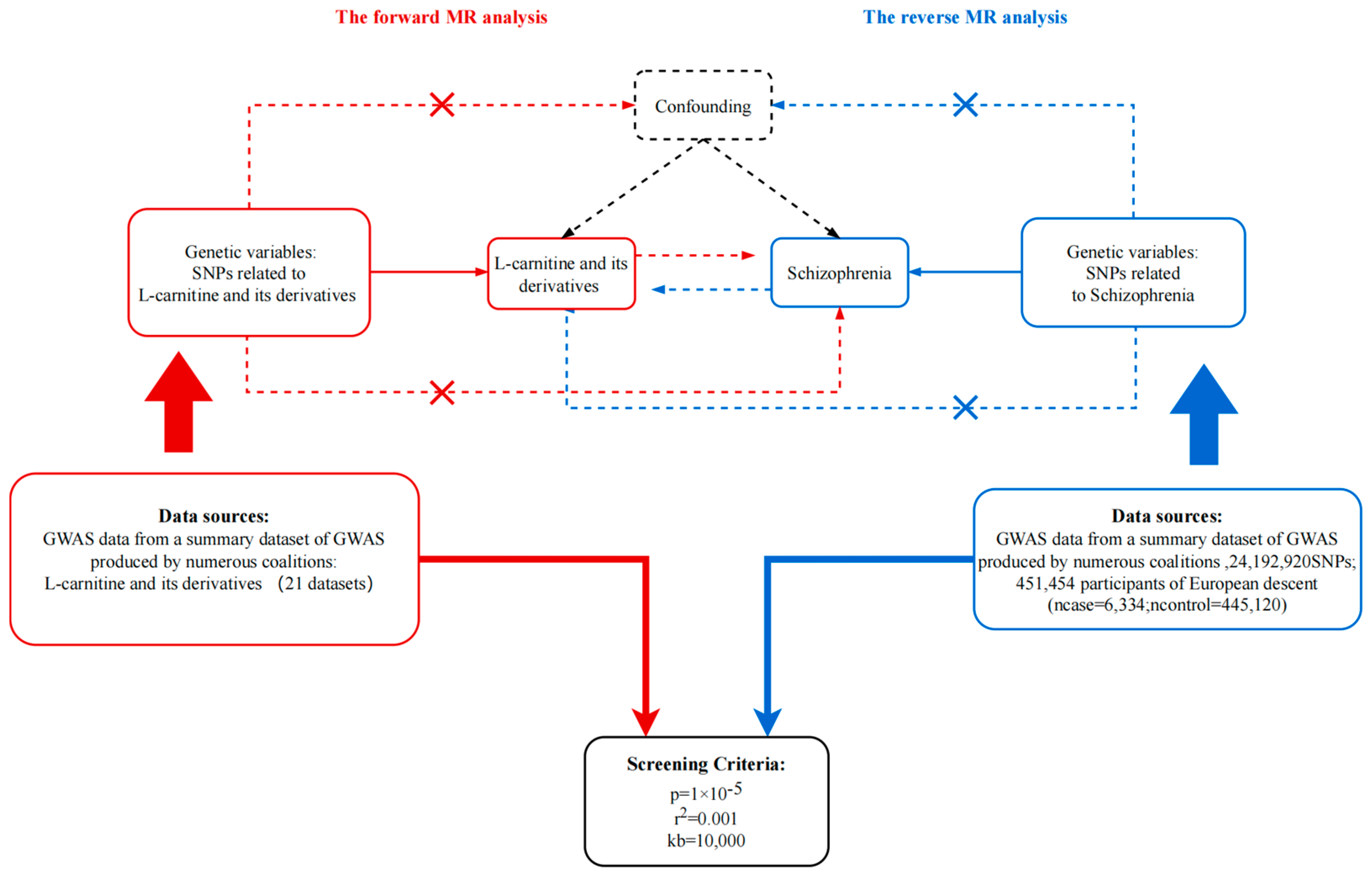
2.2. Data Sources
2.3. Identified Ivs
2.4. MR analysis
2.5. Sensitivity Analysis
2.6. Evaluation of sample overlap bias
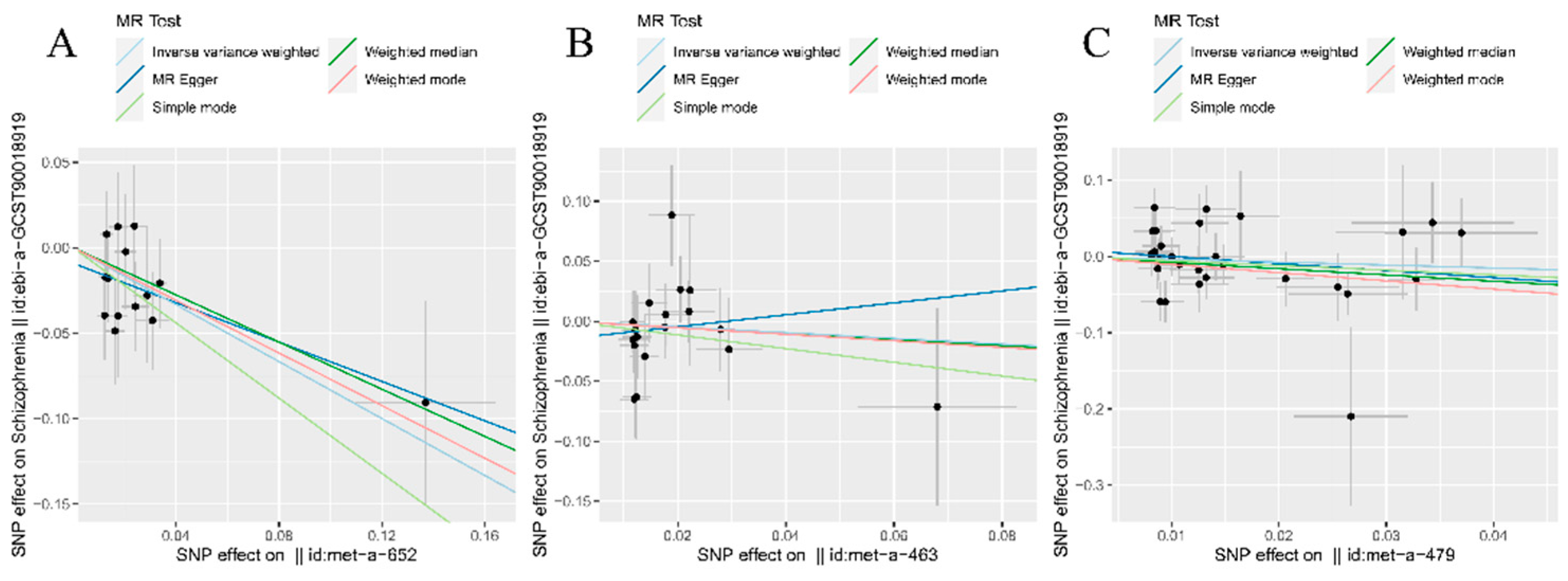
3.1. Forward analysis
3.1.1. Forward MR analysis
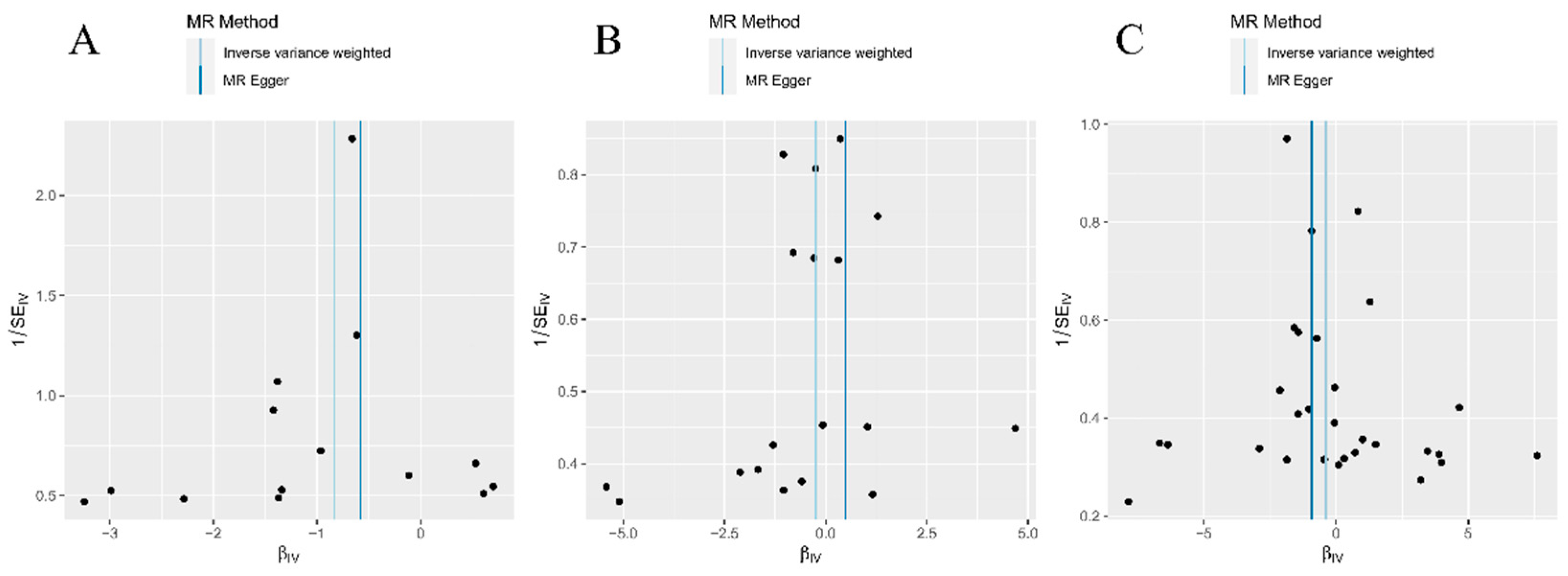
3.1.2. Forward sensitivity analysis
3.1.3. Sample overlap bias in forward analysis
3.2. Reverse analysis
| Outcome | N.SNP | PIVW | OR | 95%CI |
|---|---|---|---|---|
| Carnitine | 10 | 0.74 | 1.01 | 0.99-1.01 |
| Palmitoylcarnitine | 2 | 0.79 | 0.99 | 0.94-1.05 |
| Acetylcarnitine | 8 | 0.32 | 0.99 | 0.97-1.01 |
| Isovalerylcarnitine | 4 | 0.55 | 0.99 | 0.96-1.02 |
| Hexanoylcarnitine | 3 | 0.94 | 1.02 | 0.97-1.03 |
| Butyrylcarnitine | 3 | 0.80 | 0.99 | 0.95-1.04 |
| Propionylcarnitine | 9 | 0.37 | 0.99 | 0.98-1.01 |
| 3-dehydrocarnitine | 5 | 0.79 | 1.01 | 0.98-1.02 |
| Isobutyrylcarnitine | 4 | 0.78 | 0.99 | 0.96-1.03 |
| Octanoylcarnitine | 4 | 0.69 | 1.01 | 0.97-1.04 |
| Decanoylcarnitine | 2 | 0.79 | 1.01 | 0.96-1.05 |
| Stearoylcarnitine | 3 | 0.84 | 1.01 | 0.97-1.04 |
| Laurylcarnitine | 1 | 0.88 | 1.01 | 0.93-1.09 |
| Oleoylcarnitine | 6 | 0.88 | 1.01 | 0.98-1.02 |
| X-13431--nonanoylcarnitine | 3 | 0.89 | 1.01 | 0.96-1.04 |
| 2-methylbutyroylcarnitine | 6 | 0.88 | 0.99 | 0.97-1.02 |
| Hydroxyisovaleroyl carnitine | 7 | 0.98 | 1.00 | 0.97-1.02 |
| Glutaroyl carnitine | 3 | 0.64 | 1.00 | 0.98-1.02 |
| 2-tetradecenoyl carnitine | 4 | 0.83 | 1.00 | 0.97-1.04 |
| Succinylcarnitine | 1 | 0.82 | 0.99 | 0.97-1.03 |
| Cis-4-decenoyl carnitine | 4 | 0.62 | 1.01 | 0.98-1.04 |
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Informed Consent Statement
Data Availability Statement
Acknowledgments
References
- Saha, S.; Chant, D.; Welham, J.; McGrath, J. A systematic review of the prevalence of schizophrenia. PLoS Med 2005, 2, e141. [Google Scholar] [CrossRef]
- Jauhar, S.; Johnstone, M.; McKenna, P.J. Schizophrenia. Lancet 2022, 399, 473–486. [Google Scholar] [CrossRef] [PubMed]
- Xiu, M.H.; Li, Z.; Chen, D.C.; Chen, S.; Curbo, M.E.; Wu, H.E.; Tong, Y.S.; Tan, S.P.; Zhang, X.Y. Interrelationships Between BDNF, Superoxide Dismutase, and Cognitive Impairment in Drug-Naive First-Episode Patients With Schizophrenia. Schizophr Bull 2020, 46, 1498–1510. [Google Scholar] [CrossRef] [PubMed]
- Walsh, T.; McClellan, J.M.; McCarthy, S.E.; Addington, A.M.; Pierce, S.B.; Cooper, G.M.; Nord, A.S.; Kusenda, M.; Malhotra, D.; Bhandari, A.; et al. Rare structural variants disrupt multiple genes in neurodevelopmental pathways in schizophrenia. Science 2008, 320, 539–543. [Google Scholar] [CrossRef] [PubMed]
- Purcell, S.M.; Wray, N.R.; Stone, J.L.; Visscher, P.M.; O’Donovan, M.C.; Sullivan, P.F.; Sklar, P. Common polygenic variation contributes to risk of schizophrenia and bipolar disorder. Nature 2009, 460, 748–752. [Google Scholar] [CrossRef] [PubMed]
- Fromer, M.; Pocklington, A.J.; Kavanagh, D.H.; Williams, H.J.; Dwyer, S.; Gormley, P.; Georgieva, L.; Rees, E.; Palta, P.; Ruderfer, D.M.; et al. De novo mutations in schizophrenia implicate synaptic networks. Nature 2014, 506, 179–184. [Google Scholar] [CrossRef] [PubMed]
- Biological insights from 108 schizophrenia-associated genetic loci. Nature 2014, 511, 421–427. [CrossRef] [PubMed]
- Harrison, P.J.; Weinberger, D.R. Schizophrenia genes, gene expression, and neuropathology: on the matter of their convergence. Mol Psychiatry 2005, 10, 40–68, image 45. [Google Scholar] [CrossRef]
- Ma, L.; Rolls, E.T.; Liu, X.; Liu, Y.; Jiao, Z.; Wang, Y.; Gong, W.; Ma, Z.; Gong, F.; Wan, L. Multi-scale analysis of schizophrenia risk genes, brain structure, and clinical symptoms reveals integrative clues for subtyping schizophrenia patients. J Mol Cell Biol 2019, 11, 678–687. [Google Scholar] [CrossRef]
- Rousseau, A.F.; Dongier, A.; Colson, C.; Minguet, P.; Defraigne, J.O.; Minguet, G.; Misset, B.; Boemer, F. Serum Acylcarnitines Profile in Critically Ill Survivors According to Illness Severity and ICU Length of Stay: An Observational Study. Nutrients 2023, 15. [Google Scholar] [CrossRef]
- Di Emidio, G.; Rea, F.; Placidi, M.; Rossi, G.; Cocciolone, D.; Virmani, A.; Macchiarelli, G.; Palmerini, M.G.; D’Alessandro, A.M.; Artini, P.G.; et al. Regulatory Functions of L-Carnitine, Acetyl, and Propionyl L-Carnitine in a PCOS Mouse Model: Focus on Antioxidant/Antiglycative Molecular Pathways in the Ovarian Microenvironment. Antioxidants (Basel) 2020, 9. [Google Scholar] [CrossRef] [PubMed]
- Salic, K.; Gart, E.; Seidel, F.; Verschuren, L.; Caspers, M.; van Duyvenvoorde, W.; Wong, K.E.; Keijer, J.; Bobeldijk-Pastorova, I.; Wielinga, P.Y.; et al. Combined Treatment with L-Carnitine and Nicotinamide Riboside Improves Hepatic Metabolism and Attenuates Obesity and Liver Steatosis. Int J Mol Sci 2019, 20. [Google Scholar] [CrossRef] [PubMed]
- Sawicka, A.K.; Renzi, G.; Olek, R.A. The bright and the dark sides of L-carnitine supplementation: a systematic review. J Int Soc Sports Nutr 2020, 17, 49. [Google Scholar] [CrossRef] [PubMed]
- Chen, N.; Yang, M.; Zhou, M.; Xiao, J.; Guo, J.; He, L. L-carnitine for cognitive enhancement in people without cognitive impairment. Cochrane Database Syst Rev 2017, 3, Cd009374. [Google Scholar] [CrossRef] [PubMed]
- Hsu, P.J.; Wang, H.D.; Tseng, Y.C.; Pan, S.W.; Sampurna, B.P.; Jong, Y.J.; Yuh, C.H. L-Carnitine ameliorates congenital myopathy in a tropomyosin 3 de novo mutation transgenic zebrafish. J Biomed Sci 2021, 28, 8. [Google Scholar] [CrossRef] [PubMed]
- Flanagan, J.L.; Simmons, P.A.; Vehige, J.; Willcox, M.D.; Garrett, Q. Role of carnitine in disease. Nutr Metab (Lond) 2010, 7, 30. [Google Scholar] [CrossRef]
- Kępka, A.; Ochocińska, A.; Chojnowska, S.; Borzym-Kluczyk, M.; Skorupa, E.; Knaś, M.; Waszkiewicz, N. Potential Role of L-Carnitine in Autism Spectrum Disorder. J Clin Med 2021, 10. [Google Scholar] [CrossRef]
- Clay, H.B.; Sillivan, S.; Konradi, C. Mitochondrial dysfunction and pathology in bipolar disorder and schizophrenia. Int J Dev Neurosci 2011, 29, 311–324. [Google Scholar] [CrossRef]
- Rajasekaran, A.; Venkatasubramanian, G.; Berk, M.; Debnath, M. Mitochondrial dysfunction in schizophrenia: pathways, mechanisms and implications. Neurosci Biobehav Rev 2015, 48, 10–21. [Google Scholar] [CrossRef]
- Kriisa, K.; Leppik, L.; Balõtšev, R.; Ottas, A.; Soomets, U.; Koido, K.; Volke, V.; Innos, J.; Haring, L.; Vasar, E.; et al. Profiling of Acylcarnitines in First Episode Psychosis before and after Antipsychotic Treatment. J Proteome Res 2017, 16, 3558–3566. [Google Scholar] [CrossRef]
- Zhao, L.; Liu, H.; Wang, W.; Wang, Y.; Xiu, M.; Li, S. Carnitine metabolites and cognitive improvement in patients with schizophrenia treated with olanzapine: a prospective longitudinal study. Front Pharmacol 2023, 14, 1255501. [Google Scholar] [CrossRef] [PubMed]
- Pekala, J.; Patkowska-Sokoła, B.; Bodkowski, R.; Jamroz, D.; Nowakowski, P.; Lochyński, S.; Librowski, T. L-carnitine--metabolic functions and meaning in humans life. Curr Drug Metab 2011, 12, 667–678. [Google Scholar] [CrossRef]
- Lawlor, D.A.; Harbord, R.M.; Sterne, J.A.; Timpson, N.; Davey Smith, G. Mendelian randomization: using genes as instruments for making causal inferences in epidemiology. Stat Med 2008, 27, 1133–1163. [Google Scholar] [CrossRef] [PubMed]
- Choi, K.W.; Chen, C.Y.; Stein, M.B.; Klimentidis, Y.C.; Wang, M.J.; Koenen, K.C.; Smoller, J.W. Assessment of Bidirectional Relationships Between Physical Activity and Depression Among Adults: A 2-Sample Mendelian Randomization Study. JAMA Psychiatry 2019, 76, 399–408. [Google Scholar] [CrossRef]
- Hemani, G.; Bowden, J.; Davey Smith, G. Evaluating the potential role of pleiotropy in Mendelian randomization studies. Hum Mol Genet 2018, 27, R195–R208. [Google Scholar] [CrossRef] [PubMed]
- Shin, S.Y.; Fauman, E.B.; Petersen, A.K.; Krumsiek, J.; Santos, R.; Huang, J.; Arnold, M.; Erte, I.; Forgetta, V.; Yang, T.P.; et al. An atlas of genetic influences on human blood metabolites. Nat Genet 2014, 46, 543–550. [Google Scholar] [CrossRef] [PubMed]
- Sakaue, S.; Kanai, M.; Tanigawa, Y.; Karjalainen, J.; Kurki, M.; Koshiba, S.; Narita, A.; Konuma, T.; Yamamoto, K.; Akiyama, M.; et al. A cross-population atlas of genetic associations for 220 human phenotypes. Nat Genet 2021, 53, 1415–1424. [Google Scholar] [CrossRef]
- Zhou, X.; Lian, P.; Liu, H.; Wang, Y.; Zhou, M.; Feng, Z. Causal Associations between Gut Microbiota and Different Types of Dyslipidemia: A Two-Sample Mendelian Randomization Study. 2023, 15, 4445. [Google Scholar] [CrossRef]
- Verbanck, M.; Chen, C.Y.; Neale, B.; Do, R. Detection of widespread horizontal pleiotropy in causal relationships inferred from Mendelian randomization between complex traits and diseases. Nat Genet 2018, 50, 693–698. [Google Scholar] [CrossRef]
- Bowden, J.; Davey Smith, G.; Burgess, S. Mendelian randomization with invalid instruments: effect estimation and bias detection through Egger regression. Int J Epidemiol 2015, 44, 512–525. [Google Scholar] [CrossRef]
- Bowden, J.; Davey Smith, G.; Haycock, P.C.; Burgess, S. Consistent Estimation in Mendelian Randomization with Some Invalid Instruments Using a Weighted Median Estimator. Genet Epidemiol 2016, 40, 304–314. [Google Scholar] [CrossRef] [PubMed]
- Davey Smith, G.; Hemani, G. Mendelian randomization: genetic anchors for causal inference in epidemiological studies. Hum Mol Genet 2014, 23, R89–R98. [Google Scholar] [CrossRef]
- Hartwig, F.P.; Davey Smith, G.; Bowden, J. Robust inference in summary data Mendelian randomization via the zero modal pleiotropy assumption. Int J Epidemiol 2017, 46, 1985–1998. [Google Scholar] [CrossRef] [PubMed]
- Thaker, G.K.; Carpenter, W.T., Jr. Advances in schizophrenia. Nat Med 2001, 7, 667–671. [Google Scholar] [CrossRef] [PubMed]
- Cao, B.; Jin, M.; Brietzke, E.; McIntyre, R.S.; Wang, D.; Rosenblat, J.D.; Ragguett, R.M.; Zhang, C.; Sun, X.; Rong, C.; et al. Serum metabolic profiling using small molecular water-soluble metabolites in individuals with schizophrenia: A longitudinal study using a pre-post-treatment design. Psychiatry Clin Neurosci 2019, 73, 100–108. [Google Scholar] [CrossRef] [PubMed]
- Cao, B.; Wang, D.; Pan, Z.; McIntyre, R.S.; Brietzke, E.; Subramanieapillai, M.; Nozari, Y.; Wang, J. Metabolic profiling for water-soluble metabolites in patients with schizophrenia and healthy controls in a Chinese population: A case-control study. World J Biol Psychiatry 2020, 21, 357–367. [Google Scholar] [CrossRef] [PubMed]
- Virmani, M.A.; Biselli, R.; Spadoni, A.; Rossi, S.; Corsico, N.; Calvani, M.; Fattorossi, A.; De Simone, C.; Arrigoni-Martelli, E. Protective actions of L-carnitine and acetyl-L-carnitine on the neurotoxicity evoked by mitochondrial uncoupling or inhibitors. Pharmacol Res 1995, 32, 383–389. [Google Scholar] [CrossRef] [PubMed]
- Pennisi, M.; Lanza, G.; Cantone, M.; D’Amico, E.; Fisicaro, F.; Puglisi, V.; Vinciguerra, L.; Bella, R.; Vicari, E.; Malaguarnera, G. Acetyl-L-Carnitine in Dementia and Other Cognitive Disorders: A Critical Update. Nutrients 2020, 12. [Google Scholar] [CrossRef] [PubMed]
- Spagnoli, A.; Lucca, U.; Menasce, G.; Bandera, L.; Cizza, G.; Forloni, G.; Tettamanti, M.; Frattura, L.; Tiraboschi, P.; Comelli, M.; et al. Long-term acetyl-L-carnitine treatment in Alzheimer’s disease. Neurology 1991, 41, 1726–1726. [Google Scholar] [CrossRef]
- Sano, M.; Bell, K.; Cote, L.; Dooneief, G.; Lawton, A.; Legler, L.; Marder, K.; Naini, A.; Stern, Y.; Mayeux, R. Double-blind Parallel Design Pilot Study of Acetyl Levocarnitine in Patients With Alzheimer’s Disease. Archives of Neurology 1992, 49, 1137–1141. [Google Scholar] [CrossRef]
- Bella, R.; Biondi, R.; Raffaele, R.; Pennisi, G. Effect of acetyl-L-carnitine on geriatric patients suffering from dysthymic disorders. Int J Clin Pharmacol Res 1990, 10, 355–360. [Google Scholar]
- Garzya, G.; Corallo, D.; Fiore, A.; Lecciso, G.; Petrelli, G.; Zotti, C. Evaluation of the effects of L-acetylcarnitine on senile patients suffering from depression. Drugs Exp Clin Res 1990, 16, 101–106. [Google Scholar] [PubMed]
- Pettegrew, J.W.; Levine, J.; McClure, R.J. Acetyl-L-carnitine physical-chemical, metabolic, and therapeutic properties: relevance for its mode of action in Alzheimer’s disease and geriatric depression. Mol Psychiatry 2000, 5, 616–632. [Google Scholar] [CrossRef]
- Bertoni-Freddari, C.; Fattoretti, P.; Casoli, T.; Spagna, C.; Casell, U. Dynamic morphology of the synaptic junctional areas during aging: the effect of chronic acetyl-L-carnitine administration. Brain research 1994, 656, 359–366. [Google Scholar] [CrossRef]
- Bertoni-Freddari, C.; Fattoretti, P.; Caselli, U.; Paoloni, R. Acetylcarnitine modulation of the morphology of rat hippocampal synapses. Analytical and quantitative cytology and histology 1996, 18, 275–278. [Google Scholar]
- Bae, J.E.; Kim, J.B.; Jo, D.S.; Park, N.Y.; Kim, Y.H.; Lee, H.J.; Kim, S.H.; Kim, S.H.; Son, M.; Kim, P.; et al. Carnitine Protects against MPP(+)-Induced Neurotoxicity and Inflammation by Promoting Primary Ciliogenesis in SH-SY5Y Cells. Cells 2022, 11. [Google Scholar] [CrossRef] [PubMed]
- Ferreira, G.C.; McKenna, M.C. l-Carnitine and Acetyl-l-carnitine Roles and Neuroprotection in Developing Brain. Neurochemical Research 2017, 42, 1661–1675. [Google Scholar] [CrossRef] [PubMed]
- Scafidi, S.; Racz, J.; Hazelton, J.; McKenna, M.C.; Fiskum, G. Neuroprotection by Acetyl-L-Carnitine after Traumatic Injury to the Immature Rat Brain. Developmental Neuroscience 2011, 32, 480–487. [Google Scholar] [CrossRef]
- Xu, S.; Waddell, J.; Zhu, W.; Shi, D.; Marshall, A.D.; McKenna, M.C.; Gullapalli, R.P. In vivo longitudinal proton magnetic resonance spectroscopy on neonatal hypoxic-ischemic rat brain injury: Neuroprotective effects of acetyl-L-carnitine. Magnetic Resonance in Medicine 2015, 74, 1530–1542. [Google Scholar] [CrossRef]
- Smeland, O.B.; Meisingset, T.W.; Borges, K.; Sonnewald, U. Chronic acetyl-L-carnitine alters brain energy metabolism and increases noradrenaline and serotonin content in healthy mice. Neurochem Int 2012, 61, 100–107. [Google Scholar] [CrossRef]
- Bae, J.-E.; Kang, G.M.; Min, S.H.; Jo, D.S.; Jung, Y.-K.; Kim, K.; Kim, M.-S.; Cho, D.-H. Primary cilia mediate mitochondrial stress responses to promote dopamine neuron survival in a Parkinson’s disease model. Cell Death & Disease 2019, 10, 952. [Google Scholar] [CrossRef]
| Exposure | N.SNP | IVW | MR Egger | Weighted median | Simple mode | Weighted mode | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| OR | P | OR | P | OR | P | OR | P | OR | P | ||||||
| Carnitine | 19 | 0.43 | 0.34 | 0.10 | 0.23 | 0.38 | 0.44 | 0.46 | 0.67 | 0.41 | 0.53 | ||||
| Palmitoylcarnitine | 9 | 0.93 | 0.88 | 0.88 | 0.92 | 0.82 | 0.76 | 0.57 | 0.58 | 0.76 | 0.73 | ||||
| Acetylcarnitine | 15 | 0.88 | 0.79 | 2.37 | 0.52 | 1.27 | 0.72 | 0.46 | 0.44 | 1.35 | 0.71 | ||||
| Isovalerylcarnitine | 16 | 0.47 | 0.01 | 0.48 | 0.13 | 0.51 | 0.13 | 0.34 | 0.08 | 0.46 | 0.06 | ||||
| 2-methylbutyroylcarnitine | 24 | 1.52 | 0.37 | 0.72 | 0.82 | 2.38 | 0.19 | 6.60 | 0.17 | 5.30 | 0.20 | ||||
| 2-tetradecenoyl carnitine | 17 | 0.78 | 0.29 | 0.82 | 0.67 | 0.82 | 0.51 | 0.52 | 0.24 | 0.78 | 0.48 | ||||
| Butyrylcarnitine | 15 | 0.80 | 0.13 | 0.82 | 0.46 | 0.86 | 0.39 | 0.80 | 0.51 | 0.85 | 0.42 | ||||
| Hexanoylcarnitine | 14 | 0.78 | 0.38 | 0.69 | 0.52 | 0.48 | 0.07 | 0.68 | 0.66 | 0.46 | 0.08 | ||||
| Octanoylcarnitine | 13 | 2.11 | 0.10 | 5.08 | 0.31 | 3.38 | 0.05 | 7.46 | 0.07 | 6.38 | 0.14 | ||||
| Glutaroyl carnitine | 12 | 0.63 | 0.13 | 0.16 | 0.01 | 0.42 | 0.06 | 0.68 | 0.66 | 0.37 | 0.10 | ||||
| Laurylcarnitine | 12 | 0.74 | 0.45 | 1.37 | 0.80 | 0.96 | 0.94 | 2.25 | 0.39 | 1.92 | 0.43 | ||||
| Propionylcarnitine | 12 | 0.58 | 0.41 | 2.86 | 0.47 | 0.60 | 0.52 | 0.54 | 0.59 | 0.56 | 0.55 | ||||
| Decanoylcarnitine | 11 | 0.63 | 0.13 | 0.32 | 0.06 | 0.45 | 0.03 | 0.64 | 0.44 | 0.47 | 0.09 | ||||
| Oleoylcarnitine | 11 | 1.23 | 0.69 | 0.30 | 0.41 | 0.71 | 0.57 | 0.63 | 0.64 | 0.60 | 0.56 | ||||
| Cis-4-decenoyl carnitine | 9 | 0.56 | 0.10 | 0.63 | 0.57 | 0.44 | 0.06 | 0.47 | 0.39 | 0.41 | 0.09 | ||||
| Isobutyrylcarnitine | 9 | 1.20 | 0.54 | 0.84 | 0.78 | 0.97 | 0.94 | 1.60 | 0.50 | 0.91 | 0.83 | ||||
| X-13431--nonanoylcarnitine | 9 | 0.65 | 0.36 | 1.08 | 0.95 | 0.54 | 0.26 | 0.31 | 0.22 | 0.28 | 0.27 | ||||
| Hydroxyisovaleroyl carnitine | 8 | 0.64 | 0.27 | 0.64 | 0.53 | 0.67 | 0.43 | 0.79 | 0.72 | 0.66 | 0.47 | ||||
| Succinylcarnitine | 7 | 0.42 | 0.52 | 789.56 | 0.25 | 1.04 | 0.98 | 4.20 | 0.63 | 5.30 | 0.50 | ||||
| 3-dehydrocarnitine | 5 | 1.42 | 0.60 | 102.79 | 0.30 | 1.00 | 1.00 | 1.14 | 0.92 | 0.93 | 0.94 | ||||
| Stearoylcarnitine | 5 | 0.46 | 0.30 | 0.05 | 0.60 | 0.85 | 0.87 | 1.28 | 0.87 | 1.27 | 0.88 | ||||
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).




