Submitted:
07 November 2023
Posted:
08 November 2023
You are already at the latest version
Abstract
Keywords:
1. Introduction
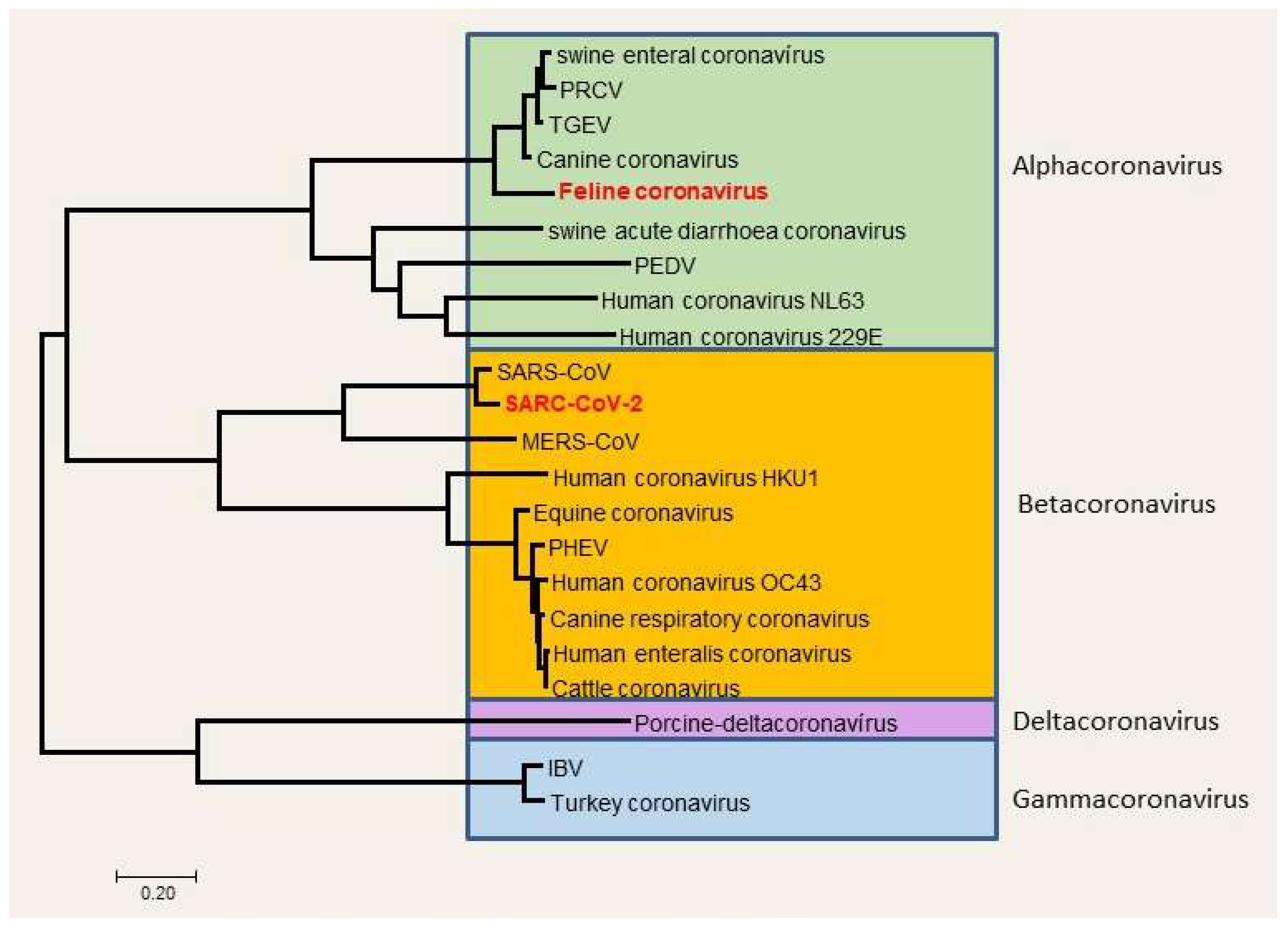
2. Materials and Methods
2.1. Ethics and animal experimentation
2.2. Clinical evaluation
2.3. Pathologic examination and sample collection
2.4. Histopathology and in situ hybridization
2.5. Molecular diagnostics, RT-qPCR
2.6. Next generation sequencing
2.7. Virus isolation on cell culture
2.8. Bacterial cultivation
3. Results
3.1. Clinical evaluation
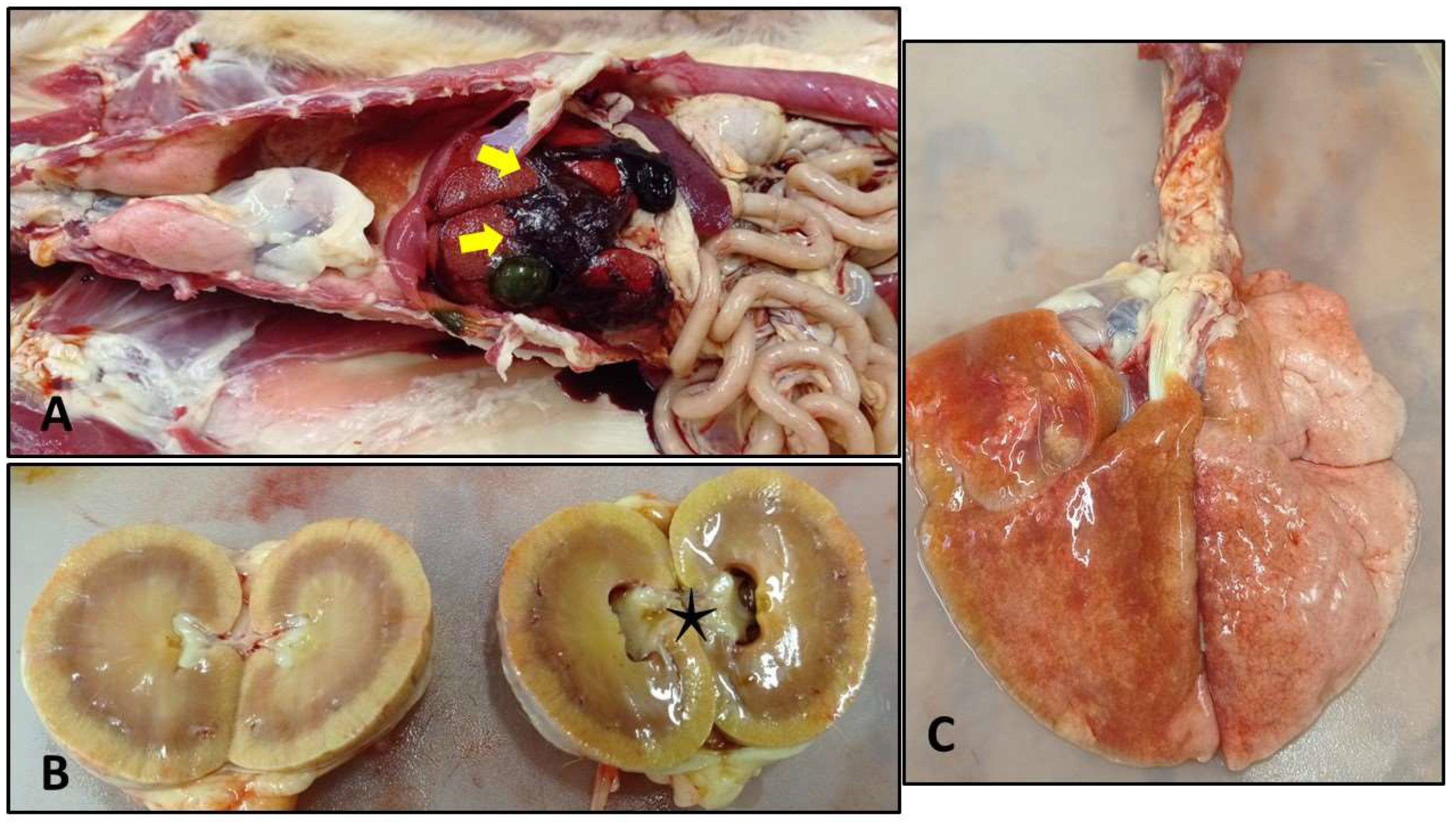
3.2. Postmortem evaluation
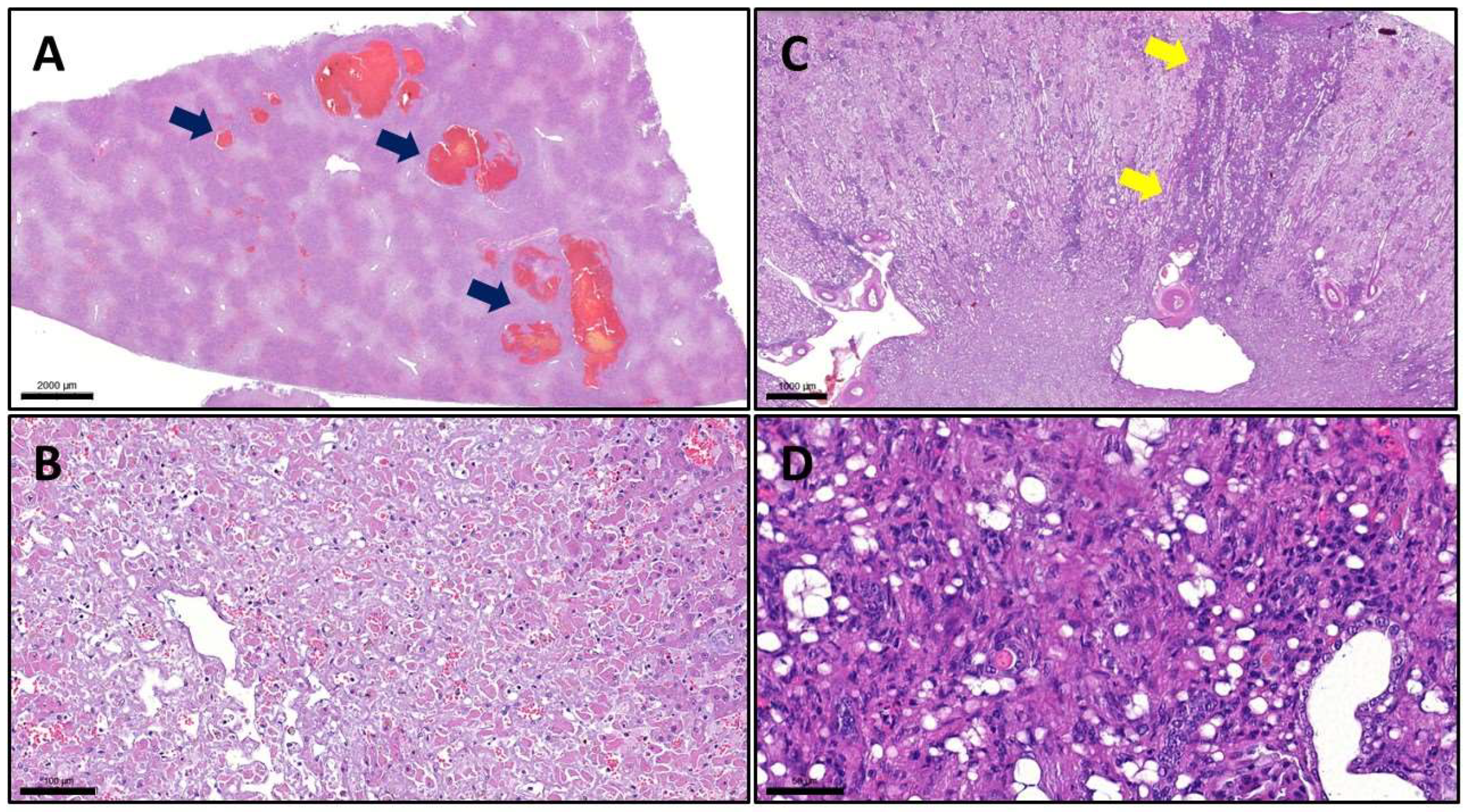
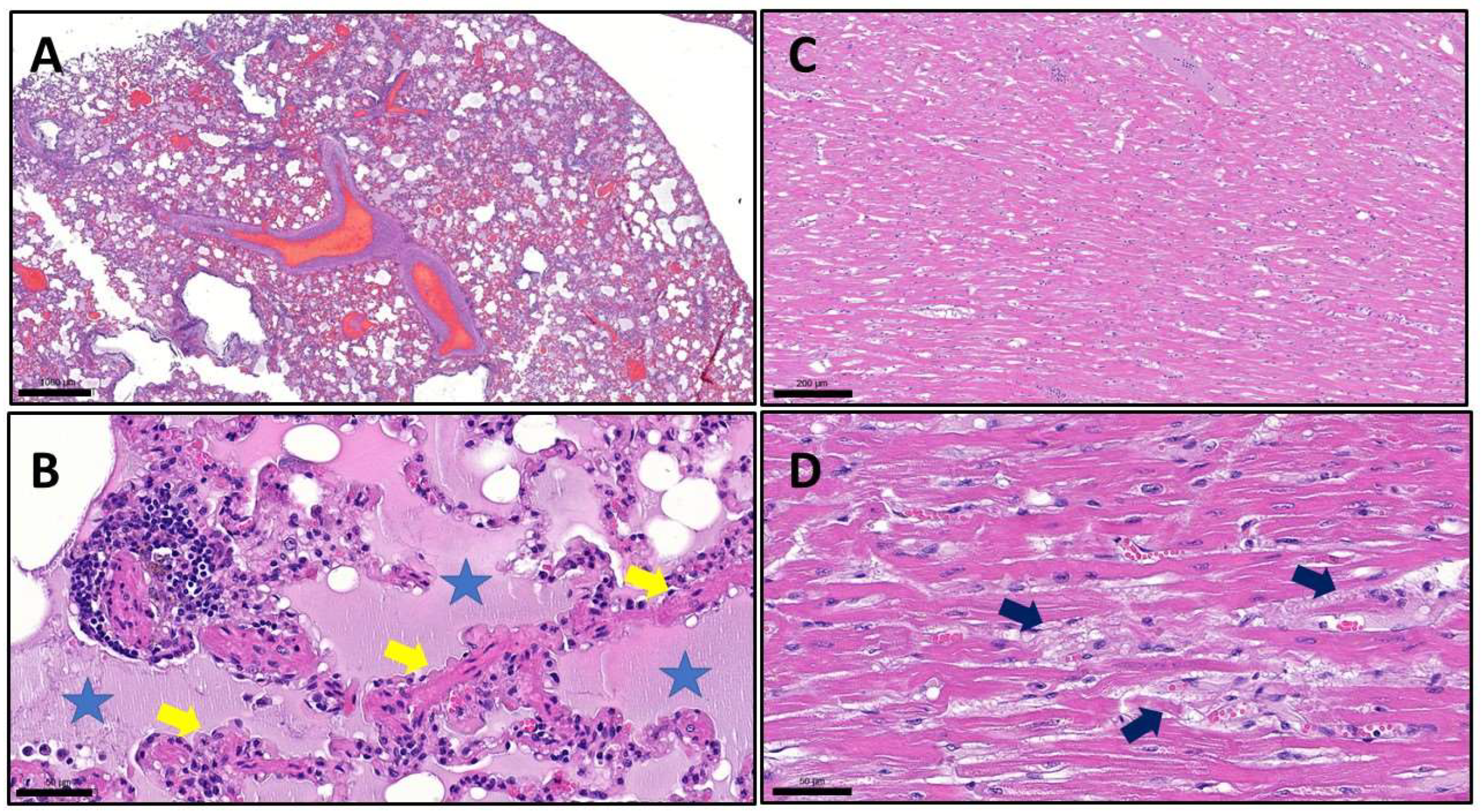
3.3. Histopathologic and ISH findings
3.4. Molecular diagnostic findings
3.5. Whole genome sequencing
3.6. Virus isolation
3.7. Bacterial cultivation
4. Discussion
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Sykes, J.; Greene, C. Infectious Diseases of the Dog and Cat, 4th ed.; Elsevier Saunders, Missouri, USA, 2012; 92.
- World Health Organization. (2020). Statement on the Second Meeting of the International Health Regulations (2005) Emergency Committee Regarding the Outbreak of Novel Coronavirus (2019-nCoV); World Health Organization: Geneva, Switzerland,. https://covid19.who.int/ (accessed , 2023). 10 October.
- Jo, W.K.; Oliveira-Filho, E.F.; Rasche, A.; Greenwood, A. ; Osterrieder, K; Drexler, J. F. Potential zoonotic sources of SARS-CoV-2 infections. Transbound. Emerg. Dis. 2020, 68, 1824–1834. [Google Scholar]
- Hobbs, E.C.; Reid, T.J. Animals and SARS-CoV-2: Species susceptibility and viral transmission in experimental and natural conditions, and the potential implications for community transmission. Transbound. Emerg. Dis. 2021, 68, 1850–1867. [Google Scholar] [CrossRef] [PubMed]
- Hosie, M.J.; Hofmann-Lehmann, R.; Hartmann, K.; Egberink, H.; Truyen, U.; Addie, D.D.; Belák, S.; Boucraut-Baralon, C.; Frymus, T.; Lloret, A.; et al. Anthropogenic Infection of Cats during the 2020 COVID-19 Pandemic. Viruses 2021, 13, 185. [Google Scholar] [CrossRef] [PubMed]
- Haake, C.; Cook, S.; Pusterla, N.; Murphy, B. Coronavirus Infections in Companion Animals: Virology, Epidemiology, Clinical and Pathologic Features. Viruses 2020, 12, 1023. [Google Scholar] [CrossRef] [PubMed]
- Hossain, G.; Javed, A.; Akter, S.; Saha, S. SARS-CoV-2 host diversity: An update of natural infections and experimental evidence. J. Microbiol. Immunol. Infect. 2021, 54, 175–181. [Google Scholar] [CrossRef] [PubMed]
- Mathavarajah, S.; Dellaire, G. Lions, tigers and kittens too: ACE2 and susceptibility to COVID-19. Evol. Med. Public Heal. 2020, 2020, 109–113. [Google Scholar] [CrossRef]
- Bienzle, D.; Rousseau, J.; Marom, D.; MacNicol, J.; Jacobson, L.; Sparling, S.; Prystajecky, N.; Fraser, E.; Weese, J.S. Risk factors for SARS-CoV-2 infection and illness in cats and dogs. Emerg. Infect. Dis. 2022, 28, 1154–1162. [Google Scholar] [CrossRef] [PubMed]
- Kaczorek-Łukowska, E.; Wernike, K.; Beer, M.; Wróbel, M.; Malaczewska, J. High Seroprevalence against SARS-CoV-2 among Dogs and Cats, Poland, 2021/2022. Animals. 2022, 12, 2016. [Google Scholar] [CrossRef] [PubMed]
- https://www.woah.org/fileadmin/Home/MM/A_Factsheet_SARS-CoV-2__1_.pdf accessed on 30th 23. 20 October.
- Balka, Gy.; Bálint, A. , Cságola, A. ; Farsang, A.; Jerzsele, A. et al. The biology of coronaviruses, with special regards to SARS-CoV-2-and COVID-19 – Literature review. Magy. Allatorvosok Lapja. 2020, 142, 259–277. [Google Scholar]
- Sila, T.; Sunghan, J.; Laochareonsuk, W.; Surasombatpattana, S.; Kongkamol, C. Suspected Cat-to-Human Transmission of SARS-CoV-2, Thailand, July–September 2021. Emerg. Infect. Dis. 2022, 28, 1485–1488. [Google Scholar] [CrossRef] [PubMed]
- Wang, F.; Flanagan, J.; Su, N.; Wang, L.-C.; Bui, S.; Nielson, A.; Wu, X.; Vo, H.-T.; Ma, X.-J.; Luo, Y. RNAscope: A Novel in Situ RNA Analysis Platform for Formalin-Fixed, Paraffin-Embedded Tissues. J. Mol. Diagn. 2012, 14, 22–29. [Google Scholar] [CrossRef] [PubMed]
- Szilasi, A.; Dénes, L.; Jakab, C.; Erdélyi, I.; Resende, T.; Vannucci, F.; Csomor, J.; Mándoki, M.; Balka, G. In situ hybridization of feline leukemia virus in a primary neural B-cell lymphoma. J. Veter- Diagn. Investig. 2020, 32, 454–457. [Google Scholar] [CrossRef]
- https://www.cdc.gov/coronavirus/2019-ncov/lab/rt-pcr-panel-primer-probes.
- Szilasi, A.; Denes, L.; Kriko, E.; Murray, C.; Mandoki, M.; Balka, G. Prevalence of feline leukaemia virus and feline immunodeficiency virus in domestic cats in Ireland. Acta Veter- Hung. 2020, 68, 413–420. [Google Scholar] [CrossRef]
- Aghazadeh, M.; Shi, M.; Barrs, V.R.; McLuckie, A.J.; Lindsay, S.A.; Jameson, B.; Hampson, B.; Holmes, E.C.; Beatty, J.A. A Novel Hepadnavirus Identified in an Immunocompromised Domestic Cat in Australia. Viruses 2018, 10, 269. [Google Scholar] [CrossRef] [PubMed]
- Woo, P.C.Y.; Lau, S.K.P.; Wong, B.H.L.; Fan, R.Y.Y.; Wong, A.Y.P.; Zhang, A.J.X.; Wu, Y.; Choi, G.K.Y.; Li, K.S.M.; Hui, J.; et al. Feline morbillivirus, a previously undescribed paramyxovirus associated with tubulointerstitial nephritis in domestic cats. Proc. Natl. Acad. Sci. 2012, 109, 5435–5440. [Google Scholar] [CrossRef] [PubMed]
- Matuck, B.F.; Dolhnikoff, M.; Duarte-Neto, A.N.; Costa Gomes, G.M.S.; Sendyk, D.I. Salivary glands are a target for SARS-CoV-2: a source for saliva contamination. J. Pathol. 2021, 254, 239–243. [Google Scholar] [CrossRef] [PubMed]
| Primer name | Sequence | Size (bp) |
|---|---|---|
| FIV for | 5’-GCGCTAGCAGCTGCCTAACCGCAAAACCAC-3’ | 163 bp |
| FIV rev | 5’-GTATCTGTGGGAGCCTCAAGGGAGAACTC-3’ | |
| FeLV for | 5’-AACAGCAGAAGTTTCAAGGCC-3’ | 150 bp |
| FeLV rev | 5’-TTATAGCAGAAAGCGCGCG-3’ | |
| FHV for | 5’-GTGCTCTGATAACCGTATGCT-3’ | 230 bp |
| FHV rev | 5’-CTAGAATGGCTACATGGGGTTAG-3’ | |
| FMV for | 5′-CAGAGACTTAATGAAATTTATGG-3′ | 155 bp |
| FMV rev | 5′-CCACCCATCGGGTACTT-3′ |
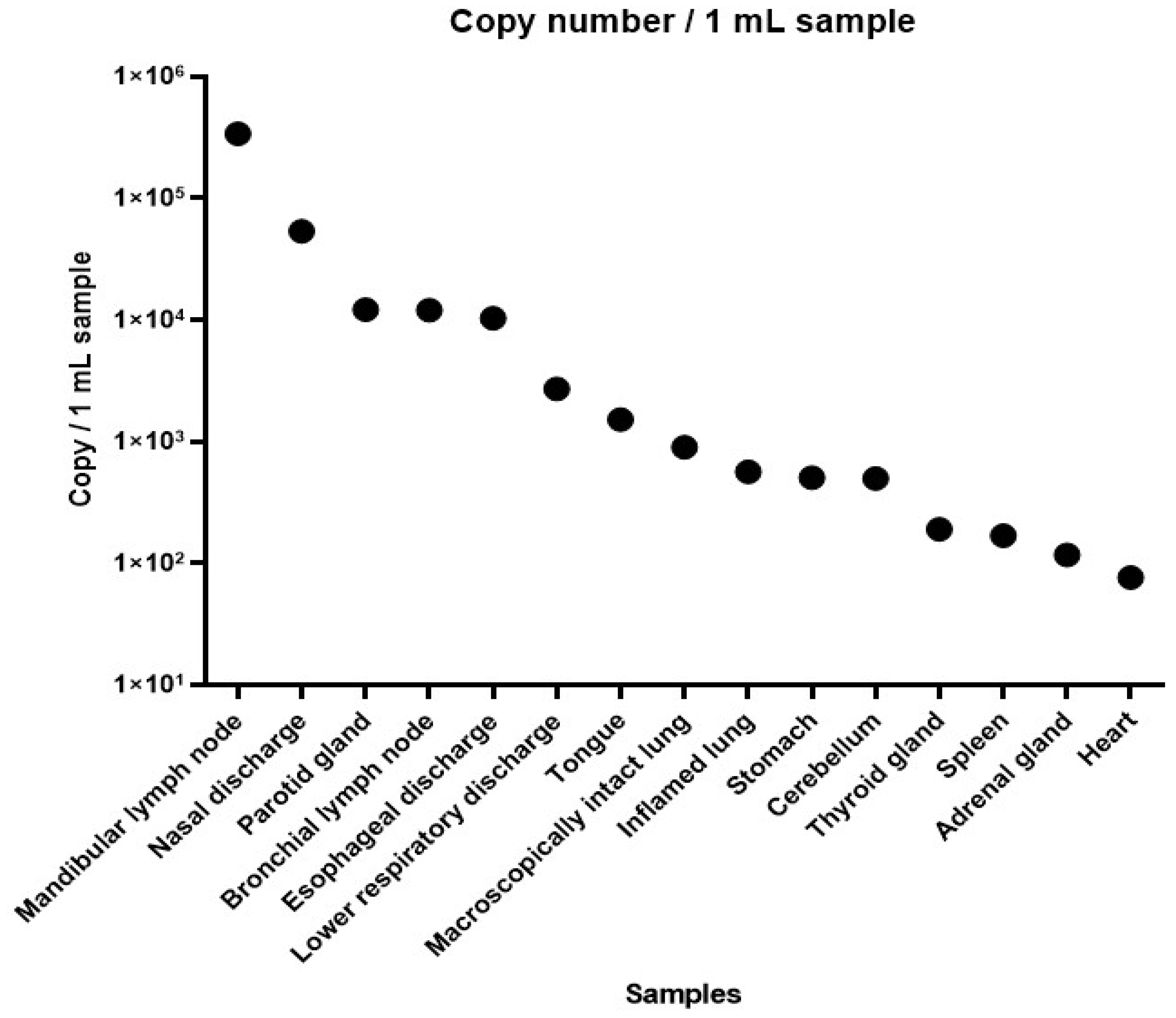
| Sample ID | Sample name | Ct value | Copy / 1 mL sample |
|---|---|---|---|
| 1 | Mesenterium | negative | na |
| 2 | Jejunum | negative | na |
| 3 | Colon | negative | na |
| 4 | Penis | negative | na |
| 5 | Mesenteric lymph node | negative | na |
| 6 | Adrenal gland | 37.36 | 1,17E+02 |
| 7 | Spleen | 36.84 | 1,68E+02 |
| 8 | Liver | negative | na |
| 9 | Kidney | negative | na |
| 10 | Bronchial lymph node# | 30.69 | 1,20E+04 |
| 11 | Heart | 37.99 | 7,63E+01 |
| 12 | Parotid gland | 30.68 | 1,21E+04 |
| 13 | Macroscopically intact lung | 34.43 | 8,98E+02 |
| 14 | Inflamed lung# | 35.09 | 5,65E+02 |
| 15 | Stomach | 35.26 | 5,05E+02 |
| 16 | Tongue# | 33.68 | 1,51E+03 |
| 17 | Mandibular lymph node# | 25.88 | 3,38E+05 |
| 18 | Thyroid gland | 36.67 | 1,90E+02 |
| 19 | Spinal cord | negative | na |
| 20 | Frontal lobe | negative | na |
| 21 | Cerebellum# | 35.28 | 4,98E+02 |
| 22 | Urine | negative | na |
| 23 | Ascites | negative | na |
| 24 | Vitreous body | negative | na |
| 25 | Lower respiratory discharge# | 32.84 | 2,70E+03 |
| 26 | Eye discharge | negative | na |
| 27 | Nasal discharge | 28.53 | 5,35E+04 |
| 28 | Esophageal discharge# | 30.91 | 1,03E+04 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




