Submitted:
19 November 2023
Posted:
20 November 2023
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
2.1. Insect Cells
2.2. Virus Stocks
2.3. Constructs
2.3.1. Rep and Cap Gene Sources
2.3.2. LacR Gene Constructs
2.3.3. LacO Promoter Constructs
2.3.4. Making Recombinant VoyBac1.1 Bacmids by T4 Ligase Ligation
2.3.5. Tn7 Locus Cloning into the VoyBac1.1 Bacmid BEV
2.3.6. Tn7L-ITR-SEAP-ITR -Tn7R Donor Plasmid and ITR-SEAP Bacmid BEV
2.4. Sucrose Cushion rAAV Purification
2.5. Affinity Purification, CE-SDS Analysis, and Empty/Full Ratios
2.6. Western Blots
2.7. rAAV Transduction Assays.
2.8. Determination of rAAV Titer by Q-PCR.
2.9. SEAP Activity Assay Assays.
2.10. Statistical Calculations.
3. Results
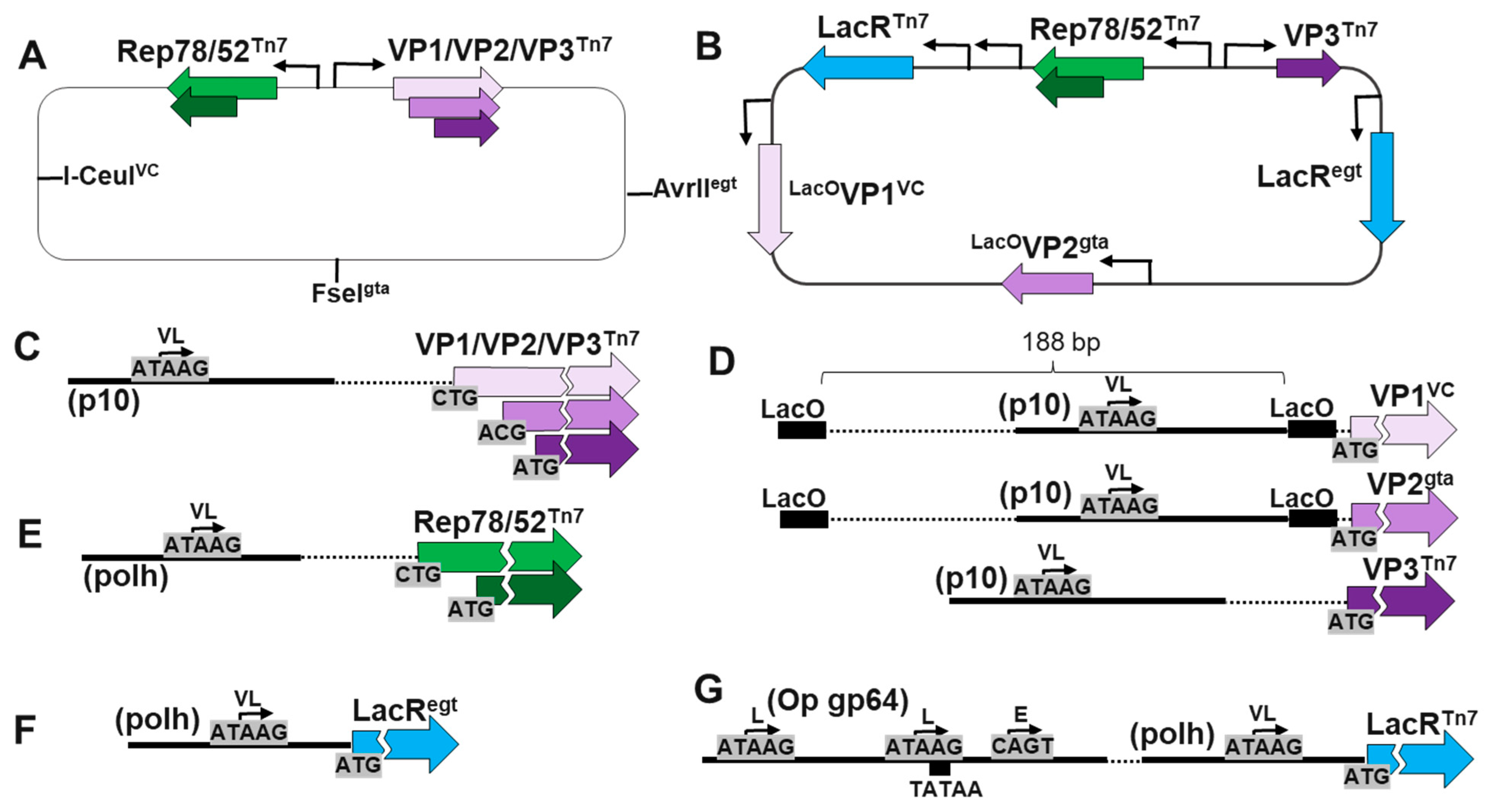
3.1. Multiple loci BEV cloning of Rep, VP1, VP2, VP3 gene cassettes.
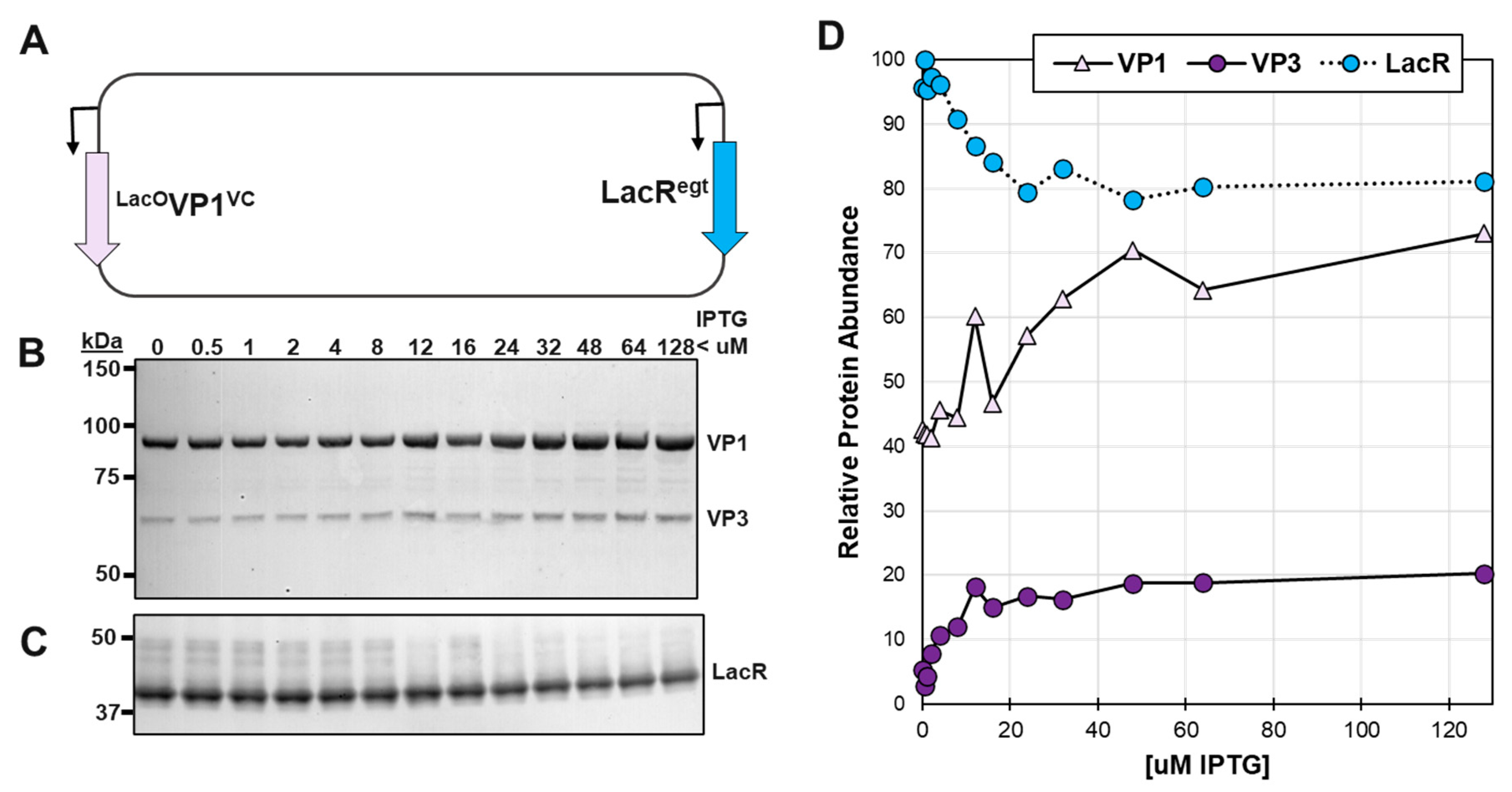
3.2. LacR was confirmed to be able to regulate expression of a LacOVP1 construct in the context of baculovirus infection.
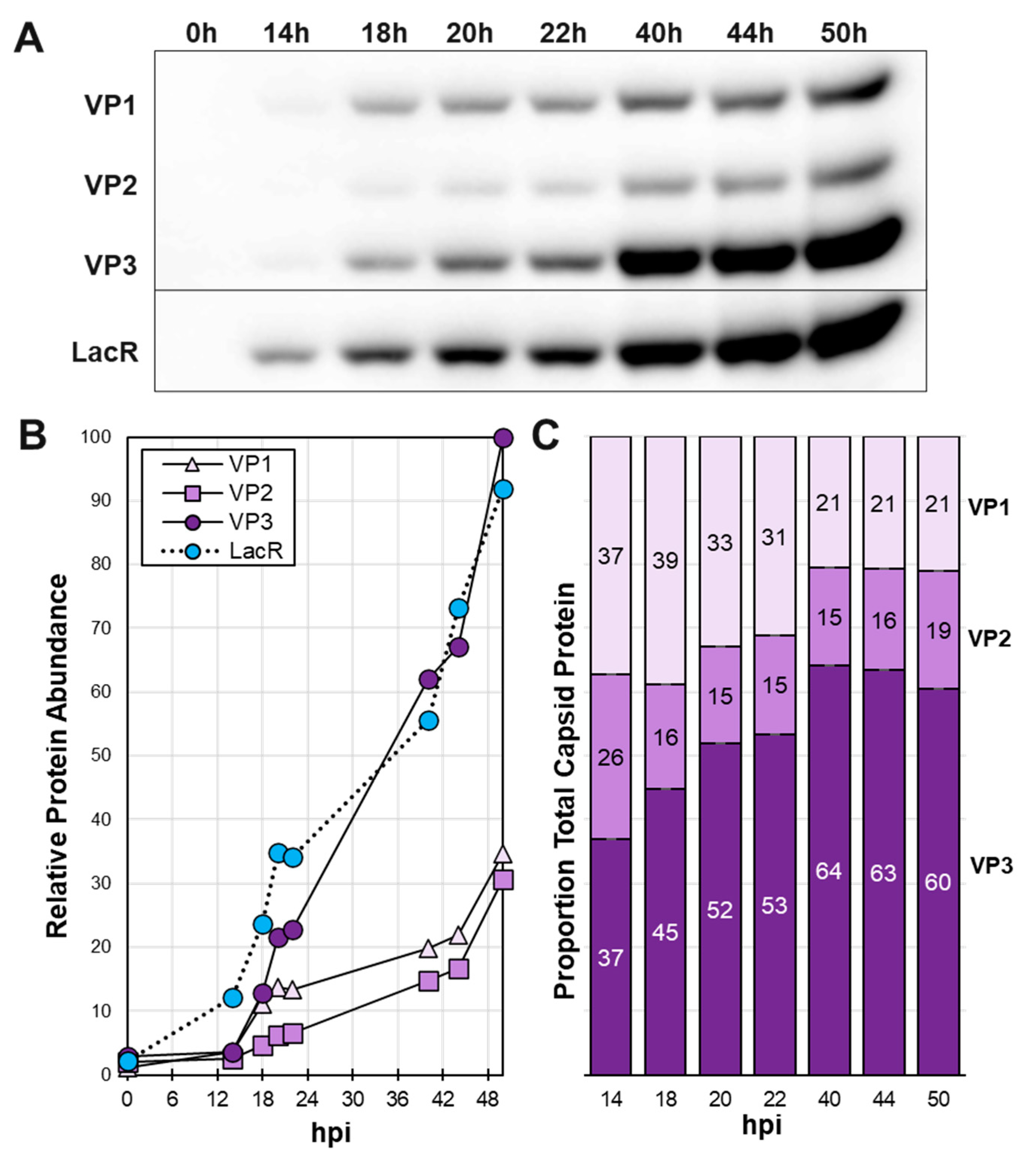
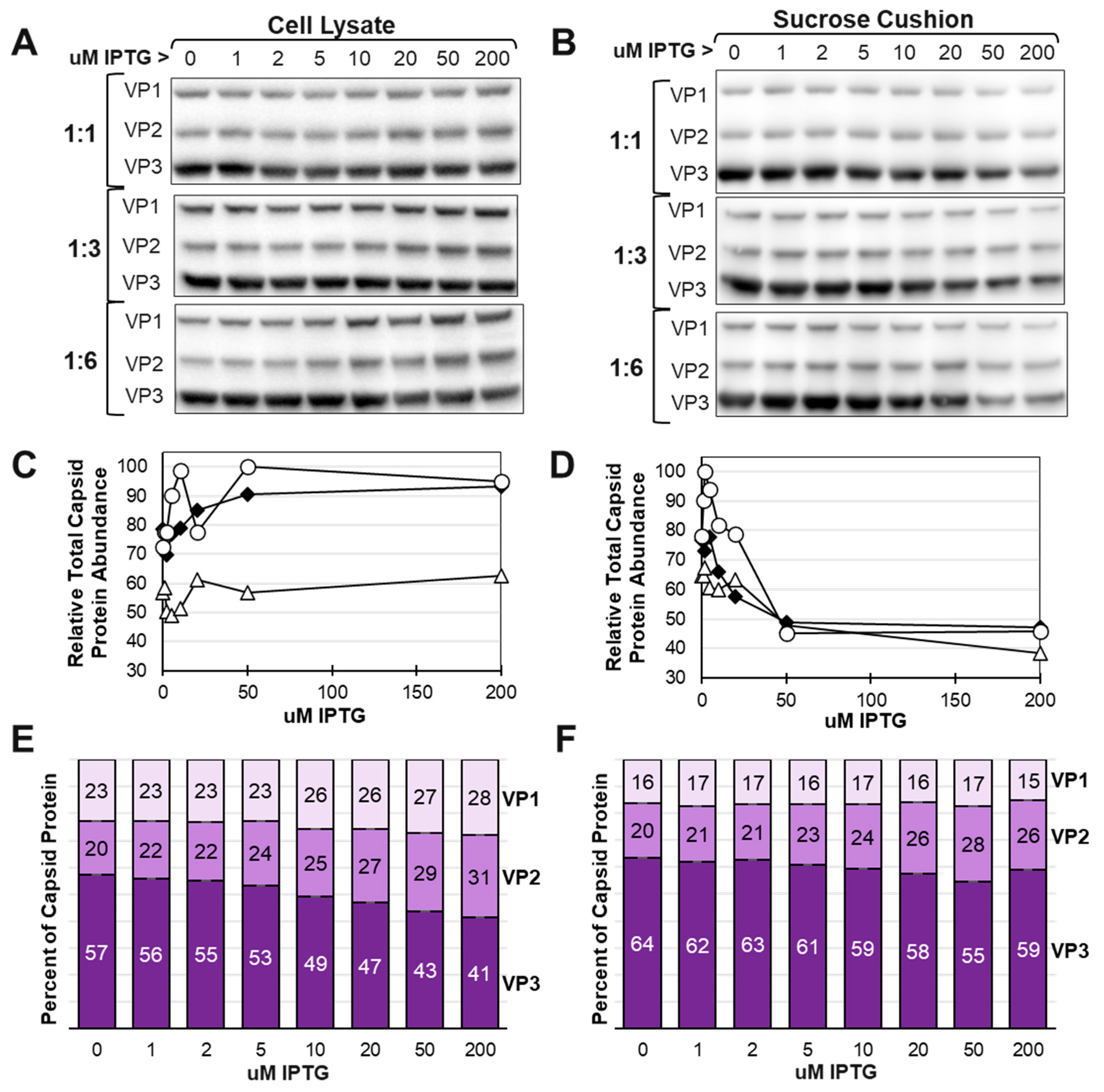
3.3. Time course of baculovirus infection in Sf9 cells reveals shifting VP ratios in inducible system.
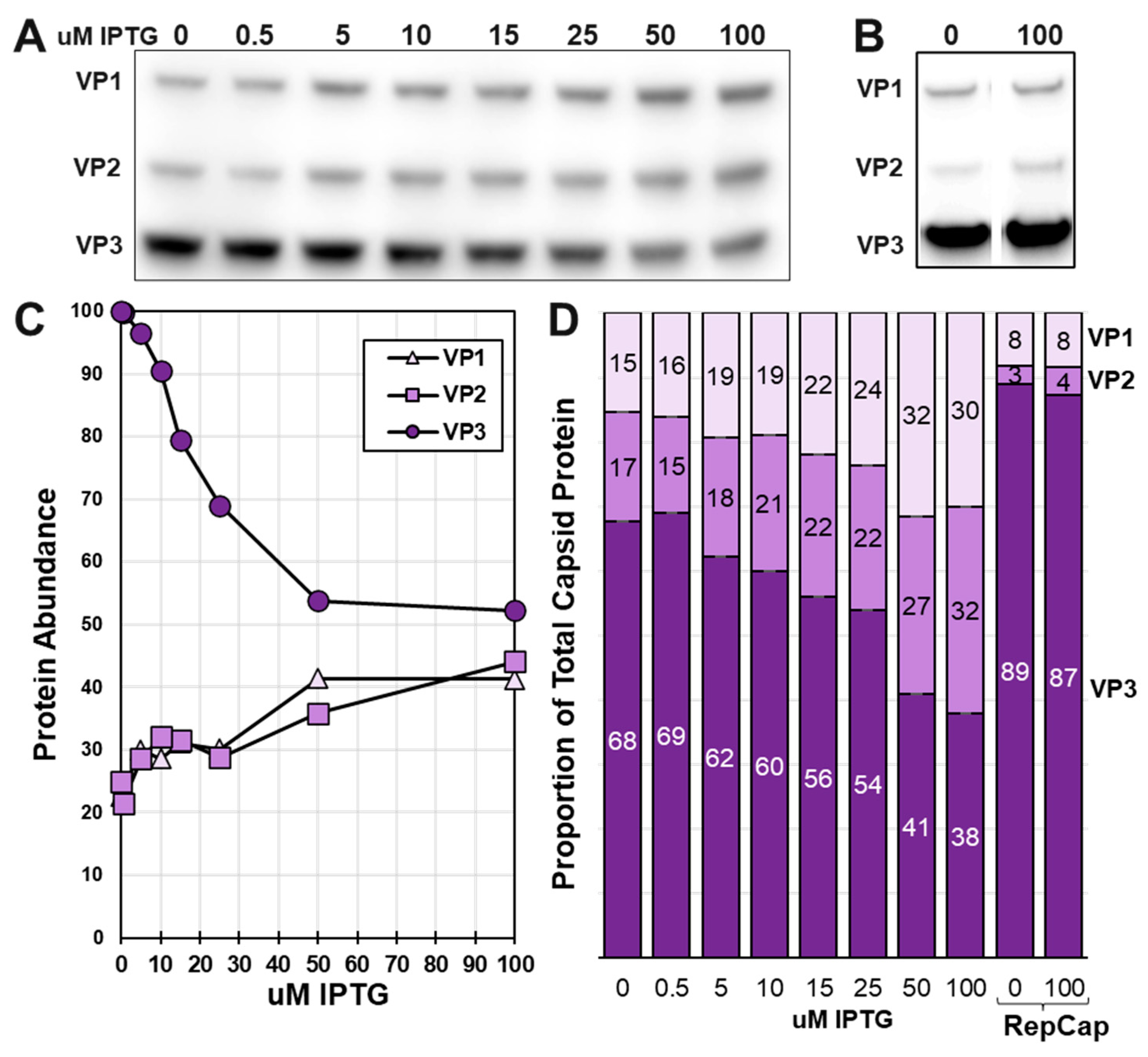
3.4. There is competition among very late promoters as IPTG induces LacR repression of LacO regulated very late promoters.
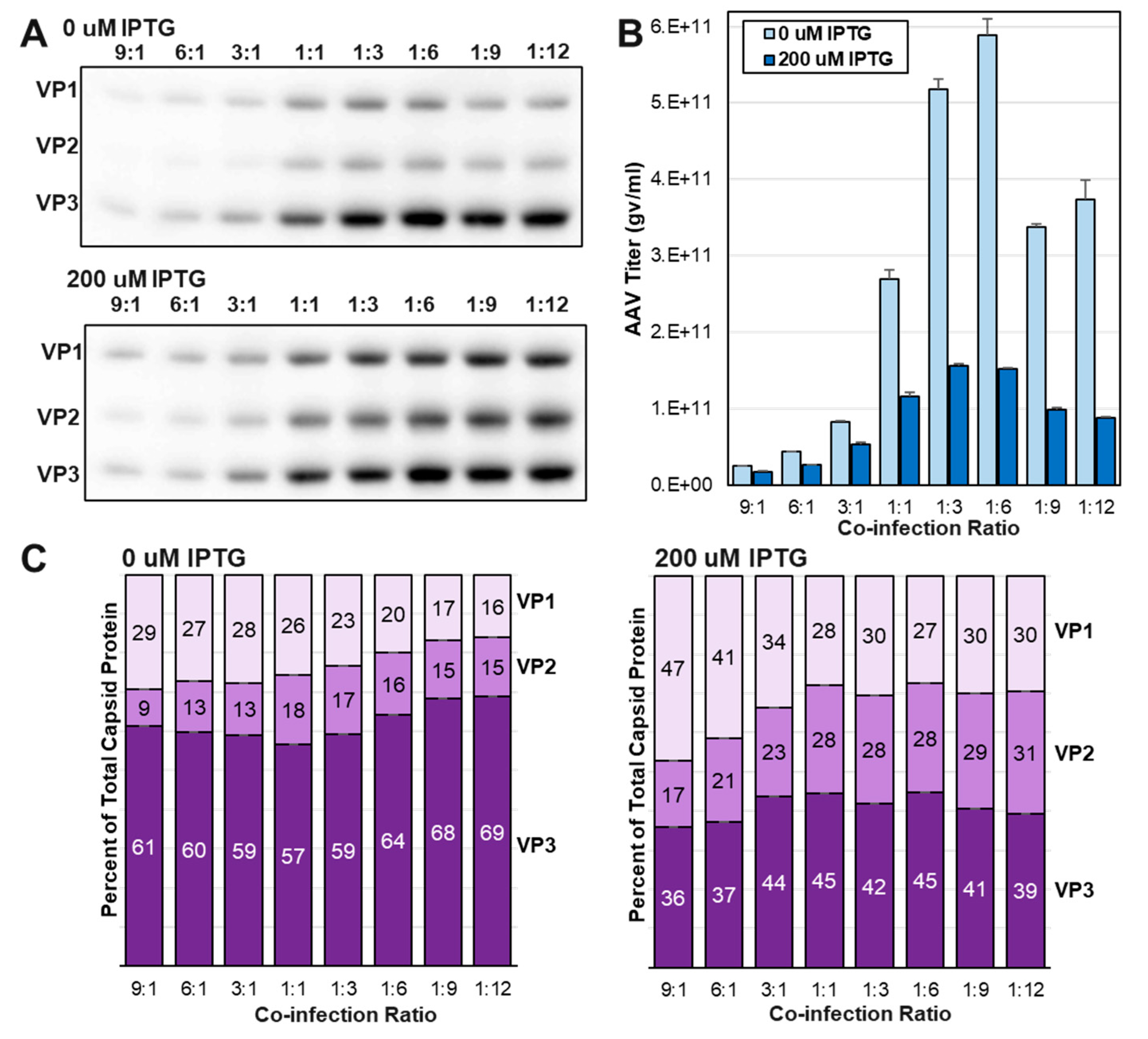
3.5. Increasing abundances of VP1 and VP2 led to reduced capsid titers at three ITR-SEAP:LacRepCap BEV co-infection ratios.
3.6. A high throughput method was developed to purify rAAV from BEV infected Sf9 cells
3.7. The yield of assembled capsids decreased as VP1 and VP2 expression was increased relative to VP3.
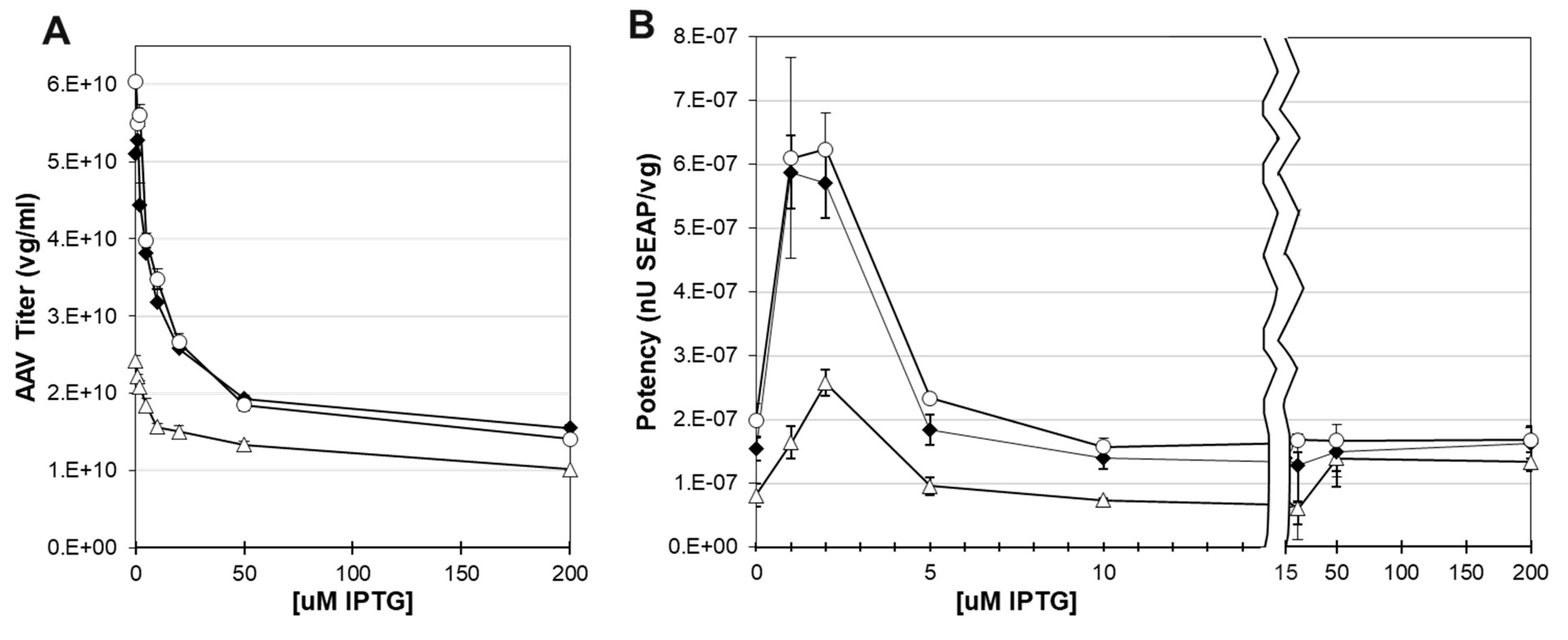
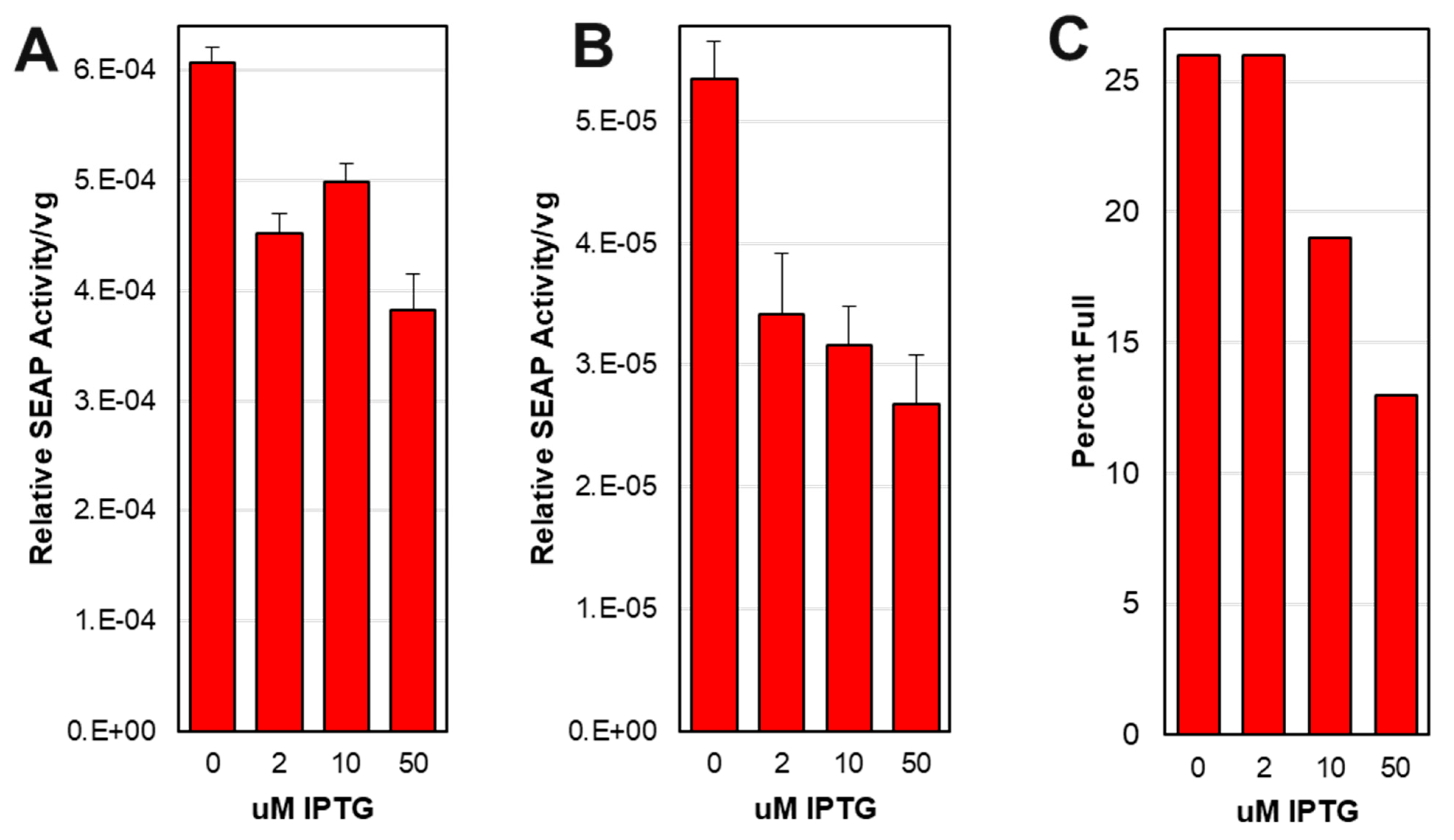
3.8. The potency of rAAV capsids was tunable using the Lac inducible system for small scale production.
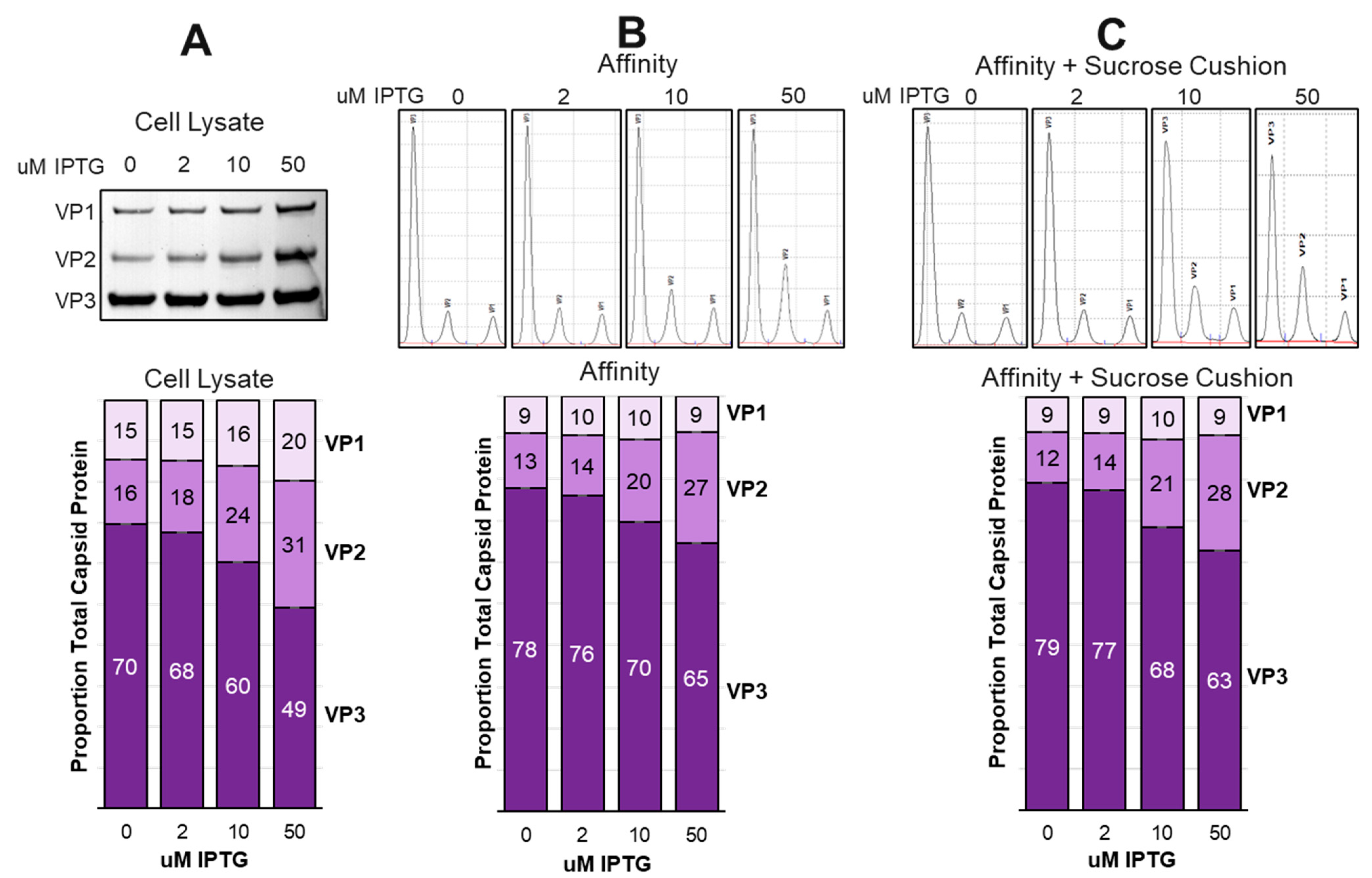
3.9. Larger scale production of rAAV in Sf9 cells confirmed that there was limited incorporation of VP1 into capsids.
3.10. Larger scale production did not show expected optimal potency at 2 uM IPTG.
4. Discussion
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Disclosures
References
- Blacklow, N.R.; Hoggan, M.D.; Rowe, W.P. Immunofluorescent studies of the potentiation of an adenovirus-associated virus by adenovirus 7. J. Exp. Med. 1967, 125, 755–765. [Google Scholar] [CrossRef] [PubMed]
- Buller, R.M.; Janik, J.E.; Sebring, E.D.; Rose, J.A. Herpes simplex virus types 1 and 2 completely help adenovirus-associated virus replication. J. Virol. 1981, 40, 241–247. [Google Scholar] [CrossRef] [PubMed]
- Ogston, P.; Raj, K.; Beard, P. Productive replication of adeno-associated virus can occur in human papillomavirus type 16 (HPV-16) episome-containing keratinocytes and is augmented by the HPV-16 E2 protein. J. Virol. 2000, 74, 3494–3504. [Google Scholar] [CrossRef] [PubMed]
- Kotin, R.M.; Menninger, J.C.; Ward, D.C.; Berns, K.I. Mapping and direct visualization of a region-specific viral DNA integration site on chromosome 19q13-qter. Genomics 1991, 10, 831–834. [Google Scholar] [CrossRef] [PubMed]
- Issa, S.S.; Shaimardanova, A.A.; Solovyeva, V.V.; Rizvanov, A.A. Various AAV Serotypes and Their Applications in Gene Therapy: An Overview. Cells 2023, 12, 785. [Google Scholar] [CrossRef] [PubMed]
- Kuzmin, D.A.; Shutova, M.V.; Johnston, N.R.; Smith, O.P.; Fedorin, V.V; et al. The clinical landscape for AAV gene therapies. Nat. Rev. Drug Discov. 2021, 20, 173–174. [Google Scholar] [CrossRef] [PubMed]
- Deverman, B.E.; Pravdo, P.L.; Simpson, B.P.; Kumar, S.R.; Chan, K.Y.; et al. Cre-dependent selection yields AAV variants for widespread gene transfer to the adult brain. Nat. Biotechnol. 2016, 34, 204–209. [Google Scholar] [CrossRef] [PubMed]
- Nonnenmacher, M.; Wang, W.; Child, M.A.; Ren, X.Q.; Huang, C.; Ren, A.Z.; Tocci, J.; Chen, Q.; Bittner, K.; Tyson, K.; Pande, N.; Chung, C.H.; Paul, S.M.; Hou, J. Rapid evolution of blood-brain-barrier-penetrating AAV capsids by RNA-driven biopanning. Mol. Ther. Methods Clin. Dev. 2020, 20. [Google Scholar] [CrossRef] [PubMed]
- Urabe, M.; Ding, C.; Kotin, R.M. Insect cells as a factory to produce adeno-associated virus type 2 vectors. Hum. Gene Ther. 2002, 13, 1935–1943. [Google Scholar] [CrossRef]
- Smith, R.H.; Levy, J.R.; Kotin, R.M. A simplified baculovirus-AAV expression vector system coupled with one-step affinity purification yields high-titer AAV stocks from insect cells. Mol. Ther. 2009, 17, 1888–1896. [Google Scholar] [CrossRef]
- Sonntag, F.; Schmidt, K.; Kleinschmidt, J.A. A viral assembly factor promotes AAV2 capsid formation in the nucleolus. Proc. Natl. Acad. Sci. USA 2010, 107, 10220–10225. [Google Scholar] [CrossRef] [PubMed]
- Maurer, A.C.; Pacouret, S.; Cepeda Diaz, A.K.; Blake, J.; Andres-Mateos, E.; et al. The assembly-activating protein promotes stability and interactions between AAV’s viral proteins to nucleate capsid assembly. Cell Rep. 2018, 23, 1817–1830. [Google Scholar] [CrossRef] [PubMed]
- Rose, J.A.; Maizel JVJr Inman, J.K.; Shatkin, A.J. Structural proteins of adenovirus-associated viruses. J. Virol. 1971, 8, 766–770. [Google Scholar] [CrossRef] [PubMed]
- Venkatakrishnan, B.; Yarbrough, J.; Domsic, J.; Bennett, A.; Bothner, B.; et al. Structure and dynamics of adeno-associated virus serotype 1 VP1-unique N-terminal domain and its role in capsid trafficking. J. Virol. 2013, 87, 4974–4984. [Google Scholar] [CrossRef] [PubMed]
- Johnson, F.B.; Ozer, H.L.; Hoggan, M.D. Structural proteins of adenovirus-associated virus type 3. J. Virol. 1971, 8, 860–863. [Google Scholar] [CrossRef] [PubMed]
- Kronenberg, S.; Kleinschmidt, J.A.; Böttcher, B. Electron cryo-microscopy and image reconstruction of adeno-associated virus type 2 empty capsids. EMBO Rep. 2001, 2, 997–1002. [Google Scholar] [CrossRef] [PubMed]
- Bosma, B.; du Plessis, F.; Ehlert, E.; Nijmeijer, B.; de Haan, M.; et al. Optimization of viral protein ratios for production of AAV serotype 5 in the baculovirus system. Gene Ther. 2018, 25, 415–424. [Google Scholar] [CrossRef]
- Kondratov, O.; Zolotukhin, S. Exploring the Comprehensive Kozak Sequence Landscape for AAV Production in Sf9 System. Viruses 2023, 15. [Google Scholar] [CrossRef]
- Summers, M.D.; Smith, G.E. A manual of methods for baculovirus vectors and insect cell culture procedures. Tex. Agric. Exp. Stn. Bull. No. 1555 1987. [Google Scholar]
- Vaughn, J.L.; Goodwin, R.H.; Tompkins, G.J.; McCawley, P. The establishment of two cell lines from the insect Spodoptera frugiperda (Lepidoptera; Noctuidae). In Vitro 1977, 13, 213–217. [Google Scholar] [CrossRef]
- Nguyen, C.; Ibe-Enwo, A.; Slack, J. A Baculovirus Expression Vector Derived Entirely from Non-Templated, Chemically Synthesized DNA Parts. Viruses 2023, 15. [Google Scholar] [CrossRef] [PubMed]
- Slack, J.M.; Blissard, G.W. Identification of two independent transcriptional activation domains in the Autographa californica multicapsid nuclear polyhedrosis virus IE1 protein. J. Virol. 1997, 71, 9579–9587. [Google Scholar] [CrossRef]
- Blissard, G.W.; Rohrmann, G.F. Baculovirus gp64 gene expression: analysis of sequences modulating early transcription and transactivation by IE1. J. Virol. 1991, 65, 5820–5827. [Google Scholar] [CrossRef] [PubMed]
- Chang, M.J.; Blissard, G.W. Baculovirus gp64 gene expression: negative regulation by a minicistron. J. Virol. 1997, 71, 7448–7460. [Google Scholar] [CrossRef] [PubMed]
- Luckow, V.A.; Lee, S.C.; Barry, G.F.; Olins, P.O. Efficient generation of infectious recombinant baculoviruses by site-specific transposon-mediated insertion of foreign genes into a baculovirus genome propagated in Escherichia coli. J. Virol. 1993, 67, 4566–4579. [Google Scholar] [CrossRef]
- Slack, J.M.; Kuzio, J.; Faulkner, P. Characterization of v-cath, a cathepsin L-like proteinase expressed by the baculovirus Autographa californica multiple nuclear polyhedrosis virus. J. Gen. Virol. 1995, 76 Pt 5, 1091–1098. [Google Scholar] [CrossRef]
- Lihoradova, O.A.; Ogay, I.D.; Abdukarimov, A.A.; Azimova, S.h.S.; Lynn, D.E.; et al. The Homingbac baculovirus cloning system: An alternative way to introduce foreign DNA into baculovirus genomes. J. Virol. Methods 2007, 140, 59–65. [Google Scholar] [CrossRef] [PubMed]
- McIntosh, N.L.; Berguig, G.Y.; Karim, O.A.; Cortesio, C.L.; De Angelis, R.; Khan, A.A.; Gold, D.; Maga, J.A.; Bhat, V.S. Comprehensive characterization and quantification of adeno associated vectors by size exclusion chromatography and multi angle light scattering. Sci. Rep. 2021, 11, 3012. [Google Scholar] [CrossRef]
- Schneider, C.; Rasband, W.; Eliceiri, K. NIH Image to ImageJ: 25 years of image analysis. Nat. Methods 2012, 9, 671–675. [Google Scholar] [CrossRef] [PubMed]
- Oehler, S.; Eismann, E.R.; Krämer, H.; Müller-Hill, B. The three operators of the lac operon cooperate in repression. EMBO J. 1990, 9, 973–979. [Google Scholar] [CrossRef]
- Chen, Y.R.; Zhong, S.; Fei, Z.; Hashimoto, Y.; Xiang, J.Z.; et al. The transcriptome of the baculovirus Autographa californica multiple nucleopolyhedrovirus in Trichoplusia ni cells. J. Virol. 2013, 87, 6391–6405. [Google Scholar] [CrossRef] [PubMed]
- Chen, S.H.; Papaneri, A.; Walker, M.; Scappini, E.; Keys, R.D.; et al. A simple, two-step, small-scale purification of recombinant adeno-associated viruses. J. Virol. Methods 2020, 281, 113863. [Google Scholar] [CrossRef] [PubMed]
- Katsuma, S.; Koyano, Y.; Kang, W.; Kokusho, R.; Kamita, S.G.; et al. The baculovirus uses a captured host phosphatase to induce enhanced locomotory activity in host caterpillars. PLoS Pathog. 2012, 8, e1002644. [Google Scholar] [CrossRef] [PubMed]
- Mietzsch, M.; Smith, J.K.; Yu, J.C.; Banala, V.; Emmanuel, S.N.; et al. Characterization of AAV-Specific Affinity Ligands: Consequences for Vector Purification and Development Strategies. Mol. Ther. Methods Clin. Dev. 2020, 19, 362–373. [Google Scholar] [CrossRef] [PubMed]
- Fuerst, T.R.; Fernandez, M.P.; Moss, B. Transfer of the inducible lac repressor/operator system from Escherichia coli to a vaccinia virus expression vector. Proc. Natl. Acad. Sci. USA 1989, 86, 2549–2553. [Google Scholar] [CrossRef] [PubMed]
- Zhao, C.; Crews, C.J.; Derdeyn, C.A.; Blackwell, J.L. Lac-regulated system for generating adenovirus 5 vaccine vectors expressing cytolytic human immunodeficiency virus 1 genes. J. Virol. Methods 2009, 160, 101–110. [Google Scholar] [CrossRef] [PubMed]
- Wyatt, L.S.; Xiao, W.; Americo, J.L.; Earl, P.L.; Moss, B. Novel nonreplicating vaccinia virus vector enhances expression of heterologous genes and suppresses synthesis of endogenous viral proteins. mBio 2017, 8, e00790-17. [Google Scholar] [CrossRef] [PubMed]
- Matthews, D.A.; Cummings, D.; Evelegh, C.; Graham, F.L.; Prevec, L. Development and use of a 293 cell line expressing lac repressor for the rescue of recombinant adenoviruses expressing high levels of rabies virus glycoprotein. J. Gen. Virol. 1999, 80 Pt 2, 345–353. [Google Scholar] [CrossRef] [PubMed]
- Jacob, F.; Monod, J. Genetic regulatory mechanisms in the synthesis of proteins. J. Mol. Biol. 1961, 3, 318–356. [Google Scholar] [CrossRef]
- Lewis, M. The lac repressor. C R Biol. 2005, 328, 521–548. [Google Scholar] [CrossRef]
- Slack, J.M.; Blissard, G.W. Measurement of membrane fusion activity from viral membrane fusion proteins based on a fusion-dependent promoter induction system in insect cells. J. Gen. Virol. 2001, 82 Pt 10, 2519–2529. [Google Scholar] [CrossRef]
- Gao, K.; Li, M.; Zhong, L.; Su, Q.; Li, J.; et al. Empty virions in AAV8 vector preparations reduce transduction efficiency and may cause total viral particle dose-limiting side-effects. Mol. Ther. Methods Clin. Dev. 2014, 1, 20139. [Google Scholar] [CrossRef]
- Wörner, T.P.; Snijder, J.; Bennett, A.; Agbandje-McKenna, M.; Makarov, A.A.; et al. Resolving heterogeneous macromolecular assemblies by Orbitrap-based single-particle charge detection mass spectrometry. Nat. Methods 2020, 17, 395–398. [Google Scholar] [CrossRef]
- Briand, L.; Marcion, G.; Kriznik, A.; Heydel, J.M.; Artur, Y.; et al. A self-inducible heterologous protein expression system in Escherichia coli. Sci. Rep. 2016, 6, 33037. [Google Scholar] [CrossRef]
- Kaneda, Y.; Kinoshita, K.; Sato, M.; Saeki, Y.; Yamada, R.; et al. The induction of apoptosis in HeLa cells by the loss of LBP-p40. Cell Death Differ. 1998, 5, 20–28. [Google Scholar] [CrossRef]
- Cronin, C.A.; Gluba, W.; Scrable, H. The lac operator-repressor system is functional in the mouse. Genes Dev. 2001, 15, 1506–1517. [Google Scholar] [CrossRef]
- Figge, J.; Wright, C.; Collins, C.J.; Roberts, T.M.; Livingston, D.M. Stringent regulation of stably integrated chloramphenicol acetyl transferase genes by E. coli lac repressor in monkey cells. Cell 1988, 52, 713–722. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).



