Submitted:
14 November 2023
Posted:
16 November 2023
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Results
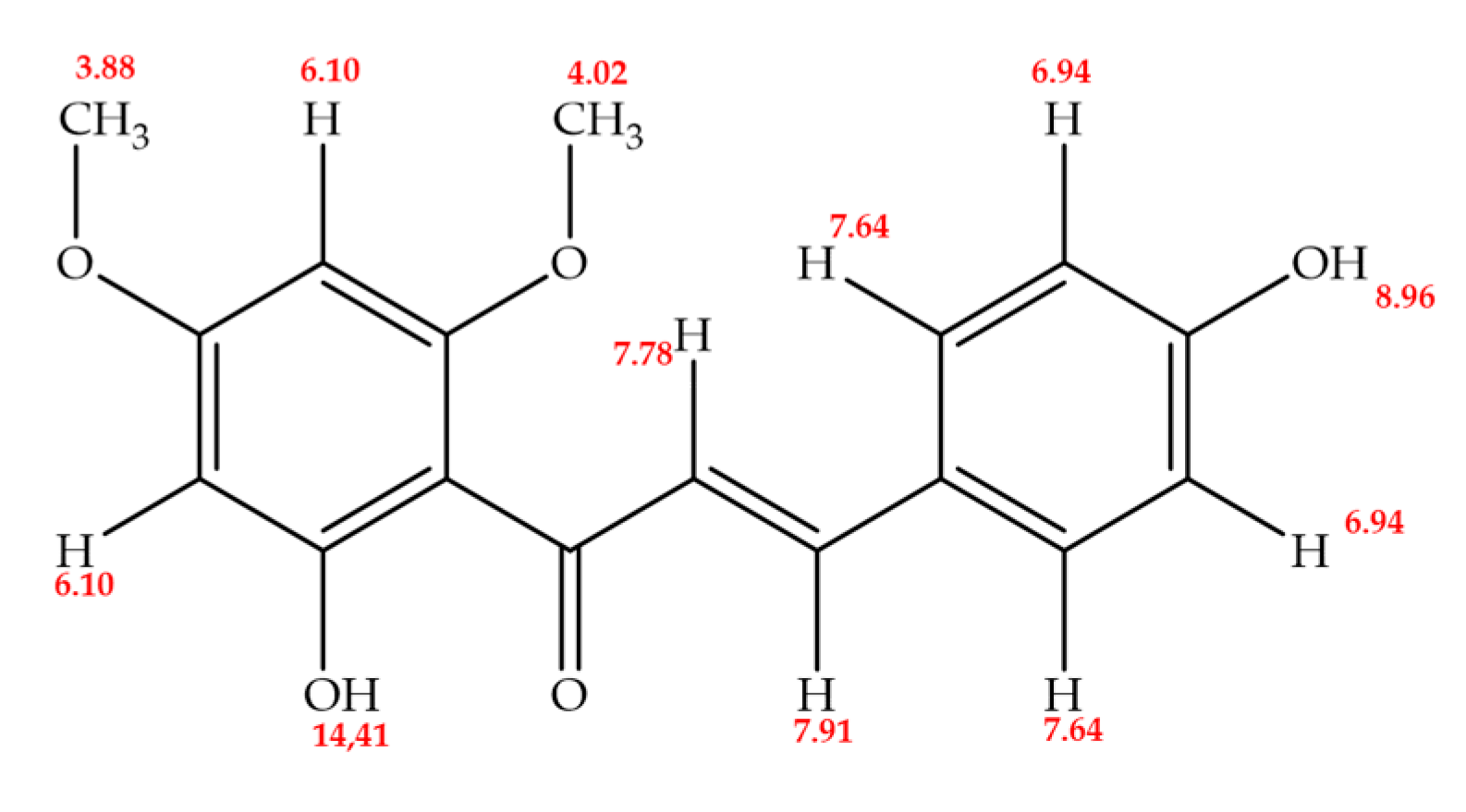
2.1. Structural analysis.
| 2’,4-dihydroxy-4´,6´-dimethoxy-chalcone (DDC) | ||||||
|---|---|---|---|---|---|---|
| Position | NMR | Displacement Analysis | ||||
| δH (J in Hz) | δC (ppm) | |||||
| C-1 C-2 C-3 C-4 C-5 C-6 C-α C-β C-β´ C-1´ C-2´ C-3´ C-4´ C-4´-OCH3 C-5´ C-6´ C-6´-OCH3 |
- 7.64 (2H, dd, J= 2.4 Hz) 6.94 (2H, dd, J= 2.4 Hz) - 6.94 (2H, dd, J= 2.4 Hz) 7.64 (2H, dd, J= 2.4 Hz) 7.78 (1H, d, J = 15.5 Hz) 7.91 (1H, d, J = 15.5 Hz) - - - 6.10 (2H, dd, J= 2.4 Hz) - 3.88 (3H, s) 6.10 (2H, dd, J= 2.4 Hz) - 4.02 (3H, s) |
127.1 130.5 115.93 159.83 115.93 130.5 124.15 142.84 192.47 105.92 166.38 93.77 168.26 55.17 90.87 162.79 55.60 |
Reactive | Band II (nm) | Band I (nm) |
Displacement (nm) |
| MeOH | 240 | 367 | - | |||
| MeOH+MeONa | 245 | 402 | B II:5, B I:35 | |||
| MeOH+AcONa | 245 | 372 | B II:5, B I:5. | |||
| MeOH+AcONa+H3BO3 | 240 | 372 | B I:5 | |||
| MeOH+AlCl3 | 240 | 383 | B I: 16 | |||
| MeOH+AlCl3+HCl | 240 | 476 | B I:9. | |||
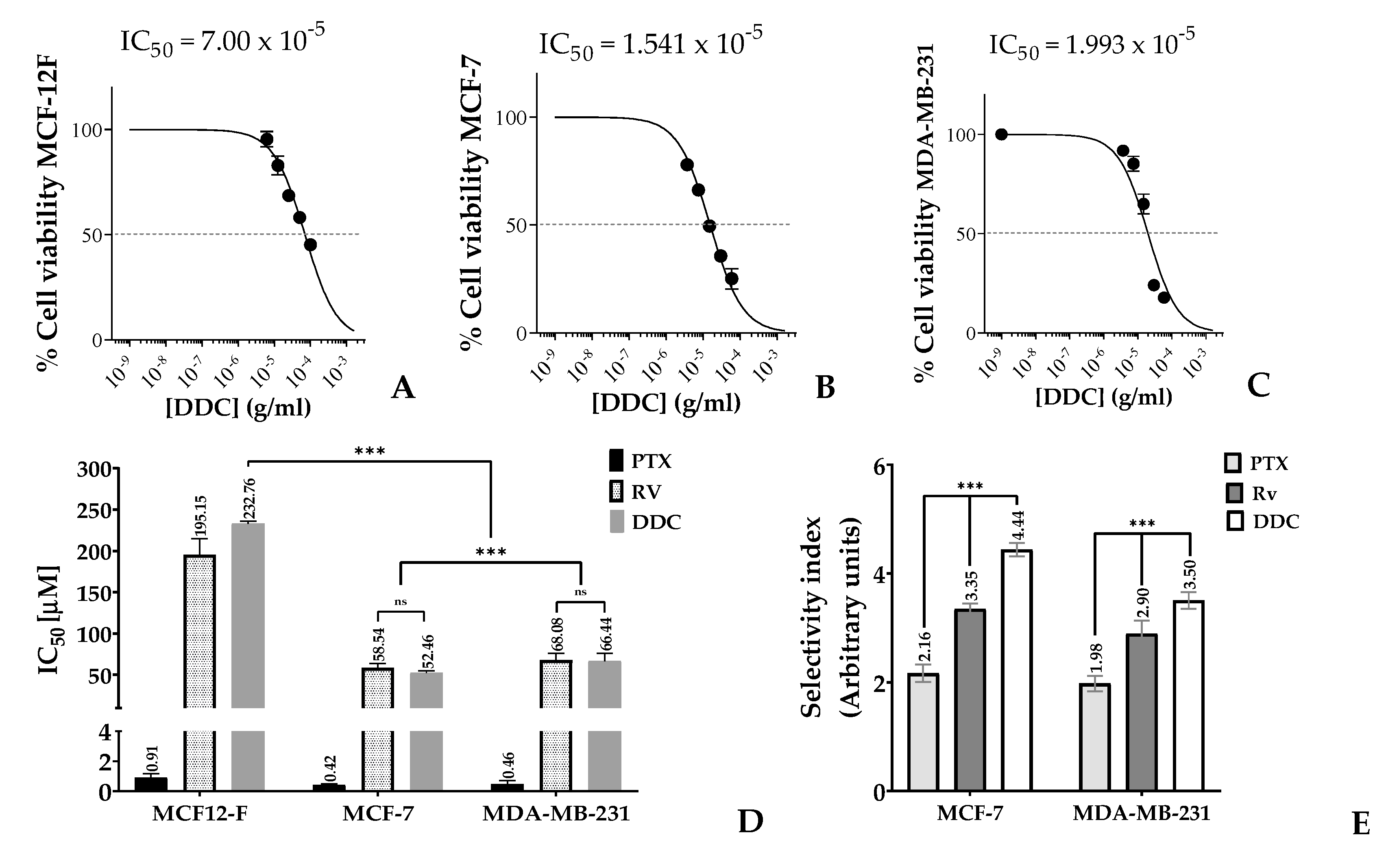
2.2. 2',4-dihydroxy-4',6'-dimethoxy chalcone induces selective cytotoxicity in BC cells
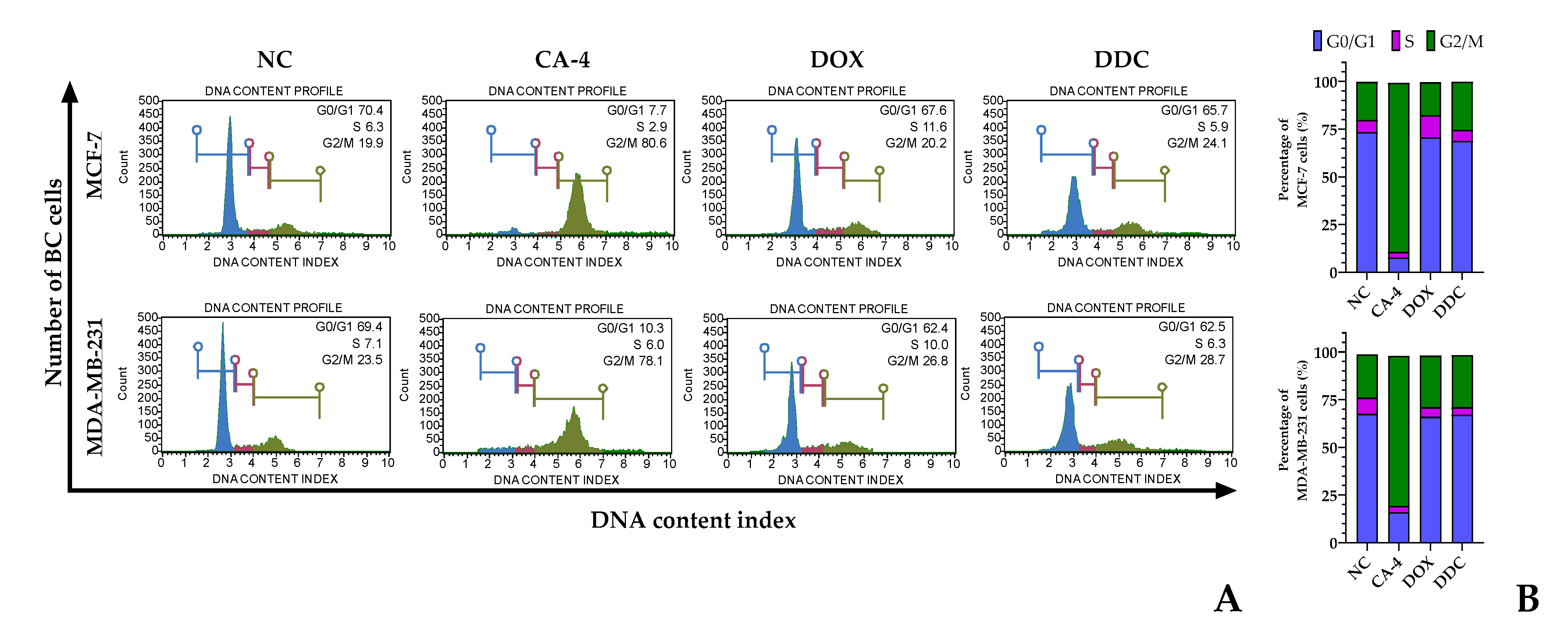
2.3. Cell cycle progression in response to the natural chalcone
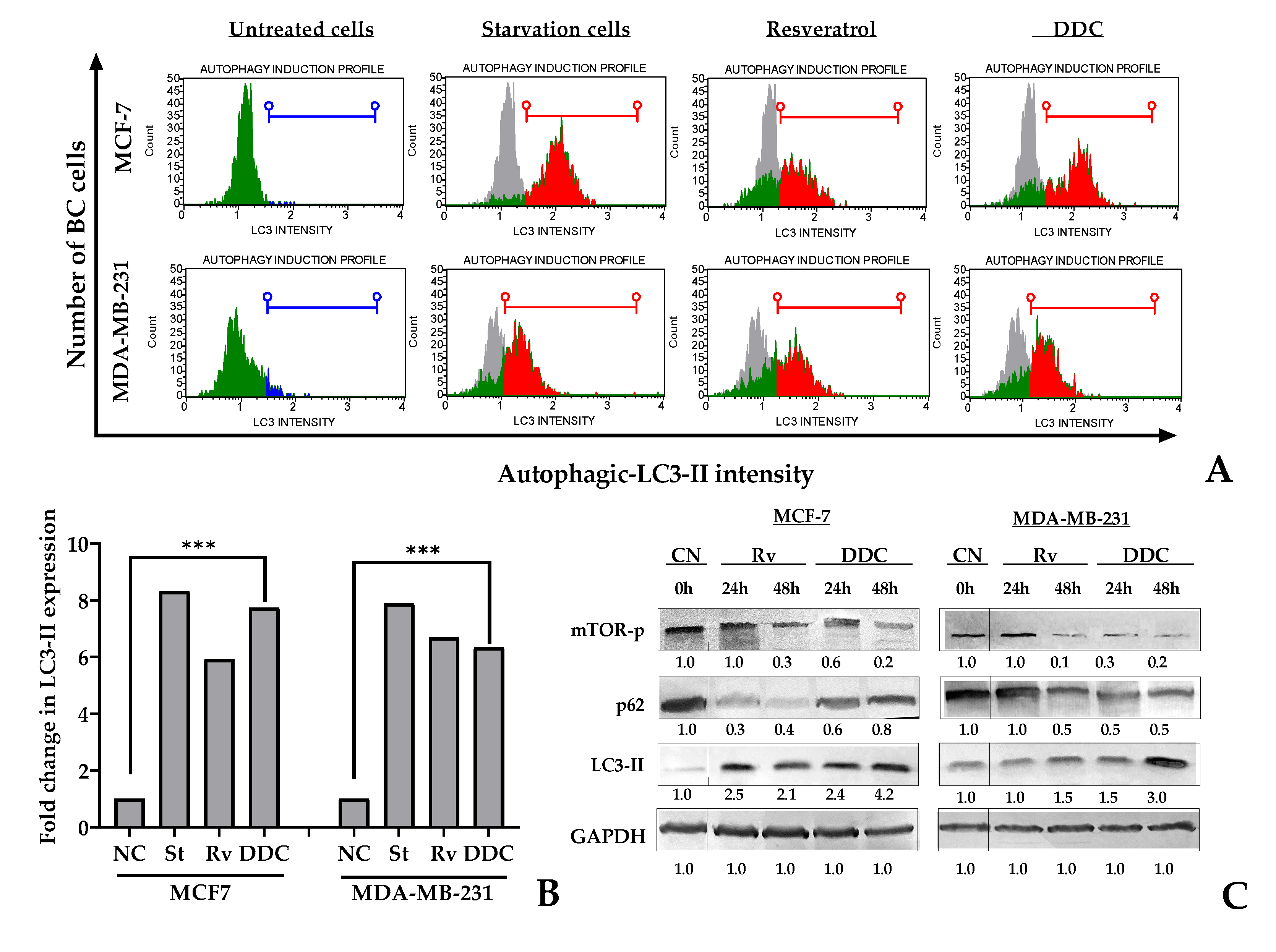
2.4. Chalcone targeting autophagy in BC cells
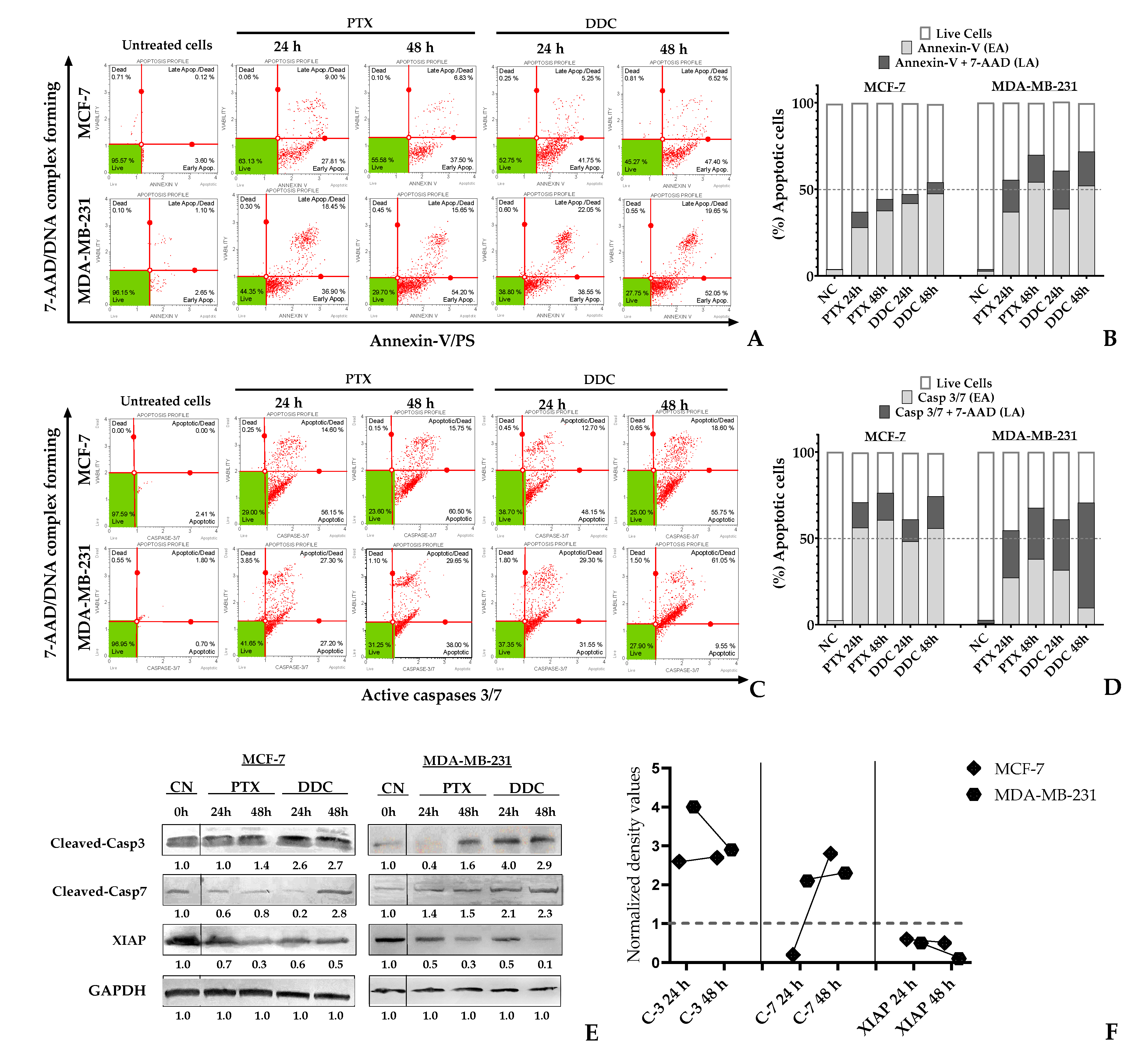
2.5. BC genotype-dependent variations in apoptosis triggering by DDC
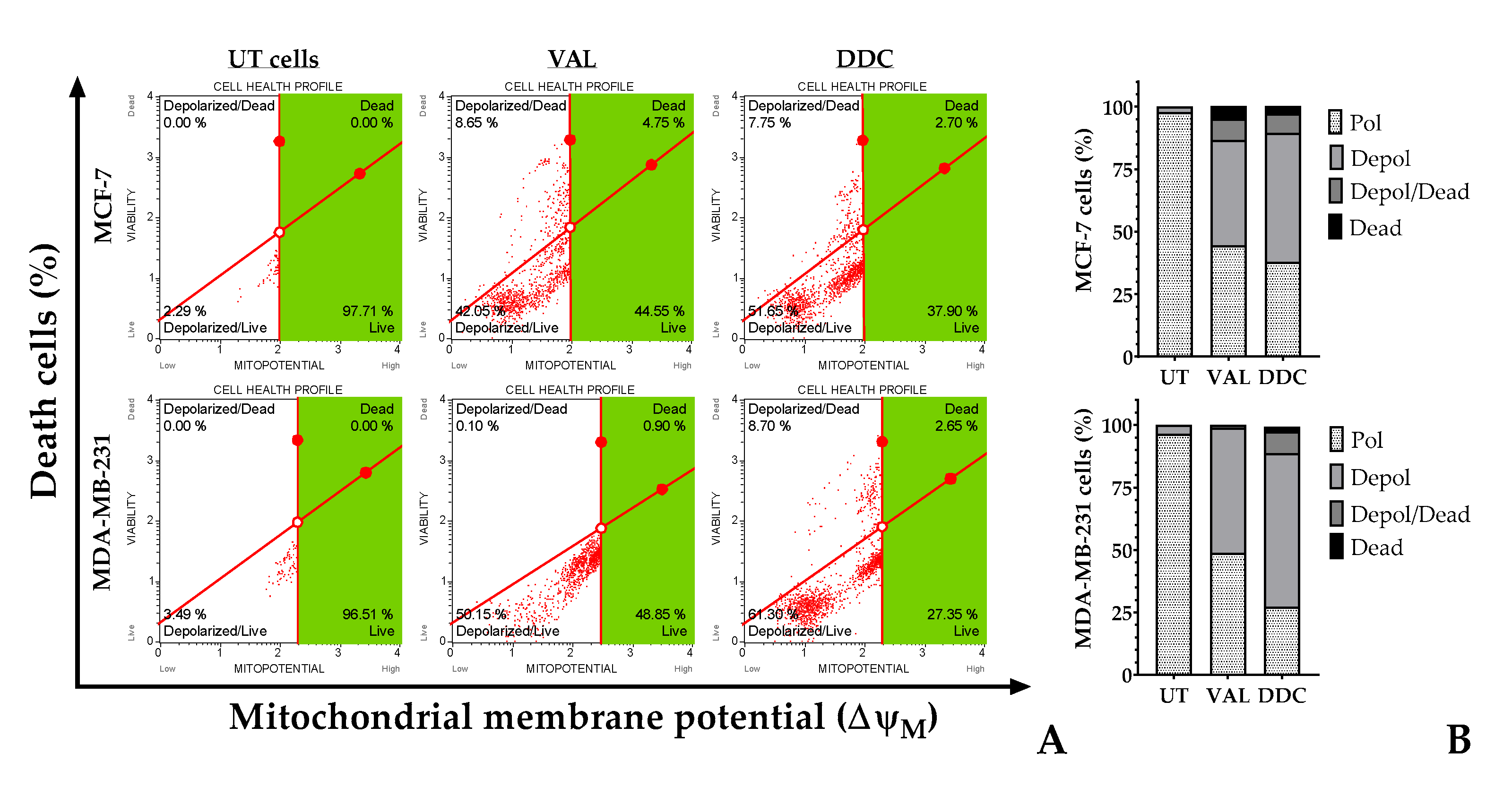
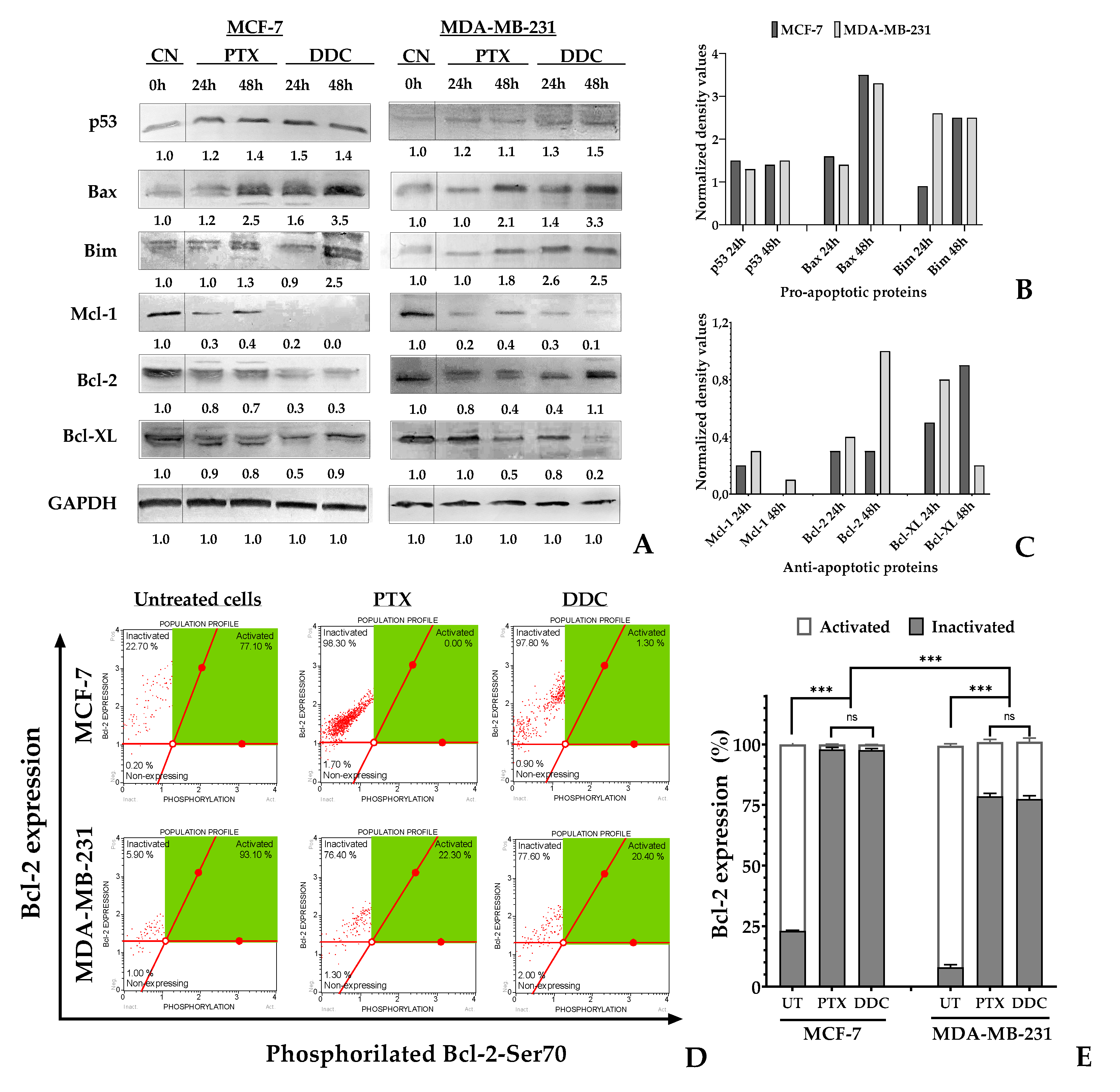
2.6. TNBC and Luminal A BC cells differ in the pathway leading to intrinsic apoptosis in response to DDC.
2.7. In silico analysis
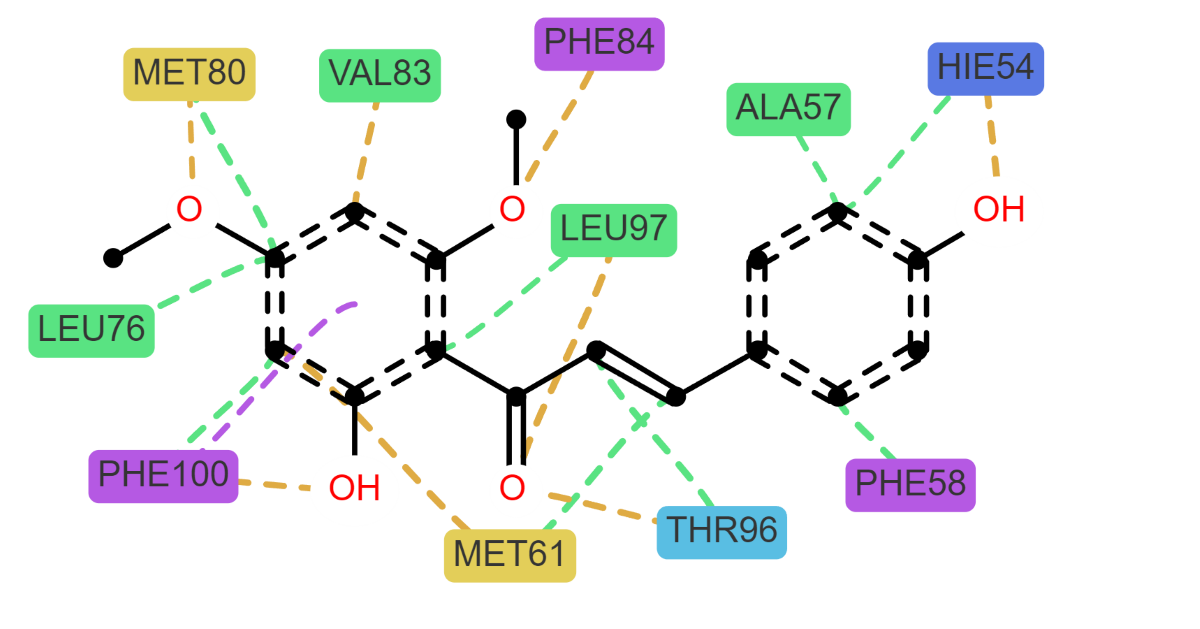
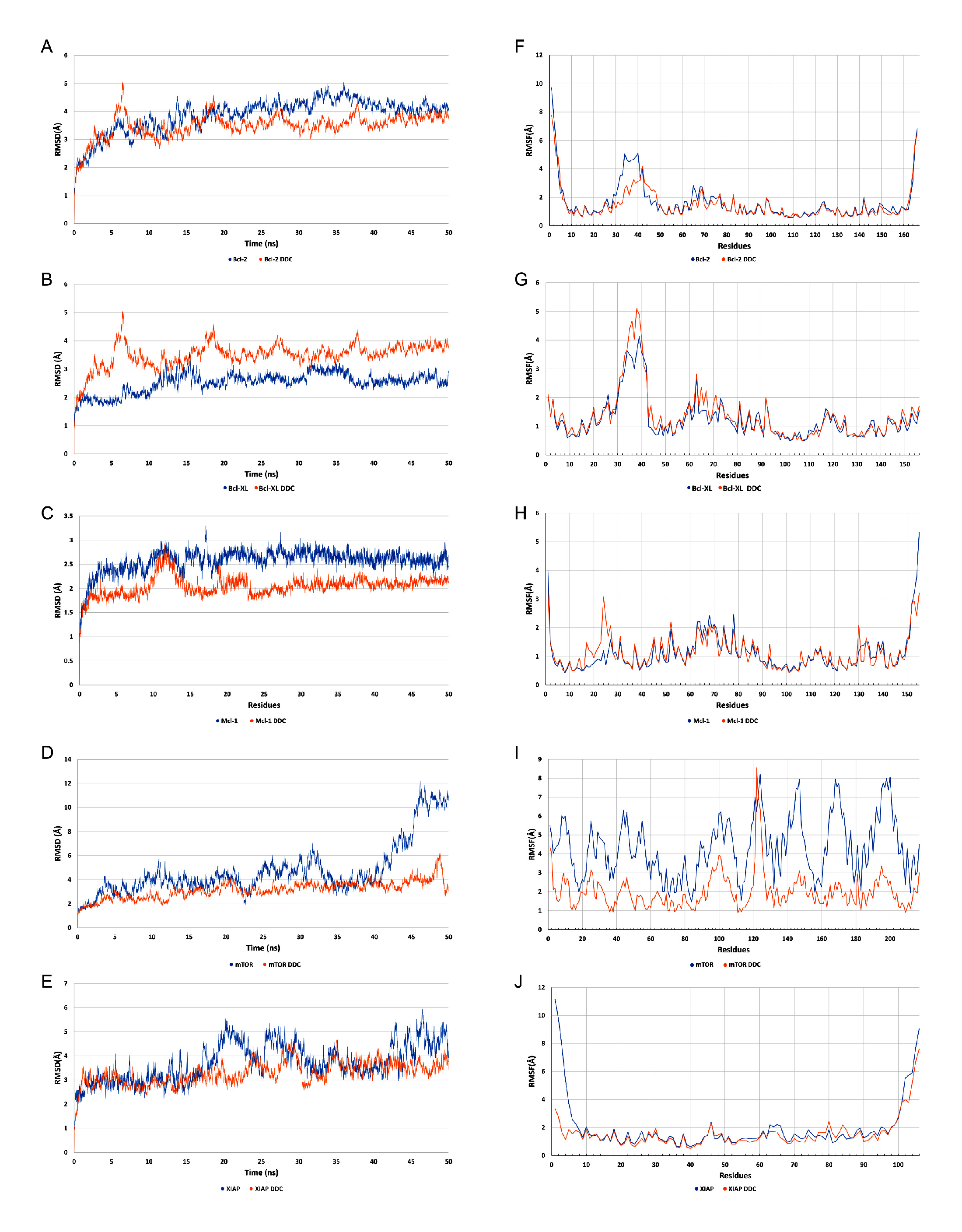
2.7.1. Docking and molecular dynamics (MD) simulation between DDC and pro-survival proteins.
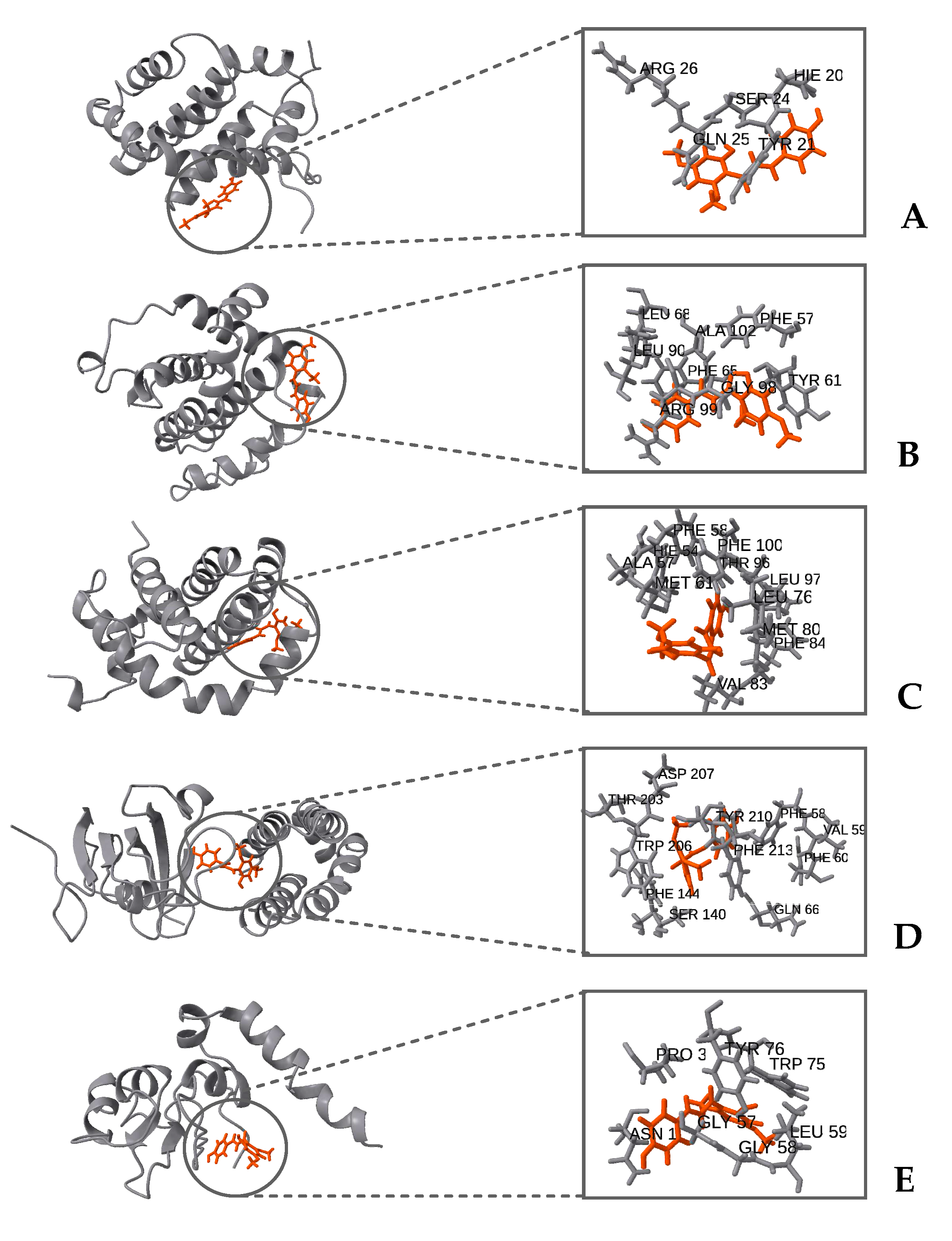
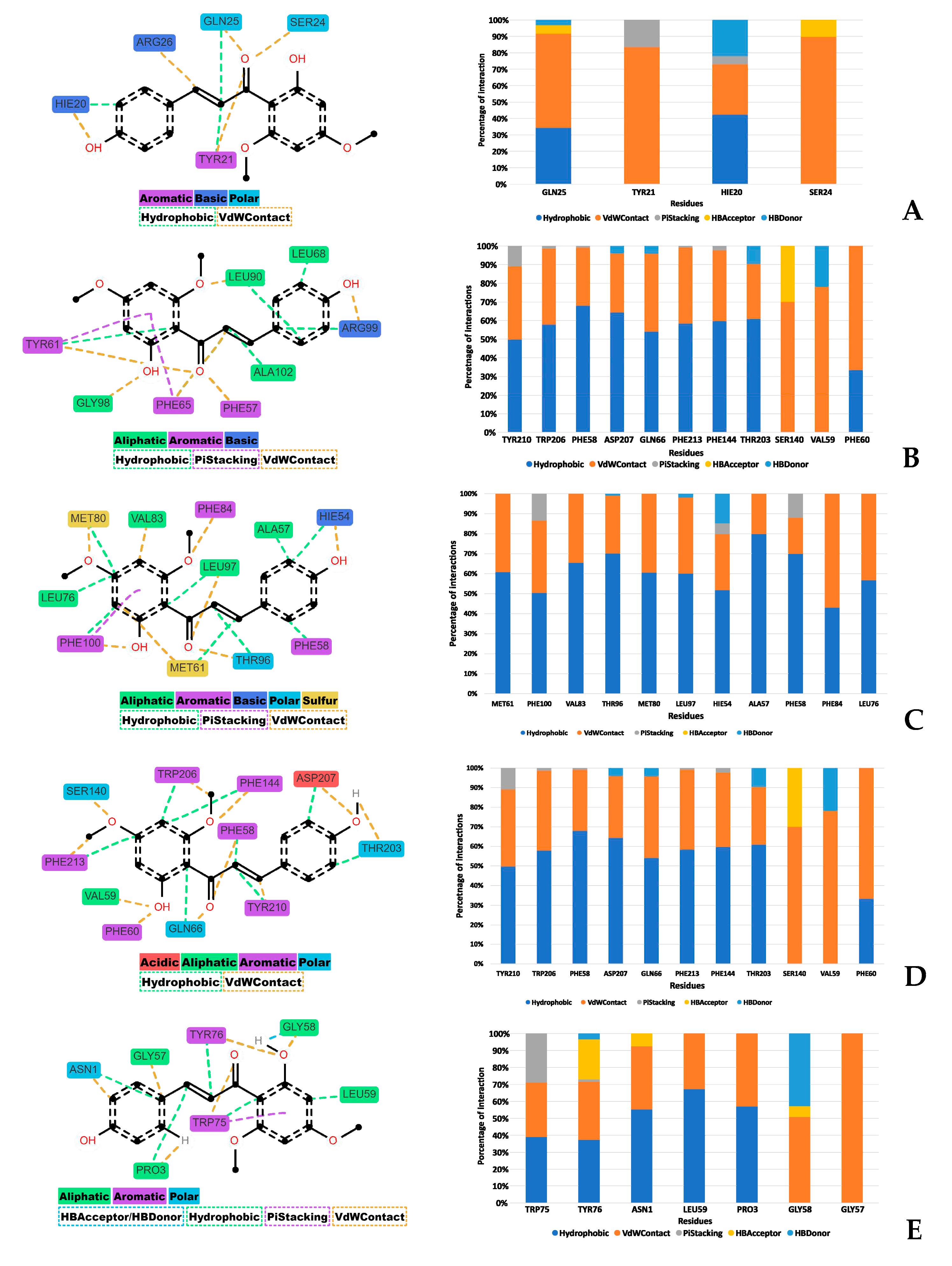
2.7.2. Interaction between DDC and pro-survival proteins analysis
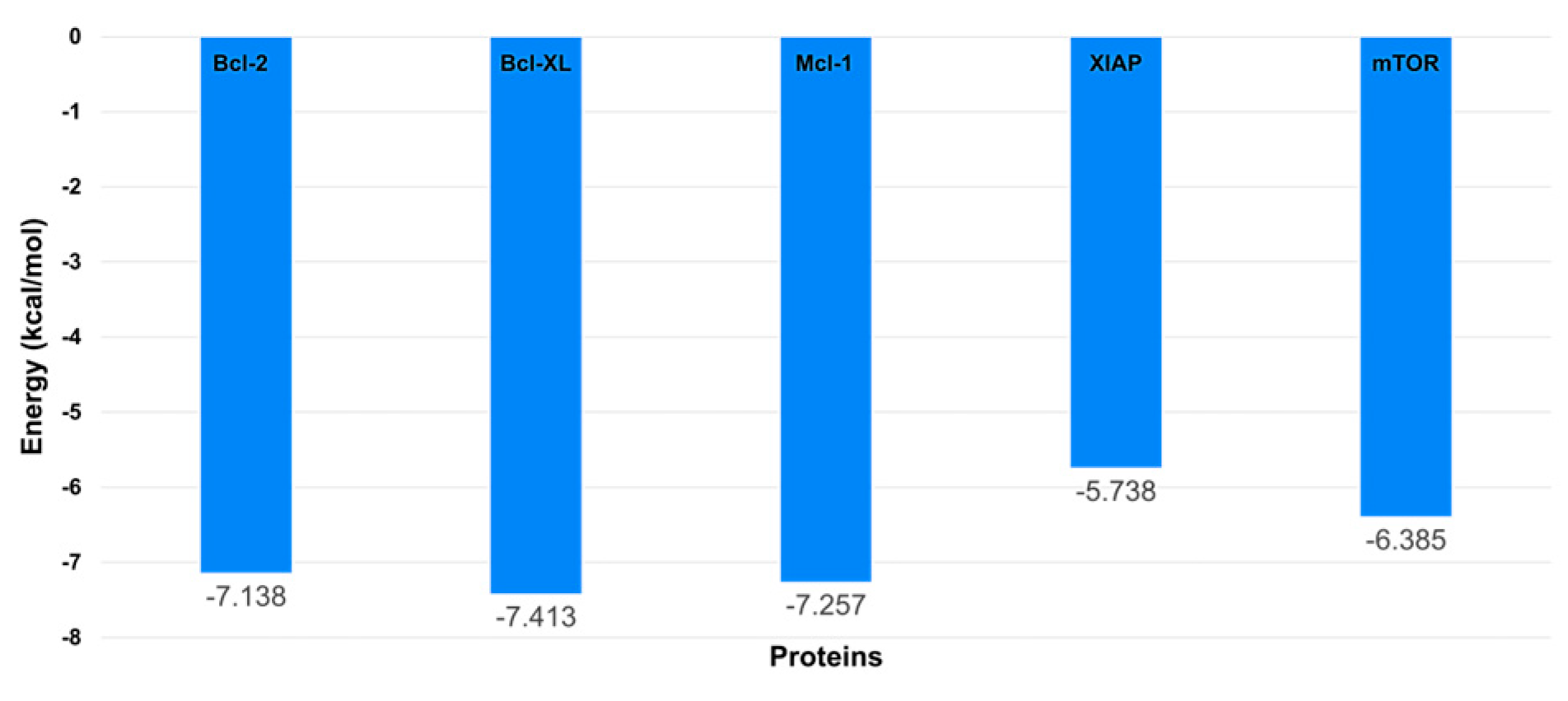
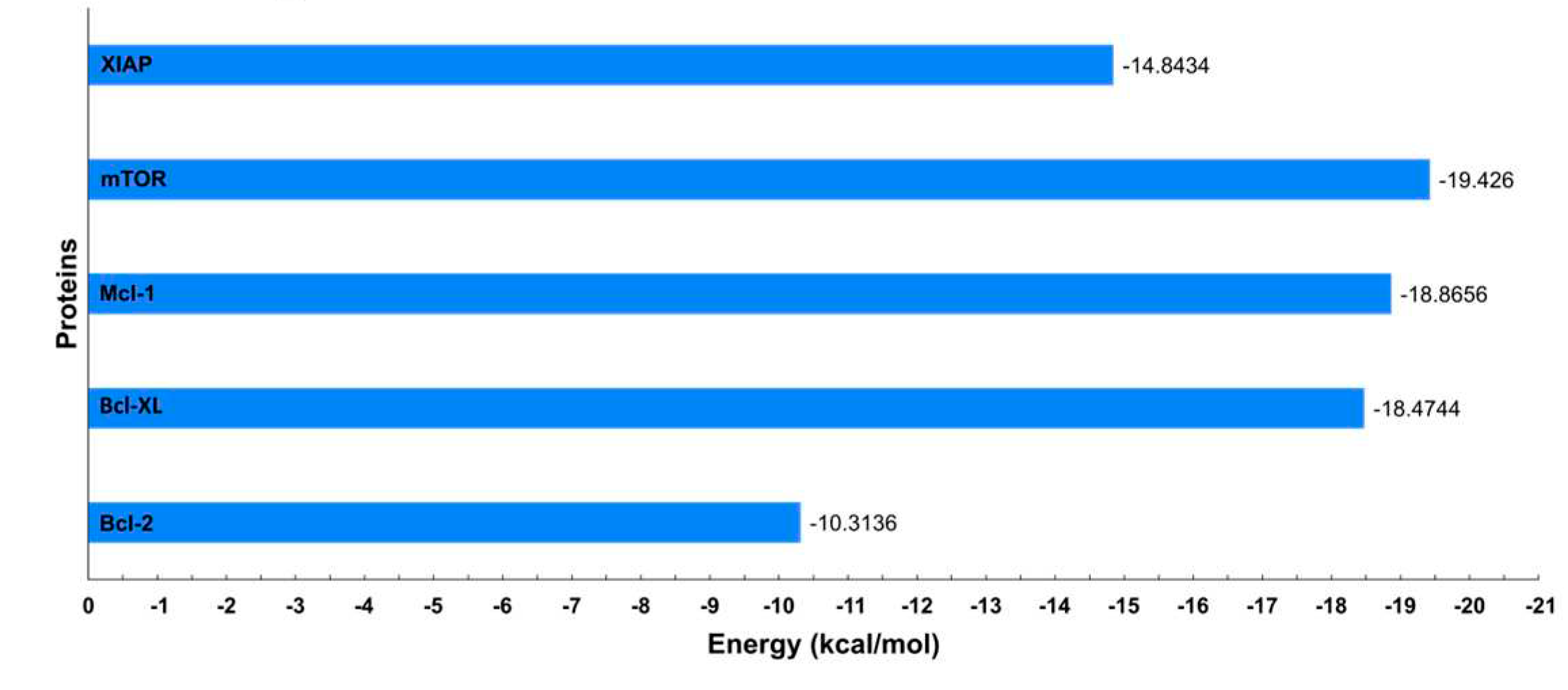
2.7.3. Binding free energy analysis via MM-PBSA
3. Discussion
4. Materials and methods
4.1. Chalcone extraction and isolation.
4.2. Structural identification
4.3. Breast cell lines and culture conditions
4.4. Cytotoxic activity and selectivity of 2',4-dihydroxy-4',6'-dimethoxychalcone
4.5. Effects of 2',4-dihydroxy-4',6'-dimethoxychalcone on cell cycle progression in BC cells
4.6. Susceptibility of BC cells to autophagy induced by the natural chalcone
4.6.1. Detection of LC3-II by flow cytometry assay
4.6.2. Western blot analysis
4.7. Apoptosis in response to 2',4-dihydroxy-4',6'-dimethoxychalcone
4.7.1. Apoptosis
4.7.2. Western Blotting.
4.8. Analysis of intrinsic pathway of apoptosis in TNBC cells
4.8.1. Changes in the outer mitochondrial membrane potential (∆ψm)
4.8.2. Pro- and anti-apoptotic proteins analysis by Western blot
4.8.3. Phospho-Bcl-2 protein detection
4.9. Statistical analysis for biological test
4.10. Molecular Docking and Molecular dynamics
4.10.1. Molecular Dynamics simulation preparations
4.10.2. Molecular dynamics configuration
4.10.3. MMPBSA
4.10.4. RMSD, RMSF and Rg
5. Conclusion
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Sample Availability
References
- Maqbool, M.; Bekele, F.; Fekadu, G. Treatment Strategies Against Triple-Negative Breast Cancer: An Updated Review. 2022. [CrossRef]
- Tran, B., Bedard, P.L. Luminal-B breast cancer and novel therapeutic targets. Breast Cancer Res 2011, 13, 221. [CrossRef]
- Panche, A.N.; Diwan, A.D.; Chandra, S.R. Flavonoids: An Overview. J Nutr Sci 2016, 5. [CrossRef]
- Falcone Ferreyra, M.L.; Rius, S.P.; Casati, P. Flavonoids: Biosynthesis, Biological Functions, and Biotechnological Applications. Front Plant Sci 2012, 3, 34352. [Google Scholar] [CrossRef] [PubMed]
- Yadav, V.R.; Prasad, S.; Sung, B.; Aggarwal, B.B. The Role of Chalcones in Suppression of NF-ΚB-Mediated Inflammation and Cancer. Int Immunopharmacol 2011, 11, 295–309. [Google Scholar] [CrossRef] [PubMed]
- Zhang, E.-H.; Wang, R.-F.; Guo, S.-Z.; Liu, B. An Update on Antitumor Activity of Naturally Occurring Chalcones. Evidence-Based Complementary and Alternative Medicine 2013, 2013, 22. [Google Scholar] [CrossRef]
- Mokbel, K. The Evolving Role of Aromatase Inhibitors in Breast Cancer. Int J Clin Oncol 2002, 7, 279–283. [Google Scholar] [CrossRef] [PubMed]
- Maggiolini, M.; Statti, G.; Vivacqua, A.; Gabriele, S.; Rago, V.; Loizzo, M.; Menichini, F.; Amdò, S. Estrogenic and Antiproliferative Activities of Isoliquiritigenin in MCF7 Breast Cancer Cells. Journal of Steroid Biochemistry and Molecular Biology 2002, 82, 315–322. [Google Scholar] [CrossRef]
- Wang, K.L.; Yu, Y.C.; Hsia, S.M. Perspectives on the Role of Isoliquiritigenin in Cancer. Cancers (Basel) 2021, 13, 1–37. [Google Scholar] [CrossRef] [PubMed]
- Girisa, S.; Saikia, Q.; Bordoloi, D.; Banik, K.; Monisha, J.; Daimary, U.D.; Verma, E.; Ahn, K.S.; Kunnumakkara, A.B. Xanthohumol from Hop: Hope for Cancer Prevention and Treatment. IUBMB Life 2021, 73, 1016–1044. [Google Scholar] [CrossRef] [PubMed]
- Yong, W.K.; Abd Malek, S.N. Xanthohumol Induces Growth Inhibition and Apoptosis in ca Ski Human Cervical Cancer Cells. Evid Based Complement Alternat Med 2015, 2015. [Google Scholar] [CrossRef]
- Shi, J.; Chen, Y.; Chen, W.; Tang, C.; Zhang, H.; Chen, Y.; Yang, X.; Xu, Z.; Wei, J.; Chen, J. Isobavachalcone Sensitizes Cells to E2-Induced Paclitaxel Resistance by down-Regulating CD44 Expression in ER+ Breast Cancer Cells. J Cell Mol Med 2018, 22, 5220–5230. [Google Scholar] [CrossRef] [PubMed]
- Jeong, Y.J.; Kang, K.J. Effect of Angelica Keiskei Extract on Apoptosis of MDA-MB-231 Human Breast Cancer Cells. Journal of the Korean Society of Food Science and Nutrition 2011, 40, 1654–1661. [Google Scholar] [CrossRef]
- Abu, N.; Ho, W.Y.; Yeap, S.K.; Akhtar, M.N.; Abdullah, M.P.; Omar, A.R.; Alitheen, N.B. The Flavokawains: Uprising Medicinal Chalcones. Cancer Cell Int 2013, 13. [Google Scholar] [CrossRef] [PubMed]
- Abu, N.; Akhtar, M.N.; Yeap, S.K.; Lim, K.L.; Ho, W.Y.; Zulfadli, A.J.; Omar, A.R.; Sulaiman, M.R.; Abdullah, M.P.; Alitheen, N.B. Flavokawain A Induces Apoptosis in MCF-7 and MDA-MB231 and Inhibits the Metastatic Process in Vitro. PLoS One 2014, 9. [Google Scholar] [CrossRef] [PubMed]
- Abu, N.; Akhtar, M.N.; Yeap, S.K.; Lim, K.L.; Ho, W.Y.; Abdullah, M.P.; Ho, C.L.; Omar, A.R.; Ismail, J.; Alitheen, N.B. Flavokawain B Induced Cytotoxicity in Two Breast Cancer Cell Lines, MCF-7 and MDA-MB231 and Inhibited the Metastatic Potential of MDA-MB231 via the Regulation of Several Tyrosine Kinases In Vitro. BMC Complement Altern Med 2016, 16. [Google Scholar] [CrossRef]
- Lin, X.; Xing, S.; Chen, K.; Yang, H.; Hu, X. Flavokavain C Suppresses Breast Cancer Cell Viability and Induces Cell Apoptosis by Triggering DNA Damage. Biol Pharm Bull 2023, 46, 684–692. [Google Scholar] [CrossRef] [PubMed]
- Yeap, S.K.; Abu, N.; Akthar, N.; Ho, W.Y.; Ky, H.; Tan, S.W.; Alitheen, N.B.; Kamarul, T. Gene Expression Analysis Reveals the Concurrent Activation of Proapoptotic and Antioxidant-Defensive Mechanisms in Flavokawain B-Treated Cervical Cancer HeLa Cells. Integr Cancer Ther 2017, 16, 373–384. [Google Scholar] [CrossRef] [PubMed]
- Phang, C.W.; Karsani, S.A.; Sethi, G.; Malek, S.N.A. Flavokawain C Inhibits Cell Cycle and Promotes Apoptosis, Associated with Endoplasmic Reticulum Stress and Regulation of MAPKs and Akt Signaling Pathways in HCT 116 Human Colon Carcinoma Cells. PLoS One 2016, 11. [Google Scholar] [CrossRef] [PubMed]
- The New York Botanical Garden. Available online: https://www.nybg.org/ (accessed on 11-05-2023).
- Taleb-Contini, S.H.; Schorr, K.; Da Costa, F.B.; De Oliveira, D.C.R. Detection of Flavonoids in Glandular Trichomes of Chromolaena Species (Eupatorieae, Asteraceae) by Reversed-Phase High-Performance Liquid Chromatography. Revista Brasileira de Ciências Farmacêuticas 2007, 43, 315–321. [Google Scholar] [CrossRef]
- Herrera-Calderon, O.; Arroyo-Acevedo, J.L.; Rojas-Armas, J.; Chumpitaz-Cerrate, V.; Figueroa-Salvador, L.; Enciso-Roca, E.; Tinco-Jayo, J.A. Phytochemical Screening, Total Phenolic Content, Antioxidant and Cytotoxic Activity of Chromolaena Laevigata on Human Tumor Cell Lines. Annu Res Rev Biol 2017, 21. [Google Scholar] [CrossRef]
- Ojo, O.; Mphahlele, M.P.; Oladeji, O.S.; Mmutlane, E.M.; Ndinteh, D.T. From Wandering Weeds to Pharmacy: An Insight into Traditional Uses, Phytochemicals and Pharmacology of Genus Chromolaena (Asteraceae). J Ethnopharmacol 2022, 291. [Google Scholar] [CrossRef] [PubMed]
- Pérez, C. Química del género Chromolaena (Compositae). 1994, 12, 141-145.
- Dhar, R.; Kimseng, R.; Chokchaisiri, R.; Hiransai, P.; Utaipan, T.; Suksamrarn, A.; Chunglok, W. 2′,4-Dihydroxy-3′,4′,6′-Trimethoxychalcone from Chromolaena Odorata Possesses Anti-Inflammatory Effects via Inhibition of NF-ΚB and P38 MAPK in Lipopolysaccharide-Activated RAW 264.7. Macrophages. 2017, 40, 43–51. [Google Scholar] [CrossRef] [PubMed]
- Barua, R.N.; Sharma, R.P.; Thyagarajan, G.; Hertz, W. Flavonoids of Chromolaena Odorata. Phytochemistry 1978, 17, 1807–1808. [Google Scholar] [CrossRef]
- Zhang, M.L.; Irwin, D.; Li, X.N.; Sauriol, F.; Shi, X.W.; Wang, Y.F.; Huo, C.H.; Li, L.G.; Gu, Y.C.; Shi, Q.W. PPARγ Agonist from Chromolaena Odorata. J Nat Prod 2012, 75, 2076–2081. [Google Scholar] [CrossRef] [PubMed]
- Mendez-Callejas, G.; Torrenegra, R.; Muñoz, D.; Celis, C.; Roso, M.; Garzon, J.; Beltran, F.; Cardenas, A. A New Flavanone from Chromolaena Tacotana (Klatt) R. M. King and H. Rob, Promotes Apoptosis in Human Breast Cancer Cells by Downregulating Antiapoptotic Proteins. Molecules 2023, 28, 58. [Google Scholar] [CrossRef] [PubMed]
- Rodriguez, J.; Gómez, L.M.; Gutierrez, A.C.; Mendez-Callejas, G.; Reyes, A.I.; Tellez, L.C.; Oscar, A.; Rodriguez, E.; Rubén, G.; Torrenegra, D. Chromolaena Tacotana (Klatt) R. M. King and H. Rob. Source of Flavonoids with Antiproliferative and Antioxidant Activity. Indian J Sci Technol 2018, 11. [Google Scholar] [CrossRef]
- Bolomsky, A.; Vogler, M.; Köse, M.C.; Heckman, C.A.; Ehx, G.; Ludwig, H.; Caers, J. MCL-1 Inhibitors, Fast-Lane Development of a New Class of Anti-Cancer Agents. Journal of Hematology & Oncology 2020 13:1 2020, 13, 1–19. [Google Scholar] [CrossRef]
- Pelz, N.F.; Bian, Z.; Zhao, B.; Shaw, S.; Tarr, J.C.; Belmar, J.; Gregg, C.; Camper, D.M. V.; Goodwin, C.M.; Arnold, A.L.; et al. Discovery of 2-Indole-Acylsulfonamide Myeloid Cell Leukemia 1 (Mcl-1) Inhibitors Using Fragment-Based Methods. J Med Chem 2016, 59, 2054–2066. [Google Scholar] [CrossRef]
- Eberhardt, J.; Santos-Martins, D.; Tillack, A.F.; Forli, S. AutoDock Vina 1.2.0: New Docking Methods, Expanded Force Field, and Python Bindings. J Chem Inf Model 2021, 61, 3891–3898. [Google Scholar] [CrossRef] [PubMed]
- Trott, O.; Olson, A.J. AutoDock Vina: Improving the Speed and Accuracy of Docking with a New Scoring Function, Efficient Optimization and Multithreading. J Comput Chem 2010, 31, 455. [Google Scholar] [CrossRef] [PubMed]
- Bouysset, C.; Fiorucci, S. ProLIF: A Library to Encode Molecular Interactions as Fingerprints. J Cheminform 2021, 13, 1–9. [Google Scholar] [CrossRef]
- Ferenczy, G.G.; Kellermayer, M. Contribution of Hydrophobic Interactions to Protein Mechanical Stability. Comput Struct Biotechnol J 2022, 20, 1946. [Google Scholar] [CrossRef] [PubMed]
- Varma, A.K.; Patil, R.; Das, S.; Stanley, A.; Yadav, L.; Sudhakar, A. Optimized Hydrophobic Interactions and Hydrogen Bonding at the Target-Ligand Interface Leads the Pathways of Drug-Designing. PLoS One 2010, 5. [Google Scholar] [CrossRef]
- Hwang, D.; Hyun, J.; Jo, G.; Koh, D.; Lim, Y. Synthesis and Complete Assignment of NMR Data of 20 Chalcones. Magnetic Resonance in Chemistry 2011, 49, 41–45. [Google Scholar] [CrossRef] [PubMed]
- Zhang, J.; Brodbelt, J.S. Structural Characterization and Isomer Differentiation of Chalcones by Electrospray Ionization Tandem Mass Spectrometry. Journal of Mass Spectrometry 2003, 38, 555–572. [Google Scholar] [CrossRef] [PubMed]
- Indrayanto, G.; Putra, G.S.; Suhud, F. Validation of In-Vitro Bioassay Methods: Application in Herbal Drug Research. Profiles Drug Subst Excip Relat Methodol 2021, 46, 273–307. [Google Scholar] [CrossRef] [PubMed]
- Riss, T.L.; Moravec, R.A.; Niles, A.L.; Duellman, S.; Benink, H.A.; Worzella, T.J.; Minor, L. Cell Viability Assays. Assay Guidance Manual 2016.
- Weerapreeyakul, N.; Nonpunya, A.; Barusrux, S.; Thitimetharoch, T.; Sripanidkulchai, B. Evaluation of the Anticancer Potential of Six Herbs against a Hepatoma Cell Line. Chinese Medicine (United Kingdom) 2012, 7, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Li, X.; Jin, L.; Ma, Y.; Jiang, Z.; Tang, H.; Tong, X. Xanthohumol Inhibits Non-Small Cell Lung Cancer by Activating PUMA-Mediated Apoptosis. Toxicology 2022, 470. [Google Scholar] [CrossRef] [PubMed]
- Rusetskaya, N.V.; Lukyanova, N.Y.; Chekhun, V.F. Molecular profile and cell cycle in MCF-7 and MCF-7/Dox cells exposed to conventional and liposomal forms of doxorubicin. Exp Oncol. 2009, 31. [Google Scholar] [PubMed]
- Sun, Z.; Zhou, D.; Yang, J.; Zhang, D. Doxorubicin Promotes Breast Cancer Cell Migration and Invasion via DCAF13. FEBS Open Bio 2022, 12, 221–230. [Google Scholar] [CrossRef] [PubMed]
- Şöhretoğlu, D.; Arroo, R.; Sari, S.; Huang, S. Flavonoids as Inducers of Apoptosis and Autophagy in Breast Cancer. Discovery and Development of Anti-Breast Cancer Agents from Natural Products 2021, 147–196. [Google Scholar] [CrossRef]
- Thorburn, A.; Thamm, D.H.; Gustafson, D.L. Autophagy and Cancer Therapy. Mol Pharmacol 2014, 85, 830–838. [Google Scholar] [CrossRef] [PubMed]
- Lin, P.H.; Chiang, Y.F.; Shieh, T.M.; Chen, H.Y.; Shih, C.K.; Wang, T.H.; Wang, K.L.; Huang, T.C.; Hong, Y.H.; Li, S.C.; et al. Dietary Compound Isoliquiritigenin, an Antioxidant from Licorice, Suppresses Triple-Negative Breast Tumor Growth via Apoptotic Death Program Activation in Cell and Xenograft Animal Models. Antioxidants 2020, 9. [Google Scholar] [CrossRef]
- Yang, L.H.; Ho, Y.J.; Lin, J.F.; Yeh, C.W.; Kao, S.H.; Hsu, L.S. Butein Inhibits the Proliferation of Breast Cancer Cells through Generation of Reactive Oxygen Species and Modulation of ERK and P38 Activities. Mol Med Rep 2012, 6, 1126–1132. [Google Scholar] [CrossRef] [PubMed]
- Michalkova, R.; Mirossay, L.; Kello, M.; Mojzisova, G.; Baloghova, J.; Podracka, A.; Mojzis, J. Anticancer Potential of Natural Chalcones: In Vitro and In Vivo Evidence. Int J Mol Sci 2023, 24, 10354. [Google Scholar] [CrossRef] [PubMed]
- Kitazumi, I.; Tsukahara, M. Regulation of DNA Fragmentation: The Role of Caspases and Phosphorylation. FEBS Journal 2011, 278, 427–441. [Google Scholar] [CrossRef] [PubMed]
- Lin, Y.F.; Lai, T.C.; Chang, C.K.; Chen, C.L.; Huang, M.S.; Yang, C.J.; Liu, H.G.; Dong, J.J.; Chou, Y.A.; Teng, K.H.; et al. Targeting the XIAP/Caspase-7 Complex Selectively Kills Caspase-3-Deficient Malignancies. J Clin Invest 2013, 123, 3861–3875. [Google Scholar] [CrossRef] [PubMed]
- Shogren-Knaak, M.; Ishii, H.; Sun, J.M.; Pazin, M.J.; Davie, J.R.; Peterson, C.L. Histone H4-K16 Acetylation Controls Chromatin Structure and Protein Interactions. Science (1979) 2006, 311, 844–847. [Google Scholar] [CrossRef] [PubMed]
- Boice, A.; Bouchier-Hayes, L. Targeting Apoptotic Caspases in Cancer. Biochim Biophys Acta Mol Cell Res 2020, 1867. [CrossRef]
- Battogtokh, G.; Cho, Y.Y.; Lee, J.Y.; Lee, H.S.; Kang, H.C. Mitochondrial-Targeting Anticancer Agent Conjugates and Nanocarrier Systems for Cancer Treatment. Front Pharmacol 2018, 9. [Google Scholar] [CrossRef] [PubMed]
- Lucantoni, F.; Salvucci, M.; Düssmann, H.; Lindner, A.U.; Lambrechts, D.; Prehn, J.H.M. BCL(X)L and BCL2 Increase the Metabolic Fitness of Breast Cancer Cells: A Single-Cell Imaging Study. Cell Death Differ 2021, 28, 1512–1531. [Google Scholar] [CrossRef] [PubMed]
- Tai, W.T.; Shiau, C.W.; Chen, H.L.; Liu, C.Y.; Lin, C.S.; Cheng, A.L.; Chen, P.J.; Chen, K.F. Mcl-1-Dependent Activation of Beclin 1 Mediates Autophagic Cell Death Induced by Sorafenib and SC-59 in Hepatocellular Carcinoma Cells. Cell Death Dis 2013, 4. [Google Scholar] [CrossRef] [PubMed]
- Ruvolo, P.P.; Deng, X.; May, W.S. Phosphorylation of Bcl2 and Regulation of Apoptosis. Leukemia 2001 15:4 2001, 15, 515–522. [Google Scholar] [CrossRef] [PubMed]
- Gopal, S.M.; Klumpers, F.; Herrmann, C.; Schäfer, L. V. Solvent Effects on Ligand Binding to a Serine Protease. Physical Chemistry Chemical Physics 2017, 19, 10753–10766. [Google Scholar] [CrossRef] [PubMed]
- Méndez-Callejas, G.M.; Leone, S.; Tanzarella, C.; Antoccia, A. Combretastatin A-4 Induces P53 Mitochondrial-Relocalisation Independent-Apoptosis in Non-Small Lung Cancer Cells. Cell Biol Int 2014, 38, 296–308. [Google Scholar] [CrossRef] [PubMed]
- Bar-On, O.; Shapira, anit; Hershko, D.D. Anti-Cancer Drugs; Wolters Kluwer Health | Lippincott Williams & Wilkins, 2007, 18.
- Park, D.; Jeong, H.; Lee, M.N.; Koh, A.; Kwon, O.; Yang, Y.R.; Noh, J.; Suh, P.G.; Park, H.; Ryu, S.H. Resveratrol Induces Autophagy by Directly Inhibiting MTOR through ATP Competition. Sci Rep 2016, 6. [Google Scholar] [CrossRef] [PubMed]
- Pettersen, E.F.; Goddard, T.D.; Huang, C.C.; Meng, E.C.; Couch, G.S.; Croll, T.I.; Morris, J.H.; Ferrin, T.E. UCSF ChimeraX: Structure Visualization for Researchers, Educators, and Developers. Protein Science 2021, 30, 70–82. [Google Scholar] [CrossRef] [PubMed]
- Frisch, M.J.; Trucks, G.W.; Schlegel, H.B.; Scuseria, G.E.; Robb, M.A.; Cheeseman, J.R.; Scalmani, G.; Barone, V.; Petersson, G.A.; Nakatsuji, H.; Li, X.; et al. Gaussian˜16 Revision C.01, 2016. Gaussian Inc. Wallingford CT.
- Case, D.A.; Aktulga, H.M.; Belfon, K.; Ben-Shalom, I.Y.; Berryman, J.T.; Brozell S.R.; Cerutti, D.S.; Cheatham, T.E.; III; Cisneros, G.A.; Cruzeiro, V.W.D.; et al. AMBER 2023. University of California, San Francisco, CA, 20323.
- Maier, J.A.; Martinez, C.; Kasavajhala, K.; Wickstrom, L.; Hauser, K.E.; Simmerling, C. Ff14SB: Improving the Accuracy of Protein Side Chain and Backbone Parameters from Ff99SB. J Chem Theory Comput 2015, 11, 3696–3713. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.; Wolf, R.M.; Caldwell, J.W.; Kollman, P.A.; Case, D.A. Development and Testing of a General Amber Force Field. J Comput Chem 2004, 25, 1157–1174. [Google Scholar] [CrossRef] [PubMed]
- Onufriev, A. V.; Izadi, S. Water Models for Biomolecular Simulations. Wiley Interdiscip Rev Comput Mol Sci 2018, 8, e1347. [Google Scholar] [CrossRef]
- Miller, B.R.; McGee, T.D.; Swails, J.M.; Homeyer, N.; Gohlke, H.; Roitberg, A.E. MMPBSA.Py: An Efficient Program for End-State Free Energy Calculations. J Chem Theory Comput 2012, 8, 3314–3321. [Google Scholar] [CrossRef] [PubMed]
- Ben-Shalom, I.Y.; Pfeiffer-Marek, S.; Baringhaus, K.H.; Gohlke, H. Efficient Approximation of Ligand Rotational and Translational Entropy Changes upon Binding for Use in MM-PBSA Calculations. J Chem Inf Model 2017, 57, 170–189. [Google Scholar] [CrossRef] [PubMed]
- Genheden, S.; Ryde, U. Comparison of the Efficiency of the LIE and MM/GBSA Methods to Calculate Ligand-Binding Energies. J Chem Theory Comput 2011, 7, 3768–3778. [Google Scholar] [CrossRef] [PubMed]
- Hou, T.; Wang, J.; Li, Y.; Wang, W. Assessing the Performance of the MM/PBSA and MM/GBSA Methods. 1. The Accuracy of Binding Free Energy Calculations Based on Molecular Dynamics Simulations. J Chem Inf Model 2011, 51, 69–82. [Google Scholar] [CrossRef] [PubMed]
- Roe, D.R.; Cheatham, T.E. PTRAJ and CPPTRAJ: Software for Processing and Analysis of Molecular Dynamics Trajectory Data. J Chem Theory Comput 2013, 9, 3084–3095. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




