Submitted:
17 November 2023
Posted:
22 November 2023
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
Experimental Conditions
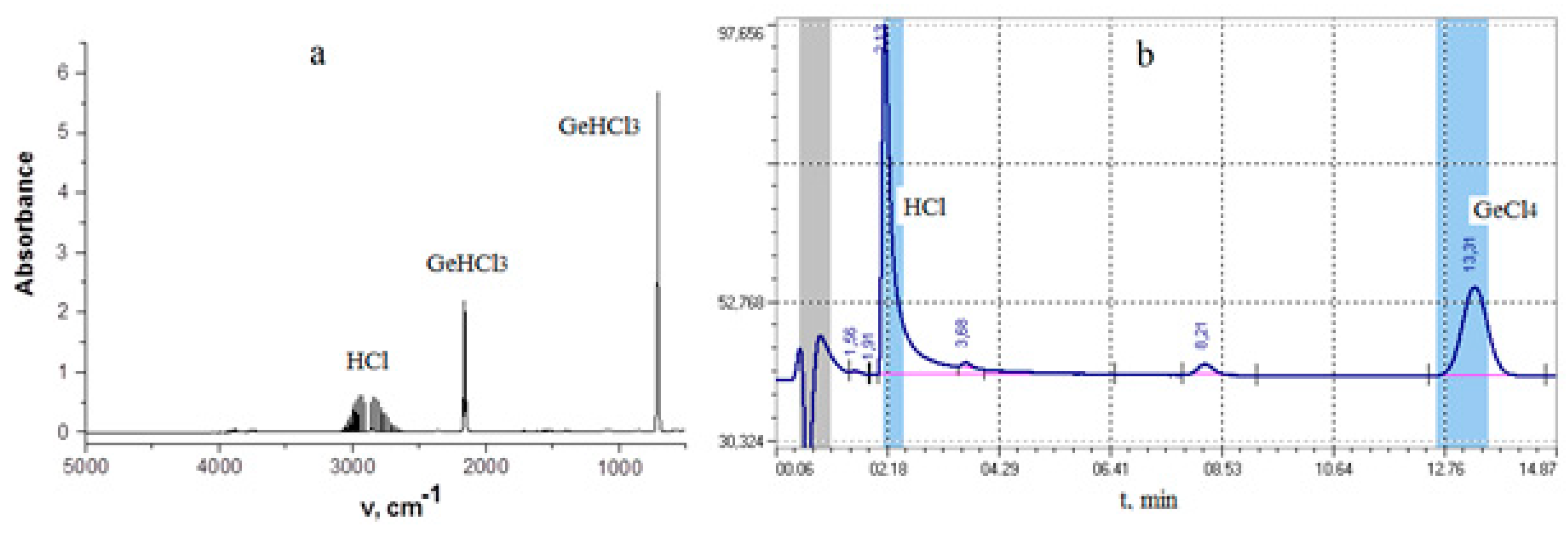
Gas chromatography analysis
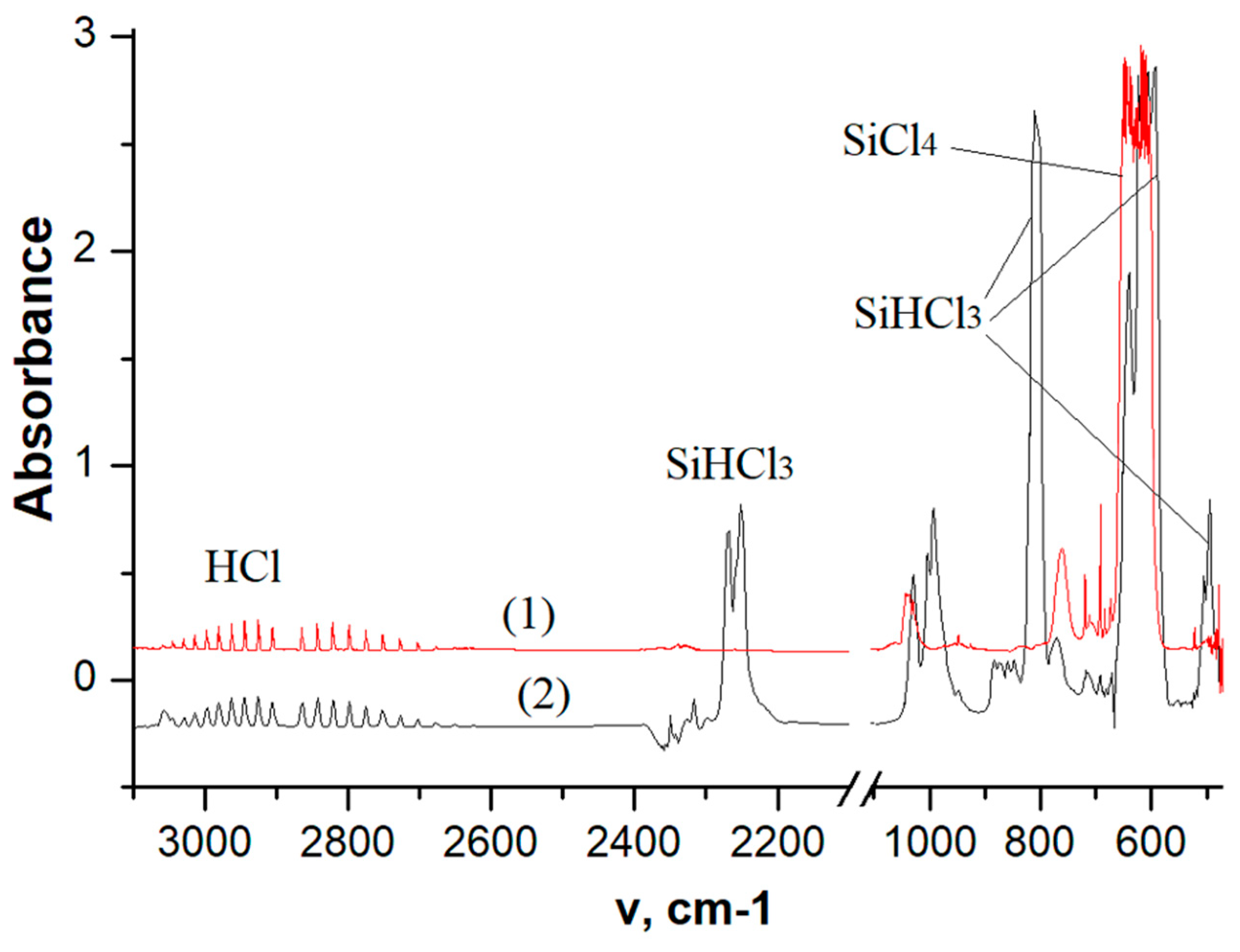
IR Spectroscopy of Exhaust Gases
Emission Spectroscopy
Gravimetric analysis
Methods for determining impurities in SiCl4, GeCl4, and chlorosilanes
Characterization of Deposits
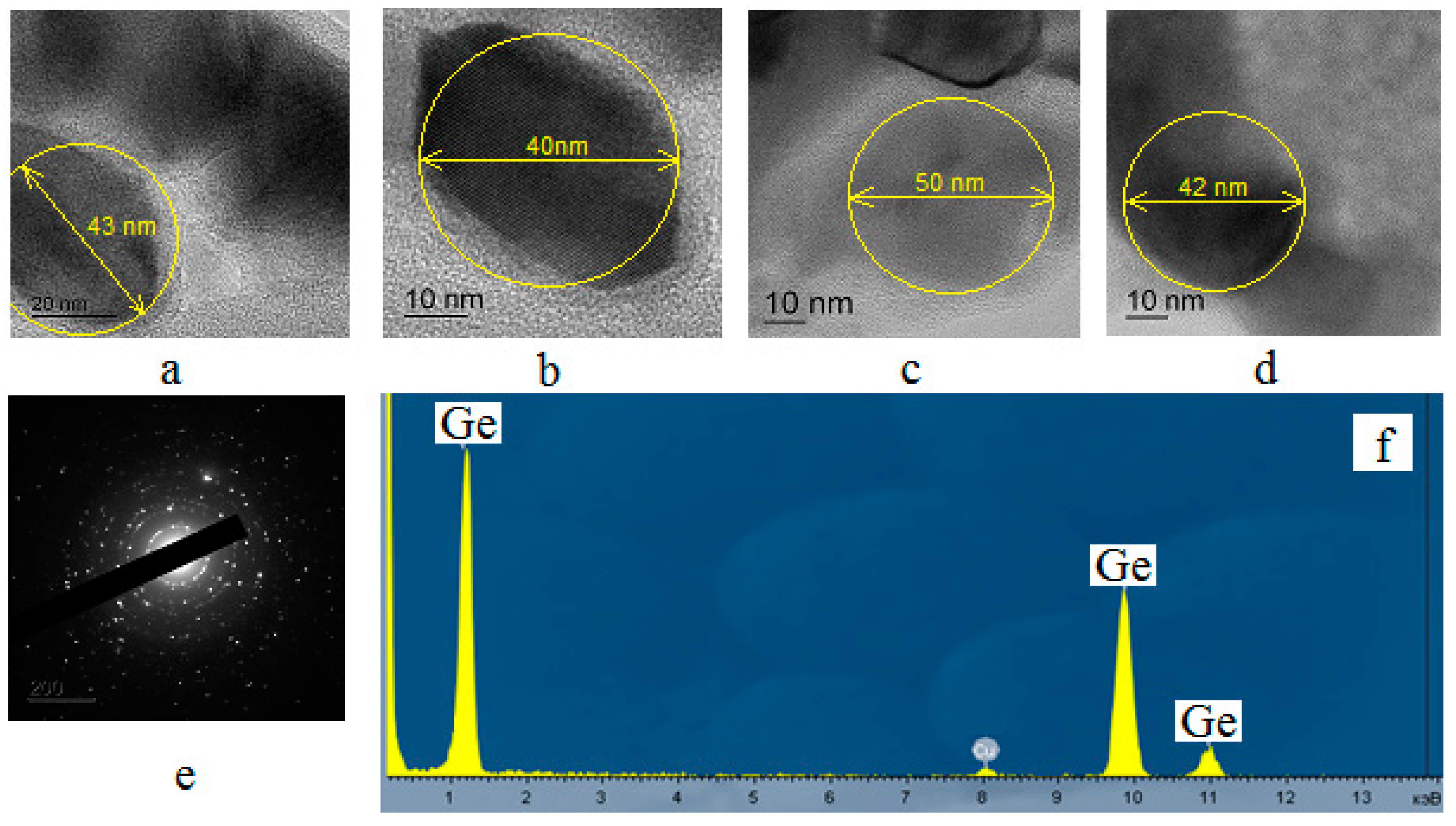
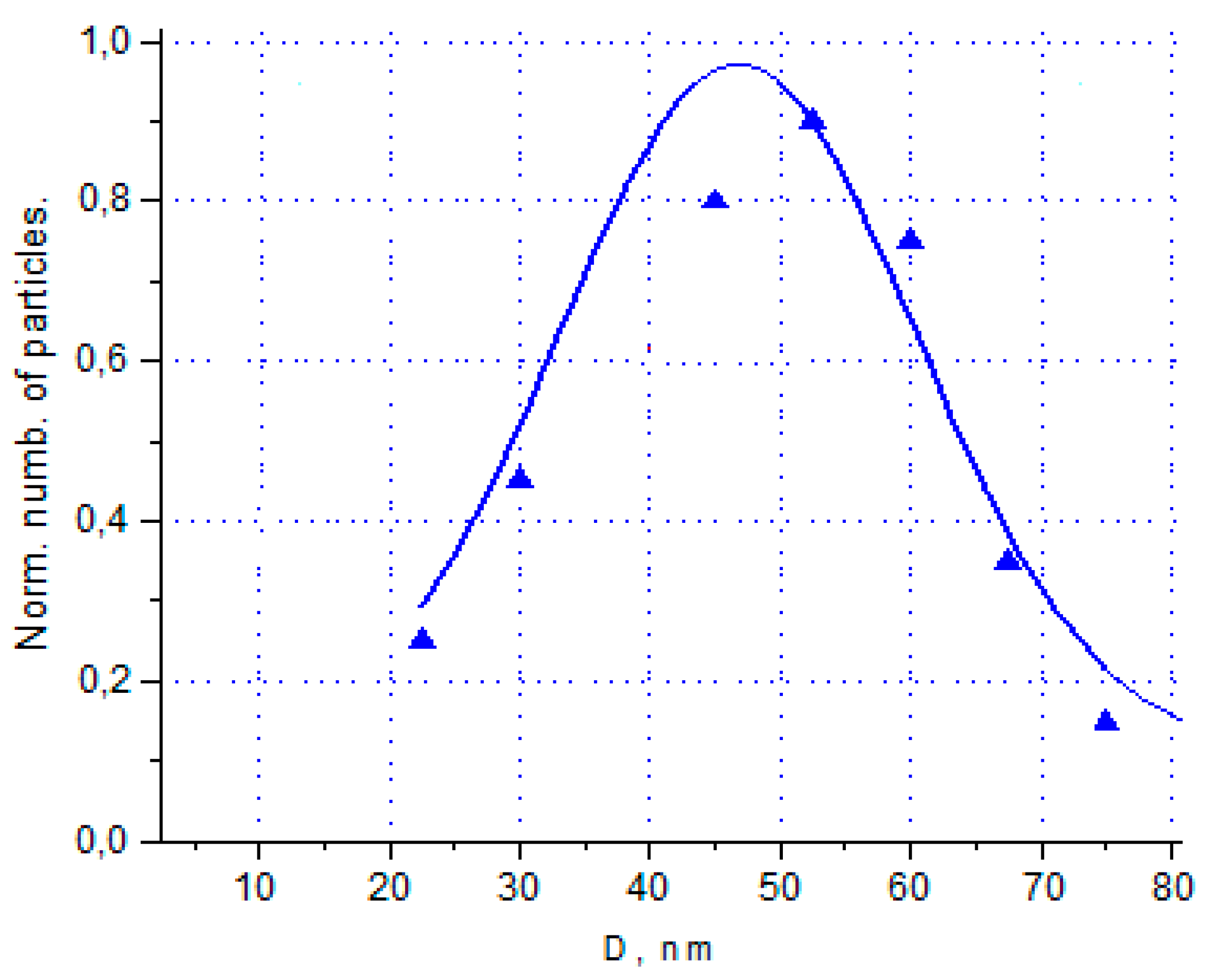
Morphological studies and analysis of elemental composition
Determination of impurity in Si and Ge
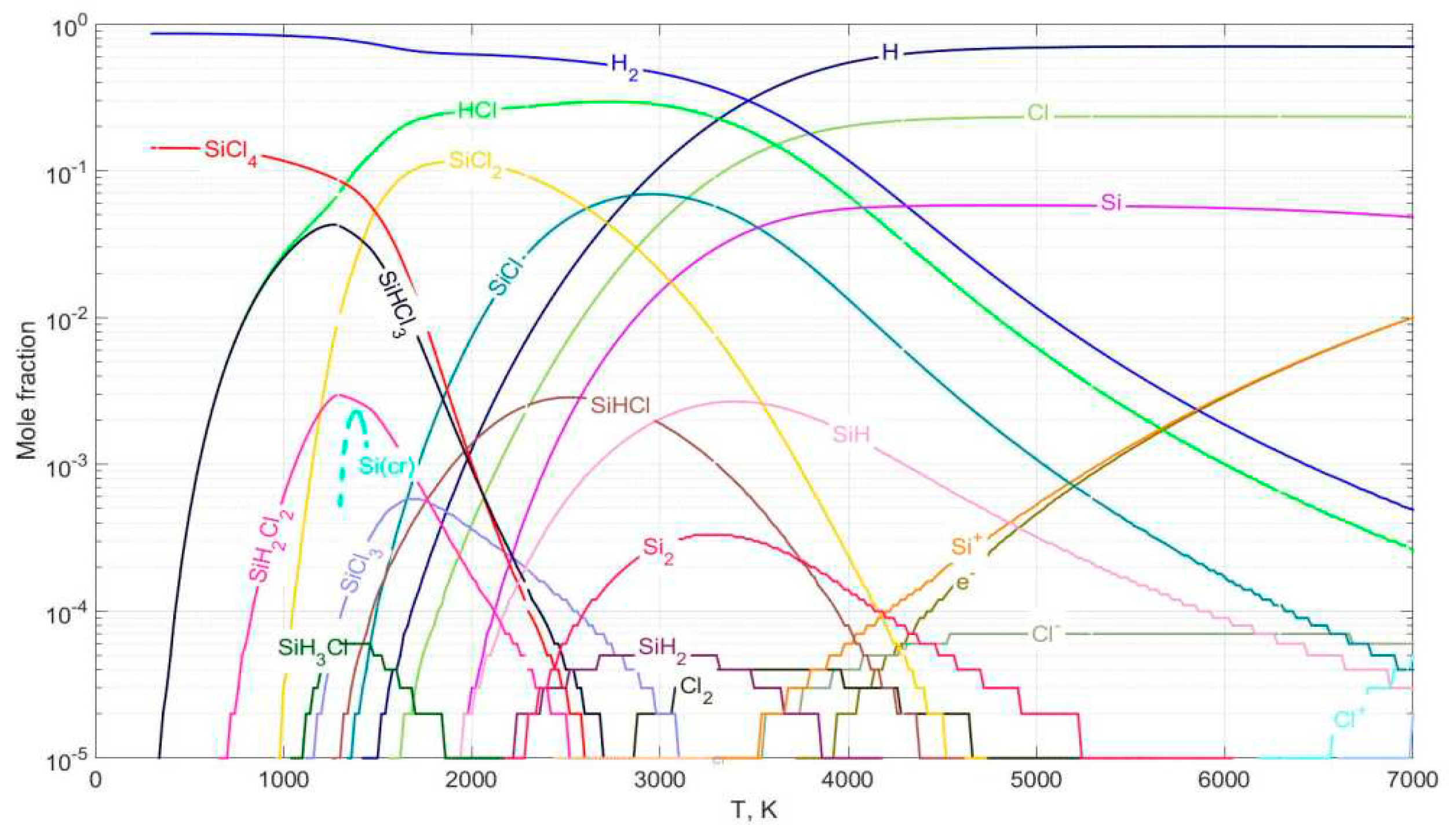
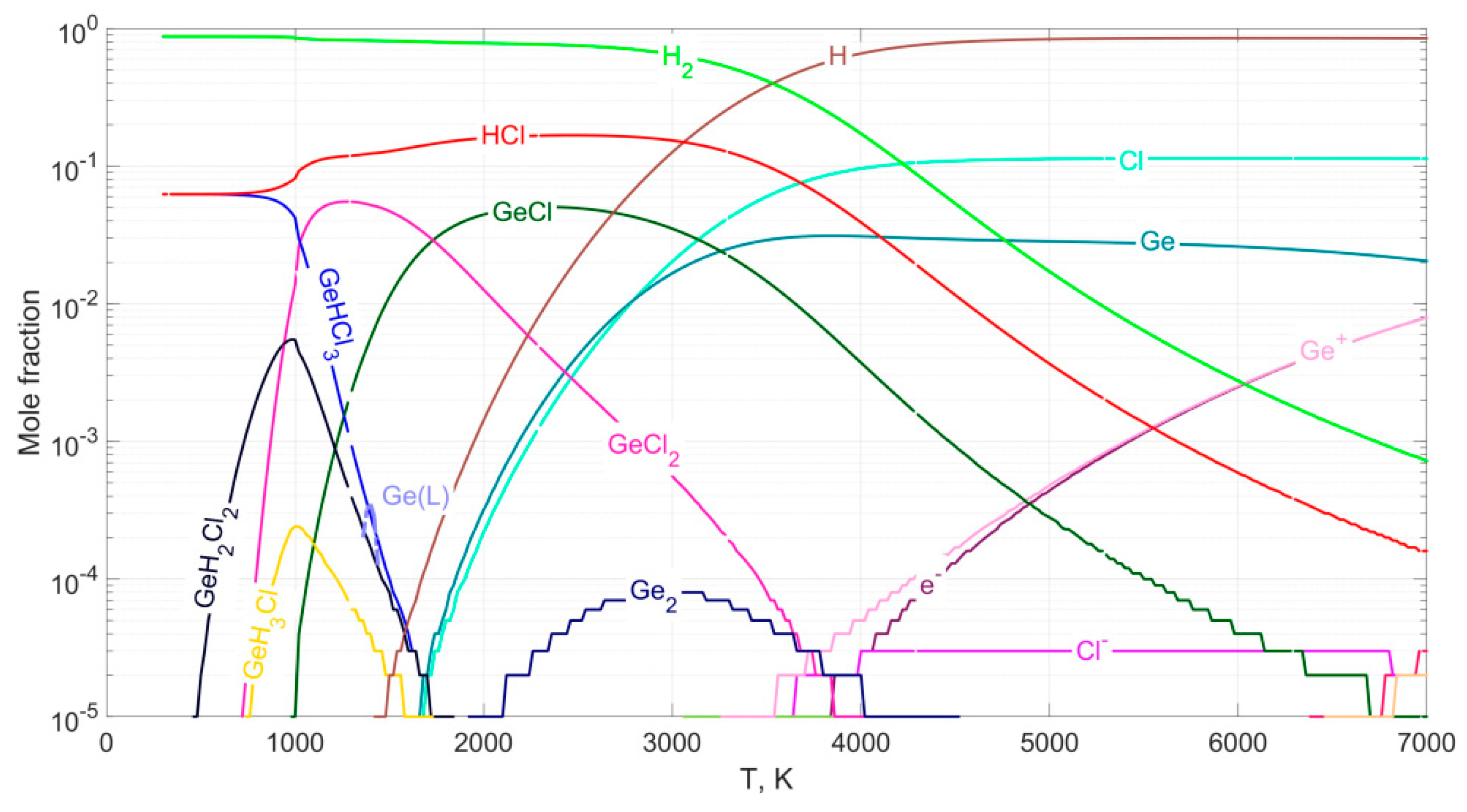
Thermodynamic calculation of chemical equilibrium
3. Discussion of the results.
Hydrogen reduction of SiCl4
Hydrogen reduction of GeCl4
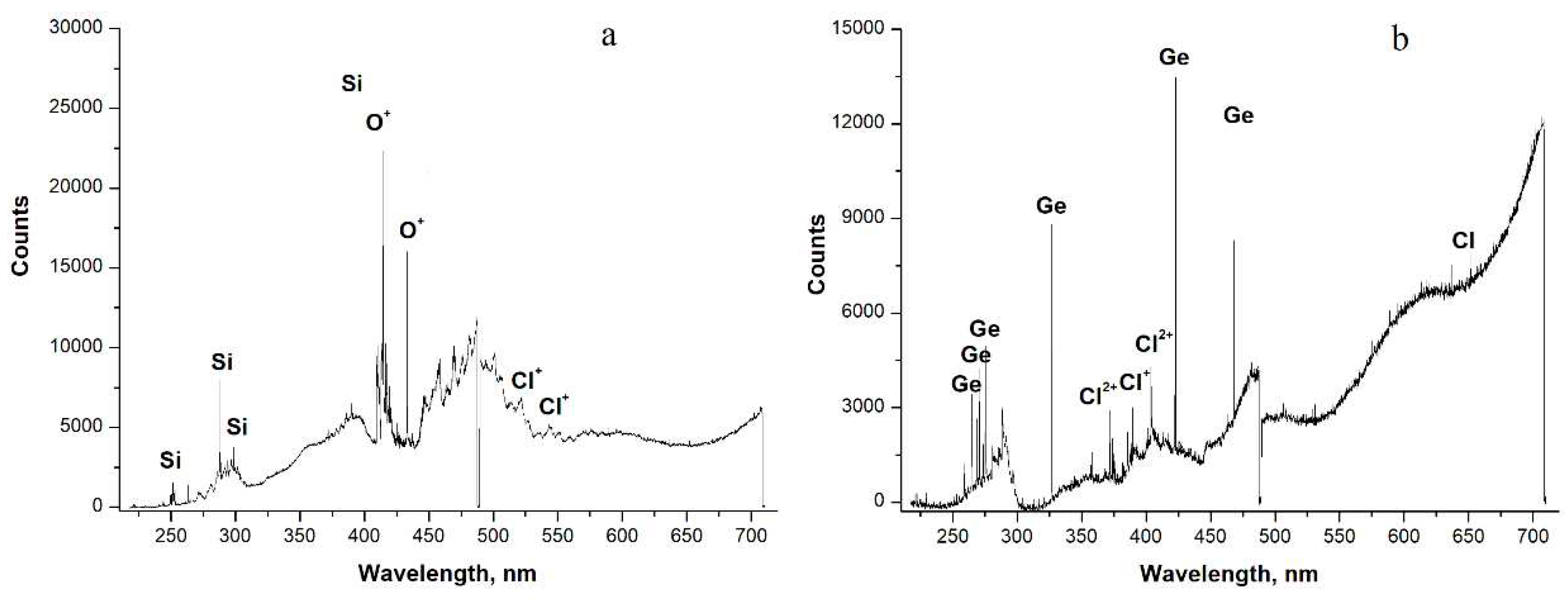
Investigation of the substances obtained
Characterization of samples obtained during the reduction of SiCl4
Samples of germanium obtained in RF-arc discharge from GeCl4
4 Conclusions
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Zulehner W (2000) Materials Science and Engineering B 73:7-15. [CrossRef]
- McHugo SA, Thompson AC, Mohammed A, Lamble G, Périchaud I, Martinuzzi S, Werner M, Rinio M, Koch W,. Hoefs HU, Haessler C (2001) Journal of Applied Physics. 89:4282-4288.
- Zhang X, Gong L, Wu B, Zhou M, Dai B (2015) Solar Energy Materials and Solar Cells 139:27-33.
- Cariou R, Tang J, Ramay N, Ruggeri R, RocaiCabarrocas P (2015) Solar Energy Materials & Solar Cells 134:15–21.
- Leal R, Dornstettera JC, Haddad F, Poulain G, Maurice JL, Cabarrocas PR (2015) 42nd Photovoltaic Specialist Conference (PVSC) New Orleans.
- Kim W, Matsuhara H, Onaka T (2005) Cryogenic Optical Systems and Instruments XI 5904:590418(12). [CrossRef]
- Fonollosa. J, Rubio R, Hartwig S, Marco S, Santander J, Fonseca L, Wollenstein J, Moreno M (2008) Sensors and Actuators B 132:498-507.
- Houssa M (2007) Germanium-Based Technologies: From Materials to Devices, Oxford:Elsevier.
- Raudorf TW, Trammel RC, Darken LS (1976) IEEE Trans. on Nucl. Sci 26:297-302.
- Pehl RH, Madden NW, Elliott JH (1979) IEEE Trans. on Nucl. Sci. 26:321-323.
- Eaglesham DJ, Cerullo M (1990) Phys. Rev. Lett. 64:1943–1947.
- Chu S, Majumdar A (2012) Nature 488:294–303.
- Bathey, BR, Cretella MC (2005) J. Mater Sci 17:3877–3896.
- Vorotyntsev A, Markov A, Petukhov A, Atlaskina M, Atlaskin A, Kapinos A, Vorotyntsev V, Pryakhina V (2021) Catal Ind 13:1–11.
- Matveev AK, Mochalov GM, Suvorov SS (2015) Method of obtaining silane and chlorosilanes RU Patent No 2608523.
- E; Claeys С (2007) Germanium-Based Technologies: From Materials to Devices Oxford:Elsevier Science.
- Jarkin VN, Kisarin OA, Kritskaya TV (2021) Izvestiya Vysshikh Uchebnykh Zavedenii. Materialy Elektronnoi Tekhniki 24:5-26. [In Russian].
- Sennikov PG, Kornev RA, Nazarov VV (2020) Sposob poluchenia visokochistogo polikrastallicheskogo kremnia Patent RF (RU) № 2739312. [In Russian].
- Lifeng W, Zhibin M, Aihua H, Jianhua W (2009) Inorganic Materials 45:1403-1407.
- Lifeng W, Zhibin M, Aihua H, Jianhua W (2010) Journal of Hazardous Materials 173:305-309.
- Zhenxi LU, Weigang Z (2014) Chinese Journal of Chemical Engineering 22: 227-233.
- Qingyon W, Hanbin C, Yuliang L, Xumei T, Zhijun H, Shuyong S, Yongxiang Y, Xiaoyan D (2010) Inorganic Materials 3:299-302.
- Gromov GN, Bolgov MV, Muravitski SA (2009) Sposob poluchenia trihlorsilana plazmohimichekim gidrirovaniem tetrahlorida kremnia I ustroistvo dlia ego osushestvlenia Patent RF (RU) № № 2350558. 685 [In Russian].
- Sarma KR, Rice MJ (1982) High pressure plasma hydrogenation of silicon tetrachloride US Patent № 4,309,259.
- Fang YZ, Ma WY, Zhou JH, Lu C, Wu JG (2008) Journal of Molecular Structure: THEOCHEM, 857:51-56.
- Menchikov LG, Ignatenko MA Pharmaceutical Chemistry Journal 46: 635 – 638.
- Gielen M, Tiekink ERT (2005) Metallotherapeutic drugs and metal-based diagnostic agents. The use of metals in medicine, Wiley.
- Ahadi AM, Hunter KI, Kramer NJ, Strunskus T, Kersten H, Faupel F, Kortshagen UR (2016) Physics Letters 108:093105.
- Gresback R, Holman Z, Kortshagen U (2007) Applied physics letters 91:093119(3).
- Whitham PJ, Strommen DP, Lundell S, Lau LD, Rodriguez R (2014) Plasma Chem Plasma Process 34:755-766.
- Lang JE, Rauleder H, Muh E (2013) Method for producing higher silanes with improved yield WO Patent № 007426 A1.
- Gusev AV, Kornev RA, Sukhanov AYu (2006) Inorganic Materials 42:1123-1026.
- Gribov LA, Smirnov VN (1961) Usp Fiz Nauk 527:527–567 [in Russian].
- https://cearun.grc.nasa.gov. Accessed on 15 April 2020.
- Smith WR, Missen RW (1982) Chemical reaction equilibrium analysis: theory and experiment, Wiley:New York.
- Shabanov SV, Gornushkin IB (2018) Appl Phys A 124:716.
- Shabanov SV, Gornushkin IB (2014) Spectrochim Acta B 100:147–172.
- Belov GV, Iorish VS, Yungman VS (2000) High Temp 38:191–196.
- Shabarova LV, Sennikov PG, Kornev RA, Plekhovich AD, Kutyin AM (2019) High Energy Chem 53:482-489.
- Shabarova LV, Plekhovich AD, Kutyin AM, Sennikov PG, Kornev RA (2019) High Energy Chem 53:482–489.
- Shabarova LV, Plekhovich AD, Kutyin AM, Sennikov PG, Kornev RA (2020) Theor Found Chem Eng 54:504–513 [in Russian].
- Sarma KR (1985) Hydrogenation Method silicon tetrachloride, Chanley C.S. Patent 4542004 USA.
- Wolf WE, Teichman R (1980) Z. anorg. allg Chem 460: 65-80.
- Sirtl E, Hunt LP, Sawyer DH (1974) J. Electrochem. Soc.: Solid-State Science and Technology 121:919-925.
- Kornev R, Sennikov P, Nazarov V, Sukhanov A, Shabarova L (2019) Plasma Physics and Technology 6:127-130.
- Sennikov PG, Kornev RA, Abrosimov NV, Bulanov AD, Gavva VA, Potapov AM (2019) Materials Science & Engineering B 244:1–5.


| Impurities | C, ppm (wt) | ||
|---|---|---|---|
| SiCl4 | Reaction products | Si | |
| Fe | 0.01 | 0.05 | 2.6 |
| Cu | 0.0005 | 0.0005 | 0.04 |
| Cr | 0.002 | 0.0008 | 2.2 |
| Mn | 0.004 | 0.002 | 2.2 |
| Ni | 0.0002 | 0.0004 | 1.3 |
| Mg | 0.01 | 0.01 | 1.6 |
| Al | 0.004 | 0.006 | <7 |
| B | --- | --- | <0.3 |
| P | --- | --- | < 2 |
| As | --- | --- | <0.3 |
| Sn | --- | --- | <0.03 |
| Impurities | GeCl4 | Ge | Impurities | GeCl4 | Ge |
|---|---|---|---|---|---|
| B | 0.3 | 0.5 | Co | 0.002 | 0.003 |
| Al | <1.9 | <1.9 | Fe | 0.03 | 8.1 |
| P | 1.2 | 1.2 | Cu | <0.2 | <0.2 |
| As | 0.5 | 1.5 | Zn | <0.1 | <0.1 |
| Sb | 0.003 | 0.003 | Cr | 0.8 | 0.7 |
| Sn | 0.1 | 0.1 | Mn | 0.01 | 0.01 |
| W | 0.1 | 170 | Mo | 0.8 | 0.6 |
| Ti | <0.2 | <0.2 | Mg | <0.4 | <0.4 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




