Submitted:
10 November 2023
Posted:
13 November 2023
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Myelodysplastic/Myeloproliferative Related Dermatosis with Indolent Clinical Outcome
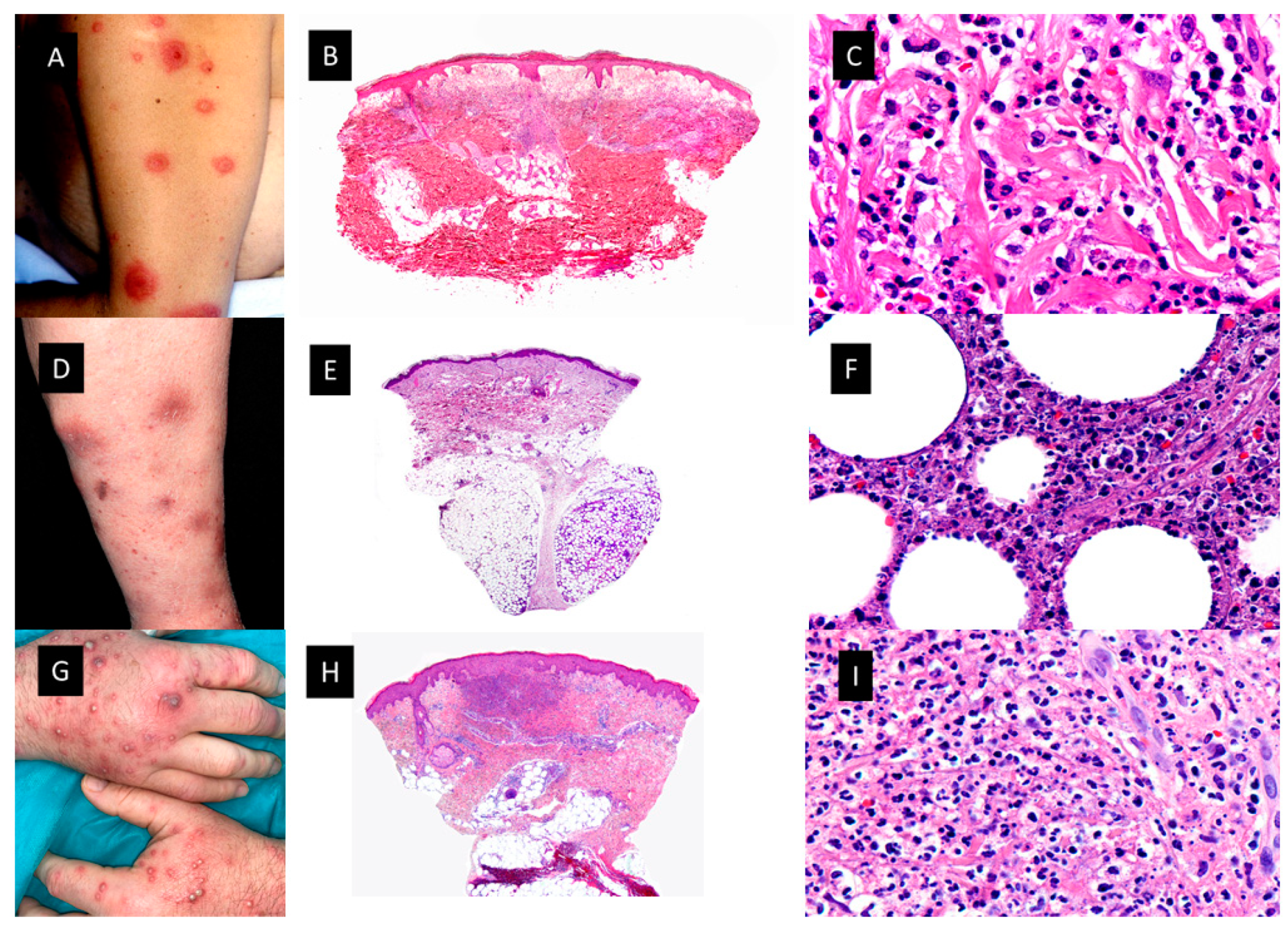
2.1. NEUTROPHILIC DERMATOSES
2.2. SWEET SYNDROME
2.2.1. HISTIOCYTOID SS (H-SS)
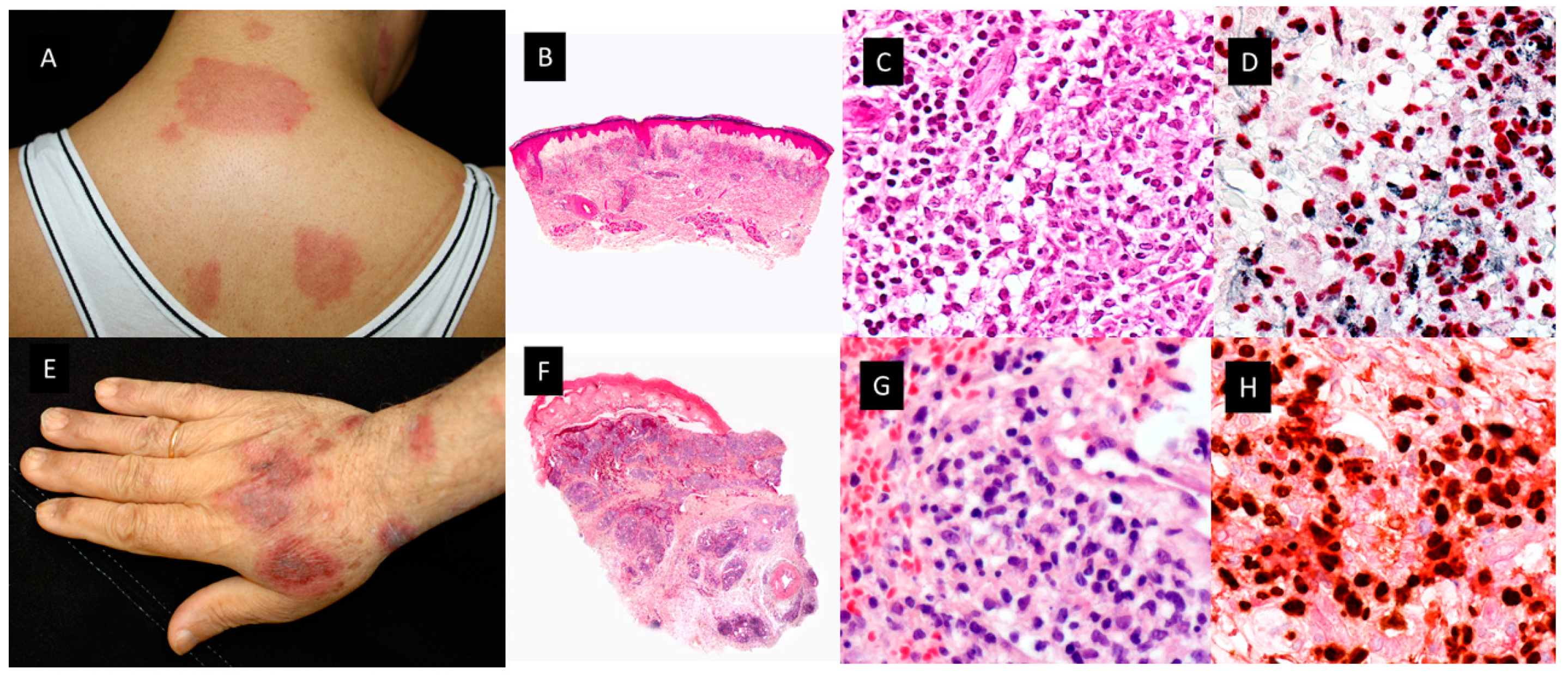
2.2.2. OTHER MORPHOLOGICAL VARIANTS OF SWEET SYNDROME
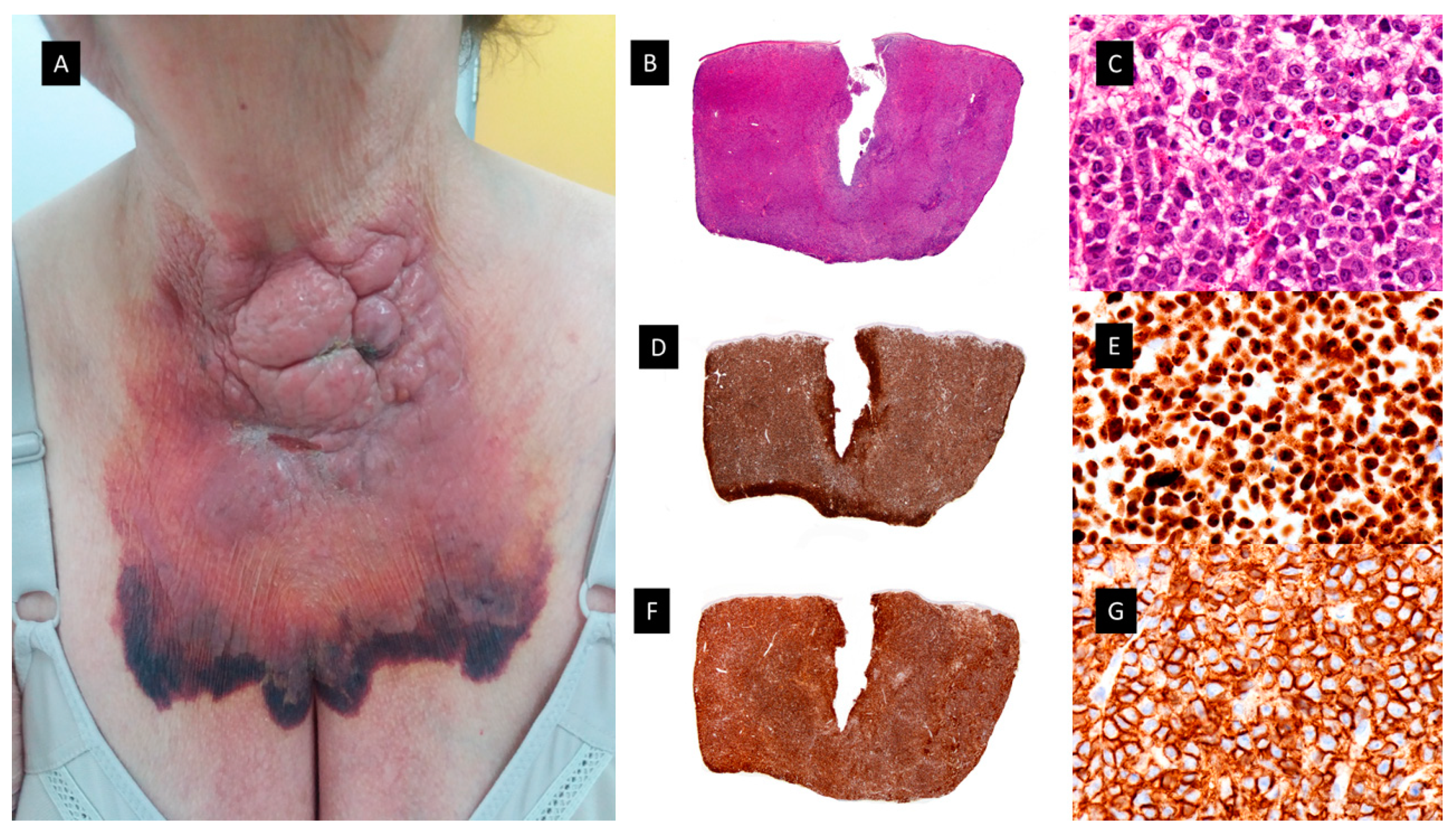
2.3. VEXAS SYNDROME
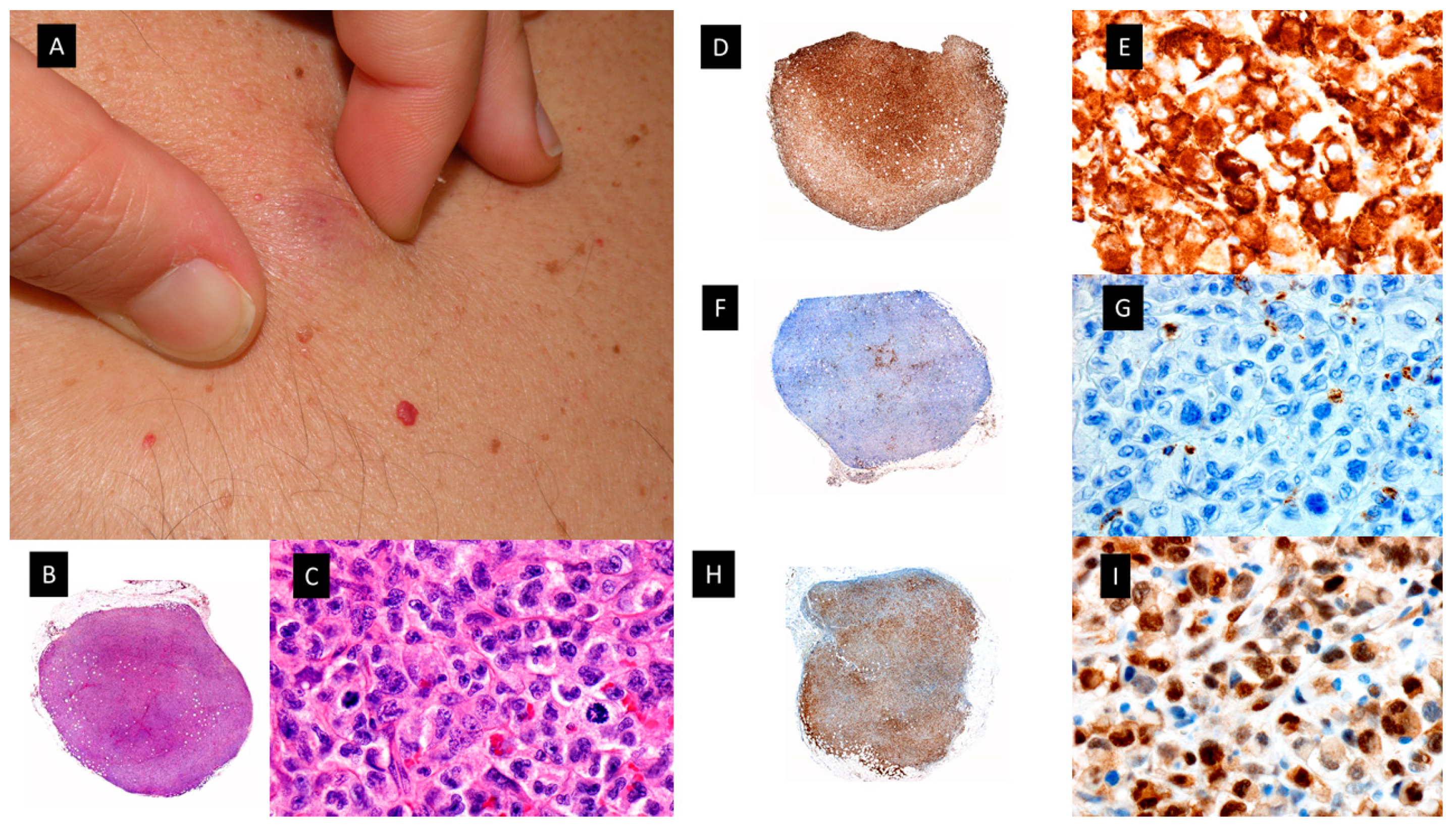
2.4. MYELODYSPLASIA CUTIS
2.5. GRANULOMATOUS DERMATOSES
2.6. MATURE PLASMACYTOID DENDRITIC CELL DERMATOSES
2.7. OTHERS MYELODYSPLASTIC/MYELOPROLIFERATIVE DERMATOSIS
3. CUTANEOUS PROCESSES RELATED TO EITHER MDS/MNP AND AGGRESSIVE CLINICAL BEHAVIOR
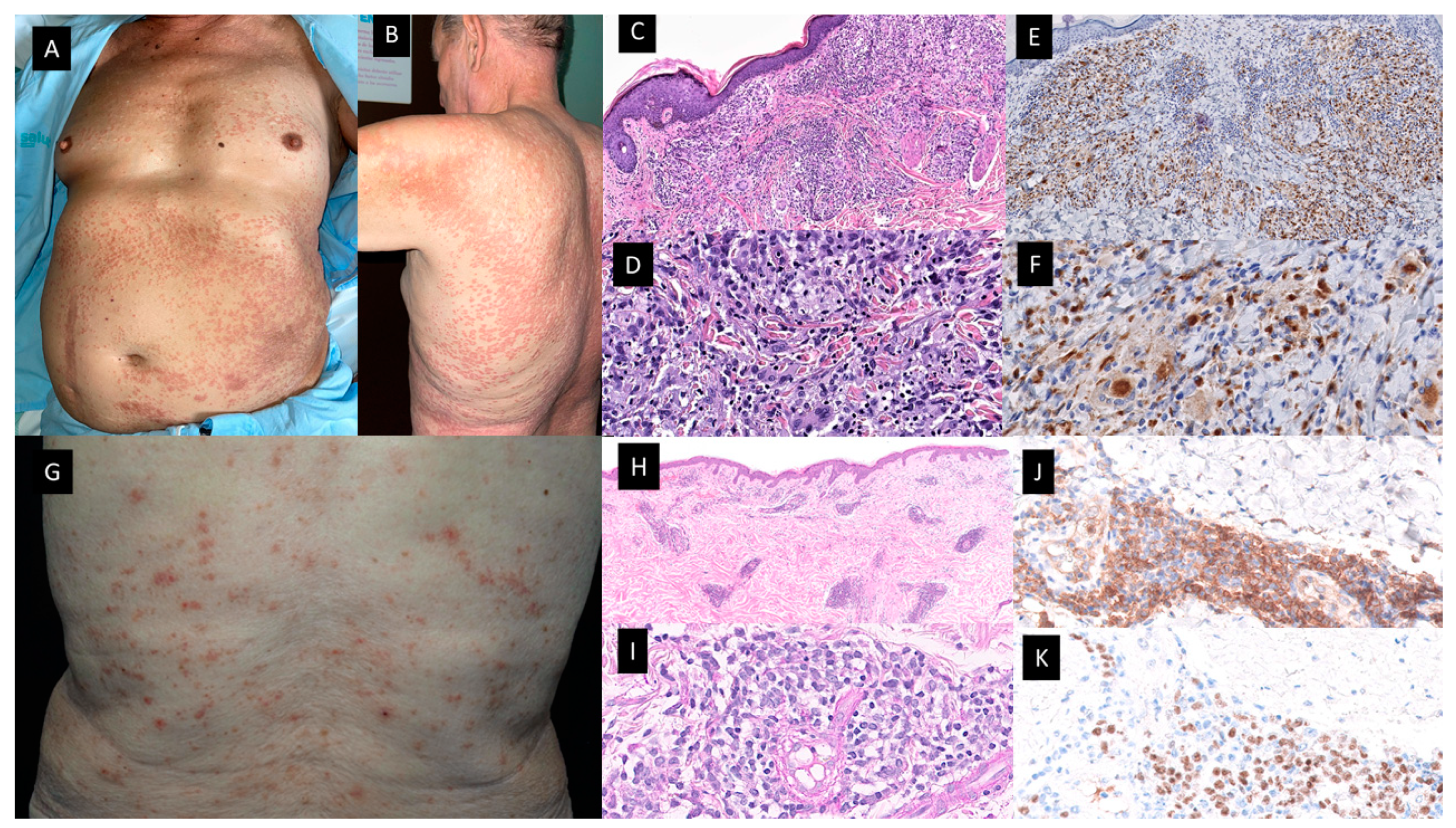
3.1. LEUKEMIA CUTIS /MYELOID LEUKEMIA CUTIS
3.2. GRANULOCYTIC SARCOMA
ALEUKEMIC LC
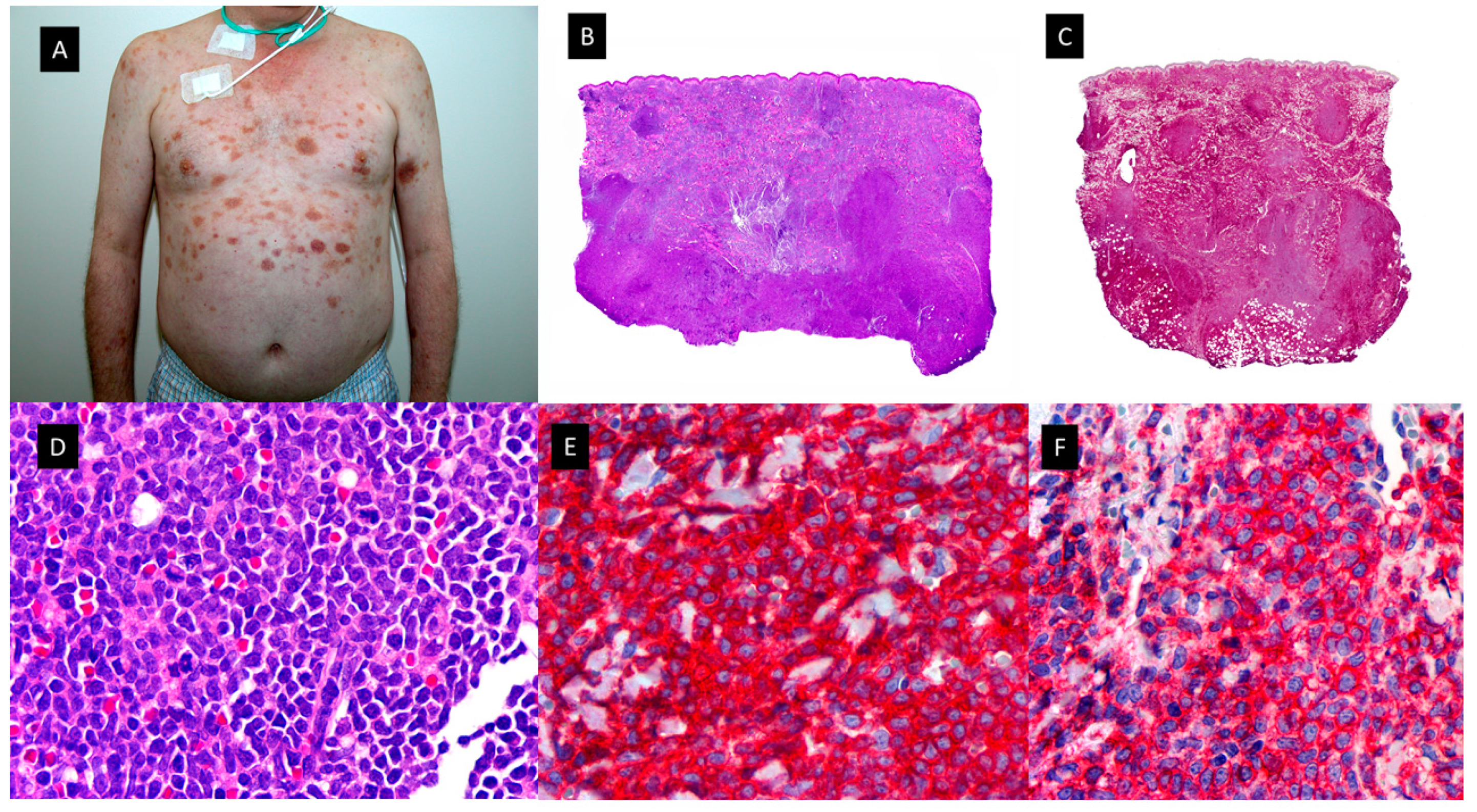
3.3. BLASTIC PLASMACYTOID DENDRITIC CELL TUMOR
3.4. HISTIOCYTOID DISORDERS/HISTIOCYTOSIS
3.4.1. HISTIOCYTIC SARCOMA
3.4.2. OTHER HISTIOCYTOSIS
4. OTHER CUTANEOUS DISORDERS IN MDS/MPN PATIENTS
4.1. CUTANEOUS MANIFESTATIONS SECUNDARY TO MICROCIRCULATION ABNORMALITIES
4.2. OTHER INFLAMMATORY/ AUTOIMMUNE DISEASES
5. CONCLUSION
Acknowledgments
References
- Khoury, J.D.; Solary, E.; Abla, O.; et al. The 5th edition of the World Health Organization Classification of Haematolymphoid Tumours: Myeloid and Histiocytic/Dendritic Neoplasms. Leukemia. 2022, 36, 1703–1719. [Google Scholar] [CrossRef] [PubMed]
- Arber, D.A.; Orazi, A.; Hasserjian, R.P.; et al. International Consensus Classification of Myeloid Neoplasms and Acute Leukemias: integrating morphologic, clinical, and genomic data. Blood. 2022, 140, 1200–1228. [Google Scholar] [CrossRef] [PubMed]
- Orazi, A.; Germing, U. The myelodysplastic/myeloproliferative neoplasms: myeloproliferative diseases with dysplastic features. Leukemia. 2008, 22, 1308–19. [Google Scholar] [CrossRef] [PubMed]
- Wagner, G.; Fenchel, K.; Back, W.; Schulz, A.; Sachse, M.M. Leukemia cutis - epidemiology, clinical presentation, and differential diagnoses. J Dtsch Dermatol Ges. 2012, 10, 27–36. [Google Scholar] [CrossRef]
- Passet, M.; Lepelletier, C.; Vignon-Pennamen, M.D.; et al. Next-Generation Sequencing in Myeloid Neoplasm-Associated Sweet's Syndrome Demonstrates Clonal Relation between Malignant Cells and Skin-Infiltrating Neutrophils. J Invest Dermatol. 2020, 140, 1873–1876. [Google Scholar] [CrossRef]
- Martin de Frémont, G.; Hirsch, P.; Gimenez de Mestral, S.; et al. Myeloid Clonal Infiltrate Identified With Next-Generation Sequencing in Skin Lesions Associated With Myelodysplastic Syndromes and Chronic Myelomonocytic Leukemia: A Case Series. Front Immunol. 2021, 12, 715053. [Google Scholar] [CrossRef] [PubMed]
- Whittington, C.P.; Ross, C.W.; Ramirez, J.A.; Lowe, L.; Brown, N.; Hristov, A.C. Myelodysplasia Cutis. Arch Pathol Lab Med. 2023. [Google Scholar] [CrossRef] [PubMed]
- Weiss, E.H.; Ko, C.J.; Leung, T.H.; et al. Neutrophilic Dermatoses: a Clinical Update. Curr Dermatol Rep. 2022, 11, 89–102. [Google Scholar] [CrossRef]
- Sweet, RD. AN ACUTE FEBRILE NEUTROPHILIC DERMATOSIS. Br J Dermatol. 1964, 76, 349–56. [Google Scholar] [CrossRef]
- Ferea, C.R.; Mihai, S.N.; Balan, G.; Badescu, M.C.; Tutunaru, D.; Tatu, A.L. Sweet Syndrome Associated with Myelodysplastic Syndrome-A Review of a Multidisciplinary Approach. Life (Basel) 2023, 13. [Google Scholar] [CrossRef]
- Cohen, P.R. Sweet's syndrome--a comprehensive review of an acute febrile neutrophilic dermatosis. Orphanet J Rare Dis. 2007, 2, 34. [Google Scholar] [CrossRef] [PubMed]
- Requena, L.; Kutzner, H.; Palmedo, G.; et al. Histiocytoid Sweet syndrome: a dermal infiltration of immature neutrophilic granulocytes. Arch Dermatol. 2005, 141, 834–42. [Google Scholar] [CrossRef]
- Alegría-Landa, V.; Rodríguez-Pinilla, S.M.; Santos-Briz, A.; et al. Clinicopathologic, Immunohistochemical, and Molecular Features of Histiocytoid Sweet Syndrome. JAMA Dermatol. 2017, 153, 651–659. [Google Scholar] [CrossRef] [PubMed]
- Vignon-Pennamen, M.D.; Juillard, C.; Rybojad, M.; et al. Chronic recurrent lymphocytic Sweet syndrome as a predictive marker of myelodysplasia: a report of 9 cases. Arch Dermatol. 2006, 142, 1170–6. [Google Scholar] [CrossRef] [PubMed]
- Ghoufi, L.; Ortonne, N.; Ingen-Housz-Oro, S.; et al. Histiocytoid Sweet Syndrome Is More Frequently Associated With Myelodysplastic Syndromes Than the Classical Neutrophilic Variant: A Comparative Series of 62 Patients. Medicine (Baltimore). 2016, 95, e3033. [Google Scholar] [CrossRef] [PubMed]
- Delaleu, J.; Kim, R.; Zhao, L.P.; et al. Clinical, pathological, and molecular features of myelodysplasia cutis. Blood. 2022, 139, 1251–1253. [Google Scholar] [CrossRef] [PubMed]
- Kamimura, A.; Yanagisawa, H.; Tsunemi, Y.; et al. Normolipemic xanthomatized Sweet's syndrome: A variant of Sweet's syndrome with myelodysplastic syndrome. J Dermatol. 2021, 48, 695–698. [Google Scholar] [CrossRef] [PubMed]
- Sutra-Loubet, C.; Carlotti, A.; Guillemette, J.; Wallach, D. Neutrophilic panniculitis. J Am Acad Dermatol. 2004, 50, 280–5. [Google Scholar] [CrossRef]
- Jagdeo, J.; Campbell, R.; Long, T.; Muglia, J.; Telang, G.; Robinson-Bostom, L. Sweet's syndrome--like neutrophilic lobular panniculitis associated with all-trans-retinoic acid chemotherapy in a patient with acute promyelocytic leukemia. J Am Acad Dermatol. 2007, 56, 690–3. [Google Scholar] [CrossRef] [PubMed]
- Chan, M.P.; Duncan, L.M.; Nazarian, R.M. Subcutaneous Sweet syndrome in the setting of myeloid disorders: a case series and review of the literature. J Am Acad Dermatol. 2013, 68, 1006–15. [Google Scholar] [CrossRef] [PubMed]
- Srisuttiyakorn, C.; Reeve, J.; Reddy, S.; Imaeda, S.; Lazova, R. Subcutaneous histiocytoid Sweet's syndrome in a patient with myelodysplastic syndrome and acute myeloblastic leukemia. J Cutan Pathol. 2014, 41, 475–9. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.; Cornejo, K.M.; Rork, J.; Rothman, K.; Deng, A. Subcutaneous Histiocytoid Sweet Syndrome in a Patient With Relapsed Acute Myeloblastic Leukemia. Am J Dermatopathol. 2018, 40, 459–462. [Google Scholar] [CrossRef]
- Lin, J.; Zhang, Q.; Chen, M. Subcutaneous histiocytoid Sweet's syndrome in a patient associated with myelodysplastic syndrome-refractory anemia. J Dermatol. 2012, 39, 99–101. [Google Scholar] [CrossRef] [PubMed]
- Beck, D.B.; Ferrada, M.A.; Sikora, K.A.; et al. Somatic Mutations in UBA1 and Severe Adult-Onset Autoinflammatory Disease. N Engl J Med. 2020, 383, 2628–2638. [Google Scholar] [CrossRef]
- Cooper, J.N.; Young, N.S. Clonality in context: hematopoietic clones in their marrow environment. Blood. 2017, 130, 2363–2372. [Google Scholar] [CrossRef] [PubMed]
- 26. Haines P, Pullarkat S, Said J. VEXAS syndrome: Vacuoles in myeloid, erythroid, and lymphoid lineages. Int J Lab Hematol. [CrossRef]
- Djerbi, N.; Zimmermann, K.; Balabanov, S.; Manz, M.G.; Becker, M.O.; Roncador, M. Intra-patient competition of VEXAS syndrome and CML clones. Blood Adv. 2023. [Google Scholar] [CrossRef]
- Nguyen, J.K.; Routledge, D.; van Der Weyden, C.; et al. VEXAS syndrome: A dermatological perspective. Australas J Dermatol. 2022, 63, 488–492. [Google Scholar] [CrossRef]
- Osio, A.; Battistella, M.; Feugeas, J.P.; et al. Myelodysplasia Cutis Versus Leukemia Cutis. J Invest Dermatol. 2015, 135, 2321–2324. [Google Scholar] [CrossRef]
- Sujobert, P.; Cuccuini, W.; Vignon-Pennamen, D.; et al. Evidence of differentiation in myeloid malignancies associated neutrophilic dermatosis: a fluorescent in situ hybridization study of 14 patients. J Invest Dermatol. 2013, 4, 1111–4. [Google Scholar] [CrossRef]
- Gottlieb GJ DR, Ackerman AB. 1:3-6. Interstitial granulomatous dermatitis with cutaneous cords and arthritis: linear subcutaneous bands in rheumatoid arthritis revisited. Dermatopathol Pract Concept. 1995, (1):3-6.
- Chu, P.; Connolly, M.K.; LeBoit, P.E. The histopathologic spectrum of palisaded neutrophilic and granulomatous dermatitis in patients with collagen vascular disease. Arch Dermatol. 1994, 130, 1278–83. [Google Scholar] [CrossRef] [PubMed]
- Rosenbach M, English JC, 3rd. Reactive Granulomatous Dermatitis: A Review of Palisaded Neutrophilic and Granulomatous Dermatitis, Interstitial Granulomatous Dermatitis, Interstitial Granulomatous Drug Reaction, and a Proposed Reclassification. Dermatol Clin. 2015, 33, 373–87. [CrossRef]
- Yoneta, K.; Fujimoto, N.; Teramura, K.; Takayama, S.; Tanaka, T. Disseminated granulomatous skin lesions associated with myelodysplastic syndrome treated successfully with tranilast: a case report and review of the literature. Eur J Dermatol. 2016, 26, 398–400. [Google Scholar] [CrossRef]
- Prieto-Torres, L.; Santonja, C.; Chamizo, C.; et al. Granulomatous Dermatitis Heralding Myelodisplastic/Myeloproliferative Neoplasms. Neoplastic or Reactive Cells? A Study of 2 Cases. Am J Dermatopathol. 2022, 44, 456–460. [Google Scholar] [CrossRef]
- Federmann, B.; Bonzheim, I.; Yazdi, A.S.; Schmidt, J.; Fend, F.; Metzler, G. Generalized palisaded neutrophilic and granulomatous dermatitis-a cutaneous manifestation of chronic myelomonocytic leukemia? A clinical, histopathological, and molecular study of 3 cases. Hum Pathol. 2017, 64, 198–206. [Google Scholar] [CrossRef]
- Kyriakou, A.; Patsatsi, A.; Papadopoulos, V.; Kioumi, A.; Efstratiou, I.; Lazaridou, E. A case of palisaded neutrophilic granulomatous dermatitis with subsequent development of chronic myelomonocytic leukemia. Clin Case Rep. 2019, 4, 695–698. [Google Scholar] [CrossRef] [PubMed]
- Enescu, C.D.; Patel, A.; Friedman, B.J. Unique Recognizable Histopathologic Variant of Palisaded Neutrophilic and Granulomatous Dermatitis that Is Associated With SRSF2-Mutated Chronic Myelomonocytic Leukemia: Case Report and Review of the Literature. Am J Dermatopathol. 2022, 44, e33–e36. [Google Scholar] [CrossRef] [PubMed]
- Tzankov, A.; Hebeda, K.; Kremer, M.; et al. Plasmacytoid dendritic cell proliferations and neoplasms involving the bone marrow : Summary of the workshop cases submitted to the 18th Meeting of the European Association for Haematopathology (EAHP) organized by the European Bone Marrow Working Group, Basel 2016. Ann Hematol. 2017, 5, 765–777. [Google Scholar]
- Wang, P.; Feng, Y.; Deng, X.; et al. Tumor-forming plasmacytoid dendritic cells in acute myelocytic leukemia: a report of three cases and literature review. Int J Clin Exp Pathol. 2017, 10, 7285–7291. [Google Scholar] [PubMed]
- Facchetti, F.; Cigognetti, M.; Fisogni, S.; Rossi, G.; Lonardi, S.; Vermi, W. Neoplasms derived from plasmacytoid dendritic cells. Mod Pathol. 2016, 29, 98–111. [Google Scholar] [CrossRef] [PubMed]
- Orazi, A.; Chiu, R.; O'Malley, D.P.; et al. Chronic myelomonocytic leukemia: The role of bone marrow biopsy immunohistology. Mod Pathol. 2006, 19, 1536–45. [Google Scholar] [CrossRef]
- Lucas, N.; Duchmann, M.; Rameau, P.; et al. Biology and prognostic impact of clonal plasmacytoid dendritic cells in chronic myelomonocytic leukemia. Leukemia. 2019, 33, 2466–2480. [Google Scholar] [CrossRef] [PubMed]
- Vitte, F.; Fabiani, B.; Bénet, C.; et al. Specific skin lesions in chronic myelomonocytic leukemia: a spectrum of myelomonocytic and dendritic cell proliferations: a study of 42 cases. Am J Surg Pathol. 2012, 36, 1302–16. [Google Scholar] [CrossRef] [PubMed]
- Machan, S.; Alonso-Dominguez, J.M.; Sánchez García, F.J.; et al. Plasmacytoid Dendritic Cell Dermatosis Associated to Myeloproliferative/Myelodysplastic Neoplasms. Am J Surg Pathol. 2022, 46, 1623–1632. [Google Scholar] [CrossRef] [PubMed]
- Dargent, J.L.; Henne, S.; Pranger, D.; et al. Tumor-forming plasmacytoid dendritic cells associated with myeloid neoplasms. Report of a peculiar case with histopathologic features masquerading as lupus erythematosus. J Cutan Pathol. 2016, 43, 280–6. [Google Scholar] [CrossRef]
- Jian, J.; Qiao, Y.; Li, Y.; Guo, Y.; Ma, H.; Liu, B. Mutations in chronic myelomonocytic leukemia and their prognostic relevance. Clin Transl Oncol. 2021, 23, 1731–1742. [Google Scholar] [CrossRef] [PubMed]
- Hirsch, C.M.; Nazha, A.; Kneen, K.; et al. Consequences of mutant TET2 on clonality and subclonal hierarchy. Leukemia. 2018, 32, 1751–1761. [Google Scholar] [CrossRef]
- Wang N, Wang F, Shan N, Sui X, Xu H. IDH1 Mutation Is an Independent Inferior Prognostic Indicator for Patients with Myelodysplastic Syndromes. Acta Haematol. 2017, 138, 143–151. [CrossRef] [PubMed]
- Brazão, C.; Mancha, D.; Antunes-Duarte, S.; Kempf, W.; Soares-de-Almeida, L. Chilblain-Like Eruption Unveiling Cutaneous Aleukemic Relapse of Acute Myeloid Leukemia. Am J Dermatopathol. Sep 13 2023. [CrossRef]
- Yazawa, H.; Saga, K.; Omori, F.; Jimbow, K.; Sasagawa, Y. The chilblain-like eruption as a diagnostic clue to the blast crisis of chronic myelocytic leukemia. J Am Acad Dermatol. 2004, 50 (2 Suppl), S42–4. [Google Scholar] [CrossRef]
- Affleck, A.G.; Ravenscroft, J.C.; Leach, I.H. Chilblain-like leukemia cutis. Pediatr Dermatol. 2007, 24, 38–41. [Google Scholar] [CrossRef]
- Faria, C.; Tzankov, A. Progression in Myeloid Neoplasms: Beyond the Myeloblast. Pathobiology. 2023, 1–21. [Google Scholar] [CrossRef] [PubMed]
- Bénet, C.; Gomez, A.; Aguilar, C.; et al. Histologic and immunohistologic characterization of skin localization of myeloid disorders: a study of 173 cases. Am J Clin Pathol. 2011, 135, 278–90. [Google Scholar] [CrossRef] [PubMed]
- Cho-Vega, J.H.; Medeiros, L.J.; Prieto, V.G.; Vega, F. Leukemia cutis. Am J Clin Pathol. Jan 2008, 129, 130–42. [Google Scholar] [CrossRef] [PubMed]
- Kaddu, S.; Zenahlik, P.; Beham-Schmid, C.; Kerl, H.; Cerroni, L. Specific cutaneous infiltrates in patients with myelogenous leukemia: a clinicopathologic study of 26 patients with assessment of diagnostic criteria. J Am Acad Dermatol. 1999, 40 Pt 1, 966–78. [Google Scholar] [CrossRef] [PubMed]
- Mathew, R.A.; Bennett, J.M.; Liu, J.J.; et al. Cutaneous manifestations in CMML: Indication of disease acceleration or transformation to AML and review of the literature. Leuk Res. 2012, 36, 72–80. [Google Scholar] [CrossRef]
- Ohno, S.; Yokoo, T.; Ohta, M. Aleukemic leukemia cutis. J Am Acad Dermatol. Feb 1990;22(2 Pt 2):374-7. [CrossRef]
- Heskel, N.S.; White, C.R.; Fryberger, S.; Neerhout, R.C.; Spraker, M.; Hanifin, J.M. Aleukemic leukemia cutis: juvenile chronic granulocytic leukemia presenting with figurate cutaneous lesions. J Am Acad Dermatol. 1983, 9, 423–7. [Google Scholar] [CrossRef]
- Gil-Mateo, M.P.; Miquel, F.J.; Piris, M.A.; Sánchez, M.; Martin-Aragonés, G. Aleukemic "leukemia cutis" of monocytic lineage. J Am Acad Dermatol. 1997, 36 (5 Pt 2), 837-40. [CrossRef]
- Aboutalebi, A.; Korman, J.B.; Sohani, A.R.; et al. Aleukemic cutaneous myeloid sarcoma. J Cutan Pathol. 2013, 40, 996–1005. [Google Scholar] [CrossRef] [PubMed]
- Iliadis, A.; Koletsa, T.; Georgiou, E.; Patsatsi, A.; Sotiriadis, D.; Kostopoulos, I. Bilateral Aleukemic Myeloid Sarcoma of the Eyelids With Indolent Course. Am J Dermatopathol. Apr 2016, 38, 312–4. [Google Scholar] [CrossRef]
- Dijkman, R.; van Doorn, R.; Szuhai, K.; Willemze, R.; Vermeer, M.H.; Tensen, C.P. Gene-expression profiling and array-based CGH classify CD4+CD56+ hematodermic neoplasm and cutaneous myelomonocytic leukemia as distinct disease entities. Blood. 2007, 109, 1720–7. [Google Scholar] [CrossRef]
- Khanlari, M.; Yin, C.C.; Takahashi, K.; et al. Bone marrow clonal hematopoiesis is highly prevalent in blastic plasmacytoid dendritic cell neoplasm and frequently sharing a clonal origin in elderly patients. Leukemia. 2022, 36, 1343–1350. [Google Scholar] [CrossRef] [PubMed]
- Yamada, T.; Hiramoto, N.; Mori, T.; et al. Coincidence of cutaneous blastic plasmacytoid dendritic cell neoplasm and myelodysplastic syndrome derived from clonal hematopoiesis. Blood Cancer J. 2023, 1, 119. [Google Scholar] [CrossRef] [PubMed]
- Emile, J.F.; Abla, O.; Fraitag, S.; et al. Revised classification of histiocytoses and neoplasms of the macrophage-dendritic cell lineages. Blood. 2016, 127, 2672–81. [Google Scholar] [CrossRef] [PubMed]
- Ansari, J.; Naqash, A.R.; Munker, R.; et al. Histiocytic sarcoma as a secondary malignancy: pathobiology, diagnosis, and treatment. Eur J Haematol. 2016, 97, 9–16. [Google Scholar] [CrossRef] [PubMed]
- Zhao, J.; Niu, X.; Wang, Z.; Lu, H.; Lin, X.; Lu, Q. Histiocytic sarcoma combined with acute monocytic leukemia: a case report. Diagn Pathol. 2015, 10, 110. [Google Scholar] [CrossRef] [PubMed]
- Mori, M.; Matsushita, A.; Takiuchi, Y.; et al. Histiocytic sarcoma and underlying chronic myelomonocytic leukemia: a proposal for the developmental classification of histiocytic sarcoma. Int J Hematol. 2010, 92, 168–73. [Google Scholar] [CrossRef] [PubMed]
- Pérez-Sáenz, M.A.; Rodriguez-Pinilla, S.M.; Salgado, R.N.; et al. Three monocytic neoplasms in a single patient. Leuk Lymphoma. 2020, 61, 2523–2526. [Google Scholar] [CrossRef] [PubMed]
- Papo, M.; Diamond, E.L.; Cohen-Aubart, F.; et al. High prevalence of myeloid neoplasms in adults with non-Langerhans cell histiocytosis. Blood. 2017, 130, 1007–1013. [Google Scholar] [CrossRef]
- Kemps, P.G.; Hebeda, K.M.; Pals, S.T.; et al. Spectrum of histiocytic neoplasms associated with diverse haematological malignancies bearing the same oncogenic mutation. J Pathol Clin Res. 2021, 7, 10–26. [Google Scholar] [CrossRef]
- Miralles, E.S.; Escribano, L.; Bellas, C.; Núñez, M.; Ledo, A. Cutaneous xanthomatous tumours as an expression of chronic myelomonocytic leukaemia? Clin Exp Dermatol. 1996, 21, 145–7. [Google Scholar] [CrossRef] [PubMed]
- Bonnet, P.; Chasset, F.; Moguelet, P.; et al. Erdheim-Chester disease associated with chronic myelomonocytic leukemia harboring the same clonal mutation. Haematologica. 2019, 104, e530–e533. [Google Scholar] [CrossRef] [PubMed]
- Fiegl, A.; Dirnhofer, S.; Juskevicius, D.; et al. Testicular Rosai-Dorfman disease clonally related to CMML - Case report and literature review. Pathol Res Pract. Jul 2023, 247, 154548. [Google Scholar] [CrossRef] [PubMed]
- Kiavash, K.; Malone, J.C. Langerhans Cell Histiocytosis Associated With Underlying Hematolymphoid Disorders in Adults: Report of 2 Cases and Review of the Literature. Am J Dermatopathol. 2018, 40, 588–593. [Google Scholar] [CrossRef]
- Billings, S.D.; Hans, C.P.; Schapiro, B.L.; et al. Langerhans cell histiocytosis associated with myelodysplastic syndrome in adults. J Cutan Pathol. 2006, 33, 171–4. [Google Scholar] [CrossRef]
- Durham, B.H.; Roos-Weil, D.; Baillou, C.; et al. Functional evidence for derivation of systemic histiocytic neoplasms from hematopoietic stem/progenitor cells. Blood. 2017, 130, 176–180. [Google Scholar] [CrossRef]
- Milne, P.; Bigley, V.; Bacon, C.M.; et al. Hematopoietic origin of Langerhans cell histiocytosis and Erdheim-Chester disease in adults. Blood. 2017, 130, 167–175. [Google Scholar] [CrossRef] [PubMed]
- Marks, R.; Lim, C.C.; Borrie, P.F. A perniotic syndrome with monocytosis and neutropenia--a possible association with a preleukaemic state. Br J Dermatol. 1969, 81, 327–32. [Google Scholar] [CrossRef] [PubMed]
- Kelly, J.W.; Dowling, J.P. Pernio. A possible association with chronic myelomonocytic leukemia. Arch Dermatol. 1985, 121, 1048–52. [Google Scholar] [CrossRef]
- Dreno, B.; Gandon, P.; Bureau, B.; Milpied, N.; Barrière, H. Skin lesions from hypersensitivity to cold during chronic myelomonocytic leukaemia. Br J Dermatol. 1986, 115, 607–9. [Google Scholar] [CrossRef]
- Nazzaro, G.; Genovese, G.; Marzano, A.V. Idiopathic chilblains in myelomonocytic leukemia: not a simple association. Int J Dermatol. 2018, 57, 596–598. [Google Scholar] [CrossRef] [PubMed]
- Pielasinski, U.; Haro, R.; Santonja, C.; Kutzner, H.; Requena, L. Essential thrombocythemia presenting as localized livedo reticularis. Am J Dermatopathol. 2013, 35, e22–5. [Google Scholar] [CrossRef] [PubMed]
- Lapeña Casado, A.; Santonja, C.; García-García, M.; et al. Essential thrombocythemia manifesting as livedoid and purpuric skin lesions: Report of two cases and literature review. J Cutan Pathol. 2023. [Google Scholar] [CrossRef] [PubMed]
- Saif, M.W.; Hopkins, J.L.; Gore, S.D. Autoimmune phenomena in patients with myelodysplastic syndromes and chronic myelomonocytic leukemia. Leuk Lymphoma. 2002, 43, 2083–92. [Google Scholar] [CrossRef] [PubMed]
- Yoshida, M.; Horiuchi, M.; Ueda, H.; et al. [Cutaneous extramedullary hematopoiesis associated with myelodysplastic/myeloproliferative neoplasm, unclassifiable]. Rinsho Ketsueki. 2015, 56, 911–4. [Google Scholar] [CrossRef] [PubMed]
| 1. NEUTROPHILIC DERMATOSES 1.1 SWEET SYNDROME (SS) 1.1.1 HISTIOCYTOID SWEET SYNDROME (H-SS) CD15+, CD43+, CD45+, Lysozyme+, MPO+ CD68/KP1+>CD68/PGM1+, MNDA+ (Double IS MNDA/MPO+) 1.1.2 OTHER MORPHOLOGICAL VARIANTS OF SS 1.2 VEXAS SYNDROME MPO+, MNDA+ |
| 1. MYELODYSPLASIA CUTIS CD33+, CD163+, CD14+, CD68/PGM1+, MPO+(Double IS CD123+/MNDA+) CD34- CD117-, CD56-, low ki67 |
| 3. GRANULOMATOUS DERMATOSES CD14+, CD68/PGM1+, CD123+ CD56-, S100-, CD1a- |
| 4. MATURE PLASMACYTOID DENDRITIC CELL DERMATOSES Double IS CD123+/SPIB + CD56-, CD34-, CD117-, MPO-, TDT-, Bcl2-, MNDA- |
| 5. OTHERS |
| 1. LEUKEMIA CUTIS/MYELOID LEUKEMIA CUTIS 1.1 GRANULOCYTIC SARCOMA CD68+, MPO+, CD33+, CD13+, high ki67 More likely to express CD34, CD117 1.2. ALEUKEMIC LEUKEMIA CUTIS No systemic involvement at diagnosis |
| 2. BLASTIC PLASMACYTOID DENDRITIC CELL TUMOR CD4+, CD56+ high Ki67, SPIB+, CD123+ MPO-, CD13-, CD33-, MNDA- |
| 3. HISTIOCYTOID DISORDERS/HISTIOCYTOSES 3.1 HISTIOCYTIC SARCOMA CD163+, CD68+, Lysozyme+, pERK+, MPO- 3.2 OTHERS ECD:CD68+, CD163+, CD1a- LCH: S100+, CD1a+, Langerin/CD203+ |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




