Submitted:
21 November 2023
Posted:
22 November 2023
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Two-carbon linker chain
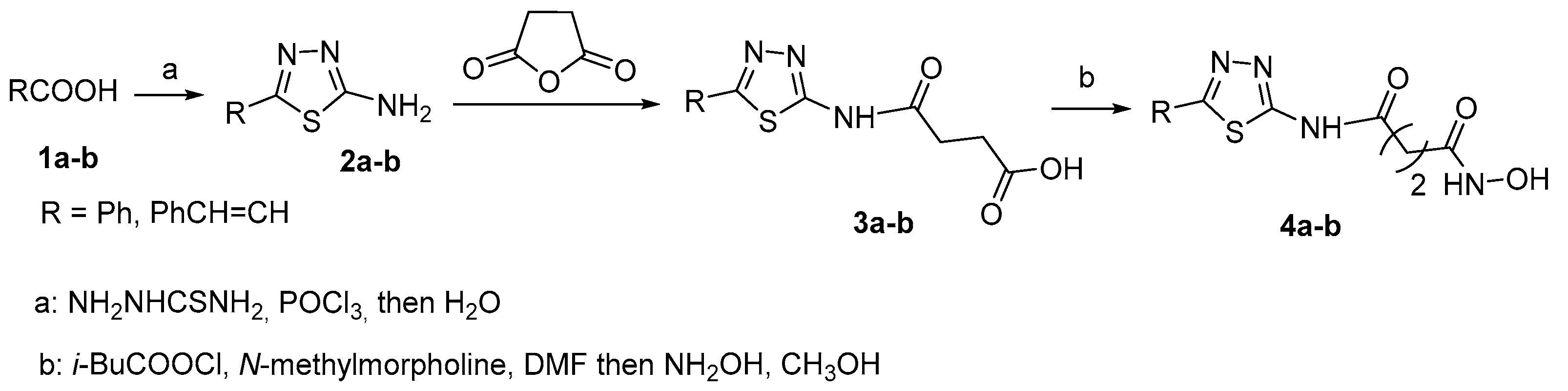
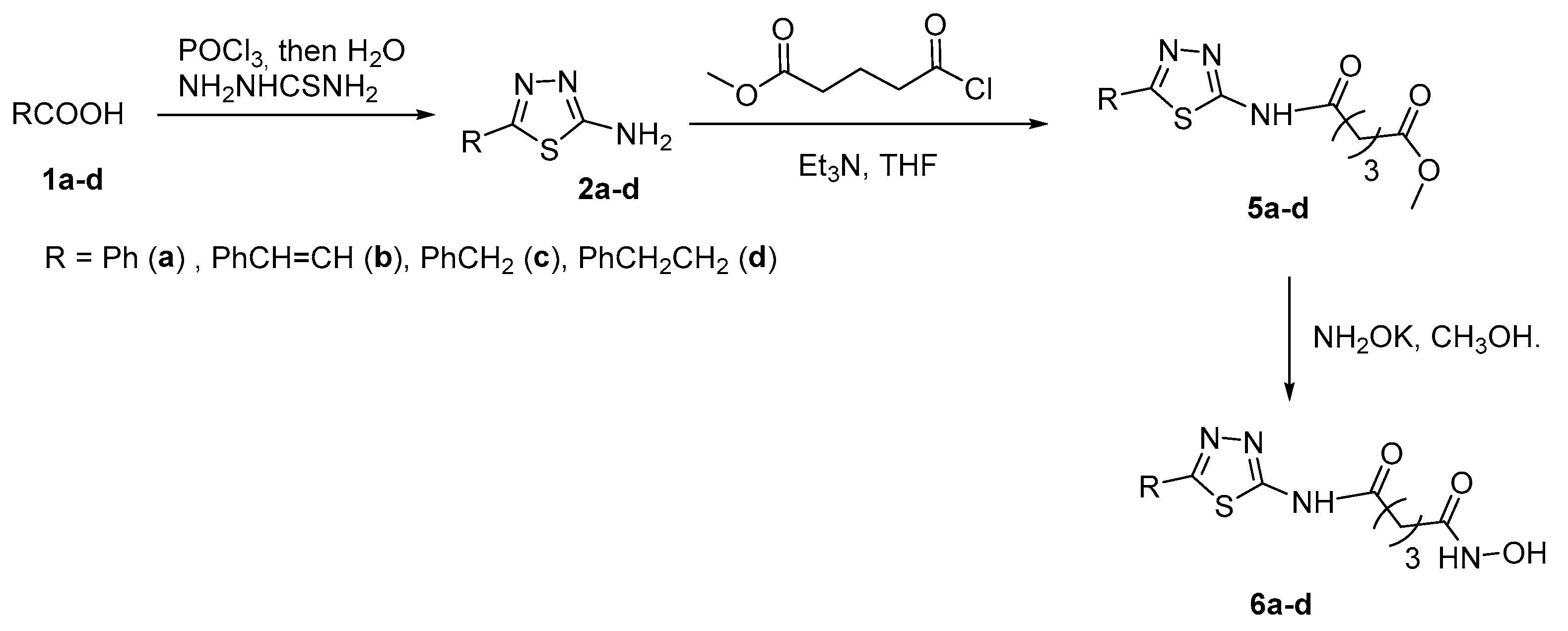
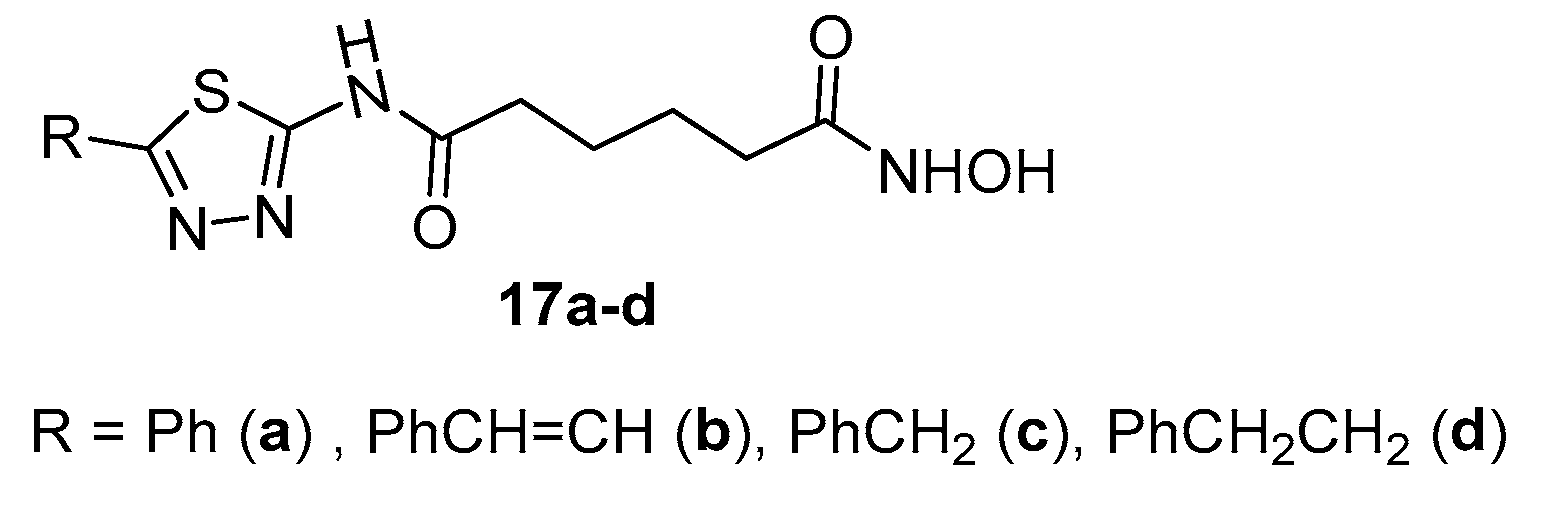
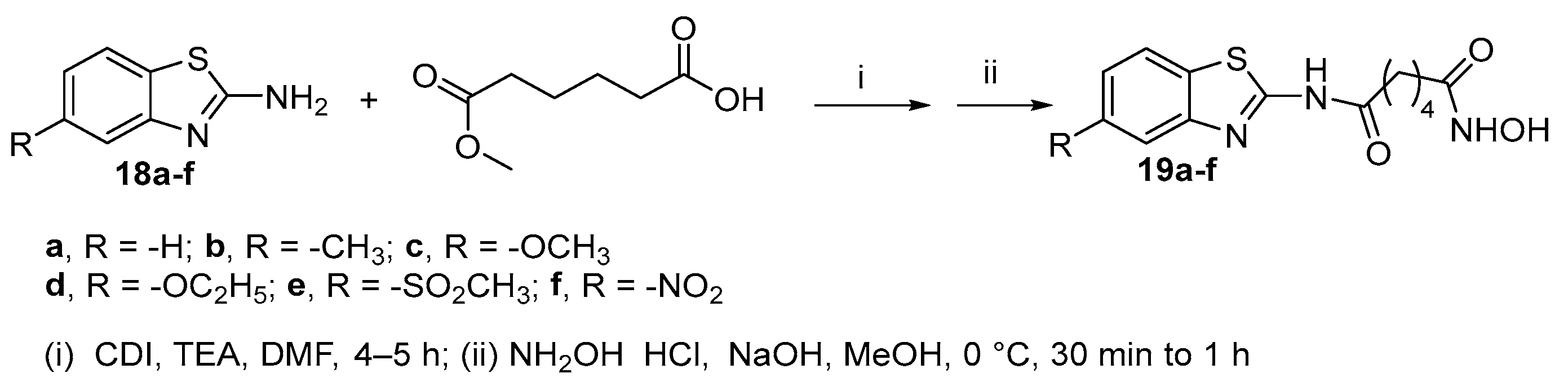
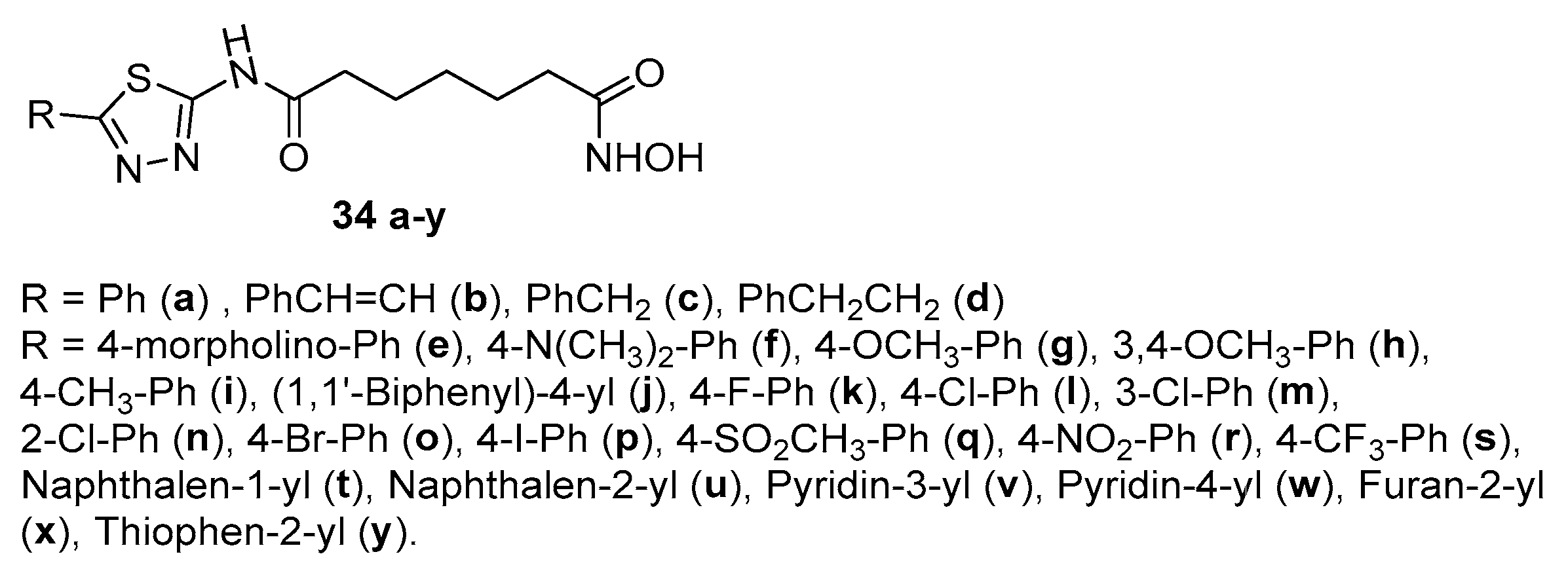
2.1. 2-amino-1,3,4-thiadiazoles in CAP group
3. Three-carbon linker chain
3.1. 2-amino-1,3,4-thiadiazoles in CAP group
3.2. Indazoles in CAP group
3.3. Benzothiazoles in CAP group
3.4. 4-Anilinothieno[2,3-d]pyrimidine derivatives in CAP group
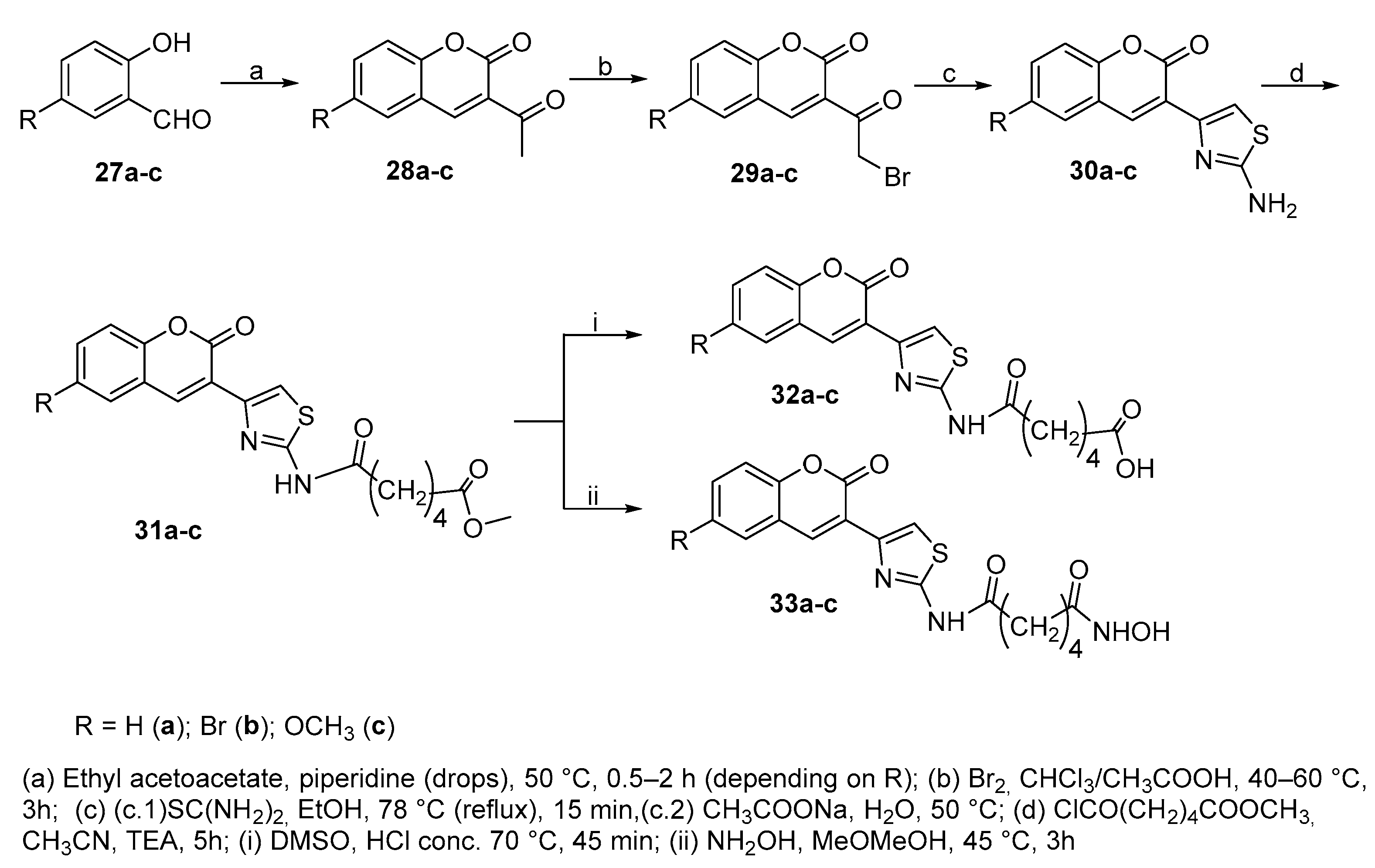
3.5. Thiazolyl-coumarin derivatives in CAP group
4. Five-carbon linker chain (5-C spacer)
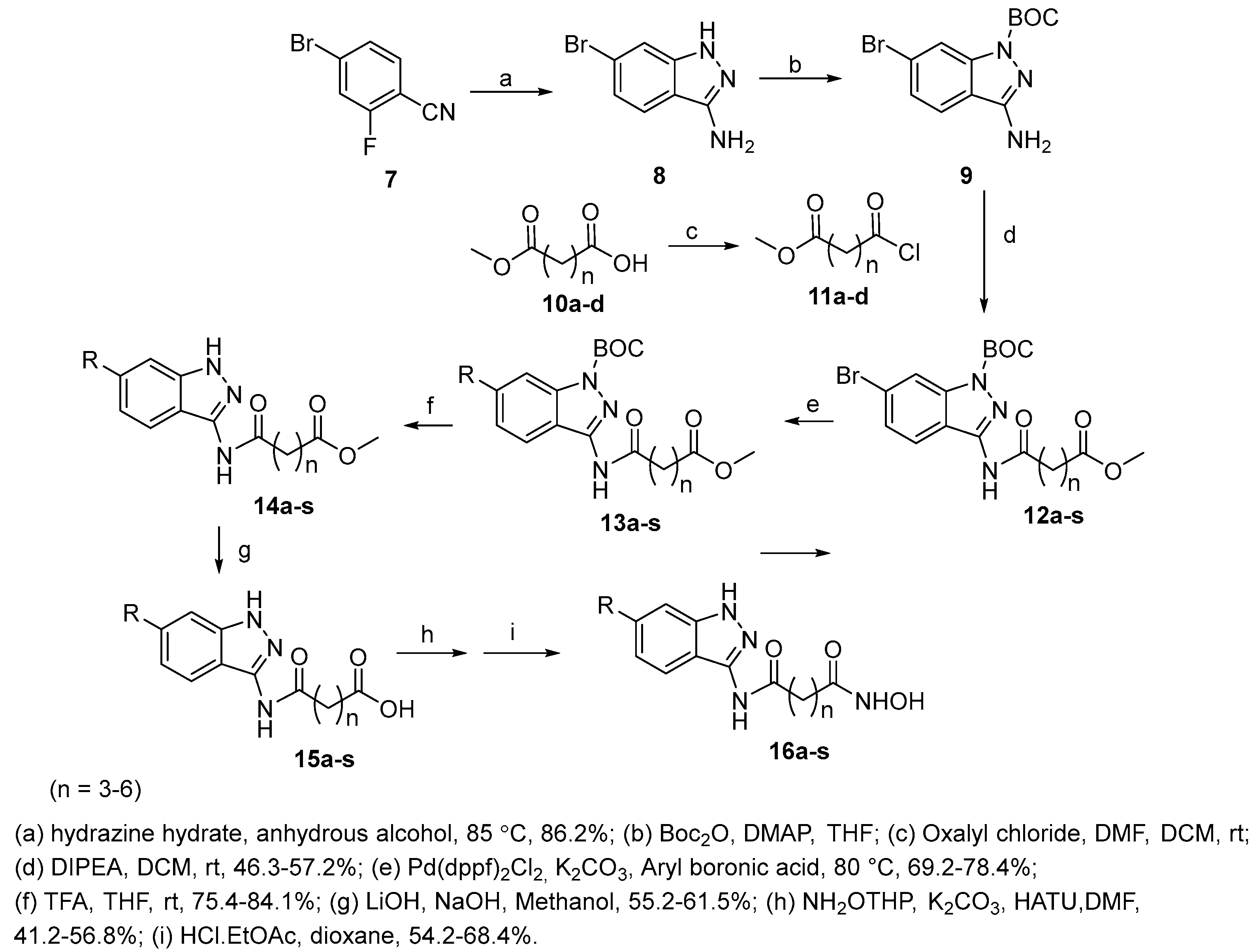
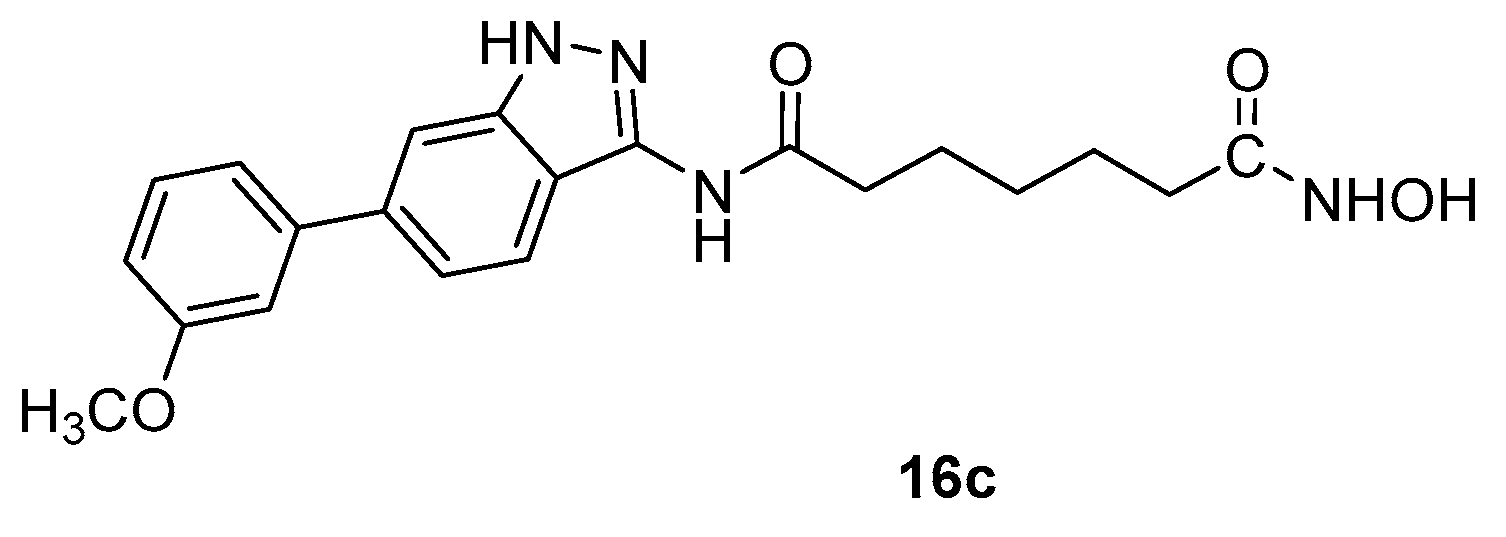
4.3. Indazole nucleus in CAP group
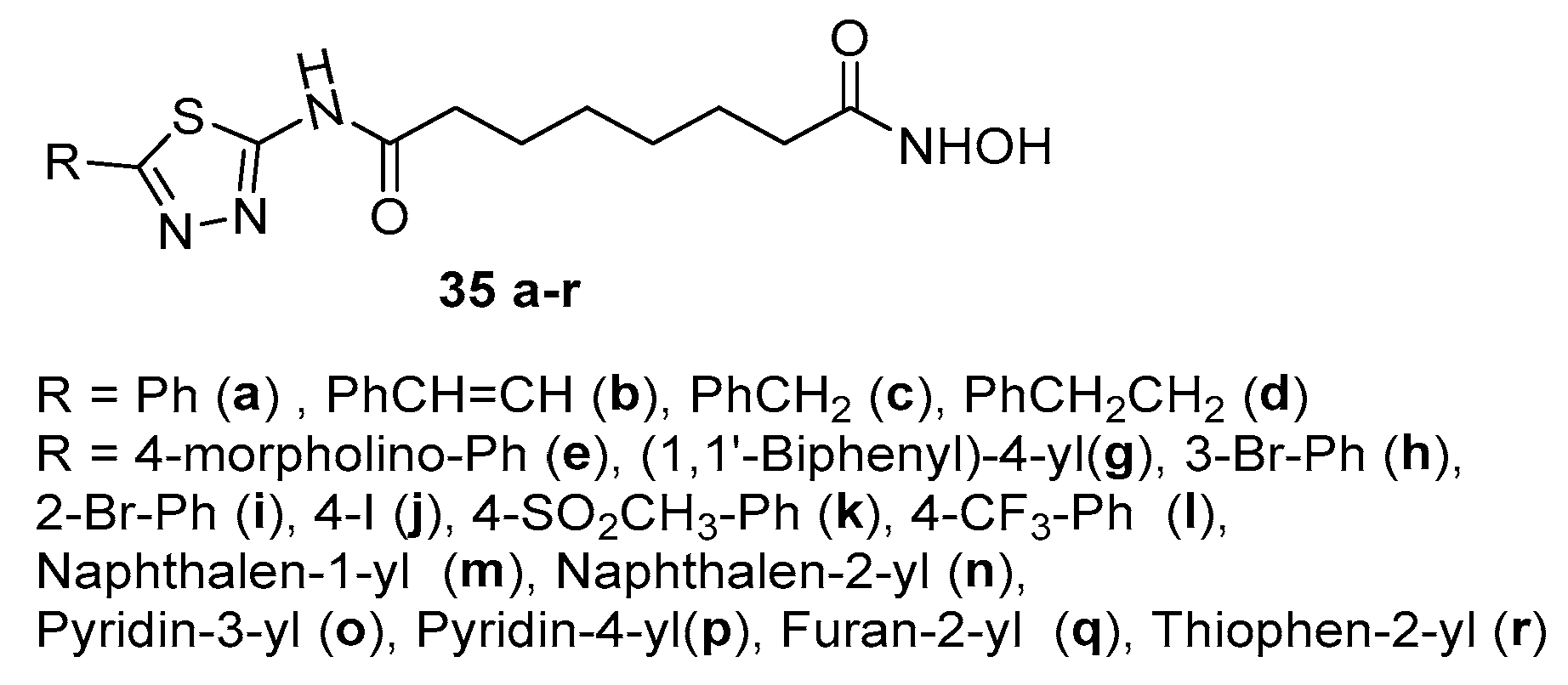
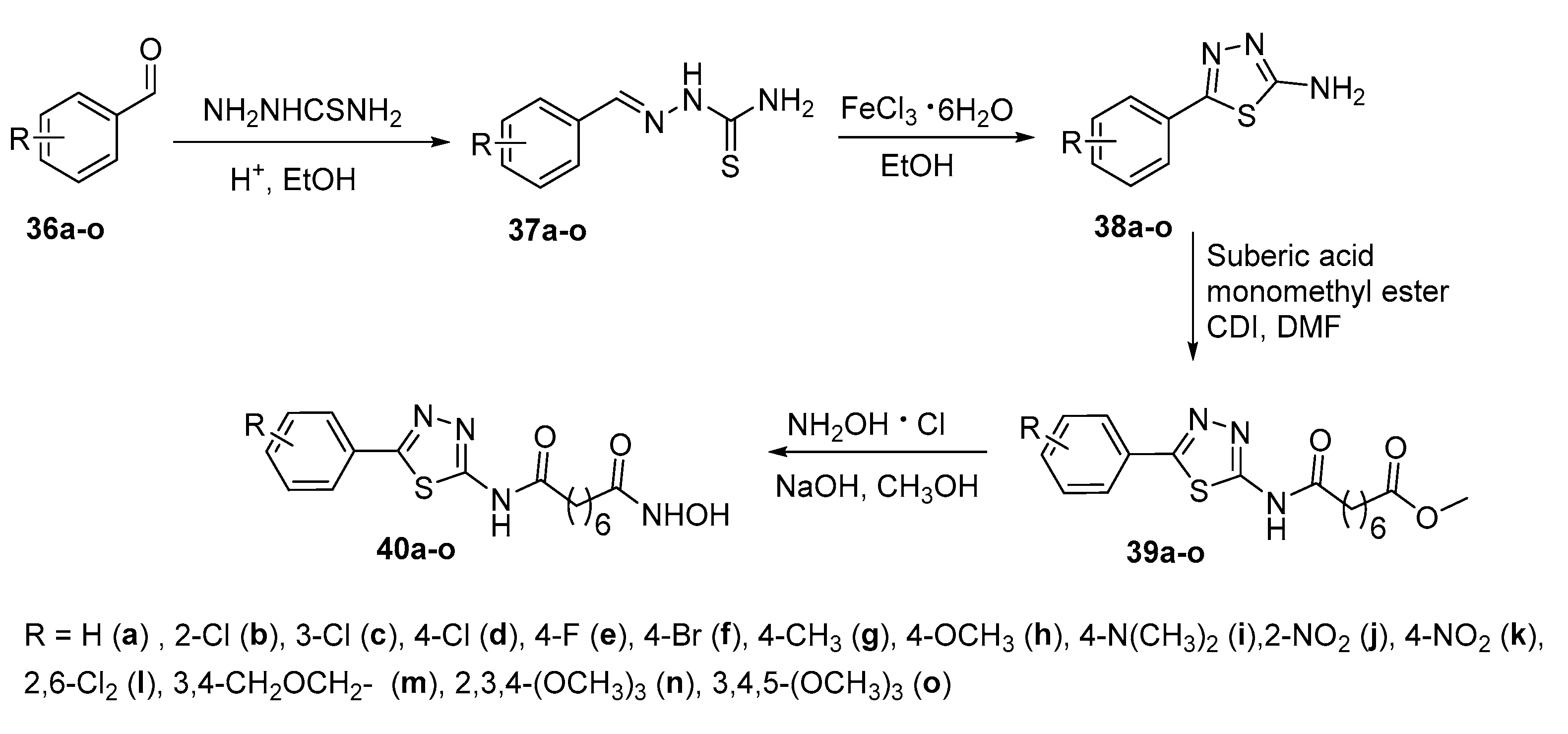
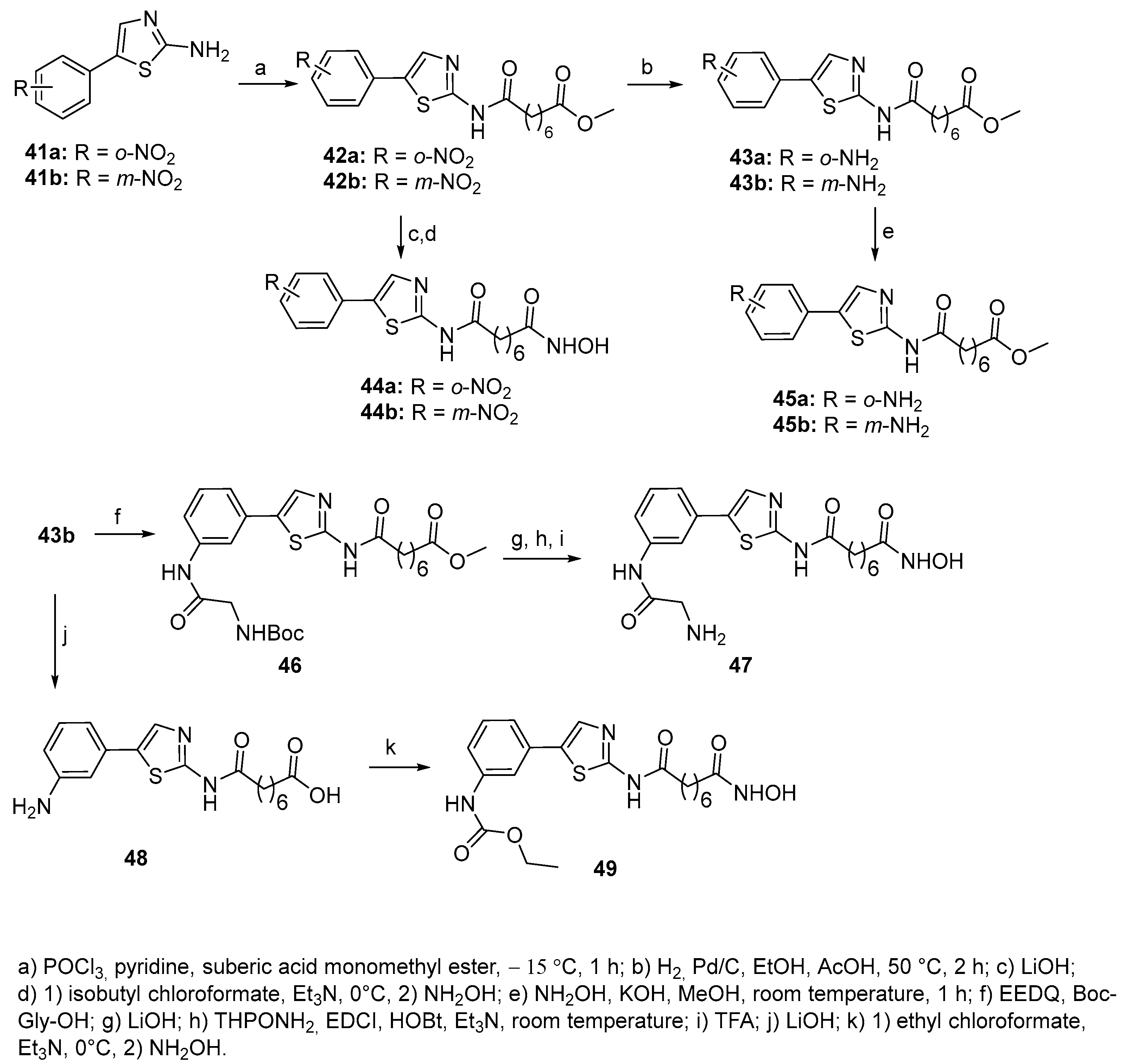
5. Six-carbon linker chain (6-C spacer)
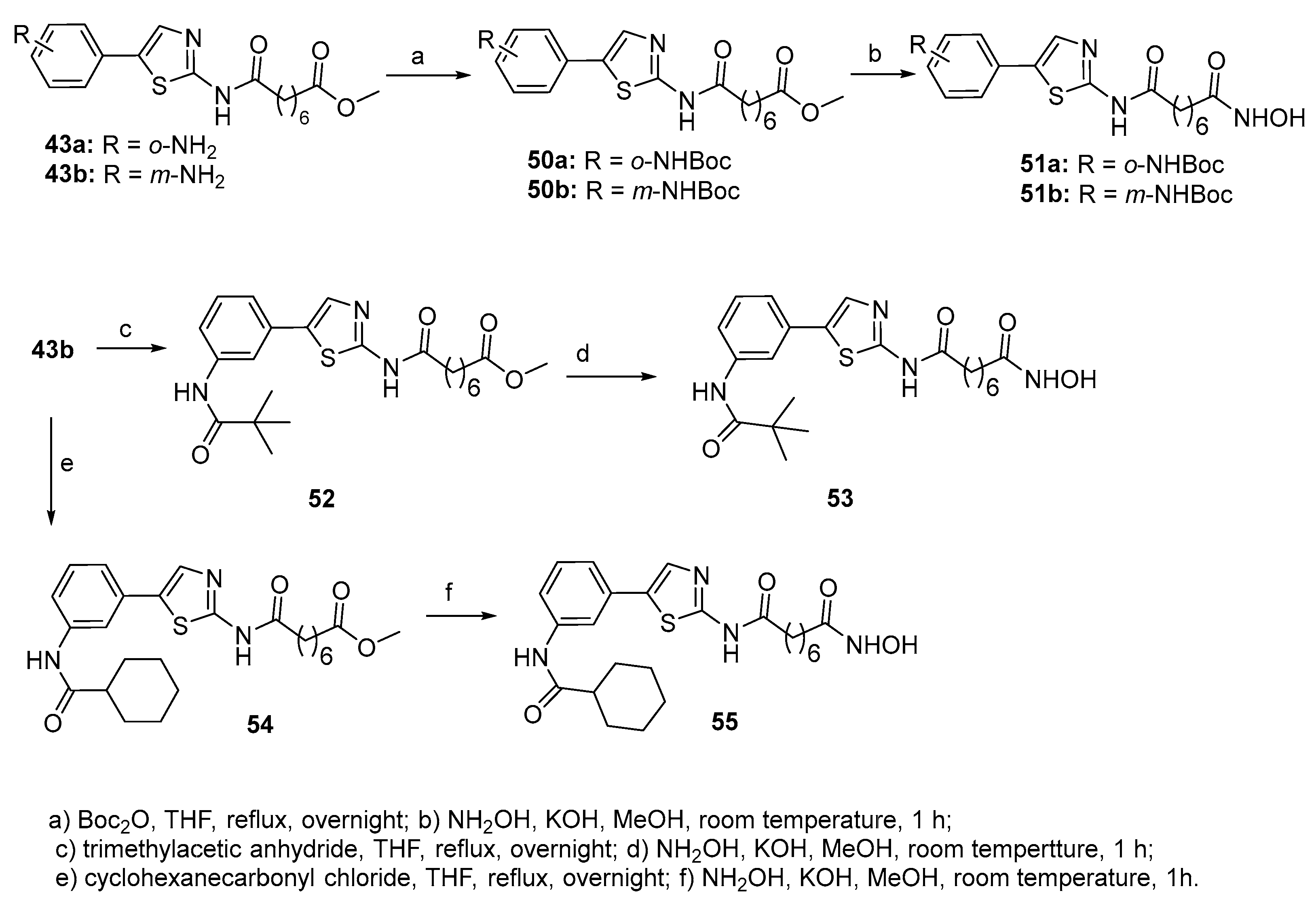
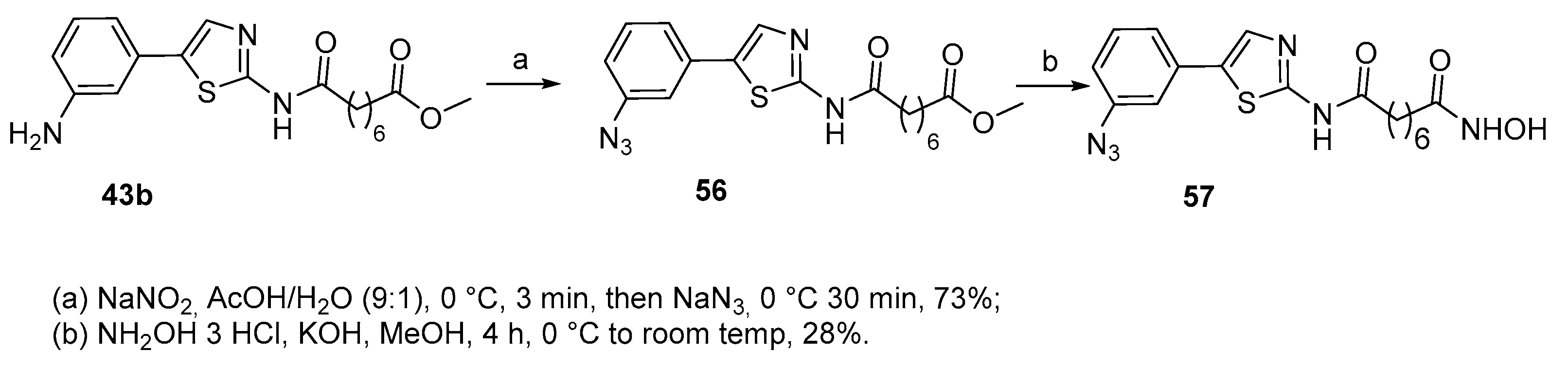
5.2. Thiazoles in CAP group
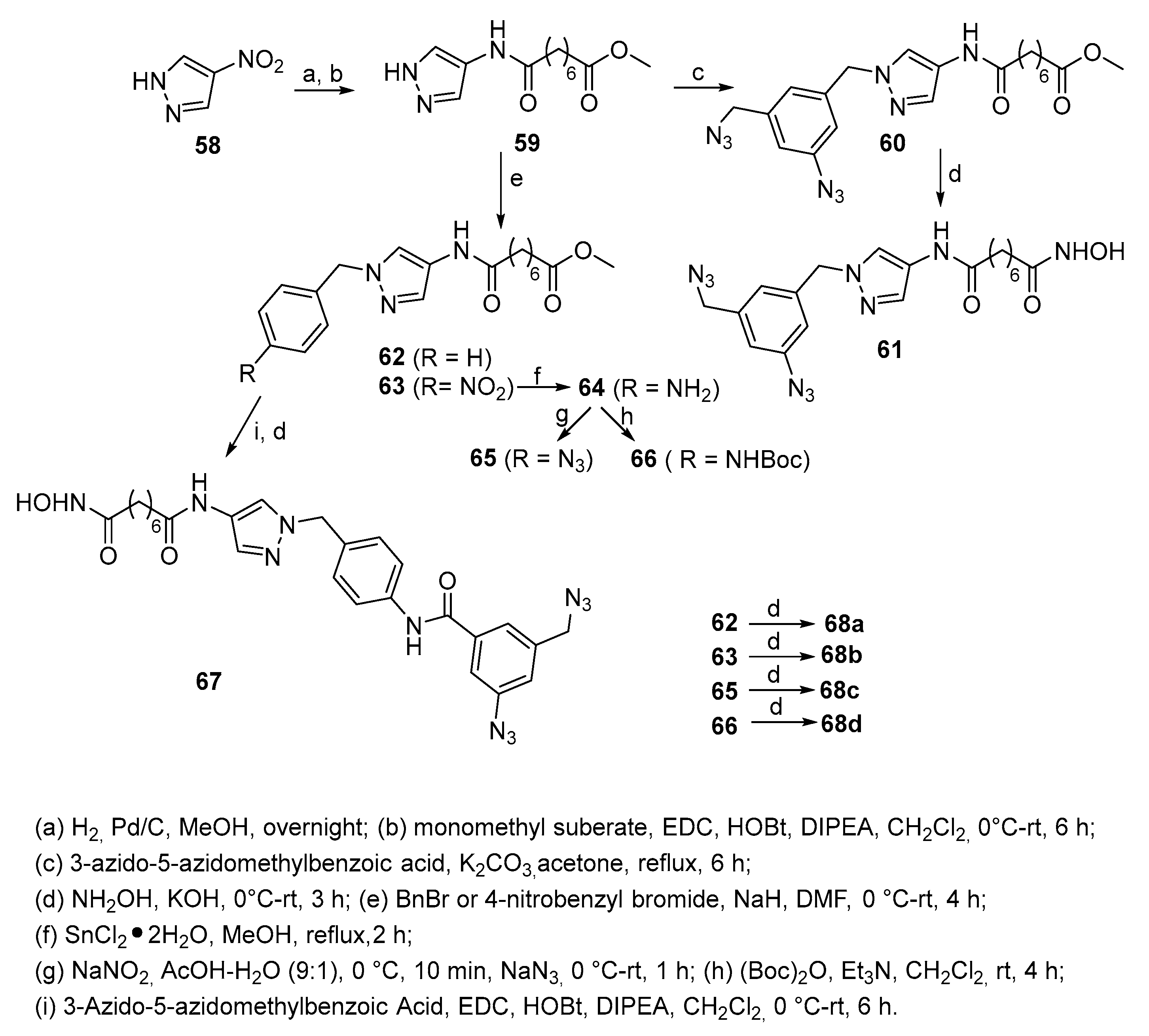
5.3. Pyrazole nucleus in CAP group
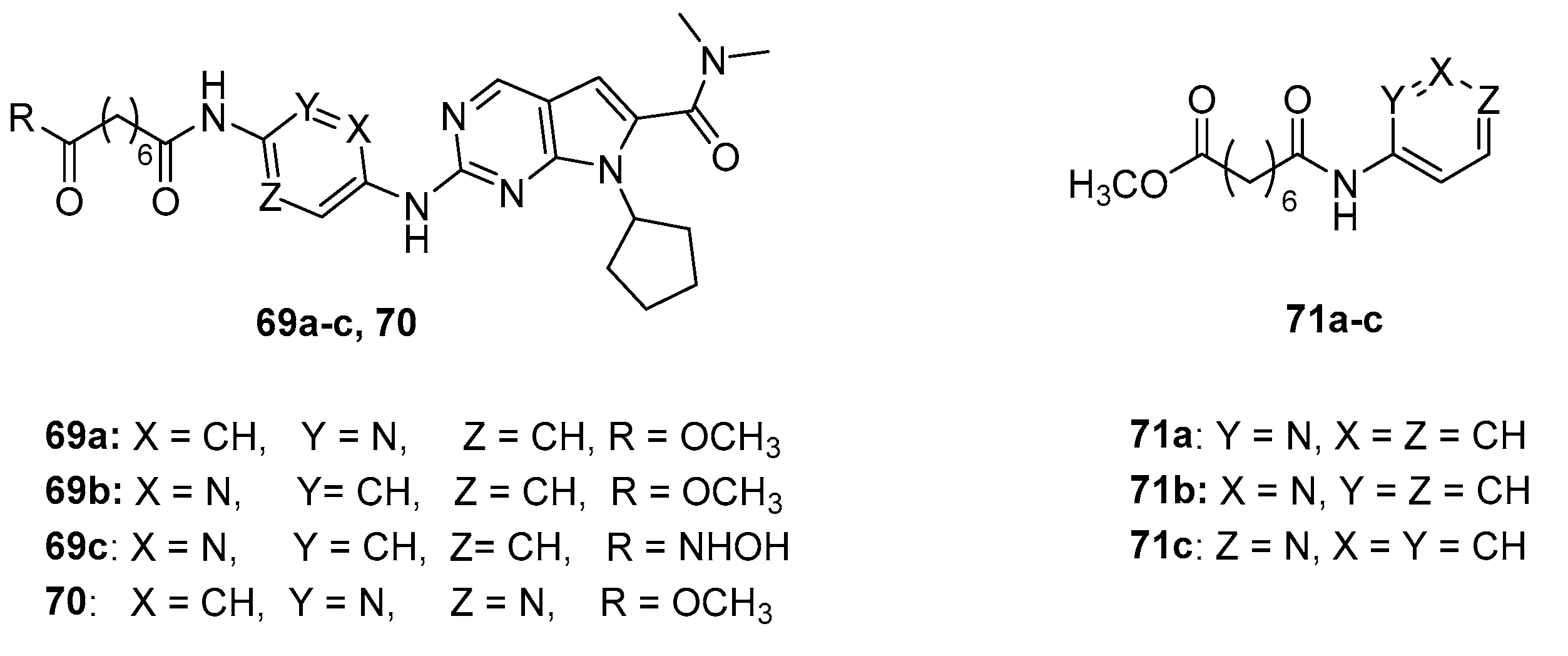
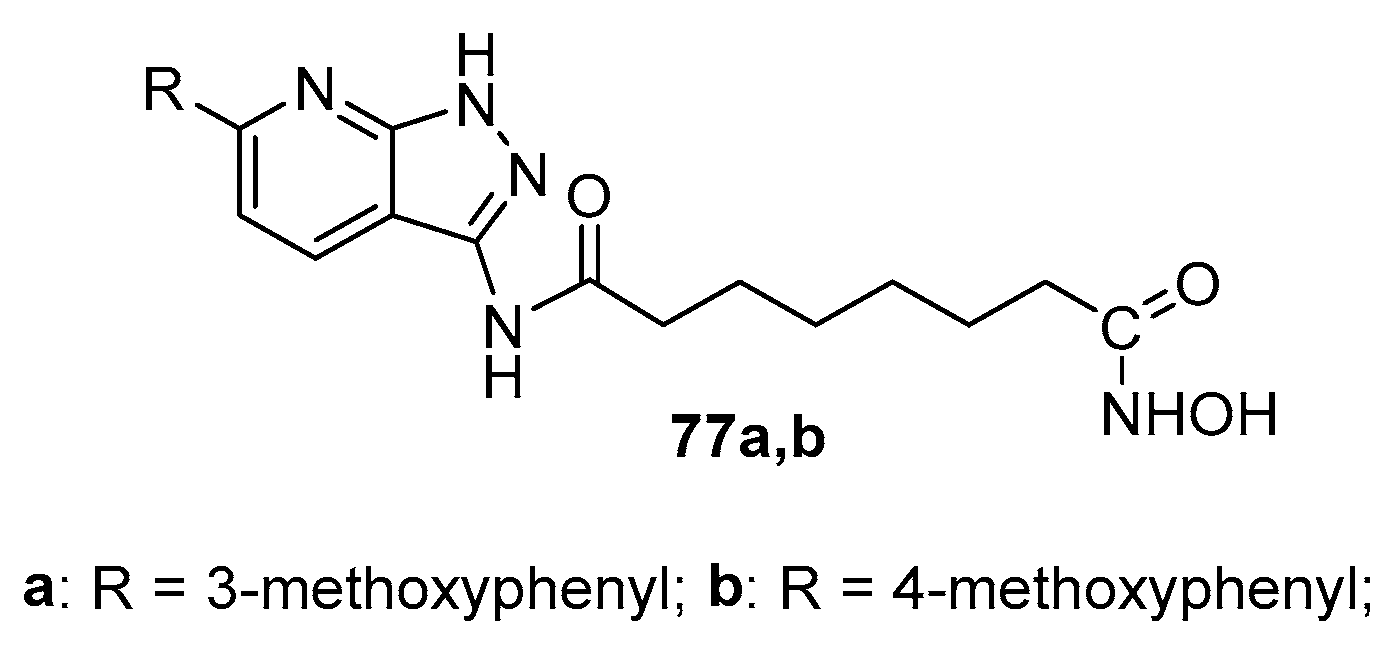
5.4. Pyridine and pyrimidine nucleus in CAP group
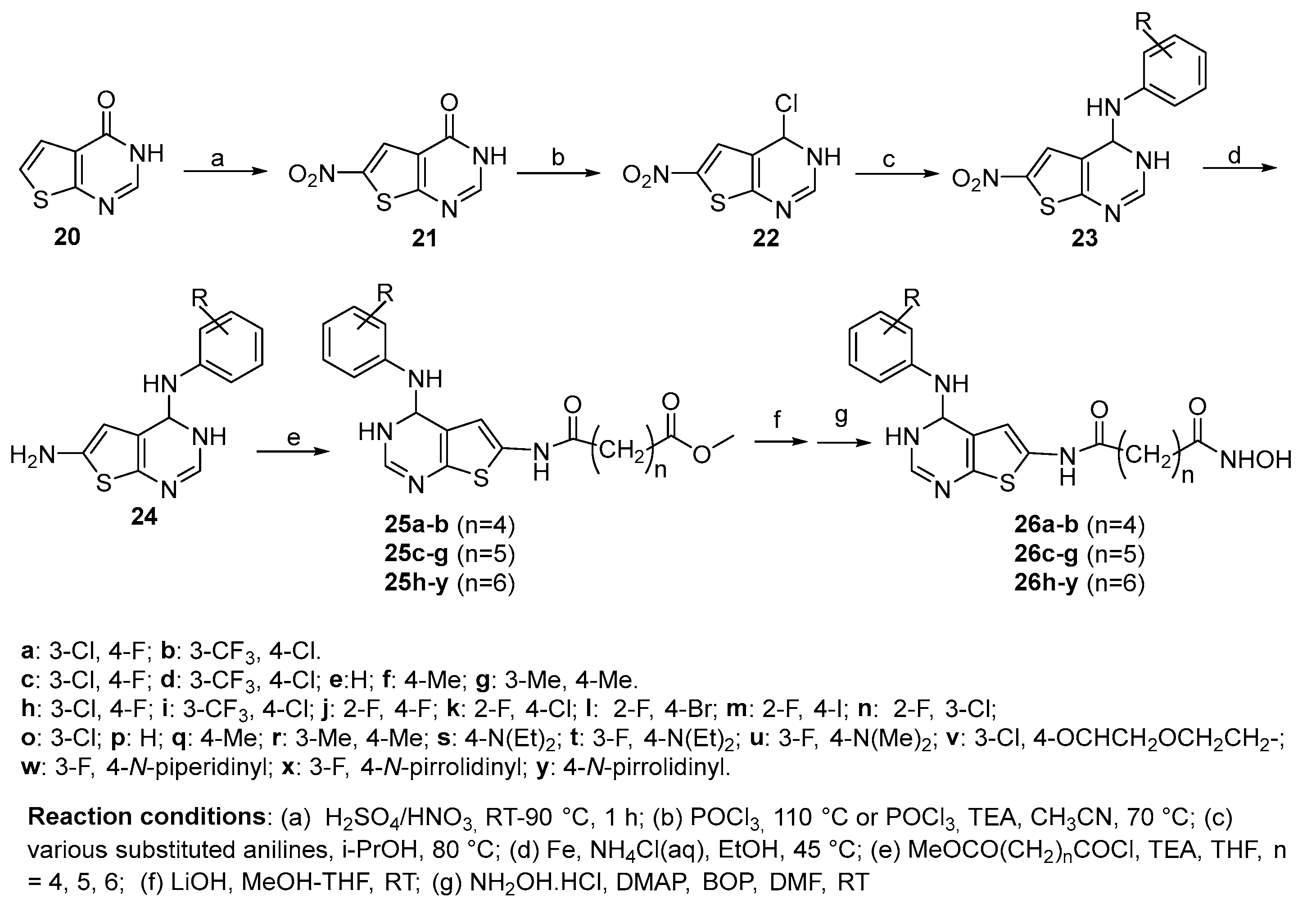
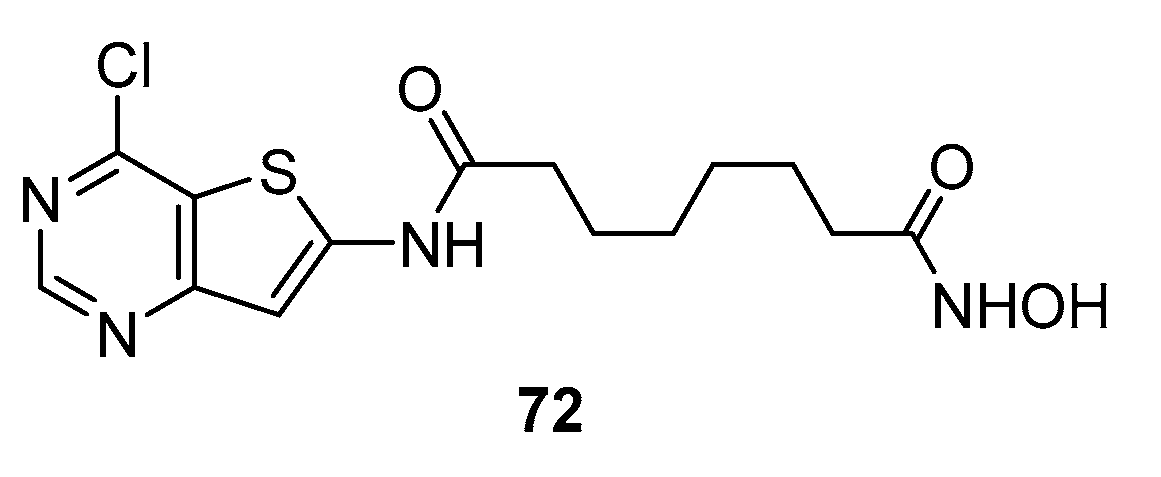
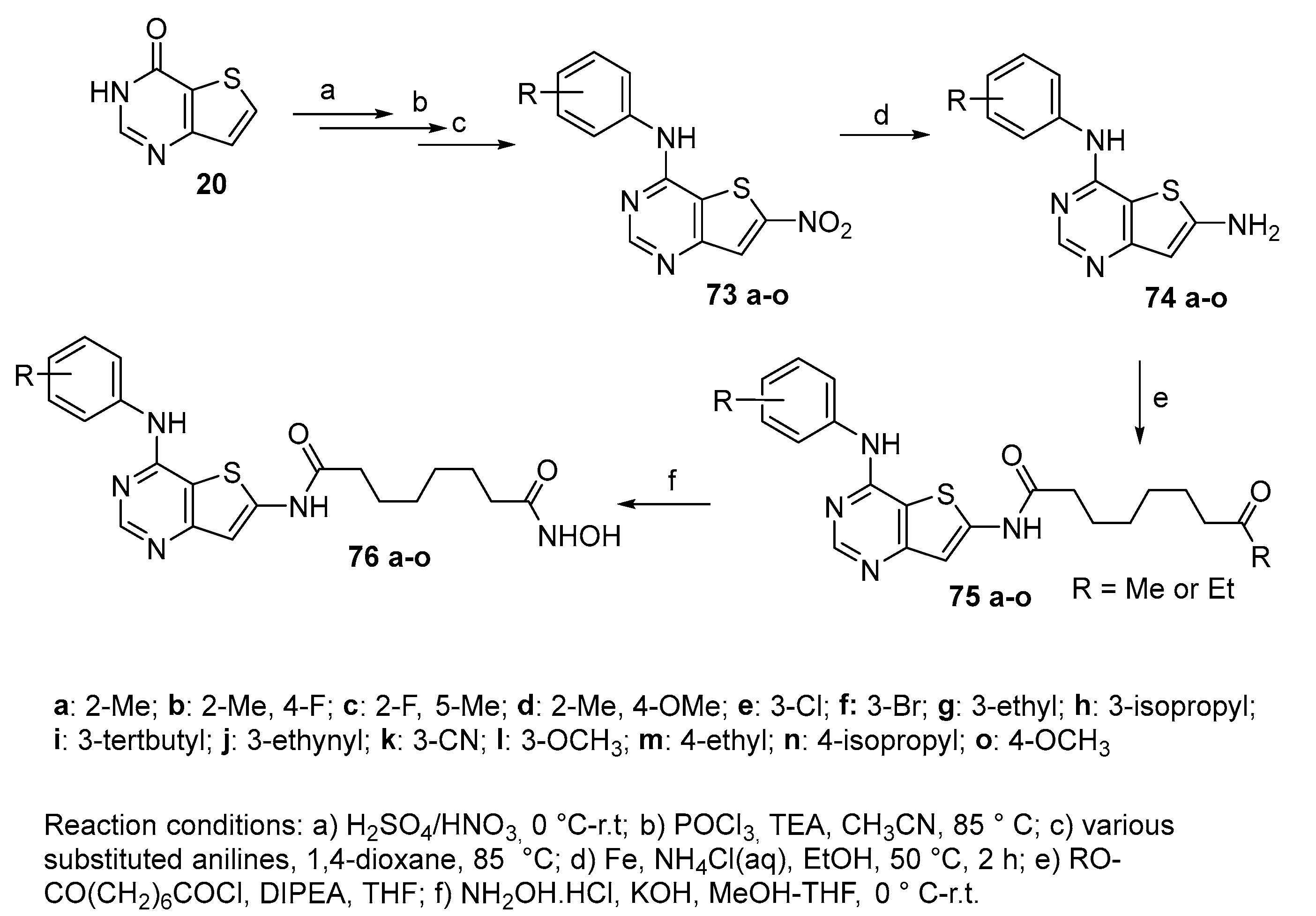
5.5. Thienopyrimidine nucleus in CAP group
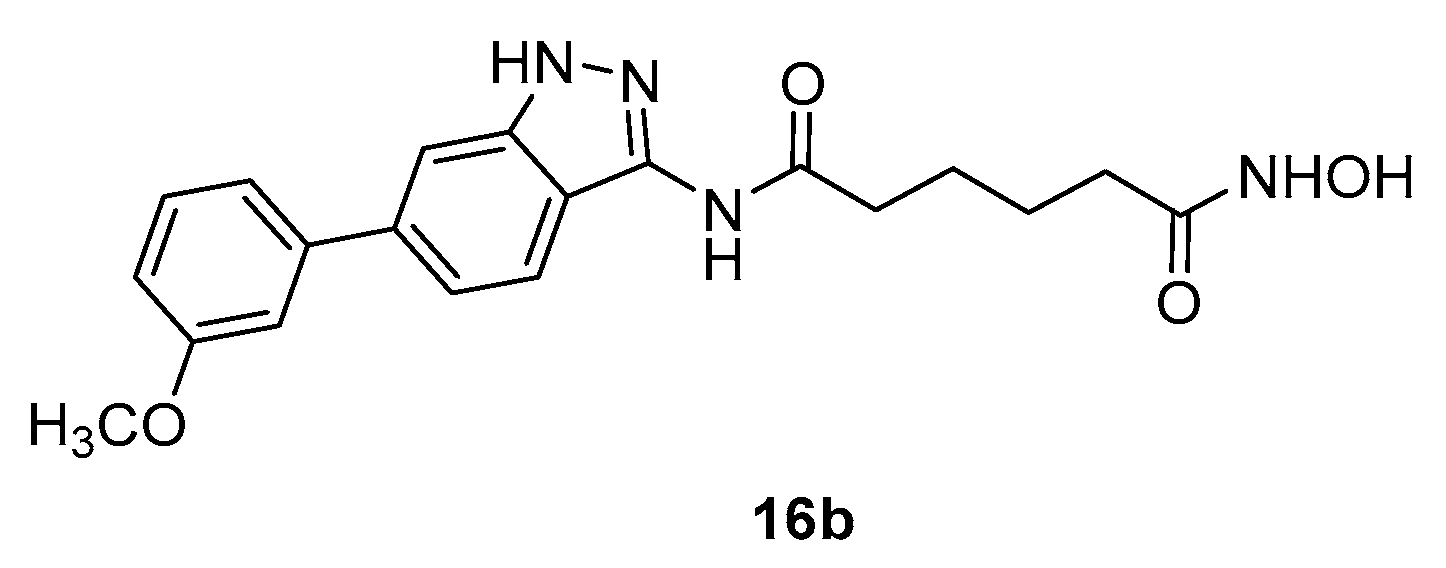
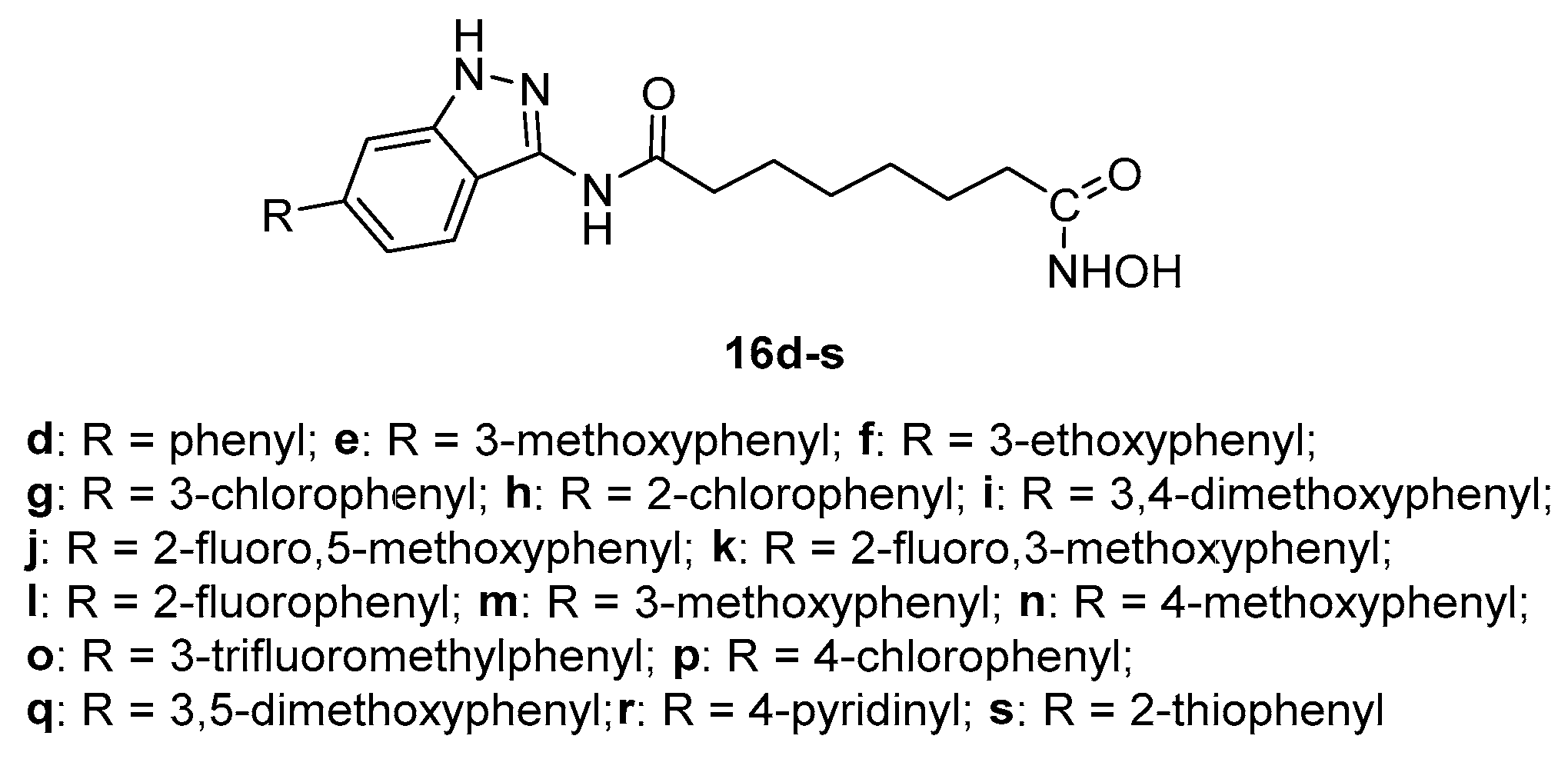
5.6. Indazole nucleus in CAP group
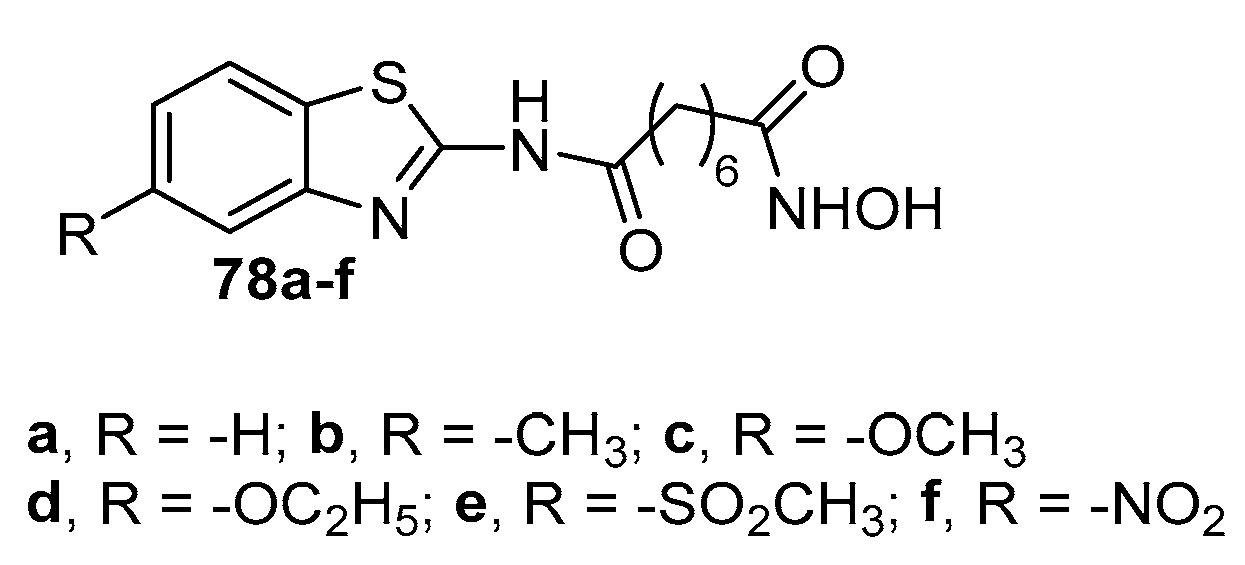
5.7. Benzothiazole moiety in CAP group
5.8. Benzoxazole moiety in CAP group
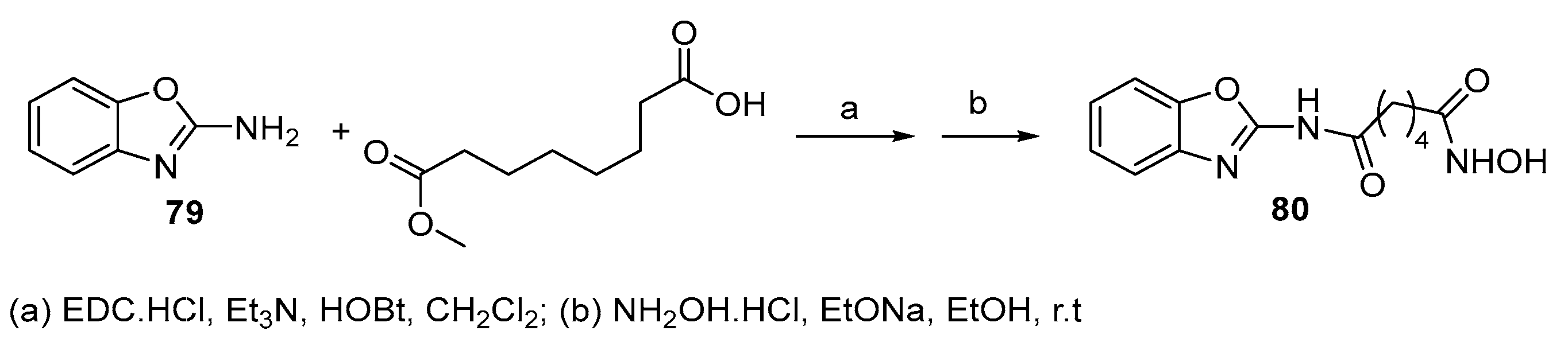
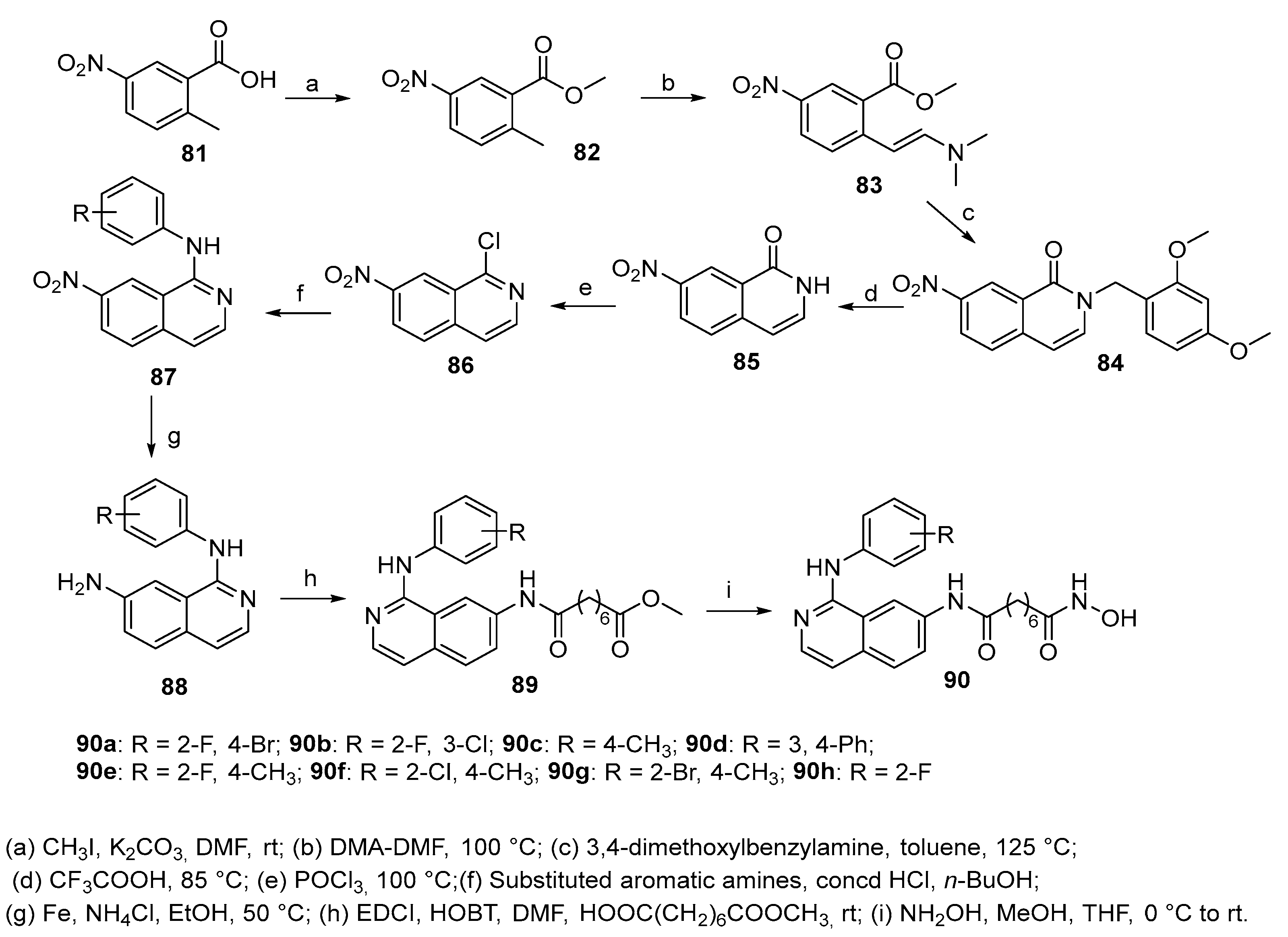
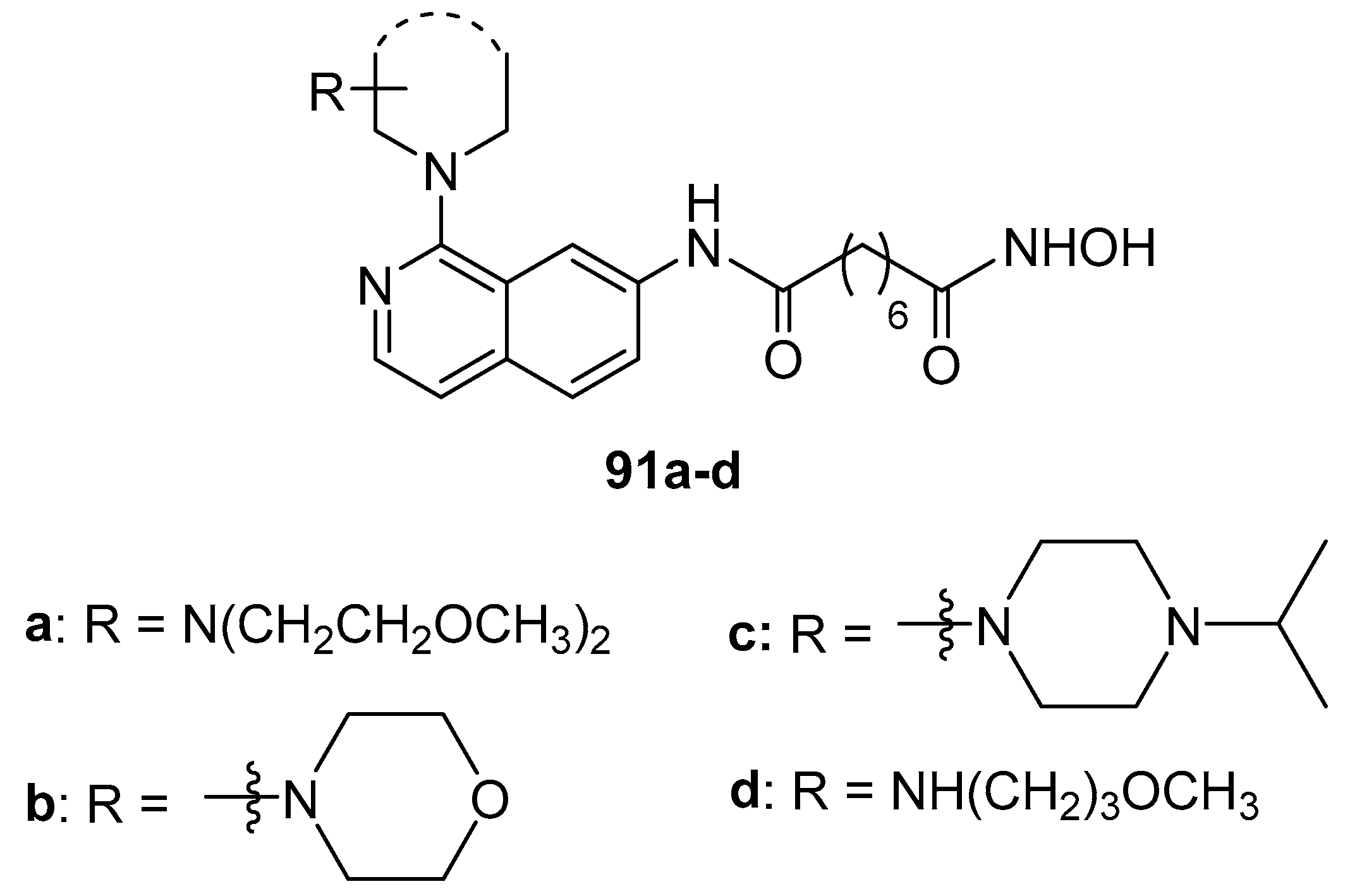
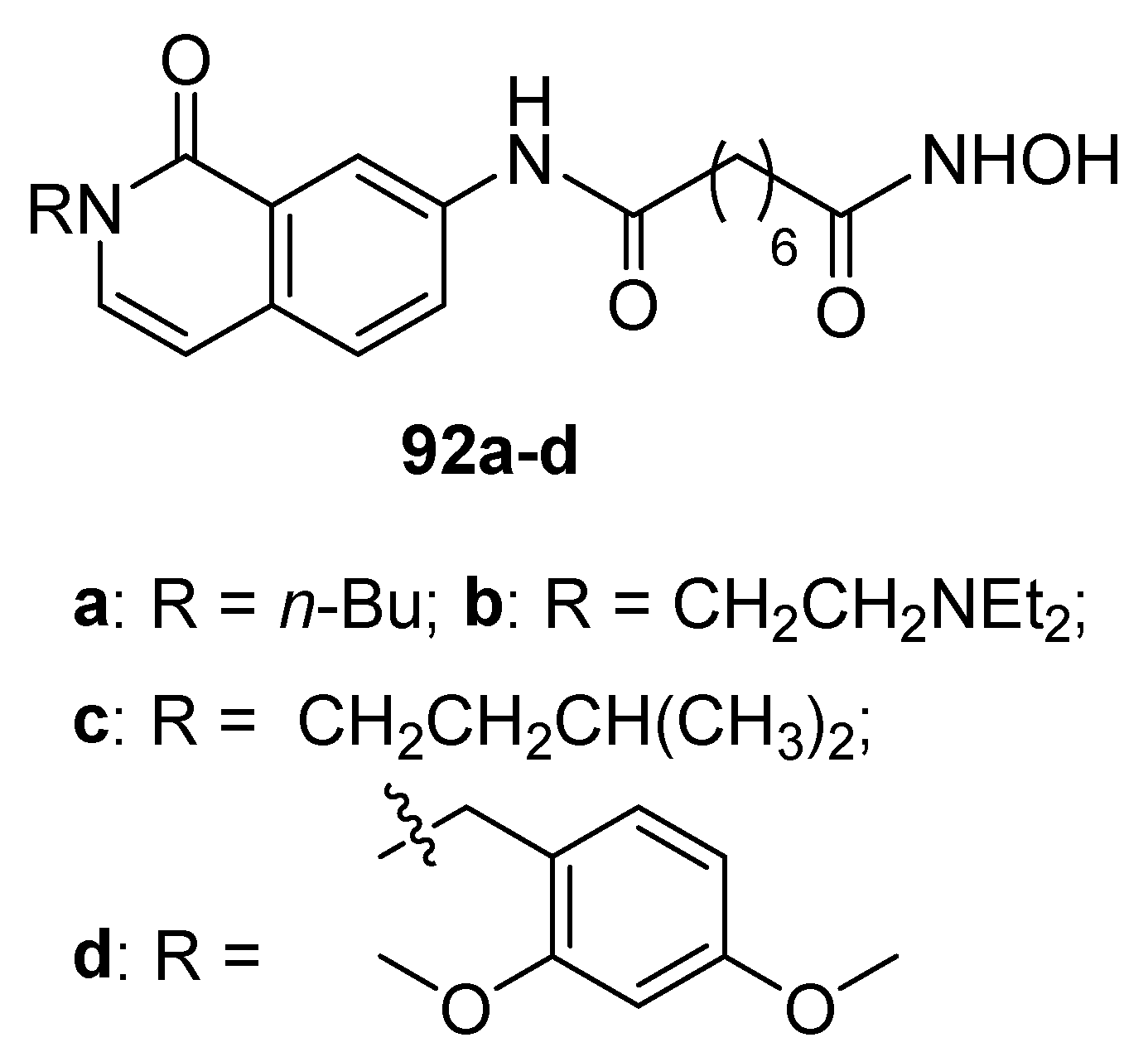
5.9. Isoquinoline moiety in CAP group
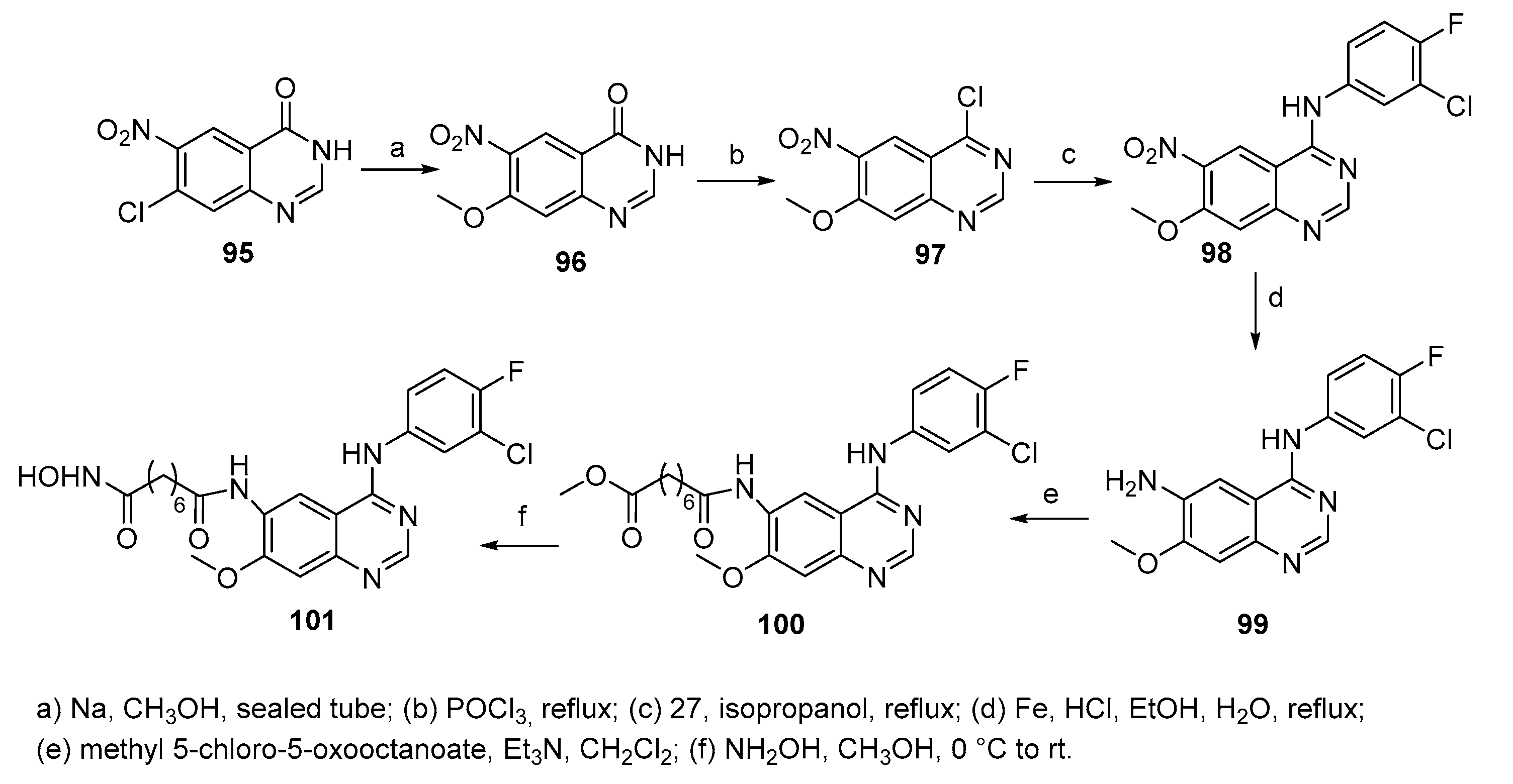
5.10. Quinazoline moiety in CAP group
6. Seven-carbon linker chain (7-C spacer)
6.1. Pyridine and pyrimidine moiety in CAP group
Author Contributions
Funding
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Witt, O.; Deubzer, H.E.; Milde, T.; Oehme, I. HDAC family: What are the cancer relevant targets? Cancer Lett. 2009, 277, 8-21. [CrossRef]
- Choudhary, C.; Kumar, C.; Gnad, F.; Nielsen, M.L.; Rehman, M.; Walther, T.C.; Olsen, J.V.; Mann, M. Lysine acetylation targets protein complexes and Co-regulates major cellular functions. Science 2009, 325, 834e840. [CrossRef]
- Vaijayanthi, T.; Pandian, G.N.; Sugiyama, H. Chemical control system of epigenetics. Chem. Rec. 2018, 18, 1833e1853. [CrossRef]
- De Ruijter, A.J.M.; van Gennip, A.H.; Caron, H.N.; Kemp, S.; van Kuilenburg, A.B.P. Histone deacetylases (HDACs): characterization of the classical HDAC family. Biochem. J. 2003, 370, 737-749. [CrossRef]
- Dokmanovic, M.; Marks, P.A. Prospects: Histone Deacetylase Inhibitors J. Cell. BioChem. 2005, 96, 293-304. [CrossRef]
- Glaser, K. B. HDAC inhibitors: Clinical update and mechanism-based potential. Biochem. Pharmacol. 2007, 74, 659-671. [CrossRef]
- Langley, B.; D’Annibale, M.A.; Suh, K.; Ayoub, I.; Tolhurst, A.; Bastan, B.; Yang, L.; Ko, B.; Fisher, M.; Cho, S.; Beal, M.F.; Ratan, R.R. Pulse Inhibition of Histone Deacetylases Induces Complete Resistance to Oxidative Death in Cortical Neurons without Toxicity and Reveals a Role for Cytoplasmic p21waf1/cip1 in Cell Cycle-Independent Neuroprotection. J. Neurosci. 2008, 28, 163-176. [CrossRef]
- Sinn, D.I.; Kim, S.J.; Chu, K.; Jung, K.H.; Lee, S.T.; Song, E.C.; Kim, J.M.; Park, D.K.; Kun Lee, S.; Kim, M.; Roh, J.K. Valproic acid-mediated neuroprotection in intracerebral hemorrhage via histone deacetylase inhibition and transcriptional activation. Neurobiol. Dis. 2007, 26, 464-472. [CrossRef]
- Petri, S.; Kiaei, M.; Kipiani, K.; Chen, J.; Calingasan, N.Y.; Crow, J.P.; Beal, M.F. Additive neuroprotective effects of a histone deacetylase inhibitor and a catalytic antioxidant in a transgenic mouse model of amyotrophic lateral sclerosis. Neurobiol. Dis. 2006, 22, 40-49. [CrossRef]
- Hahnen, E.; Hauke, J.; Trankle, C.; Eyupoglu, I.Y.; Wirth, B.; Blumcke, I. Histone deacetylase inhibitors: possible implications for neurodegenerative disorders Expert Opin. Invest. Drugs 2008, 17, 169-184. [CrossRef]
- Gregoretti, I.V.; Lee, Y.M.; Goodson, H.V. Molecular evolution of the histone deacetylase family: functional implications of phylogenetic analysis. J. Mol. Biol. 2004, 338, 17–31. [CrossRef]
- Smith, B.C.; Hallows, W.C.; Denu, J.M. Mechanisms and molecular probes of sirtuins. Chem. Biol. 2008, 15, 1002–1013. [CrossRef]
- Mann, B.S.; Johnson, J.R.; Cohen, M.H.; Justice, R.; Pazdur, R. FDA approval summary: vorinostat for treatment of advanced primary cutaneous T-cell lymphoma. Oncologist 2007, 12, 1247–1252. [CrossRef]
- Finnin, M.S.; Donigian, J.R.; Cohen, A.; Richon, V.M.; Rifkind, R.A.; Marks, P.A.; Breslow, R.; Pavletich, N.P. Nature 1999, 401, 188-193. [CrossRef]
- Guan, P.; Sun, F.; Hou, X.; Wang, F.; Yi, F.; Xu, W.; Fang, H. Design, synthesis and preliminary bioactivity studies of 1,3,4-thiadiazole hydroxamic acid derivatives as novel histone deacetylase inhibitors. Bioorg. Med. Chem., 2012, 20, 3865-3872. [CrossRef]
- Liu, J.; Zhou, J.; He, F.; Gao, L.; Wen, Y.; Gao, L.; Wang, P.; Kang, D.; Hu, L. Design, synthesis and biological evaluation of novel indazole-based derivatives as potent HDAC inhibitors via fragment-based virtual screening. Eur. J. Med. Chem. 2020, 192, 112189. [CrossRef]
- Oanh, D.T.K.; Hai, H.V.; Park, S.H.; Kim, H.-J.; Han, B.-W.; Kim, H.-S.; Hong, J.-T.; Han, S.-B.; Hue, V.T.M.; Nam, N.-H.. Benzothiazole-containing hydroxamic acids as histone deacetylase inhibitors and antitumor agents. Bioorg.Med. Chem. Lett. 2011, 21, 7509–7512. [CrossRef]
- Yang, W.; Li, L.; Ji, X.; Wu, X.; Su, M.; Sheng, L.; Zang, Y.; Li, J.; Liu, H. Design, synthesis and biological evaluation of 4-anilinothieno[2,3-d]pyrimidine-based hydroxamic acid derivatives as novel histone deacetylase inhibitors. Bioorg. Med. Chem. 2014, 22, 6146–6155. [CrossRef]
- Pardo-Jiménez, V.; Navarrete-Encina, P.; Díaz-Araya, G. Synthesis and Biological Evaluation of Novel Thiazolyl-Coumarin Derivatives as Potent Histone Deacetylase Inhibitors with Antifibrotic Activity. Molecules 2019, 24, 739; [CrossRef]
- Guan, P.; Wang, L.; Hou, X.; Wan, Y.; Xu, W.; Tang, W.; Fang, H. Improved antiproliferative activity of 1,3,4-thiadiazole-containing histone deacetylase (HDAC) inhibitors by introduction of the heteroaromatic surface recognition motif. Bioorg. Med. Chem. 2014, 22, 5766–5775. [CrossRef]
- Nam, N.-H.; Huong, T.L.; Dung, D.T.M.; Dung, P.T.P.; Oanh, D.T.K.; Park, S.H.; Kim, K.; Han, B.W.; Yun, J.; Kang, J.S.; Kim, Y.; Han, S.-B. Synthesis, bioevaluation and docking study of 5-substitutedphenyl-1,3,4-thiadiazole-based hydroxamic acids as histone deacetylase inhibitors and antitumor agents. J. Enzyme Inhib. Med. Chem. 2014; 29, 611-618. [CrossRef]
- Kozikowski, A.P.; Chen, Y.; Gaysin, A.M.; Savoy, D.N.; Billadeau, D.D.; Kim, K.H. Chemistry, Biology, and QSAR Studies of Substituted Biaryl Hydroxamates and Mercaptoacetamides as HDAC Inhibitors—Nanomolar-Potency Inhibitors of Pancreatic Cancer Cell Growth. ChemMedChem 2008, 3, 487–501. [CrossRef]
- Kozikowski, A.P.; Chen, Y.; Gaysin, A.; Chen, B.; D'Annibale, M.A.; Suto, C.M.; Brett C. Langley, B.C. Functional Differences in Epigenetic Modulators Superiority of Mercaptoacetamide-Based Histone Deacetylase Inhibitors Relative to Hydroxamates in Cortical Neuron Neuroprotection Studies J. Med. Chem. 2007, 50, 3054-3061. [CrossRef]
- He, B.; Velaparthi, S.; Pieffet, G.; Pennington, C.; Mahesh, A.; Holzle, D.L.; Brunsteiner, M.; van Breemen, R.; Blond, S.Y.; Petukhov, P.A. Binding Ensemble Profiling with Photoaffinity Labeling (BEProFL) Approach: Mapping the Binding Poses of HDAC8 Inhibitors. J. Med. Chem. 2009, 52, 7003–7013. [CrossRef]
- Glaser, K.B.; Li, J.; Pease, L.J.; Staver, M.J.; Marcotte, P.A.; Guo, J.; Frey, R.R.; Garland, R.B.; Heyman, H.R.; Wada, C.K.; Vasudevan, A.; Michaelides, M.R.; Davidsen, S.K.; Curtin, M.L. Differential protein acetylation induced by novel histone deacetylase inhibitors. Biochem Biophys Res Commun. 2004, 325, 683–690. [CrossRef]
- Neelarapu, R.; Holzle, D.L.; Velaparthi, S.; Bai, H.; Brunsteiner, M.; Blond, S.Y.; Petukhov, P.A. Design, Synthesis, Docking, and Biological Evaluation of Novel Diazide-Containing Isoxazole-and Pyrazole-Based Histone Deacetylase Probes. J. Med. Chem. 2011, 54, 4350–4364. [CrossRef]
- Vaidya, A.S.; Neelarapu, R.; Madriaga, A.; Bai, H.; Mendonca, E.; Abdelkarim, H.; van Breemen, R.B.; Blond, S.Y.; Petukhov, P.A. Novel histone deacetylase 8 ligands without a zinc chelating group: Exploring an 'upside-down' binding pose. Bioorg. Med. Chem. Lett. 2012, 22, 6621–6627. [CrossRef]
- Albrow, V.E.; Grimley, R.L.; Clulow, J.; Rose, C.R.; Sun, J.; Warmus, J.S.; Tate, E.W.; Jonesd, L.H.; Storer, R.I.. Design and development of histone deacetylase (HDAC) chemical probes for cell-based profiling. Mol. BioSyst., 2016, 12, 1781—1789. [CrossRef]
- Li, Y.; Luo, X.; Guo, Q.; Nie, Y.; Wang, T.; Zhang, C.; Huang, Z.; Wang, X.; Liu, Y.; Chen, Y.; Zheng, J.; Yang, S.; Yan Fan, Y.; Xiang, R. Discovery of N1-(4-((7-Cyclopentyl-6-(dimethylcarbamoyl)-7 H-pyrrolo[2,3- d]pyrimidin-2-yl)amino)phenyl)- N8-hydroxyoctanediamide as a Novel Inhibitor Targeting Cyclin-dependent Kinase 4/9 (CDK4/9) and Histone Deacetlyase1 (HDAC1) against Malignant Cancer. J. Med. Chem. 2018, 61, 3166-3192. [CrossRef]
- Remiszewski, S.W:, Sambucetti, L.C.; Atadja, P.; Bair, K.W.; Cornell, W.D.; Green, M.A.; Howell, K.L.; Jung, M.; Kwon, P.; Trogani, N.; Walker, H. Inhibitors of Human Histone Deacetylase: Synthesis and Enzyme and Cellular Activity of Straight Chain Hydroxamates. J. Med. Chem. 2002, 45, 753–757. [CrossRef]
- Wang, J.; Su, M.; Li, T.; Gao, A.; Yang, W.; Sheng, L.; Zang, Y.; Li, J.; Liu, H. Design, synthesis and biological evaluation of thienopyrimidine hydroxamic acid based derivatives as structurally novel histone deacetylase (HDAC) inhibitors. Eur. J. Med. Chem. 2017, 128, 293-299. [CrossRef]
- Mantzourani, C.; Gkikas, D.; Kokotos, A.; Nummela, P.; Theodoropoulou, M.A.; Wu, K.-C.; Fairlie, D.P.; Politis, P.K.; Ristimäki, A.; Kokotos, G. Synthesis of benzoxazole-based vorinostat analogs and their antiproliferative activity. Bioorg. Chem. 2021, 114, 105132. [CrossRef]
- Yang, W.; Li, L.; Wang, Y.; Wu, X.; Li, T.; Yang, N.; Su, M.; Sheng, L.; Zheng, M.; Zang, Y.; Li, J.; Liu, H. Design, synthesis and biological evaluation of isoquinoline-based derivatives as novel histone deacetylase inhibitors. Bioorg. Med. Chem. 2015, 23, 5881-5890. [CrossRef]
- Salmi-Smail, C.; Fabre, A.; Dequiedt, F.; Restouin, A.; Castellano, R.; Garbit, S.; Roche, P.; Morelli, X.; Brunel, J.M.; Collette, Y. Modified Cap Group Suberoylanilide Hydroxamic Acid Histone Deacetylase Inhibitor Derivatives Reveal Improved Selective Antileukemic Activity. J. Med. Chem. 2010, 53, 3038–3047. [CrossRef]
- Cai, X.; Zhai, H.-X.; Wang, J.; Forrester, J.; Qu, H.; Yin, L.; Lai, C.-J.; Bao, R.; Qian, C. Discovery of 7-(4-(3-Ethynylphenylamino)-7-methoxyquinazolin-6-yloxy)-N-hydroxyheptanamide (CUDC-101) as a Potent Multi-Acting HDAC, EGFR, and HER2 Inhibitor for the Treatment of Cancer. J. Med. Chem. 2010, 53, 2000–2009. [CrossRef]
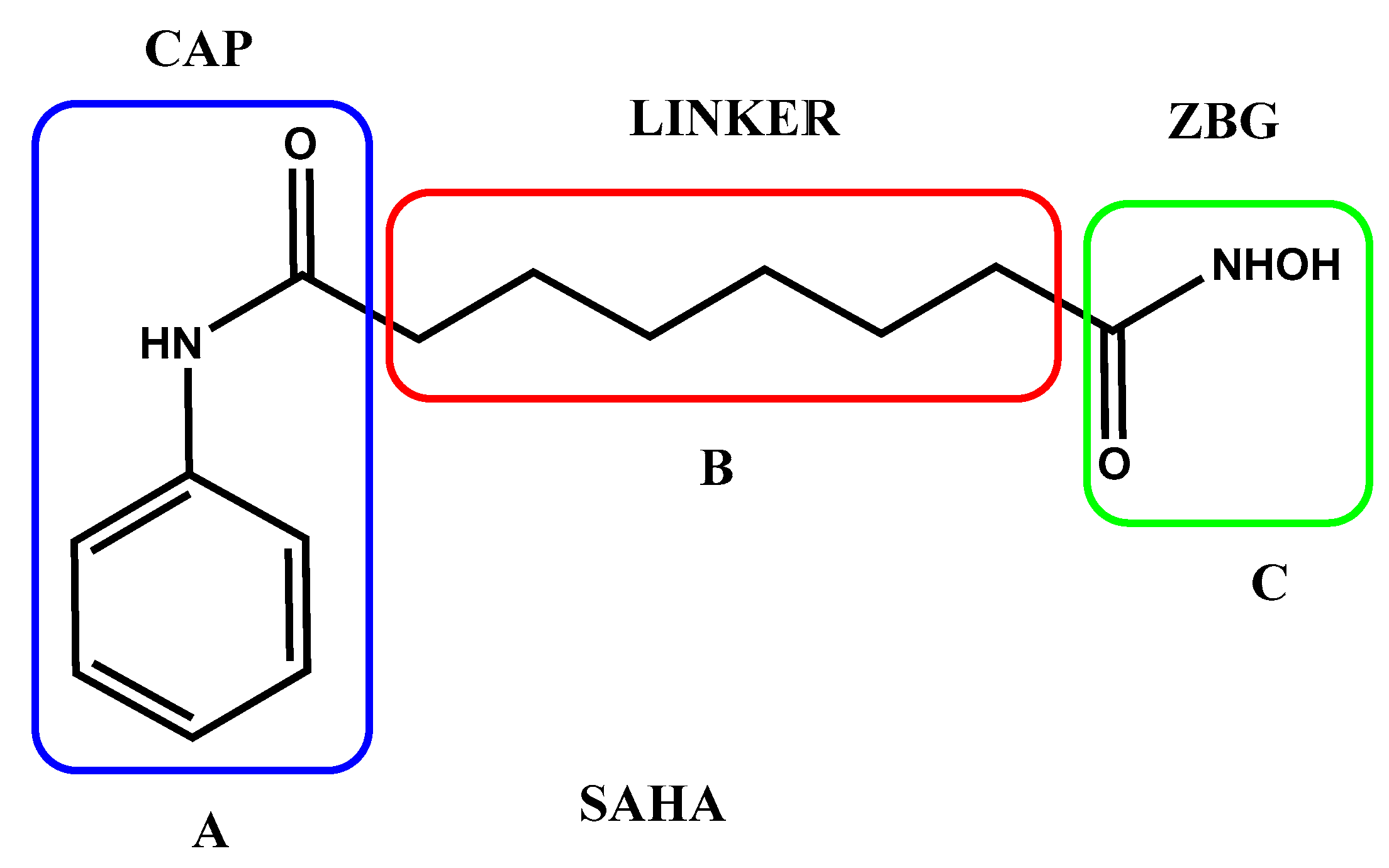
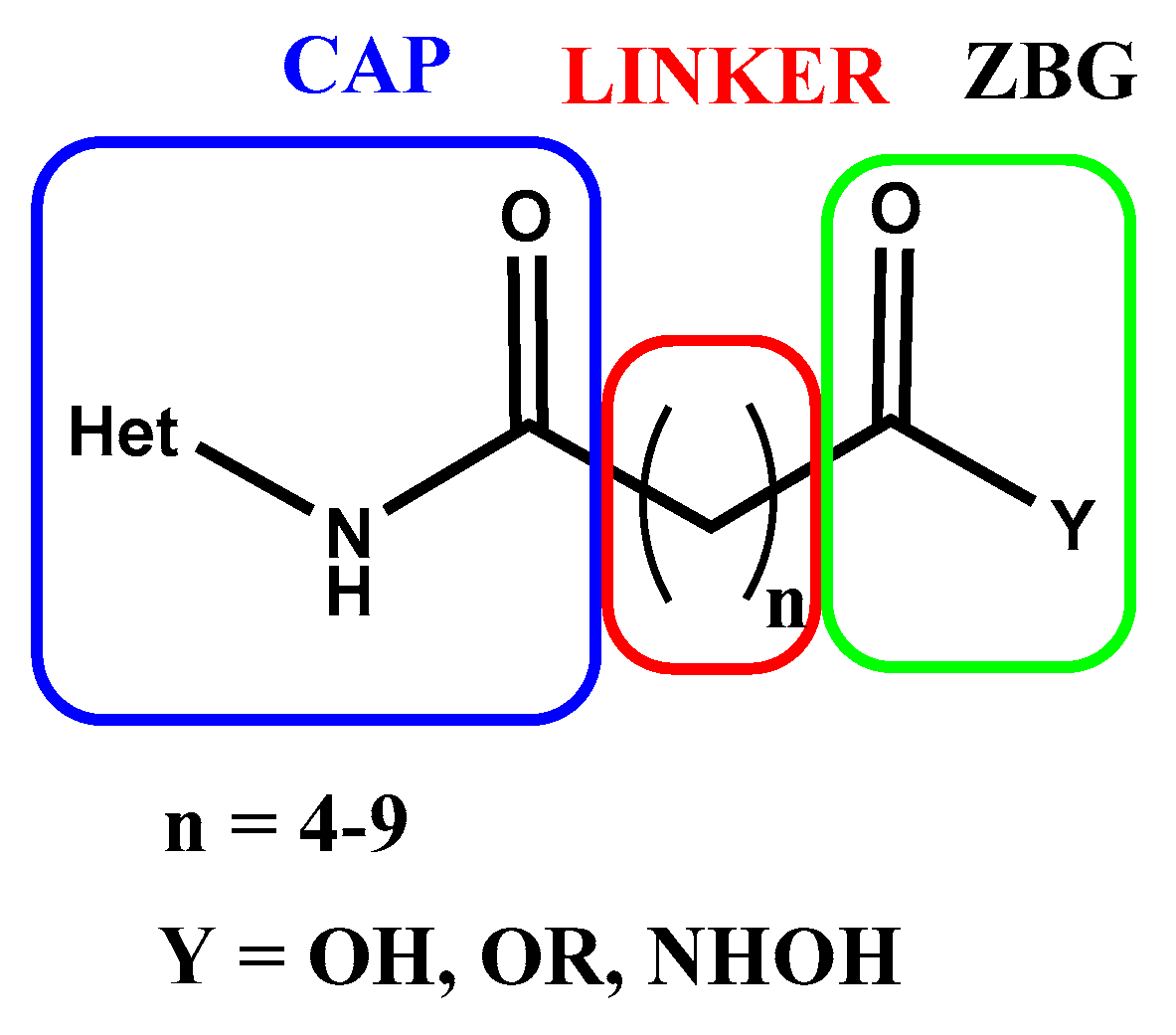
- Boga, C.; Micheletti, G. Design and Synthesis of Organic Molecules as Antineoplastic Agents. Molecules 2020, 25, 2808. [CrossRef]
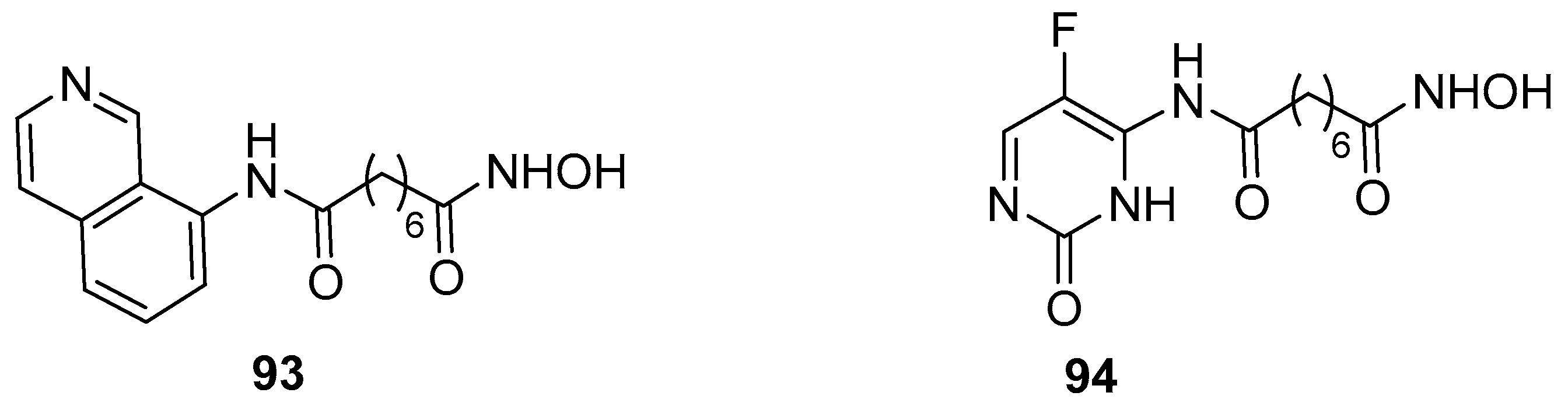
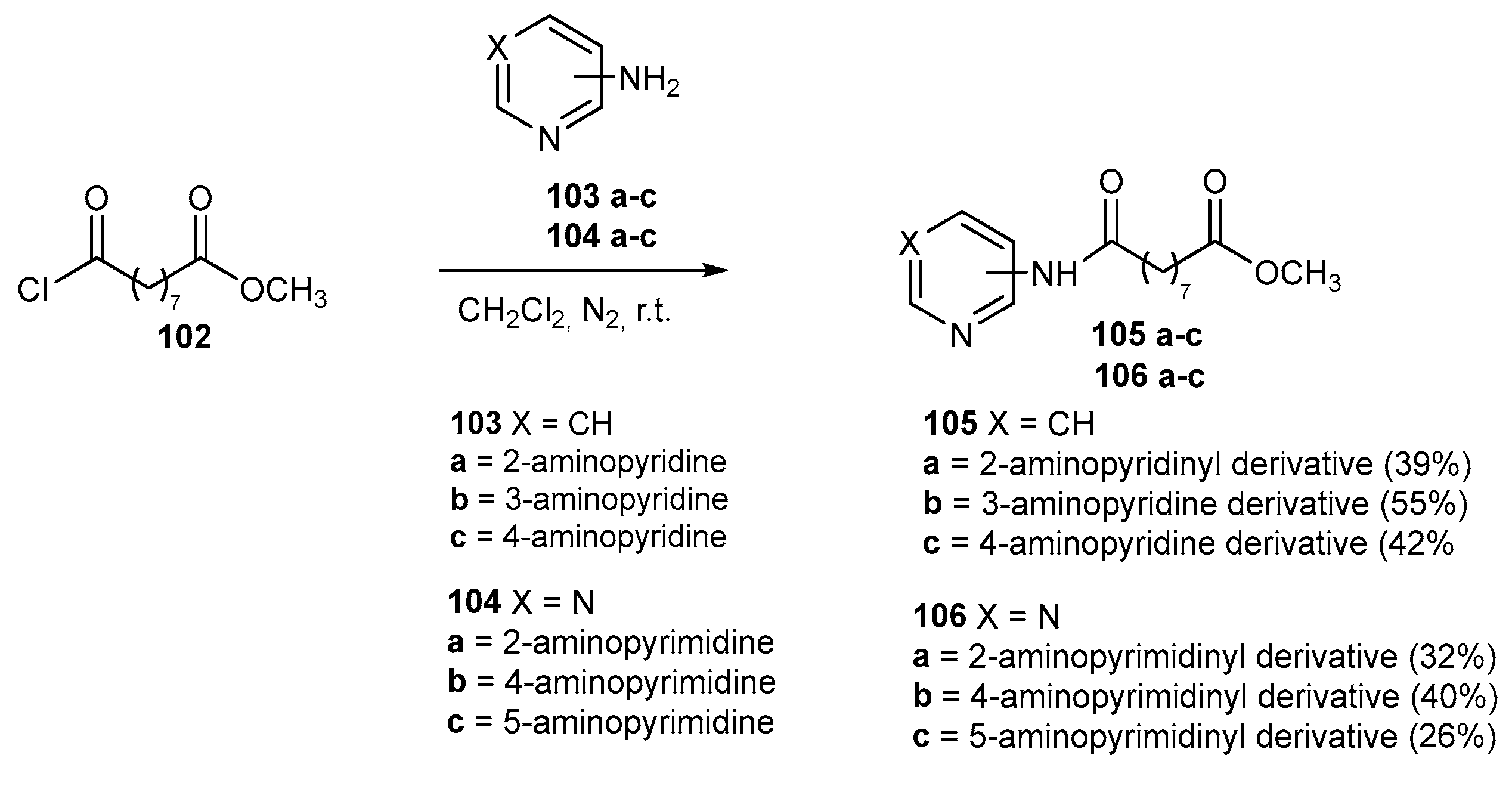
| Compound | R | HDAC1 IC50 (nM) |
HDAC3 IC50(nM) | HDAC6 IC50(nM) |
|---|---|---|---|---|
| 26c | 3-Cl, 4-F | 35.89 ± 16.34 | 37.67 ± 1.61 | 23.99 ± 0.72 |
| 26d | 3-CF3, 4-Cl | 40.84 ± 8.23 | 48.26 ± 1.78 | 30.00 ± 1.14 |
| 26e | H | 11.77 ± 0.50 | 20.77 ± 0.64 | 26.99 ± 4.95 |
| 26f | 4-CH3 | 14.01 ± 1.32 | 9.33 ± 0.10 | 19.68 ± 1.96 |
| 26g | 3-CH3, 4-CH3 | 29.82 ± 11.51 | 14.74 ± 0.03 | 16.87 ± 3.02 |
| SAHA | 93.34 ± 2.78 | 158.17 ± 6.66 | 78.98 ± 13.19 |
| Compound | HDAC3 IC50 ± SD (nM) |
HDAC8 IC50 ± SD (nM) |
|---|---|---|
| SAHA | 27± 1.0 | 440± 21 |
| 61 | 128 ± 9.8 | 17± 3 |
| 67 | 432± 52 | 487± 80 |
| 68a | 44± 5.8 | 76± 5.0 |
| 68b | 59± 1.0 | 82± 9.0 |
| 68c | 22± 1.3 | 28± 3.0 |
| 68d | 191± 18 | 147± 15 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




