Submitted:
15 February 2024
Posted:
19 February 2024
Read the latest preprint version here
Abstract
Keywords:
1. Introduction
2. Simulation Details
3. Results and Discussion
3.1. Subatomic Particle Dynamics Simulations for Particle Nature
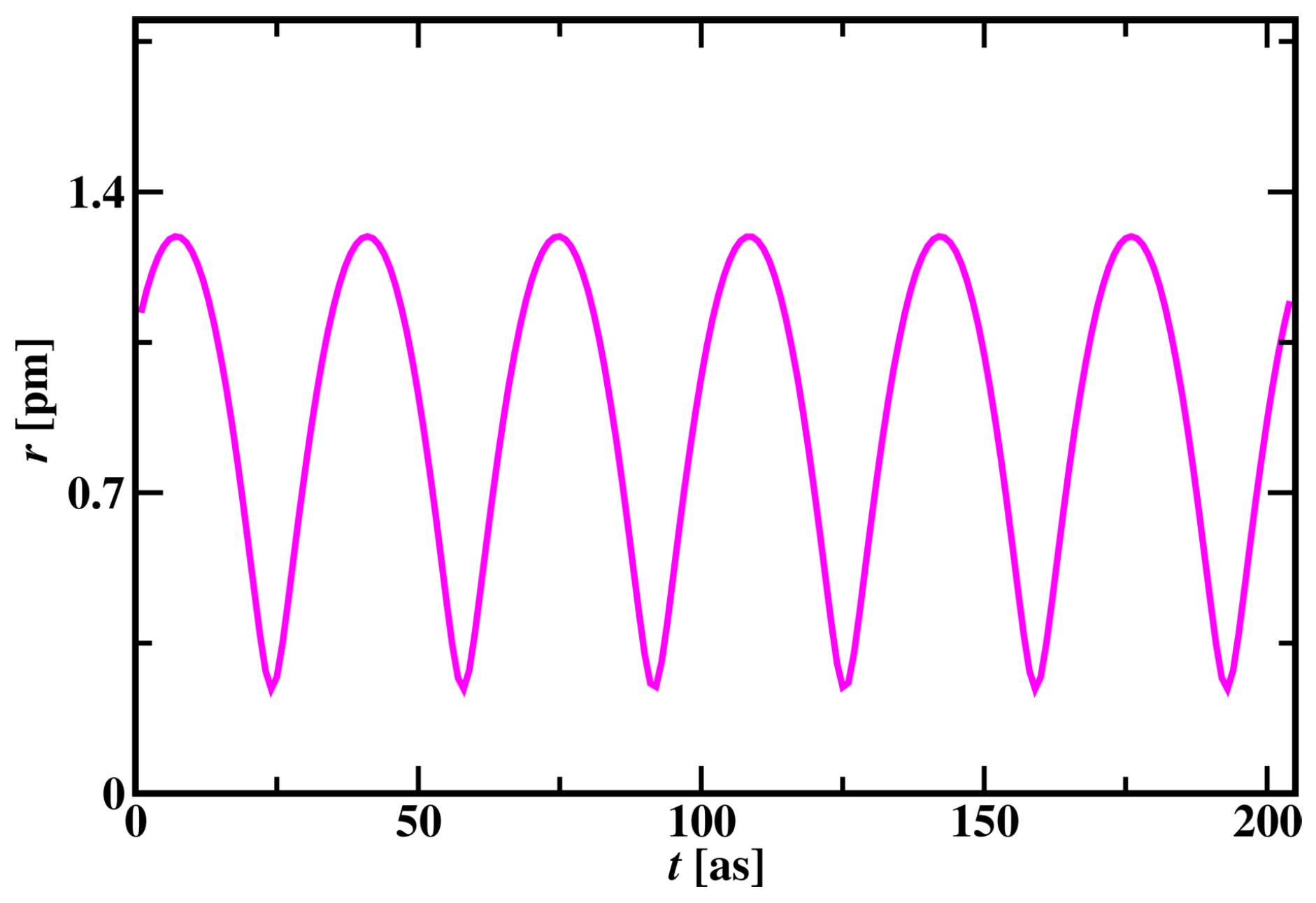
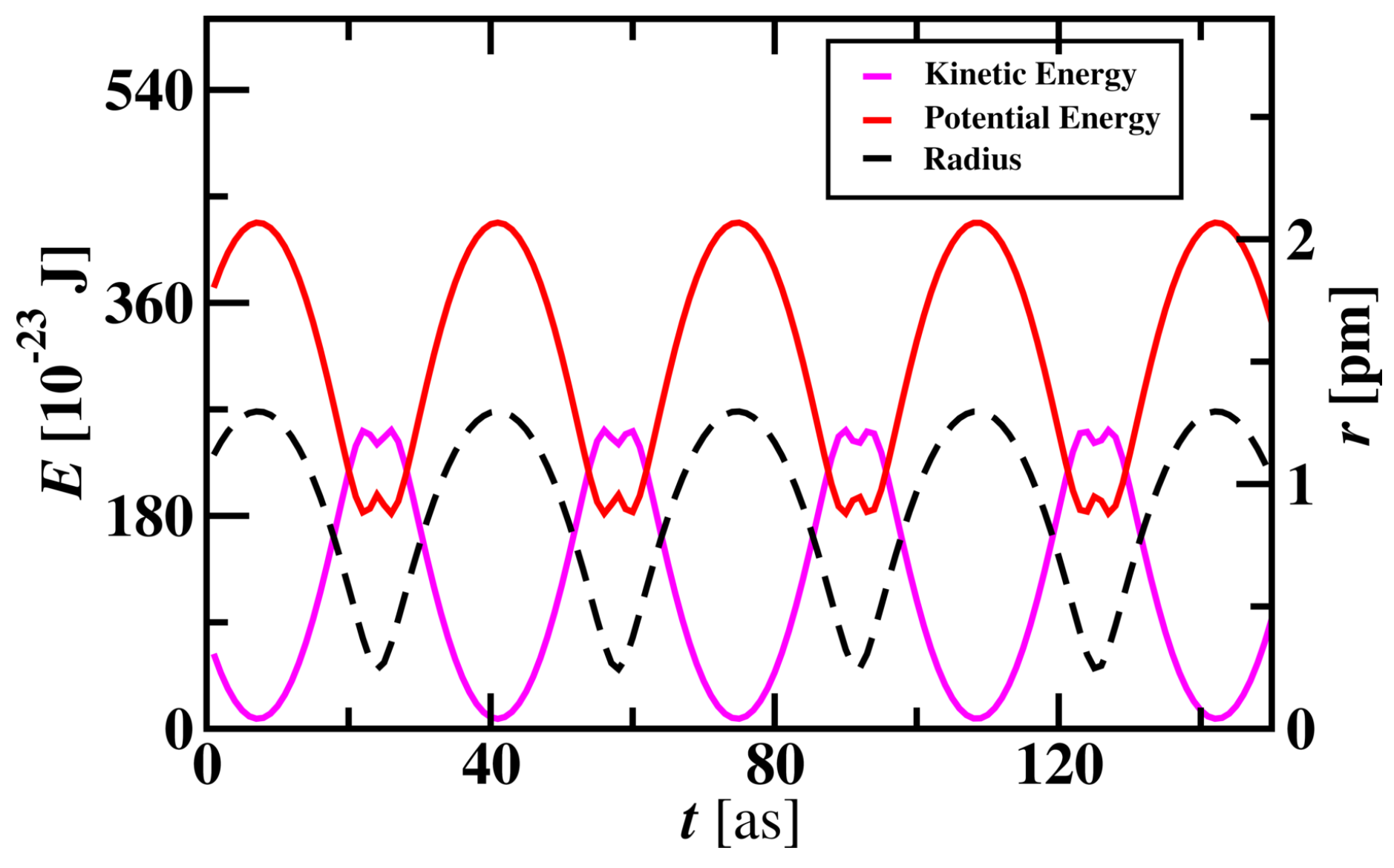
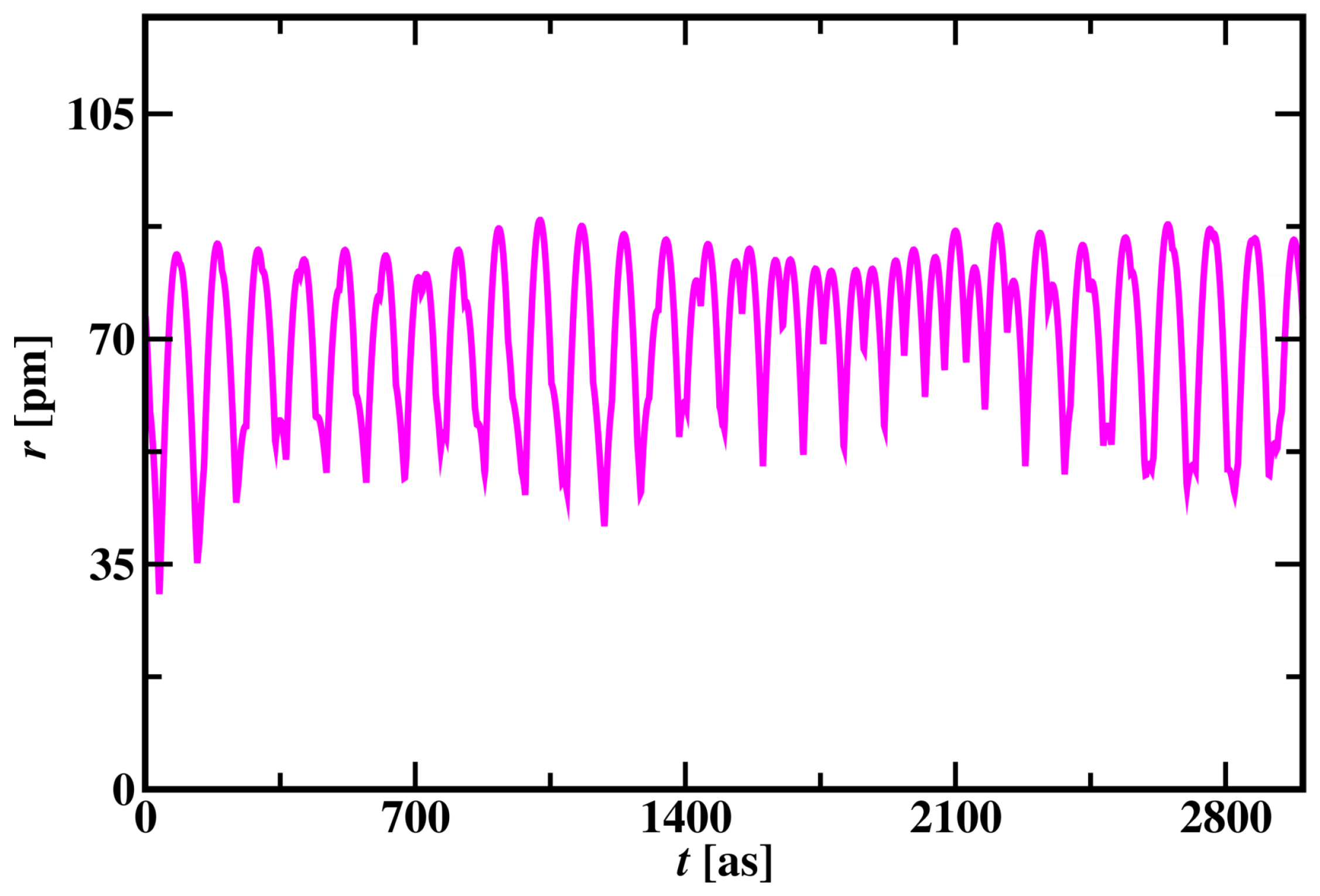
3.1.1. Hydrogen Atom Simulation
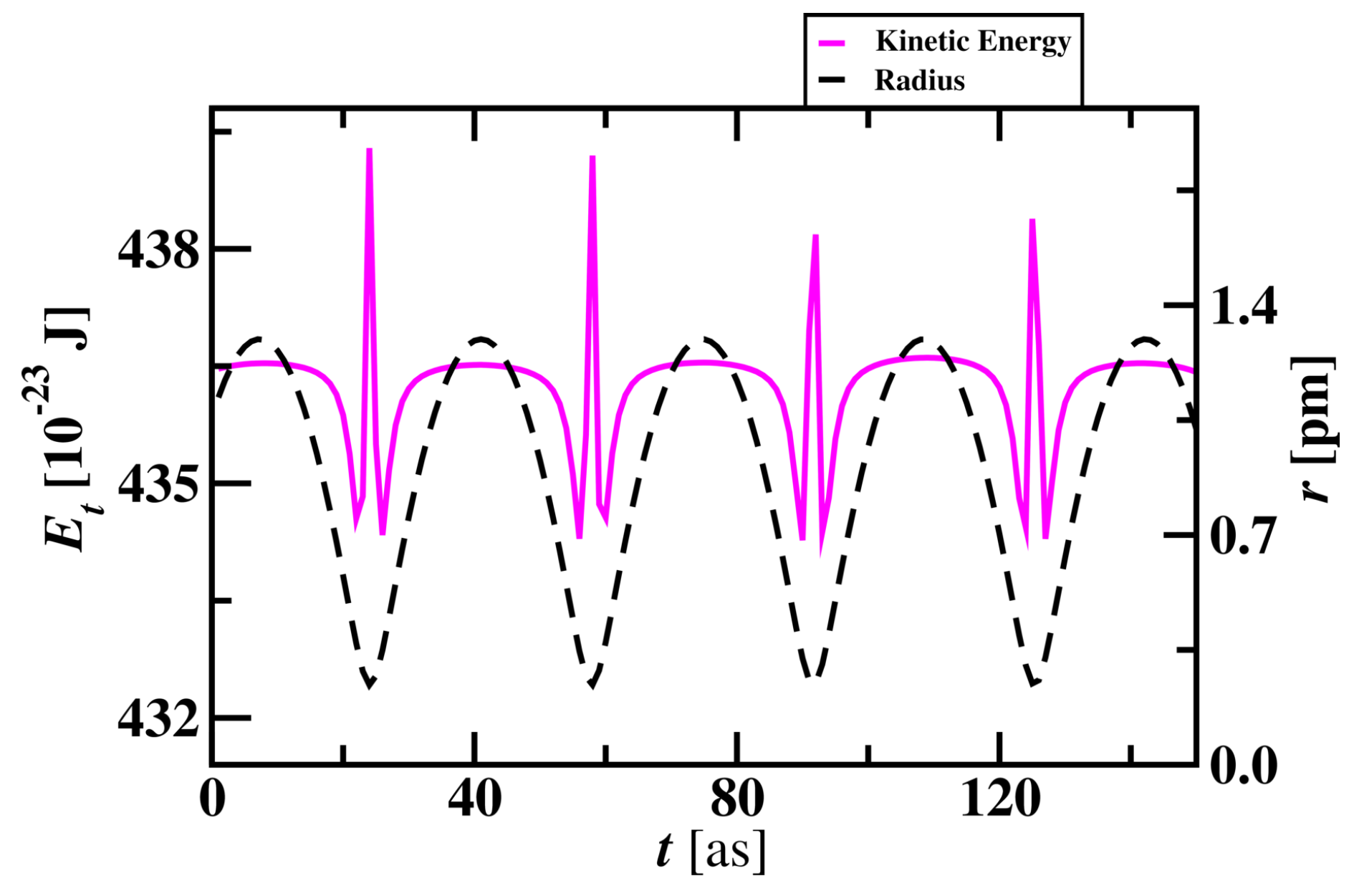
3.1.2. Randomness in Hydrogen Atom Simulation
3.1.3. Particle Nature of Carbon Atom Simulation
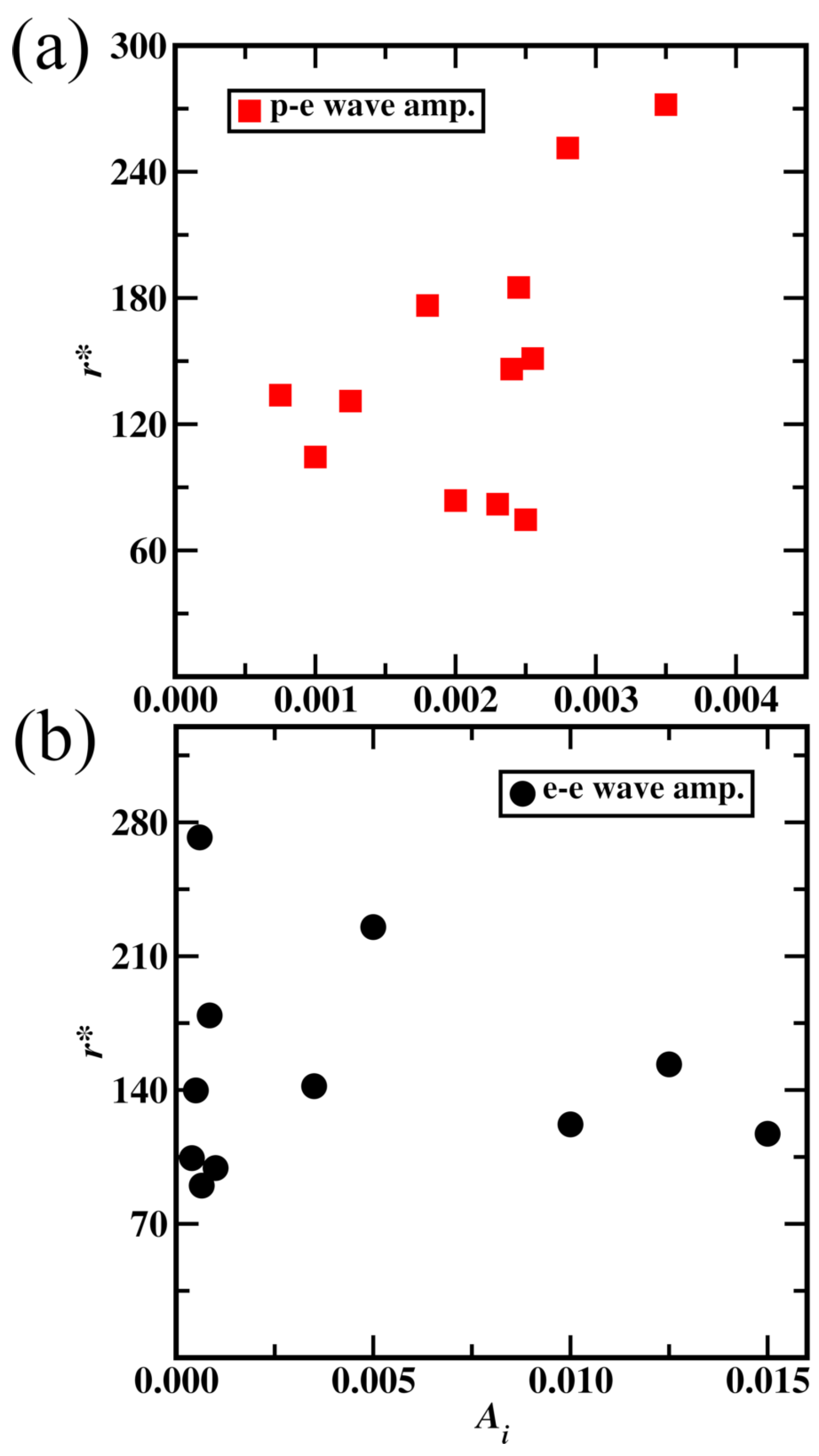
3.2. Wave Nature Subatomic Particle Dynamics Simulations
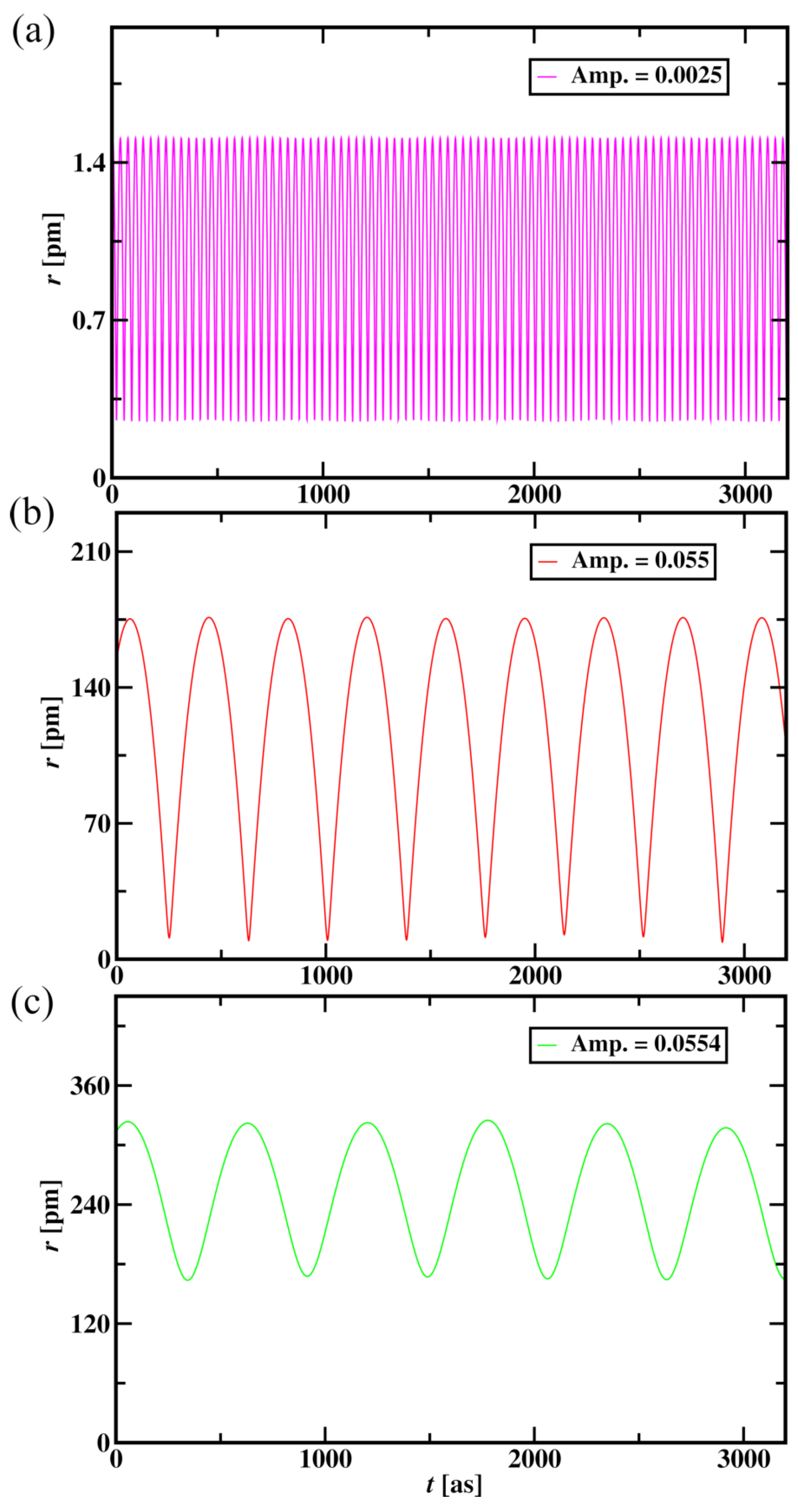
3.2.1. Wave Nature Simulation in Hydrogen Atom
3.2.2. Wave Nature of Carbon Atom Simulation
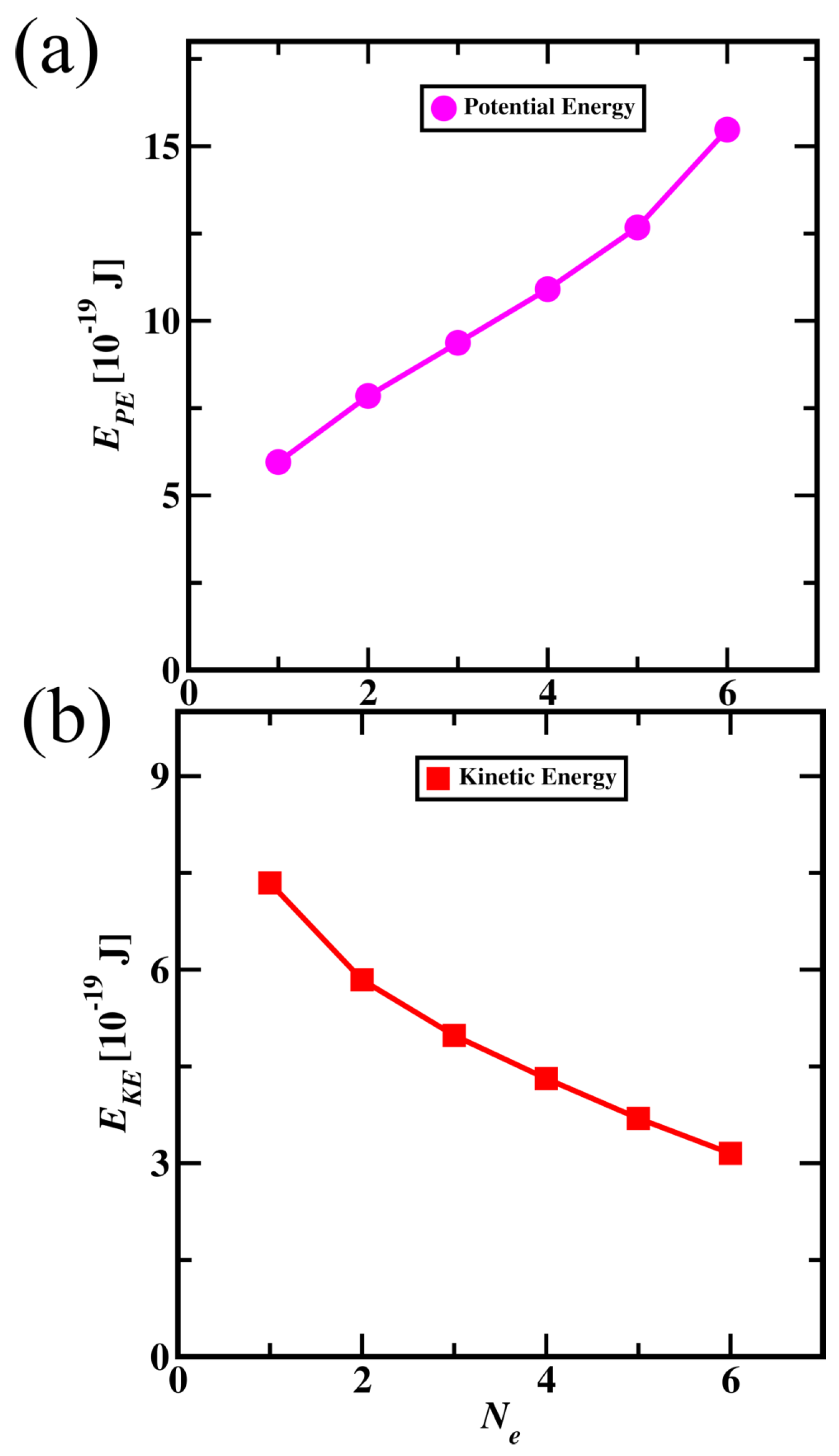
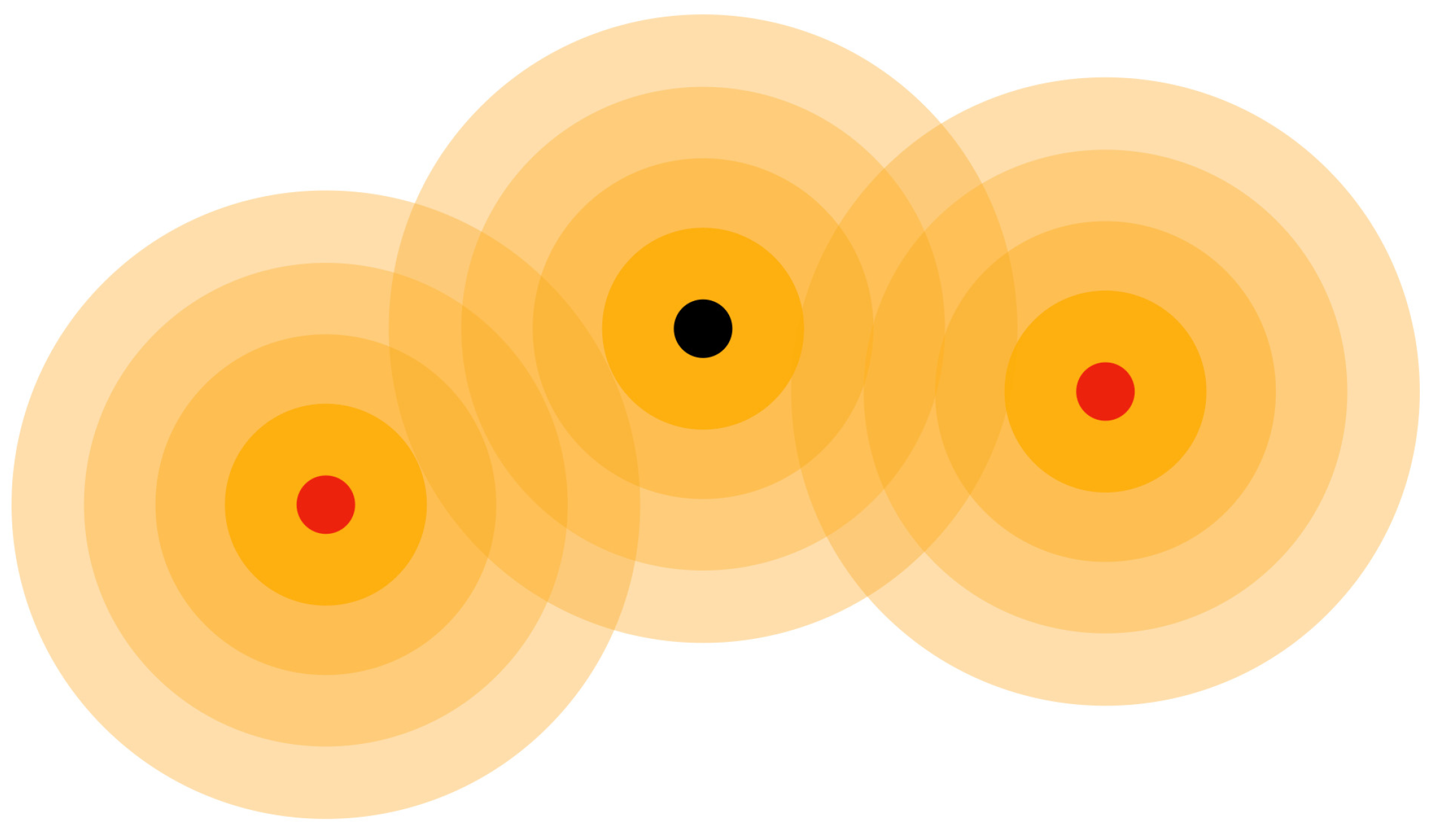
3.3. Subatomic Particle Dynamics Simulations for Molecules
3.4. Uranium Atom Simulation
4. Conclusion
Supplementary Materials
Acknowledgments
References
- Coulomb, C.A. Premier mémoire sur l’électricité et le magnétisme. Histoire de l’Academie royale des sciences 1785, 569. [Google Scholar]
- Kane, C.; Balents, L.; Fisher, M.P. Coulomb interactions and mesoscopic effects in carbon nanotubes. Physical review letters 1997, 79, 5086. [Google Scholar] [CrossRef]
- Cea, T.; Guinea, F. Coulomb interaction, phonons, and superconductivity in twisted bilayer graphene. Proceedings of the National Academy of Sciences 2021, 118, e2107874118. [Google Scholar] [CrossRef] [PubMed]
- Alder, B.J.; Wainwright, T.E. Studies in Molecular Dynamics. I. General Method. The Journal of Chemical Physics 1959, 31, 459–466. [Google Scholar] [CrossRef]
- Karplus, M.; Petsko, G.A. Molecular dynamics simulations in biology. Nature 1990, 347, 631–639. [Google Scholar] [CrossRef] [PubMed]
- Rapaport, D.C. The art of molecular dynamics simulation; Cambridge university press, 2004. [Google Scholar]
- Laio, A.; Parrinello, M. Escaping free-energy minima. Proceedings of the National Academy of Sciences 2002, 99, 12562–12566. [Google Scholar] [CrossRef] [PubMed]
- Corkum, P.B.; Krausz, F. Attosecond science. Nature Physics 2007, 3, 381–387. [Google Scholar] [CrossRef]
- Krausz, F.; Ivanov, M. Attosecond physics. Reviews of Modern Physics 2009, 81, 163–234. [Google Scholar] [CrossRef]
- Calegari, F.; Ayuso, D.; Trabattoni, A.; Belshaw, L.; De Camillis, S.; Anumula, S.; Frassetto, F.; Poletto, L.; Palacios, A.; Decleva, P.; others. Ultrafast electron dynamics in phenylalanine initiated by attosecond pulses. Science 2014, 346, 336–339. [CrossRef]
- Ciappina, M.F.; Pérez-Hernández, J.A.; Landsman, A.S.; Okell, W.A.; Zherebtsov, S.; Förg, B.; Schötz, J.; Seiffert, L.; Fennel, T.; Shaaran, T.; others. Attosecond physics at the nanoscale. Reports on Progress in Physics 2017, 80, 054401. [CrossRef]
- Smirnova, O.; Mairesse, Y.; Patchkovskii, S.; Dudovich, N.; Villeneuve, D.; Corkum, P.; Ivanov, M.Y. High harmonic interferometry of multi-electron dynamics in molecules. Nature 2009, 460, 972–977. [Google Scholar] [CrossRef] [PubMed]
- de Broglie, L. Recherches sur la théorie des quanta [Researches on the quantum theory]. Annales de Physique 1927, 3, 22–128. [Google Scholar]
- Bohm, D. A suggested interpretation of the quantum theory in terms of "hidden" variables. I and II. Physical Review 1952, 85, 166–179. [Google Scholar] [CrossRef]
- Valentini, A. Signal-locality, uncertainty, and the subquantum H-theorem. I. Physical Review A 2001, 63, 033605. [Google Scholar] [CrossRef]
- Norsen, T. The pilot-wave perspective on spin. American Journal of Physics 2014, 82, 337–348. [Google Scholar] [CrossRef]
- Einstein, A. Die Feldgleichungen der Gravitation. Sitzungsberichte der Preussischen Akademie der Wissenschaften zu Berlin 1915, pp. 844–847.
- Magueijo, J. Faster than the Speed of Light; Harvard University Press, 2003. [Google Scholar]
- Couder, Y.; Fort, E. Single-particle diffraction and interference at a macroscopic scale. Physical review letters 2006, 97, 154101. [Google Scholar] [CrossRef] [PubMed]
- Harris, D.M.; Moukhtar, J.; Fort, E.; Couder, Y.; Bush, J.W. Wavelike statistics from pilot-wave dynamics in a circular corral. Physical Review E 2013, 88, 011001. [Google Scholar] [CrossRef] [PubMed]
- Couder, Y.; Fort, E. Probabilities and trajectories in a classical wave-particle duality. Journal of Physics: Conference Series. IOP Publishing, 2012, Vol. 361, p. 012001. [CrossRef]
- Fort, E.; Eddi, A.; Boudaoud, A.; Moukhtar, J.; Couder, Y. Path-memory induced quantization of classical orbits. Proceedings of the National Academy of Sciences 2010, 107, 17515–17520. [Google Scholar] [CrossRef]
- Pucci, G.; Harris, D.M.; Faria, L.M.; Bush, J.W. Walking droplets interacting with single and double slits. Journal of Fluid Mechanics 2018, 835, 1136–1156. [Google Scholar] [CrossRef]
- Swope, W.C.; Andersen, H.C.; Berens, P.H.; Wilson, K.R. A computer simulation method for the calculation of equilibrium constants for the formation of physical clusters of molecules: Application to small water clusters. The Journal of chemical physics 1982, 76, 637–649. [Google Scholar] [CrossRef]
- Humphrey, W.; Dalke, A.; Schulten, K. VMD – Visual Molecular Dynamics. Journal of Molecular Graphics 1996, 14, 33–38. [Google Scholar] [CrossRef] [PubMed]

| (Joules) | (picometers) | |
|---|---|---|
| Parameterised based on Carbon atom | 7.837 * | 0.7777 |
| Probability of Randomly Placing Electron | Average Radius (pm) | Average Total Energy, J | Average Potential Energy, J | Average Kinetic Energy, J |
|---|---|---|---|---|
| 0 (Normal Hydrogen Atom) | 0.8875 | 436.97 | 314.58 | 121.4 |
| 0.000001 | 3.00 | 1453.32 | 974.18 | 479.14 |
| 0.00001 | 65.32 | 31323.31 | 21066.33 | 10256.99 |
| 0.0001 | 41.77 | 20374.48 | 13472.27 | 6902.20 |
| 0.001 | 14.00 | 6997.81 | 4520.54 | 2477.34 |
| 0.01 | 6.34 | 4418.42 | 2051.65 | 2366.77 |
| Average Velocity (m/s) | Average Velocity/Light Velocity | |
|---|---|---|
| Electron 1 | 835747.25 | 0.0028 |
| Electron 2 | 989235.65 | 0.0033 |
| Electron 3 | 977732.33 | 0.0033 |
| Electron 4 | 1213998.64 | 0.0040 |
| Electron 5 | 1058082.83 | 0.0035 |
| Electron 6 | 980335.11 | 0.0033 |
| Aether Wave Amplitude | Average Radius (pm) | Average Potential Energy ( J) | Average Kinetic Energy ( J) |
|---|---|---|---|
| 0 (Particle Nature) | 0.8875 | 314.58 | 121.4 |
| 0.0025 | 1.03 | 355.33 | 152.27 |
| 0.055 | 41.18 | 13288.73 | 8556.51 |
| 0.0554 | 239.94 | 77371.72 | 38671.99 |
| Carbon Atom Particle Nature | Carbon Atom Wave Nature | |
|---|---|---|
| Average Radius (pm) | 71.48 | 70.00 |
| Potential Energy | Kinetic Energy | Total Energy | ||||
|---|---|---|---|---|---|---|
| ( J) | ( J) | ( J) | ||||
| Particle Nature | Wave Nature | Particle Nature | Wave Nature | Particle Nature | Wave Nature | |
| Core Electron 1 | 59548.52 | 38095.44 | 73459.19 | 49875.38 | 133007.71 | 87970.82 |
| Core Electron 2 | 78471.76 | 52407.55 | 58419.72 | 43061.08 | 136891.49 | 95468.63 |
| Core Electron 3 | 93705.87 | 66293.05 | 49802.98 | 39838.54 | 143508.85 | 106131.59 |
| Core Electron 4 | 109062.78 | 83036.78 | 43116.09 | 36906.08 | 152178.86 | 119942.86 |
| Core Electron 5 | 126742.76 | 105527.48 | 36944.32 | 34075.01 | 163687.09 | 139602.49 |
| Valence Electron | 154750.70 | 150474.13 | 31521.12 | 29943.54 | 186271.82 | 180417.67 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the author. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).




