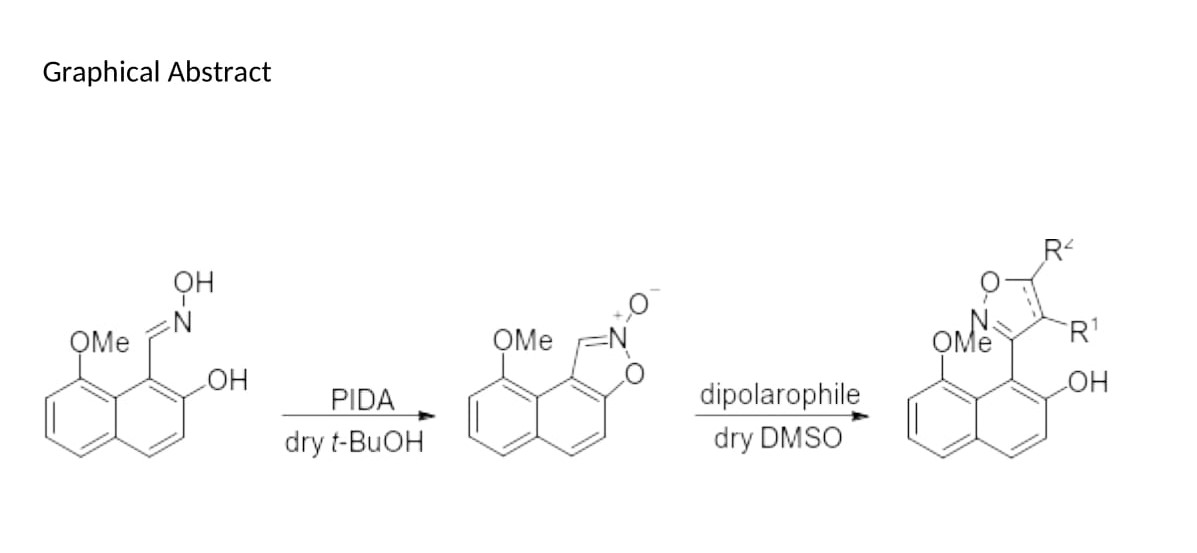
2. Results & Discussion
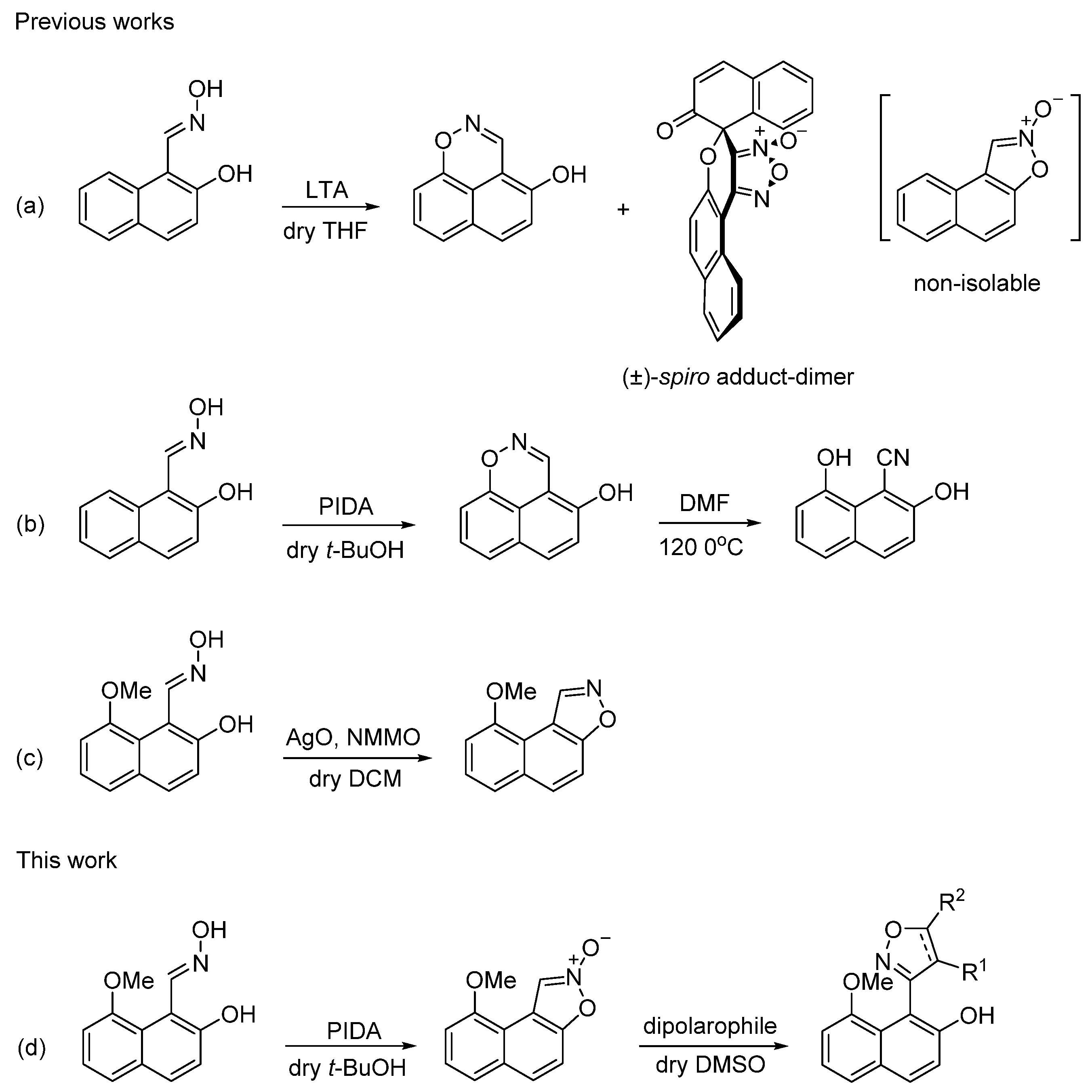
Following up our previous observations about the instability of naphtho [1,2-
d]isoxazole 2-oxide [
49] (
Scheme 1a) and the effect of OMe group at the C-8 position of (
E)-2-hydroxy-1-naphthaldehyde oxime on its selective oxidation to 9-methoxynaphtho [1,2-
d]isoxazole [
62] (
Scheme 1c) we became interested in investigating the outcome of a PIDA oxidation of (
E)-2-hydroxy-8-methoxy-1-naphthaldehyde oxime, a reagent used successfully for the oxidative cyclization of 2-hydroxyaryl ketoximes into 3-arylbenzo[
d]isoxazole 2-oxides [
48]. The key structure in our recent work (
Scheme 1c), 2-hydroxy-8-methoxy-1-naphthaldehyde (
6) was prepared from 1-naphthaldehyde by
o- and
peri-methoxylation from Pd(OAc)
2, K
2S
2O
8 and 3-(trifluoromethyl)aniline in a closed vessel, that provided 2,8-dimethoxy-1-naphthaldehyde in 30% yield. The latter was selectively demethylated by AlCl
3 to (
6) in 15% yield [
62]. The low overall yield (4.5%) of this reaction and the inconvenience of using a sealed vessel, steered us towards an alternative route to (
6). The high yielding (80%) synthesis of naphtho [1,8-
de][
1,
2]oxazin-4-ol (
1) followed by its equally high yielding (80%) ring opening to 2,8-dihydroxy-1-naphthonitrile (
Scheme 1b), provided attractive starting materials that we decided to use towards our target (
6).
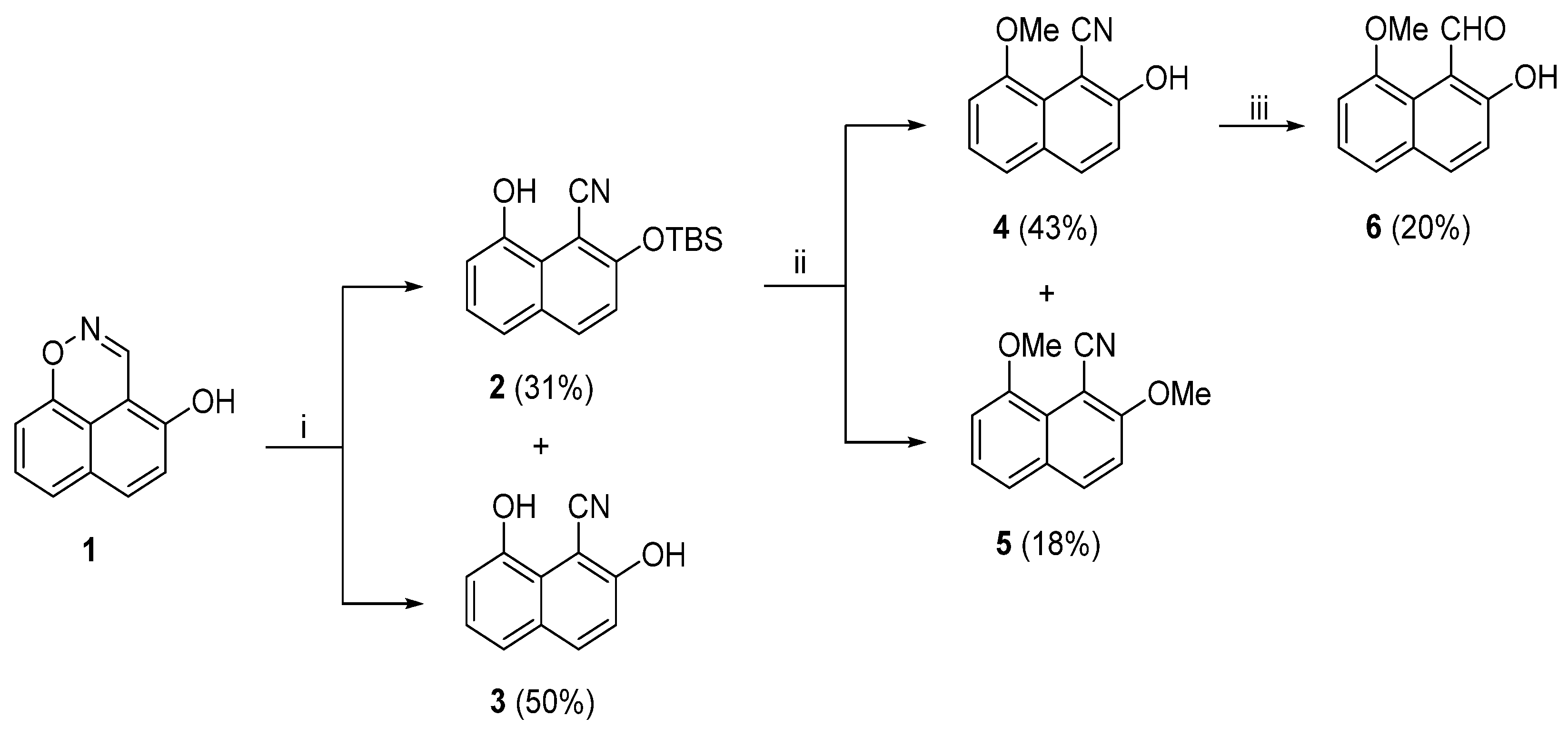
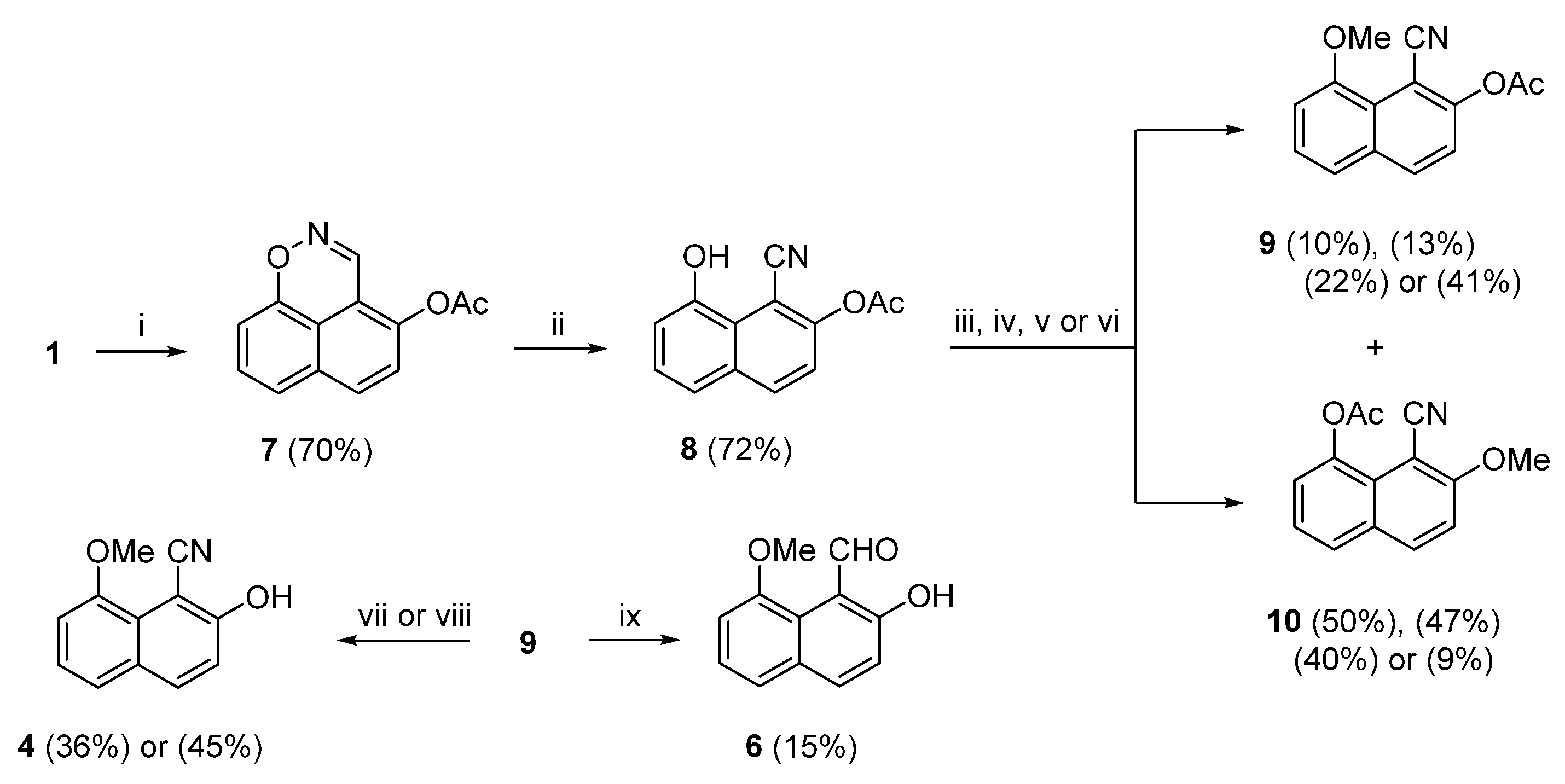
At the start of our investigation, we reacted (
1) with TBSCl and imidazole in dry DMF at room temperature, reaction conditions used by Bencivenni and co-workers [
63] to silylate 1,7-naphthalenediol. After the disappearance of the starting material spot and appearance of a new spot on TLC, the reaction was heated at 120
oC for a short period of time, according to the reaction conditions used in our previous report (
Scheme 1b) [
10]. The outcome was the expected ring-opened 2-TBS protected 1-naphthonitrile (
2) and the unprotected 1-naphthonitrile (
3), in 31% and 50% yields, respectively. Compound (
3) obviously resulted from (
2) by cleavage of its TBS-group. After separating by column chromatography, (
2) was methylated using MeI and K
2CO
3 in dry acetone, reaction conditions used by Bencivenni and co-workers [
63], to methylate the OH group and desilylate the OTBS group of 7-[(
tert-butyldimethylsilyl)oxy]naphthalen-1-ol. Our reaction was over after 1 hour stirring at room temperature. Column chromatography furnished two compounds identified as selectively methylated nitrile (
4) in a 43% yield and dimethylated nitrile (
5) in an 18% yield. Nitrile (
4) was then reduced to aldehyde (
6) with DIBAL in toluene, reaction conditions used for the reduction of 6-(methylamino)-2-naphthonitrile to 6-(methylamino)-2-naphthaldehyde [
64], at 0
oC and then room temperature, in our case. The overall yield of (
6) from (
1) by the four step route, as described in
Scheme 2, was only 2.8%.
This low overall yield probably due to byproducts (
3) and (
5) and to the low yield of the last step, turned our attention to a four-step route to (
6) from (
1) (
Scheme 3). Thus, according to an old published procedure [
8] , (
1) was acetylated with acetic anhydride to afford naphtho [1,8-
de][
1,
2]oxazin-4-yl acetate (
7) in 70% yield. Ring opening of (
7) in DMF at 120
oC, reaction conditions used previously [
10], afforded 1-naphthonitrile (
8) in 72% yield. Next, we tried to methylate the OH group of (
8), under various reaction conditions, without cleaving the acetyl protective group. Dimethyl sulphate with K
2CO
3, in dry acetone, was first used, according to an earlier report [
65]. The two products obtained were separated by column chromatography to afford the 8-methoxy nitrile (
9) and 8-acetoxy-1-acetoxy nitrile (
10), in 10% and 50% yields, respectively. The origin of (
10) may well have been accounted from an intermolecular acetylation between two molecules of (
8), followed by methylation of the resulting 8-acetoxy-1-cyanonaphthalen-2-olate. The methylation outcome of (
8) remained the same, under different conditions but with varying yields of (
9) and (
10). Therefore, methylation of (
8) was first tried with MeI and K
2CO
3 in dry THF at room temperature, a second attempt was with 50 mg of (
8), MeI, NaH and 5 mL dry THF at room temperature and a final one with 50 mg of (
8), MeI, NaH but this time 50 mL dry THF at room temperature, that afforded (
9) and (
10), in 13% and 47%, 22% and 40%, and, 41% and 9%, respectively. It was interesting to note that the solvent effect in the last two attempts apparently favored (
9) (41% yield) compared to (
10) (9% yield). Moreover, the MeO group in (
9) was properly placed for further deprotection and reduction to the target (
6). Thus, reacting (
9) with either DIBAL (1.2 M in toluene) in dry THF at −78
oC [
64] or with PtO
2 in an equal volume of HCO
2H/H
2O at 55−60
oC [
66] , the deprotected nitrile (
4) was obtained in in 36% and 45% yields, respectively. The reduction of (
9) to (
6) proved difficult, either under the standard DIBAL conditions (1.2 M in toluene) [
64] or an attempted modification (initiating the reaction at 0
oC and allowing a long period at room temperature), it gave disappointingly a 15% yield and an overall 3.1% yield from (
1) (
Scheme 3), marginally higher than that of
Scheme 2.
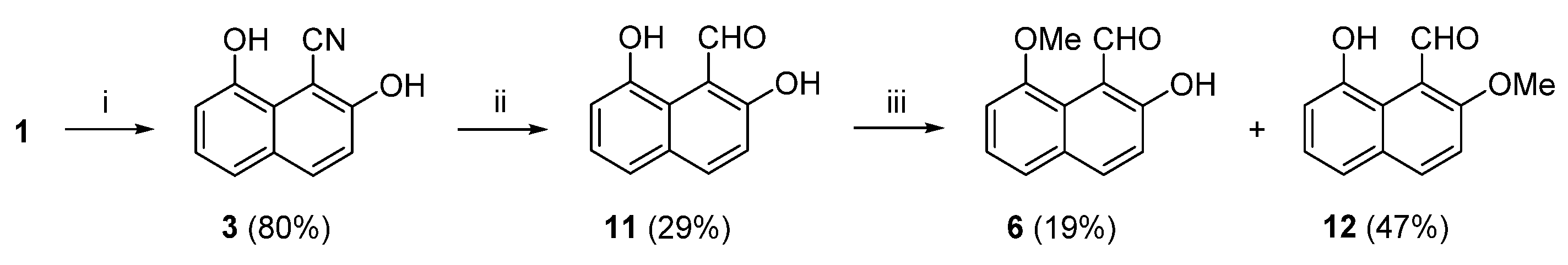
In the next attempt to synthesize (
6) from (
1) we planned the simpler three-step route shown in
Scheme 4. We had wrongly anticipated that the 6-membered intramolecular hydrogen bond in 1-naphthaldehyde (
11) between the CHO oxygen atom and the OH hydrogen atom, would be strong enough to survive in DMF and thus encourage selective methylation at the
peri position. The synthetic route started from (
1) with heating in DMF at 120
oC according to our published procedure [
10]. Reduction of the CN group in (
3) with calcium hypophosphite in the presence of base and nickel(II) acetate tetrahydrate led to a disappointing 29% yield of 1-naphthaldehyde (
11). Using these reactants, Estelle Métay, Marc Lemaire and co-workers [
67] reduced 1-naphthonitrile to 1-naphthaldehyde in 85% yield while aryl nitriles bearing OH groups are tolerant to these reaction conditions. In the last step, methylation of (
11) took place in dry DMF and at room temperature in the presence of MeI and K
2CO
3 to afford, after column chromatography, the target compound (
6) and side product (
12) in 19% and 47% yields, respectively. These reaction conditions have been used to methylate 2-chloro-8-hydroxy-1-naphthaldehyde to 2-chloro-8-methoxy-1-naphthaldehyde, in very good yield [
15]. The formation of product (
12) implies that in a DMF solution of (
11) there could exist an intramolecular pseudo hydrogen bond between the CHO group and the
peri OH group, stronger than that between the CHO group and the
ortho OH group. The outcome of this effort was a 4.4% overall yield of (
6), more or less as in the route described in
Scheme 3.
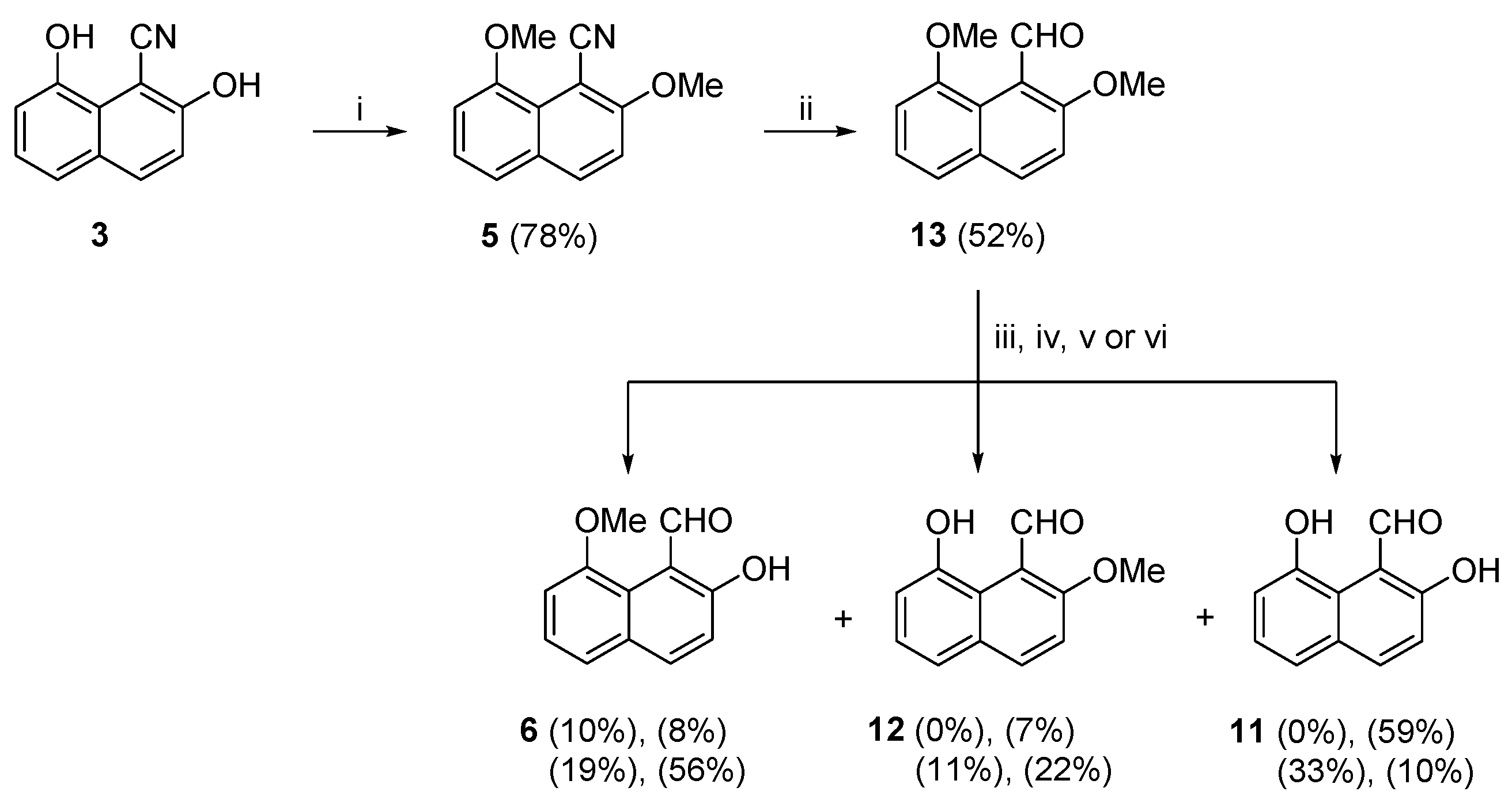
In the fourth attempt towards synthesizing (
6) from (
1), we planned to alkylate (
3), reduce the resultant nitrile to the corresponding aldehyde and then selectively demethylate to produce the target compound (
6) (
Scheme 5). The synthetic route began by the dimethylation of (
3) with MeI and Na
2CO
3, in aqueous acetone, under mild heating [
63], that gave the dimethoxy nitrile (
5) in a satisfying 78% yield. 1-Naphthonitrile (
5) was reduced to the aldehyde (
13) in moderate yield, by a modification of the standard conditions of DIBAL (1.2 M in toluene) [
64], as described earlier. Aldehyde (
13) was then subjected to, what we hoped to be, selective deprotection of the 2-OMe group to the target compound (
6) in a useful yield. In the first selective demethylation attempt of (
13) to (
6), MgBr
2.etherate and KI in MeCN were heated in a closed vessel, by applying a reported method [
68]. The reaction, after column chromatography, gave (
6) as the only product albeit in very low yield. The next demethylation attempt of (
13) entailed the use of BBr
3 in DCM at room temperature for 18 hours, which, after column chromatography, gave products (
6), (
12) and (
11) in 8%, 7% and 59%, respectively. These reaction conditions have been used to demethylate 1,2-
bis-(4-methoxyphenyl)propane to 1,2-
bis-(4-hydroxy-phenyl)propane [
69]. The selective demethylation was again repeated with BBr
3 in DCM, only this time at room temperature for 1 hour and at −15
oC for 1 hour, respectively. In our third and fourth reaction trials on the selective demethylation of compound (
13), BBr
3 in DCM was used again but this time at room temperature for 1 hour and at −15
oC for 1 hour, respectively. The yields of products (
6), (
12) and (
11) were obtained in 19%, 11% and 33%, and, 56%, 22% and 10% yields, respectively. The results are not clear-cut but we can assume that at low temperature, in compound (
13), one of the lone pairs of the
peri OMe group is intramolecularly engaged in a pseudo hydrogen bond with the CHO group, for most of the time. This H bonding interaction engages the
peri OMe group, consequently allowing a relatively easier demethylation of its more available
o-OMe counterpart. The moderate yields of the last two steps of this reaction sequence towards (
6) diminished its value, despite an overall increase of the yield to 18.2%.
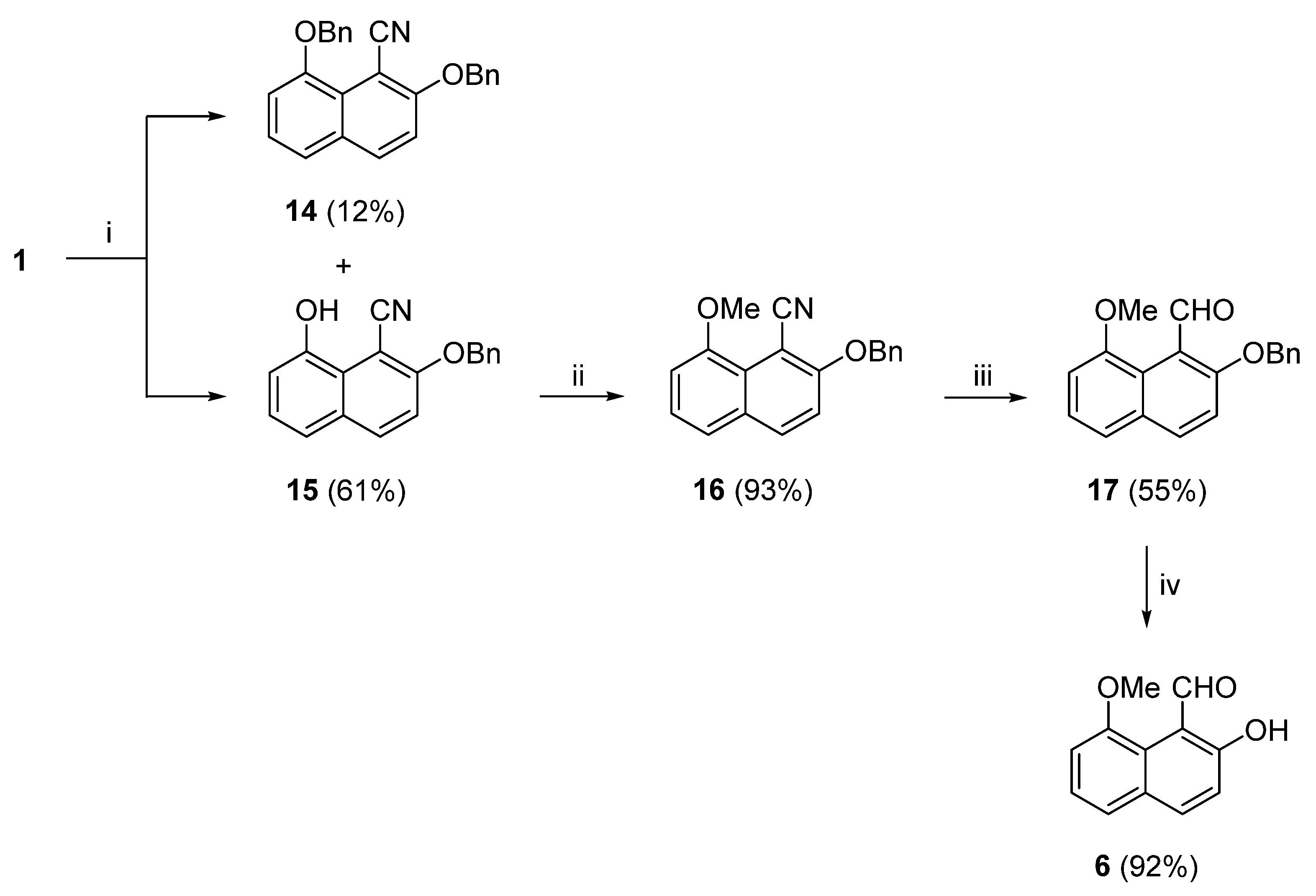
In the final attempt to synthesize (
6) as efficiently as possible from (
1), we decided to start with an
O-benzylation of (
1), and then ring open the resulting 4-(benzyloxy)naphtho [1,8-
de][
1,
2]oxazine (by heating in DMF) to the targeted precursor 2-(benzyloxy)-8-hydroxy-1-naphthonitrile (
15) (
Scheme 6). Further, we envisaged methylation of the
peri OH group, reduction of nitrile to aldehyde and debenzylation of the OBn group. For the benzylation step, we slightly modified the reaction conditions applied by Luo and Zheng and co-workers [
70]. to convert 2-hydroxy-1-naphthaldehyde to 2-(benzyloxy)-1-naphthaldehyde. Starting from oxazine (
1) we used benzyl bromide, instead of benzyl chloride, in the presence of K
2CO
3 and KI and stirred the reaction in acetone, at room temperature for 18 hours, instead of under reflux. TLC analysis showed two new spots and no starting material. Column chromatography separation and NMR analysis identified these two compounds as (
14) and (
15) in 12% and 61% yields, respectively. To our surprise, the intermediate 4-(benzyloxy)naphtho [1,8-
de][
1,
2]oxazine was not detected. Apparently, the oxazine suffered ring opening in the presence of the base. Indeed, this result was experimentally verified by stirring (
1) with K
2CO
3 and KI in acetone to find out that after 4 hours 2,8-dihydroxy-1-naphthonitrile was obtained as a single product. It may be argued that benzylation could take place either sequentially, first on (
1) and then on ring-opened (
14) to (
15) or on the ring-opened (
3). The higher yield of (
15) points to the former process. In the second step of this reaction sequence, 2-(benzyloxy)-8-hydroxy-1-naphthonitrile (
15) was subjected to methylation by MeI and K
2CO
3 in dry acetone under reflux [
63] to afford the 8-OMe derivative (
16) in excellent yield. Reduction of the nitrile group of (
16) was accomplished by a modification of the standard conditions of DIBAL (1.2 M in toluene) [
64] (see earlier comments) 1-naphthaldehyde (
17) was, thus, obtained, in a moderate 55% yield. In the last step of this reaction sequence, debenzylation took place by H
2 and Pd/C as catalyst, according to the procedure used for the deprotection of (
R)-3-(4-(benzyloxy)-3-fluorophenyl)-5-(hydroxymethyl)oxazolidin-2-one to (
R)-3-(3-fluoro-4-hydroxyphenyl)-5-(hydroxymethyl)-oxazolidin-2-one [
71]. Catalytic hydrogenation has been used the first time for the deprotection of a 2-(benzyloxy)naphthalene. The yield of 1-naphthaldehyde (
6) was excellent. The overall yield of target compound (
6) from (
1) by this four step route, as described in
Scheme 6, was increased to 28.7%, 10.5% higher than the method of
Scheme 5.
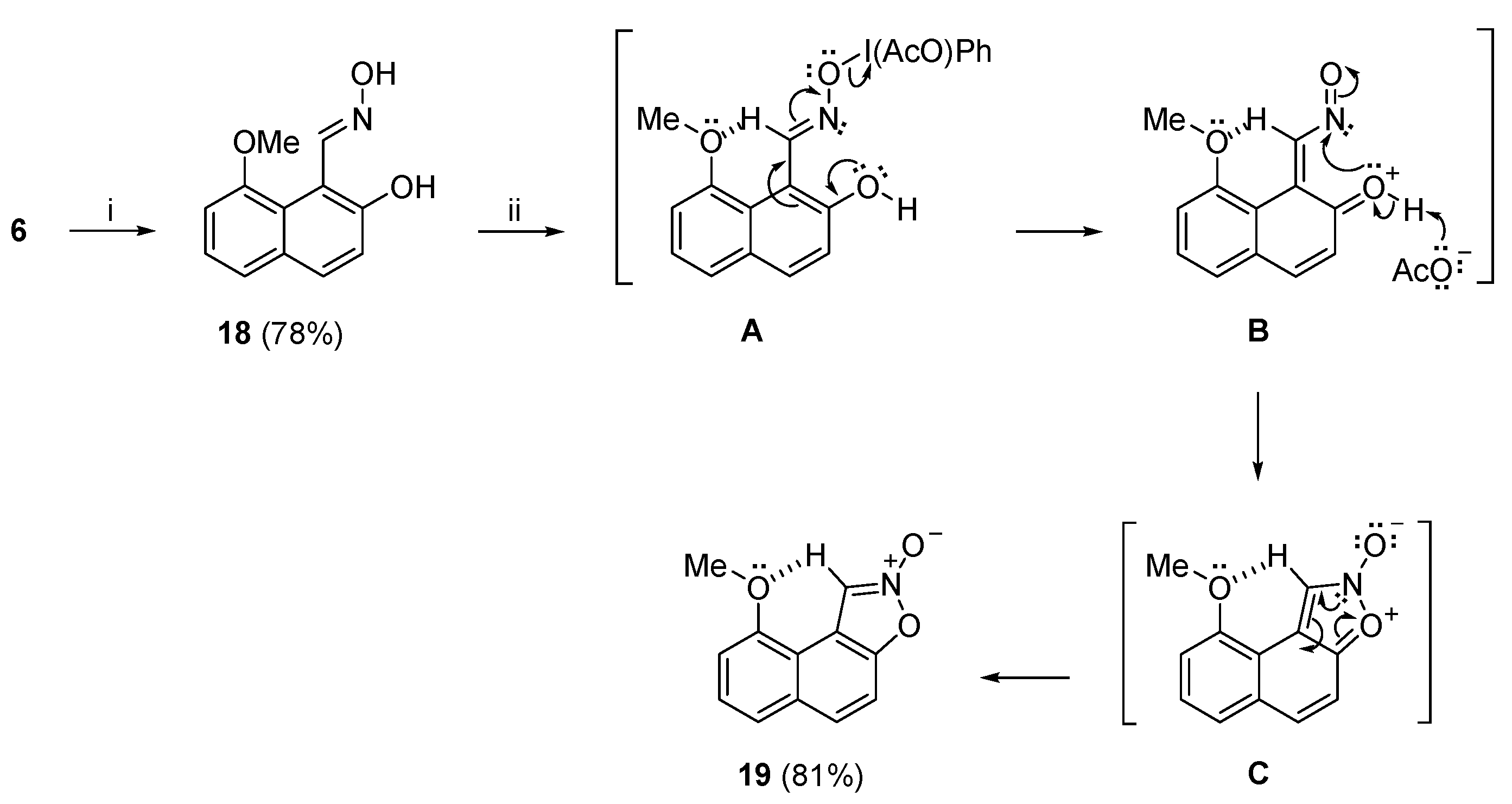
Having established a viable route from (
1) to target compound (
6), we moved forward to our next goal, that is the synthesis of oxime (
18) and its reaction with PIDA. Aldehyde (
6) was condensed with hydroxylamine hydrochloride in the presence of base in MeOH, first at 0
oC and then at room temperature, according to our article by Tzeli, Tsoungas and co-workers [
62], to afford (
E)-oxime (
18) in 78% yield. The latter compound was then subjected to oxidation with PIDA in non-nucleophilic dry
t-BuOH at room temperature, to afford isoxazole-2-oxide (
19) in very good yield (
Scheme 7). We propose that in the first step of the reaction, the oxygen atom of the oxime group acted as a nucleophile towards electrophilic iodine atom of PIDA and displaced acetate ion to form organoiodo complex
A. The O−I bond was much weaker than the N−O bond and therefore spontaneous cleavage of the former bond lead to the formation of
o-naphthoquinone nitrosomethide intermediate
B. The latter underwent 6π-electrocyclization to the non-aromatic dipolar naphthoisoxazole-
N-oxide
C which aromatized to sTable 9-methoxynaphtho [1,2-
d]isoxazole 2-oxide (
19), in very good yield. The stability of (
19) was apparently due to the intramolecular pseudo hydrogen bonding between the OMe
peri substituent and the sp
2 C−1 hydrogen atom, that impeded ring opening of the isoxazole ring. That could well serve as a rationale for the non-isolable naphtho [1,2-
d]isoxazole 2-oxide, lacking this particular stabilizing factor (
Scheme 1a) [
49].
When we recorded the
1H NMR spectrum of isoxazole 2-oxide (
19) in DMSO-
d6, that took only a few minutes, we found that the peaks in the spectrum corresponded to the structure of this compound. Next day we set forth to record the
13C NMR spectrum of the compound. As the normal practice is, in these cases, we recorded again the
1H NMR spectrum of the sample before setting up the machine to record its
13C NMR spectrum overnight. To our surprise, the initial
1H NMR spectrum of isoxazole 2-oxide (
19) was different from this spectrum which we found later that it was nitrile oxide (
20) [see
Supplementary Materials Figure S59 for superimposed and
Figure S60 for stacked
1H NMR spectra of (
19) and (
20)]. This novel isomerization of (
19) to (
20) was also detected by
1H NMR spectroscopy that was measured by a time-course plot (see
Supplementary Materials Figure S61). We recorded the
1H and
13C NMR spectra of 2-oxide (
19) in CDCl
3 and confirmed that the compound is stable in this solvent even after 16 hours. We stirred a sample of isoxazole 2-oxide (
19) in DMSO-
d6 and after 6 hours recorded the
1H and
13C NMR spectra. As with the previously recorded NMR spectra of nitrile oxide (
20), in the
1H NMR spectrum in DMSO-
d6, the OH proton at high field was not visible and there was a range of 7.90–7.02 ppm with a total of 5 protons belonging to the naphthalen-2-ol ring. The characteristic singlet of the methyl group was found at 3.92 ppm. The
13C NMR spectrum in DMSO-
d6 showed 11 signals. According to Koyama, Takata and co-workers [
72] quaternary signals of nitrile groups usually turn up under the residual DMSO signal so that for nitrile oxide (
20) the total number of carbon atoms was 12, as expected. High-resolution mass spectrometry analysis confirmed the expected molecular ion at m/z = 216.0661 [M+H]
+ (ESI) that took up a proton and was calculated for C
12H
10NO
3 m/z = 216.0661. In the mass spectrum of this compound there was no peak at m/z = 431.1243 [M+H]
+ corresponding to the dimerization of (
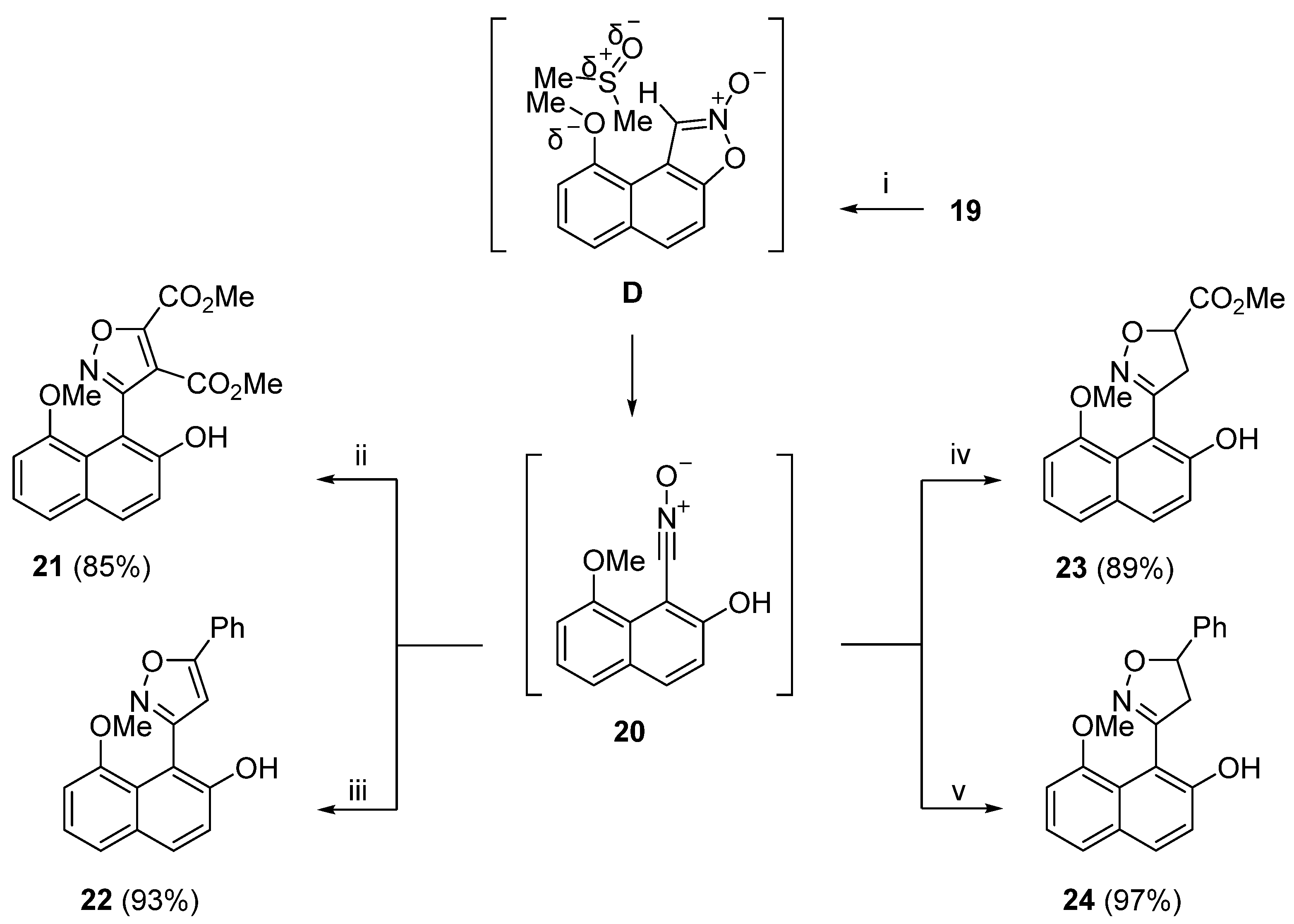
20) to 3,4-bis(2-hydroxy-8-methoxynaphthalen-1-yl)-1,2,5-oxadiazole 2-oxide. At this point we did not know whether compound (
20) was the isocyanate isomer of the nitrile oxide, namely, 1-isocyanato-8-methoxynaphthalen-2-ol. Confirmation that (
20) was the nitrile oxide came from 1,3-dipolar cycloaddition reactions of in situ generated nitrile oxide (
20) with various dipolarophiles, that produced substituted isoxazoles (
Scheme 8). The reactions took place by dissolving isoxazole 2-oxide (
19) in DMSO under an atmosphere of nitrogen, addition of the appropriate dipolarophile (DMAD, phenylacetylene, methyl acrylate or styrene) and stirring at room temperature for 18 hours. Substituted isoxazoles (
21) to (
24) were produced in 85%, 93%, 89% and 97% yields, respectively. It was suggested that when isoxazole 2-oxide (
19) was dissolved in DMSO, solvated species
D could have disrupted the intramolecular pseudo hydrogen bond between the MeO group and the C-1 hydrogen atom, allowing for the ring opening of the isoxazole ring to the nitrile oxide moiety (
20) which then underwent 1,3-dipolar cycloaddition with the dipolarophiles (
Scheme 8).
3. Materials and Methods
Organic solutions were concentrated by rotary evaporation at 40 °C under 15 Torr. Melting points were taken on a Büchi 510 apparatus and are uncorrected. 1H and 13C NMR spectra were measured in CDCl3 or DMSO-d6 on a 400 MHz Brüker Avance spectrometer. 1H chemical shifts are reported in ppm from an internal standard TMS, residual CHCl3 (7.26 ppm) or DMSO (2.50 ppm). 13C NMR chemical shifts are reported in ppm from an internal standard TMS, residual CHCl3 (77.00 ppm) or DMSO (39.43 ppm). High resolution ESI mass spectra were measured on a Thermo Fisher Scientific Orbitrap XL system. IR spectra were acquired on an Agilent Cary 630 FTIR spectrophotometer as solids and are reported in wave numbers (cm−1). Analytical thin layer chromatography (TLC) was performed with TLC plates (Merck 70-230 mesh silica gel). TLC visualization took place under a 254 nm UV light source. Purification of reaction products was generally done by flash column chromatography using Carlo Erba Reactifs-SDS silica gel 60. Solvents, reagents, and catalysts were used as received from the manufacturers (Acros, Aldrich, Alfa-Aesar, Fluka and Merck) except for DCM, EtOAc and hexane that were dried and purified according to recommended procedures.
Synthesis of 2-[(tert-butyldimethylsilyl)oxy]-8-hydroxy-1-naphthonitrile (2) and 2,8-dihydroxy-1-naphthonitrile (3)
To a solution of compound (1) (500 mg, 2.2 mmol, 1 equiv) in dry DMF (15 mL), under an atmosphere of N2, was added imidazole (374 mg, 5.5 mmol, 2.5 equiv) and TBSCl (398 mg, 2.64 mmol, 1.2 equiv) and the reaction was left stirring at room temperature for 2 h (TLC analysis had shown the absence of the starting material spot and the presence of a new spot). The reaction mixture was then heated at 120 oC for 0.5 h. TLC examination revealed the absence of the starting material spot and the presence of two new spots. To the cooled reaction mixture water (100 mL) was added and extracted with EtOAc (3 × 20 mL) and the combined organic extracts washed with brine (20 mL), dried over anhydrous Na2SO4, and concentrated in vacuum. The acquired crude residue was purified by flash column chromatography (25% EtOAc in hexane) to give title compounds (2) and (3).
Compound (2): (204 mg, 31%) as a yellow oil; Rf = 0.61 (25% EtOAc in hexane); 1H NMR (400 MHz, CDCl3) δ: a broad singlet corresponding to OH is not visible, 7.76 (d, J = 8.9 Hz, 1H), 7.26 (d, J = 8.2 Hz, 1H), 7.18 (t, J = 7.5 Hz, 1H), 7.00–6.90 (m, 2H), 1.01 (s, 9H), 0.25 (s, 6H); 13C NMR (100.6 MHz, CDCl3) δ: 159.85, 150.81, 134.82, 130.30, 125.70, 123.58, 120.95, 120.16, 118.59, 113.10, 95.08, 25.67, 18.33; IR (solid): 3161, 3060, 2955, 2922, 2855, 2215, 2118, 1729 cm−1; HRMS (ESI): m/z [M−H]− calcd. for C17H21NO2Si: 298.1263, found: 298.1264.
Compound (
3): (203 mg, 50%) as a yellow solid, m.p. = 166–167
oC (lit. [
10], m.p. = 167–168
oC); R
f = 0.1 (20% ethyl acetate in hexane);
1H NMR (400 MHz, DMSO-
d6) δ: 11.24 (s, 1H), 10.31 (s, 1H), 7.93 (d, J = 9.0 Hz, 1H), 7.32 (d, J = 7.9 Hz, 1H), 7.25–7.15 (m, 2H), 6.92 (dd, J = 7.6, 1.1 Hz, 1H) (in agreement with the
1H NMR data that were previously reported for this compound) [
10] .
Synthesis of 2-hydroxy-8-methoxy-1-naphthonitrile (4) and 2,8-dimethoxy-1-naphthonitrile (5)
To a solution of compound (2) (100 mg, 0.334 mmol, 1 equiv) in dry acetone (10 mL), under an atmosphere of N2, was added oven-dried K2CO3 (50 mg, 0.367 mmol, 1.1 equiv) and MeI (70 mg, 0.501 mmol, 1.5 equiv) and the reaction was left stirring at room temperature for 1 h (TLC analysis had shown complete conversion of the starting material and the presence of two new spots). Water (20 mL) was added, reaction mixture extracted with EtOAc (3 × 10 mL) and the combined organic extracts were washed with brine (10 mL), dried over anhydrous Na2SO4, and concentrated under vacuum. The acquired crude residue was purified by flash column chromatography (11% EtOAc in hexane) to give title compounds (4) and (5).
Compound (4): (28.6 mg, 43%) as a yellow solid, m.p. = 172−174 oC; Rf = 0.09 (20% EtOAc in hexane); 1H NMR (400 MHz, CDCl3) δ: a broad singlet corresponding to OH is not visible, 7.90 (d, J = 9.0 Hz, 1H), 7.42–7.33 (m, 2H), 7.22 (d, J = 9.0 Hz, 1H), 6.97 (d, J = 7.3 Hz, 1H), 4.05 (s, 3H); 13C NMR (100.6 MHz, CDCl3) δ: 160.44, 153.86, 135.17, 129.76, 125.50, 123.62, 121.22, 117.51, 117.41, 107.86, 89.31, 55.88; IR (solid): 3072, 2920, 2848, 2216, 1705, 1600, 1513 cm−1; HRMS (ESI): m/z [M+Na]+ calcd. for C12H9NO3Na: 222.0531, found: 222.0528.
Compound (5): (12.8 mg, 18%) as a colorless amorphous solid (hexane), m.p. = 147−149 oC; Rf = 0.21 (20% EtOAc in hexane); 1H NMR (400 MHz, CDCl3) δ: 7.83 (d, J = 9.2 Hz, 1H), 7.25 (d, J = 8.2 Hz, 1H), 7.19 (t, J = 7.9 Hz, 1H), 7.13 (d, J = 9.3 Hz, 1H), 6.80 (dd, J = 7.5, 1.2 Hz, 1H), 3.93 (s, 3H), 3.89 (s, 3H); 13C NMR (100.6 MHz, CDCl3) δ: 162.94, 154.29, 134.80, 129.71, 125.23, 123.75, 121.06, 116.96, 112.37, 107.72, 92.62, 56.85, 55.82; IR (solid): 2921, 2848, 2218, 2091, 1915, 1826, 1593 cm−1; HRMS (ESI): m/z [M+H]+ calcd. for C13H12NO2: 214.0863, found: 214.0858.
Synthesis of 2-hydroxy-8-methoxy-1-naphthaldehyde (6)
To a stirred solution of (
4) (200 mg, 1 mmol,) in dry toluene (10 mL), under an atmosphere of N
2, at 0
oC, was added DIBAL (1.67 mL of a 1.2 M in toluene, 2 mmol) and the reaction mixture was left stirring for 0.5 h and then for 18 h at room temperature (TLC analysis had shown absence of starting material spot and presence of a new spot). The solvent was removed under reduced pressure, water (20 mL) was carefully added to the residue and the resulting mixture cooled to 0
oC followed by dropwise addition of 1 M aqueous HCl until pH = 1. The aqueous solution was extracted with EtOAc (3 × 10 mL), the combined organic extracts washed with brine (20 mL), dried over anhydrous Na
2SO
4 and concentrated under vacuum. The acquired crude residue was purified by flash column chromatography (17% EtOAc in hexane) to give the title compound (40 mg, 20%) as a yellow solid, m.p. = 66–69
oC (lit. [
62], m.p. = 68–70
oC); R
f = 0.54 (20% ethyl acetate in hexane);
1H NMR (400 MHz, CDCl
3) δ: 14.15 (s, 1H), 11.22 (s, 1H), 7.87 (d, J = 9.0 Hz, 1H), 7.38 (dd, J = 8.0, 1.3 Hz, 1H), 7.31 (t, J = 7.9 Hz, 1H), 7.10 (d, J = 9.0 Hz, 1H), 7.04 (dd, J = 7.8, 1.2 Hz, 1H), 3.99 (s, 3H) (in agreement with the
1H NMR data that were previously reported for this compound) [
62].
Synthesis of Naphtho[1,8-de][1,2]oxazin-4-yl acetate (
7)
A solution of compound (1) (500 mg, 2.7 mmol) in freshly distilled acetic anhydride (10 mL), under an atmosphere on N2, was stirred at room temperature for 18 h (TLC had shown absence of starting material spot and presence of one new spot). Ice water (30 mL) was added and the reaction mixture was stirred for 0.5 h at room temperature. The reaction mixture was then extracted with EtOAc (3 x 15 mL), the combined organic extracts were washed with brine (10 mL), dried over anhydrous Na2SO4 and concentrated under vacuum. The acquired crude residue was purified by flash column chromatography (17% EtOAc in hexane) to give the title compound (429 mg, 70%) as a brown oil; Rf = 0.05 (20% EtOAc in hexane); 1H NMR (400 MHz, DMSO-d6) δ: 8.71 (s, 1H), 7.99 (d, J = 9.1 Hz, 1H), 7.58–7.51 (m, 2H), 7.41 (d, J = 9.1 Hz, 1H), 7.06–6.97 (m, 1H), 2.33 (s, 3H); 13C NMR (100.6 MHz, DMSO-d6) δ: 169.25, 166.97, 151.25, 144.24, 140.41, 130.81, 128.31, 124.15, 119.87, 119.22, 107.86, 105.71, 20.71; IR (solid): 2924, 2682, 2217, 1926, 1749, 1510 cm−1; HRMS (ESI): m/z [M+Na]+ calcd. for C13H9NO3Na: 250.0480, found: 250.0477.
Synthesis of 1-cyano-8-hydroxynaphthalen-2-yl acetate (8)
A solution of compound (7) (400 mg, 2.16 mmol) in DMF (8 mL) was heated at 120 °C for 45 min. (TLC analysis had shown absence of starting material spot and presence of a new spot). Water (30 mL) was added and the reaction mixture was extracted with Et2O (3 × 15 mL). The combined organic extracts were dried (Na2SO4) and the solvent was removed under vacuum. The residue was purified by flash column chromatography (50% EtOAc in hexane) to give the title compound (267 mg, 72 %) as a brown solid; m.p. = 165–167 oC; Rf = 0.13 (20% ethyl acetate in hexane); 1H NMR (400 MHz, DMSO-d6) δ: 10.85 (s, 1H), 8.22 (d, J = 9.0 Hz, 1H), 7.55–7.42 (m, 3H), 7.07 (dd, J = 7.5, 1.3 Hz, 1H), 2.41 (s, 3H); 13C NMR (100.6 MHz, DMSO-d6) δ: 168.71, 153.79, 152.42, 134.84, 132.59, 127.94, 122.16, 121.74, 119.42 (2C), 115.41, 111.74, 98.99; IR (solid): 3338, 2921, 2855, 2229, 2070, 1906, 1753, 1583 cm−1; HRMS (ESI): m/z [M+Na]+ calcd. for C13H9NO3Na: 250.0480, found: 250.0471.
Procedure A for the Synthesis of 1-cyano-8-methoxynaphthalen-2-yl acetate (9) and 8-cyano-7-methoxynaphthalen-1-yl acetate (10)
To a solution of compound (8) (50 mg, 0.22 mmol, 1 equiv) in dry acetone (5 mL), under an atmosphere of N2, was added oven-dried K2CO3 (34 mg, 0.24 mmol, 1,1 equiv) and dimethyl sulfate (31 mg, 0.24 mmol, 1.1 equiv) and the reaction was left stirring for 24 h at room temperature. (TLC had shown complete conversion of the starting material and the presence of two new spots (visualized under a UV lamp). Water (20 mL) was added, the reaction mixture was extracted with EtOAc (3 × 10 mL) and the combined organic extracts were washed with brine (10 mL), dried over anhydrous Na2SO4, and concentrated in vacuo. The acquired crude residue was purified by flash column chromatography (20% EtOAc in hexane) to give the title compounds (9) and (10).
Compound (9): (5.3 mg, 10%) as a yellow solid, m.p. = 94−96 oC; Rf = 0.25 (20% EtOAc in hexane); 1H NMR (400 MHz, DMSO-d6) δ: 8.30 (d, J = 8.9 Hz, 1H), 7.66 (dd, J = 8.3, 1.2 Hz, 1H), 7.64–7.52 (m, 2H), 7.24 (dd, J = 7.8, 1.1 Hz, 1H), 4.00 (s, 3H), 2.42 (s, 3H); 13C NMR (100.6 MHz, DMSO-d6) δ: 168.64, 154.19, 153.83, 134.96, 132.20, 127.80, 122.83, 122.14, 121.08, 115.17, 108.49, 98.76, 55.95, 20.65; IR (solid): 2924, 2855, 2223, 1578 cm−1; HRMS (ESI): m/z [M+H]+ calcd. for C14H12NO3: 242.0812, found: 242.0813.
Compound (10): (26.5 mg, 50%) as a yellow solid, m.p. = 132−134 oC; Rf = 0.11 (20% EtOAc in hexane); 1H NMR (400 MHz, DMSO-d6) δ: 8.36 (d, J = 9.3 Hz, 1H), 7.95 (dd, J = 8.2, 1.3 Hz, 1H), 7.66 (d, J = 9.3 Hz, 1H), 7.54–7.50 (m, 1H), 7.43 (dd, J = 7.6, 1.2 Hz, 1H), 4.08 (s, 3H), 2.42 (s, 3H); 13C NMR (100.6 MHz, DMSO-d6) δ: 169.86, 163.70, 143.96, 136.24, 129.33, 127.36, 124.87 (2C), 122.97, 116.10, 113.69, 88.92, 57.09, 20.89; IR (solid): 2942, 2850, 2219, 2101, 1747, 1592 cm−1; HRMS (ESI): m/z [M+Na]+ calcd. for C14H11NO3Na: 264.0631, found: 264.0625.
Procedure B for the Synthesis of Compounds (9) and (10)
To a solution of compound (8) (50 mg, 0.22 mmol, 1 equiv) in dry THF (5 mL), under an atmosphere of nitrogen, was added oven-dried K2CO3 (34 mg, 0.24 mmol, 1.1 equiv) and MeI (34 mg, 0.24 mmol, 1.1 equiv) and the reaction mixture was left stirring for 18 h at room temperature (TLC had shown complete conversion of the starting material and the presence of two new spots). Water (20 mL) was added and the reaction mixture was extracted with EtOAc (3 × 10 mL) and the combined organic extracts were washed with brine (10 mL), dried over anhydrous Na2SO4, and concentrated under reduced pressure. The acquired crude residue was purified by flash column chromatography (20% EtOAc in hexane) to give compounds (9) and (10).
Compound (9): (6.9 mg, 13%) as a yellow solid, m.p. = 94−96 oC; Rf = 0.25 (20% EtOAc in hexane); (the 1H NMR data were in agreement with those reported in Procedure A for this compound).
Compound (10): (25 mg, 50%) as a yellow solid, m.p. = 132−134 oC; Rf = 0.11 (20% EtOAc in hexane); (the 1H NMR data were in agreement with those reported in Procedure A for this compound).
Procedure C for the Synthesis of Compounds (9) and (10)
To a solution of compound (8) (50 mg, 0.22 mmol, 1 equiv) in dry THF (5 mL), under an atmosphere of N2, was added NaH (5.8 mg, 0.24 mmol, 1.1 equiv) and MeI (34 mg, 0.24 mmol, 1.1 equiv) and the reaction mixture was left stirring for 18 h at room temperature (TLC analysis showed the absence of starting material and presence of two new spots). Water (20 mL) was added to the reaction mixture and then extracted with EtOAc (3 × 10 mL). The combined organic extracts were washed with brine (10 mL), dried over anhydrous Na2SO4 and removed under reduced pressure. The acquired crude residue was purified by flash column chromatography (20% EtOAc in hexane) to give title compounds (9) and (10).
Compound (9): (11.7 mg, 22%) as a yellow solid, m.p. = 94−96 oC; Rf = 0.25 (20% EtOAc in hexane); (the 1H NMR data were in agreement with those reported in Procedure A for this compound).
Compound (10): (21 mg, 40%) as a yellow solid, m.p. = 132−134 oC; Rf = 0.11 (20% EtOAc in hexane); (the 1H NMR data were in agreement with those reported in Procedure A for this compound).
Procedure D for the Synthesis of Compounds (9) and (10)
To a solution of compound (8) (50 mg, 0.22 mmol, 1 equiv) in dry THF (50 mL), under an atmosphere of N2, was added NaH (5.8 mg, 0.24 mmol, 1.1 equiv) and MeI (34 mg, 0.24 mmol, 1.1 equiv) and the reaction was left stirring for 18 h at room temperature (TLC analysis had shown the absence of the starting material spot and the presence of two new spots). Water (20 mL) was added to the reaction mixture, extracted with EtOAc (3 × 10 mL) and the combined organic extracts washed with brine (10 mL), dried over anhydrous Na2SO4 and evaporated under vacuum. The acquired crude residue was purified by flash column chromatography (20% EtOAc in hexane) to give products (9) and (10).
Compound (9): (21.8 mg, 41%) as a yellow solid, m.p. = 94−96 oC; Rf = 0.25 (20% EtOAc in hexane); (the 1H NMR data were in agreement with those reported in Procedure A for this compound).
Compound (10): (4.8 mg, 9%) as a yellow solid, m.p. = 132−134 oC; Rf = 0.11 (20% EtOAc in hexane); (the 1H NMR data were in agreement with those reported in Procedure A for this compound).
Attempted Reduction of 1-cyano-8-methoxynaphthalen-2-yl acetate (9) with DIBAL
To solution of compound (
9) (50mg, 0.27 mmol, 1 equiv) in dry THF, under an atmosphere of N
2, that was cooled to −78
oC, DIBAL (0.86 mL, of a 1.2 M in toluene, 0.864 mmol, 3.2 equiv) was added dropwise and the reaction mixture was left stirring at that temperature for 0.5 h and then for 1 h at room temperature (TLC analysis had shown the absence of the starting material spot and the presence of one new spot). A saturated aq. solution of NH
4Cl (5 mL) was added dropwise, followed by 1 N HCl aq. (25 mL) and EtOAc (25 mL) and the reaction mixture was left stirring for 1 h. The organic phase was separated and the aqueous phase was extracted with EtOAc (3 × 10 mL), the combined organic phases were washed with brine (20 mL), dried over anhydrous Na
2SO
4 and evaporated under vacuum. The acquired crude residue was purified by flash column chromatography (20% EtOAc in hexane) to give 2-hydroxy-8-methoxy-1-naphthonitrile (
4) (23 mg, 36%) as a yellow solid, m.p. = 172−174
oC; R
f = 0.09 (20% EtOAc in hexane); [the
1H NMR data were in agreement with those reported for this compound synthesized from compound (
2) (
Scheme 2)].
Attempted Reduction of 1-cyano-8-methoxynaphthalen-2-yl acetate (9) with PtO2
Compound (
9) (30 mg, 0.16 mmol, 1 equiv) was dissolved in a stirred 1/1 solution of HCOOH/H
2O (4 mL), under an atmosphere of N
2. PtO
2 (3.6mg, 0.016 mmol, 0.1 equiv) was added and the reaction mixture was heated at 55
oC for 2 h (TLC analysis had shown the absence of the starting material spot and the presence of one new spot). The cooled reaction mixture was filtered through celite, water (10 mL) was added to the filtrate and the aqueous solution was extracted with EtOAc (3 × 10 mL). The combined organic phases were washed with brine (20 mL), dried over anhydrous Na2SO4 and evaporated under vacuum. The acquired crude residue was purified by flash column chromatography (20% EtOAc in hexane) to give 2-hydroxy-8-methoxy-1-naphthonitrile (
4) (29 mg, 45%) as a yellow solid, m.p. = 172−174
oC; R
f = 0.09 (20% EtOAc in hexane) [the
1H NMR data were in agreement with those reported for this compound synthesized from compound (2) (
Scheme 2)].
Synthesis of 2-hydroxy-8-methoxy-1-naphthaldehyde (6)
To a solution of compound (
9) (250 mg, 1 mmol) in dry toluene (10 mL) at 0
oC, under an atmosphere of N
2, was added DIBAL (1.67 mL of a 1.2 M in toluene, 2 mmol) and the reaction mixture was stirred for 0.5 h and then left stirring for 18 h at room temperature. Upon completion of the reaction (TLC examination), the solvent was removed under vacuum and water (20 mL) was carefully added to the residue. The resulting mixture was cooled to 0
oC, followed by the dropwise addition of 1 M HCl aq. until pH = 1. The aqueous solution was extracted with EtOAc (3 × 10 mL) and the combined organic extracts were washed with brine (20 mL), dried over anhydrous Na
2SO
4 and the solvent removed under vacuum. The acquired crude residue was purified by flash column chromatography (17% EtOAc in hexane) to give the title compound (31.5 mg, 15%) as a yellow solid, m.p. = 67–69
oC (lit. [
62], m.p. = 68–70
oC); R
f = 0.54 (20% ethyl acetate in hexane); (the
1H NMR data were in agreement with those reported for this compound synthesized from (4) (
Scheme 2) and with those previously reported) [
62].
Synthesis of 2,8-dihydroxy-1-naphthonitrile (3)
A solution of compound (
1) (500 mg, 2.7 mmol) in dry DMF (15 mL) was heated at 120
oC for 0.5 h (TLC analysis had shown the absence of starting material and the presence of one new spot), the reaction was left to cool to room temperature, then water (100 mL) was added. The reaction mixture was extracted with EtOAc (3 × 20 mL) and the combined organic extracts were washed with brine (20 mL), dried over anhydrous Na2SO4, and concentrated under vacuum. The acquired crude residue was purified by flash column chromatography (25% EtOAc in hexane) to give the title compound as a yellow solid (400 mg, 80%), m.p. = 166–167
oC (lit. [
10], m.p. = 167–168
oC); R
f = 0.1 (20% ethyl acetate in hexane);
1H NMR (400 MHz, DMSO-
d6) δ: 11.24 (s, 1H), 10.31 (s, 1H), 7.93 (d, J = 9.0 Hz, 1H), 7.32 (d, J = 7.9 Hz, 1H), 7.25–7.15 (m, 2H), 6.92 (dd, J = 7.6, 1.1 Hz, 1H) (in agreement with the
1H NMR data that were previously reported for this compound) [
10].
Synthesis of 2,8-dihydroxy-1-naphthaldehyde (11)
A closed vessel with a magnetic stir bar was charged with Ni(OAc)2·4H2O (20 mg, 0.11 mmol, 0.2 equiv), followed by water (1 mL) and the mixture was stirred at room temperature for a few minutes. Ca(H
2PO
2)
2 (90 mg, 0.54 mmol, 1 equiv) was then added, followed by Ca(OAc)
2.H
2O (40 mg, 0.22 mmol, 0.4 equiv), compound (3) (100 mg, 0.54 mmol, 1 equiv) and EtOH (1 mL). The reaction mixture was heated at 100
oC for 24 h. Upon completion (TLC analysis) the reaction was left to cool to room temperature and water (10 mL) was added. The reaction mixture was then extracted with EtOAc (3 × 10 mL) and the combined organic extracts were washed with brine (10 mL), dried over anhydrous Na
2SO
4, and concentrated under vacuum. The acquired crude residue was purified by flash column chromatography (17% EtOAc in hexane) to give the title compound (29 mg, 29%) as a yellow solid, m.p. = 194–196
oC (lit. [
14], m.p. = 195–197
oC); R
f = 0.51 (33% EtOAc in hexane);
1H NMR (400 MHz, CDCl
3) δ: a broad singlet corresponding to OH is not visible, 11.33 (s, 1H), 7.92 (d, J = 9.1 Hz, 1H), 7.42 (d, J = 7.9 Hz, 1H), 7.23 (d, J = 7.7 Hz, 1H), 7.14 (d, J = 9.0 Hz, 1H), 6.98 (d, J = 7.4 Hz, 1H) (in agreement with the
1H NMR data that was previously reported for this compound [
14].
Synthesis of 2-hydroxy-8-methoxy-1-naphthaldehyde (6) and 8-hydroxy-2-methoxy-1-naphthaldehyde (12)
To a solution of (11) (25 mg, 0.133 mmol, 1 equiv) in dry DMF (5 mL), under an atmosphere of N2, oven-dried K2CO3 (19 mg, 0.134 mmol, 1.05 equiv) was added, followed by MeI (19 mg, 0.134 mmol, 1.05 equiv) and the reaction was stirred at room temperature for 2 h (TLC had shown the absence of the starting material and the presence of two new spots). Water (50 mL) was added, the reaction mixture was extracted with EtOAc (3 × 20 mL) and the combined organic extracts were washed with brine (20 mL), dried over anhydrous Na2SO4, and concentrated under vacuum. The acquired crude residue was purified by flash column chromatography (11% EtOAc in hexane) to give the title products (6) and (12).
Compound (
6): (5.1 mg, 19%) as a yellow solid, m.p. = 67–68
oC (lit. [
62], m.p. = 68–70
oC); R
f = 0.54 (20% ethyl acetate in hexane); (the
1H NMR data were in agreement with those reported for this compound synthesized from (
4) (
Scheme 2) and with those previously reported) [
62].
Compound (
12): (12.5 mg, 47%) as a yellow solid, m.p. = 110-112
oC); R
f = 0.28 (20% ethyl acetate in hexane); 1H NMR (400 MHz, CDCl
3) δ: 12.00 (s, 1H), 10.59 (s, 1H), 8.12 (d, J = 9.3 Hz, 1H), 7.35 (d, J = 7.7 Hz, 1H), 7.29–7.26 (m, 1H), 7.22 (d, J = 9.2 Hz, 1H), 7.14 (dd, J = 7.6, 1.4 Hz, 1H), 4.08 (s, 3H) (in agreement with the
1H NMR data that were previously reported for this compound) [
15].
Synthesis of 2,8-dimethoxy-1-naphthonitrile (5)
To a stirred solution of compound (
3) (300 mg, 1.62 mmol, 1 equiv) in acetone (10 mL), was added MeI (460 mg, 3.28 mmol, 2.02 equiv) followed by Na
2CO
3 (175 mg, 1.65 mmol, 1.02 equiv) and H
2O (2 mL). The reaction mixture was gently heated for 3 h during which time TLC analysis had shown complete conversion of the starting material and the presence of one new spot. The solvents were removed under reduced pressure and the oily residue was triturated with hexane to give the title compound (270 mg, 78%) as a colorless solid (hexane), m.p. = 147–149
oC; R
f = 0.21 (20% ethyl acetate in hexane); [the
1H NMR data were in agreement with those reported for this compound synthesized from compound (
2) (
Scheme 2)].
Synthesis of 2,8-dimethoxy-1-naphthaldehyde (13)
To a stirred solution of compound (
5) (210 mg, 1 mmol) in dry toluene (10 mL) at 0
oC, under an atmosphere of N
2, was added DIBAL (1.67 mL of a 1.2 M in toluene, 2 mmol) and the reaction mixture stirred for 0.5 hours and then left stirring at room temperature for 18 h. Upon completion of the reaction (TLC analysis) the solvent was removed under reduced pressure, cold water (20 mL) was carefully added to the residue and the resulting mixture was cooled to 0
oC, followed by the dropwise addition of 1 M HCl aq. until pH = 1. The aqueous solution was extracted with EtOAc (3 × 10 mL) and the combined organic extracts were washed with brine (20 mL), dried over anhydrous Na
2SO
4 and concentrated under vacuum. The acquired crude residue was purified by flash column chromatography (17% EtOAc in hexane) to give the title compound as a light brown oil (112 mg, 52%), R
f = 0.37 (20% ethyl acetate in hexane);
1H NMR (400 MHz, CDCl
3) δ: 10.75 (s, 1H), 7.85 (d, J = 9.1 Hz, 1H), 7.40 (dd, J = 8.2, 1.0 Hz, 1H), 7.34–7.25 (m, 2H), 6.86 (dd, J = 7.6, 1.0 Hz, 1H), 3.93 (s, 3H), 3.92 (s, 3H) (in agreement with the
1H NMR data that were previously reported for this compound) [
16].
Procedure F for the Synthesis of 2-hydroxy-8-methoxy-1-naphthaldehyde (6), 8-hydroxy-2-methoxy-1-naphthaldehyde (12) and 2,8-dihydroxy-1-naphthaldehyde (11)
To an oven-dried closed vessel with a magnetic stirrer bar, under an atmosphere of N
2, was added compound (
13) (50 mg, 0.231 mmol, 1 equiv), MgBr
2 diethyl etherate (119 mg, 0.462 mmol, 2 equiv), KI (76.5 mg, 0.642 mmol, 2 equiv) and MeCN (10 mL). The vessel was sealed and then heated with stirring at 150
oC for 2 h. TLC analysis of the cooled reaction mixture showed the absence of the starting material and the presence of three new spots, one intense and two very faint (visualized under a UV lamp). The solvent was removed under vacuum, cooled water (20 mL) was carefully added to the residue and the resulting cooled mixture was acidified by adding dropwise 1 M HCl aq. to pH = 1. The aqueous mixture was extracted with EtOAc (3 × 10 mL), the combined organic extracts washed with brine (10 mL), dried over anhydrous Na
2SO
4 and concentrated under reduced pressure. The acquired crude residue was purified by flash column chromatography (11% EtOAc in hexane) to give title compound (
6) (4.6 mg, 10%) as a yellow solid, m.p. = 67–69
oC (lit. [
62], m.p. = 68–70
oC); R
f = 0.54 (20% EtOAc in hexane); (the
1H NMR data were in agreement with those reported for this compound synthesized from compound (
4) (
Scheme 2) and with those previously reported) [
62].
Procedure G for the Synthesis of 2-hydroxy-8-methoxy-1-naphthaldehyde (6), 8-hydroxy-2-methoxy-1-naphthaldehyde (12) and 2,8-dihydroxy-1-naphthaldehyde (11)
A solution of compound (13) (50 mg, 0.231 mmol, 1 equiv) in dry DCM (10 mL), over an atmosphere of N2, was cooled to 0 oC and then BBr3 (690 μL of a 1 M solution in DCM, 0.693 mmol, 3 equiv) was added dropwise. The reaction was left stirring at room temperature for 18 h (TLC analysis had shown complete conversion of the starting material and the presence of three new spots) and then cooled to 0 oC, quenched slowly with cold water (30 mL) and extracted with DCM (3 × 50 mL). The combined organic extracts were washed with brine (20 mL), dried over anhydrous Na2SO4 and concentrated reduced pressure. The acquired crude residue was purified by flash column chromatography (11% EtOAc in hexane) to give title compounds (6), (12) and (11).
Compound (
6): (3.7 mg, 8%) as a yellow solid, m.p. = 67–68
oC (lit. [
62], m.p. = 68–70
oC); R
f = 0.54 (20% EtOAc in hexane); (the
1H NMR data were in agreement with those reported for this compound synthesized from (
4) (
Scheme 4) and with those previously reported) [
62].
Compound (
12): (3.2 mg, 7%) as a yellow solid, m.p. = 110-112
oC); R
f = 0.28 (20% ethyl acetate in hexane);
1H NMR (400 MHz, CDCl
3) δ: 12.00 (s, 1H), 10.59 (s, 1H), 8.12 (d, J = 9.3 Hz, 1H), 7.35 (d, J = 7.7 Hz, 1H), 7.29–7.26 (m, 1H), 7.22 (d, J = 9.2 Hz, 1H), 7.14 (dd, J = 7.6, 1.4 Hz, 1H), 4.08 (s, 3H) (in agreement with the
1H NMR data that were previously reported for this compound) [
73].
Compound (
11): (25.6 mg, 59%) as a yellow solid, m.p. = 194–196
oC (lit. [
14], m.p. = 195–197
oC); R
f = 0.51 (33% EtOAc in hexane); (the
1H NMR data were in agreement with those reported for this compound synthesized from compound (
3) and with those previously reported) [
14].
Procedure H for the Synthesis of 2-hydroxy-8-methoxy-1-naphthaldehyde (6), 8-hydroxy-2-methoxy-1-naphthaldehyde (12) and 2,8-dihydroxy-1-naphthaldehyde (11)
A solution of compound (13) (50 mg, 0.231 mmol, 1 equiv) in dry DCM (10 mL), over an atmosphere of N2, was cooled to 0 oC and then BBr3 (230 μL of a 1M solution in DCM, 0.231 mmol, 1 equiv) was added dropwise and the reaction was left stirring at room temperature for 1 h. TLC analysis had shown complete conversion of the starting material and the presence of three new spots (visualized under a UV lamp). The reaction mixture was cooled to 0 oC, quenched slowly with cold water (30 mL), extracted with DCM (3 × 20 mL), the combined organic extracts washed with brine (20 mL), dried over anhydrous Na2SO4 and concentrated under vacuum. The acquired crude residue was purified by flash column chromatography (11% EtOAc in hexane) to give title compounds (6), (12) and (11).
Compound (
6): (3.7 mg, 19%) as a yellow solid, m.p. = 67–68
oC (lit. [
62], m.p. = 68–70
oC); R
f = 0.54 (20% EtOAc in hexane); (the
1H NMR data were in agreement with those reported for this compound synthesized from (
4) and with those previously reported) [
62].
Compound (
12): (5 mg, 11%) as a yellow solid, m.p. = 110-112
oC; R
f = 0.28 (20% ethyl acetate in hexane); (the
1H NMR data were in agreement with those reported for this compound synthesized from compound (
13) (
Scheme 5) and with those previously reported) [
15].
Compound (
11): (14.3 mg, 33%) as a yellow solid, m.p. = 194–196
oC (lit. [
14], m.p. = 195–197
oC); R
f = 0.51 (33% EtOAc in hexane); (the
1H NMR data were in agreement with those reported for this compound synthesized from compound (
3) (
Scheme 4) and with those previously reported) [
14].
Procedure I for the Synthesis of 2-hydroxy-8-methoxy-1-naphthaldehyde (6), 8-hydroxy-2-methoxy-1-naphthaldehyde (12) and 2,8-dihydroxy-1-naphthaldehyde (11)
A solution of A solution of compound (13) (50 mg, 0.231 mmol, 1 equiv) in dry DCM (10 mL), over an atmosphere of N2, was cooled to −15 oC and then BBr3 (230 μL of a 1 M solution in DCM, 0.231 mmol, 1 equiv) was added dropwise over a period of 10 min. The reaction was allowed to slowly reach room temperature and was left stirring for 1 h after which TLC analysis had shown complete conversion of the starting material and the presence of three new spots (visualized under a UV lamp). The reaction mixture was cooled to 0 oC, quenched slowly with cold water (30 mL) and extracted with DCM (3 × 20 mL). The combined organic extracts were washed with brine (20 mL), dried over anhydrous Na2SO4 and concentrated under reduced pressure. The acquired crude residue was purified by flash column chromatography (11% EtOAc in hexane) to give title compounds (6), (12) and (11).
Compound (
6): (25.9 mg, 56%) as a yellow solid, m.p. = 67–68
oC (lit. [
62], m.p. = 68–70
oC); R
f = 0.54 (20% EtOAc in hexane); (the
1H NMR data were in agreement with those reported for this compound synthesized from (
4) and with those previously reported) [
62].
Compound (
12): (10 mg, 22%) as a yellow solid, m.p. = 110-112
oC); R
f = 0.28 (20% ethyl acetate in hexane); (the
1H NMR data were in agreement with those reported for this compound synthesized from compound (
13) (
Scheme 5) and with those previously reported) [
15].
Compound (
11): (4.3 mg, 10%) as a yellow solid, m.p. = 194–196
oC (lit. [
14], m.p. = 195–197
oC); R
f = 0.51 (33% EtOAc in hexane); (the
1H NMR data were in agreement with those reported for this compound synthesized from compound (
3) (
Scheme 4) and with those previously reported) [
14].
Procedure J for the Synthesis of 2,8-bis(benzyloxy)-1-naphthonitrile (14) and 2-(benzyloxy)-8-hydroxy-1-naphthonitrile (15)
To a stirred solution of compound (1) (500 mg, 2.7 mmol, 1 equiv) in dry acetone (20 mL), under an atmosphere of N2, was added benzyl bromide (462 mg, 2.7 mmol, 1 equiv), oven-dried K2CO3 (391 mg, 2.83 mmol, 1.05 equiv) and KI (89 mg, 0.54 mmol, 0.2 equiv). The reaction was left stirring at room temperature for 18 h, after which TLC analysis indicated the absence of the starting material and the presence of two new spots. The solvent was removed under vacuum, the residue was dissolved in EtOAc (30 mL) and washed with water (3 × 10 mL). The organic extract was dried over anhydrous Na2SO4 and concentrated under reduced pressure. The acquired crude residue was purified by flash column chromatography (20% EtOAc in hexane) to give the title compounds (14) and (15).
Compound (14): (118 mg, 12%) as an orange oil; Rf = 0.38 (20% EtOAc in hexane); 1H NMR (400 MHz, CDCl3) δ: 7.90 (d, J = 9.1 Hz, 1H), 7.65–7.60 (m, 2H), 7.58–7.51 (m, 2H), 7.48–7.24 (m, 9H), 6.99 (d, J = 7.6 Hz, 1H), 5.39 (d, J = 3.6 Hz, 4H); 13C NMR (100.6 MHz, CDCl3) δ: 162.11, 153.05, 136.39, 136.06, 134.61, 130.02, 128.83 (2C), 128.61 (2C), 128.22, 128.07, 127.84 (2C), 127.02 (2C), 125.34, 125.28, 121.32, 116.78, 114.08, 109.59, 93.78, 71.32, 71.11.; IR (solid): 3067, 3027, 2922, 2887, 2214, 2094, 1737, 1677, 1591 cm−1; HRMS (ESI): m/z [M+H]+ calcd. for C25H20NO2: 366.1489, found: 366.1485.
Compound (15): (450 mg, 61%) as a colorless solid, m.p. = 136–138 oC; Rf = 0.49 (20% ethyl acetate in hexane); 1H NMR (400 MHz, CDCl3) δ: 7.85–7.77 (m, 2H), 7.59 (d, J = 8.8 Hz, 1H), 7.44 (d, J = 8.8 Hz, 1H), 7.33–7.25 (m, 6H), 5.46 (s, 2H); 13C NMR (100.6 MHz, CDCl3) δ: 156.03, 139.59, 135.71, 131.33, 129.68, 129.25 (2C), 128.10, 127.01, 126.31 (2C), 124.92, 124.43, 123.94, 120.35, 120.21, 110.81, 47.79; IR (solid): 3052, 2920, 2845, 2216, 2117, 1886, 1752 cm−1; HRMS (ESI): m/z [M+H]+ calcd. for C18H14NO2: 276.1019, found: 216.1018.
Synthesis of 2-(benzyloxy)-8-methoxy-1-naphthonitrile (16)
Prepared according to the experimental procedure for the synthesis of (
5) from (
3)
Scheme 5. Used as starting material compound (
15) (400 mg, 5 mmol, 1 equiv), MeI (724 mg, 5.1 mmol, 1.02 equiv) Na
2CO
3 (540 mg, 5.1 mmol, 1.02 equiv), acetone (15 mL) and H
2O (3 mL), that gave the title compound (384 mg, 93%) as a yellow solid, m.p. = 100–103
oC; R
f = 0.32 (20% EtOAc in hexane);
1H NMR (400 MHz, CDCl
3) δ: 7.89 (d, J = 9.1 Hz, 1H), 7.55–7.48 (m, 2H), 7.42–7.30 (m, 5H), 7.26–7.24 (m, 1H), 6.94 (dd, J = 7.1, 1.7 Hz, 1H), 5.40 (s, 2H), 4.04 (s, 3H);
13C NMR (100.6 MHz, CDCl
3) δ: 161.96, 154.32, 136.08, 134.55, 129.87, 128.84 (2C), 128.23, 127.02 (2C), 125.39, 125.15, 121.02, 116.80, 114.15, 107.71, 93.69, 71.32, 55.82; IR (solid): 2919, 2844, 2212, 2101, 1737, 1595 cm
−1; HRMS (ESI): m/z [M+H]
+ calcd. for C
19H
16NO
2: 290.1176, found: 290.1177.
Synthesis of 2-(benzyloxy)-8-methoxy-1-naphthaldehyde (17)
Prepared according to the experimental procedure for the synthesis of compound (
13) from (
5)
Scheme 5. Used as starting material compound (
16) (100 mg, 0.34 mmol, 1 equiv), DIBAL (580 μL of a 1.2 M in toluene, 0.68 mmol, 2 equiv) and dry toluene (10 mL), that gave the title compound (55.6 mg, 55%) as a yellow oil; R
f = 0.46 (20% EtOAc in hexane);
1H NMR (400 MHz, CDCl
3) δ: 10.76 (s, 1H), 7.81 (d, J = 9.0 Hz, 1H), 7.46–7.27 (m, 8H), 6.86 (dd, J = 7.7, 1.1 Hz, 1H), 5.23 (s, 2H), 3.94 (s, 3H);
13C NMR (100.6 MHz, CDCl
3) δ: 194.98, 155.01, 153.70, 136.89, 131.66, 130.49, 128.73 (2C), 128.10, 127.41 (2C), 124.80, 124.06, 123.44, 121.09, 116.39, 106.62, 72.30, 56.05; IR (solid): 2924, 2843, 2210, 1711, 1596 cm
−1; HRMS (ESI): m/z [M+H]
+ calcd. for C
19H
17O
3: 293.1172, found: 293.1168.
Synthesis of 2-hydroxy-8-methoxy-1-naphthaldehyde (6)
To a stirred solution of compound (
17) (20 mg, 0.099 mmol) in anhydrous MeOH (5 mL), under an atmosphere of N
2, 5% Pd/C (1 mg, 5%) was added and the reaction was purged with H
2. Stirring at room temperature was continued for 18 h (TLC analysis showed complete conversion of the starting material and the presence of a new spot. The reaction mixture was filtered and the solvent removed under vacuum. To the remaining residue EtOAc (20 mL) was added and then washed with brine (10 mL). The organic layer was dried over anhydrous Na
2SO
4 and concentrated under reduced pressure. The acquired crude product was purified by flash column chromatography (20% EtOAc in hexane) to give the title compound (18 mg, 92%) as a yellow solid, m.p. = 67–69
oC (lit. [
62], m.p. = 68–70
oC); R
f = 0.54 (20% ethyl acetate in hexane); (the
1H NMR data were in agreement with those reported for this compound synthesized from compound (
4) (
Scheme 2) and with those previously reported) [
62].
Synthesis of (E)-2-hydroxy-8-methoxy-1-naphthaldehyde oxime (18)
To a stirred solution of compound (
6) (200 mg, 0.99 mmol, 1 equiv) in MeOH (20 mL), NH
2OH.HCl (76.4 mg, 0.0011 mmol, 1.1 equiv) was added, and the resulting mixture was cooled to 0
oC, followed by the dropwise addition of an aqueous saturated Na
2CO
3 solution until pH = 8. The reaction mixture was left stirring at room temperature for 1 h. TLC analysis had shown the absence of the starting material and the presence of a new spot on the baseline. The solution was cooled to 0
oC, followed by the dropwise addition of MeCO
2H until pH = 5. The solvent was evaporated under vacuum, water (25 mL) was added to the residue and the mixture was extracted with EtOAc (3 × 10 mL). The combined organic extracts were washed with brine (10 mL), dried over anhydrous Na
2SO
4 and concentrated under reduced pressure. The acquired light yellow solid was purified by flash column chromatography (20% EtOAc in hexane) to give the title compound (164 mg, 78 %) as a yellow solid, m.p. = 142–143 °C (lit. [
62], m.p. = 141–143
oC); R
f = 0.32 (20% EtOAc in hexane);
1H NMR (400 MHz, DMSO-
d6) δ: 12.06 (s, 1H), 11.45 (s, 1H), 9.65 (s, 1H), 7.83 (d, J = 9.0 Hz, 1H), 7.46 (d, J = 6.8 Hz, 1H), 7.29 (t, J = 7.9 Hz, 1H), 7.18 (d, J = 8.9 Hz, 1H), 7.08 (d, J = 7.9 Hz, 1H), 3.94 (s, 3H) (in agreement with the
1H NMR data that were previously reported for this compound) [
62].
Synthesis of 9-methoxynaphtho [1,2-d]isoxazole 2-oxide (19)
To a stirred solution of (E)-oxime (18) (150 mg, 0.70 mmol, 1 equiv) in dry t-BuOH (15 mL), under an atmosphere of N2, PIDA (450 mg, 1.40 mmol, 2 equiv) was added, and the resulting mixture was stirred at room temperature for 0.5 h. TLC analysis had shown the absence of the starting material and the presence of a new spot. Water (15 mL) was added followed by dropwise addition of 5% NaHCO3 aq. solution until pH = 7−8. The solvents were evaporated under vacuum and to the residue water (20 mL) was added and the resulting mixture was extracted with EtOAc (3 × 10 mL). The combined organic extracts were washed with brine (10 mL), dried over anhydrous Na2SO4 and concentrated under reduced pressure. The acquired residue was purified by flash column chromatography (17% EtOAc in hexane) to give the title compound (120 mg, 81 %) as a yellow solid, m.p. = 91–93 oC; Rf = 0.44 (20% EtOAc in hexane); 1H NMR (400 MHz, CDCl3) δ: 8.10 (s, 1H), 7.89 (d, J = 8.9 Hz, 1H), 7.52 (d, J = 8.1 Hz, 1H), 7.47 (t, J = 7.9 Hz, 1H), 7.36 (d, J = 9.0 Hz, 1H), 7.02 (d, J = 7.6 Hz, 1H), 4.07 (s, 3H); 13C NMR (100.6 MHz, CDCl3) δ: 155.15, 149.72, 131.86, 129.33, 126.12, 121.23, 117.39, 112.25, 111.30, 108.43, 106.61, 55.79; IR (solid) 3098, 2919, 2840, 2365, 2209, 2107, 1743, 1568 cm−1; HRMS (ESI): m/z [M+H]+ calcd. for C12H10NO3: 216.0661, found: 216.0664.
In Situ Generation of 2-hydroxy-8-methoxy(naphthalen-1-yl)nitrile oxide (20)
A solution of isoxazole 2-oxide (19) (5 mg, 0.023 mmol) in dry DMSO-d6 (0.5 mL), under an atmosphere of N2, was stirred at room temperature for 6 h. TLC analysis revealed the absence of the starting material and the presence of a new spot. The solution was transferred to an NMR tube and the 1H and 13C NMR spectra of the new compound were recorded. Rf = 0.2 (20% EtOAc in hexane); 1H NMR (400 MHz, DMSO-d6) δ: 11.29 (s, 1H), 7.92 (d, J = 9.0 Hz, 1H), 7.46 (dd, J = 8.2, 1.0 Hz, 1H), 7.39 – 7.20 (m, 2H), 7.07 (dd, J = 7.8, 1.0 Hz, 1H), 3.98 (s, 3H).; 13C NMR (100.6 MHz, DMSO-d6) δ: 161.86, 153.80, 133.31, 129.39, 124.90, 124.85, 121.69, 117.97, 108.13, 89.16, 56.89.; HRMS (ESI): m/z [M+H]+ calcd. for C12H10NO3: 216.0661, found: 216.0661.
Procedure K for the Synthesis of Dimethyl 3-(2-hydroxy-8-methoxynaphthalen-1-yl)isoxazole-4,5-dicarboxylate (21), 8-methoxy-1-(5-phenylisoxazol-3-yl)naphthalen-2-ol (22), methyl 3-(2-hydroxy-8-methoxynaphthalen-1-yl)-4,5-dihydroisoxazole-5-carboxylate (23) and 8-methoxy-1-(5-phenyl-4,5-dihydroisoxazol-3-yl)naphthalen-2-ol (24)
To a solution of 2-oxide (19) (5 mg, 0.023 mmol, 1 equiv) in dry DMSO (1.5 mL), under an atmosphere of N2, was added DMAD, phenylacetylene, methyl acrylate or styrene (0.069 mmol, 3 equiv) and the resulting mixture was stirred in room temperature for 18 h. TLC analysis had shown the absence of the starting material and the presence of a new spot (visualized under a UV lamp). The reaction was quenched with water (15 mL) and the resulting mixture was extracted with EtOAc (3 × 10 mL). The combined organic extracts were washed with brine (10 mL), dried over anhydrous Na2SO4 and concentrated under vacuum. The acquired residue was purified by flash column chromatography (17% EtOAc in hexane) to give the title compounds (21), (22), (23) and (24).
Compound (21): (7 mg, 85%) as a yellow solid, m.p. = 149−151 oC; Rf = 0.09 (20% EtOAc in hexane); 1H NMR (400 MHz, CDCl3) δ: a singlet corresponding to OH is not visible, 7.83 (d, J = 8.9 Hz, 1H), 7.41 (d, J = 8.1 Hz, 1H), 7.32–7.21 (m, 1H), 6.79 (d, J = 7.7 Hz, 1H), 4.05 (s, 3H), 3.62 (s, 3H), 3.38 (s, 3H); 13C NMR (100.6 MHz, CDCl3) δ: 162.22, 160.46, 159.17, 157.22, 154.43, 153.44, 132.89, 130.44, 124.20, 124.02, 121.53, 118.75, 118.29, 106.58, 103.82, 55.28, 53.63, 52.24; IR (solid) 3352, 2922, 2848, 2364, 2119, 1717, 1613, 1520 cm−1; HRMS (ESI): m/z [M+H]+ calcd. for C18H16NO7: 358.0921, found: 358.0916.
Compound (22): (6.2 mg, 93%) as a colorless solid, m.p. = 108−110 oC; Rf = 0.49 (20% EtOAc in hexane); 1H NMR (400 MHz, CDCl3) δ: 8.61 (s, 1H), 7.88–7.79 (m, 3H), 7.55–7.41 (m, 4H), 7.37–7.29 (m, 2H), 6.90 (d, J = 7.6 Hz, 1H), 6.55 (s, 1H), 3.72 (s, 3H); 13C NMR (100.6 MHz, CDCl3) δ: 168.01, 167.26, 154.77, 153.98, 132.36, 130.87, 130.38, 129.24 (2C), 127.69, 125.99 (2C), 124.09, 123.55, 121.63, 118.84, 107.62, 105.24, 103.92, 55.28; IR (solid) 3205, 2919, 2845, 2363, 2123, 1732, 1606 cm−1; HRMS (ESI): m/z [M+H]+ calcd. for C20H16NO3: 318.1125, found: 318.1125.
Compound (23): (6.2 mg, 89%) as a yellow solid, m.p. = 118−120 oC; Rf = 0.06 (20% EtOAc in hexane); 1H NMR (400 MHz, CDCl3) δ: a singlet corresponding to OH is not visible, 7.76 (d, J = 8.9 Hz, 1H), 7.40 (d, J = 8.0 Hz, 1H), 7.29 (t, J = 7.9 Hz, 1H), 7.22 (d, J = 8.9 Hz, 1H), 6.90 (d, J = 7.8 Hz, 1H), 5.20 (t, J = 8.6 Hz, 1H), 3.88 (s, 3H), 3.86 (s, 3H), 3.58 (d, J = 8.6 Hz, 2H); 13C NMR (100.6 MHz, CDCl3) δ: 171.38, 158.64, 154.38, 153.34, 132.27, 130.65, 124.30, 124.11, 121.69, 118.73, 108.01, 104.68, 56.29, 52.95, 45.17; IR (solid) 3173, 2928, 2848, 2364, 2122, 1899, 1736, 1607, 1516 cm−1; HRMS (ESI): m/z [M+H]+ calcd. for C16H16NO5: 302.1023, found: 302.1028.
Compound (24): (7.1 mg, 97%) as a colorless solid, m.p. = 205−207 oC; Rf = 0.34 (20% EtOAc in hexane); 1H NMR (400 MHz, CDCl3) δ: 8.31 (s, 1H), 7.67 (d, J = 8.9 Hz, 1H), 7.41 (d, J = 7.5 Hz, 2H), 7.36 (t, J = 7.5 Hz, 2H), 7.30 (t, J = 7.8 Hz, 2H), 7.23–7.14 (m, 2H), 6.73 (d, J = 7.7 Hz, 1H), 5.73 (dd, J = 10.4, 7.4 Hz, 1H), 3.54 (dd, J = 16.3, 10.3 Hz, 1H), 3.37–3.29 (m, 4H); 13C NMR (100.6 MHz, CDCl3) δ 159.05, 154.30, 153.37, 140.97, 131.97, 130.56, 128.74 (2C), 128.02, 125.77 (2C), 124.03, 123.93, 121.37, 118.53, 107.33, 105.34, 81.42, 55.20, 49.03; IR (solid) 3135, 2922, 2848, 2363, 2110, 1917, 1754, 1605, 1514 cm−1; HRMS (ESI): m/z [M+H]+ calcd. for C20H18NO3: 320.1281, found: 320.1279.