Submitted:
09 November 2023
Posted:
13 November 2023
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
2.1. Naphthoquinones
2.2. Microorganisms
2.3. Antifungal activity assays
2.4. Antifungal mechanism of action
2.5. Toxicity assays
2.6. Statistical analysis
3. Results
3.1. Antifungal Activity
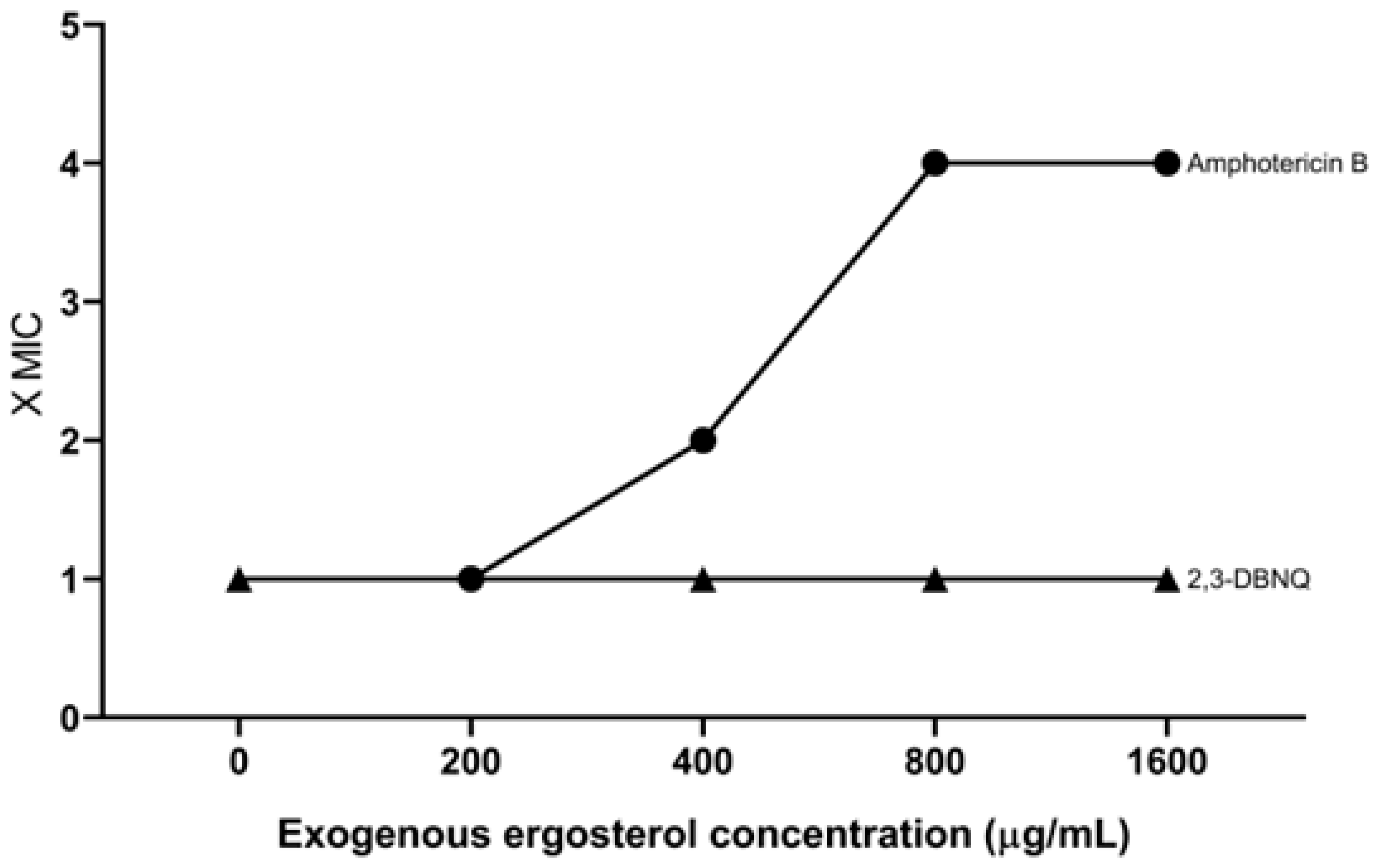
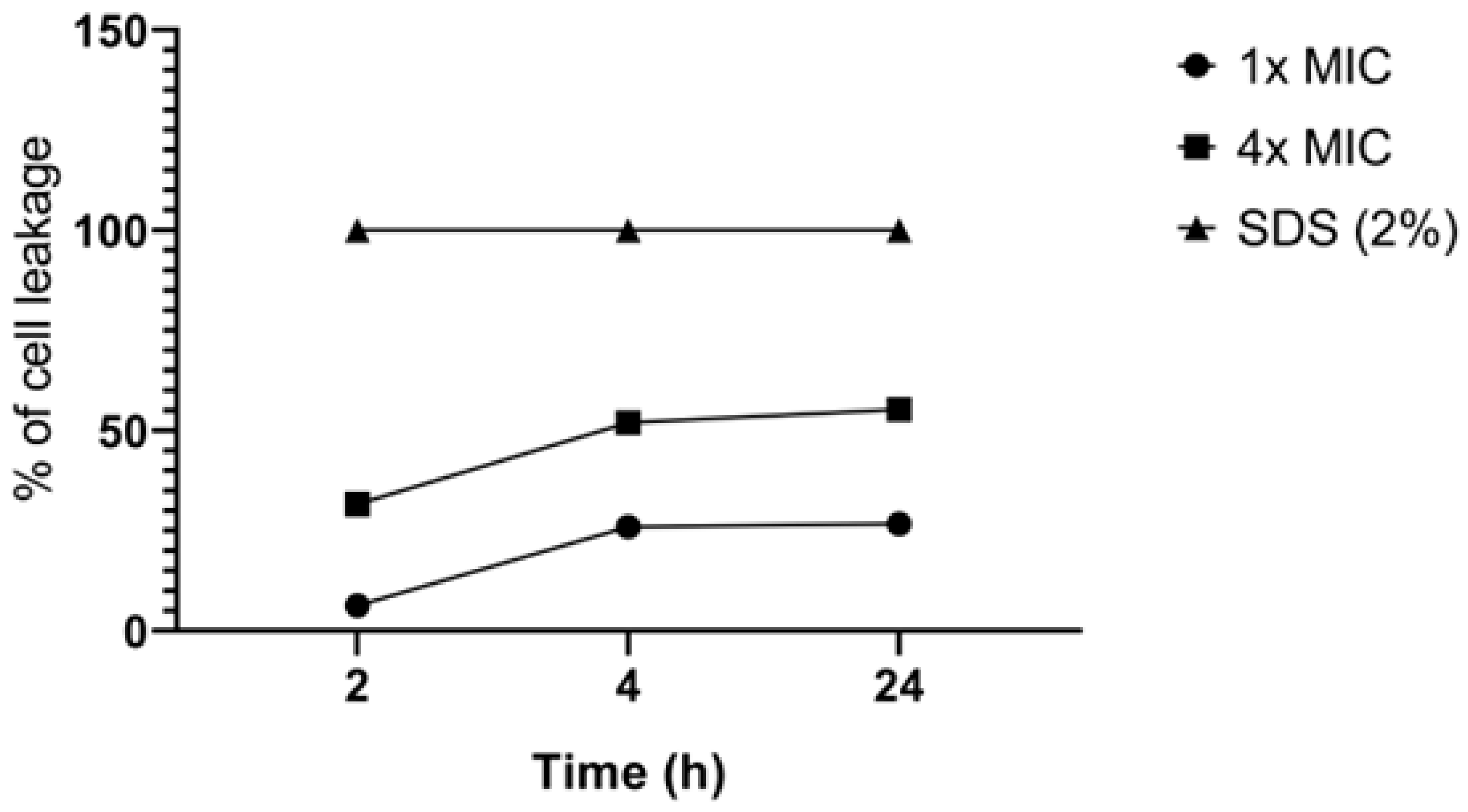
3.2. Mechanisms of Action 2,3-DBNQ
3.3. Toxicity assays
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Declaration of generative ai and ai-assisted technologies in the writing process
References
- Ozturk I, Tunçel A, Yurt F, Biyiklioglu Z, Ince M, Ocakoglu K. Antifungal photodynamic activities of phthalocyanine derivatives on Candida albicans. Photodiagnosis Photodyn Ther 2020;30. [CrossRef]
- Hendrickson JA, Hu C, Aitken SL, Beyda N. Antifungal Resistance: a Concerning Trend for the Present and Future. Curr Infect Dis Rep 2019;21:1–8. [CrossRef]
- CDC C for D and CP. Candida auris | Candida auris | Fungal Diseases | CDC. Candida auris: A Drug-Resistant Yeast That Spreads in Healthcare Facilities 2018. https://www.cdc.gov/fungal/candida-auris/ (accessed August 2, 2022).
- Forsberg K, Woodworth K, Walters M, Berkow EL, Jackson B, Chiller T, et al. Candida auris: The recent emergence of a multidrug-resistant fungal pathogen. Med Mycol 2019;57:1–12. [CrossRef]
- Chowdhary A, Prakash A, Sharma C, Kordalewska M, Kumar A, Sarma S, et al. A multicentre study of antifungal susceptibility patterns among 350 Candida auris isolates (2009–17) in India: role of the ERG11 and FKS1 genes in azole and echinocandin resistance. Journal of Antimicrobial Chemotherapy 2018;73:891–9. [CrossRef]
- Lockhart SR. Candida auris and multidrug resistance: Defining the new normal. Fungal Genetics and Biology 2019;131:103243. [CrossRef]
- Labbozzetta M, Poma P, Occhipinti C, Sajeva M, Notarbartolo M. Antitumor Effect of Glandora rosmarinifolia (Boraginaceae) Essential Oil through Inhibition of the Activity of the Topo II Enzyme in Acute Myeloid Leukemia. Molecules 2022;27:4203. [CrossRef]
- Aminin D, Polonik S. 1,4-Naphthoquinones: Some biological properties and application. Chem Pharm Bull (Tokyo) 2020;68:46–57. [CrossRef]
- Ferreira M do PSBC, Cardoso MF do C, da Silva F de C, Ferreira VF, Lima ES, Souza JVB. Antifungal activity of synthetic naphthoquinones against dermatophytes and opportunistic fungi: Preliminary mechanism-of-action tests. Ann Clin Microbiol Antimicrob 2014;13:1–6. [CrossRef]
- Clinical and Laboratory Standards Institute (CLSI). Reference method for broth dilution antifungal susceptibility testing of yeasts; CLSI-M27. 4th ed. Wayne, USA: Clinical and Laboratory Standards Institute; 2017.
- Clinical and Laboratory Standards Institute (CLSI). Reference method for broth dilution antifungal susceptibility testing of filamentous fungi; CLSI-M38. 3rd ed. Wayne, USA: Clinical and Laboratory Standards Institute; 2017.
- Bona EAM De, Pinto FG da S, Fruet TK, Jorge TCM, Moura AC de. Comparação de métodos para avaliação da atividade antimicrobiana e determinação da concentração inibitória mínima (cim) de extratos vegetais aquosos e etanólicos. Arq Inst Biol (Sao Paulo) 2014;81:218–25. [CrossRef]
- Escalante A, Gattuso M, Pérez P, Zacchino S. Evidence for the mechanism of action of the antifungal phytolaccoside B isolated from Phytolacca tetramera Hauman. J Nat Prod 2008;71:1720–5. [CrossRef]
- Ansar Ahmed S, Gogal RM, Walsh JE. A new rapid and simple non-radioactive assay to monitor and determine the proliferation of lymphocytes: an alternative to [3H]thymidine incorporation assay. J Immunol Methods 1994;170:211–24. [CrossRef]
- Meyer BN, Ferrigni NR, Putnam JE, Jacobsen LB, Nichols DE, McLaughlin JL. Brine shrimp: A convenient general bioassay for active plant constituents. Planta Med 1982;45:31–4. [CrossRef]
- Sánchez Perera LM, Mancebo Dorvigny B, Regalado Veloz AI. Inhibition of seed germination, toxicity on Artemia salina and phytochemical prospecting with from Cuban plants as indicator of antitumo activity. Macedonian Pharmaceutical Bulletin 2018;63:29–36. [CrossRef]
- Chouychai W, Thongkukiatkul A, Upatham S, Lee H, Pokethitiyook P, Kruatrachue M. Phytotoxicity assay of crop plants to phenanthrene and pyrene contaminants in acidic soil. Environ Toxicol 2007;22:597–604. [CrossRef]
- Vaezi A, Moghadaszadeh M, Nasri E, Gharibi S, Diba K, Matkowski A, et al. In vitro activity of juglone (5-hydroxy-1,4-naphthoquinone) against both fluconazole-resistant and susceptible Candida isolates. Rev Iberoam Micol 2022;39:50–3. [CrossRef]
- Yang J, Xia X, Guo M, Zhong L, Zhang X, Duan X, et al. 2-Methoxy-1,4-naphthoquinone regulated molecular alternation of Fusarium proliferatum revealed by high-dimensional biological data. RSC Adv 2022;12:15133–44. [CrossRef]
- López V, Les F. Fungal Quinones: Benzo-, Naphtho-, and Anthraquinones. Natural Secondary Metabolites, Cham: Springer International Publishing; 2023, p. 607–26. [CrossRef]
- Futuro DO, Ferreira PG, Nicoletti CD, Borba-Santos LP, Da Silva FC, Rozental S, et al. The antifungal activity of naphthoquinones: An integrative review. An Acad Bras Cienc 2018;90:1187–214. [CrossRef]
- Sánchez-Calvo JM, Barbero GR, Guerrero-Vásquez G, Durán AG, Macías M, Rodríguez-Iglesias MA, et al. Synthesis, antibacterial and antifungal activities of naphthoquinone derivatives: a structure–activity relationship study. Medicinal Chemistry Research 2016;25:1274–85. [CrossRef]
- Chen X, Li W, Chen J, Zhang X, Zhang W, Duan X, et al. Transcriptomics Integrated with Metabolomics Reveals 2-Methoxy-1, 4-Naphthoquinone-Based Carbon Dots Induced Molecular Shifts in Penicillium italicum. Journal of Fungi 2022;8:420. [CrossRef]
- Guo M, Liu J, Xu Z, Wang J, Li T, Lei H, et al. 2-Methoxy-1,4-naphthoquinone Induces Metabolic Shifts in Penicillium Digitatum Revealed by High-Dimensional Biological Data. J Agric Food Chem 2020;68:9697–706. [CrossRef]
- de Almeida PDO, dos Santos Barbosa Jobim G, dos Santos Ferreira CC, Rocha Bernardes L, Dias RB, Schlaepfer Sales CB, et al. A new synthetic antitumor naphthoquinone induces ROS-mediated apoptosis with activation of the JNK and p38 signaling pathways. Chem Biol Interact 2021;343:109444. [CrossRef]
- Mahalapbutr P, Leechaisit R, Thongnum A, Todsaporn D, Prachayasittikul V, Rungrotmongkol T, et al. Discovery of Anilino-1,4-naphthoquinones as Potent EGFR Tyrosine Kinase Inhibitors: Synthesis, Biological Evaluation, and Comprehensive Molecular Modeling. ACS Omega 2022;7:17881–93. [CrossRef]
- Lima Bezerra JJ, Johanes I, Vieira Pinheiro AA. Anticancer potential and toxicity of the genus Handroanthus Mattos (Bignoniaceae): A systematic review. Toxicon 2022;217:131–42. [CrossRef]
- Emadi A, Ross AE, Cowan KM, Fortenberry YM, Vuica-Ross M. A Chemical Genetic Screen for Modulators of Asymmetrical 2,2′-Dimeric Naphthoquinones Cytotoxicity in Yeast. PLoS One 2010;5:e10846. [CrossRef]
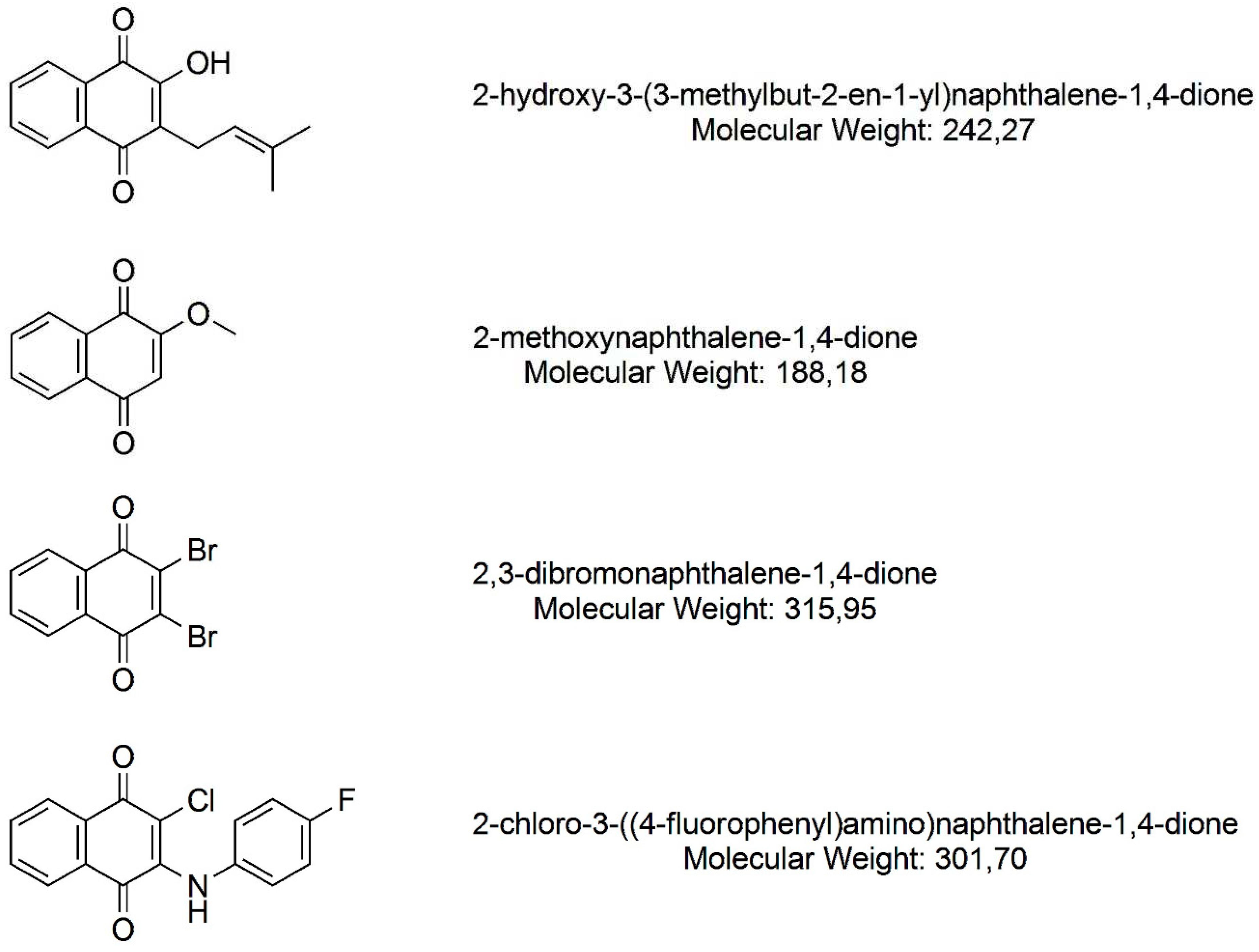
| Microorganisms | Species | Strain designation |
|---|---|---|
| Yeasts | Candida albicans | ATCC 60193 |
| Candida albicans | ATCC 36323 | |
| Candida krusei | ATCC 34135 | |
| Candida tropicalis | ATCC 13803 | |
| Candida glabrata | ATCC 2001 | |
| Candida parapsilosis | ATCC 22019 | |
| Subcutaneous pathogenic fungi | Sporothrix brasiliensis | CFP 00551 |
| Sporothrix schenckii | CFP 00746 | |
| Dermatophytes | Trichophyton mentagrophytes | ATCC 9533 |
| Trichophyton rubrum | ATCC 28189 | |
| Opportunistic filamentous fungi | Fusarium oxysporum | LM 5643 |
| Microorganisms | Strain designation | lapachol | 2-MNQ | 2,3-DNBQ | 2-ClFNQ | Amphotericin B | Fluconazol* |
|---|---|---|---|---|---|---|---|
| Minimum Inhibitory Concentration (MIC) µg/mL | |||||||
| Candida albicans | ATCC 60193 | >800 | 25 | 3,125 | 200 | 2 | 2 |
| ATCC 36232 | >800 | 12,5 | <1,56 | 100 | 2 | 2 | |
| Candida krusei | ATCC 34135 | >800 | 12,5 | <1,56 | 50 | 4 | 16 |
| Candida tropicalis | ATCC 13803 | >800 | 12,5 | 3,125 | 200 | 4 | 2 |
| Candida glabrata | ATCC 2001 | >800 | 25 | 6,25 | 400 | 4 | 2 |
| Candida parapsilosis | ATCC 22019 | >800 | 6,25 | <1,56 | 100 | 2 | 2 |
| Sporothrix brasiliensis | CFP 00551 | >800 | 50 | 3,125 | 25 | 2 | 64 |
| Sporothrix schenckii | CFP 00746 | >800 | 25 | 3,125 | 12,5 | 8 | 64 |
| Trichophyton mentagrophytes | ATCC 9533 | >800 | 6,25 | <1,56 | 25 | 8 | 16 |
| Trichophyton rubrum | ATCC 28189 | >800 | <1,56 | <1,56 | 50 | 2 | 2 |
| Fusarium oxysporum | LM 5634 | >800 | 50 | <1,56 | 100 | 16 | 32 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




