1. Introduction
In enzymology, the term "serine/threonine protein kinase" refers to a category of enzymes within the transferase family. These enzymes are responsible for transferring phosphates to the oxygen atom of either a serine or threonine side chain in proteins, a biochemical process known as phosphorylation. Protein phosphorylation holds substantial importance in numerous cellular processes and serves as a crucial posttranslational modification[
1,
2] .
Serine/threonine kinases play pivotal roles in various cellular processes, contributing to the regulation of fundamental biological functions[
1,
2] . Here's an overview of their involvement:
Serine/threonine kinases are key regulators of cell cycle progression, controlling the transitions between different phases of the cell cycle. They influence processes such as DNA replication and cell division, thereby impacting cell proliferation[
3].
Programmed Cell Death (Apoptosis):
These kinases are involved in the intricate signaling pathways that govern apoptosis, a highly regulated and essential process for the removal of damaged or unnecessary cells. Serine/threonine kinases can either promote or inhibit apoptosis, depending on the context and specific signaling pathways involved[
1].
- -
Cell Differentiation:
Serine/threonine kinases contribute to the regulation of cell differentiation, the process by which cells become specialized and acquire distinct functions. They influence signaling pathways that guide cells towards specific fates, determining their roles in tissues and organs[
4,
5].
- -
Embryonic Development:
During embryonic development, serine/threonine kinases play crucial roles in orchestrating the intricate processes of cell division, differentiation, and tissue morphogenesis. They contribute to the formation and patterning of tissues and organs[
6].
- -
Signal Transduction Pathways:
Serine/threonine kinases are integral components of various signaling pathways that relay information from the cell surface to the nucleus. These pathways are critical for cells to respond to extracellular signals and adapt to changes in their environment[
7,
8].
Examples of serine/threonine kinases include members of the cyclin-dependent kinase (CDK) family, mitogen-activated protein kinase (MAPK) family, and protein kinase B (Akt), among others[
2,
9].
This brief theoretical study seeks to explore the potential role of Amentoflavone, a natural substance through an in silico approach employing Molecular Docking [
10], by Mcule Database [
11].
General speaking, Amentoflavone, categorized as a biflavonoid, belongs to the flavonoid class characterized by the coupling of two apigenin molecules. Specifically, amentoflavone features the coupling of these apigenin units at the 8 and 3′ positions, resulting in the formation of a structure termed 3′,8″-biapigenin. This compound is present in various plants, including Ginkgo biloba. Flavonoids, such as biflavonoids like amentoflavone, are renowned for their potential health benefits and antioxidant properties. Ginkgo biloba, a plant with a history in traditional herbal medicine, contains amentoflavone as one of its phytochemical constituents, contributing to the overall bioactivity of the plant [
12,
13].
3. Results and Discussion
Serine/threonine-protein kinases are a class of enzymes within the transferase family. They play crucial roles in cellular processes, including the regulation of cell cycle progression, programmed cell death (apoptosis), cell differentiation, and embryonic development. These kinases catalyze the transfer of phosphate groups to serine or threonine residues in proteins, a process known as phosphorylation. This posttranslational modification influences protein activity and function, contributing to the intricate signaling pathways that govern various biological functions. Examples of serine/threonine-protein kinases include cyclin-dependent kinases, mitogen-activated protein kinases, and protein kinase B (Akt). Understanding their functions is essential for unraveling the complexities of cell biology and has implications for therapeutic development in diseases associated with dysregulated cellular processes[
1,
2,
3,
4,
5,
6,
7,
8].
This study specifically delves into the potential interaction of Amentoflavone with Serine/threonine-protein kinases using Molecular Docking [
10] via the Mcule Database[
11].
By employing Molecular Docking to compare binding energies across various serine/threonine-protein kinases, Amentoflavone emerges as a potentially key molecule influencing several biological processes.
Table 1 illustrates the binding energy scores (kcal/mol) of amentoflavone with the investigated serine/threonine-protein kinases. These scores provide valuable insights into the strength and specificity of the interactions, shedding light on the potential significance of Amentoflavone in modulating the activities of these kinases and, consequently, various cellular processes.
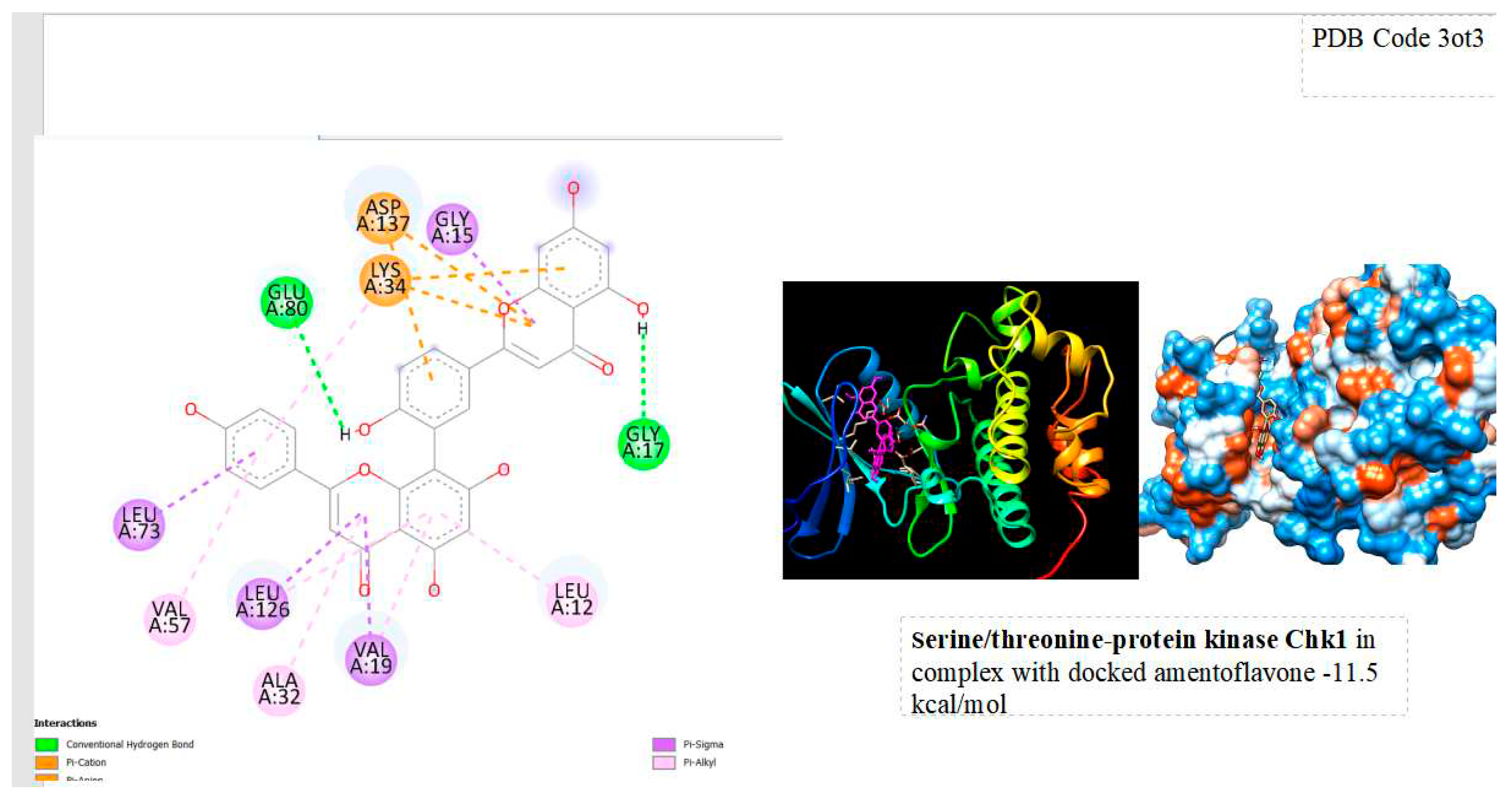
Figure 1.
displays the docking outcomes of Serine/threonine-protein kinase Chk1 in conjunction with Amentoflavone within the Ligand Binding Site, as analyzed by Autodock Vina through the Mcule Database. On the left side, 2D diagrams illustrate the residue interactions between the protein and Amentoflavone. Meanwhile, the right side exhibits the Ligand Binding Site of the protein, highlighting the specific location of Amentoflavone.
Figure 1.
displays the docking outcomes of Serine/threonine-protein kinase Chk1 in conjunction with Amentoflavone within the Ligand Binding Site, as analyzed by Autodock Vina through the Mcule Database. On the left side, 2D diagrams illustrate the residue interactions between the protein and Amentoflavone. Meanwhile, the right side exhibits the Ligand Binding Site of the protein, highlighting the specific location of Amentoflavone.
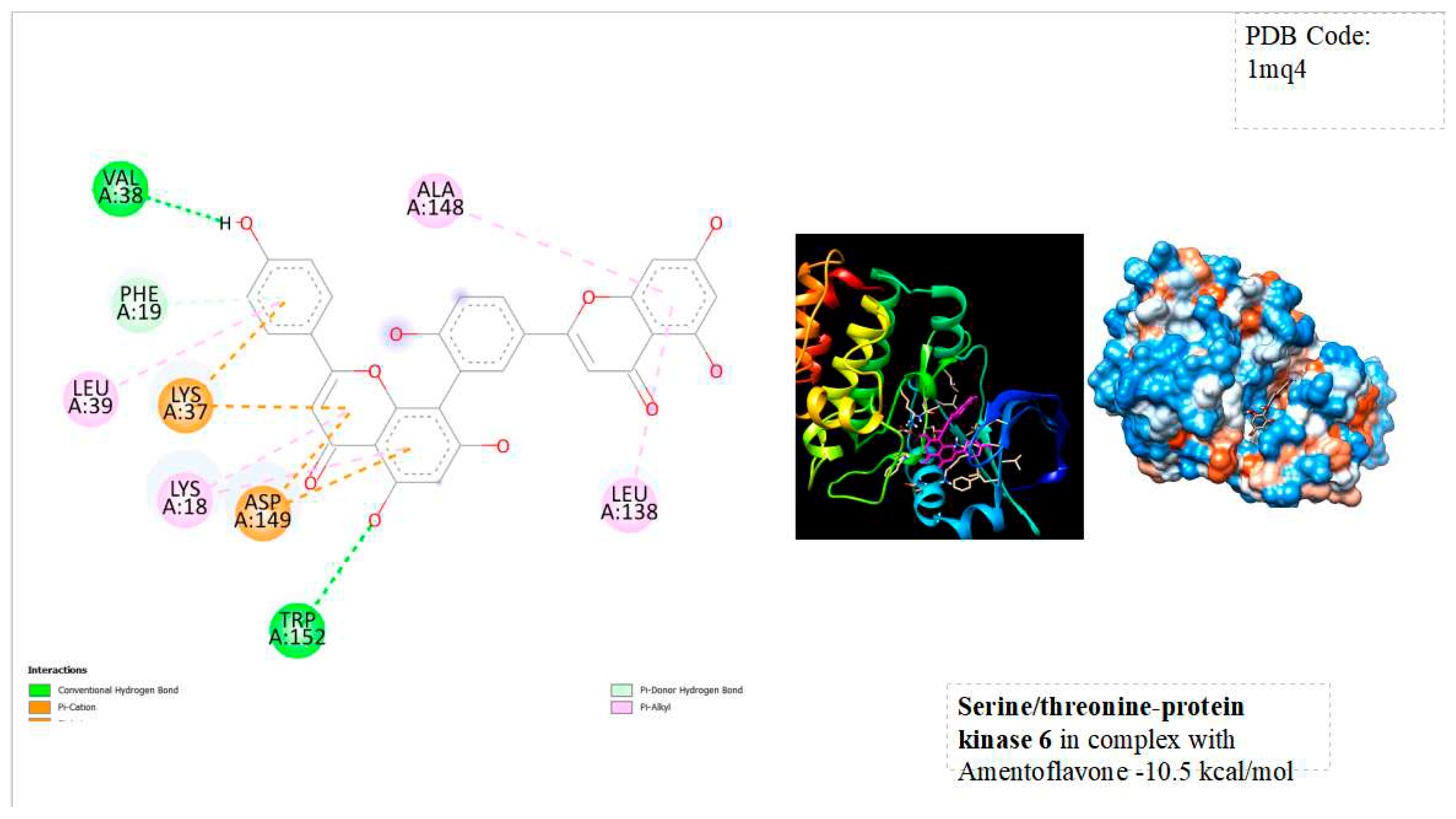
Figure 2.
displays the docking outcomes of Serine/threonine-protein kinase 6 in conjunction with Amentoflavone within the Ligand Binding Site, as analyzed by Autodock Vina through the Mcule Database. On the left side, 2D diagrams illustrate the residue interactions between the protein and Amentoflavone. Meanwhile, the right side exhibits the Ligand Binding Site of the protein, highlighting the specific location of Amentoflavone.
Figure 2.
displays the docking outcomes of Serine/threonine-protein kinase 6 in conjunction with Amentoflavone within the Ligand Binding Site, as analyzed by Autodock Vina through the Mcule Database. On the left side, 2D diagrams illustrate the residue interactions between the protein and Amentoflavone. Meanwhile, the right side exhibits the Ligand Binding Site of the protein, highlighting the specific location of Amentoflavone.
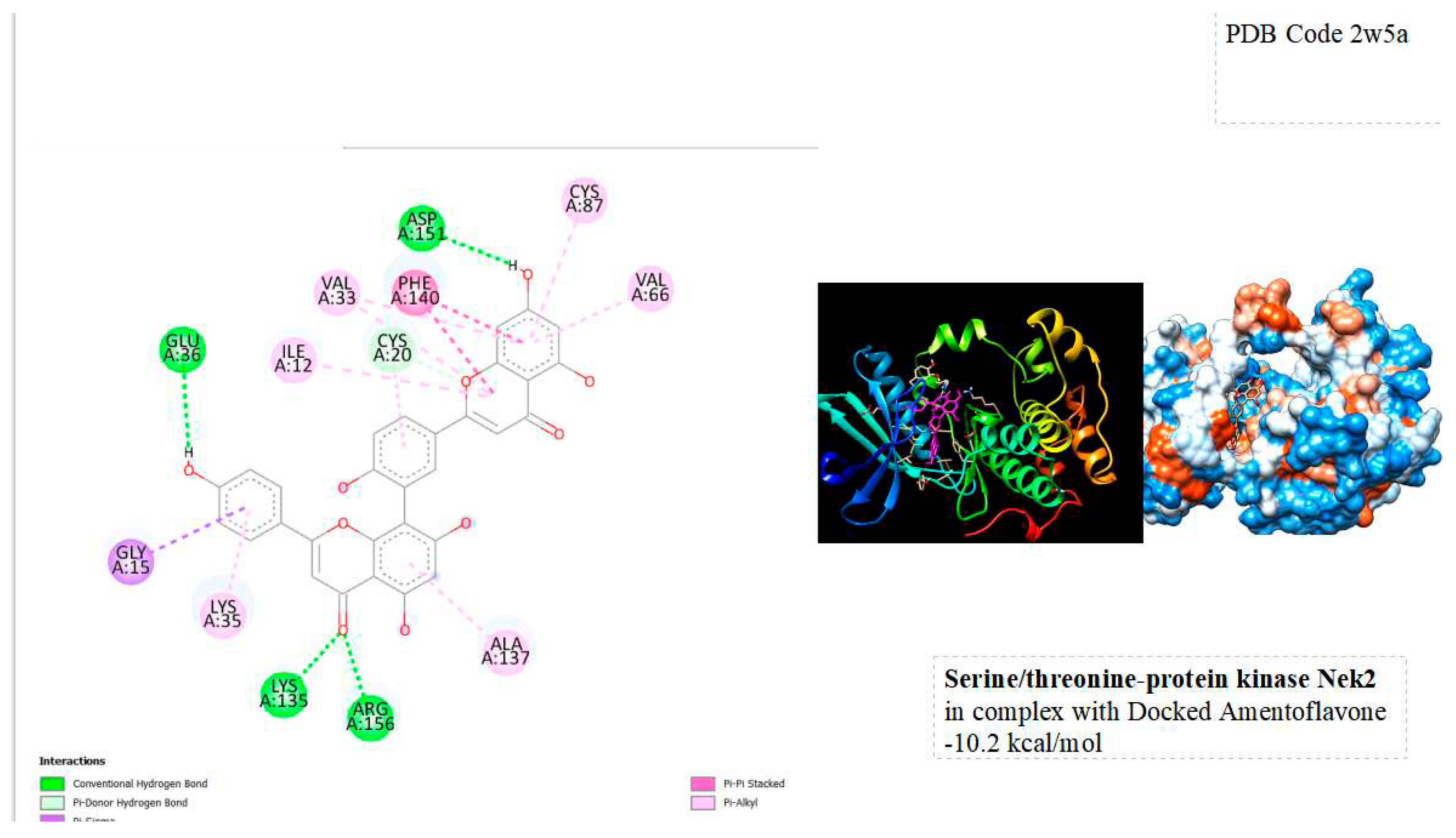
Figure 3.
displays the docking outcomes of Serine/threonine-protein kinase Nek2 in conjunction with Amentoflavone within the Ligand Binding Site, as analyzed by Autodock Vina through the Mcule Database. On the left side, 2D diagrams illustrate the residue interactions between the protein and Amentoflavone. Meanwhile, the right side exhibits the Ligand Binding Site of the protein, highlighting the specific location of Amentoflavone.
Figure 3.
displays the docking outcomes of Serine/threonine-protein kinase Nek2 in conjunction with Amentoflavone within the Ligand Binding Site, as analyzed by Autodock Vina through the Mcule Database. On the left side, 2D diagrams illustrate the residue interactions between the protein and Amentoflavone. Meanwhile, the right side exhibits the Ligand Binding Site of the protein, highlighting the specific location of Amentoflavone.
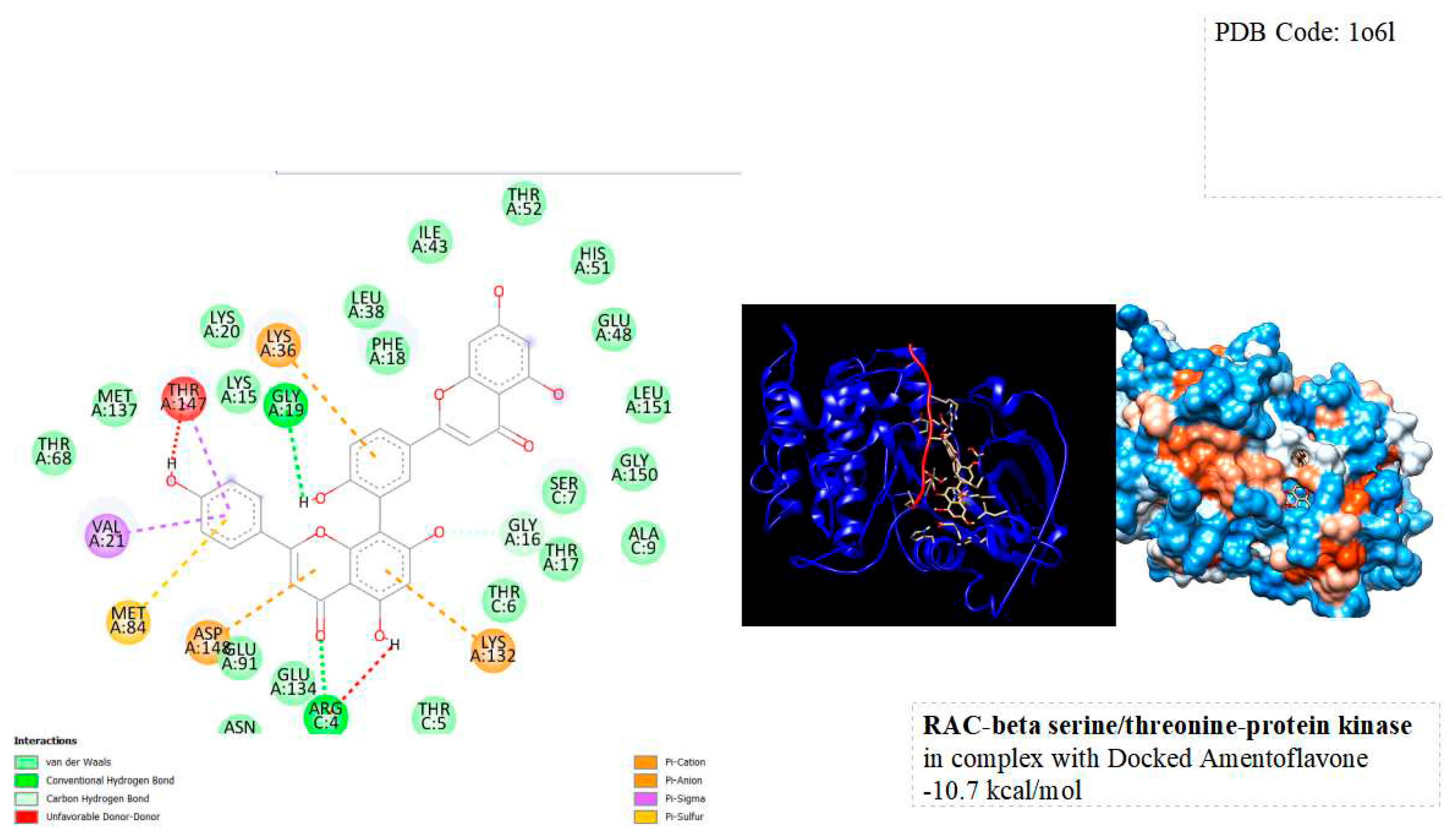
Figure 4.
displays the docking outcomes of RAC-Beta Serine/threonine-protein kinase in conjunction with Amentoflavone within the Ligand Binding Site, as analyzed by Autodock Vina through the Mcule Database. On the left side, 2D diagrams illustrate the residue interactions between the protein and Amentoflavone. Meanwhile, the right side exhibits the Ligand Binding Site of the protein, highlighting the specific location of Amentoflavone.
Figure 4.
displays the docking outcomes of RAC-Beta Serine/threonine-protein kinase in conjunction with Amentoflavone within the Ligand Binding Site, as analyzed by Autodock Vina through the Mcule Database. On the left side, 2D diagrams illustrate the residue interactions between the protein and Amentoflavone. Meanwhile, the right side exhibits the Ligand Binding Site of the protein, highlighting the specific location of Amentoflavone.
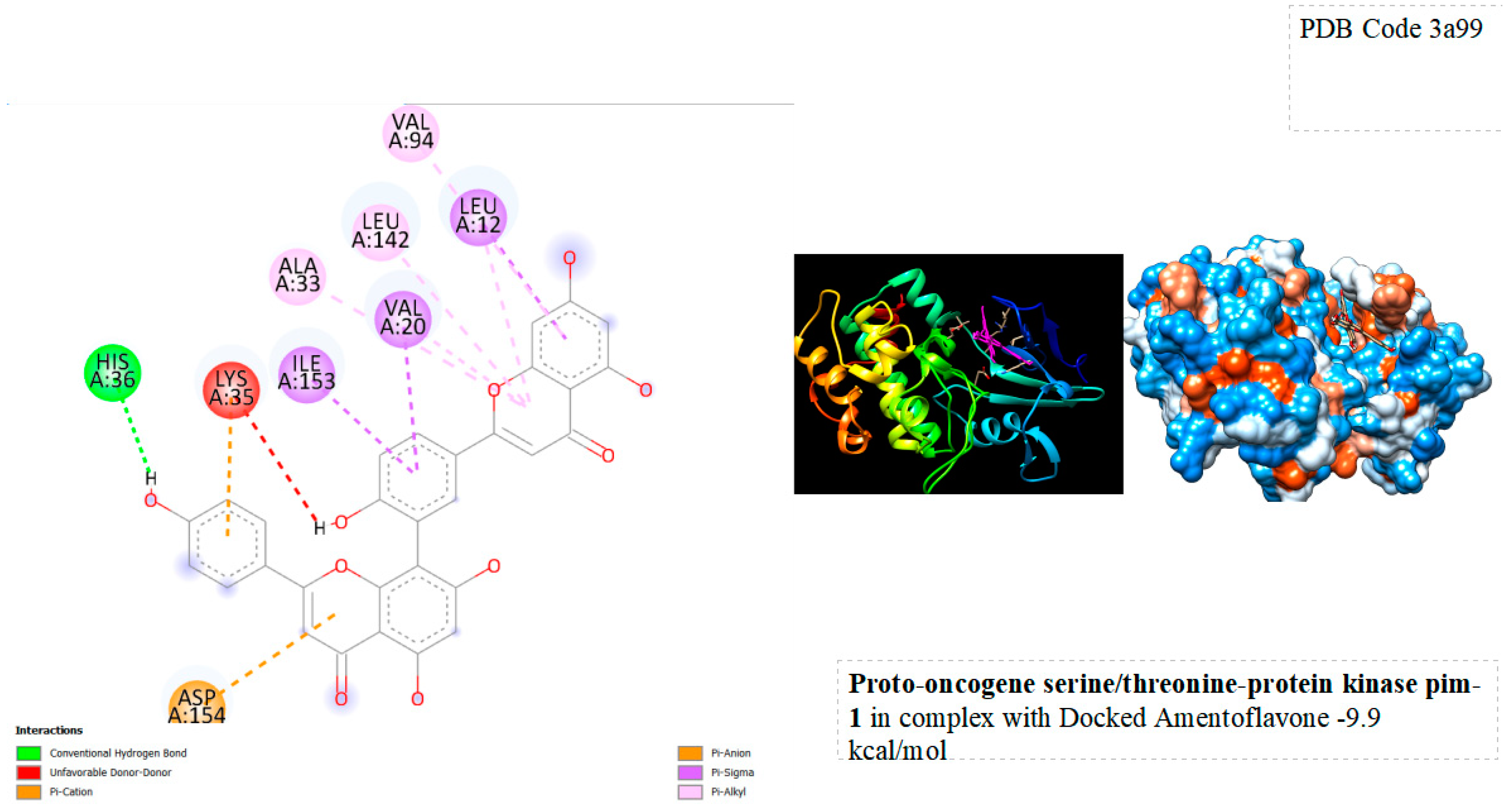
Figure 5.
displays the docking outcomes of Proto-oncogene Serine/threonine-protein kinase pim-1 in conjunction with Amentoflavone within the Ligand Binding Site, as analyzed by Autodock Vina through the Mcule Database. On the left side, 2D diagrams illustrate the residue interactions between the protein and Amentoflavone. Meanwhile, the right side exhibits the Ligand Binding Site of the protein, highlighting the specific location of Amentoflavone.
Figure 5.
displays the docking outcomes of Proto-oncogene Serine/threonine-protein kinase pim-1 in conjunction with Amentoflavone within the Ligand Binding Site, as analyzed by Autodock Vina through the Mcule Database. On the left side, 2D diagrams illustrate the residue interactions between the protein and Amentoflavone. Meanwhile, the right side exhibits the Ligand Binding Site of the protein, highlighting the specific location of Amentoflavone.
Table 1.
Comparison of Histone deacetylases with Curcumin evaluated by Mcule Database with Autodock Vina.
Table 1.
Comparison of Histone deacetylases with Curcumin evaluated by Mcule Database with Autodock Vina.
| serine/threonine-protein kinases |
Binding Energies ( kcal/mol) |
| RAC-beta serine/threonine-protein kinase |
-10.7 |
| Serine/threonine-protein kinase Chk1 |
-11.5 |
| Serine/threonine-protein kinase 6 |
-10.5 |
| Serine/threonine-protein kinase Nek2 |
-10.2 |
Proto-oncogene serine/threonine-protein kinase
pim-1 |
-9.9 |
| Serine/threonine-protein kinase PLK1 |
-8.4 |